Abstract
Maternal diet plays a critical role in health development. Perinatal overnutrition induces metabolic dysfunctions and obesity in the offspring. Obesity is associated with endocannabinoid system (ECS) over activation and oxidative stress. Liver ECS activation induces hepatic steatosis, inflammation and fibrosis while the antagonism of cannabinoid receptors ameliorates these alterations. Here, we investigated the effect of perinatal maternal high-fat diet (HF, 29% of calories as fat) on the ECS and antioxidant system in liver of male and female adult rat offspring (180 days old). Maternal HF diet increased hepatic cannabinoid receptors, ECS metabolizing enzymes and triglyceride content, with male offspring more affected. ECS changes are likely independent of estradiol serum levels but associated with increased hepatic content of estrogen receptor, which can stimulate the expression of ECS components. Differently, maternal HF diet decreased the activity of the antioxidant enzymes glutathione peroxidase, superoxide dismutase and catalase, and increased oxidative stress markers in both sexes. Alterations in the redox homeostasis were associated with mitochondria damage but not with liver fibrosis. Our data suggest that maternal HF diet induces ECS over activation in adulthood, and that male offspring are at higher risk to develop liver disease compared with female rats.
Similar content being viewed by others
Introduction
Environmental conditions during critical developmental periods such as gestation and lactation predict the lifelong health of the offspring, a phenomenon known as “programming”1. Maternal exposure to under or overnutrition, stress, endocrine disruptors and other types of insults may program metabolic dysfunctions in the offspring including diabetes, cardiovascular diseases, dyslipidemia and obesity2,3. We have shown that maternal high-fat (HF) diet during gestation and lactation induces obesity in the offspring associated with thyroid and adrenal dysfunction, hyperleptinemia with hypothalamic leptin resistance, hepatic dysfunction, dyslipidemia, hyperphagia and increased appetite for fat across different periods of life4,5,6,7,8.
Obesity is an important risk factor for liver dysfunctions such as non-alcoholic fatty liver disease (NAFLD)9. We previously demonstrated that maternal HF diet increased hepatic triglycerides, total lipid content, free cholesterol in parallel to reduced cholesterol ester and higher liver weight of adolescent rat offspring, which could predispose to later NAFLD5.
Obesity and NAFLD are associated with dysfunctions in the endocannabinoid system (ECS) in humans and animal models10,11. ECS comprises the main endogenous cannabinoids (ECs), anandamide (AEA) and 2-aracdonoylglycerol (2-AG), which are produced from arachidonic acid from membrane phospholipids. ECs regulate energy and lipid metabolism through central and peripheral mechanisms via cannabinoid receptors subtypes 1 and 2 (CB1 and CB2), and ECs are degraded by fatty acid amide hydroxylase (FAAH) and monoacylglycerol lipase (MAGL)11,12.
CB1 and CB2 are expressed in the central nervous system (CNS), heart, uterus, intestine, brown and white adipose tissue, cells of the immune system and liver13,14,15. In the liver, ECs increase lipid accumulation and reactive oxygen species (ROS)16. CB1 activation increases oxidative stress and decreases antioxidant activity, whereas CB2 receptors are related to the suppression of ROS generation in different tissues17. On the other hand, CB1 antagonism improves liver steatosis, decreases serum triglyceride levels and increases serum high-density lipoprotein (HDL)18,19.
Hepatic diseases are also associated with the accumulation of cellular lesions caused by deregulation of redox homeostasis, which may result from increased ROS, such as superoxide and hydrogen peroxide (H2O2), or decreased antioxidant defense of the enzymes superoxide dismutase (SOD), glutathione peroxidase (GPx) and catalase (Cat)20. The disarrangement of this system leads to cellular oxidative stress, with decreased total thiols and increased residues of carbonylated proteins21 and derivatives from lipid peroxidation like 4-hydroxynonenal (4-HNE)22.
Although studies have demonstrated a relationship between ECS and the cellular redox homeostasis in liver22,23, the interconnection between ECS and oxidative stress in metabolic programming is mostly unknown. We hypothesized that maternal HF diet consumption during perinatal phase would program hepatic dysfunctions associated with dysregulation of ECS and redox homeostasis in the liver of adult rat offspring. Because ECS is known to be regulated by sex steroids24,25, we further hypothesized that liver ECS programming would occur in a sex dependent manner and liver ECS would be differentially regulated by estradiol levels in female adult rats.
Results
Maternal HF diet increased body weight in male and female adult offspring at 180 days old (p = 0.017, p = 0.014, respectively) as well as subcutaneous fat pad mass (p = 0.025; p = 0.001, respectively) (Table 1). Besides, maternal HF diet increased the brown adipose tissue mass, and serum levels of glucose and triglycerides (p = 0.01; p = 0.01; p = 0.04, respectively) in male adult offspring, without differences in females. In female adult offspring from HF dams, we observed diminished estradiol levels (p = 0.0007), without differences in males. No differences were observed in aspartate aminotransferase (AST) and alanine aminotransferase (ALT) in male or female adult offspring (Table 1).
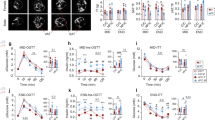
In the liver of male offspring, maternal HF diet increased CB1 and CB2 protein content (p = 0.02; p = 0.008, respectively) (Fig. 1a,b), FAAH content (p = 0.01) (Fig. 1c) and MAGL content (p = 0.003) (Fig. 1d). However, female HF offspring only presented increase in MAGL (p = 0.03) (Fig. 1i).
Effect of maternal HF diet on hepatic protein expression of CB1 (a,f), CB2 (b,g), FAAH (c,h), MAGL (d,i) in both male and female offspring at 180-day-old. Representative western blot bands for each protein were showed (e,j). Data is expressed as mean ± S.E.M, n = 6–7 per group; *p < 0.05, **p < 0.001, by Student’s t test.
Because we found reduced plasma estrogen levels in the female HF offspring, we evaluated the influence of estradiol circulating levels on the liver ECS components. We performed ovariectomy (OVX) and estradiol treatment in OVX female control rats (OVXE). The estradiol levels were higher in OVXE (p = 0.04) compared with OVX rats (Fig. 2a), as expected. However, we did not observe alterations in liver ECS between OVX and OVXE rats (Fig. 2b-e).
Effect of ovariectomy and estradiol treatment on estradiol levels (a) and hepatic protein expression of CB1 (b), CB2 (c), FAAH (d) and MAGL (e) of adult rats. Representative western blot bands for each protein were showed (f). Data is expressed as mean ± S.E.M, n = 6–9 per group; *p < 0.05, by Student’s t test.
We also evaluated the protein expression of ERα in liver of C and HF offspring, and the ERα expression was higher in HF male offspring (p = 0.04) compared with sex-matched controls, without changes in female offspring (Fig. 3a,b).
ECS changes in the liver occurred in parallel to increased hepatic triglycerides content (p = 0.04) only in male offspring (Fig. 4e). Maternal HF diet did not change hepatic total lipid content, free cholesterol, cholesterol ester and NEFA (Fig. 4a-j) in male or female offspring.
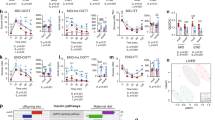
We also evaluated the main enzymes of antioxidant system in the liver of adult offspring. Maternal HF diet decreased Cat activity (Fig. 5a,d) (p = 0.001), and SOD activity (Figs. 5c,f) both in male and female offspring (p = 0.04; p = 0.0006, respectively), while it decreased GPx activity (p = 0.04) only in male rats (Fig. 5b). Regarding the oxidative stress biomarkers, maternal HF diet decreased liver thiol content (p = 0.0007) (Fig. 6a), and increased protein carbonyl and 4-HNE content (p = 0.04; p = 0.03) in male rats (Fig. 6b,c). In female rats, maternal HF diet only increased the 4-HNE content (p = 0.02) (Fig. 6h). However, maternal HF diet did not change the H2O2 generation in membrane or microsomal fraction in both male and female offspring.
Effect of maternal HF diet on hepatic activity of antioxidant enzymes, catalase (a,d), glutathione peroxidase (GPx) (b,e), superoxide dismutase (SOD) (c,f) in both male and female offspring at 180-day-old. Data is expressed as mean ± S.E.M, n = 8 per group; *p < 0.05, **p < 0.001, ***p < 0.0001, by Student’s t test.
Effect of maternal HF diet on hepatic biomarkers of oxidative stress: thiol content (a,f), total protein bound carbonyl (b,g) and 4-HNE (c,h) in both male and female offspring at 180-day-old. Representative western blot bands for each protein were showed (d,i). Data is expressed as mean ± S.E.M., n = 7–8 per group; *p < 0.05, ***p < 0.0001, by Student’s t test.
We quantified the percentage of injured mitochondria by Transmission Electron Microscopy and we observed that maternal HF diet was able to induce injury in both male and female offspring (p = 0.005; p = 0.001, respectively) (Fig. 7a,b).
To evaluate the impact of ECS and redox homeostasis changes on liver morphology, we performed the picrosirius red staining of collagen fibers to investigate fibrosis. However, we observed no effect of maternal HF diet on collagen fibers content in male or female offspring (Fig. 8).
Discussion
In the current study, we investigated the ECS in liver of the male and female adult rat offspring from HF fed dams in parallel to the prooxidant and antioxidant profile. We observed that male HF offspring phenotype at adulthood corroborated previously data published by our group6,8 and others26. Male and female offspring from HF dams displayed higher body weight and adiposity. This phenotype is observed already at weaning in this model, which is associated with changes in the milk composition from HF fed dams with higher lipid and protein content4.
The main finding of the present study is that maternal HF diet during perinatal life induced sex-specific long-term changes in the liver ECS components of the adult offspring. Maternal HF diet increased protein expression of CB1 and CB2 receptor in male offspring. It has been shown that upregulation of CB1 in liver is related with hepatic fibrosis, steatosis and lipid accumulation in humans, animal models and hepatocytes in vitro27,28,29, and on the other side, the blockage of CB1 receptor reverses these dysfunctions28.
Different of CB1, the role of CB2 receptor on metabolic parameters is controversial or dual30. In obese children with NAFLD, a polymorphism of the CB2 gene (Cnr2) is associated with severity of liver inflammation and inflammatory steatohepatitis (NASH)31. In obese women, CB2 mRNA levels in liver correlates positively with hepatic Acetyl-coenzyme A carboxylase 1 (ACC1) gene expression, a key lipogenic enzyme of the de novo synthesis of fatty acids32. Besides, it is associated with increased PPARγ, IL6, TNFα, resistin, suggesting a deleterious role promoting liver lipid accumulation and inflammation. Paradoxically, CB2 mRNA levels were also positively correlated with the levels of the adipocyte-derived hormone adiponectin in the liver32, which presents beneficial effect on steatosis, insulin sensitivity, oxidative stress and inflammation33. In cirrhotic human liver, CB2 activation develops an antifibrogenic role during chronic liver injury34. On the other hand, in Cnr2 −/− HFD-fed mice model, the absence of CB2 receptors decreases the steatosis and liver triglyceride concentration caused by HF diet, suggesting that CB2 activation contributes to liver inflammation and hepatic steatosis35. Thus, increased hepatic CB1 and CB2 in HF male offspring may represent a higher risk to develop liver steatosis and fibrosis.
Increased triglyceride hepatic content indicates liver injury probably induced by HF diet36,37,38. In nonhuman primate model, fetuses from obese mothers fed HF diet have increased liver triglycerides content39, suggesting that developing fetuses of different species are highly vulnerable to excess of lipids in maternal diet. In fact, the analysis of lipid profile showed that HF male offspring had increased hepatic content of triglycerides and hipertrigliceridemia. However, maternal HF diet did not affect serum levels of AST and ALT neither the amount of collagen fibers in the liver, which are markers of severe hepatic damage. Thus, we speculate that the profile found in liver ECS of the offspring may contribute to a more drastic metabolic phenotype under a second nutritional hit, such as a high-fat diet challenge in adulthood, as previously demonstrated by other studies40,41.
Interestingly, we observed a marked sex-specific regulation of the hepatic ECS by maternal HF diet. Sex-specific programming is an interesting topic and it has been demonstrated in several experimental models, including the maternal HF diet rat model42,43. However, very little is known regarding the direct ECS regulation by sex steroids, even in control rats, mice or humans24. In our previous studies7,8, we characterized that the ECS is differentially regulated in male and female HF offspring in other tissues, such as white and brown adipose tissue and hypothalamus. Interestingly, ECS changes can occur even before the major physiological alterations in sex hormone levels during puberty, since we observed that maternal HFD induces ECS sex-specific changes already at birth8 and also at weaning7. In the present study, maternal HF diet increased the ECS expression in liver of adult male offspring with minor changes in females. In adulthood, ECS changes occurred in parallel to decreased serum levels of estradiol in female offspring. To test whether this serum alteration was associated with the sex-dependent ECS changes, we developed the OVX model. However, the modulation of estradiol levels at adulthood did not affect ECS expression in the liver. It is possible that liver ECS changes induced by maternal HF diet occurred before the age analyzed in the present study, similar to the profile we observed in other peripheral tissues and may not be directly associated with changes in serum estradiol. A possible mechanism would be the modulation of estrogen receptor (ER) expression in the liver, rather than estradiol serum levels. It has been reported the presence of response elements to estrogen in Cnr1 and Faah promoter region44,45, and estrogen has a stimulatory effect on ECS components25. Here, we showed that maternal HF diet increased the ER levels in liver of male offspring but not in female offspring, which may contribute to the increased CB1, CB2 and FAAH in males.
To our knowledge, only two studies have been conducted to characterize the effect of maternal HF diet on hepatic expression of the ECS components. The first one conducted by Ramirez et al., showed that maternal hypercaloric-hypoproteic palatable diet decreases mRNA levels of FAAH and MAGL in the liver of female adult rat offspring46, rather than changes in male offspring as we showed here. Although interesting, these results cannot be directly compared with the present study because there are relevant differences in the offspring phenotype, such as decreased offspring body weight. In adult offspring programmed by perinatal undernutrition, it was demonstrated decreased hepatic mRNA levels of CB1, FAAH and MAGL only in female offspring47. However, of note, the present study focused on the regulation of the protein content of ECS components instead of transcript levels. Another study recently published by Gandhi et al., showed that maternal HF diet in baboons decreases CB2 and FAAH protein content in fetal liver, but the impact of ECS changes on liver morphology and function was not investigated48. The different profiles found in liver ECS among these studies may arise from different ages and species used in the experiments.
In this study, we showed that maternal HF diet increased FAAH and MAGL protein expression in the liver of offspring. MAGL inhibition suppresses oxidative stress and inflammation in the liver49, and FAAH inhibitor decreases ROS production and improves hepatic antioxidant response22. Thus, the increased ECS enzymes may contribute to impaired redox homeostasis in the liver of HF offspring. These findings suggest an interplay of ECS and redox system and may be important components of study for therapeutic targets in metabolic diseases.
Liver injury is frequently associated with oxidative stress, which is characterized by an imbalance between prooxidant and antioxidant systems resulting in increased intracellular ROS availability50. We observed decreased activity of the antioxidant enzymes Cat, GPx and SOD in both male and female HF offspring, suggesting a deleterious impact of maternal HF diet on antioxidant defense. In fact, we also observed alterations in markers of oxidative stress such as decreased thiol levels and increased total protein bound carbonyl and 4-HNE content in HF offspring. These biochemical alterations occurred in parallel to increased mitochondrial damage in the liver.
There are some evidences that ECS can modulate ROS production by alterations in the activity of prooxidant and antioxidant enzymes. Jia, et al.51 demonstrated that treatment of neuronal cells with AEA led to decreased intracellular ROS and NADPH oxidase 2 (Nox2), which were reversed by the CB1 antagonist. They also demonstrated that under oxidative stress, AEA increases SOD and GPx activity, suggesting improvement of the antioxidant defense. In mice presenting NAFLD, CB1 antagonism improves liver oxidative stress52. Moreover, CB2 agonists protect against hepatic injury induced by ischemia and decrease tissue free radical damage, suggesting that CB2 activation could be a protective pathway53,54.
Although maternal HF diet induced minor changes in the liver ECS of female offspring, it induced significant changes in the antioxidant profile and oxidative stress markers in both male and female adult rats. These findings suggest that alteration in additional pathways, independent on the ECS, contribute to the oxidative balance in liver of female offspring.
We consider as main limitation of the present study the lack of measurement of the tissue levels of the major endocannabinoids AEA and 2-AG in the offspring liver. In addition, a more detailed analysis of molecular and morphological markers of liver injury would provide a better picture of the impact of maternal HF diet on liver metabolism and inflammatory status.
In conclusion, we demonstrated that maternal HF diet induced overweight, increased adiposity, dyslipidemia, over expression of the ECS and impairment of the antioxidant capacity in liver of adult offspring, with male rats more affected. Of note, these alterations did not impact the levels of liver fibrosis or serum hepatic enzymes, which could possibly be triggered by second hits such as postnatal exposure to obesogenic factors. The present data is a contribution to characterize the impact of maternal HF diet on liver ECS in parallel to oxidative stress markers in the offspring, and the comprehension of these pathways may contribute to insights into new pharmacological targets for treatment of liver disease, considering sex differences in the liver ECS regulation.
Methods
Animals
The handling of the experimental animals followed the American Physiological Society’s guiding principles55 and was previously approved by the Animal Care and Use Committee of the Carlos Chagas Filho Biophysics Institute (process number 123/14 and 089/17). For the experimental programming model induced by maternal HF diet consumption, 60-day-old female Wistar rats (180–220 g) were obtained from the Center of Rat Reproduction of the Federal University of Rio de Janeiro, Rio de Janeiro, Brazil. Progenitor rats received control or experimental diets as detailed in the section “experimental groups and diets”.
A separate subset of female rats underwent a bilateral ovariectomy (OVX) to investigate the role of modulation of the estradiol serum levels on ECS components in the liver at adulthood. Rats were anesthetized with ketamine/xylazine (30/6 mg/kg, intramuscular). A midline incision was made to expose the lower abdominal cavity. Rats were randomly divided into 2 groups: OVX and OVX treated with estradiol (OVXE). In the OVXE group, the rats were administered with 7 µg/kg/day of β-estradiol-3-benzoate (Sigma-Aldrich, USA) by subcutaneous injection for 7 days beginning 10 days after surgery, while group OVX received vehicle (soy oil) in the same volume. We evaluated the ECS in liver samples from both groups by western blotting.
For all animal procedures, rats were housed under controlled conditions in a 12-h light-dark cycle (lights on from 7 a.m. to 7 p.m.) and at room temperature of 23 ± 2 °C. Water and diets were offered ad libitum.
Experimental groups and diets
Female progenitor rats were randomly chosen and divided in two groups: control group (C) which received a standard diet (9% of the calories as fat), and a high-fat group (HF) which received a HF diet (28.6% of the calories as fat) as previously described by our group4,5,6,7,8. Progenitor rats received the diets during 8 weeks before mating and throughout gestation (21 days) and lactation (21 days). After mating, pregnant rats were housed in individual standard rat cages until delivery. At birth, the litters were adjusted to three male and three female pups per each dam, a number that maximizes lactation performance56. All pups were weaned at 21-day-old and fed a control chow diet until adulthood. Body weight was recorded weekly from weaning to 180-day-old, when offspring were euthanized to collect and weigh the retroperitoneal and subcutaneous fat pads, liver, and blood samples. For each experimental procedure, except for electronic microscopy, at least 6 animals from different litters were used for each group to avoid litter effects on the statistical analysis.
Plasma analyses
Blood samples were centrifuged (1233 × g for 15 min, 4 °C) for plasma separation. Glucose, triglycerides, AST and ALT concentration were determined using commercial kits (Gold Analisa®, Belo Horizonte, MG, Brazil). The AST/ALT intra and interassay variation coefficients were 1.5% and 5.9%/ 2.8% and 5.3%, respectively. The limit of detection was 400 U/L. Plasma insulin concentration was determined by radioimmunoassay (Linco Research, MO, USA). The insulin intra- and interassay variation coefficients were 9.8 and 12.2%, respectively. Plasma estradiol levels were determined using a radioimmunoassay kit (MP Diagnostics, 25 USA) with intra-assay variation of 15,7%.
Western blotting
Protein content of ECS targets and 4-HNE was determined by western blotting in liver homogenate as previously described with some adaptations7,8. Each sample was obtained from a different rat from a different litter (n = 7). Anti-CB1, CB2, FAAH, MAGL and 4-HNE were assayed. Liver samples were collected and snap frozen in liquid nitrogen, followed by lysis (pH 6.4 lysis buffer: 50 mM HEPES, 1 mM MgCl2, 4 10 mM EDTA, 1% Triton X-100, containing a protease inhibitor cocktail (Roche®)). After centrifugation, total protein content was determined using a BCA™ Protein Assay Kit (Thermo Scientific®, Rockford, IL, USA). Total liver protein (15ug) were separated by 12% SDS-PAGE, and then transferred to a polyvinylidene difluoride membrane (Hybond-P, 0.45 µm, PVDF, Amersham Biosciences, BKM, England) using Semi-dry system (Bio-rad laboratories, Hercules, CA, USA). The non-specific binding was blocked for 90 min with 5% BSA with continuous shaking and then membranes were incubated overnight at 4 °C with primary antibodies followed by 90 min incubation with the secondary antibody. The specification of each antibody was described in Table 2. Immunoreacted proteins were visualized by ECL prime kit and Image Quant LAS 4000 equipment followed by densitometric analyses (GE Healthcare, Buckingham, Shire, UK). Glyceraldehyde-3-phosphate dehydrogenase (GAPDH) or cyclophilin was used for normalization.
Antioxidant enzyme activities
Approximately 100 mg of liver from both male and female offspring were homogenized in 5 mM Tris HCl, 0.9% NaCl (pH 7.4) containing protease inhibitor cocktail (Roche Diagnostics, IN, USA). The homogenate was centrifuged at 720 × g for 10 min, 4 °C and the supernatant was used for enzyme activity assays. Total protein content was quantified using a PierceTM BCA Protein Assay Kit (Thermo Scientific, Rockford, USA). All the enzymatic assays were performed in an UV spectrophotometer (PerkinElmer, LAMBDA™, Shelton, CT, USA) at 37 °C. Catalase activity was measured according the method previously described57. Glutathione peroxidase activity was measured by NADPH oxidation at 340nm58 and superoxide dismutase activity was assayed by the reduction of cytochrome C at 550nm59.
Thiol content and carbonyl groups quantification
Total reduced thiols were determined using 5,5-dithionitrobenzoic acid (DTNB). Thiol residues react with DTNB, cleaving the disulfide bond to give 2-nitro-5-thiobenzoate (NTB−), which ionizes to the NTB2− dianion in water at neutral and alkaline pH. NTB2− was quantified in a spectrophotometer by measuring the absorbance at 412 nm and data was expressed as nmol of reduced DTNB/mg protein60. The proteins bound to carbonyl were evaluated based on the reaction with dinitrophenilhidrazine (DNPH), as previously described61. The carbonyl groups were measured at 370 nm.
Thin-layer chromatography (TLC)
Liver total lipids were extracted by the method previously described62 with some modifications. After incubation in chloroform-methanol-water solution (2:1:0.8, v/v), samples were centrifuged (1500 × g for 20 min at 4 °C). Then, the chloroform was added to the collected supernatant. After centrifugation (1500 × g for 20 min) the ordic phase was removed and dried in nitrogen. The extracted lipids were analyzed by TLC for neutral lipids, using a DC Silicagel 60 plate (Merck Millipore, HE, Germany). The plates were submerged for 10 s in Charring solution (3% CuSO4 and 8% H3PO4 v/v), then they were dried and heated to 110 °C for 10 min. TLC plates were analyzed by densitometry (ImageQuant LAS 4000, GE Healthcare, Buckingham, Shire, UK). We analyzed free cholesterol, cholesterol ester, non-esterification fatty acid and triglycerides.
H2O2 generation
Liver tissue was homogenized in a 50 mM sodium phosphate buffer, pH 7.2, containing 0.25 M sucrose, 0.5 mM dithiothreitol, 1 mM ethylene glycol tetra-acid (EGTA), 5 mg/mL aprotinin, and 34.8 mg/mL phenyl methane sulfonyl fluoride (PMSF). The homogenates were centrifuged at 3000 × g for 15 min at 4 °C. The membrane fraction was obtained by the resuspension of the pellet in assay buffer (0.5 mL 50 mM sodium phosphate buffer, pH 7.2, containing 0.25 M sucrose, 2 mM MgCl2, 5 mg/ml aprotinin and 34,8 mg/ml phenylmethanesulfonyl fluoride (PMSF)). To obtain the microsomal fraction, the supernatant was centrifuged at 100,000 × g for 35 min at 4 °C and the pellets were resuspended in assay buffer. Then, the supernatant was centrifuged at 100,000 × g for 35 min at 4 °C and the pellets were resuspended in assay buffer and were used for analyses. Both fractions were incubated in 150 mM sodium phosphate buffer (pH 7.4) containing SOD (100 U/ml; Sigma, USA), horseradish peroxidase (0.5 U/ml, Roche, Indianapolis, IN), Amplex red (50 µM; Molecular Probes, Eugene, OR), 1 mM EGTA in the presence or absence of 1 mM NADPH. The fluorescence was immediately measured in a microplate reader (Victor X4; PerkinElmer, Norwalk, CT) at 30 °C, using excitation at 530 nm and emission at 595 nm63.
Transmission Electron Microscopy (TEM)
The fragments of liver were processed and analyzed qualitatively using a JEM1011 (JEOL) microscope. The fragments were washed in PBS and fixed for 24 h in a solution containing 2.5% glutaraldehyde in 0.1 M sodium cacodylate buffer (pH 7.2). Then, the samples were washed for 10 min in the same buffer. This wash step was repeated 3 times. The tissue was post-fixed for 1 h with a 1% osmium tetroxide (OsO4) solution in 0.1 M sodium cacodylate buffer (pH 7.2), dehydrated in an acetone series (30, 50, 70, 90 and 100%) and embedded in Poly/Bed(r) 812 resin (Ted Pella Inc, Redding, CA, USA). After polymerization, ultrathin sections were obtained and contrasted with uranyl acetate-lead citrate for ultrastructural observation. Were collected in 3 grids with 7 micrometers between them, acquired 10 images of random fields in each grid, quantified the total number of mitochondria and the number of injured mitochondria to obtain the percentage of injured mitochondria.
Picrosirius red collagen staining
For the histological analysis, the liver was collected, fixed with 4% formalin, cleared and embedded in histological paraffin. The fixed samples were sectioned using a microtome in semi serials cuts 5 µm thick. After deparaffinization, the sections were rehydrated and incubated with 0.2% phosphomolybdic acid (Merk, Darmstadt, Germany) for 1 min and then incubated with picrosirius red solution for 90 min. After this, the sections were washed with 0.01 N of chloridric acid for 2 min and then in 70% alcohol for 45 seconds. The sections were then dehydrated with two successive washes of 5 min with 95% and 100% alcohol. After two washes of 100% xylol for 5 min, the blades were mounted and visualized in microscope (Axiovert 100-Carl Zeiss). The number of collagen fibers were quantified (15 fields for animal) using the Image Pro Plus® version 4.5 software (Media Cybernetics, Silver Spring, MD, USA).
Data analysis
All data are presented as the mean ± standard error from mean (S.E.M.). Data sets were tested for normality using the Kolmogorov–Smirnov test, and the differences between control and HF offspring per each sex were analyzed by Mann-Whitney test or Student’s t test when appropriated. A value of p < 0.05 was considered statistically significant. Statistical analyses were performed in GraphPad Prism version 6.0 for Windows (GraphPad Software Inc., San Diego, CA, USA).
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author.
References
Kwon, E. J. & Kim, Y. J. What is fetal programming?: a lifetime health is under the control of in utero health. Obstetrics & gynecology science 60, 506–519, https://doi.org/10.5468/ogs.2017.60.6.506 (2017).
Elshenawy, S. & Simmons, R. Maternal obesity and prenatal programming. Mol Cell Endocrinol 435, 2–6, https://doi.org/10.1016/j.mce.2016.07.002 (2016).
Desai, M., Jellyman, J. K. & Ross, M. G. Epigenomics, gestational programming and risk of metabolic syndrome. Int J Obes (Lond) 39, 633–641, https://doi.org/10.1038/ijo.2015.13 (2015).
Franco, J. G. et al. Maternal high-fat diet induces obesity and adrenal and thyroid dysfunction in male rat offspring at weaning. J Physiol 590, 5503–5518, https://doi.org/10.1113/jphysiol.2012.240655 (2012).
Oliveira, L. S. et al. Perinatal maternal high-fat diet promotes alterations in hepatic lipid metabolism and resistance to the hypolipidemic effect of fish oil in adolescent rat offspring. Molecular nutrition & food research 60, 2493–2504, https://doi.org/10.1002/mnfr.201600171 (2016).
Franco, J. G. et al. Resveratrol treatment rescues hyperleptinemia and improves hypothalamic leptin signaling programmed by maternal high-fat diet in rats. Eur J Nutr 55, 601–610, https://doi.org/10.1007/s00394-015-0880-7 (2016).
Almeida, M. M. et al. Perinatal maternal high-fat diet induces early obesity and sex-specific alterations of the endocannabinoid system in white and brown adipose tissue of weanling rat offspring. The British journal of nutrition 118, 788–803, https://doi.org/10.1017/S0007114517002884 (2017).
Dias-Rocha, C. P. et al. Maternal high-fat diet induces sex-specific endocannabinoid system changes in newborn rats and programs adiposity, energy expenditure and food preference in adulthood. The Journal of nutritional biochemistry 51, 56–68, https://doi.org/10.1016/j.jnutbio.2017.09.019 (2018).
Ahima, R. S. Digging deeper into obesity. The Journal of clinical investigation 121, 2076–2079, https://doi.org/10.1172/JCI58719 (2011).
Matias, I. & Di Marzo, V. Endocannabinoids and the control of energy balance. Trends Endocrinol Metab 18, 27–37, https://doi.org/10.1016/j.tem.2006.11.006 (2007).
Ruby, M. A. et al. Acute overactive endocannabinoid signaling induces glucose intolerance, hepatic steatosis, and novel cannabinoid receptor 1 responsive genes. PloS one 6, e26415, https://doi.org/10.1371/journal.pone.0026415 (2011).
D’Addario, C. et al. Endocannabinoid signaling and food addiction. Neurosci Biobehav Rev 47, 203–224, https://doi.org/10.1016/j.neubiorev.2014.08.008 (2014).
Roche, R. et al. Presence of the cannabinoid receptors, CB1 and CB2, in human omental and subcutaneous adipocytes. Histochemistry and cell biology 126, 177–187, https://doi.org/10.1007/s00418-005-0127-4 (2006).
Nunez, E. et al. Cannabinoid CB2 receptors are expressed by perivascular microglial cells in the human brain: an immunohistochemical study. Synapse 53, 208–213, https://doi.org/10.1002/syn.20050 (2004).
Wang, M., Meng, N., Chang, Y. & Tang, W. Endocannabinoids signaling: Molecular mechanisms of liver regulation and diseases. Frontiers in bioscience 21, 1488–1501 (2016).
Basu, P. P., Aloysius, M. M., Shah, N. J. & Brown, R. S. Jr. Review article: the endocannabinoid system in liver disease, a potential therapeutic target. Alimentary pharmacology & therapeutics 39, 790–801, https://doi.org/10.1111/apt.12673 (2014).
Han, K. H. et al. CB1 and CB2 cannabinoid receptors differentially regulate the production of reactive oxygen species by macrophages. Cardiovascular research 84, 378–386, https://doi.org/10.1093/cvr/cvp240 (2009).
Despres, J. P., Golay, A. & Sjostrom, L. & Rimonabant in Obesity-Lipids Study, G. Effects of rimonabant on metabolic risk factors in overweight patients with dyslipidemia. The New England journal of medicine 353, 2121–2134, https://doi.org/10.1056/NEJMoa044537 (2005).
Hussien, N. I., El-Kerdasy, H. I. & Ibrahim, M. E. Protective effect of rimonabant, a canabinoid receptor 1 antagonist, on nonalcoholic fatty liver disease in a rat model through modulation of the hepatic expression of activin A and follistatin. Can J Physiol Pharmacol 95, 1433–1441, https://doi.org/10.1139/cjpp-2017-0070 (2017).
Kasapoglu, M. & Ozben, T. Alterations of antioxidant enzymes and oxidative stress markers in aging. Experimental gerontology 36, 209–220 (2001).
Frankenfeld, S. P. et al. The anabolic androgenic steroid nandrolone decanoate disrupts redox homeostasis in liver, heart and kidney of male Wistar rats. PloS one 9, e102699, https://doi.org/10.1371/journal.pone.0102699 (2014).
Biernacki, M. et al. Crosstalk between liver antioxidant and the endocannabinoid systems after chronic administration of the FAAH inhibitor, URB597, to hypertensive rats. Toxicology and applied pharmacology 301, 31–41, https://doi.org/10.1016/j.taap.2016.04.006 (2016).
Lipina, C. & Hundal, H. S. Modulation of cellular redox homeostasis by the endocannabinoid system. Open biology 6, 150276, https://doi.org/10.1098/rsob.150276 (2016).
Craft, R. M., Marusich, J. A. & Wiley, J. L. Sex differences in cannabinoid pharmacology: a reflection of differences in the endocannabinoid system? Life Sci 92, 476–481, https://doi.org/10.1016/j.lfs.2012.06.009 (2013).
Maia, J. et al. The endocannabinoid system expression in the female reproductive tract is modulated by estrogen. J Steroid Biochem Mol Biol, https://doi.org/10.1016/j.jsbmb.2017.07.023 (2017).
Tellechea, M. L., Mensegue, M. F. & Pirola, C. J. The Association between High Fat Diet around Gestation and Metabolic Syndrome-related Phenotypes in Rats: A Systematic Review and Meta-Analysis. Sci Rep 7, 5086, https://doi.org/10.1038/s41598-017-05344-7 (2017).
Osei-Hyiaman, D. et al. Endocannabinoid activation at hepatic CB1 receptors stimulates fatty acid synthesis and contributes to diet-induced obesity. The Journal of clinical investigation 115, 1298–1305, https://doi.org/10.1172/JCI23057 (2005).
Teixeira-Clerc, F. et al. CB1 cannabinoid receptor antagonism: a new strategy for the treatment of liver fibrosis. Nature medicine 12, 671–676, https://doi.org/10.1038/nm1421 (2006).
De Gottardi, A., Spahr, L., Ravier-Dall’Antonia, F. & Hadengue, A. Cannabinoid receptor 1 and 2 agonists increase lipid accumulation in hepatocytes. Liver international: official journal of the International Association for the Study of the Liver 30, 1482–1489, https://doi.org/10.1111/j.1478-3231.2010.02298.x (2010).
Soethoudt, M. et al. Cannabinoid CB2 receptor ligand profiling reveals biased signalling and off-target activity. Nat Commun 8, 13958, https://doi.org/10.1038/ncomms13958 (2017).
Rossi, F. et al. Cannabinoid receptor type 2 functional variant influences liver damage in children with non-alcoholic fatty liver disease. PLoS One 7, e42259, https://doi.org/10.1371/journal.pone.0042259 (2012).
Auguet, T. et al. Endocannabinoid receptors gene expression in morbidly obese women with nonalcoholic fatty liver disease. Biomed Res Int 2014, 502542, https://doi.org/10.1155/2014/502542 (2014).
Rodriguez, A., Ezquerro, S., Mendez-Gimenez, L., Becerril, S. & Fruhbeck, G. Revisiting the adipocyte: a model for integration of cytokine signaling in the regulation of energy metabolism. Am J Physiol Endocrinol Metab 309, E691–714, https://doi.org/10.1152/ajpendo.00297.2015 (2015).
Julien, B. et al. Antifibrogenic role of the cannabinoid receptor CB2 in the liver. Gastroenterology 128, 742–755 (2005).
Deveaux, V. et al. Cannabinoid CB2 receptor potentiates obesity-associated inflammation, insulin resistance and hepatic steatosis. PLoS One 4, e5844, https://doi.org/10.1371/journal.pone.0005844 (2009).
Anstee, Q. M. & Goldin, R. D. Mouse models in non-alcoholic fatty liver disease and steatohepatitis research. International journal of experimental pathology 87, 1–16, https://doi.org/10.1111/j.0959-9673.2006.00465.x (2006).
Bradbury, M. W. & Berk, P. D. Lipid metabolism in hepatic steatosis. Clinics in liver disease 8, 639–671, xi, https://doi.org/10.1016/j.cld.2004.04.005 (2004).
Armitage, J. A., Taylor, P. D. & Poston, L. Experimental models of developmental programming: consequences of exposure to an energy rich diet during development. The Journal of physiology 565, 3–8, https://doi.org/10.1113/jphysiol.2004.079756 (2005).
McCurdy, C. E. et al. Maternal high-fat diet triggers lipotoxicity in the fetal livers of nonhuman primates. The Journal of clinical investigation 119, 323–335 (2009).
Mouralidarane, A. et al. Maternal obesity programs offspring non-alcoholic fatty liver disease through disruption of 24-h rhythms in mice. Int J Obes (Lond) 39, 1339–1348, https://doi.org/10.1038/ijo.2015.85 (2015).
Mouralidarane, A. et al. Maternal obesity programs offspring nonalcoholic fatty liver disease by innate immune dysfunction in mice. Hepatology 58, 128–138, https://doi.org/10.1002/hep.26248 (2013).
Aiken, C. E. & Ozanne, S. E. Sex differences in developmental programming models. Reproduction 145, R1–13, https://doi.org/10.1530/REP-11-0489 (2013).
Lecoutre, S. et al. Depot- and sex-specific effects of maternal obesity in offspring’s adipose tissue. The Journal of endocrinology 230, 39–53, https://doi.org/10.1530/JOE-16-0037 (2016).
Proto, M. C. et al. Interaction of endocannabinoid system and steroid hormones in the control of colon cancer cell growth. Journal of cellular physiology 227, 250–258, https://doi.org/10.1002/jcp.22727 (2012).
Grimaldi, P. et al. The faah gene is the first direct target of estrogen in the testis: role of histone demethylase LSD1. Cell Mol Life Sci 69, 4177–4190, https://doi.org/10.1007/s00018-012-1074-6 (2012).
Ramirez-Lopez, M. T. et al. Exposure to a Highly Caloric Palatable Diet during the Perinatal Period Affects the Expression of the Endogenous Cannabinoid System in the Brain, Liver and Adipose Tissue of Adult Rat Offspring. PLoS One 11, e0165432, https://doi.org/10.1371/journal.pone.0165432 (2016).
Ramirez-Lopez, M. T. et al. Long-Term Effects of Prenatal Exposure to Undernutrition on Cannabinoid Receptor-Related Behaviors: Sex and Tissue-Specific Alterations in the mRNA Expression of Cannabinoid Receptors and Lipid Metabolic Regulators. Frontiers in behavioral neuroscience 10, 241, https://doi.org/10.3389/fnbeh.2016.00241 (2016).
Gandhi, K. et al. Effect of maternal high-fat diet on key components of the placental and hepatic endocannabinoid system. Am J Physiol Endocrinol Metab 314, E322–E333, https://doi.org/10.1152/ajpendo.00119.2017 (2018).
Cao, Z. et al. Monoacylglycerol lipase controls endocannabinoid and eicosanoid signaling and hepatic injury in mice. Gastroenterology 144, 808–817 e815, https://doi.org/10.1053/j.gastro.2012.12.028 (2013).
Ahmadi, A. & Shadboorestan, A. Oxidative stress and cancer; the role of hesperidin, a citrus natural bioflavonoid, as a cancer chemoprotective agent. Nutrition and cancer 68, 29–39, https://doi.org/10.1080/01635581.2015.1078822 (2016).
Jia, J. et al. Anandamide protects HT22 cells exposed to hydrogen peroxide by inhibiting CB1 receptor-mediated type 2 NADPH oxidase. Oxidative medicine and cellular longevity 2014, 893516, https://doi.org/10.1155/2014/893516 (2014).
Jorgacevic, B. et al. Rimonabant Improves Oxidative/Nitrosative Stress in Mice with Nonalcoholic Fatty Liver Disease. Oxidative medicine and cellular longevity 2015, 842108, https://doi.org/10.1155/2015/842108 (2015).
Batkai, S. et al. Cannabinoid-2 receptor mediates protection against hepatic ischemia/reperfusion injury. FASEB journal: official publication of the Federation of American Societies for Experimental Biology 21, 1788–1800, https://doi.org/10.1096/fj.06-7451com (2007).
Pacher, P. & Mechoulam, R. Is lipid signaling through cannabinoid 2 receptors part of a protective system? Progress in lipid research 50, 193–211, https://doi.org/10.1016/j.plipres.2011.01.001 (2011).
World Medical, A. & American Physiological, S. Guiding principles for research involving animals and human beings. Am J Physiol Regul Integr Comp Physiol 283, R281–283, https://doi.org/10.1152/ajpregu.00279.2002 (2002).
Fischbeck, K. L. & Rasmussen, K. M. Effect of repeated reproductive cycles on maternal nutritional status, lactational performance and litter growth in ad libitum-fed and chronically food-restricted rats. J Nutr 117, 1967–1975 (1987).
Aebi, H. Catalase in vitro. Methods in enzymology 105, 121–126 (1984).
Flohe, L. & Gunzler, W. A. Assays of glutathione peroxidase. Methods in enzymology 105, 114–121 (1984).
Crapo, J. D., McCord, J. M. & Fridovich, I. Preparation and assay of superoxide dismutases. Methods in enzymology 53, 382–393 (1978).
Fortunato, R. S. et al. Sexual dimorphism of thyroid reactive oxygen species production due to higher NADPH oxidase 4 expression in female thyroid glands. Thyroid: official journal of the American Thyroid Association 23, 111–119, https://doi.org/10.1089/thy.2012.0142 (2013).
Levine, R. L. et al. Determination of carbonyl content in oxidatively modified proteins. Methods in enzymology 186, 464–478 (1990).
Bligh, E. G. & Dyer, W. J. A rapid method of total lipid extraction and purification. Canadian journal of biochemistry and physiology 37, 911–917, https://doi.org/10.1139/o59-099 (1959).
Fortunato, R. S. et al. Functional consequences of dual oxidase-thyroperoxidase interaction at the plasma membrane. The Journal of clinical endocrinology and metabolism 95, 5403–5411, https://doi.org/10.1210/jc.2010-1085 (2010).
Acknowledgements
This work was supported by Fundação de Amparo à Pesquisa do Estado do Rio de Janeiro (FAPERJ) and Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq). We thank the Biology Institute of Universidade Federal Fluminense for allowing us to use the transmission electron microscope (JEM 1011) and the technical support biologist Alan Moraes for acquiring images. We also thank the technical support Juliana Pena Gonçalves and Luela Luam Pereira Dias da Mouta from Laboratory of Molecular Endocrinology, Carlos Chagas Filho Biophysics Institute, Federal University of Rio de Janeiro, Rio de Janeiro, Brazil.
Author information
Authors and Affiliations
Contributions
R.A.M., M.M.A., C.P.D.R. and I.H.T. have designed the experimental rat models and performed the biochemical, hormonal and molecular analyses; L.d.B.F. participated in the ovariectomy and western blotting experiments; L.L.S. and A.F.P.S have performed the thin-layer chromatography; C.B.V.A. performed mitochondria morphology analysis and picrosirius collagen staining; R.A.M., R.S.F. and A.F.P.S have performed the oxidative balance analyses; and R.A.M., R.S.F., C.C.P.M. and I.H.T. wrote and revised the final manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Miranda, R.A., De Almeida, M.M., Rocha, C.P.D. et al. Maternal high-fat diet consumption induces sex-dependent alterations of the endocannabinoid system and redox homeostasis in liver of adult rat offspring. Sci Rep 8, 14751 (2018). https://doi.org/10.1038/s41598-018-32906-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-32906-0
Keywords
This article is cited by
-
Fish oil supplementation during pregnancy decreases liver endocannabinoid system and lipogenic markers in newborn rats exposed to maternal high-fat diet
European Journal of Nutrition (2024)
-
Maternal high-fat diet up-regulates type-1 cannabinoid receptor with estrogen signaling changes in a sex- and depot- specific manner in white adipose tissue of adult rat offspring
European Journal of Nutrition (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.