Abstract
Sauropod dinosaurs were the largest terrestrial vertebrates; yet despite a robust global fossil record, the paucity of cranial remains complicates attempts to understand their paleobiology. An assemblage of small diplodocid sauropods from the Upper Jurassic Morrison Formation of Montana, USA, has produced the smallest diplodocid skull yet discovered. The ~24 cm long skull is referred to cf. Diplodocus based on the presence of several cranial and vertebral characters. This specimen enhances known features of early diplodocid ontogeny including a short snout with narrow-crowned teeth limited to the anterior portion of the jaws and more spatulate teeth posteriorly. The combination of size plus basal and derived character expression seen here further emphasizes caution when naming new taxa displaying the same, as these may be indicative of immaturity. This young diplodocid reveals that cranial modifications occurred throughout growth, providing evidence for ontogenetic dietary partitioning and recapitulation of ancestral morphologies.
Similar content being viewed by others
Introduction
With their titanic bodies and long necks and tails, sauropods are perhaps the most recognizable non-avian dinosaurs. Diplodocus is one of the best-known sauropod taxa, represented by over 100 specimens since its discovery in 18781. Whereas the postcrania of Diplodocus are well represented, cranial remains are exceedingly rare. An adult Diplodocus might attain a body length in excess of 30 m1, but its skull was well under 1 m2. To date only three of these skulls are hypothesized to be from immature animals3,4,5, thus biasing our understanding of this taxon’s ontogeny, ecology, and evolution towards adult specimens. While few, these immature skulls reveal insights into cranial allometry through ontogeny and suggest that Diplodocus and its Diplodocidae kin underwent radical ontogenetic change. Such changes would have significant effects on the ecology of immature Diplodocidae and the life history of these animals.
The smallest of the three immature skulls (CM 11255) is 29.2 cm in length, slightly over half of the adult cranial length3. Although this specimen reveals critical ontogenetic components, the cranial attributes of much younger diplodocids have remained unknown. Here we describe a new immature cf. Diplodocus skull (CMC VP14128), which, with a total cranial length of ~24 cm, represents the smallest known example. This important new specimen reveals hitherto unknown aspects of immature diplodocid anatomy, and shows that juveniles are not merely smaller versions of adults (sensu Whitlock et al.3). Our primary objectives are to test the taxonomic identity of the specimen using phylogenetic analyses, comparative qualitative and quantitative methods, and discuss the ecological implications of cranial ontogeny in diplodocids. We use immature and mature to refer specifically to developmental history, while juvenile, sub-adult, and adult are used as maturational colloquialisms.
Material and Methods
Specimens
CMC VP14128 was collected in 2010 from the Mother’s Day Quarry (MDQ) of south central Montana (MOR locality no. M-166). The MDQ is a monodominant bone bed containing the remains of at least sixteen small diplodocines6,7 (recorded femur lengths between 59.5 cm–120 cm5,8). While five partial braincases have previously been collected from the MDQ, CMC VP14128 is the only complete skull found at the site. The skull of CMC VP14128 is preserved in four major segments (Fig. 1). CMC VP14128 also preserves one half of the proatlas and at least four anterior cervical vertebrae that were clustered with the cranial remains. A second, similarly sized, isolated, and less distorted braincase (CMC VP14129) was also found in the opposite side of the same field jacket.
Skeletal reconstruction of CMC VP14128 to scale with a mature D. carnegii (dark grey). Grey bones are missing, while those in ivory are those present in CMC VP14128. Skeletal reconstruction based on the Diplodocus by S. Hartman. Silhouettes by S. Hartman and PhyloPic (Creative Commons Attribution-ShareAlike 3.0 Unported; http://phylopic.org/image/3cb1d5bf-7db5-4db2-82a6-4c39f6a4441b/; https://creativecommons.org/licenses/by-sa/3.0/), modifications made. Skeletal reconstruction of CMC VP14128 redrawn from D. carnegii skeletal by S. Hartman (http://www.skeletaldrawing.com/sauropods-and-kin/diplodocus). Human scale is Andrew Carnegie at his natural height of 1.6 m. Skeletal and silhouettes to scale. (B) CMC VP14128 in right lateral view with accompanying schematic. (C) CMC VP14128 in left lateral view with accompanying schematic. Schematics by DCW. The four portions of the skull numbered on accompanying schematics. Lateral views and schematics to scale. a: angular, al: alisphenoid, aof: antorbital fenestra, d: dentary, f: frontal, h: hyoid, l: lacrimal, m: maxilla, n: nasal, oc: occipital condyle, os: orbitosphenoid, p: parietal, paof: preantorbital fenestra, pf: prefrontal, pm: premaxilla, po: postorbital, pro: prootic, q: quadrate, sa: surangular, sq: squamosal. L and r before bone denotes if it is left or right.
Skull length estimates
Anteroposterior skull length was estimated using a linear regression of skull length to lower jaw length in diplodocid genera (Fig. S3). Scaling the anterior and posterior portions of the lower jaw of the immature Diplodocus SMM P84.15.3 results in a total jaw length of 19.9 cm, which indicates approximately 5 cm of missing cranial bone. As the cranial dimensions of smaller (and presumably younger) specimens do not develop isometrically (sensu Whitlock et al.3), from CMC VP14128 it appears that the surangular is proportionally smaller and that most of the lower jaw is represented. This approach produces a conservative estimate of 1 cm of missing bone, which results in a jaw length of 15.6 cm and a skull length of 24.3 cm. This shorter estimate is supported by the preservation of the paired dentaries, posterior portions of the jaw and associated ceratobranchial.
Transformation grid
A transformation grid (Fig. 2E) highlighting shape changes between CMC VP14128 and an adult Diplodocus was produced using the program tpsSplin8. Landmarks were placed on the line drawings of CMC VP14128 and CM 11255 presented in Fig. 2, using the programs tpsUtil9 and tpsDig26410 (see Fig. S4).
Cranial ontogeny in Diplodocus sp. (A) CMC VP14128; (B) CM 11255 (redrawn from Whitlock et al.3); (C) CM 11161 (redrawn from Wilson and Sereno69). Skull drawings by K. Scannella. Skulls to scale. (D) Silhouettes of CMC VP14128 and a mature D. carnegii to illustrate body length differences between skulls of A and C size. Diplodocus silhouette by S. Hartman and PhyloPic (Creative Commons Attribution-ShareAlike 3.0 Unported; http://phylopic.org/image/3cb1d5bf-7db5-4db2-82a6-4c39f6a4441b/; https://creativecommons.org/licenses/by-sa/3.0/), modifications made. Skeletal reconstruction of CMC VP14128 redrawn from D. carnegii skeletal by S. Hartman (http://www.skeletaldrawing.com/sauropods-and-kin/diplodocus). (E) Transformation grid highlighting the ontogenetic cranial changes. Adult skull is the same in part C (Wilson and Sereno69).
Phylogenetic analyses
We tested the taxonomic identity of CM VP14128 by including it in the phylogenetic analyses of Whitlock11 and Tschopp et al.12. As CMC VP14128 is predominantly represented by cranial material, in addition to the full analyses incorporating all skeletal characters, we also phylogenetically analyzed CMC VP14128 using only cranial characters. An approach such as thus is certainly not fool-proof, nor should the results be unanimously accepted to represent unequivocal relationships – we simply took this approach to examine what, if any, effects might occur from such extensive missing data. We also combined the matrices (excluding redundant characters) into a single data set – 540 characters for the cranial + postcranial matrix (115 scorable characters – 21% accounted for, 79% missing), and 153 characters for the cranial only matrix (98 scorable characters – 64% accounted for, 36% missing).
Although cranial and vertebral characters identify CMC VP14128 as a diplodocid, it possesses features seen in more inclusive clades, including an extended tooth row, proportionally small antorbital fenestra, and dorsoventrally tall maxilla and premaxilla, which are seen in derived Eusauropoda and basal Macronaria. Paradoxically, if portions of the skull and vertebrae (such as the spatulate teeth and extended tooth row versus the centrum which lack a strong ventral curvature and possess posteriorly elongate postzygapophyses, see below) of CMC VP14128 had been discovered separately, they could have been misidentified.
Furthermore, recent analyses indicate that diplodocids, and potentially all sauropods, underwent allometric growth, and consequently radical ontogenetic trajectories3,5,13,14. The documentation of specimens that fill voids in our understanding of sauropod ontogeny is critical. As Rozhdestvensky15 noted, derived immature individuals can appear more morphologically similar to basal adults than to their own adult form; thus, this developmental aphorism reveals the multifaceted importance of such specimens.
Within the Morrison Formation, some relatively small body size diplodocids have been used to establish new genera (including Suuwassea16 and Kaatedocus17), and the taxonomy of some has changed in different analyses (e.g. Suuwassea11,12,16,18,19,20). When examined using phylogenetic analyses, while there is more recent taxonomic consensus (again Suuwassea11,12,20) some previous analyses placed such taxa in more basal positions18,19. A similar phenomenon has also been seen in other dinosaurian clades21,22,23,24,25,26. Therefore, CMC VP14128 present the opportunity to assess the phylogenetic position of a demonstrably immature animal in a numerical cladistic analysis. Given the presence of plesiomorphic characters, we predict that it will be recovered in a position basal to Diplodocus. For this purpose, we used recent phylogenetic analyses as a starting point11,12 and analyzed the matrices using parsimony and Bayesian algorithms.
Institutional Abbreviations
AMNH, American Museum of Natural History, New York, NY; BYU, Brigham Young University, Provo, UT; CM, Carnegie Museum of Natural History, Pittsburgh, PA; CMC, Cincinnati Museum Center, Cincinnati, OH; HMNS, Houston Museum of Natural Science, Houston, TX; HMN, Humbolt Museum für Naturkunde, Berlin, Germany; MOR, Museum of the Rockies, Bozeman, MT; SMA, Sauriermuseum Aathal, Aathal, Switzerland; SMM, Science Museum of Minnesota, Saint Paul, MN; USNM, United States National Museum, Washington D.C.
Results
Description and comparisons
Skull
Most of the bones are identifiable, but portions of the maxillae, jugals, nasals, and lacrimals are damaged or missing.
The cranium of CMC VP14128 has diagnostic features of Diplodocidae including: a long prefrontal, paroccipital process with a rounded ventrolateral end, external nares that are retracted and dorsally-facing, a tooth row that does not extend the full length of the maxilla and dentary, low coronoid eminence, and absence of a squamosal-quadratojugal contact. Additionally, the morphology of the basal tubera (robust, triangular, and protruding posteroventrally from the basicranium), the presence of subnarial and maxillary foramina, and the largely peg-like teeth, while not exclusive to Diplodocidae, such morphologies and their combination are commonly observed in diplodocines.
To narrow down the taxonomic identity of CMC VP14128, we compared it with the four Morrison Formation genera of Diplodocinae: Barosaurus, Diplodocus, Galeamopus, and Kaatedocus. Diplodocus is known from eight complete skulls (CM 11255, CM 3452, CM 11161, MOR 7029, SMM P84.15.3, USNM 2672, USNM 2763), Kaatedocus from three skull (AMNH 7530 SMA 0004, SMA D-16/312,17), and Galeamopus is reported from at least three partial skulls (AMNH 969, SMA 0011, and HMNS 17512,27). Fragmentary cranial remains of Barosaurus are reported from the Howe Quarry12 and recently have been recovered from the Aaron Scott Site in the San Raphael Swell of Utah (CMC VP15544). A tooth row not restricted to the anteriormost portion of the skull is present in CMC VP14128, Kaatedocus (SMA 0004), and Galeamopus (SMA 0011). The premaxillae of the newest and smallest specimen, CMC VP14128, express the Massopoda condition of four teeth - seen in Diplodocus and Kaatedocus. In contrast, the reconstructed skull of the Galeamopus pabsti holotype has five27. The exact morphology of the prefrontal in CMC VP14128 is difficult to determine due to taphonomic distortion, but it appears to exhibit the typical diplodocid posterior hook27. The anterior portion of the antorbital fenestra seems to be dorsally situated to the preantorbital fossa as seen in CM 11255, Galeamopus12,27 and possibly in Kaatedocus17, although the damaged margin in CMC VP14128 makes this observation tentative. The posterior margin of the postorbital is gracile and more forked – as in Diplodocus and Kaatedocus, in contrast to less forked in Galeamopus12,27; whereas the dorsomedial process is long and tapered as in Diplodocus and Kaatedocus3,4,17, but not Galeamopus27. The squamosal in CMC VP14128 has a tapered and long anterior process approaching the quadrate, as in Diplodocus and Galeamopus, and not Kaatedocus – an autapomorphy of this genus17. Due to taphonomic damage and distortion, the morphology of the sagittal nuchal crest cannot be accurately discerned in CMC VP14128 (distinct and narrow vs. wide11,12). In contrast, a distinct crest is found in both Kaatedocus and Galeamopus hayi12,27, and CMC VP14129 does exhibit this feature (see Materials and Methods). Therefore the weight of evidence indicates a referral to Diplodocus over the other taxa.
Mandible
The relative proportions of the dentary and surangular are very similar to the lower jaw of the larger, and slightly more mature Diplodocus (CM 112553). Assuming similar proportions of mandibular bones in a mature Diplodocus to CMC VP14128, the skull would have been disproportionally stretched (see Material & Methods). Instead, CMC VP14128 indicates that in early ontogeny, the bones of the lower jaw were not directly proportionate to those of an adult (e.g. dentary to total jaw length). Regarding the dentary, the dorsoventral thickness post symphysis is more uniform as in Diplodocus and Kaatedocus3,12, opposed to strongly tapered as in Galeamopus27. One of the most interesting features of the lower jaw is the tooth row (Fig. 1B,C). As expressed in the upper jaw, the lower tooth row extends more posteriorly than seen in more mature specimens. In the right dentary of CMC VP14128, the tooth row (which is posteriorly obscured and damaged) is located along the anterior most ~1.5 cm. The left tooth row, however, clearly spans ~6.5 cm of the dentary; approximately 46% the length of the dentary compared to 22% in adults. Comparable to the adult condition, CM 11255 was hypothesized3 to have had 10–11 dentary teeth, whereas CMC VP14128 possesses 13. This dental variation may represent intraspecific variation28, considering that the dentary formula of CMC VP 14128 is the same as in immature camarasauromorphs28 and that ontogenetic dental formula reduction is documented in other dinosaurs29,30.
Dentition
The dental formula of CMC VP14128 is 4.8/13; the dental formula of the more mature CM 11255 has a formula of 4.8–9/10–11 dentary teeth, which is comparable to the adult condition of CM 11161 that has a formula of 4.9/11–1231.
The premaxillary teeth of CMC VP14128 exhibit the typical diplodocine condition: long, slightly inclined, pointed, and narrow-crowned – the so-called peg-like condition. However, from the second maxillary tooth posteriorly, the teeth are apicobasally short, with mesiodistally wide and more labial convex crowns. Several teeth have a Camarasaurus-like distal occlusal wear facet (Fig. 3). This relatively basal tooth morphology is consistent with the overall basal-expression form of the cranium.
The dental morphotypes in CMC VP14128. Pre- and maxillary teeth of CMC VP14128 in right and left lateral. Drawing by K. Scannella. Red outlines highlight the zoomed in views on the right. Note the combination of diplodocid peg and camarasaurid spatulate tooth forms. Camarasaurus sp. with the spatulate tooth form (SMA 0002). Diplodocus longus with the peg tooth form (USNM 2672). Camarasaurus and Diplodocus skull modified from McIntosh70. Skulls not to scale.
Maturational state of CMC VP 14128
CMC VP14128 establishes the immature condition for many features of the skull, jaws, dentition, and anterior cervical vertebrae far beyond what has been previously known. We summarize those changes here, and make note of immature features that correspond to the plesiomorphic character states of Diplodocoidea (for a greater discussion on the possibility of ontogenetic recapitulation, please consult the Supplementary Information).
Size
The estimated skull length of CMC VP14128 is 24.29 cm, which is ~40% the length of the largest adult Diplodocus skull (USNM 2673, ~60 cm). Cranial size differences observed between CMC VP14128 and adult skulls attest to changing body size through ontogeny (Fig. 2). The cranial size difference between CMC VP14128 and CM 11255 – ~5 cm – seems minor, yet the understanding of sauropodomorph paleobiology is dependent on their relative scale. Understanding minor skeletal nuances can have vast ontogenetic repercussions. A mere 25 cm difference in femoral length separates a 6 m 6-year-old from a 27 m 24-year-old Diplodocus (5).
While we do not have complete Diplodocus specimens, we have composites and referable material enabling us to draw some conclusions about adult proportions. Specifically, here we use D. carnegii CM 84 which is a composite, but represents the informal standard for the genus32. Nevertheless, using a ratio from CM 84 assumes isometric growth – contrary to the ontogenetic record of Dinosauria – therefore we should view the resulting estimates as nothing more than generalized proportions.
Using this adult cranial:body length ratio predicts a maximum body length of 9 m for CMC VP14128 and 10.9 m for CM 11255, a difference that would be even greater with a more realistic allometric skull-body length ratio; yet even this isometric trend indicates a minimum difference of nearly 2 m in body length, expressed in ~5 cm of cranial length-difference. While we await more specimens to fill in these crucial ontogenetic intervals, assuming size covaries with age at this locality (see Discussion), we hypothesize that CMC VP14128 was within the recorded MDQ ages of two – six years of age5 and had a body length well under the isometrically calculated 9 m (Fig. 1).
Tooth count
CMC VP14128 has a high dentary tooth count – 13 – in contrast to the lower tooth count – 11 – seen in larger, presumably more mature specimens, such as CM 11161 (see description above). This variation in tooth count, while limited in sample size, may be indicating a trend of dentary tooth count reduction, which is seen in other immature dinosaurs29,30,33. Therefore, the high dentary tooth count of CMC VP14128 indicates its juvenile growth stage. Also, basal eusauropods tend to have a dentary tooth count higher than 1134,35.
Neurocentral synostosis
Three cervical vertebrae with neural arches are preserved with CMC VP14128. In two of the cervical vertebrae (labeled 2 and 3 in Fig. S1) the arches are completely separate from their centra. One vertebra (labeled 1 in Fig. S1) has a fused arch with sutural contacts that are seen on the anterior- and posterior-most margins. The corresponding sutures in a skeletally mature Diplodocus (such as CM 84) are completely closed.
Cervical centrum pneumatization
The cervical centra of CMC VP14128 are excavated by shallow, simple, and weakly divided fossae, typical for young animals, as compared to highly complex fossae and foramina with numerous accessory laminae in adults such as D. carnegii CM 845,13,36,37,38.
Cervical rib histology
In the absence of chronologically-informative bones (such as sauropod dorsal ribs that provide an almost complete growth record39,40,41), a cervical rib of CMC VP14128 was sectioned to obtain an estimate of the relative maturity of the specimen based on patterns of remodeling – in like manner to the Histologic Ontogenetic Stage42. However, we must cautiously note that the origin and development of cervical ribs is still ongoing research (JRH and DCW in prep.). As cervical ribs incorporate a complex developmental relationship of metaplastic and osteogenic processes43,44, at this time we should only compare rates of secondary remodeling (Fig. S2).
Progressing through maturity in diplodocid cervical ribs, there are dramatic changes in tissue composition. In the smallest specimen (SMA 0009) the tissue is composed entirely of highly vascular primary tissue. Progressing to CMC VP14128 the tissue is composed of secondary reconstructions and primary tissue – features indicative of metaplasia45. Finally, within a sub-adult Diplodocus (MOR 592), the tissue consists of regular bony tissues - a core of Haversian bone, periosteally grading from secondary to primary osteons (see greater discussion in the Supplementary Material).
While the ontogenetic development of cervical ribs must be studied in further detail, this analysis supports the hypothesis that they develop via metaplasia from a collagenous to an osseous tissue43,44. Thus, the cervical rib of CMC VP14128, conforming to this developmental pathway, further supports our maturational interferences of immaturity for this specimen.
Systematic Paleontology
Saurischia Seeley 1887
Sauropodomorpha von Huene 1932
Sauropoda Marsh 1878
Diplodocoidea Marsh 1884
Flagellicaudata Harris and Dodson 2004
Diplodocidae Marsh 1884
cf. Diplodocus Marsh 1878.
Comparative description
The Morrison Formation preserves three sauropod clades: Diplodocoidea, Camarasauridae, and Brachiosauridae. The lack of only spatulate teeth, an inclined posterior portion of the premaxilla, projecting external naris, cervical ribs shorter than centrum, and rectangular not rhomboidal cervical vertebrae profiles in CMC VP14128 are more diplodocoid than macronarian morphologies. Although many of the diagnostic characters in the sauropod skull are proportionally or ontogenetically variable46 some of the morphologies within CMC VP14128 are different from those expressed in the adult. While a few traits could be outside of the typical adult expressions, we hypothesize that characters/conditions of CMC VP14128 will at least largely associate with a known genus (as previously demonstrated in Europasaurus47).
CMC VP14128 is referable to Diplodocidae based on the presence of: a long posterior process of the prefrontal, teeth that do not span the length of the maxilla and dentary, a low coronoid eminence on the mandible, lack of crown-to-crown occlusion, cervical rib length that is shorter than the corresponding centrum length, and external nares that are retracted and face dorsally. CMC VP14128 is not referable to Apatosaurus given the presence of a basipterygoid recess, a basipterygoid process that lacks an anteroventral flare, and enlarged cervical ribs that do not project below the centrum. Likewise, CMC VP14128 is not referable to Barosaurus, based on the presence of long postzygapophyses, anteroposteriorly narrow neural spines, and a strongly posteriorly-angled centrum cotyles. CMC VP14128 has its strongest affinities with the slender diplodocines, including Diplodocus, Galeamopus, and Kaatedocus; however, the distribution of shared features is inconsistent, obscuring its lower-level identity. However, based on the number of shared characteristics, CMC VP14128 is most referable to Diplodocus than either Galeamopus or Kaatedocus.
However, it must be stated that specimens previously assigned to Diplodocus, and how we phylogenetically recognize and identify this genus are being reexamined12,32. Some historically recognized Diplodocus specimens are now being referred to other genera – such as USNM 2673 possibly representing Galeamopus12 and even CM 11255 to Barosaurus48. Additionally, some Morrison Formation taxa have little to any known or described cranial material (Barosaurus, Dystrophaeus, Haplocanthosaurus, Supersaurus, Suuwassea), therefore there are several taxa we cannot adequately compare CMC VP14128 to or assess. Additionally, while Whitlock11 identified three cranial autapomorphies for Diplodocus (preantorbital fenestra with well-defined fossa, pterygoid medial to ectopterygoid on transverse palatal hook, teeth inclined anteriorly relative to axis of jaw) since no skulls to date are unquestionably associated with post-crania, Tschopp et al.12 questioned these characters. While the latest phylogenetic analysis of Diplodocidae would advocated that no unambiguous diplodocinae cranial synapomorphies are recognized12, the exact taxonomic assignment of CMC VP14128 within Diplodocinae remains uncertain. With the current lack of no known diplodocinae synapomorphies12, one could taxonomically identify CMC VP14128 simply as diplodocinae indeterminate. However, given the predominance of similar morphologies between CMC VP14128 and Diplodocus sp. in comparison to the other Morrison Formation diplodocids, we tentatively opt to refer CMC VP14128 to cf. Diplodocus. While both identifications (diplodocinae indeterminate and cf. Diplodocus) are testable hypotheses, we currently believe that it is more fruitful and more constructive for future works to support/refute our identification of CMC VP14128 as cf. Diplodocus versus reanalyzing starting from a subfamily level identification.
Phylogenetic analyses
Separate data sets
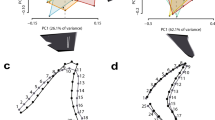
The cranial + postcranial parsimony and Bayesian analyses of the Whitlock11 data set recovered CMC VP14128 as the basalmost member of Dicraeosauridae, while the cranial only analysis recovered CMC VP14128 as the sister species of Dicraeosauridae, and Dicraeosaurus + Amargasaurus, respectively. The cranial + postcranial parsimony and Bayesian analyses of the Tschopp et al.12 data set recovered CMC VP14128, as a diplodocine more derived than Diplodocus, while the cranial only analysis recovered CMC VP14128 in a polytomy within Flagellicaudata.
Combined data set
In the combined cranial + postcranial parsimony and Bayesian typology, there is a degree of basal uncertainty (no united Macronaria), yet there is a structured and organized Diplodocoidea (Fig. 4). In the parsimony typology, CMC VP14128 is recovered as the sister species to Diplodocus, while in the Bayesian analysis, it is recovered as a derived member of Flagellicaudata and basal to Diplodocidae. For the combined cranial only analyses, the typologies are similar to that of the cranial + postcranial analyses. There are distinct Rebbachisauridae, Dicraeosauridae, and Diplodocidae branches, and CMC VP14128 is recovered as the sister taxon to Diplodocus. Apart from minor changes in posterior probability, the Bayesian topology is very similar to the earlier analysis12. It likewise recovers CMC VP14128 in a flagellicaudatan polytomy in which Dicraeosauridae forms one branch. Yet we would caution that while these two analyses superficially produce similar results, the low support for groupings indicates that these relations are not definitive.
Dendrograms of parsimony (left column) and Bayesian (right column) phylogenetic analyses. (A–C) Consist of cranial and postcranial characters, while (D,E) consist of only cranial characters. (A and D) CMC VP14128 coded into the matrix of Whitlock11. (B and E) CMC VP14128 coded into the matrix of Tschopp et al.12. (C and F) CMC VP14128 coded into a combined matrix.
The minutia between the parsimony and Bayesian analyses do vary, but in general the encompassing skeletal analyses do little to elucidate the taxonomic identify of CMC VP14128. In the analysis of Whitlock11, CMC VP14128 is recovered in the same dicraeosaurid position, while in the analysis of Tschopp et al.12 taxonomy is slightly more refined as CMC VP14128 is recovered as a derived diplodocine versus a flagellicaudatan polytomy, and finally in the combined analysis, CMC VP14128 is still recovered as a diplodocoid. Certainly the encompassing analyses do reveal much more overall taxonomic resolution, but said resolution is largely irrespective to CMC VP14128. A cranial only approach could appear redundant or unnecessary, yet given the vastly differing phylogenetic placements between the matrices used herein, we would suggest that an elemental or regional styled analyses can be used to further check or verify specimens that are recovered in seemingly unusual or suspect positions.
Discussion
Phylogeny
Parsimony analyses of CMC VP14128 produced a conflict. One resulted in only a generic level association with Diplodocus, while the other advocated a unique basal position. If our cranial-only analyses represent true phylogenetic patterns, then relatively small morphological matrices (in terms of number of characters, percentage of each character region, and/or taxa) can be sensitive to inclusion of juvenile specimens5, as demonstrated by the more basal positioning of CMC VP14128. This is expected as even a few characters that change with ontogeny will have a large effect on the outcome of the analysis. In the larger12 and combined analyses, CMC VP14128 is recovered amongst other flagellicaudatans (including diplodocids), albeit in large polytomies, agreeing with our previous diagnosis. This suggests including additional characters can override the effect of a few characters that are ontogenetically biased, but as paleontological phylogenetic analyses are often based on small datasets, this bias can be significant.
In regard to the plesiomorphic characters in CMC VP14128, we theorize these might be evidence of recapitulation; i.e., the ephemeral presence of plesiomorphic characters that transition into their derived states by adulthood. Recapitulation has been documented in other basal49,50 and derived47,51,52,53 immature sauropods. Morphologies in CMC VP14128 – such as the posterior tooth row, lower jaw proportions, and rounded snout – could be recapitulatory in origin (see Supplementary Material); yet given the relationship between cranial attributes and feeding strategy, these morphologies could also derive from ontogenetic ecomorphological relationships (see below). More specimens and analyses are need to substantiate either possibility, and we note that these hypotheses are not mutually exclusive.
Despite the presence in CMC VP14128 of phylogenetically plesiomorphic features that are not seen in adult Diplodocus, this does not mean that the entire skeleton of CMC VP14128 is plesiomorphic, (or that it is a misidentified basal taxon), since the specimen does have characters of Diplodocus. Instead, this combination of characters is evidence that 1: not all parts of the skeleton develop at the same rate, and 2: regardless of rate, ontogenetic skeletal changes followed the phylogenetic progression from plesiomorphic to derived states. Recognition of this phenomenon strongly cautions against diagnosing new taxa based on small statured specimens displaying a combination of basal and derived characters, which might instead be evidence of immaturity. Interestingly, similar findings were reported by Tschopp et al.12 regarding “Elosaurus” parvus (CM 566). While originally thought to be a valid taxon, “Elosaurus” is now regarded as an immature Brontosaurus parvus12 (however, while this analysis believes this maturational inference is highly likely, we cautiously note that this hypothesis has not been histologically tested). Including CM 566 into a specimen-level analysis, Tschopp et al.12 recovered the specimen not only within the species B. parvus, but also as the basalmost specimen. We would agree with Tschopp et al.12 that more studies are needed to address this issue, and this further highlights the multifaceted relationship between ontogenetically variable characters and taxonomic recovery.
Ecomorphology and behavior implications
The unique cranial and dental characters seen in the immature CMC VP14128 suggests resource partitioning between juvenile and adult Diplodocus. Like the immature Diplodocus CM 112553, CMC VP14128 has a mediolaterally narrow snout, in stark contrast to more mature individuals that express a wide and squared snout. These differences have been hypothesized to indicate dietary niche-partitioning through ontogeny3, and the narrow snout of CMC VP14128 is consistent with the hypothesis that immature diplodocids had a selective feeding strategy, while fully mature animals were ground-level browsers3. CMCVP14128 also has spatulate teeth and an extended tooth row. Spatulate teeth are more efficient for coarse vegetation and bulk feeding, and non-spatulate teeth are beneficial for softer foliage and browsing54,55. This combination of dental morphologies in CMC VP14128 may indicate that very immature diplodocids were feeding on a greater variety of plant materials, and orally processed them differently than their more mature counterparts.
However, previous studies show that nutrition selectivity is problematic in animals exceeding a certain size and that there is a selection pressure towards browsing for large herbivores56,57. Another recent analysis58 showed that narrow, short, and rounded macronarian snouts were more efficient for forested environments while broader and longer diplodocoid snouts suggest open, ground-level browsing. This could also be interpreted as indicators of different ecological environments, implying macronarians would have been adapted for more forested environments while diplodocids were more specialized for open environments. Therefore, the plesiomorphic macronarian-like characters seen in CMC VP14128 could also indicate that juvenile diplodocids lived in more forested environments than the adults that (restricted and protected by their size) were most likely browsing in more open habitats. Foraging in forests would have provided the juveniles with protective cover from predators, a danger that colossal adults would not share.
The skull and tooth morphology of Diplodocus suggests that these animals transitioned through distinct feeding roles over their lifespan. This inference is supported by a study of Alligator mississippiensis tooth change through ontogeny59. Dental changes coincided with changes in diet59,60,61, thus dental allometry could be attributed to dietary partitioning.
Though it is currently unknown which specific plant species Morrison Formation sauropods ate, based on δ13C relationships between modern plant equivalents and tooth enamel implies that Diplodocus may have fed on ferns and horsetail, while Camarasaurus was generalized, feeding on a wide range of foliage, including ferns, horsetails, conifers, and cycads62. An immature Diplodocus may have fended for itself (or possibly as part of an age-segregated herd63) and fed on differing foliage to gain more nutrients during critical development.
The ontogenetic change in dental morphologies observed within Diplodocus also gives evidence for the lack of parental care in sauropods (along with nest structures and histology14,64). If adults fed hatchlings - potentially the foliage they ate - then there would be no reason for the changes in tooth morphology. Evidence of precocial juvenile sauropods was also found in a recent histologic assessments of a very immature titanosaur. Furthermore, the extreme size differences between parent and hatchlings could have resulted in high infant mortalities due to trampling.
The differences in cranial morphology between CMC VP14128 and more mature Diplodocus specimens (such as CM 11255; Whitlock et al.3) highlights extreme cranial changes that occurred rapidly over a short increase in size. In addition to the narrow snout, the proportionally enlarged braincase and extremely large orbits are both infantile attributes observed in many other immature vertebrates. The more box-like cranial condition and the extended tooth row in CMC VP14128 are reminiscent of the co-occurring camarasaurid condition. If these characters represents recapitulation, we could hypothesize that dietary generalization is the more basal condition and increasing specialization the more derived.
Possible dwarfism?
Instead of a juvenile, CMC VP14128 could represent an unknown Morrison Formation dwarf taxon. Two separate investigations of age determinant histology and morphology of the MDQ material produced different results5,41. The bone tissue types in the first analysis indicate average sized immature diplodocids with typically expressed tissues5, yet many of the specimens of the second analysis exhibit tissues indicating skeletal maturity41. In comparing histology, this is not simply the case of incorrect tissue interpretations (DCW and KW pers. obs.) – the varying tissue morphologies indicate a more complex story for the MDQ.
The regional geology does not support the presence of an island depositional system – an extensive marine unit with localized terrestrial deposits. Nevertheless, regional geography or environment could explain a possible size difference. In the case of stegosaur specimens from the northern extent of the Morrison Formation, larger overall size (Stegosaurus) appears correlated with arid environments, and smaller size with wetter climates (Hesperosaurus65). Based on the coastal setting adjacent to the Sundance Seaway, this trend in stegosaur size was hypothesized to be environmentally correlated65. A similar pattern might hold true for the MDQ diplodocids.
Other Morrison Formation localities in Montana record more typical sized specimens of Apatosaurus and Diplodocus5,66. This regional size difference, and size difference within the MDQ41, may represent regional variation. Such regional differences are present in a modern group of herbivores, cervids. The North American coastal White-tailed Deer (Odocoileus virginianus) have an average body mass 77 kg, while their interior counterparts have an average body mass in excess of 100 kg67. The smallest sub-species, the Key Deer (Odocoileus virginianus clavium) represents an example of island dwarfing has an average body mass of 34 kg67. Therefore, these size differences between localities may indicate variably-sized regional diplodocid populations. In spite of these findings CMC VP14128 is almost certainly not a regional dwarf because it lacks autapomorphies. The morphologies we assigned to ontogeny could hypothetically be used to erect autapomorphies, however we strongly caution that the onus is on such an analysis to demonstrate that no attributes are related to ontogenetic variation as proposed herein. Alternatively, opposed to the dwarf morphotype observed by Waskow et al.41, CMC VP14128 may represent the larger morph present at the site.
Conclusions
Within Dinosauria, there are small bodied taxa that display basal and derived characters and occupy unusual basal phylogenetic positions. The validity and position of such taxa has been disputed21,22,23,24,25,26,68,69, and regarding sauropodomorph phylogeny, we would advocate that the combination of basal and derived characters and basal phylogenetic recovery should be recognized as an indicator of an immature ontogimorph – instead of a distinct taxon. In light of the current wealth of information pertaining to dinosaur ontogeny, we can no longer assume that all morphological differences correspond with phylogenetic distinctiveness. Accounting for ontogeny could prove as test for our phylogenies. Recognizing the ontogenetic age of immature specimens provides important insights into the life history of these animals. The immature Diplodocus specimen CMC VP14128 extends our understanding of the ontogeny of the genus and the evolution of diplodocids into new areas, where:
-
(1)
The combination of basal and derived characters in the juvenile is broadly congruent with the phylogenetic transition from eusauropods to diplodocoids.
-
(2)
The plesiomorphic tooth morphology is retained in immature Diplodocus and lost with maturity, and we predict this growth pattern will be seen in all other diplodocoids.
-
(3)
As first proposed by Whitlock et al.3, tooth and skull morphology indicate that during growth Diplodocus inhabited different trophic levels/niches, where juveniles were generalists (i.e., browsers; Fig. 5) and more mature individuals were specialists (i.e., ground-level browsing), a pattern that we predict is ancestral for Diplodocoidea.
Figure 5 Life reconstruction of CMC VP14128. Note the cranial morphologies interpreted to denote differing feeding strategies: in CMC VP14128 the narrow snout with posteriorly elongated and morphologically varied tooth row for bulk feeding vs. the widened snout with anteriorly restricted peg-shaped teeth for ground-level browsing in adults. Also note the camouflaged ontogenetic color change suggesting young diplodocids may have sought forested refuge. Reconstruction by A. Atuchin.
References
Foster, J. Jurassic West: The Dinosaurs of the Morrison Formation and their World. (Indiana University Press, 2007).
Berman, D. & McIntosh, J. S. Skull and relationships of the Upper Jurassic sauropod Apatosaurus (Reptilia, Saurischia). Ann. Carnegie Museum 8, 1–35 (1978).
Whitlock, J. A., Wilson, J. A. & Lamanna, M. C. Description of a nearly complete juvenile skull of Diplodocus (Sauropoda: Diplodocoidea) from the Late Jurassic of North America. J. Vertebr. Paleontol. 30, 442–457 (2010).
Erickson, B. History of the Poison Creek expedition 1976–1990: With description of Haplocanthosaurus postcranials and a subadult diplodocid skull. Sci. Museum Minnesota Monogr. 8, 1–34 (2014).
Woodruff, D. C., Fowler, D. W. & Horner, J. R. A new multi-faceted framework for deciphering diplodocid ontogeny. Palaeontol. Electron. 20.3.43A, 1–53 (2017).
Myers, T. S. & Storrs, G. W. Taphonomy of the Mother’s Day Quarry, Upper Jurassic Morrison Formation, south-central Montana, USA. Palaios 22, 651–666 (2007).
Storrs, G. W., Oser, S. E. & Aull, M. Further analysis of a Late Jurassic dinosaur bone-bed from the Morrison Formation ofMontana, USA, with a computed three-dimensional reconstruction. Earth Environ. Sci. Trans. R. Soc. Edinburgh 103, 443–458 (2013).
Rohlf, F. TpsSplin ver 1.22. Stony Brook, NY (2016).
Rohlf, F. TpsUtil ver. 1.64. Stony Brook, NY (2013).
Rohlf, F. J. tpsDig2, digitize landmarks and outlines, version 2.10. Dep. Ecol. Evol. State Univ. New York Stony Brook (2015).
Whitlock, J. A. A phylogenetic analysis of Diplodocoidea (Saurischia: Sauropoda). Zool. J. Linn. Soc. 161, 872–915 (2011).
Tschopp, E., Mateus, O. & Benson, R. B. J. A specimen-level phylogenetic analysis and taxonomic revision of Diplodocidae (Dinosauria, Sauropoda). PeerJ 3, e857 (2015).
Carballido, J. L. & Sander, P. M. Postcranial axial skeleton of Europasaurus holgeri (Dinosauria, Sauropoda) from the Upper Jurassic of Germany: Implications for sauropod ontogeny and phylogenetic relationships of basal Macronaria. J. Syst. Palaeontol. 12, 335–387 (2014).
Rogers, K. C., Whitney, M., D’Emic, M. & Bagley, B. Precocity in a tiny titanosaur from the Cretaceous of Madagascar. Science. 352, 450–453 (2016).
Rozhdestvensky, A. K. Growth changes in Asian dinosaurs and some problems of their taxonomy. Paleontol. žurnal 3, 95–109 (1965).
Harris, J. D. The significance of Suuwassea emilieae (Dinosauria: Sauropoda) for flagellicaudatan intrarelationships and evolution. J. Syst. Palaeontol. 4, 185–198 (2006).
Tschopp, E. & Mateus, O. The skull and neck of a new flagellicaudatan sauropod from the Morrison Formation and its implication for the evolution and ontogeny of diplodocid dinosaurs. J. Syst. Palaeontol. 11, 853–888 (2013).
Rauhut, O. W. M., Remes, K., Fechner, R., Cladera, G. & Puerta, P. Discovery of a short-necked sauropod dinosaur from the Late Jurassic period of Patagonia. Nature 435, 670–672 (2005).
Lovelace, D. M., Hartman, S. A. & Wahl, W. R. Morphology of a specimen of Supersaurus (Dinosauria, Sauropoda) from the Morrison Formation of Wyoming, and a re-evaluation of Diplodocid phylogeny. Arq. do Mus. Nac. 65, 527–544 (2007).
Whitlock, J. A. & Harris, J. D. The dentary of Suuwassea emilieae (Sauropoda: Diplodocoidea). J. Vertebr. Paleontol. 30, 1637–1641 (2010).
Carr, T. D. & Williamson, T. E. Diversity of late Maastrichtian Tyrannosauridae (Dinosauria: Theropoda) from western North America. Zool. J. Linn. Soc. 142, 479–523 (2004).
Fowler, D. W., Woodward, H. N., Freedman, E. A., Larson, P. L. & Horner, J. R. Reanalysis of ‘Raptorex kriegsteini’: A juvenile tyrannosaurid dinosaur from Mongolia. PLoS One 6 (2011).
Campione, N. E., Evans, D. C. Jr, J, S., Cuthbertson, R. & Holliday, C. Cranial growth and variation in Edmontosaurs (Dinosauria: Hadrosauridae): implications for latest Cretaceous megaherbivore diversity in North America. PLoS One 6, e25186 (2011).
Frederickson, J. A. & Tumarkin-Deratzian, A. R. Craniofacial ontogeny in Centrosaurus apertus. PeerJ 2, e252 (2014).
Scannella, J. B., Fowler, D. W., Goodwin, M. B. & Horner, J. R. Evolutionary trends in Triceratops from the Hell Creek Formation, Montana. Proc. Natl. Acad. Sci. 111, 10245–10250 (2014).
Fowler, E. A. F. & Horner, J. R. A new brachylophosaurin hadrosaur (Dinosauria: Ornithischia) with an intermediate nasal crest from the Campanian Judith River Formation of northcentral Montana. PLoS One 10 (2015).
Tschopp, E. & Mateus, O. Osteology of Galeamopus pabsti sp. nov. (Sauropoda: Diplodocidae), with implications for neurocentral closure timing, and the cervico-dorsal transition in diplodocids. PeerJ 5, e3179 (2017).
Gilmore, C. W. A nearly complete articulated skeleton of Camarasaurus, a saurischian dinosaur from the Dinosaur National Monument, Utah. Mem. Carnegie Museum (1925).
Carr, T. D. Craniofacial ontogeny in Tyrannosauridae (Dinosauria, Coelurosauria). J. Vertebr. Paleontol. 19, 497–520 (1999).
Carr, T. D., Varricchio, D. J., Sedlmayr, J. C., Roberts, E. M. & Moore, J. R. A new tyrannosaur with evidence for anagenesis and crocodile-like facial sensory system. Sci. Rep. 7 (2017).
Yu, C. The skull of Diplodocus and the phylogeny of the Diplodocidae. PhD thesis, (University of Chicago, 1993).
Tschopp, E. & Mateus, O. Case 3700: Diplodocus Marsh, 1878 (Dinosauria, Sauropoda): proposed designation of D. carnegii Hatcher, 1901 as the type species. Bull. Zool. Nomencl. 73, 17–24 (2016).
Wang, S. et al. Extreme ontogenetic changes in a ceratosaurian theropod. Curr. Biol. 27, 144–148 (2017).
He, X., Li, K. & Cai, K. The Middle Jurassic dinosaur fauna from Dashanpu, Zigong, Sichuan: Omeisaurus tianfuensis. Sauropod dinosaurs (2). Sichuan Publ. House Sci. Technol (1988).
Ouyang, H. & Ye, Y. The first mamenchisaurian skeleton with a complete skull Mamenchisaurus youngi. Sichuan Publ. House Sci. Technol (2002).
Wedel, M. J. The evolution of vertebral pneumaticity in sauropod dinosaurs. J. Vertebr. Paleontol. 23, 344–357 (2003).
Wedel, M. J. Vertebral pneumaticity, air sacs, and the physiology of sauropod dinosaurs. Paleobiology 29, 243–255 (2003).
Wedel, M. J. In The sauropods: evolution and paleobiology (eds Rogers, K. C. & Wilson, J.) 201–228 (University of California Press, 2005).
Waskow, K. & Sander, P. M. Growth record and histological variation in the dorsal ribs of Camarasaurus sp. (Sauropoda). J. Vertebr. Paleontol. 34, 852–869 (2014).
Waskow, K. & Mateus, O. Dorsal rib histology of dinosaurs and a crocodylomorph from western Portugal: Skeletochronological implications on age determination and life history traits. Comptes Rendus Palevol 16, 425–439 (2017).
Waskow, K., Wiersma, K., Tschopp, E., Woodruff, D. C. & Sander, P. M. Histological evidence for dwarfism and slow growth in Late Jurassic diplodocoid sauropods from the Upper Jurassic Mother’s Day Quarry (Morrison Formation, Montana, USA). Paleobiology.
Klein, N. & Sander, M. Ontogenetic stages in the long bone histology of sauropod dinosaurs. Paleobiology 34, 247–263 (2008).
Cerda, I. A. et al. Novel insight into the origin of the growth dynamics of sauropod dinosaurs. PLoS One 12 (2017).
Klein, N., Christian, A. & Sander, P. M. Histology shows that elongated neck ribs in sauropod dinosaurs are ossified tendons. Biol. Lett. 8, 1032–1035 (2012).
Horner, J. R., Woodward, H. N. & Bailleul, A. M. Mineralized tissues in dinosaurs interpreted as having formed through metaplasia: A preliminary evaluation. Comptes Rendus - Palevol 15, 183–203 (2016).
Chure, D., Britt, B. B., Whitlock, J. A. & Wilson, J. A. First complete sauropod dinosaur skull from the Cretaceous of the Americas and the evolution of sauropod dentition. Naturwissenschaften 97, 379–391 (2010).
Marpmann, J. S., Carballido, J. L., Sander, P. M. & Knötschke, N. Cranial anatomy of the Late Jurassic dwarf sauropod Europasaurus holgeri (Dinosauria, Camarasauromorpha): Ontogenetic changes and size dimorphism. J. Syst. Palaeontol. 13, 221–263 (2015).
Melstrom, K. M., D’emic, M. D., Chure, D. & Wilson, J. A. A juvenile sauropod dinosaur from the Late Jurassic of Utah, U.S.A., presents further evidence of an avian style air-sac system. J. Vertebr. Paleontol. 36 (2016).
Reisz, R. R., Scott, D., Sues, H. D., Evans, D. C. & Raath, M. A. Paleontology: Embryos of an early Jurassic prosauropod dinosaur and their evolutionary significance. Science (80-.). 309, 761–764 (2005).
Reisz, R. R., Evans, D. C., Sues, H. D. & Scott, D. Embryonic skeletal anatomy of the sauropodomorph dinosaur Massospondylus from the lower Jurassic of South Africa. J. Vertebr. Paleontol. 30, 1653–1665 (2010).
Chiappe, L. M., Salgado, L. & Coria, R. A. Embryonic skulls of titanosaur sauropod dinosaurs. Science 293, 2444–2446 (2001).
García, R. A. & Cerda, I. A. Dentition and histology in titanosaurian dinosaur embryos from Upper Cretaceous of Patagonia, Argentina. Palaeontology 53, 335–346 (2010).
García, R. A. et al. Paleobiology of titanosaurs: Reproduction, development, histology, pneumaticity, locomotion and neuroanatomy from the South American Fossil Record. Ameghiniana 52, 29–68 (2015).
Fiorillo, A. R. Dental microwear patterns of the sauropod dinosaurs Camarasaurus and Diplodocus: evidence for resource partitioning in the Late Jurassic of North America. Hist. Biol. 13, 1–16 (1998).
D’Emic, M. D., Whitlock, J. A., Smith, K. M., Fisher, D. C. & Wilson, J. A. Evolution of High Tooth Replacement Rates in Sauropod Dinosaurs. PLoS One 8 (2013).
Hummel, J. et al. In vitro digestibility of fern and gymnosperm foliage: implications for sauropod feeding ecology and diet selection. Proc. R. Soc. B Biol. Sci. 275, 1015–1021 (2008).
Westoby, M. An Analysis of Diet Selection by Large Generalist Herbivores. Am. Nat. 108, 290–304 (1974).
Whitlock, J. A. Inferences of diplodocoid (Sauropoda: Dinosauria) feeding behavior from snout shape and microwear analyses. PLoS One 6 (2011).
Gignac, P. M. Biomechanics and the ontogeny of feeding in the American alligator: reconciling factors contributing to intraspecific niche differentiation in a large-bodied vertebrate. (Florida State University, 2010).
Dodson, P. Functional and ecological significance of relative growth in Alligator. J. Zool. 175, 315–355 (1975).
Bailleul, A. M., Scannella, J. B., Horner, J. R. & Evans, D. C. Fusion patterns in the skulls of modern archosaurs reveal that sutures are ambiguous maturity indicators for the Dinosauria. PLoS One 11 (2016).
Tütken, T. In Biology of the Sauropod dinosaurs: understanding the life of giants (eds Klein, N., Remes, K., Gee, C. T. & Sander, P. M.) 57–79 (Indiana University Press, 2011).
Myers, T. S. & Fiorillo, A. R. Evidence for gregarious behavior and age segregation in sauropod dinosaurs. Palaeogeogr. Palaeoclimatol. Palaeoecol. 274, 96–104 (2009).
Chiappe, L. M. et al. Nest Structure for Sauropods: Sedimentary Criteria for Recognition of Dinosaur Nesting Traces. Palaios 19, 89–95 (2004).
Maidment, S. C. R., Woodruff, D. C. & Horner, J. R. A new specimen of the ornithischian dinosaur Hesperosaurus mjosi from the Upper Jurassic Morrison Formation ofMontana, USA, and implications for growth and size in Morrison stegosaurs. J. Vertebr. Paleontol. e1406366 (2018).
Woodruff, D. C. & Rogers, K. C. Diplodocid Sauropods From The Late Jurassic Morrison Formation Of Montana. SVP Conf. 2014 Abstract (2014).
Geist, V. Deer of the world: their evolution, behaviour, and ecology. (Stackpole books, 1998).
Scannella, J. B. & Horner, J. R. Torosaurus Marsh, 1891, is Triceratops Marsh, 1889 (Ceratopsidae: Chasmosaurinae): Synonymy through ontogeny. J. Vertebr. Paleontol. 30, 1157–1168 (2010).
Wilson, J. & Sereno, P. C. Early Evolution and Higher Level Phylogeny of Sauropod Dinosaurs. J. Vertebr. Paleontol. 18, 1–68 (1998).
McIntosh, J. S. In The Dinosauria (eds Weishampel, D. B., Dodson, P. & H., O.) 345–401 (University of California Press, 1990).
Acknowledgements
Thanks to the Cincinnati Museum Center Dinosaur Field School participants, the U.S. Bureau of Land Management (permit M 99231), and to S. Oser for initiating this research. K. Curry Rogers provided invaluable descriptive assistance. J. Horner and the Museum of the Rockies provided financial support associated with transportation and preparation. E.T. Lamm created the histologic thin sections of CMC VP14128. K. Scannella produced the cranial drawings of CMC VP14128 used in Figs 2, 3, 4, and SI Fig. 4. A. Atuchin produced the exquisite life restoration of CMC VP14128 in Fig. 5. Thanks to C. Ancell and B. Harmon for the preparation and casting. E. Tschopp and J. Whitlock provided invaluable reviews that greatly improved this work.
Author information
Authors and Affiliations
Contributions
G.W.S. collected the specimen, D.C.W. described the specimen, D.C.W., T.D.C., G.W.S., K.W., J.B.S., K.K.N. and J.P.W. wrote the manuscript, T.D.C. and K.K.N. created and analyzed the phylogenetic models, J.B.S. created the transformation grid, J.P.W. photographed the specimen and created the photogrammetric models.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Woodruff, D.C., Carr, T.D., Storrs, G.W. et al. The Smallest Diplodocid Skull Reveals Cranial Ontogeny and Growth-Related Dietary Changes in the Largest Dinosaurs. Sci Rep 8, 14341 (2018). https://doi.org/10.1038/s41598-018-32620-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-32620-x
Keywords
This article is cited by
-
Coupling length: a generalized gleno-acetabular distance measurement for interpreting the size and gait of quadrupedal trackmakers
Swiss Journal of Geosciences (2022)
-
Exceptionally simple, rapidly replaced teeth in sauropod dinosaurs demonstrate a novel evolutionary strategy for herbivory in Late Jurassic ecosystems
BMC Ecology and Evolution (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.