Abstract
Family history was one of the major risk factors for developing adverse health outcomes such as metabolic syndrome (MetS), type 2 diabetes mellitus (DM) and hypertension (HTN). Our aim was to examine the relationship between different family histories and cardiometabolic events, including DM, stroke, myocardial infarction (MI), and HTN. Participants who attended the health examinations at the Tri-Service General Hospital from 2010 to 2016 were enrolled in the study and were categorized into four groups by representing different family history. A multivariable logistic regression model was used for the associations between various family history with the cardiometabolic events. Subjects with family history of DM were divided into quartiles by percentage body fat (PBF) to be analyzed for these adverse outcomes. In the cross-sectional analysis, subjects with family history of DM had significant association with MetS (OR = 1.34 [95%CI: 1.17–1.54]) and DM (OR = 3.03 [95%CI: 2.44–3.76]), and those with family history of HTN were positively associated with HTN (OR = 1.60 [95%CI: 1.41–1.81]). Notably, those with family history of DM in higher PBF quartiles had substantially increased association of cardiometabolic events (MetS: OR = 15.20 [95%CI: 9.87–23.39]; DM: OR = 3.35 [95%CI: 1.91–5.90]; HTN: 2.81 [95%CI: 1.84–4.29]). Individuals with family history of DM were positively associated with MetS and DM, and those with family history of HTN was associated with HTN. Family history assessment was requested especially in obese population for screening adverse health outcomes.
Similar content being viewed by others
Introduction
The prevalence of chronic disorders such as metabolic syndrome (MetS), type 2 diabetes mellitus (DM), and hypertension (HTN) had increased in the past decades worldwide1,2. A high prevalence of obesity population was noted in Taiwan which was contributed to MetS and DM and became emerging economies and public health problems3,4. These cardiometabolic complications were multifactorial and a wide spectrum of different factors including environments, lifestyles, and genetic5,6,7.
Family history, was indisputably one of the major risk factors for chronic diseases like cancer, cardiovascular disease, DM, and was considered as an important genomic tool for preventive medicine and public health recently8,9. There were several advantages for family history assessment including inexpensive, greater acceptability, and a reflection of shared genetic and environmental risk factors10. Previous studies had reported the impact of family history on the risk of developing adverse health outcomes. In a Korean study, young adults with a family history of DM had an increased risk of DM and MetS11. The incidence of DM was increased among individuals with a family history of DM in Caucasians12.
Emerging studies reported that increased adiposity was associated with risks of cardiometabolic diseases13,14. High percentage body fat (PBF) was correlated with increased risk of DM even having a normal body mass index (BMI)15. In the present study, we hypothesized that participants who had family history with high PBF would have more closely association with cardiometabolic events than those with low PBF. Moreover, we examined the associations between combinations of different family histories and cardiometabolic risks. Our main goal was to ascertain the relevance and usefulness of PBF in the relationship between family histories with the cardiometabolic events.
Results
Demographic characteristics of the study population
There were 13561 participants with family history of DM, 3775 with family history of MI, 5460 with family history of stroke, and 18399 with family history of HTN. The mean age of each subgroup was 40.68 ± 13.58 (DM), 42.10 ± 13.92 (MI), 42.56 ± 14.18 (stroke), and 42.69 ± 14.37 (HTN). All characteristics of participants in different family history subgroups were shown in Table 1.
Association between various family history and cardiometabolic events
In Table 2, a multivariable logistic regression analysis was performed for the association between various family history and MetS, DM, and HTN. The odds ratios (ORs) for MetS in subjects with family history of DM were 1.31 (95%CI: 1.17–1.47), 1.35 (95%CI: 1.17–1.55) and 1.34 (95%CI: 1.17–1.54) in each adjusted model. However, other family history was not significantly associated with MetS. The association between DM and family history of DM were remained significant after adjustment for various covariables with ORs of 2.46 (95%CI: 2.02–3.01), 3.05 (95%CI: 2.46–3.78), and 3.03 (95%CI: 2.44–3.76) in each model. In the outcome of HTN, only those with family history of HTN had significant difference with ORs of 1.60 (95%CI: 1.42–1.79), 1.60 (95%CI: 1.42–1.81), and 1.60 (95%CI: 1.41–1.81) after multivariable adjustment.
Subjects with family history of DM in PBF quartiles and cardiometabolic events
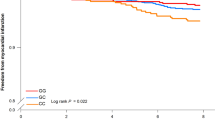
Participants with family history of DM were divided into quartiles by PBF. Associations between these people and cardiometabolic events were analyzed in Table 3. The ORs for MetS in subjects with different PBF quartiles after full adjustment were Q2: 2.82, Q3: 5.08 and Q4: 15.20 (95%CI: 2.03–3.91, 3.53–7.33, 9.87–23.39). Only subjects in the highest quartile of PBF were significantly associated with DM with ORs of 3.35 (95%CI: 1.91–5.90) after fully adjusting. The ORs for HTN in subjects with different PBF quartiles after fully adjusting were Q1: 1.50, Q2: 1.81, and Q3: 2.81 (95%CI: 1.08–2.09, 1.26–2.61, 1.84–4.29).
Association between various combinations of family history and DM
In Table 4, we categorized participants into various combinations of family history (DM, stroke, HTN, and MI). Associations between various combinations of family history and DM were analyzed by a univariate and a multivariate logistic regression model, respectively. Combinations which composed of family history of DM were significantly associated with DM in univariate model: C2 (DM): ORs = 3.461 (95%CI: 2.614–4.584), C6 (DM + MI): ORs = 2.636 (95%CI: 1.239–5.608), C10 (DM + HTN): ORs = 1.963 (95%CI: 1.419–2.717), and C14 (DM + HTN + MI): ORs = 2.944 (95%CI: 1.855–4.673). After fully adjusting for pertinent covariates, combinations of family history were associated with DM in C2 (DM): ORs = 4.366 (95%CI: 3.231–5.901), C6 (DM + MI): ORs = 2.782 (95%CI: 1.253–6.174), C10 (DM + HTN): ORs = 2.445 (95%CI: 1.735–3.445), C12 (DM + stroke + HTN): ORs = 2.836 (95%CI: 1.361–5.908), and C14 (DM + HTN + MI): ORs = 4.748 (95%CI: 2.910–7.745). However, subjects with family history combinations of (DM + stroke), (DM + stroke + MI), and (DM, stroke, HTN, MI) were not significantly associated with DM.
Association between different cut-off values of PBF and the presence of MetS, DM, and HTN in subjects with family history of DM
To predict the cardiometabolic risks in subjects with family history of DM, we assessed the cut-off values of PBF for the presence of MetS, DM, and HTN by using receiver operating characteristic (ROC) curve analysis. The cut-off values of PBF for MetS, DM, and HTN were 29.05, 32.65, and 29.75, respectively. After fully adjusting for covariates, family history of DM was significantly associated with MetS, DM, and HTN with ORs of 4.351 (95%CI: 3.290–5.753), 2.033 (95%CI: 1.355–3.048), and 1.763 (95%CI: 1.327–2.343), respectively (Table 5).
Discussion
In the current study, we observed that those with family history of DM had significant association with MetS and DM, and those with family history of HTN were positively associated with HTN. Subjects with family history of DM in higher PBF quartiles were associated with these cardiometabolic events. In addition, family history combinations which contained family history of DM had significant association with the presence of DM. Subjects who had family history of DM with different cut-off values of PBF could predict the risks of MetS, DM, and HTN. Our finding was the first to examine the association between various family history and cardiometabolic events in Taiwanese general population.
Previous researches had reported the important role of not only family history of HTN but also family history of DM in predicting the risks of different adverse health outcomes. In a long-term prospective study, both paternal and maternal hypertension were significantly associated with higher blood pressure and with the development of HTN over the adult life course16. Ranasinghe et al. had demonstrated that the prevalence of HTN was higher in individuals with family history of HTN17. The family history of DM was suggested as a useful tool to evaluate the risks of cardiometabolic disorders such as MetS, DM and cardiovascular diseases6,18,19. In a Korean study composed of a young adult population, those with family history of DM had increased risks of MetS and DM11. These results were consistent with our findings that the prevalence of MetS and DM was greater in subjects with family history of DM, and those with family history of HTN were correlated with the presence of HTN.
Several studies had addressed the synergistic effect of both positive family history of DM and obesity on the risk of developing DM. Hilding et al. demonstrated that exposure to both family history of DM and BMI showed the strongest effect on the risk of developing pre-diabetes and DM and conveyed a higher risk than either alone20. In an American study composed of a Indians population, family history of DM was correlated with higher incidence of DM with increased BMI than in those without family history of DM21. Obesity had been indicated to be associated with family history of DM and might be substantial part of the association between family history and the risk of DM22,23. In a Japanese cohort study, a family history of DM was associated with the incident risk of DM, and this association was independent of interactions with obesity and lifestyle factors24. Generalized adiposity reflected by BMI contributed to the association between family history of DM and the risk of developing DM25. A similar finding observed by Rice et al. represented that total body fat shared common familial determinants26. Consistent with our findings, combined family history of DM and higher PBF had increased risk for predicting MetS, DM and HTN than those in lower PBF quartiles. It was tempting to speculate that existing family history increased cardiometabolic risks and ensuing higher PBF could result in metabolic derangement and endothelial dysfunction that harbored a predisposing milieu for cardiometabolic diseases.
Several limitations were noted among the current study. First, a causal inference was not suitable in the study due to a cross-sectional design. A longitudinal analysis was necessary for further researches to examine the association between family history and the risks of adverse health outcomes. Second, the results might be influenced by recall bias because self-reported data was performed in the study to categorize family history which could result in some misclassification. Thus, we validated the family history collected by the questionnaire to eliminate inaccurate information. Third, the study sample was obtained from adult population in Taiwan. The limited ethnicity distribution of participants might not present the effect of family history on adverse health outcomes in terms of racial differences. Next, we only recruited study sample from a single hospital, which could limit generalization to the Taiwanese general population. Last, detail family histories in father and mother and familial risk classification were not recorded. It cannot be provided because our dataset included no information on detail family histories in first-degree relative or second degree relative. Familial risk classifications were not categorized in our analyzed models.
Conclusion
By assessing the Taiwanese adult population, the current study highlighted that family history of DM was significantly associated with cardiometabolic events. Distinctly, body fat accumulation obviously contributed to increased risks of MetS, DM and HTN, especially in offspring of patients with DM. Prevention strategies of cardiovascular diseases would benefit from giving more attention to lower body fat percentage in individuals with family history. The potential role of reducing PBF in the prevention of cardiometabolic diseases warranted more longitudinal surveys to explore the clinical applications.
Methods
Subjects Enrollment
Subjects enrolled in the study were derived from the health examinations at TSGH, from 2010 to 2016. Participants aged 20 years old and older attended comprehensive examinations including laboratory data, body composition and detailed self-reported questionnaires. All protocols in this retrospective study were approved by the Institutional Review Board (IRB) of TSGH. The IRB waived the need to obtain individual informed consent because the data were analyzed anonymously. All methods were performed in accordance with the relevant guidelines and regulations of TSGH.
Study Design
According to the flowchart shown in Fig. 1, participants who attended the health examinations at the TSGH from 2010 to 2016 and finished biochemical examination, body composition measurement, and questionnaire of family history were included (N = 27341). Eligible participants were divided into subgroups based on various family history including type 2 diabetes mellitus (DM) (N = 13561), stroke (N = 3775), myocardial infarction (MI) (N = 5460), and hypertension (HTN) (N = 18399). In the next step, a multivariable logistic regression model was performed for the association among different family histories and the presences of MetS, DM and HTN.
Family History Assessment
Family history was obtained from participants who attended the health examinations at TSGH by using a detailed self-reported questionnaire. A question “What family history do you have” was used for assessing various kinds of family history.
Definition of MetS
Several organizations had various criteria for diagnosing MetS. The World Health Organization (WHO) first reported its definition in 199827. The National Cholesterol Education Program (NCEP) Adult Treatment Panel III (ATP III) presented another definition in 2001 and updated in 200528. Subsequently, the International Diabetes Foundation (IDF) demonstrated an new criteria in 200529. In the current study, we adopted the nationwide standard published by the Taiwan Health Promotion Administration of the Ministry of Health and Welfare in 200730. A subject with at least 3 of the abnormal components was diagnosed of MetS: (1) blood pressure ≥130/85 mmHg; (2) waist circumference (WC) >90 cm for males and >80 cm for females; (3) fasting plasma glucose ≥100 mg/dL; (4) HDL-C<40 mg/dL for males and <50 mg/dL for females; and (5) triglyceride ≥150 mg/dL.
Definition of DM
Subjects with type 2 DM was diagnosed by the American Diabetes Association criteria: (1) fasting plasma glucose ≥126 mg/dL; (2) 2 hours plasma glucose ≥200 mg/dL during oral glucose tolerance test 75 g; (3) hemoglobin A1c tes t ≥ 6.5%; and (4) random plasma glucose ≥200 mg/dL31.
Definition of HTN
The definition of HTN in the current study was adopted by the guidelines which blood pressure ≥ 140/90 mmHg or subjects taking antihypertensive agents32.
Measurement of Body Composition
PBF was measured by bioelectrical impedance analysis (BIA) (InBody720, Biospace, Inc., Cerritos, CA, USA), an useful method for assessing body composition33. The procedure of BIA was simple and noninvasive, and the results were reproducible and rapidly obtained.
Assessments of Covariates
These pertinent characteristics included demographic factors (age, gender), biochemistry data (body mass index (BMI), proteinuria, serum total cholesterol, uric acid, creatinine, aspartate aminotransferase (AST), albumin, highly sensitive C-reactive protein (hsCRP)), and personal history (cigarette smoking, alcoholic consumption). A self-reported questionnaire was used to obtain age, gender and personal history. BMI was calculated by a formula that the weight divided by the square of the height (kg/m2) of a participant. Subjects were asked to fast at least 8 hours before health examinations for collecting blood samples. Biochemistry data was analyzed by different standard measurements. Total cholesterol was analyzed by an enzymatic colorimetric method (Roche Diagnostics, Indianapolis, IN, USA). The latex-enhanced nephelometry was used to detect hsCRP. Serum uric acid was measured by the Hitachi 737 automated multichannel chemistry analyzer (Boehringer Mannheim Diagnostics, Indianapolis, IN, USA).
Statistical Analysis
We classified subjects into different family history and compared the distribution of characteristics and covariates across subgroups by using ANOVA for continuous variables and the chi-squared test for categoric variables. Statistical significance was defined as a two-sided p-value of ≤0.05. Multivariable models were adjusted as follows: Model 1 included age, gender and BMI. Model 2 included Model 1 plus proteinuria, serum total cholesterol, uric acid, creatinine, AST, albumin and hsCRP. Model 3 included Model 2 plus history of cigarette smoking and alcoholic consumption. A logistic regression model was investigated for the association between family history and the risk of developing MetS, DM and HTN. Analyses in the current study were conducted by Statistical Package for the Social Sciences, version18.0 (SPSS Inc., Chicago, IL, USA) for Windows.
References
Ford, E. S., Giles, W. H. & Mokdad, A. H. Increasing Prevalence of the Metabolic Syndrome Among US Adults. Diabetes care 27, 2444–2449, https://doi.org/10.2337/diacare.27.10.2444 (2004).
Malik, V. S., Willett, W. C. & Hu, F. B. Global obesity: trends, risk factors and policy implications. Nature reviews. Endocrinology 9, 13–27, https://doi.org/10.1038/nrendo.2012.199 (2013).
Jiang, Y. D., Chang, C. H., Tai, T. Y., Chen, J. F. & Chuang, L. M. Incidence and prevalence rates of diabetes mellitus in Taiwan: analysis of the 2000–2009 Nationwide Health Insurance database. Journal of the Formosan Medical Association = Taiwan yi zhi 111, 599–604, https://doi.org/10.1016/j.jfma.2012.09.014 (2012).
Hwang, L. C., Bai, C. H. & Chen, C. J. Prevalence of obesity and metabolic syndrome in Taiwan. Journal of the Formosan Medical Association = Taiwan yi zhi 105, 626–635, https://doi.org/10.1016/s0929-6646(09)60161-3 (2006).
Han, J. C., Lawlor, D. A. & Kimm, S. Y. Childhood obesity. Lancet (London, England) 375, 1737–1748, https://doi.org/10.1016/s0140-6736(10)60171-7 (2010).
Valdez, R., Yoon, P. W., Liu, T. & Khoury, M. J. Family history and prevalence of diabetes in the U.S. population: the 6-year results from the National Health and Nutrition Examination Survey (1999–2004). Diabetes care 30, 2517–2522, https://doi.org/10.2337/dc07-0720 (2007).
Haskell, W. L. Cardiovascular disease prevention and lifestyle interventions: effectiveness and efficacy. J Cardiovasc Nurs 18, 245–255 (2003).
Yoon, P. W. et al. Can family history be used as a tool for public health and preventive medicine? Genetics in medicine: official journal of the American College of Medical Genetics 4, 304–310, doi: 10.109700125817-200207000-00009 (2002).
Valdez, R., Yoon, P. W., Qureshi, N., Green, R. F. & Khoury, M. J. Family history in public health practice: a genomic tool for disease prevention and health promotion. Annual review of public health 31, 69–87 61 p following 87, https://doi.org/10.1146/annurev.publhealth.012809.103621 (2010).
Burke, W. Taking family history seriously. Ann Intern Med 143, 388–389 (2005).
Moon, J. H. et al. Increased risk of metabolic disorders in healthy young adults with family history of diabetes: from the Korea National Health and Nutrition Survey. Diabetology & Metabolic Syndrome 9, 16, https://doi.org/10.1186/s13098-017-0210-8 (2017).
Harrison, T. A. et al. Family history of diabetes as a potential public health tool. Am J Prev Med 24, 152–159 (2003).
Shea, J. L., King, M. T. C., Yi, Y., Gulliver, W. & Sun, G. Body fat percentage is associated with cardiometabolic dysregulation in BMI-defined normal weight subjects. Nutrition, Metabolism and Cardiovascular Diseases 22, 741–747, https://doi.org/10.1016/j.numecd.2010.11.009 (2012).
Zhu, S., Wang, Z., Shen, W., Heymsfield, S. B. & Heshka, S. Percentage body fat ranges associated with metabolic syndrome risk: results based on the third National Health and Nutrition Examination Survey (1988–1994). The American Journal of Clinical Nutrition 78, 228–235, https://doi.org/10.1093/ajcn/78.2.228 (2003).
Gomez-Ambrosi, J. et al. Body adiposity and type 2 diabetes: increased risk with a high body fat percentage even having a normal BMI. Obesity (Silver Spring, Md.) 19, 1439–1444, https://doi.org/10.1038/oby.2011.36 (2011).
Wang, N. Y. et al. Blood pressure change and risk of hypertension associated with parental hypertension: the Johns Hopkins Precursors Study. Archives of internal medicine 168, 643–648, https://doi.org/10.1001/archinte.168.6.643 (2008).
Ranasinghe, P., Cooray, D. N., Jayawardena, R. & Katulanda, P. The influence of family history of Hypertension on disease prevalence and associated metabolic risk factors among Sri Lankan adults. BMC Public Health 15, 576, https://doi.org/10.1186/s12889-015-1927-7 (2015).
Das, M., Pal, S. & Ghosh, A. Family history of type 2 diabetes and prevalence of metabolic syndrome in adult Asian Indians. Journal of Cardiovascular Disease Research 3, 104–108, https://doi.org/10.4103/0975-3583.95362 (2012).
Park, J. W. et al. Family history of diabetes and risk of atherosclerotic cardiovascular disease in Korean men and women. Atherosclerosis 197, 224–231, https://doi.org/10.1016/j.atherosclerosis.2007.03.037 (2008).
Hilding, A. et al. The impact of family history of diabetes and lifestyle factors on abnormal glucose regulation in middle-aged Swedish men and women. Diabetologia 49, 2589–2598, https://doi.org/10.1007/s00125-006-0402-5 (2006).
Knowler, W. C., Pettitt, D. J., Savage, P. J. & Bennett, P. H. Diabetes incidence in pima indians: contributions of obesity and parental diabetes1. American Journal of Epidemiology 113, 144–156, https://doi.org/10.1093/oxfordjournals.aje.a113079 (1981).
Lapidus, L., Bengtsson, C., Lissner, L. & Smith, U. Family history of diabetes in relation to different types of obesity and change of obesity during 12-yr period. Results from prospective population study of women in Goteborg, Sweden. Diabetes care 15, 1455–1458 (1992).
van Dam, R. M., Boer, J. M., Feskens, E. J. & Seidell, J. C. Parental history of diabetes modifies the association between abdominal adiposity and hyperglycemia. Diabetes care 24, 1454–1459 (2001).
Sakurai, M. et al. Family history of diabetes, lifestyle factors, and the 7‐year incident risk of type 2 diabetes mellitus in middle‐aged Japanese men and women. Journal of Diabetes Investigation 4, 261–268, https://doi.org/10.1111/jdi.12033 (2013).
van ‘t Riet, E. et al. Role of Adiposity and Lifestyle in the Relationship Between Family History of Diabetes and 20-Year Incidence of Type 2 Diabetes in U.S. Women. Diabetes care 33, 763–767, https://doi.org/10.2337/dc09-1586 (2010).
Rice, T., Bouchard, C., Perusse, L. & Rao, D. C. Familial clustering of multiple measures of adiposity and fat distribution in the Quebec Family Study: a trivariate analysis of percent body fat, body mass index, and trunk-to-extremity skinfold ratio. International journal of obesity and related metabolic disorders: journal of the International Association for the Study of Obesity 19, 902–908 (1995).
Alberti, K. G. & Zimmet, P. Z. Definition, diagnosis and classification of diabetes mellitus and its complications. Part 1: diagnosis and classification of diabetes mellitus provisional report of a WHO consultation. Diabetic medicine: a journal of the British Diabetic Association 15, 539–553, https://doi.org/10.1002/(sici)1096-9136(199807)15:7<539::aid-dia668>3.0.co;2-s (1998).
Third Report of the National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III) final report. Circulation 106 3143-3421 (2002).
Zimmet, P., Magliano, D., Matsuzawa, Y., Alberti, G. & Shaw, J. The metabolic syndrome: a global public health problem and a new definition. Journal of atherosclerosis and thrombosis 12, 295–300 (2005).
Health Promotion Administration, Ministry of Health and Welfare (2015).
Standards of Medical Care in Diabetes-2016: Summary of Revisions. Diabetes care 39 Suppl 1, S4–5, https://doi.org/10.2337/dc16-S003 (2016).
Chiang, C. E. et al. 2015 guidelines of the Taiwan Society of Cardiology and the Taiwan Hypertension Society for the management of hypertension. Journal of the Chinese Medical Association: JCMA 78, 1–47, https://doi.org/10.1016/j.jcma.2014.11.005 (2015).
Sergi, G., De Rui, M., Stubbs, B., Veronese, N. & Manzato, E. Measurement of lean body mass using bioelectrical impedance analysis: a consideration of the pros and cons. Aging clinical and experimental research 29, 591–597, https://doi.org/10.1007/s40520-016-0622-6 (2017).
Acknowledgements
This research did not receive any specific grant from any funding agency in the public, commercial or not-for-profit sector.
Author information
Authors and Affiliations
Contributions
Yuan-Yuei Chen contributed to the design of the study, was responsible for the management and retrieval of data, contributed to initial data analysis and interpretation, drafted the initial manuscript. Yuan-Yuei Chen, Wen-Hui Fang, Chung-Ching Wang, Tung-Wei Kao, Yaw-Wen Chang, Hui-Fang Yang, Chen-Jung Wu, Yu-Shan Sun, Wei-Liang Chen decided upon the data collection methods. Yuan-Yuei Chen and Wei-Liang Chen were also responsible for the data analysis decisions. Wei-Liang Chen conceptualized and designed the study, supervised all aspects of the study, critically reviewed and revised the manuscript, and approved the final manuscript as submitted. All authors meet the ICMJE criteria for authorship.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Chen, YY., Fang, WH., Wang, CC. et al. Association of Percentage Body Fat and Metabolic Health in Offspring of Patients with Cardiovascular Diseases. Sci Rep 8, 13831 (2018). https://doi.org/10.1038/s41598-018-32230-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-32230-7
Keywords
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.