Abstract
This study aims to explore the feeding ecology of two terrestrial papionins, Papio and Theropithecus from the Shungura Formation in Ethiopia, the most complete stratigraphic and paleontological record of the African Plio-Pleistocene. Two aspects were evaluated using Dental Microwear Texture Analysis: differences in diet between the extinct genera and their extant relatives, and any potential dietary fluctuations over time. Amongst more than 2,500 cercopithecid dental remains, 154 Theropithecus molars and 60 Papio molars were considered. Thirty-nine extant wild baboons and 20 wild geladas were also considered. The results show that diets of extinct monkeys from Member G already differed between genera as it is the case for their extant representatives. The shearing facets on the Theropithecus molars display significant variations in microwear textures, suggesting several dietary shifts over time. Two events point to higher intakes of herbaceous monocots (tougher than dicots foliages), at about 2.91 Ma (between members B and C) and at 2.32 Ma (between members E and F). These two events are separated by an inverse trend at about 2.53 Ma (between members C and D). Some of these variations, such as between members E and F are supported by the enamel carbon isotopic composition of herbivorous mammals and with paleovegetation evidence.
Similar content being viewed by others
Introduction
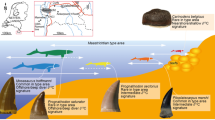
Papio and Theropithecus are terrestrial cercopithecids whose extinct relatives were more diverse and widespread. For example, the modern gelada (Theropithecus gelada) is a refugee species in the Ethiopian High Plateau, with a restricted geographical range and likely a reduced set of habitats compared to its extinct relatives (Fig. 1). The present work aims to investigate the ecological segregation between these two primate genera found in eastern African Plio-Pleistocene sites. The second goal is to track changes in the feeding habits of these cercopithecids that reflect variations in food resources over time, in the context of environmental changes during the Plio-Pleistocene at a regional scale. To do so, dental microwear textures of specimens of Papio and Theropithecus from the Shungura Formation were analyzed. Shungura is a geological formation situated within the Lower Omo Valley, on the northern part of the Turkana Basin in southwestern Ethiopia (Fig. 1). Its sedimentary deposits have yielded major Plio-Pleistocene paleontological and archeological records, including numerous hominin and lithic remains. It is subdivided into 12 geological members, from the lowermost to the uppermost: Basal, A to H, and J to L. The main volcanic tuffs of the formation, forming the base of each member, allow for long distance stratigraphic correlations. These and other ash layers have been dated by radiochronological methods, including 40K/40Ar and 40Ar/39Ar, and by magnetostratigraphy. These methods and extrapolations from their results are indicative of a time interval of 3.6 Ma to 1.05 Ma for the deposition of the formation. This in turn led to a particularly accurate chronostratigraphic framework for testing evolutionary hypotheses. Unlike all other African Plio-Pleistocene sites, this formation displays a good chronological continuity of its sedimentary deposits, notably between 3 Ma and 2 Ma, documenting a critical period in hominin evolution marked by the transition between Australopithecus and Homo and by the emergence of Paranthropus.
Geographical context of the study. Localization of the Plio-Pleistocene deposits of the Shungura Formation; geographical extent of the subsistence area of the extant gelada (in striped orange; IUCN 2017); and localization of gelada populations from Guassa and Indetu, respectively studied by Fashing et al.35 and Abu et al.77. See also Welch et al.78 for an updated and more detailed geographical distribution of the gelada.
The inter-specific and temporal variations in the dental microwear textures of these terrestrial monkeys were compared with enamel stable carbon isotopic data and paleovegetation evidence. This provides insights into the available vegetation in the cercopithecid habitats along the ancestral Omo River and therefore allows for identifying environmental trends.
Most primate remains found in the Shungura Formation belong to Papio and Theropithecus. The latter taxon has a unique dietary specialization among extant primates: it forages predominantly on the herbaceous layer and mostly on monocotyledons. Today, Papio is represented by several species throughout Africa. Up to six species have been recognized by now, but the phylogeny and systematics have been debated for decades1,2,3,4. These extant monkeys are terrestrial and have a wide spectrum of feeding habits5,6,7,8,9,10,11, which include the aerial and subterranean parts of herbaceous vegetation, and also flowers, leaves, fruits and seeds from the shrub to the arboreal layers, as well as animal matter (see references and Fig. 2, p. 556 in Scott et al.12).
From the fossil remain to the dental microwear textures. (a) Fossil mandible (L 199-5) attributed to Theropithecus brumpti and recovered in Member C of the Shungura Formation. (b) Right lower second molar from the mandible in occlusal view with the localization of the two scanned facets. (c) Photosimulations of the two types of facets on the lower third molar (respectively facet 8 and facet 12) of a dubious specimen, OMO 18-1968-2238, that were not included in the study due to potential postmortem alteration. Pairs of photosimulations showing microwear textures differing between the two types of facets and that were included in the analyses; (d) OMO 28-1968-1273, lower third molar with facets 5 and 11; (e) OMO 18-1969-510, upper second molar with facets 3 and 9. Scale bars represent 50 µm.
As fossil Papio and Theropithecus present the same different dental morphologies met between their extant relatives, evidence that diets differed between extinct taxa was expected. Based on enamel stable carbon isotopic composition, Lee-Thorp et al.13 discovered that the Swartkrans baboons fed on C3 vegetation, whereas the sympatric species of Theropithecus mostly consumed C4 vegetation. Taking into account the differences in dental morphology, this result was to be anticipated. Other studies have also examined stable carbon isotopes in the enamel of fossil Theropithecus14,15,16,17,18,19. Although this dietary proxy provides information on the proportions of plants using C3 and C4 photosynthetic pathways in the diet, it cannot distinguish the type of organs consumed. Dental Microwear Texture Analysis (DMTA) offers a complementary source of data that gives information on the mechanical properties of the eaten organs for more complete inferences of the dietary habits of these fossil representatives. Indeed, each organ has its own mechanical properties and inner composition20,21,22. We expect mature foliages of silica-bearing herbaceous monocots to generate less complex and more anisotropic microwear textures on primate teeth than the consumption of rhizomes, bulbs or small seeds from ear12. There is no doubt that underground storage organs are ingested with grit that contribute to tooth wear. We also expect primates foraging on a wide spectrum of food types (with different mechanical properties) to have more variable dental microwear textures than a monkey focusing for instance only on foliages. Such variations has been found on different modern ungulates with different feedings habits but also on captive animals fed with different fodders23,24,25,26,27.
Materials and Methods
Materials
All the fossil specimens considered in this study were collected by the International Omo Research Expedition (IORE, 1967–1976) and the Omo Group Research Expedition (OGRE, since 2006) in the Shungura Formation, Lower Omo Valley, Ethiopia. They were all gathered between Member B and the lower part of Member G (stratigraphic units 1–13), covering a time span of 3.44 Ma to 2.05 Ma (see Appendices 1 and 2).
The sediments constituting these stratigraphic entities were mostly deposited in a fluviatile context, with the recurrence of a sequence of sands and sandstones at the bottom, silts, and clays/clayey silts at the top. These were respectively interpreted as channel deposits, levee deposits, and flood plain deposits28. These sediments include volcanic tuff deposits, for which the sedimentary facies strongly suggest underwater deposition in a fluviatile context.
The specimens studied here were essentially found in the sandy horizons, with the major exception of the specimens from OMO 33 that came from a sandy-tuffitic deposit, again in a fluviatile context. The Shungura Formation also includes a large number of archeological occurrences in Member F and the lower part of Member G29,30, almost exclusively containing lithics made from quartz31. However, most of these elements occur in silts or clays. They do not co-occur with fossil concentrations, and are not associated with significant faunal assemblages. The material studied here was not discovered in such archeological contexts, but in natural assemblages. The radiometric 40Ar/39Ar dates from McDougall and Brown32 and McDougall et al.33 are used here for dating the boundaries between geological members.
More than 2,500 dental remains of Shungura cercopithecids attributed with certainty to the genus Theropithecus and 173 attributed with certainty to the genus Papio are stored at the Authority for Research and Conservation of Cultural Heritage in Addis Ababa. Among all of these specimens, 297 dental remains of Theropithecus and 110 of Papio were selected for analysis because their molars display apparent functional dental facets without post-mortem damage. Initially, observations were made with the naked eye or with a 10 × handheld lens. When non-occlusal and occlusal surfaces showed similar alterations, this was a hint of post mortem damage that allowed the exclusion of the specimens before molding. The next step happened after molding and scanning (Fig. 2). Dental microwear textures between shearing and crushing facets are expected to differ from each other, as the two facets contribute to different phases during mastication. So contrasted dental microwear textures were indeed a clear hint that the dietary signal was preserved. When the two facets showed identical pitted enamel surfaces without any scratches, the specimen was rejected from the analysis.
Then, among those, DMTA has been performed on 154 Theropithecus individuals and 60 Papio individuals (see Appendices 1 and 2). Post-mortem damaged molars are identifiable using the following characteristics: homogeneous rough textures with similar small pits and an absence of apparent scratches on both Phase I and Phase II dental facets. Furthermore, scratches and pits of different size were preserved on some specimens but the flanks of these scratches did show erosion.
Among the papionins recovered in the Shungura Formation, the species Papio (Dinopithecus) quadratirostris has been identified from Member D to the lower part of Member G. However, Frost34 suggested that this species, or at least the genus Papio, is present from Member A to Member L. Here, only the specimens attributed to the genus Papio were considered, meaning that no other papionin material was included.
Two species of Theropithecus are represented (Appendix 1): T. brumpti (n = 42) and T. oswaldi (n = 6). It is worth mentioning that the specific attribution of many specimens of Theropithecus that we considered is uncertain (n = 106; see Appendix 1). Theropithecus brumpti has been recovered in members A to D and, starting from Member E, it is associated with T. oswaldi. Thus, specimens from the lower members can most probably be attributed to T. brumpti, but specimens from Member E to the lower part of Member G can belong to either species. It was decided to work at the generic level as a specific level attribution for these fossil taxa cannot be made with certainty on isolated teeth. However, specific differences between T. oswaldi and T. brumpti from the Shungura Formation were explored. Due to the low sample size when considering only specimens co-occurring in the same member, it was decided to compare the two species of Theropithecus by including all specimens assigned to the specific level, regardless of the member in which they were found (Appendix 4).
Extant and adult wild-shot specimens of T. gelada (n = 22), P. hamadryas anubis (n = 15), P. hamadryas cynocephalus (n = 18), and P. hamadryas hamadryas (n = 6) were also sampled (Appendix 1). Among the gelada samples, nine specimens come from Guassa (housed at the Authority for Research and Conservation of Cultural Heritage in Addis Ababa), an ecosystem believed to be almost intact where the behavioral and feeding ecologies of geladas, as well as the structure and composition of the vegetation have been documented35,36,37. The other specimens of geladas come from the surroundings of Debark, to the west of the Simien Mountain National Park, and from different areas not located in Ethiopia (housed in the MNHN collections in Paris, France and the Naturmuseum Senckenberg, Frankfort, Germany).
Dental Microwear Texture Analysis
DMTA has been discussed in detail by Scott et al.38,39; see also Calandra and Merceron40. The molars were cleaned with cotton swabs soaked in alcohol in order to remove grit, dust, and glue residues remaining on their occlusal facets. After cleaning, dental impressions were made with a silicone material (polyvinyl siloxane ISO 4823, President Regular Body, Coltène-Whaledent Corp.). Scans were made directly from the silicon molds with a Leica DCM8 white-light scanning confocal microscope (Leica Microsystems; Fig. 2) with a ×100 long-distance lens (Numerical Aperture = 0.90; working distance = 0.9 mm), housed at the PALEVOPRIM laboratory. For each specimen, one Phase I occlusal facet (facets 1–8)41,42 and one Phase II facet (Facets 9 to 13)41,42 were scanned on either the upper or lower molars (M1, M2, M3). The choice of the molar is made based on the stage of wear (stage 3–4 sensu Meikle43, or stage 2–4 sensu Venkataraman et al.36). The scanning process generates 350 × 264 μm point clouds with a vertical sampling lower than 0.002 μm and a lateral sampling (x, y) of 0.129 μm. These surfaces have been saved as plu files by the LeicaScan software (Leica Microsystems). They are then opened with LeicaMap software (Mountain technology, Leica Microsystems) to remove aberrant peaks with automatic operators including a morphological filter23. These surfaces were leveled and a 200 × 200 μm area was selected and saved as a Digital Elevation Model (.sur) to be used for DMTA.
The four most significant dental microwear texture variables were considered: Asfc, HAsfc, epLsar and Tfv, which have been described in detail by Ungar et al.44 and Scott et al.38,39. Complexity (area-scale fractal complexity, Asfc) is a measure of the roughness at a given scale. Heterogeneity of complexity (heterogeneity of area-scale fractal complexity, HAsfc) quantifies the variation of complexity observed within a scan. HAsfc is obtained by subdividing the surface to a 9 × 9 grid, hence 81 cells, in which complexity is calculated. Anisotropy (epLsar or exact proportion of length-scale anisotropy of relief) measures the orientation concentration of surface roughness. Complexity (Asfc), heterogeneity of complexity (HAsfc) and anisotropy (epLsar) were calculated with Toothfrax software (www.surfract.com). Textural fill volume (Tfv) is the result of an algorithm that fills a surface with square cuboids of different volumes (10 and 2 µm-side cuboids). Tfv does not depend on the surface shape but on its finer texture. It was calculated with Sfrax software (www.surfract.com).
Among Primates, the complexity of dental microwear textures is higher for taxa consuming hard food items, such as fruits with hard exocarp or hard seeds12,38,39. This relationship has been shown experimentally on captive primates45,46 but also on captive ewes24. High complexity can be correlated with the consumption of possibly mechanically challenging Underground Storage Organs (USOs) that may also carry grit and dust on their surface. Although quartz grains present on the surface of some food items do scratch the enamel and contribute to tooth wear47,48, a recent controlled food test concluded that dust load on foods (simulating natural Harmattan wind-blown dust loads in present day western Africa) does not overwhelm the dietary signal23. Anisotropy (epLsar) is higher for species consuming blades of herbaceous monocots or mature leaves from woody dicots12,38,39. Similarly, based on a controlled food test on ewes, Merceron et al.23 showed that mature grasses generate significantly higher anisotropy on teeth than is generated by soft herbaceous dicots. High HAsfc values are linked with high variability in both size and nature of wear-causing particles, indicating a broader diet in terms of fracture properties12,39. Ramdarshan et al.24 showed that a mixture of foliage and seeds produces higher heterogeneity of complexity than is produced by a dietary bolus composed exclusively of herbaceous dicots.
Statistical analyses
Intergeneric variations
To investigate potential differences in terms of diets between extinct Papio and Theropithecus and their extant relatives, consideration was given only to geological members for which there were at least seven specimens per genus as well as the two extant samples of Papio and Theropithecus. These geological members with substantial specimen numbers are E, F, and G (Table 1). The four dental microwear texture variables were considered (Asfc, HAsfc, epLsar, and Tfv) but only on Phase II molar facets, as these facets have been considered more significant for discriminating primates with contrasting diets49.
Prior to the analysis, variables were rank-transformed to avoid normality assumption violations in parametric tests50,51. One-way analyses of variance (ANOVAs) were performed for each parameter (Table 2). Finally, for each significant parameter, pairwise comparisons were performed using the combination of Tukey’s HSD (Honest Significant Difference) and the less conservative Fisher’s LSD (Least Significant Difference; Table 3; Appendix 3) tests.
Chronological variations
For each of the two genera (Theropithecus and Papio) and for each of the two sets of dental facets (Phase I and Phase II facets), four Principal Component Analyses (PCAs) were generated, followed by a set of ANOVAs to identify significant variations between stratigraphic samples (Appendices 6 and 7). Only the PCA on Phase I molar facets of Theropithecus shows significant variations along the first and the third Principal Components (PC). As for generic variations, pairwise comparisons on ranked PC1 and PC3 coordinates were performed with the tandem HSD-LSD (Appendices 8 and 9). As the coordinates along the most informative component, PC1, of the PCA conducted on Phase I facets of Theropithecus display significant variations across geological time, this linear combination of the four dental microwear texture parameters (Fig. 3) will be referred to as the Wear Textural Index (WTI).
Temporal paleodietary fluctuations across the geological members of the Shungura Formation. Symbols on the curves represent means and error bars represent standard errors of means. Asterisks pinpoint significant differences on PC1 coordinates for the Phase I (Wear Textural Index) of Theropithecus. A single asterisk means a moderate difference (only LSD is significant) and two asterisks mean a substantial difference (both LSD and HSD are significant).
Results
Differences between modern and fossil Papio and Theropithecus
The two-way nested ANOVAs (using genus and sample as factors) ran on ranked data detect variations in all surface parameters, between genera in complexity Asfc, heterogeneity of complexity HAsfc and textural fill volume Tfv, and between samples for heterogeneity of complexity HAsfc and anisotropy epLsar (Table 2). The samples in question were of extant species of Theropithecus and Papio along with extinct papionins grouped by genus and stratigraphic provenance.
When considering whole genera, Papio differ from Theropithecus in having significantly higher Asfc and HAsfc, and lower Tfv, but there is no perceptible difference for epLsar (Tables 1 and 2). It should be noted that this result combines both extant and extinct representatives of the two genera.
When considering differences between samples, Theropithecus gelada exhibit a lower HAsfc compared to extant Papio species. Extinct Theropithecus from members E and F differ from their extant relatives in having a moderately lower epLsar, whereas Theropithecus from G do not differ from T. gelada based on this variable (Table 3). Extinct Papio from members E and F differ from the three extant species in having a moderately lower HAsfc. Papio from Member G differ from Papio hamadryas hamadryas in having a lower HAsfc and from P. hamadryas anubis in having lower HAsfc and epLsar, but they do not differ from P. hamadryas cynocephalus (Table 3). When considering extinct papionins, Papio from members E, F, and G do not differ from Theropithecus from members E and F. Papio from members E and G show a moderately higher HAsfc than Theropithecus from Member G whereas Papio from Member F do not differ from Theropithecus from G (Table 3).
Chronological variations
Before drawing any interpretations on the temporal dietary fluctuations of Shungura Theropithecus, it was important to check whether the two species identified in the formation (i.e. T. brumpti and T. oswaldi) had different dietary preferences that could affect the signal, as T. oswaldi was not recovered from the lowest geological members (B to D). Hence, two sets of ANOVAs were performed on each texture parameter (complexity Asfc, heterogeneity of complexity HAsfc, anisotropy epLsar, and textural fill volume Tfv) for the two types of facets (Phase I and Phase II). None of the ANOVAs resulted in a significant difference between the two species when considering all attributed specimens whatever the member in which they were found (Appendix 4).
ANOVAs performed on coordinates obtained from the four PCAs revealed that only PC1 and PC3 coordinates from the analysis on Phase I facets of Theropithecus show significant differences between geological members (Appendices 6 and 7). PC coordinates obtained from both Phase I and Phase II Papio molar facets show no variations over time. The same is true for Phase II Theropithecus facets that also display no variations along the stratigraphic sequence (Appendices 6 and 7). This last result might seem unexpected because both Phase I and Phase II molar facets are involved successively during the mastication of the dietary bolus, but they do not show the same trends. However, as Phases I and II molar facets involve different kinds of masticatory movements41,42, they may carry different dietary signals49.
PC1 for Phase I Theropithecus facets accounts for 33.2% of the total variance and is mainly explained by the complexity Asfc (45.5% of variance), followed by the heterogeneity of complexity HAsfc (37.2%), the anisotropy epLsar (17.2%) and the textural fill volume Tfv (0.1%). Asfc, HAsfc, and Tfv are positively correlated with PC1, whereas epLsar is negatively correlated with PC1 (Appendix 6). Pairwise comparisons show fluctuations of PC1 coordinates between successive geological members with one major difference between members C and D (highlighted by significant HSD, see Appendix 8). Specimens from Member D exhibit higher values than specimens from Member C. Two minor differences are also shown between B and C and between E and F, for which only LSD is significant. Specimens from Member C show lower PC1 values than specimens from Member B. Specimens from Member F display lower PC1 values than Member E specimens (Fig. 3). As Tfv represents a negligible amount of the variance on PC1, this principal component is mainly explained by the complexity Asfc and heterogeneity of complexity HAsfc, which are positively correlated to the axis, and by anisotropy epLsar, which is negatively correlated to PC1 (Appendix 6). Therefore, the previously described differences can be interpreted as a slight decrease in both Asfc and HAsfc along with a slight increase in epLsar from Member B to Member C, followed by a substantial increase of Asfc and HAsfc with a marked decrease in epLsar from Member C to Member D, and then another slight decrease in both complexity Asfc and heterogeneity of complexity HAsfc and an increase in anisotropy epLsar from Member E to Member F (Fig. 3).
PC3 for Phase I facets of Theropithecus accounts for 19.7% of the total variance and is mostly explained by heterogeneity of complexity HAsfc (56.6% of variance), followed by complexity Asfc (21.5%), textural fill volume Tfv (11.6%) and anisotropy epLsar (10.3%). Complexity Asfc and textural fill volume Tfv are positively correlated with PC3, whereas heterogeneity of complexity HAsfc and anisotropy epLsar are negatively correlated with PC3 (Appendix 6). Pairwise comparisons highlight variations of PC3 coordinates between consecutive geological members with two minor differences (shown by significant LSD, see Appendices 8 and 9) between members D and E and then between the following Member F and the lower part of Member G. Specimens from Member E exhibit a lower mean PC3 value than Member D specimens. Theropithecus recovered from the lower part of Member G display a higher mean PC3 value than specimens found in Member F. PC3 is negatively explained by heterogeneity of complexity HAsfc and anisotropy epLsar, and positively explained by complexity Asfc and textural fill volume Tfv. Hence, the previously mentioned variations can be seen as a slight increase in mean heterogeneity of complexity HAsfc and anisotropy epLsar along with a slight decrease in complexity Asfc and textural fill volume Tfv from Member D to Member E, followed by a slight decrease in means of heterogeneity of complexity HAsfc and anisotropy epLsar with a slight increase of means of complexity Asfc and textural fill volume Tfv from Member F to the lower part of Member G.
Discussion
Diet of extant geladas and implications for the interpretation of paleodietary proxies applied to extinct Theropithecus
During the wet season, the gelada diet was first thought to be almost exclusively composed of herbaceous monocots, mainly grass blades, representing more than 90% of their diet. During the dry season, they were shown to heavily rely on Underground Storage Organs (USOs), which represent more than 50% of their diet during the dry months52.
But most of these short-term studies had focused on populations living in disturbed areas, subject to high livestock grazing pressure, resulting in a dusty habitat dominated by short grasses35. Two long-term studies recently came out: one in an almost intact tall-grass afroalpine ecosystem at Guassa Plateau (Fig. 1)35, and the other in a disturbed area, Indetu (Fig. 1)53. The latter study showed that leaves of herbaceous monocotyledons represent between 38.1% in September to 71.4% of the diet in November, or 51.7% yearly. This is supplemented by USOs that compose up to 46.6% of the diet in February, or 34.8% yearly. The study has also reported that leaves of herbaceous dicots (forbs) compose up to 18.4% of the diet in April53, but only 7.1% of the yearly diet overall. Conversely, the study done at Guassa has shown a higher contribution of herbaceous dicots (forbs), representing 37.8% of the annual diet and an intake of herbaceous monocotyledons accounting for 56.8% of their yearly diet35. They also observed high seasonal dietary variations with herbaceous monocotyledon blades and inflorescences being heavily consumed during the wet season, whereas the intake of underground organs is higher for the dry season. Even during the rainiest month, herbaceous monocotyledons do not represent more than 75% of the diet, and underground organs do not account for more than 28% of the diet during the driest month35. As forbs represent from 20% to 60% of the gelada’s yearly diet, and because edible forbs account for only 7.4% of the ground cover at Guassa, the authors considered forbs as a preferred food source35. Conversely, Underground Storage Organs, the consumption of which is negatively correlated with rainfall, are considered as fallback foods (FBFs)35,36,54,55,56,57. Because extant Theropithecus show broader seasonal variations in their diet than previously thought, special attention should be paid to seasonal consumption of mechanically challenging FBFs, notably due to the potential rapid overprinting of wear marks on dental facets45,46,58. USOs consumed by Guassa geladas as fallback foods during the dry season may not be particularly hard but they are covered in grit36. However, geladas have been observed to clean USOs with their hand before ingestion. Also, the tunic of corms/tubers is spat out, probably to avoid grit and limit tooth wear35. The observations from Fashing et al.35 on intact ecosystems compared to the studies on disturbed areas52,53 raise one important potential bias of uniformitarian approaches19. As the distribution area of extant geladas overlaps anthropic habitats shaped by millennia of tree felling, pastoralism and agriculture, its dietary habits should not be directly transposed onto its extinct relatives spread across Africa during the Plio-Pleistocene.
The specific monocot-dominated diet of geladas was also proposed for its extinct relatives, T. brumpti and T. oswaldi, based on 2D dental microwear studies and on stable carbon isotope studies13,14,15,16,17,18,19,37,59,60. Among these, Cerling et al.16 examined stable carbon isotopes analyses on the enamel of T. brumpti and T. oswaldi (n = 41) from the Turkana Basin and from Olorgesaillie in southern Kenya. T. brumpti specimens exhibit a mean δ13C value indicating a diet comprising between 55% and 75% of C4 plants, whereas T. oswaldi specimens show a diet even more focused on C4 plants, representing between 70% and 90% of its consumption. The present study did not detect any significant difference between the two species (Appendix 5). DMTA might reach its limits to discriminate two grass-eating monkeys, notably taking into account the low sample size regarding T. oswaldi. Alternatively, it can be hypothesized that the two species of Theropithecus shared food resources with similar mechanical properties between 2.5 and 2.0 Ma, an interval of time not explored by Cerling et al.16. It is also worth pointing out here that many herbaceous monocots are not C4 but C3 plants. As recalled by Souron51, the C3 component in mammal paleodiets is frequently assumed to come from trees, bushes or shrubs, and C4 from herbaceous monocots. However, growing evidence indicates that, during the Plio-Pleistocene, C3 herbaceous monocots such as grasses, sedges and rushes were more abundant in the tropical lowlands of Africa than they are today61,62,63,64. Therefore, the C3 component of their diet can instead come from the herbaceous layer, with either herbaceous monocots or dicots (forbs), rather than from woody dicots. Of note is the fact that, although herbaceous monocots in modern African tropical lowland ecosystems are mainly C4, this is not the case in the Highlands inhabited by extant geladas, where C3 monocotyledons represent the dominant part of the herbaceous layer35,65,66,67. Although several sets of enamel stable carbon isotopes analyses on extinct specimens of Theropithecus point to a mixed C3/C418 to dominant C4 diet16, it is surprising that there is hitherto only one stable carbon isotope data published on modern gelada, which is below −10‰68. There is no doubt that supplementary analysis would confirm the very low proportion of C4 because the Highlands where the extant geladas live are too high in altitude to favor C4 grasses at the expense of C3 ones.
DMTA on extant primates12,69,70 and notably geladas are scarce12,37. Scott et al.12 showed that modern geladas have low complexity and high anisotropy, suggesting the consumption of tough plants, presumably herbaceous monocots. Later, Shapiro et al.37 presented slightly different textures. The extant geladas they analyzed show lower values in anisotropy compared to the specimens sampled by Scott et al.12. This fits with the lower amount of herbaceous monocots consumed by the Guassa population studied by Shapiro et al.37. The sample of extant geladas used here also comes partly from Guassa and as such the values of anisotropy and complexity that were obtained are similar to the values found by Shapiro et al.37. This supports the conclusions of Fashing et al.35 that this species has a broader diet including fewer leaves of herbaceous monocots than previously thought.
It is not unexpected to find such values of anisotropy in the extinct specimens of Theropithecus from the Shungura Formation. Moreover, it is worth mentioning that the extant sample of geladas does not differ from the samples of modern baboons in anisotropy (epLsar) but rather in complexity (Asfc) and in heterogeneity of complexity (HAsfc; Tables 1 and 3).
Dietary differences between terrestrial papionins from the Shungura Formation
The only enamel stable carbon isotope analysis exploring niche partitioning among terrestrial papionins was conducted by Lee-Thorp et al.13 on T. darti and P. robinsoni from Swartkrans in South Africa. The authors detected that the former species included high proportions of C4 vegetation in its diet, presumably herbaceous monocots, whereas the latter included only C3 vegetation. The present study also explores differences between species of Theropithecus and Papio in the Shungura Formation.
Three geological members (E, F, and G) provide enough preserved material to compare differences in dental microwear textures between the two genera (Appendix 1b).
As expected due to their different diets, Theropithecus gelada and all extant Papio species considered here differ from one another. Nonetheless, although extant Papio exhibit a higher heterogeneity of complexity, they do not differ from T. gelada when considering anisotropy. This was unexpected given the higher consumption of tough herbaceous monocots by the latter taxon. Hence, from a mechanical perspective, extant Theropithecus and Papio only differ in size and variability of wear-causing particles and therefore by a broader diet for Papio.
Theropithecus from members E and F, having lower anisotropy than their extant relatives, probably ate fewer tough herbaceous monocots and maybe more herbaceous dicots, whereas Theropithecus from Member G might have had a similar diet to T. gelada from Guassa35.
Papio from members E and F show a lower heterogeneity of complexity than the three extant species. This can be interpreted as a more restricted diet for the two samples of extinct Papio or a seasonal bias in the fossil records where more specimens died at a given season than at another one. Conversely, Papio from Member G do not differ from P. hamadryas cynocephalus, but probably had a more restricted diet than P. hamadryas anubis and P. hamadryas hamadryas, as well as incorporating fewer tough plants than P. hamadryas cynocephalus.
The main goal of this analysis was to track potential ecological segregation between extinct Papio and Theropithecus recovered in the Shungura Formation. Although the two genera do not differ in members E and F, Papio from Member G differ from the sympatric Theropithecus in having a higher heterogeneity of complexity, reflecting a broader diet in terms of mechanical properties of the food.
When considering the results and the previous isotope analyses, it is possible to conclude that the ecological segregation between the extant geladas and baboons is nested in their evolutionary history, at least from 2.27 Ma onwards. This corresponds to the very base of Member G.
Temporal variations in the diets of terrestrial papionins from the Shungura Formation
Phase I facets of Theropithecus show significant differences between geological members. As Phase I molar facets contribute to the shearing of food items, it can be hypothesized that changes in dental microwear textures on Phase I facets over time would reflect the abundance of tough vegetation, possibly herbaceous monocots in the diet of Theropithecus. Considering Papio, neither Phase I nor Phase II molar facets show differences in microwear textures, suggesting no significant changes in feeding resources over time. This is not unexpected as extant Papio has a varied diet and thus changes in environmental conditions would not impact the whole food stock, but only a portion of it. Conversely, environmental changes would have higher impacts on species like the modern geladas with a narrower range of foods most of them being issued from the herbaceous layer.
Here, instead of exploring variations through geological times for all of the surface texture parameters (see Materials and Methods), a synthetic linear combination was computed to summarize the entire dental microwear texture into a single attribute, rendering fluctuations through time easier to interpret. The WTI (first PC of the PCA computed from textures on Phase I molar facets of Theropithecus) shows significant variations, suggesting fluctuations in the dietary habits of those terrestrial papionins (Fig. 3; Appendix 8).
The first putative dietary change is observed between members B and C at about 2.91 Ma (Fig. 3). Theropithecus from Member B potentially had a more diverse diet than their relatives from Member C. The former might have consumed more hard and brittle foods, which are more mechanically challenging, such as USOs, stems or seeds of herbaceous plants. Geladas at Guassa have been shown to sometimes consume invertebrates, so such food items might also have been eaten by the specimens of Theropithecus recovered in Member B that were analyzed here. Conversely, the specimens from Member C probably had a more restricted diet including fewer hard but more tough food items. In the herbaceous layer, such mechanical properties are notably found for the blades of herbaceous monocots, which are soft and tough36, so Theropithecus recovered in Member C may have ingested more of these plants. Cerling et al.20 analyzed the enamel stable carbon isotope of T. brumpti and T. oswaldi from the Turkana Basin and from Olorgesaillie in southern Kenya. In the current study, the only time window in common with the latter study runs from 3.44 Ma (the base of Member B) to 2.53 Ma (the top of Member C), in which only T. brumpti occurs. Cerling et al.20 found that T. brumpti includes from 55% to 75% of C4 plants in its diet. This is consistent with the results here. However, they did not check for fine temporal dietary fluctuations during this time interval.
The second piece of putative dietary shift evidence is the most significant of all the fluctuations observed (Fig. 3). It happened between the geological members C and D at about 2.53 Ma. Specimens of Theropithecus from Member D had a clearly more diverse diet than the earlier individuals from Member C, and also ate more hard and likely fewer tough foods. When considering food items available both above and underground, Theropithecus specimens from Member D probably had a similar diet to their relatives from Member B as they might have eaten a variety of mechanically challenging foods such as seeds, stems, and USOs from both herbaceous dicots and monocots, as well as invertebrate prey.
The third probable dietary shift happened at about 2.32 Ma, between members E and F (Fig. 3). Theropithecus from Member F likely ingested less quantities of hard foods but more of tougher foods, and their diet was also more restrictive than their relatives from Member E. Similarly to Member C specimens, these Theropithecus may have eaten more herbaceous monocot blades, with fewer hard items such as seeds, stems, or USOs. They probably also ate some leaves of herbaceous dicots.
Environmental proxies and diets of Theropithecus
In order to make inferences on vegetal resources available during the Plio-Pleistocene along the ancestral Omo River, it is essential to combine proxies. The aim here is to test whether the fluctuations in dental microwear textures observed on Theropithecus reflect behavior, or mirror environmental changes occurring at the regional scale. It can be supposed that ephemeral local changes in available resources potentially impacting few individuals are unlikely to be detectable through a random sampling of fossil specimens. This is true whatever the proxy used, either from dental microwear texture or from stable isotope analyses. Only long-lasting regional changes that deeply affect feeding resources will potentially be noticeable from these paleodietary proxies.
DMTA on shearing facets of Shungura Theropithecus indicated several potential dietary shifts. The first one occurred around 2.91 Ma, between members B and C (Fig. 3). In the latter member, the diet of Theropithecus may have incorporated more blades of herbaceous monocots (Fig. 3). By conducting a high-resolution serial isotopic analysis of the ever-growing canines of two hippopotamid specimens from Shungura units B-9 (ca. 3 Ma) and C-9 (ca. 2.5 Ma), Souron et al.71 detected an environmental shift that could be linked with the dietary change observed in Theropithecus. In addition to higher δ13C values, the specimen from the later unit displays a higher intra-tooth amplitude of the δ18O values than the older specimen71. This may reflect a higher seasonality of rainfall in Member C71. Yet, these results should be considered with great caution, given the number of specimens involved (n = 2). Using enamel stable carbon isotopes on one suid genus (Kolpochoerus) and one bovid genus (Tragelaphus), Bibi et al.72 showed a trend towards the incorporation of more C4 plants between units B-10 and C-4/C-5 (2.97 Ma-2.74 Ma). Another isotopic study conducted by Negash et al.73 showed that, in members B and C, reduncins, alcelaphins and aepycerotins all included C4 plants in their diet to different degrees. However, both DMTA and the molar mesowear score do not support a dietary shift towards more grazing, but rather constant browsing habits for tragelaphins in members B and C74. This apparent discrepancy reflects either the consumption of soft, C4-growing monocotyledons that are less tough and poorer in biosilica, or the consumption of C4 dicots such as Amaranthaceae sensu lato (including Chenopodiaceae) that are regionally abundant, especially increasing through the 3.4 Ma-2.6 Ma interval61,63. Some of the Amaranthaceae are C4 and succulent dicots75 and were reported in the pollen fossil records in the Shungura Formation by Bonnefille and Dechamps76. Although pollen assemblages might be subject to different biases, these authors concluded that the vegetation in unit B-10 (ca. 3 Ma) comprises the highest proportion of arboreal taxa (27.7% of harvested pollen) along with the lowest frequency of grass pollen, which still represents no less than 43% of collected pollen76. The authors identified a shift in the stratigraphic unit C-9 (ca. 2.5 Ma) towards a higher proportion of grass pollen (60%) along with the decrease in abundance of montane forest taxa, which represent 21% of the collected pollens in the stratigraphic unit C-7 and no more than 9.5% in C-976. Dental microwear textures of Theropithecus are congruent with the presence of C4 plant-eating ungulates in members B and C, a significant shift around 2.8 Ma in dietary habits towards further grazing habits of certain mammals71,72,73, as well as also being congruent with the pollen evidence. This would support the occurrence of a regional environmental shift around 3 Ma-2.5 Ma characterized by the spread of more open habitats, such as grasslands or shrub land, at the expense of the tree cover.
The second and main putative dietary shift inferred from dental microwear textures of Theropithecus occurred between members C and D at 2.53 Ma. This is characterized by a probable higher consumption of mechanically challenging foods (Fig. 3). Because USOs are predominantly consumed during the dry season by extant geladas at Guassa, though representing no more than 28% of the diet during the driest month35, and because these USOs account for a higher portion of the diet, frequently more than 50%52, in highly seasonal habitats53, this dietary shift could be explained by a harsher seasonality in Member D.
Dental microwear textures of Theropithecus from members E and F highlight another potential dietary change around 2.32 Ma, with a diet more focused on blades from herbaceous monocots for specimens recovered in Member F (Fig. 3). Negash et al.73 invoked a shift towards higher δ13C values when considering all bovid tribes except for tragelaphins, which displayed the opposite trend. This implies a greater C4 intake between Member D (2.53 Ma-2.4 Ma) and Member F (2.32 Ma-2.27 Ma) deposits. The fact that herbaceous monocots seem to be more consumed by both Theropithecus and most of the bovids from Member F suggests that these plants were more abundant in the milieu. It is worth mentioning that based on the combination of DMTA and the stable carbon isotopic composition on tooth enamel74, the tragelaphins kept browsing during Member F deposition, suggesting a more significant ecological segregation with reduncins. What is surprising is that Bonnefille and Dechamps76 have described the Member E as “an almost treeless vegetation […] evidenced by the pollen spectrum of E-4” (p. 206). Interestingly, in F-1, fewer grasses have been identified than in the previous member and there is a slight return of montane forest taxa at 8% and species restricted to riverine forests76. However, even though fewer grass pollens were recovered in the stratigraphic unit F-1, their proportion of 57% is higher than in stratigraphic unit B-10 at 43% and equivalent to unit C-9 at 60%. Therefore, the herbaceous layer probably comprised more monocots in Member F than in Member B.
During the transition from Member F to the lower part of Member G, aepycerotins and tragelaphins both show a decrease in mean δ13C, implying less C4 consumption73. Bibi et al.72 did not detect such a shift when considering tragelaphins from various sites along the lower part of the Member G. Based on direct evidence from pollens gathered by Bonnefille and Dechamps76, in Member G grass pollen accounts for only 53% of the pollinic spectrum and the frequency of riparian forest taxa is the highest, indicating a wetter environment. The WTI on Theropithecus does not reflect significant changes in diet between Member F and the lower part of Member G (Fig. 3). However, the PC3 coordinates (mainly explained by HAsfc and Asfc; Appendix 6) of the analysis vary significantly with a more homogenous, harder, and less tough diet in Member G than in Member F (Appendices 9 and 10).
Conclusions
The present study is the first to apply DMTA on a broad sample of terrestrial papionins, more precisely of Papio and Theropithecus found in the Shungura Formation. Two aspects were evaluated: differences in diet between contemporaneous extinct Papio and Theropithecus in comparison to the diets of their extant relatives, and potential dietary fluctuations along the stratigraphic sequence.
The results here show that, even by 2.27 Ma onwards (lower part of Member G), extinct representatives of the genera Papio and Theropithecus already differed in diet, paralleling the situation showed for their extant relatives. The former probably had a more mixed diet including a greater number of hard food items. When performing a linear combination of all the texture parameters, neither the Phase I nor Phase II molar facets of Papio nor the Phase II facets of Theropithecus displayed temporal variations along the Shungura Formation, from Member B to lower part of Member G. Conversely, the Phase I facets of Theropithecus specimens suggested several dietary shifts along the stratigraphic sequence. Two events were recognized at about 2.91 Ma (between Member B and Member C) and 2.32 Ma (between Member E and Member F) respectively, possibly indicating higher intakes of herbaceous monocots. These two shifts are separated by an inverse trend at about 2.53 Ma (between Member C and Member D), characterized by a higher reliance on mechanically challenging foods.
Together with other environmental proxies, these potential dietary variations complement the understanding of environmental changes that happened during this period. Throughout this time span, terrestrial cercopithecines may have shared habitats with early hominins. The dietary habits of early human ancestors are still a matter of debate. The contribution of animal matter and challenging foods, such as USOs, silica-bearing vegetation, hard seeds, and the contribution of exogenous particles are the subject of discussion among paleoanthropologists. Beyond the differences in molar morphology that would limit direct comparisons of dental microwear textures, the authors here support the idea that the terrestrial cercopithecines analyzed in the present study constitute an appropriate model to best interpret the niche partitioning between extinct hominins.
References
Disotell, T. R., Honeycutt, R. L. & Ruvolo, M. Mitochondrial DNA phylogeny of the Old-World monkey tribe Papionini. Mol. Biol. Evol. 9, 1–13 (1992).
Frost, S. R., Marcus, L. F., Bookstein, F. L., Reddy, D. P. & Delson, E. Cranial allometry, phylogeography, and systematics of large-bodied papionins (primates: Cercopithecinae) inferred from geometric morphometric analysis of landmark data. Anat. Rec. A. Discov. Mol. Cell. Evol. Biol. 275A, 1048–1072 (2003).
Newman, T. K., Jolly, C. J. & Rogers, J. Mitochondrial phylogeny and systematics of baboons (Papio). Am. J. Phys. Anthropol. 124, 17–27 (2004).
Zinner, D., Groeneveld, L. F., Keller, C. & Roos, C. Mitochondrial phylogeography of baboons (Papio spp.) – Indication for introgressive hybridization? BMC Evol. Biol. 9, 83 (2009).
Hall, K. R. L. Ecology and behavior of baboons, patas, and vervet monkeys in Uganda. Baboon Med. Res. 43–61 (1965).
Nagel, U. A Comparison of Anubis Baboons, Hamadryas Baboons and Their Hybrids at a Species Border in Ethiopia. Folia Primatol. (Basel) 19, (104–165 (1973).
Post, D. G., Hausfater, G. & McCuskey, S. Feeding Behavior of Yellow Baboons (Papio cynocephalus): Relationship to Age, Gender and Dominance Rank. Folia Primatol. (Basel) 34, 170–195 (1980).
Rhine, R. J., Norton, G. W., Wynn, G. M., Wynn, R. D. & Rhine, H. B. Insect and meat eating among infant and adult baboons (Papio cynocephalus) of Mikumi National Park, Tanzania. Am. J. Phys. Anthropol. 70, 105–118 (1986).
Norton, G. W., Rhine, R. J., Wynn, G. W. & Wynn, R. D. Baboon Diet: A Five-Year Study of Stability and Variability in the Plant Feeding and Habitat of the Yellow Baboons (Papio cynocephalus) of Mikumi National Park, Tanzania. Folia Primatol. (Basel) 48, 78–120 (1987).
Rowe, N. Pictorial guide to the living primates. (Pogonias Press, 1996).
Kunz, B. K. & Linsenmair, K. E. Changes in Baboon Feeding Behavior: Maturity-dependent Fruit and Seed Size Selection within a Food Plant Species. Int. J. Primatol. 28, 819–835 (2007).
Scott, R. S., Teaford, M. F. & Ungar, P. S. Dental microwear texture and anthropoid diets. Am. J. Phys. Anthropol. 147, 551–579 (2012).
Lee-Thorp, J. A., van der Merwe, N. J. & Brain, C. K. Isotopic evidence for dietary differences between two extinct baboon species from Swartkrans. J. Hum. Evol. 18, 183–189 (1989).
Codron, D. et al. Utilization of savanna-based resources by Plio-Pleistocene baboons. South Afr. J. Sci. 101, 245–249 (2005).
Cerling, T. E. et al. Diet of Paranthropus boisei in the early Pleistocene of EastAfrica. Proc. Natl. Acad. Sci. 108, 9337–9341 (2011).
Cerling, T. E., Chritz, K. L., Jablonski, N. G., Leakey, M. G. & Manthi, F. K. Diet of Theropithecus from 4 to 1 Ma in Kenya. Proc. Natl. Acad. Sci. 110, 10507–10512 (2013).
Levin, N. E., Haile-Selassie, Y., Frost, S. R. & Saylor, B. Z. Dietary change among hominins and cercopithecids in Ethiopia during the early Pliocene. Proc. Natl. Acad. Sci. 112, 12304–12309 (2015).
Wynn, J. G. et al. Dietary flexibility of Australopithecus afarensis in the face of paleoecological change during the middle Pliocene: Faunal evidence from Hadar, Ethiopia. J. Hum. Evol. 99, 93–106 (2016).
Souron, A. Morphology, diet, and stable carbon isotopes: On the diet of Theropithecus and some limits of uniformitarianism in paleoecology. Am. J. Phys. Anthropol. (2018).
Berthaume, M. A. Food mechanical properties and dietary ecology. Am. J. Phys. Anthropol. 159, 79–104 (2016).
Dominy, N. J., Vogel, E. R., Yeakel, J. D., Constantino, P. & Lucas, P. W. Mechanical Properties of Plant Underground Storage Organs and Implications for Dietary Models of Early Hominins. Evol. Biol. 35, 159–175 (2008).
Lucas, P. W., Turner, I. M., Dominy, N. J. & Yamashita, N. Mechanical defences to herbivory. Ann. Bot. 86, 913–920 (2000).
Merceron, G. et al. Untangling the environmental from the dietary: dust does not matter. Proc R Soc B 283, 20161032 (2016).
Ramdarshan, A. et al. Seeds, browse, and tooth wear: a sheep perspective. Ecol. Evol. 6, 5559–5569 (2016).
Scott, J. R. Dental microwear texture analysis of extant African Bovidae. mammalia 76, 157–174 (2012).
Ramdarshan, A., Blondel, C., Gautier, D., Surault, J. & Merceron, G. Overcoming sampling issues in dental tribology: Insights from an experimentation on sheep. Palaeontol. Electron. https://doi.org/10.26879/762 (2017).
Schulz, E. et al. Dietary abrasiveness is associated with variability of microwear and dental surface texture in rabbits. PLoS One 8, e56167 (2013).
Heinzelin, J. de. The Omo group: archives of the international Omo Research Expedition (Musée Royale de l’Afrique Centrale, 1983).
Maurin, T., Delagnes, A. & Boisserie, J.-R. Spatial behaviours of Early Oldowan toolmakers in the Shungura Formation (Lower Omo Valley, Ethiopia): Proposal for an integrated approach. Comptes Rendus Palevol 13, 737–746 (2014).
Maurin, T., Bertran, P., Delagnes, A. & Boisserie, J.-R. Early hominin landscape use in the Lower Omo Valley, Ethiopia: Insights from the taphonomical analysis of Oldowan occurrences in the Shungura Formation (Member F). J. Hum. Evol. 111, 33–53 (2017).
Delagnes, A. et al. Archaeological investigations in the Lower Omo Valley (Shungura Formation, Ethiopia): New data and perspectives. J. Hum. Evol. 61, 215–222 (2011).
McDougall, I. & Brown, F. H. Geochronology of the pre-KBS Tuff sequence, Omo Group, Turkana Basin. J. Geol. Soc. 165, 549–562 (2008).
McDougall, I. et al. New single crystal 40Ar/39Ar ages improve time scale for deposition of the Omo Group, Omo–Turkana Basin, East Africa. J. Geol. Soc. 169, 213–226 (2012).
Frost, S. Fossil cercopithecidae of the Afar depression, Ethiopia: species systematics and comparison to the Turkana basin. (The City University of New York, Graduate Faculty in Anthropology, 2001).
Fashing, P. J., Nguyen, N., Venkataraman, V. V. & Kerby, J. T. Gelada feeding ecology in an intact ecosystem at Guassa, Ethiopia: Variability over time and implications for theropith and hominin dietary evolution. Am. J. Phys. Anthropol. 155, 1–16 (2014).
Venkataraman, V. V. et al. Effects of dietary fracture toughness and dental wear on chewing efficiency in geladas (Theropithecus gelada). Am. J. Phys. Anthropol. 155, 17–32 (2014).
Shapiro, A. E., Venkataraman, V. V., Nguyen, N. & Fashing, P. J. Dietary ecology of fossil Theropithecus: Inferences from dental microwear textures of extant geladas from ecologically diverse sites. J. Hum. Evol. 99, 1–9 (2016).
Scott, R. S. et al. Dental microwear texture analysis shows within-species diet variability in fossil hominins. Nature 436, 693–695 (2005).
Scott, R. S. et al. Dental microwear texture analysis: technical considerations. J. Hum. Evol. 51, 339–349 (2006).
Calandra, I. & Merceron, G. Dental microwear texture analysis in mammalian ecology. Mammal Rev. 46, 215–228 (2016).
Kay, R. F. & Hiiemae, K. M. Jaw movement and tooth use in recent and fossil primates. Am. J. Phys. Anthropol. 40, 227–256 (1974).
Kay, R. F. The functional adaptations of primate molar teeth. Am. J. Phys. Anthropol. 43, 195–215 (1975).
Meikle, W. E. Molar wear stages in Theropithecus gelada. Kroeber Anthropol. Soc. Pap. 50, 21–25 (1977).
Ungar, P. S., Brown, C. A., Bergstrom, T. S. & Walker, A. Quantification of Dental Microwear by Tandem Scanning Confocal Microscopy and Scale-Sensitive Fractal Analyses. Scanning 25, 185–193 (2003).
Teaford, M. F. & Oyen, O. J. In vivo and in vitro turnover in dental microwear. Am. J. Phys. Anthropol. 80, 447–460 (1989).
Teaford, M. F., Ungar, P. S., Taylor, A. B., Ross, C. F. & Vinyard, C. J. In vivo rates of dental microwear formation in laboratory primates fed different food items. Biosurface Biotribology, https://doi.org/10.1016/j.bsbt.2017.11.005 (2017).
Lucas, P. W. et al. Mechanisms and causes of wear in tooth enamel: implications for hominin diets. J. R. Soc. Interface 10, 20120923 (2013).
Constantino, P. J., Borrero-Lopez, O., Pajares, A. & Lawn, B. R. Simulation of enamel wear for reconstruction of diet and feeding behavior in fossil animals: A micromechanics approach. BioEssays 38, 89–99 (2016).
Krueger, K. L., Scott, J. R., Kay, R. F. & Ungar, P. S. Technical note: Dental microwear textures of “Phase I” and “Phase II” facets. Am. J. Phys. Anthropol. 137, 485–490 (2008).
Conover, W. J. & Iman, R. L. Rank Transformations as a Bridge Between Parametric and Nonparametric Statistics. Am. Stat. 35, 124–129 (1981).
Sokal, R. R. & Rohlf, F. J. Biometry. The principles and practice of statistics in biological research. New York: W. E. Freeman and Company, p. 887 (1969).
Iwamoto, T. The ecology of Theropithecus gelada. In Theropithecus: The rise and fall of a primate genus 441–452 (1993).
Abu, K., Mekonnen, A., Bekele, A. & Fashing, P. J. Diet and activity patterns of Arsi geladas in low-elevation disturbed habitat south of the Rift Valley at Indetu, Ethiopia. Primates, https://doi.org/10.1007/s10329-017-0640-9 (2017).
Liem, K. F. Adaptive Significance of Intra- and Interspecific Differences in the Feeding Repertoires of Cichlid Fishes. Am. Zool. 20, 295–314 (1980).
Lambert, J. E., Chapman, C. A., Wrangham, R. W. & Conklin-Brittain, N. L. Hardness of cercopithecine foods: Implications for the critical function of enamel thickness in exploiting fallback foods. Am. J. Phys. Anthropol. 125, 363–368 (2004).
Lambert, J. E. Seasonality, fallback strategies, and natural selection: a chimpanzee and Cercopithecoid model for interpreting the evolution of the hominin diet. In Evolution of the human diet: The known, the unknown, and the unknowable 324–343 (2007).
Marshall, A. J., Boyko, C. M., Feilen, K. L., Boyko, R. H. & Leighton, M. Defining fallback foods and assessing their importance in primate ecology and evolution. Am. J. Phys. Anthropol. 140, 603–614 (2009).
Grine, F. E. Dental evidence for dietary differences in Australopithecus and Paranthropus: a quantitative analysis of permanent molar microwear. J. Hum. Evol. 15, 783–822 (1986).
Teaford, M. F. Dental microwear and diet in extant and extinct Theropithecus: preliminary analyses. in Theropithecus: The life and death of a primate genus. Cambridge: Cambridge University Press. p 331–349 (1993).
El-Zaatari, S., Grine, F. E., Teaford, M. F. & Smith, H. F. Molar microwear and dietary reconstructions of fossil cercopithecoidea from the Plio-Pleistocene deposits of South Africa. J. Hum. Evol. 49, 180–205 (2005).
Bonnefille, R. Cenozoic vegetation, climate changes and hominid evolution in tropical Africa. Glob. Planet. Change 72, 390–411 (2010).
Rossouw, L. & Scott, L. Phytoliths and pollen, the microscopic plant remains in Pliocene volcanic sediments around Laetoli, Tanzania. In Paleontology and geology of Laetoli: Human evolution in context 201–215 (Springer, 2011).
Feakins, S. J. et al. Northeast African vegetation change over 12 m.y. Geology 41, 295–298 (2013).
Albert, R. M. et al. Vegetation landscape at DK locality, Olduvai Gorge, Tanzania. Palaeogeogr. Palaeoclimatol. Palaeoecol. 426, 34–45 (2015).
Tieszen, L. L., Senyimba, M. M., Imbamba, S. K. & Troughton, J. H. The distribution of C3 and C4 grasses and carbon isotope discrimination along an altitudinal and moisture gradient in Kenya. Oecologia 37, 337–350 (1979).
Livingstone, D. A. & Clayton, W. D. An altitudinal cline in tropical African grass floras and its paleoecological significance. Quat. Res. 13, 392–402 (1980).
Young, H. J. & Young, T. P. Local distribution of C3 and C4 grasses in sites of overlap on Mount Kenya. Oecologia 58, 373–377 (1983).
Levin, N. E., Simpson, S. W., Quade, J., Cerling, T. E. & Frost, S. R. Herbivore enamel carbon isotopic composition and the environmental context of Ardipithecus at Gona, Ethiopia. Geol Soc Am Spec Pap 446, 215–234 (2008).
Merceron, G. et al. Folivory or fruit/seed predation for Mesopithecus, an earliest colobine from the late Miocene of Eurasia? J. Hum. Evol. 57, 732–738 (2009).
Percher, A. M. et al. Dental microwear textural analysis as an analytical tool to depict individual traits and reconstruct the diet of a primate. Am. J. Phys. Anthropol. https://doi.org/10.1002/ajpa.23337 (2017).
Souron, A., Balasse, M. & Boisserie, J.-R. Intra-tooth isotopic profiles of canines from extant Hippopotamus amphibius and late Pliocene hippopotamids (Shungura Formation, Ethiopia): Insights into the seasonality of diet and climate. Palaeogeogr. Palaeoclimatol. Palaeoecol. 342–343, 97–110 (2012).
Bibi, F., Souron, A., Bocherens, H., Uno, K. & Boisserie, J.-R. Ecological change in the lower Omo Valley around 2.8 Ma. Biol. Lett. 9, 20120890 (2013).
Negash, E. W., Alemseged, Z., Wynn, J. G. & Bedaso, Z. K. Paleodietary reconstruction using stable isotopes and abundance analysis of bovids from the Shungura Formation of South Omo, Ethiopia. J. Hum. Evol. 88, 127–136 (2015).
Blondel, C. et al. Feeding ecology of Tragelaphini (Bovidae) from the Shungura Formation, Omo Valley, Ethiopia: Contribution of dental wear analyses. Palaeogeogr. Palaeoclimatol. Palaeoecol. doi:10.1016/j.palaeo.2018.01.027 (2018).
Ehleringer, J. R., Cerling, T. E. & Helliker, B. R. C4 photosynthesis, atmospheric CO2, and climate. Oecologia 112, 285–299 (1997).
Bonnefille, R. & Dechamps, R. Data on fossil flora. In Ann. Mus. roy. Afr. Centr., Tervuren, Sci. geol 85, 191–207 (1983).
Abu, K., Mekonnen, A., Bekele, A. & Fashing, P. J. Diet and activity patterns of Arsi geladas in low-elevation disturbed habitat south of the Rift Valley at Indetu, Ethiopia. Primates 1–9 https://doi.org/10.1007/s10329-017-0640-9 (2017).
Welch, C., Frost, T. B. & Kerby, J. Where theWorld’s Only Grass-Eating Monkeys Thrive. Nationa Geographic (2017). Available at: https://www.nationalgeographic.com/magazine/2017/04/gelada-monkeys-grass-eating-guassa-ethiopia-bleeding-heart/. (Accessed: 14th May 2018).
Acknowledgements
We thank the Authority for Research and Conservation of Cultural Heritage (ARCCH) in Addis Ababa, Ethiopia, for granting us permission to sample primate dental remains from the Omo collections. The authors are also grateful to J. Cuisin from the Muséum National d’Histoire Naturelle, Paris, France; O. Röhrer-Ertl from the Zoologische Staatssammlung of Munich; G. Storch and J. Anger from the Senckenberg Museum of Frankfurt, Germany for providing access to the cranio-dental collections. This study was financed by the ANR TRIDENT Project (ANR-13-JSV7-0008-01, PI: G. Merceron, http://anr-trident.prd.fr/). This research was also funded within the framework of the Omo Group Research Expedition (Agence Nationale pour la Recherche: projects EVAH ANR-09-BLAN-0238 and OLD ANR-CE27-0009-02). This study was undertaken as part of the Master thesis of C-A. Plastiras and F. Martin. C-A. Plastiras (Aristotle University of Thessaloniki, Greece) thanks the ERASMUS + program which granted his stay at the University of Poitiers for his Master thesis. The authors thank G. Reynaud and M. Pourade (PALEVOPRIM, Poitiers) for administrative guidance, and A. Percher (ISEM, Montpellier), A. Ramdarshan and E. Berlioz (PALEVOPRIM, Poitiers) for fruitful discussions. S. Ramdarshan (https://srtranslations.wordpress.com/) corrected the English of the manuscript.
Author information
Authors and Affiliations
Contributions
G.M. and J.-R.B. designed the research; G.M., F.M. and C.-A.P. acquired and analyzed the data; F.M. and G.M. wrote the paper. All authors contributed to the discussion and reviewed the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Martin, F., Plastiras, CA., Merceron, G. et al. Dietary niches of terrestrial cercopithecines from the Plio-Pleistocene Shungura Formation, Ethiopia: evidence from Dental Microwear Texture Analysis. Sci Rep 8, 14052 (2018). https://doi.org/10.1038/s41598-018-32092-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-32092-z
Keywords
This article is cited by
-
Identifying the Impact of Soil Ingestion on Dental Microwear Textures Using a Wild Boar Experimental Model
Journal of Archaeological Method and Theory (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.