Abstract
We evaluated automated OCT-derived drusen volume measures in a population-based study (n = 4,512) aged ≥40 years, and its correlation with conventional color fundus photographs (CFP)-derived early AMD features. Participants had protocol-based assessment to capture medical and ocular history, genotyping for SNPs in CFH, ARMS2, and CETP, CFP-based AMD grading and automated drusen volume based on SD-OCT using built-in software (Cirrus OCT advanced RPE analysis software). Significantly fewer eyes with early AMD features (drusen, hyperpigmentation, soft or reticular drusen) had drusen volume = 0 mm3 (p < 0.001). In eyes with drusen volume > 0 mm3, increasing AMD severity was associated with increase in drusen volume (correlation coefficient 0.17, p < 0.001). However 220 (59.14%) of 372 participants with AMD based on CFP grading had drusen volume = 0 mm3. Factors associated with drusen volume included age (OR 1.42 per 5 years, 95% confidence interval [CI] 2.76, 4.48), systolic blood pressure (OR1.00, 95% CI 1.00, 1.01), ethnic Malay (OR 1.54, 95% CI 1.29, 1.83) and Chinese (OR 1.66, 95% CI 1.37, 2.01) compared to Indian. The ARMS2 rs10490924 T allele was associated with increased drusen volume in subjects with AMD (multivariable adjusted OR1.54, 95% CI 1.08, 2.19). Automated OCT-derived drusen volume is correlated with CFP-based AMD grading in many, but not all subjects. However the agreement is not good. These two modalities provide complementary information and should be incorporated into future studies.
Similar content being viewed by others
Introduction
Drusen are extracellular deposits accumulating within Bruch’s membrane or between the retinal pigment epithelium (RPE) and Bruch’s membrane on the apical side of the RPE, and are considered the hallmark of age-related macular degeneration (AMD). Increasing severity of baseline drusen characteristics and pigmentary changes have been shown to be important predictors for progression of AMD1,2. The gold standard method for qualitative and quantitative assessment of drusen has traditionally been based on assessment of high quality stereoscopic color fundus photographs (CFP), typically using modifications of the Wisconsin AMD grading system3. This method has been employed in many population-based studies around the world, including the Beaver Dam and Blue Mountains Eye Studies4. However, there are limitations of CFP-based grading, both due to technical reasons as well as due to the labor intensive, time-consuming nature of CFP assessment.
Optical coherence tomography (OCT), particularly spectral domain OCT (SD-OCT) is increasingly used in clinical studies and trials to evaluate AMD5. Recently, fully automated measurements of drusen volume from OCT have become possible with commercially available software. Several, mostly smaller clinic-based, studies have validated the accuracy and reproducibility of these automated OCT-based algorithms5,6,7,8,9. A few population-based studies have further correlated OCT-derived drusen measures with demographic risk factors10, genetic variants11 and risk of AMD progression12.
To date, there have been no studies correlating fully automated OCT-derived drusen volume measurements with conventional CFP grading in population studies in Asians. Previous clinical and epidemiological studies have suggested that drusen characteristics based on CFP grading in Asians and the risk they pose on AMD progression may be different from those in white populations13,14. In this study, we investigated the correlation of automated OCT-derived drusen volume with CFP-derived Wisconsin AMD grading and specific AMD features from a large population-based study. In addition, we evaluated associations of AMD risk factors with OCT-derived drusen volume.
Methods
Study Population
Data for this analysis were derived from the Singapore Epidemiology of Eye disease (SEED) program, a population-based cohort study of eye diseases in adults aged 40 years and older living in Singapore. Specifically, this report analyzes SD-OCT measurements, which were incorporated into the examination protocols since 2009. The SEED program was approved by the Institutional Review Board of the Singapore Eye Research Institute, Singapore and conducted in accordance with the Declaration of Helsinki. All participants provided written informed consent.
Detailed methodology and protocols for the examinations have been described elsewhere15,16. Briefly the sampling frame was composed of adults aged 40–80 years living in designated study areas in south-western Singapore during each stipulated study period, derived from a computer-generated list provided by the Singapore Ministry of Home Affairs using an age-stratified (by 10-year age groups) random sampling method.
Ophthalmic examination and AMD grading
All participants had an interview, systemic examination, and laboratory investigations to determine socioeconomic, ocular, and systemic risk factors. After pupil dilation with 1.0% tropicamide, CFP were taken according to standardized protocol. Specifically, CFP of Early Treatment for Diabetic Retinopathy Study17 standard fundus fields 1 (centered on the optic disc) and 2 (centered on the fovea) were obtained for both eyes using a digital retinal camera (Canon CR-1 Mark-II Nonmydriatic Digital Retinal Camera, Canon). The Centre for Vision Research, University of Sydney, performed AMD grading using the Wisconsin Age-Related Maculopathy Grading System modified for magnification3,18.
AMD status was classified per eye: early AMD was defined as presence of either any soft drusen (distinct or indistinct) plus pigmentary abnormalities, reticular drusen, or large soft drusen >125 µm in diameter with drusen area >500 µm-diameter. Late AMD was defined as the presence of neovascular AMD or geographic atrophy (GA). Neovascular AMD included serous or hemorrhagic detachment of the RPE or sensory retina, and the presence of subretinal or sub-RPE hemorrhages or subretinal fibrous scar tissue3,15,16,19,20. GA was characterized by sharply edged, roughly round or oval areas of RPE hypopigmentation, with clearly visible choroidal vessels. The minimum diameter of GA was 175 um, or larger3,15,16,19,20. All CFP were graded initially in a masked manner by two graders and discrepancies were adjudicated by a senior retinal specialist (PM).
Optical Coherence Tomography Acquisition
Each eye was scanned after pupil dilation with Cirrus OCT 4000 with 512 A-scans × 128 B-scans over a 6- × 6-mm2 area, centered on the fovea. RPE analysis was performed using Cirrus Version 6.0 Advanced RPE Analysis algorithm. Quantitative measurement of drusen volume was obtained for the retinal region centered on the fovea with diameter of 5 mm, derived from the distance between the real RPE and the interpolated RPE floor. Six hundred and sixty-one eyes were excluded because of low signal strength (<6) or presence of pathology which was thought could affect the accuracy of RPE-fit line which represents the logarithm-determined Bruch’s membrane (vitreomacular traction, epiretinal membranes or extensive peripapillary atrophy).
Assessment of Risk Factors
A detailed interviewer-administered questionnaire was used to collect information about medical history (including hypertension, diabetes, angina, myocardial infarction and stroke), cigarette smoking (defined as current, past and never). The following information was collected: demographic information, socioeconomic characteristics (education, income level and occupation), family and medical history, and lifestyle factors (smoking, alcohol). We defined age as the age at examination. We categorized cigarette smoking into current smokers, former smokers or nonsmokers. Alcohol consumption was categorized into drinkers (those who reported having drunk alcohol in the past 3 months, irrespective of quantity) and non-drinkers. The participant’s height was measured in centimeters using a wall-mounted measuring tape. Weight was measured in kilograms using a digital scale (SECA, model 7822321009: Vogel & Halke, Hamburg, Germany). We calculated body mass index (BMI) using weight divided by the square of body height. Blood pressure was taken with the participant seated and after 5 minutes of rest. Systolic and diastolic blood pressure and pulse rate were measured with a digital automatic blood pressure monitor (Dinamap model Pro Series DP110X-RW, 100V2; GE Medical Systems Information Technologies Inc., Milwaukee, USA). Blood pressure was measured on two occasions 5 minutes apart. If the blood pressures differed by more than 10 mmHg in systolic or 5 mmHg in diastolic, a third measurement was taken. The blood pressure of the participant was defined as the mean between the two closest readings. A 40 mL sample of non-fasting venous blood was collected for the assessment of Hemoglobin A1c (HbA1c), serum glucose and lipid levels. All serum biochemistry tests were performed in the Singapore General Hospital Laboratory on the same day.
Genotyping
We extracted DNA from venous blood samples from all participants. Genotyping was performed using Illumina Human OmniExpres or Human Hap610-Quad Beadchip. For the current analysis, we evaluated the following four SNPs: CFH rs900292, ARMS2 rs10490924, CETP rs3764261 and rs2303790 (D442G). Genetic ancestry was inferred using principal component (PC) analysis to account for spurious associations resulting from ancestral differences of individual SNPs. Details of the PC analysis is reported in detail elsewhere21,22.
Statistical Methods
All the statistical analysis was performed using R software version 3.2.223. In order to account for the highly skewed distribution of drusen volume, the dataset was dichotomized into 2 categories: Drusen volume = 0 (n = 7,312) and Drusen volume > 0 mm3 (n = 1, 301) and the association between exposure variables (e.g. age, gender). Relationship between drusen volume and CFP-derived AMD outcomes (early and late AMD) and specific early AMD features was first assessed through trend test which tests the hypothesis of an increased trend of drusen volume with each ordered individual CFP grading parameter, using non-parametric Cuzik’s trend test23,24 for continuous response, and Cochran-Armitage test25,26 for binary response of drusen volume (cutoff at 0 mm3). As Cuzik’s trend test assumes no correlation in the dataset, only the worse eye is selected from each participant for the test (n = 3795 and n = 717 for Drusen volume = 0 and > 0 mm3, respectively). In addition, the strength of association between Drusen volume and AMD was also evaluated using Spearman rank correlation test. In regression analysis, drusen volume of both eyes (outcome variable) was assessed using logistic regression analysis with generalized estimating equations to account for the inter-correlation between left and right eyes. The association is firstly assessed with each single exposure factor, and then, the association was further controlled for the factors which are significant in the univariable model, including age, gender, ethnicities, LDL, anti-cholesterol medication, history of cardiovascular disease, creatinine, glomerular filtration rate, blood pressure and genetic factors, plus factors and AMD status. The association between genetic factors (including CFH, ARMS2, CETP and D442G) and drusen volume was assessed through logistic regressions in participants stratified by AMD status. Univariable and multivariable logistic regression models were constructed after no adjustment or adjusting for age, gender and principal components 1–2 from principal component analysis of genetic data.
Results
We included 4,512 subjects of Chinese (n = 1, 294) and Malay (n = 1, 346) and Indian (n = 1, 872) ethnicity. The mean age was 59.1 years. Of the 4,521 participants, 348 had early AMD and 24 subjects had late AMD in the worse eye. Bilateral AMD was present in 92 participants. For CFH, 486 subjects (14.79%) were homozygous and 1, 552 subjects (47.25%) were heterozygous for rs800292_A respectively. For ARMS2, 479 subjects (14.69%) were homozygous and 1, 562 (47.91%) were heterozygous for the rs104909274_T. For CETP, 151 subjects (4.60%) were homozygous and 1, 027 subjects (31.24%) were heterozygous for rs3764261_A; whereas 1 subject (0.03%) was homozygous and 54 subjects (1.67%) were heterozygous for D442G. Detailed baseline characteristics are summarized in Table 1.
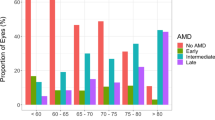
Drusen volume was markedly skewed towards 0 mm3. Drusen volume was 0 mm3 in 3,795 (84.11%) of 4,512 participants, and 3,575 (94.20%) had no AMD according to CFP grading. In the remaining 717 participants with drusen volume > 0 mm3, only 152 (21.20%) had AMD based on CFP grading. Despite this skewed deviation, the proportion of eyes with drusen volume = 0 mm3 significantly decreased from no AMD (86.36%) to early AMD (62.25%) to late AMD (12.5%), p < 0.001 The correlation coefficient between drusen volume and AMD status in all 4,512 participants is 0.21 (p < 0.001). In participants in whom early AMD features evaluated were present (any drusen, pigmentation, soft and reticular drusen), there were also significantly lower percentage of participants with drusen volume = 0 mm3 than that in participants with no AMD feature (p < 0.001). Similarly, with increasing drusen size, number and area, significantly fewer proportion of participants have drusen volume = 0 mm3 p < 0.001. Among the remaining 717 participants with drusen volume > 0 mm3, there was a significant trend of increasing drusen volume as AMD severity increases (correlation coefficient 0.17, p < 0.001). Among the individual early AMD features evaluated, drusen volume was significantly increased in eyes with reticular drusen, p = 0.005. (Table 2)
Next, the association of drusen volume with possible AMD risk factors was assessed using logistic regression. In the univariate regression model, drusen volume was associated with age, total cholesterol, LDL-cholesterol, anti- hyperlipidemia medication, glomerular filtration rate, systolic blood pressure, history of cardiovascular disease, and ethnic Malay (compared to Indian ethnicity) and presence of any AMD. (Table 3) In the multivariable regression model, higher drusen volume was associated with older age (OR 1.42 per 5 years, 95% CI 2.76, 4.48), systolic blood pressure (OR 1.00, 95% CI 1.00, 1.01), ethnic Malay (OR 1.54, 95% CI 1.29, 1.83) and Chinese (OR 1.66, 95% CI 1.37, 2.01) compared to Indian and presence of any AMD (OR 3.52, 95% CI 2.76, 4.48) remained significantly associated with higher drusen volume.
Amongst the AMD risk genotypes evaluated, presence of the ARMS2 rs10490924 T allele was associated with increased drusen volume (multivariable adjusted OR1.54, 95% CI 1.08, 2.19) in subjects with AMD (n = 347). (Table 4) None of the genotypes evaluated was significantly associated with increased drusen volume in subjects without AMD.
Discussion
In this population-based study using data from CFP and SD-OCT-derived drusen volume measured from 4512 subjects, we showed automated measurement of drusen volume increased with AMD severity based on traditional Wisconsin AMD grading parameters using CFP. We note that drusen volume was markedly skewed towards 0 mm3, and not surprisingly, majority of participants with no AMD based on CFP grading had drusen volume = 0 mm3. Interestingly, 220 (59.14%) of 372 participants with AMD based on CFP grading also had drusen volume = 0 mm3. (Fig. 1) This discrepancy is likely due to eyes with small drusen and shallow RPE distortion27,28. Conversely, 152 participants had drusen volume > 0 mm3 but no AMD based on CFP grading. Several small studies have previously compared drusen area and volume measured based on SD-OCT versus CFP and reported different levels of agreement between these two modalities. Jain et al. evaluated drusen area reported mean agreement in 82% in 12 eyes with non-neovascular AMD27. Freeman et al. also reported ‘strong and significant correlation’ between drusen volume and AREDS-determined drusen area in 36 eyes with non-exudative AMD, but added that the correlation is not perfect28. In contrast, Yehoshua et al. reported only fair agreement between drusen area measurements obtained from SD-OCT images and CFP based on 50 eyes with drusen secondary to non-exudative AMD29. Some disagreement between the two modalities could be explained by the different properties of drusen being detected, namely pigmentary changes on CFP as opposed to distortion in RPE geometry on SD-OCT. As a result, CFP may be less accurate, particularly in eyes with large drusen areas whereas SD-OCT may be less sensitive in eyes with small drusen and shallow RPE distortion28,29. The findings from the current study with the largest sample size comparing these two modalities further highlight that these two imaging modalities provide complementary information regarding RPE changes in AMD.
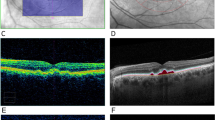
Examples of comparison between cross-sectional OCT through red dashed arrow, color fundus photographs (CFP) and OCT-derived drusen volume map. In (a), soft drusen can be seen on CFP and RPE elevation can be seen in corresponding cross-sectional OCT. This area was reflected as increased drusen volume (in turquoise) on the enface map. In (b), only hyperpigmentation can be seen on CFP. The corresponding cross-sectional OCT showed shallow and irregularly elevated RPE. A wider area of elevated drusen volume is highlighted in turquoise in the enface drusen volume map. In (c), cuticular (hard) drusen are seen on CFP, and very mild elevation of RPE is seen in a localized area. No significant elevation of drusen volume was reflect on the enface map.
For several decades CFP-based grading has been the most widely-used imaging modality to assess AMD in population and clinical studies3,19,20,30,31,32,33. Features assessed include drusen characteristics as well as pigmentary changes for early AMD, whereas signs of pigment epithelial detachment, choroidal neovascularization and GA are the key features assessed for late AMD. AREDS 1 and AREDS2 reported that baseline drusen characteristics (including drusen area and maximum drusen size) and pigmentary abnormalities are strongly predictive of subsequent risk of development of neovascular AMD2,34. We recently also validated the predictive value of the AREDS simplified severity scale in the six-year incidence of late AMD in Singaporean Malays20 and in the 5- and 10-year incidence of AMD in the Blue Mountains Eye Study35. However, reliable CFP quantification may be challenging due to difficulty in identifying clear drusen borders, particularly in photographs with low contrast. Assessment of maximum drusen size based on estimation by visual comparison to a set of standardized circles lacks precision. Furthermore, categorical grading is somewhat arbitrary. Finally, detailed grading of these parameters by trained professional graders, usually in centralized reading centers (e.g. University of Wisconsin or University of Sydney) remains time-consuming, labor intensive and costly. With the advent of SD-OCT, detailed cross-sectional imaging of drusen and the RPE can be obtained, thus enabling a three-dimensional rather than two-dimensional assessment of the macula, which may improve the precision of AMD feature grading20. Several groups have evaluated manual and subsequently customized semi-automated and automated quantification strategies and reported satisfactory repeatability and reproducibility, particularly for drusen volume5,6,7,8,9,36. More recently, fully automated drusen-volume quantification software has become commercially available (Carl Zeiss Meditec Inc, Dublin, CA, USA). This software allows for much higher throughput quantitative assessments of drusen volume, with highly reproducible results29,37. Incorporating such automated drusen volume measurement in future population study protocols is likely a natural trend. The current study is the largest population study to-date to provide information on the agreement between CFP and automated drusen volume, and also provides information on other factors which may influence drusen volume.
Regarding factors which may influence drusen volume, the strongest association was seen with the presence of any AMD (OR 3.52, p < 0.001). However, we also observed that increasing drusen volume was associated with older age, systolic blood pressure, ethnic Malay and Chinese compared to Indian ethnicity. These results are in keeping with previous report by Diniz et al. in non-AMD subjects aged 50 + years in which perifoveal drusen area was associated with increasing age, systolic blood pressure and years of smoking38. Ethnic variations in AMD lesions have also been reported in previous studies based on CFP14. In the current study, we report that Chinese and Malay ethnicity were associated with higher drusen volume compared to Indian ethnicity. Thus further studies are warranted to further evaluate differences between ethnic groups.
The pathogenesis of AMD is complex and multi-factorial. One of the key mechanisms involves thickening of the RPE-BM complex and deposition of extracellular materials above or below the RPE, which is thought to lead to reduced transport of nutrients and lowered oxygen tension. We hypothesize that increased drusen volume may be observed in eyes affected by this process, which may in turn be related to an individual’s genetic background. The association between OCT-derived drusen measurements with SNPs in eight AMD-associated genes (SYN3, LIPC, ARMS2, C3, CFB, CETP, CFI and CFH) was evaluated in 216 subjects (432 eyes) with intermediate AMD from the Amish population11. In that study, a significant association of the CFH risk allele GG for SNP rs120238333 with drusen area as well as drusen volume and area of RPE atrophy was reported, but none of the other 7 AMD-associated gene SNPS tested were associated with any of the drusen measurements. In the current study, we evaluated the association between drusen volume and four AMD-related SNPS. We did not find an association between CFH rs900292 and drusen volume. However in subjects with AMD, we found that ARMS2 rs10490924_T was associated with increased drusen volume. Studies with larger sample size will be needed to further evaluate the potential association between ARMS2 rs104900292 and drusen volume.
Strengths of the current study include its population-based sample, the adoption of standardized Wisconsin AMD grading methodology for CFP identical to those used in the landmark Beaver Dam and Blue Mountains Eye studies. The use of standardized questionnaires to capture baseline demographic data allowed us to evaluate the influence of a comprehensive list of medical and social risk factors on drusen volume. The availability of genotype data in a large sample size provided a unique opportunity to evaluate the influence of AMD-associated genetic polymorphisms on drusen volume. There are limitations, however, that need to be discussed. The automated drusen volume measurement algorithm used in this study is based on automatic SD-OCT segmentation of the outer border of the RPE layer and compared with the interpolated RPE floor which correspond to a virtual RPE layer in contact with Bruch’s membrane and free of deformation. Segmentation error may occur in cases of mild RPE distortion or low RPE reflectivity. As the minimum cutoff for detection of RPE elevation is 19.5μm, small drusen could be missed. Differences are likely to exist between several custom semi-automated and automated algorithms for drusen measurement. However, the use of a commercially available, FDA-approved automated drusen quantification tool in SD-OCT makes the current approach attractive and broadly applicable in clinical practice. Finally, to account for the highly right-skewed drusen volume distribution, we analyzed the relationship between drusen volume and CFP grading using non-parametric Cuzik’s trend test23,24.
In conclusion, using population-based data with >8,000 eyes, we showed automated SD-OCT measurement of drusen volume using a commercial algorithm (Cirrus OCT; Carl Zeiss Meditec, Inc.) correlated closely with conventional CFP-derived AMD severity and individual AMD features. These two imaging modalities offer unique but complementary information and should be incorporated in future population and clinical studies. Our results suggest automated OCT-derived drusen volume can potentially be used in large scale population studies and big data analyses, such as UK Biobank, to screen for early AMD.
References
Age-Related Eye Disease Study Research, G. A randomized, placebo-controlled, clinical trial of high-dose supplementation with vitamins C and E, beta carotene, and zinc for age-related macular degeneration and vision loss: AREDS report no. 8. Arch Ophthalmol 119, 1417–1436 (2001).
Vitale, S. et al. Evaluating the Validity of the Age-Related Eye Disease Study Grading Scale for Age-Related Macular Degeneration: AREDS2 Report 10. JAMA Ophthalmol 134, 1041–1047, https://doi.org/10.1001/jamaophthalmol.2016.2383 (2016).
Klein, R. et al. The Wisconsin age-related maculopathy grading system. Ophthalmology 98, 1128–1134 (1991).
Wong, W. L. et al. Global prevalence of age-related macular degeneration and disease burden projection for 2020 and 2040: a systematic review and meta-analysis. Lancet Glob Health 2, e106–116, https://doi.org/10.1016/S2214-109X(13)70145-1 (2014).
Schmidt-Erfurth, U., Klimscha, S., Waldstein, S. M. & Bogunovic, H. A view of the current and future role of optical coherence tomography in the management of age-related macular degeneration. Eye (Lond) 31, 26–44, https://doi.org/10.1038/eye.2016.227 (2017).
de Sisternes, L. et al. Individual Drusen Segmentation and Repeatability and Reproducibility of Their Automated Quantification in Optical Coherence Tomography Images. Transl Vis Sci Technol 6, 12, https://doi.org/10.1167/tvst.6.1.12 (2017).
Nittala, M. G., Ruiz-Garcia, H. & Sadda, S. R. Accuracy and reproducibility of automated drusen segmentation in eyes with non-neovascular age-related macular degeneration. Invest Ophthalmol Vis Sci 53, 8319–8324, https://doi.org/10.1167/iovs.12-10582 (2012).
Chen, Q. et al. Automated drusen segmentation and quantification in SD-OCT images. Med Image Anal 17, 1058–1072, https://doi.org/10.1016/j.media.2013.06.003 (2013).
Chiu, S. J. et al. Validated automatic segmentation of AMD pathology including drusen and geographic atrophy in SD-OCT images. Invest Ophthalmol Vis Sci 53, 53–61, https://doi.org/10.1167/iovs.11-7640 (2012).
Diniz, B. et al. Drusen and RPE atrophy automated quantification by optical coherence tomography in an elderly population. Eye (Lond) 29, 272–279, https://doi.org/10.1038/eye.2014.260 (2015).
Chavali, V. R. et al. Association of OCT derived drusen measurements with AMD associated-genotypic SNPs in Amish population. J Clin Med 4, 304–317, https://doi.org/10.3390/jcm4020304 (2015).
Folgar, F. A. et al. Drusen Volume and Retinal Pigment Epithelium Abnormal Thinning Volume Predict 2-Year Progression of Age-Related Macular Degeneration. Ophthalmology 123, 39–50 e31, https://doi.org/10.1016/j.ophtha.2015.09.016 (2016).
Cheung, C. M. & Wong, T. Y. Clinical Relevance and Application of the Age-Related Eye Disease Study Severity Scale for Age-Related Macular Degeneration. JAMA Ophthalmol 134, 1047–1048, https://doi.org/10.1001/jamaophthalmol.2016.2407 (2016).
Joachim, N. et al. Ethnic variation in early age-related macular degeneration lesions between white Australians and Singaporean Asians. Invest Ophthalmol Vis Sci 55, 4421–4429, https://doi.org/10.1167/iovs.14-14476 (2014).
Rosman, M. et al. Singapore Malay Eye Study: rationale and methodology of 6-year follow-up study (SiMES-2). Clin Experiment Ophthalmol 40, 557–568, https://doi.org/10.1111/j.1442-9071.2012.02763.x (2012).
Sabanayagam, C. et al. Singapore Indian Eye Study-2: methodology and impact of migration on systemic and eye outcomes. Clin Exp Ophthalmol, https://doi.org/10.1111/ceo.12974 (2017).
Grading diabetic retinopathy from stereoscopic color fundus photographs–an extension of the modified Airlie House classification. ETDRS report number 10. Early Treatment Diabetic Retinopathy Study Research Group. Ophthalmology 98, 786–806 (1991).
Mitchell, P., Wang, J. J., Foran, S. & Smith, W. Five-year incidence of age-related maculopathy lesions: the Blue Mountains Eye Study. Ophthalmology 109, 1092–1097 (2002).
Cheung, C. M. et al. Prevalence, racial variations, and risk factors of age-related macular degeneration in Singaporean Chinese, Indians, and Malays. Ophthalmology 121, 1598–1603, https://doi.org/10.1016/j.ophtha.2014.02.004 (2014).
Cheung, C. M. G. et al. Six-Year Incidence of Age-Related Macular Degeneration in Asian Malays: The Singapore Malay Eye Study. Ophthalmology, https://doi.org/10.1016/j.ophtha.2017.03.056 (2017).
Tham, Y. C. et al. Aggregate Effects of Intraocular Pressure and Cup-to-Disc Ratio Genetic Variants on Glaucoma in a Multiethnic Asian Population. Ophthalmology 122, 1149–1157, https://doi.org/10.1016/j.ophtha.2015.01.024 (2015).
Cornes, B. K. et al. Identification of four novel variants that influence central corneal thickness in multi-ethnic Asian populations. Hum Mol Genet 21, 437–445, https://doi.org/10.1093/hmg/ddr463 (2012).
Lui, K. J. & Cumberland, W. G. A Wilcoxon-type test for trend. Stat Med 14, 445–446 (1995).
Johnston, S. C., Mendis, S. & Mathers, C. D. Global variation in stroke burden and mortality: estimates from monitoring, surveillance, and modelling. Lancet Neurol 8, 345–354, https://doi.org/10.1016/S1474-4422(09)70023-7 (2009).
Armitage, P. Tests for linear trends in proportions and frequencies. Vol. 11 375–386 (International BiometricSociety, 1955).
Chuang-Stein, C. a. A. A. Tutorial in biostatistics: A review of tests for detecting a monotone dose-response relationship with ordinal response data. Vol. 16 2599–2618 (John Wiley & Sons, Ltd. 1997).
Jain, N. et al. Quantitative comparison of drusen segmented on SD-OCT versus drusen delineated on color fundus photographs. Invest Ophthalmol Vis Sci 51, 4875–4883, https://doi.org/10.1167/iovs.09-4962 (2010).
Freeman, S. R. et al. Optical coherence tomography-raster scanning and manual segmentation in determining drusen volume in age-related macular degeneration. Retina 30, 431–435, https://doi.org/10.1097/IAE.0b013e3181bd2f94 (2010).
Yehoshua, Z. et al. Comparison of drusen area detected by spectral domain optical coherence tomography and color fundus imaging. Invest Ophthalmol Vis Sci 54, 2429–2434, https://doi.org/10.1167/iovs.12-11569 (2013).
Klein, R., Klein, B. E. & Cruickshanks, K. J. The prevalence of age-related maculopathy by geographic region and ethnicity. Prog Retin Eye Res 18, 371–389 (1999).
Mitchell, P., Smith, W., Attebo, K. & Wang, J. J. Prevalence of age-related maculopathy in Australia. The Blue Mountains Eye Study. Ophthalmology 102, 1450–1460 (1995).
Fisher, D. E. et al. Incidence of Age-Related Macular Degeneration in a Multi-Ethnic United States Population: The Multi-Ethnic Study of Atherosclerosis. Ophthalmology 123, 1297–1308, https://doi.org/10.1016/j.ophtha.2015.12.026 (2016).
Goldberg, J., Flowerdew, G., Smith, E., Brody, J. A. & Tso, M. O. Factors associated with age-related macular degeneration. An analysis of data from the first National Health and Nutrition Examination Survey. Am J Epidemiol 128, 700–710 (1988).
Age-Related Eye Disease Study Research, G. A randomized, placebo-controlled, clinical trial of high-dose supplementation with vitamins C and E and beta carotene for age-related cataract and vision loss: AREDS report no. 9. Arch Ophthalmol 119, 1439–1452 (2001).
Liew, G., Joachim, N., Mitchell, P., Burlutsky, G. & Wang, J. J. Validating the AREDS Simplified Severity Scale of Age-Related Macular Degeneration with 5- and 10-Year Incident Data in a Population-Based Sample. Ophthalmology 123, 1874–1878, https://doi.org/10.1016/j.ophtha.2016.05.043 (2016).
Yu, Y. et al. Association of variants in the LIPC and ABCA1 genes with intermediate and large drusen and advanced age-related macular degeneration. Invest Ophthalmol Vis Sci 52, 4663–4670, https://doi.org/10.1167/iovs.10-7070 (2011).
Gregori, G. et al. Spectral domain optical coherence tomography imaging of drusen in nonexudative age-related macular degeneration. Ophthalmology 118, 1373–1379, https://doi.org/10.1016/j.ophtha.2010.11.013 (2011).
Diniz, B. et al. Drusen and RPE atrophy automated quantification by optical coherence tomography in an elderly population. Eye (Lond) 29, 300, https://doi.org/10.1038/eye.2014.307 (2015).
Acknowledgements
We thank Prof Cheung Yin Bun for his advice on statistical analysis. National Medical Research Council grants no. 0796/2003 and Biomedical Research Council Grant no. 501/25-5.
Author information
Authors and Affiliations
Contributions
C.M.G. Cheung (literature review, study design, data analysis, writing of manuscript, decision to submit), Y.S. (data analysis). Y.C. Tham, C. Sabanayagam, K. Neelam (study design, data collection, review of manuscript), J.J. Wang, P. Mitchell, C.Y. Cheng, T.Y. Wong (study design, critical review of manuscript), C.Y.L. Cheung (study design, critical review of manuscript)
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Cheung, C.M.G., Shi, Y., Tham, Y.C. et al. Correlation of Color Fundus Photograph Grading with Risks of Early Age-related Macular Degeneration by using Automated OCT-derived Drusen Measurements. Sci Rep 8, 12937 (2018). https://doi.org/10.1038/s41598-018-31109-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-31109-x
This article is cited by
-
Drusen morphometrics on optical coherence tomography in eyes with age-related macular degeneration and normal aging
Graefe's Archive for Clinical and Experimental Ophthalmology (2023)
-
Hypothetical pathogenesis of age-related macular degeneration and pachychoroid diseases derived from their genetic characteristics
Japanese Journal of Ophthalmology (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.