Abstract
The nature-nurture debate regarding the origin of mental lines is fundamental for cognitive neuroscience. We examined natural-nurture effects on the mental time line, applying three different challenges to the directionality of time representation. We tested (1) patients with left-neglect and healthy participants, who are (2) left-to-right or right-to-left readers/writers, using (3) a lateralized left-right button press or a vocal mode in response to a mental time task, which asks participants to judge whether events have already happened in the past or are still to happen in the future. Using lateralized responses, a spatial-temporal association of response code (STEARC) effect was found, in concordance with the cultural effects. With vocal responses (no lateralization), past and future events showed similar results in both cultures. In patients with neglect, who have a deficit of spatial attention in processing the left side of space, future events were processed more slowly and less accurately than past events in both cultures. Our results indicate the existence of a “natural” disposition to map past and future events along a horizontal mental time line, which is affected by the different ways in which spatial representation of time is introduced.
Similar content being viewed by others
Introduction
Representation of magnitudes plays a major role in our everyday life. Numbers, time and even emotions quantified in our brain, are hypothesized to be represented on “mental lines”1,2,3, spatially relying on a continuous left-to-right orientation axis2,4,5. Studies of numerosity in archaic cultures and in children demonstrate a common origin of the mental number line (MNL), that may be modified as based on cultural effects6,7. A strong evidence for the association between space and numbers is the so-called Spatial-Numerical Association of Response Codes (SNARC) effect8. This effect represents the finding that responses are faster for relatively small numbers while given with the left-hand side and faster for relatively large numbers while given with the right-hand side9. For instance, while participants are required to perform an odd–even classification task on numbers from 1 to 9 by pressing a left or right key, responses are faster and more accurate when participants respond with the left key to small numbers and with the right key to large numbers, compared to the opposite instructions8. This finding supports the existence of a horizontal spatial representation of numbers along the MNL where, in Western cultures, numbers are progressively positioned from left-to-right (L → R), while a reverse SNARC is reported in right-to-left (R → L) readers/writers8,10.
An important contribution to the understanding of the spatial representation of number is provided by neuropsychological studies in brain damaged patients. Such studies demonstrated that patients with a right brain lesion and a contra-lesional visuo-spatial deficit (i.e., left neglect) show a rightward bias in bisecting a number sequence from 0 to 911. This bias was an expression of the deficit in considering the smaller numbers, hypothesized to be represented in the leftmost part of the (horizontal) MNL. However, other studies have found that in patients with right brain damage the ‘rightward’ bias in MNL is not correlated with the attentional bias in visual space12,13,14,15. Specifically, Aiello and colleagues16 found that patients with right hemispheric lesion disregard small numbers both when these are mapped on the left side of the mental number line and on the right side of an imagined clock face. Similar results were found while the right hemisphere was suppressed by electrical stimulation17. The hemispheric specialization explanation for the MNL is in line with studies identifying a MNL-like behavior in neonates or animals, in whom cultural-biased cannot account for the number-space effect18. These findings have led to the conclusion that the right hemisphere supports the representation of small numerical magnitudes independently from their mapping on the left or the right side of the MNL.
Similarly to the MNL, several studies pointed on the spatial nature of time representation that may be regarded as a mental time line (MTL) (e.g.1,19,20,21,22; for reviews5,23,24). Moreover, a temporal equivalent of the SNARC effect, that is the Spatial TEmporal Association of Response Codes (STEARC) effect, has also been reported. For example, facilitation was found between short temporal durations and left-hand responses, and long temporal durations and right-hand responses25,26. A similar effect has also been obtained with temporal concepts: faster responses were found when words with meaning referring to the past were presented on the left and words with meaning referring to the future on the right space27,28. Like in the SNARC effect10,29, the STEARC effect is prompted by L → R reading and writing direction (left/past and right/future), while a reversed pattern (i.e., left/future and right/past facilitation effect) has been found in R → L readers/writers participants (e.g.7,30).
Here, we asked whether the left-right directionality of the MTL is inherent to the cognitive system or, alternatively, whether it emerges only when an explicit horizontal representation is applied. To this aim, we tested participants in a mental time (MT) task, requiring them to determine whether a series of events were located in the past or in the future1,31,32,33. We challenged the habitual STEARC effect using three different modifications of the left-to-right axis: (1) we tested participants who are either L → R or R → L writers and readers; (2) participants responded either with both hands or by vocal response, and (3) we included participants with or without left neglect. These manipulations enabled us to examine the factors that influence the spatial representation of past and future events on the MTL.
Results
Experiment 1. Mental time task using manual responses in healthy participants from L → R and R → L cultures
In the mental time (MT) task, brief descriptions of personal (e.g., car license, first child) or non-personal world events (e.g., Obama’s election, Chernobyl disaster) are presented. For each event, participants were required to indicate whether the event had already happened (past event) or is yet to happen (future event)1,31,32,33. In order to examine the directionality of the mental time line (MTL), we first used here a bimodal experimental settings involving 28 participants who are either L → R or R → L readers/writers responding either for past events with the left hand and for future events with the right hand and vice-versa.
Response times
Analysis of response times (RTs) showed a main effect of Group (L → R vs R → L) [F(1,26) = 7.20, p < 0.05, η2p = 0.22], since R → L participants responded faster than L → R participants (mean ± SD, 1188 ± 555 vs 1620 ± 357 msec). Relevantly, the interaction Group (L → R vs R → L) × Laterality (past/left and future/right vs past/right and future/left) was also significant [F(1,26) = 12.51, p < 0.01, η2p = 0.33] (Fig. 1). Duncan post-hoc tests revealed that, in the L → R group, responses were faster in the past/left and future/right condition (i.e., in the condition congruent with L → R culture) with respect to the opposite condition (1476 ± 341 vs 1764 ± 317 msec, p < 0.001; Fig. 1, left). In the R → L group the other way around was found with participants faster for a condition congruent with the R → L culture, though this difference between responses provided in the past/left and future/right and past/right and future/left conditions did not reach significance (1242 ± 613 vs 1134 ± 497 msec, p = 0.18; Fig. 1, right). Moreover, comparing the two groups, the R → L group was faster than the L → R one (1134 ± 497 vs 1764 ± 317 msec, p < 0.01) in responding to the past/right and future/left condition (i.e., in the condition congruent with R → L culture).
Experiment 1. Significant interaction between Group (L → R or R → L readers/writes) and Laterality (past/future-left/right) in lateralized responses. In the L → R culture group, responses were faster in the past/left-future/right condition while in the R → L group the other way around was found. Values are indicated in milliseconds (msec); error bars depict standard errors of the mean (SEM); asterisks indicate significant differences (p < 0.05).
Error rates
The main effect of Group (L → R vs R → L) [F(1,26) = 7.55, p < 0.05, η2p = 0.23] was significant: R → L participants made more errors than L → R participants (9 vs 4%).
Results of Experiment 1 show that a spatial factor – reading/writing direction – influences judgments related to past and future events, when the MT-task required an explicit spatial coding through left/right lateralized responses. Such space/time association emerged and a facilitation was found when L → R participants responded with the left hand to the past and the right hand to the future, and vice versa for R → L participants, though the latter did not reach significance, hinting on an additional factor in this experimental group (see Discussion for possible interpretations). The present data thus support a culturally-influenced organization of the MTL, where past and future are represented from left-to-right in L → R readers and writers, and vice versa in R → L ones.
Two hypotheses may explain these results. First, there is an intrinsic (“natural”) organization of the MTL (past in the left and future in the right), as it is found for other magnitudes in very young children18 and even in animals34 (or vice versa). Second, cultural factors influence the directionality of the effects. To allow the spatial representation of time to emerge, in Experiment 1, spatiality was introduced through motor response. In Experiment 2, we tested patients with left-neglect, impaired in the spatial processing of the left visual field35,36,37,38,39,40. Therefore, on the one hand we removed the spatial dimension from the MT-task (using vocal instead of motor response), on the other hand we introduced space in the group of neglect patients due to their spatial deficit. Following a right brain lesion, neglect patients show a disorder characterized by an unbalanced distribution of attention in space, with a deficit in orienting attention to the contralesional space and an attentional bias for the ipsilesional space. Based on this view, two opposite results can be predicted. According with the deficit in orienting attention towards stimuli in contralesional space41, a significant impairment in representing events associated with the left portion of space is expected (i.e., slower response times and higher error rate to past events, assuming a L → R MTL). Instead, if neglect reflects an ipsilesional attentional bias, consisting of enhanced attention to the right42, patients should spend more time in exploring the right space where future events are located (i.e., slower response times and higher error rate to future events, assuming a L → R MTL). If natural effects are prominent compared to cultural ones, the above reported scenarios should affect both L → R and R → L readers/writers neglect patients in the same way while vocal responses are given. If the reading/writing directionality plays instead a key role, left neglect will affect the performance of L → R and R → L readers/writers differently.
Experiment 2. Mental time task using vocal responses in healthy participants and patients with left neglect from L → R and R → L cultures
To distinguish between these two hypotheses, in Experiment 2 participants were required to give vocal (i.e., non-lateralized) responses to past/future events. Moreover, we verified whether, without a lateralized motor response, the spatial encoding of past and future events emerged when an attentional spatial deficit is present, as happens following a brain lesion in neglect patients, and how this interacts with cultural factors. To this aim, in Experiment 2, we examined 7 left-neglect patients (NP) and 7 healthy controls (HC) who are solely L → R readers and writers, and 7 NP and 7 HC who are solely R → L readers/writers. In addition to the audio version of the MT-task, we performed lesion analysis to ensure homogeneity between the two patients groups.
Lesion Analysis
The lesion overlay of R → L patients revealed three primary regions of overlap: in the right putamen and claustrum (x = 53, y = 126, z = 77) appearing in four out of the seven patients, at the right insula (x = 59, y = 125, z = 89) appearing in five patients, and in the right caudate (x = 75, y = 115, z = 93) appearing in four patients, such that every R → L patient exhibited a lesion in at least one of these three foci (Fig. 2A). The lesion overlay of the L → R patients revealed two primary foci of convergence: five out of the seven patients exhibited a lesion in the right posterior limb of the internal capsule (x = 68, y = 115, z = 77) and four patients exhibited a lesion in the right middle temporal gyrus (x = 29, y = 104, z = 71), such that every L → R patient exhibited a lesion in at least one of these two foci (Fig. 2B). Taken together, underlying lesions in most patients from both R → L and L → R cultures were located in the right basal ganglia, as well as the right temporal/insular cortex. The spatial distribution of lesions is within the normal range for neglect patients, since the most frequent sites of damage in the right hemisphere involve temporal cortex and subcortical nuclei, along with inferior parietal lobule and ventral frontal region43,44.
Lesion analysis. The spatial distribution of ischemic lesions (transformed into standard MNI stereotaxic space) in all R → L (A) and L → R (B) patients is demonstrated by overlaying the lesion masks onto a T1-weighted atlas. Voxels are color-coded in accordance with the number of patients with a lesion affecting a specific voxel. Arrowheads are directed at foci of lesion map convergences (x, y, z coordinates denoted in brackets).
Behavioural Analysis
Response times
The main effects of Group (L → R HC, R → L HC, L → R NP, and R → L NP) [F(3,24) = 8.79, p < 0.001, η2p = 0.49] and Time (past vs future) [F(1,24) = 44.64, p < 0.001, η2p = 0.65] were significant, as well as their interaction [F(3,24) = 9.27, p < 0.001, η2p = 0.54] (Fig. 3). Both L → R and R → L control groups did not show a significant difference between responses to past and future events (L → R HC 1286 ± 306 vs 1292 ± 301 msec, p = 0.94; R → L HC 1389 ± 157 vs 1514 ± 173 msec, p = 0.16). Conversely, both neglect patients groups were significantly slower in responding to future than to past events (L → R NP 2680 ± 1022 vs 2181 ± 1013 msec, p < 0.001; R → L NP 3066 ± 868 vs 2529 ± 708 msec, p < 0.001). Overall, R → L NPs responded slower than respective HC in both past and future events (p < 0.05 for both comparisons), and L → R NPs were slower than respective HC in particular when considering future events (p < 0.05).
Experiment 2. Significant interaction between Group (L → R or R → L readers/writes) and Time (past/future) in non-lateralized responses. Similar results were found in both the L → R and R → L culture groups: responses were faster in healthy controls (HCs) than in neglect patients (NPs). Note the similarity of response times between the two groups, as in NPs in both cultures future responses were slower than past ones. Values are indicated in milliseconds (msec); error bars depict standard errors of the mean (SEM); asterisks indicate significant differences (p < 0.05).
Error rates
Overall, error rates were slightly higher for future than for past events (6% vs 4%), though main effect of Time (past and future) did not reach significance [F(1,24) = 3.30, p < 0.08] and the interaction Group × Time was not significant (p = 0.16). Means showed that both patients groups made more errors for future than past responses (L → R NPs 10% vs 5%; R → L NPs 10% vs 6%) whereas this was not the case for healthy controls (L → R HCs 1% vs 2%; R → L HCs 1% vs 1%).
Crawford’s test
The previous analyses revealed that both neglect patients groups were significantly slower in responding to future than to past events. We conducted an additional analysis to verify whether this effect was limited to a few patients or, alternatively, whether it emerged in most of our patients. To this aim, we used the Crawford’s test45, a modified t-test, to compare the RTs of each NP with respective HC. The mean values suggest that this effect was present in all patients and the Crawford’s test was significant in 5 out of 7 L → R NP and in 6 out of 7 R → L NP (p < 0.05).
Overall, our data highlight the role played by spatial factors in the emergence of spatio-temporal association. The STEARC effect emerges only when a spatial factor is involved, such as when responses are given in a lateralized manner (Experiment 1) or when an attentional spatial bias is present, as happens in left neglect patients (Experiment 2). These findings are discussed below in the framework of attentional influence on the MTL.
Discussion
In the present study, we tested factors that influence the MTL and its directionality, requiring participants to determine whether a series of events were located in the past or the future. Three different factors were checked: response modality (motor vs verbal), cultural influence (L → R vs R → L reading/writing direction), and spatial attention (participants with vs without left neglect). In healthy participants, we found a spatial-temporal association of response codes (STEARC) effect in concordance with reading/writing direction when responses were given in a lateralized manner (motor MT-task). In left-neglect, the left-right axis is interrupted following a spatial attention deficit, and a significant difficulty for future processing has been found, in terms of both slower response times and higher error rate, in line with previous studies46. Notably, similar results in processing past and future events in healthy participants elicited by Experiment 2 (vocal MT-task) speak against the possibility that uncontrolled factors could make future events intrinsically more difficult than past events.
In Experiment 1, healthy participants showed facilitation in responding to past stimuli with the left key and to future ones with the right key in L → R culture (STEARC effect). The opposite pattern (facilitation for future stimuli while responded with the left key and to past ones with the right key) was found in R → L culture. These results are in line with the interpretation of the STEARC effect as related to a conflict emerging at the stage of response selection (e.g.26), in analogy with similar effects found in other domains, like sound pitches (SMARC effect47) or numbers (e.g., SNARC effect8). For example, regarding the SNARC effect, and in accordance with findings by Gevers and colleagues48, this conflict emerges when two alternative response codes are possible: one automatically triggered through pre-existing associations linking a stimulus to its response, and the other generated on the basis of task instructions (dual-route account; e.g.49). If both routes converge on the same response, a response can be activated fast; conversely, if the two routes elicit different responses, the automatic response should be inhibited before the correct response can be given.
According to these theories, the effect found here can be explained by a conflict between responses elicited by a culture-dependent time representation and task-related responses. Indeed, facilitation was registered when L → R participants responded with the left hand to the past and the right hand to the future, and vice versa for R → L participants. However, this latter effect did not reach significance probably because R → L readers/writers participants in Experiment 1 were also exposed to the opposed direction of language and mathematical schemes (see also30,50,51). Note that “hybrid” reading/writing habits for text and numbers is known to decrease the SNARC effect10,52 as may be the case also for time53. Alternatively, weaker effect in R → L participants may suggest that there is an intrinsic (“natural”) organization of the MTL (past in the left and future in the right), that can be influenced but not overwritten by cultural habits. This directionality from left to right of past and future events is in line with previous studies (for review see5).
In Experiment 2, when a lateralized response was not required and a verbal response was given, past and future events were processed similarly by healthy participants. Space-time association was found by several studies25,26,27,33,54,55,56,57. More in detail, such a space-time association is present when responses are given in a lateralized manner by a manual response code (Experiment 1 reported here), when there is a disruption of lateralization as occurs following left neglect (Experiment 2 reported here, see also46), and when a lateralization is induced by shifting visuo-spatial attention through prismatic adaptation33. In this latter study, we showed that prism-induced manipulations of spatial attention influenced processing of past and future concepts. In particular, we obtained a facilitation in responding to past/future events according to the direction of the attentional shift, by using a verbal/non-lateralized response (as in Experiment 2 reported here): leftward and rightward shift of spatial attention facilitated indication of past and future events, respectively. Thus, our data highlight how spatial attention prioritizes processing of particular locations in time, which failed to emerge before prismatic modulation, as shown here in Experiment 2.
In order to eliminate the hand-related effect (i.e., removing space from the MT-task), we used here audio presentation of stimuli and vocal response modes, which have proven efficient in our previous studies33,46. Notably, in Experiment 2 the lack of effects in healthy participants is due to the experimental manipulation that, through acoustical binaural presentation and vocal responses, made space not a dimension of the task. Similarly, Magnani and colleagues58 found that a spatial representation of auditory time (i.e., in a task where participants classified the duration of auditory stimuli as “short” or “long”) emerges selectively when space becomes a relevant dimension, enforcing a spatial-encoding of auditory stimuli. More specifically, in their experiment an underestimation of the duration of auditory stimuli presented on the left, relative to that presented on the right, was found only when participants were first required to localize stimuli. Taken together, the current as well as previous results point on the emergence of a space-time association (i.e., a horizontal spatial representation of time) under certain conditions requiring lateralization.
Regarding the alteration of the MTL following a spatial attention impairment in patients with left-neglect, we found longer response times and higher error rates for future events in patients from both L → R and R → L cultures. Several explanations may account for these findings. The first explanation assumes a MTL oriented from right-to-left. Both patients’ group with left neglect (L → R and R → L) faced difficulty in processing future events, suggesting the future to lie in the disturbed side, namely the left. However, this hypothesis is not in-line with previous studies suggesting a L → R MTL5,39,40, though most of these studies adopted an allocentric point of view while the current study adopts an egocentric one. The difference in significance between L → R and R → L participants in Experiment 1 also suggests a L → R MTL.
An alternative interpretation of our results can be provided by the hemispheric-specialization theory proposed in the domain of number representation. According to this theory, the right hemisphere represents small numerical magnitudes and the left hemisphere represents large magnitudes, independently from their mapping on the left or the right side of the mental number line16,34. Following an analogous mental time and mental number lines, future impairment in our patients implies that the right hemisphere accounts for future processing and the left hemisphere for the past.
A third explanation is related to the potential character of the future vs real events in the past. Future events, as used here, can be regarded as potential events which might never happen, while past events can be labelled as factual events which have surely occurred in the past. Thus, our MT-task may require participants to judge whether something is factual or potential. The difficulty of neglect patients in processing future events can be in line with a recent study revealing that participants were faster in responding to potential events with the left hand and to factual events with the right hand, than with the opposite mapping59. The dimension of factuality-potentiality may therefore be also mapped onto space, similarly to time and number, with a left-potential and right-factual mapping. This may lead to impairment in potential-events processing in our patients.
A final explanation refers to the hypothesis that neglect reflects a rightward attentional bias42: neglect patients would spend more time in exploring the right space than left space33. Assuming a L → R MTL, longer and pathological exploration of the right-space would lead to slower response times to future events46. Though longer exploration may reduce errors, higher error rate found here may be related to false recognitions.
In conclusion, our results suggest that spatial components may interfere with the processing of time. This interference may be further influenced by other factors such as reading/writing direction. Additional research may explore such hierarchical nature-nurture influences on the MTL and other mental lines, and provide theoretical and experimental frameworks for their potential interpretations.
Methods
Experiment 1
Participants
28 right-handed healthy participants (mean age ± SD: 62 ± 6 years old) took part in Experiment 1, half of them L → R readers/writers (Italian speakers) and half R → L readers/writers (Hebrew speakers living in Israel who were also exposed to L → R language and mathematical schemes). Participants did not show any cognitive impairment as measured by the Mini Mental State Examination (MMSE score ≥29)60. All participants were right-handed, had normal or corrected-to-normal vision, and no history of psychiatric diseases. Participants involved in all reported experiments were naive to the purpose of the study and provided written informed consent. All the reported experiments were carried out in accordance with the 2008 Helsinki Declaration and approved by the Ethical Committees of Hadassah Medical Center and the University of Bologna.
Stimuli and procedure
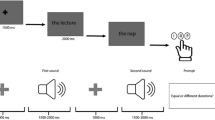
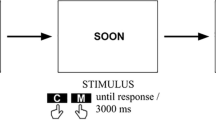
During the MT-task, participants sat in front of a 15-inch color monitor, at a distance of about 60 cm. Brief descriptions of personal (e.g., car license, first child) or non-personal world events (e.g., Obama’s election, Chernobyl disaster) were presented on the computer screen (similar to1,31,32,33), including 24 stimuli (half personal and half non-personal), equally distributed between past and future events (Fig. 4; for a complete list of stimuli, see Table 1). For each event, participants were required to indicate whether the event had already happened (past event) or was yet to happen (future event).
Each stimulus appeared in the center of the computer screen and remained visible until a response was given, with an inter-stimulus interval of 1000 msec. The experiment had two blocks, differing in the mapping of the left and right keys to “past” or “future” judgments (i.e., mapping 1 past events/left key and future events/right key; mapping 2 past events/right key and future events/left key; motor MT-task). The order of blocks was counterbalanced over participants (within-participants design). E-Prime 2.0 software was used for stimulus presentation and response collection.
Statistical analysis
Data on response times (RTs) and error rates (ERs) were entered in separate repeated-measures analysis of variance (ANOVA) with Time (past vs future) and Laterality (past/left and future/right vs past/right and future/left) as within-subject factors, and Group (R → L vs L → R) as between-subject factor. Duncan post-hoc tests were performed on significant interactions and the magnitude of effect size was expressed by η2p.
Data availability
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
Experiment 2
Participants
7 left-neglect patients (NP) and 7 healthy controls (HC) who are solely L → R readers and writers (Italian speakers), and 7 NP and 7 HC who are solely R → L readers/writers (Hebrew or Arabic speakers) participated in Experiment 2 (mean age ± SD: 66 ± 7.84 years old, for neuropsychological details see Table 2). All patients were recruited for the experiment within 6 months from the onset of illness. The day before the experimental session, all patients were submitted to Mini Mental State Examination (MMSE) and a battery of tests assessing the presence of neglect (Behavioral Inattention Test Battery – conventional part, star cancellation, and line bisection) assuring that all patients had an active left-neglect at the time of testing (Table 2). Healthy participants and patients did not show significant cognitive impairment. L → R patients with a right brain lesion and without neglect have also been tested and showed the same pattern of results of controls [46; see also Supplementary Information], excluding that the difference between neglect patients and controls can be explained by a brain lesion per se or by differences in past and future events related to uncontrolled effects.
Stimuli and procedure
The stimuli and procedure were the same as outlined in Experiment 1, except that here participants were presented with audio stimuli and provided a vocal response (“past” vs. “future”; verbal MT-task). This response modality removes spatialization of stimulus’ representation (for the same procedure, see33). Patients with neglect were not tested using motor responses due to left hand weakness.
Lesion analysis
Lesion tracing was completed using MRIcron software (http://people.cas.sc.edu/rorden/mricron/index.html). Lesions were visually identified as having altered signal intensity in relation to equivalent contralateral tissue. Lesions were traced by a trained image analyst and confirmed by an experienced neurologist. Brain images and lesion tracings were normalized to standard stereotaxic space (MNI space), using an extension of the Statistical Parametric Mapping (SPM) software, named “Clinical Toolbox”, that transforms a lesion image to a dilated lesion mask, applies it to its corresponding brain image to ensure normalization is driven by intact brain tissue61,62, and outputs a normalized binary lesion map as well as the normalized CT/MRI image. Lesion maps were superimposed, and the extant of overlap between patients was estimated and projected onto a T1 template MNI image.
Behavioural analysis
Data on RTs and ERs were entered in separate repeated-measures ANOVAs with Time (past vs future) as within-subject factor, and Group (L → R HC, R → L HC, L → R NP, and R → L NP) as between-subject factor. Duncan post-hoc tests were performed on significant interactions and the magnitude of effect size was expressed by η2p.
References
Arzy, S., Adi-Japha, E. & Blanke, O. The mental time line: an analogue of the mental number line in the mapping of life events. Consciousness and Cognition 18, 781–785 (2009).
Dehaene, S. The Number Sense. Oxford: University Press (2011).
Holmes, K. J. & Lourenco, S. F. Common spatial organization of number and emotional expression: A mental magnitude line. Brain Cognition 77, 315–323 (2011).
Hubbard, E. M., Piazza, M., Pinel, P. & Dehaene, S. Interactions between number and space in parietal cortex. Nature Review Neuroscience 6, 435–448 (2005).
Bonato, M., Zorzi, M. & Umiltà, C. When time is space: evidence for a mental time line. Neuroscience and Biobehavioral Reviews 36, 2257–2273 (2012).
Dehaene, S., Izard, V., Spelke, E. & Pica, P. Log or linear? Distinct intuitions of the number scale in Western and Amazonian indigene cultures. Science 320, 1217–1220 (2008).
Fuhrman, O. et al. How Linguistic and Cultural Forces Shape Conceptions of Time: English and MandarTime 3D. Cognitive Science 35, 1305–1328 (2011).
Dehaene, S., Bossini, S. & Giraux, P. The mental representation of parity and number magnitude. Journal of Experimental Psychology: General 122, 371–396 (1993).
Dehaene, S., Dupoux, E. & Mehler, J. Is numerical comparison digital? Analogical and symbolic effects in two-digit number comparison. Journal of Experimental Psychology: Human Perception and Performance 16, 626–641 (1990).
Shaki, S., Fischer, M. H. & Petrusic, W. M. Reading habits for both words and numbers contribute to the SNARC effect. Psychonomic Bulletin and Review 16, 328–331 (2009).
Zorzi, M., Priftis, K. & Umiltà, C. Neglect disrupts the mental number line. Nature 417, 138–139 (2002).
Rossetti, Y. et al. Does action make the link between number and space representation? Visuo-manual adaptation improves number bisection in unilateral neglect. Psychological Science 15, 426–430 (2004).
Doricchi, F., Guariglia, P., Gasparini, M. & Tomaiuolo, F. Dissociation between physical and mental number line bisection in right hemisphere brain damage. Nature Neuroscience 8, 1663–1665 (2005).
Doricchi, F. et al. Spatial orienting biases in the decimal numeral system. Current Biology 19, 682–687 (2009).
Loetscher, T. & Brugger, P. Random number generation in neglect patients reveals enhanced response stereotypy, but no neglect in number space. Neuropsychologia 47, 276–279 (2009).
Aiello, M. et al. No inherent left and right side in human “mental number line”: evidence from right brain damage. Brain 135, 2492–2505 (2012).
Arshad, Q. et al. Bidirectional Modulation of Numerical Magnitude. Cerebral Cortex 26, 2311–2324 (2016).
de Hevia, M. D., Izard, V., Coubart, A., Spelke, E. S. & Streri, A. Representations of space, time, and number in neonates. Proceedings of the National Academy of Sciences 111, 4809–4813 (2014).
Lakoff, G. & Johnson, M. Philosophy in the flesh: The embodied mind and its challenge to western thought. New York: Basic Books (1999).
Casasanto, D. & Boroditsky, L. Time in the mind: using space to think about time. Cognition 106, 579–593 (2008).
Bender, A. & Beller, S. Mapping spatial frames of reference onto time: A review of theoretical accounts and empirical findings. Cognition 132, 342–382 (2014).
Anelli, F., Candini, M., Cappelletti, M., Oliveri, M. & Frassinetti, F. The remapping of time by active tool-use. Plos One 10(12), art. 0146175 (2015).
Núñez, R. & Cooperrider, K. The tangle of space and time in human cognition. Trends in Cognitive Sciences 17, 220–229 (2013).
Winter, B., Marghetis, T. & Matlock, T. Of magnitudes and metaphors: Explaining cognitive interactions between space, time, and number. Cortex 64, 209–224 (2015).
Ishihara, M., Keller, P. E., Rossetti, Y. & Prinz, W. Horizontal spatial representations of time: evidence for the STEARC effect. Cortex 44(4), 454–461 (2008).
Vallesi, A., Binns, M. A. & Shallice, T. An effect of spatial–temporal association of response codes: understanding the cognitive representations of time. Cognition 107, 501–527 (2008).
Torralbo, A., Santiago, J. & Lupiánez, J. Flexible conceptual projection of time onto spatial frames of reference. Cognitive Sciences 30, 745–757 (2006).
Santiago, J., Lupianez, J., Perez, E. & Funes, M. J. Time (also) flies from left to right. Psychonomic Bullettin and Review 14, 512–516 (2007).
Shaki, S., Fischer, M. H. & Göbel, S. M. Direction counts: A comparative study of spatially directional counting biases in cultures with different reading directions. Journal of Experimental Child Psychology 112, 275–281 (2012).
Ouellet, M., Santiago, J., Israeli, Z. & Gabay, S. Is the future the right time? Experimental Psychology 57, 308–314 (2010).
Arzy, S., Molnar-Szakacs, I. & Blanke, O. Self in time: imagined self-location influences neural activity related to mental time travel. Journal of Neuroscience 28, 6502–6507 (2008).
Anelli, F., Ciaramelli, E., Arzy, S. & Frassinetti, F. Age-related effects on future mental time travel. Neural Plasticity art. 1867270 (2016a).
Anelli, F., Ciaramelli, E., Arzy, S. & Frassinetti, F. Prisms to travel in time: investigation of time-space association through prismatic adaptation effect on mental time travel. Cognition 156, 1–5 (2016b).
Rugani, R., Vallortigara, G., Priftis, K. & Regolin, L. Number-space mapping in the newborn chick resembles humans’mental number line. Science 347, 534–536 (2015).
Basso, G., Nichelli, P., Frassinetti, F. & di Pellegrino, G. Time perception in a neglected space. NeuroReport 7(13), 2111–2114 (1996).
Danckert, J. et al. Neglected time: Impaired temporal perception of multiseconds intervals in unilateral neglect. Journal of Cognitive Neuroscience 19(10), 1706–1720 (2007).
Magnani, B., Oliveri, M., Mancuso, G., Galante, E. & Frassinetti, F. Time and spatial attention: Effects of prism adaptation on temporal deficits in brain damaged patients. Neuropsychologia 49(5), 1016–1023 (2011).
Oliveri, M., Magnani, B., Filipelli, A., Avanzi, S. & Frassinetti, F. Prismatic adaptation effects on spatial representation of time in neglect patients. Cortex 49(1), 120–130 (2013).
Saj, A., Fuhrman, O., Vuilleumier, P. & Boroditsky, L. Patients With Left Spatial Neglect Also Neglect the “ Left Side” of Time. Psychological Science 25, 207–214 (2014).
Bonato, M., Saj, A. & Vuilleumier, P. Hemispatial Neglect Shows That (Before) Is (Left). Neural Plasticity, art. 2716036 (2016).
Heilman, K. M. & Valenstein, E. Mechanisms underlying hemispatial neglect. Annual Neurology 5, 166–170 (1979).
Bartolomeo, P. & Chokron, S. Orienting of attention in left unilateral neglect. Neuroscience Biobehavioral Review 26, 217–234 (2002).
Corbetta, M., Kincade, M., Lewis, C., Snyder, A. Z. & Sapir, A. Neural basis and recovery of spatial attention deficits in spatial neglect. Nature Neuroscience 8, 1603–1610 (2005).
Corbetta, M. & Shulman, G. L. Spatial neglect and attention networks. Annual Review of Neuroscience 34, 569–599 (2011).
Crawford, J. R., Garthwaite, P. H. & Porter, S. Point and interval estimates of effect sizes for the case-controls design in neuropsychology: rationale, methods, implementations, and proposed reporting standards. Cognitive Neuropsychology 27, 245–260 (2010).
Anelli, F., Avanzi, S., Arzy, S., Mancuso, M. & Frassinetti, F. Effects of spatial attention on mental time travel in patients with neglect. Cortex 101, 192–205 (2018).
Rusconi, E., Kwan, B., Giordano, B. L., Umiltà, C. & Butterworth, B. Spatial representation of pitch height: The SMARC effect. Cognition 99(2), 113–129 (2006).
Gevers, W., Lammertyn, J., Notebaert, W., Verguts, T. & Fias, W. Automatic response activation of implicit spatial information: Evidence from the SNARC effect. Acta Psychologica 122, 221–233 (2006).
De Jong, R., Liang, C.-C. & Lauber, E. Conditional and unconditional automaticity: A dual-process model of effects of spatial stimulus-response correspondence. Journal of Experimental Psychology: Human Perception and Performance 20, 731–750 (1994).
Nachshon, I. Cross-cultural difference in directionality. International Journal of Psychology 16, 199–211 (1981).
Nachshon, I. Directional preferences of bilingual children. Perceptual and Motor Skills 56, 747–750 (1983).
Shaki, S. & Fischer, M. H. Multiple spatial mappings in numerical cognition. Journal of Experimental Psychology: Human Perception and Performance 38, 804–809 (2012).
Vallesi, A., Weisblatt, Y., Semenza, C. & Shaki, S. Cultural modulations of space–time compatibility effects. Psychonomic Bulletin & Review 21, 666–669 (2014).
Ulrich, R. & Maienborn, C. Left–right coding of past and future in language: The mental timeline during sentence processing. Cognition 117, 126–138 (2010).
Flumini, A. & Santiago, J. Time (also) flies from left to right… if it is needed! In Knauff, M., Pauen, M., Sebanz, N. & Wachmuz, I. (Eds), Proceedings of the 35th Annual Conference of the Cognitive Science Society (pp. 2315–2320). Austin, TX: Cognitive Science Society (2013).
Ulrich, R. et al. With the past behind and the future ahead: Back-to-front representation of past and future sentences. Memory & Cognition 40(3), 483–495 (2012).
Macnamara, A., Keage, H. A. & Loetscher, T. Mapping of non-numerical domain on space: a systematic review and meta-analysis. Experimental Brain Research 236, 335–346 (2018).
Magnani, B., Pavani, F. & Frassinetti, F. Changing auditory time with prismatic goggles. Cognition 125, 233–243 (2012).
Aguirre, R. & Santiago, J. Do potential past and future events activate the left-right mental timeline? Psicologica 38, 231–255 (2017).
Folstein, M. F., Folstein, S. E. & McHugh, P. R. Mini-Mental State. A practical method for grading the cognitive state of patients for the clinician. Journal of Psychiatric Research 12(3), 189–198 (1975).
Brett, M., Leff, A. P., Rorden, C. & Ashburner, J. Spatial normalization of brain images with focal lesions using cost function masking. NeuroImage 14(2), 486–500 (2001).
Rorden, C., Bonilha, L., Fridriksson, J., Bender, B. & Karnath, H. O. Age-specific CT and MRI templates for spatial normalization. NeuroImage 61, 957–965 (2012).
Acknowledgements
The authors gratefully acknowledge Prof. Anna M. Borghi and two anonymous reviewers for their helpful comments on a previous version of this article. This work was supported by RFO (Ricerca Fondamentale Orientata, Ministry of University and Research), by a grant from Ministero della Salute (Ricerca Finalizzata PE Estero Theory-enhancing, Grant no. PE-2016-02362477, URL Ministero della Salute: http://www.salute.gov.it/portale/ministro/p4_10_1_1_atti_1_1.jsp?lingua=italiano&id=135), and by the Israeli Science Foundation (ISF, Grant no. 315/16)) and the Agnes Ginges center of Neurogenetics - Orion Foundation. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
F.A., F.F. and S.A. developed the study concept and contributed to the study design. F.A., G.P.F., Y.S., E.M., C.F. and S.A. recruited participants. F.A., G.P.F. and Y.S. carried out testing and data collection. F.A. performed data analyses. G.P.F. performed the lesion analysis. F.A., F.F. and S.A. drafted the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Anelli, F., Peters-Founshtein, G., Shreibman, Y. et al. Nature and nurture effects on the spatiality of the mental time line. Sci Rep 8, 11710 (2018). https://doi.org/10.1038/s41598-018-29584-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-29584-3
This article is cited by
-
Horizontal mapping of time-related words in first and second language
Scientific Reports (2024)
-
The categorical use of a continuous time representation
Psychological Research (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.