Abstract
Diabetes mellitus is a group of disorders characterized by prolonged high levels of circulating blood glucose. Type 1 diabetes is caused by decreased insulin production in the pancreas whereas type 2 diabetes may develop due to obesity and lack of exercise; it begins with insulin resistance whereby cells fail to respond properly to insulin and it may also progress to decreased insulin levels. The brain is an important target for insulin, and there is great interest in understanding how diabetes affects the brain. In addition to the direct effects of insulin on the brain, diabetes may also impact the brain through modulation of the inflammatory system. Here we investigate how perturbation of circulating insulin levels affects the expression of Hes3, a transcription factor expressed in neural stem and progenitor cells that is involved in tissue regeneration. Our data show that streptozotocin-induced β-cell damage, high fat diet, as well as metformin, a common type 2 diabetes medication, regulate Hes3 levels in the brain. This work suggests that Hes3 is a valuable biomarker helping to monitor the state of endogenous neural stem and progenitor cells in the context of diabetes mellitus.
Similar content being viewed by others
Introduction
There is concern that diabetes and, more generally, aberrant insulin regulation, have a negative impact on brain function1. In fact, even common medication for diabetes such as metformin may impact the brain in ways that are not yet well understood as metformin has been shown to alter the self-renewal and differentiation properties of neural stem cells in vitro and in vivo2,3. It is, therefore, important to identify molecular mechanisms by which diabetes mellitus may affect the brain, either directly via alterations in the levels of circulating glucose and insulin or indirectly through effects on the immune/inflammatory system. Here we investigated how streptozotocin-induced β-cell damage, high fat diet, and metformin administration regulate the expression of Hairy and Enhancer of Split 3 (Hes3) in the adult mouse brain.
We focused on Hes3 because (a) we previously showed that it is involved in various paradigms of brain damage and regeneration4,5,6,7, (b) it is also expressed in neural stem cells4, and (c) Hes3 is regulated by insulin4,5,6. Hes3 belongs to the Hes superfamily of basic helix-loop-helix (bHLH) transcription factors that include the Hes and Hey (Hes-related with YRPW motif) members8,9,10. Hes1 and Hes5 are direct targets of Notch signaling and their expression is often used as an indicator of canonical Notch signaling activity10. In contrast, Hes3 is an indirect target of Notch signaling; following Notch receptor activation, a pathway involving phosphatidylinositol-4, 5-bisphosphate 3 (PI3) kinase, Protein kinase B (Akt), mechanistic target of rapamycin (mTOR), and Signal transducer and activator of transcription 3 - Serine (STAT3-Ser) phosphorylation leads to Hes3 expression, which can be used as an indicator of the activity of this non-canonical Notch signaling branch4.
Hes3 is of interest because emerging data shows that it is an important regulator of regeneration in both the pancreas and brain. In cultured mouse insulinoma cells (MIN6), Hes3 knockdown and overexpression studies revealed that Hes3 regulates the expression of pancreatic and duodenal homeobox 1 (Pdx1), an important gene in pancreatic islet health and insulin production; it also regulates the expression of insulin itself11. Hes3 null mice are more sensitive to pancreatic islet damage by the toxin streptozotocin (STZ; used to model type 1 diabetes), compared to wild type (WT) mice, and regenerate beta cell mass less efficiently11,12. In the brain, Hes3 is expressed in putative neural stem cells (NSCs) and progenitor cells4,6. Cultured NSCs also express Hes3; expression is lost following their differentiation13,14,15. Various pharmacological treatments that induce Hes3 expression promote cell survival in culture and the number of Hes3-expressing cells in vivo4,5,6,13,14,15,16,17. This is followed by powerful neuroprotection and disease modification in models of ischemic stroke and Parkinson’s disease4,5,6. Hes3 null mice exhibit lower levels of myelin basic protein (MBP) in the brain, indicating insufficient numbers of oligodendrocytes or reduced myelination7. In summary, Hes3 plays important roles in various tissues and organs, including the brain, where it protects them from damage and enables them to regenerate efficiently.
In this work we demonstrate, for the first time, that the expression of Hes3 in the brain is regulated in mice subjected to streptozotocin-induced β-cell damage, high fat diet, and metformin administration. We establish Hes3 as a biomarker to monitor the brain in animal models that are widely used to study various aspects of diabetes mellitus. Future studies will address whether Hes3 is also regulated in diabetes patients, which parameters of insulin deregulation and/or diabetes mellitus are primarily responsible for Hes3 regulation, and the roles that Hes3 plays in the progression of diabetes-related phenotypes.
Results
Streptozotocin-induced β–cell damage and high fat diet regulate Hes3 expression in the brain
As described in the introduction, we hypothesized that brain Hes3 expression would be altered in mouse models of diabetes, in which insulin signaling is perturbed. Such a result would provide novel information, at the molecular level, of how such perturbations might be affecting the brain. We used streptozotocin (STZ) to induce insulin deficiency. STZ-induced β–cell damage is an established model to study type 1 diabetes in rodents. High dose STZ induces hyperglycemia and leads to insulin deficiency resulting from selective β-cell damage in the pancreas18,19. Furthermore, we studied mice fed a high-fat diet (HFD) as there is a general agreement that feeding a high calorie diet results in impaired glucose homeostasis and at least a pre-diabetic state comprising hyperglycemia, hyperinsulinemia and insulin resistance20.
To measure Hes3 expression, we prepared mRNA extracts from mouse brains where the olfactory bulb and all parts caudal to the cortex were removed. Mice were carefully age-matched because, as we observed using PCR analysis, expression of Hes3 (both isoforms: Hes3a and Hes3b) drops with age (Fig. S1a). A polyclonal antibody against Hes3 further confirmed the reduction in Hes3 expression with age (Fig. S1b,c). The data are consistent with a role of Hes3 in the NSC/progenitor cell population.
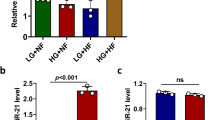
In the pancreas, the toxin streptozotocin (STZ) induces a powerful increase in Hes3 expression, possibly in an effort to promote regeneration of pancreatic islet cells11,12,21. Here we addressed whether similar effects can also be observed in the brain. STZ is used to damage pancreatic islet cells and produce animal models for the study of type 1 diabetes that exhibit reduced production and systemic circulation of insulin18,19,22. Consistent with published studies, mice treated with STZ exhibited increased glucose levels and reduced insulin levels (Fig. S1d,e). In these mice, Hes3a and Hes3b mRNA levels in the brain were significantly increased; in contrast, the mRNA levels of the canonical Notch signaling targets Hes1 and Hes5 were not significantly altered (Fig. 1a). These data show that intraperitoneal administration of STZ leads to Hes3 expression changes in the brain.
Streptozotocin-induced β–cell damage and high fat diet regulate Hes3 expression in the brain. (a) STZ regulates Hes3a and Hes3b levels (N = 6–7). (b,c) HFD (short and long feeding) regulates Hes/Hey gene expression (N = 4–7). (d) HFD (16 weeks) regulates Hes3 expression in different brain areas. The heatmap shows average gene expression for Hes3a and Hes3b in different brain areas (BR, HPT, CBL; N = 5–8). [Data are means ± SEM. Mann-Whitney test; *p < 0.05. HPRT was used as a reference gene]. See also Fig. S1.
HFD in mice leads to a complex condition that involves elevated circulating insulin (an activator of Hes3) as well as complex inflammatory responses (some of which promote and others that are predicted to oppose Hes3 expression)11,15. It is therefore difficult to predict the effect of particular HFD paradigms on brain Hes3; here we investigated this question using established HFD protocols. In both the short and long HFD feeding groups the mice gained BW and exhibited increased insulin and glucose levels (Fig. S1f–h). In the short HFD feeding group, Hes3a, Hes3b, Hes1, but not Hes5 expression were reduced, relative to the short normal diet (ND) feeding group; in the long HFD feeding group, Hes3a and Hes3b expression showed a tendency to decrease, compared to the long ND feeding group, but did not reach statistical significance. Again, Hes1 but not Hes5 levels also dropped (Fig. 1b,c). We used a separate group of mice, fed a HFD for an intermediate period (16 weeks), to identify particular brain areas where Hes3 mRNA levels are regulated. With this group, we dissected distinct brain areas: Hypothalamus (HPT), Cerebellum (CBL), and the Remaining Brain without HPT, CBL, and olfactory bulbs that we denote here as “RB”. HPT is involved in metabolic regulation and it expresses Hes323. Hes3 is highly expressed in the CBL although its roles are unknown; RB would provide an overview of Hes3 regulation in the remaining brain areas. In this group of mice we measured significant reductions in both Hes3a and Hes3b levels in RB, HPT, and CBL (Significant changes: RB: Hes3a and Hes3b; HPT: Hes3a; CBL: Hes3a and Hes3b) (Fig. 1d). These results show that streptozotocin-induced β-cell damage and HFD induce significant alterations in the expression of Hes3 in the brain.
Metformin regulates Hes3 expression in vivo and in vitro
We hypothesized that metformin administration would alter brain Hes3 expression. To show this could have important clinical connotations as many patients are prescribed metformin. Metformin is a widely prescribed medication for type 2 and some cases of type 1 diabetes mellitus that opposes hyperglycemia and improves insulin sensitivity24,25. It regulates a variety of signaling pathways, suggesting that it may have additional effects. Indeed, metformin also appears to have anti-inflammatory effects irrespective of diabetes mellitus status26,27, anti-tumor effects28,29, as well as several effects in the living brain including the promotion of neurogenesis and improved spatial memory formation30,31. Because it regulates signaling pathways that intercept with Hes3, such as mTOR4,32,33, we addressed the effect of metformin on Hes3 expression in the living brain.
We isolated RNA from RB, HPT, and CBL as described above. Metformin affected the expression of multiple Hes/Hey genes in the living brain (Significant changes: RB: Hes6, Hey1, Hey2, HeyL; HPT: Hes3b, Hes5, Hey2, HeyL; CBL: No significant changes) (Figs 2a; S2a–d). Metformin did not significantly affect BW; fasting glucose levels were slightly but significantly reduced; fasting insulin levels were slightly but not significantly increased (Fig. S2e–h). Overall, metformin reduced Hes3 expression in the hypothalamus of the adult mouse brain.
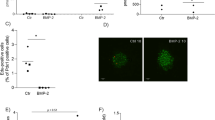
Metformin regulates Hes3 expression in vivo and in vitro. (a) Metformin in drinking water for 2 months regulates Hes/Hey gene mRNA levels in the brain (N = 5–12; CTRL: Control; MET: Metformin). The heatmap shows average gene expression levels in different areas of the brain. (b) Metformin reduces cell number (DAPI-stained nuclei counts) in a dose-dependent manner (N = 4; 72 h, 500 µM). (c) Metformin (500 µM) reduces EdU incorporation in vitro (N = 3; 72 h; 1-tailed t-test). (d) Brightfield images of control and metformin-treated (500 µM, 72 hours) primary fNSC cultures. [Scale bar: 30 μm]. (e,f) Metformin (500 µM) regulates Hes3a and Hes3b mRNA levels in vitro (N = 3, 72 h). [Data are means ± SEM. Mann-Whitney test unless noted otherwise; *p < 0.05. HPRT was used as a reference gene; for the in vitro experiments, data were collected from at least 3 separate experiments]. See also Fig. S2.
To address whether metformin acts directly on NSCs, we used established culture systems of primary mouse fetal NSCs (fNSCs). Dose response experiments demonstrated that 500 µM metformin reduced cell number and 5-ethynyl-2′-deoxyuridine (EdU) incorporation; cell morphology changed to a more differentiated appearance with longer processes (Figs 2b–d; S2i). Together, these results suggest that metformin opposes self-renewal and promotes cell differentiation. Time course experiments revealed a time-dependent increase in Hes3a and Hes3b mRNA levels; at 72 hours we observed increased mRNA levels of Hes3a and Hes3b but not Hes1 or Hes5 (Fig. 2e,f).
These data show that metformin increases Hes3 levels in cultured NSCs, although it decreases Hes3 expression in vivo. We speculate that the expression increase in vitro may be a response to the stress caused to the cells by metformin or due to changes induced by the onset of the differentiation process. In support of this, we show a transient increase in immunoreactivity for Hes3 when cultured NSCs start expressing markers of neurons (TUJ1) and oligodendrocytes (CNPase) (Fig. S2j). Overall, our data show effects of metformin on Hes3 expression in NSCs isolated from the fetal mouse brain. Future experiments may address whether the changes in Hes3 expression mediate effects of metformin to brain function.
Exendin-4 (Ex-4) is another commonly used type-2 diabetes medication. We previously showed that it induces Hes3 expression in a cultured mouse insulinoma cell line11. Unlike metformin, Ex-4 promoted cultured NSC growth at concentrations (200 nM) that induced Hes3 expression (Fig. S3a,b). Also unlike metformin, Ex-4 did not reduce EdU incorporation (Fig. S3c,d). qPCR analysis showed that Ex-4 significantly increased Hes3a and Hes3b mRNA levels, in a time-dependent manner (Fig. S3e). At 72 h of treatment (the time point when Hes3 expression induction reached significance), we did not observe a significant change in Hes1 expression; however, we did measure a significant induction of Hes5 expression (Fig. S3f). These results dissociate the increase in Hes3 expression from cell growth and are consistent with the hypothesis that the increase in Hes3 expression by metformin may be part of a stress response to the treatment.
Hes3 null mice exhibit a quasi-normal phenotype
We established Hes3 expression as an indicator that streptozotocin-induced β-cell damage, high fat diet, and metformin administration affect the brain. It is not yet clear what consequences these changes may have in the health of the experimental models, despite the fact that we previously showed, using Hes3 null mice, that the complete lack of Hes3 leads to increased sensitivity and impaired regeneration in the pancreas11,12 (using the STZ model) and in the brain7 (following cuprizone-induced damage to oligodendrocytes). Extensive future work with conditional genetic mouse models may address the roles of Hes3 in different tissues and organs, in the progression of diabetic and brain-related phenotypes. However, in order to obtain first-level information on possible Hes3 roles, we performed an extensive phenotypic analysis of the Hes3 null (“knockout”) mouse strain.
Homozygous Hes3 null mice are generally healthy and breed normally9 but also have phenotypes that become obvious under stress5,7,11,12. We performed particular phenotypic analyses of the Hes3 null mice following either a ND or a HFD. A summary of the results is presented in Table 1. The full report is provided at https://www.mouseclinic.de (Click on “phenomap” and search for project “Hes3_KO”).
First, we investigated whether the expression of other members of the Hes/Hey gene family is altered in the brains of the Hes3 null mice. We expected this because members of this family are known to suppress the expression of other members10. Because Hes1 and Hes5 are established mediators of canonical Notch signaling, such a result would demonstrate that key pathways in neural stem cell biology are also affected.
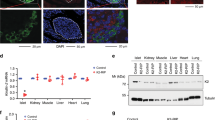
We found that in ND, male Hes3 null mice showed significant alterations in the expression of other Hes/Hey gene family members in the brain, compared to WT; these alterations were much less pronounced in HFD (Figs 3a; S4a–g). These results show that the lack of Hes3 alters the equilibrium of the other Hes/Hey genes and that this outcome is affected by diet.
Hes3 null mice exhibit a quasi-normal phenotype. (a) Heatmap of the average qPCR gene expression levels of different Hes/Hey genes in WT and Hes3 null mice in the brain areas indicated under ND and under HFD conditions (for 16 weeks; N = 4–8). (b) MIF expression in RB of WT and Hes3 null mice under ND and HFD (qPCR; N = 3–6). (c) MIF expression in HPT of WT and Hes3 null mice under ND and HFD (qPCR; N = 4–7). (d) BW progression in mice fed a ND (for 16 weeks; N = 5, 5, same cohort as in (a)) or a HFD (for 16 weeks; N = 11, 9). (e) Hes3 null mice exhibit lower scores (and AUC values) in the ipGTT assay in HFD (after 24 weeks of HFD; see https://www.mouseclinic.de) (N = 11,9). (f) Hes3 null mice exhibit a trend towards higher rectal and body surface temperature than controls [Same cohort of mice as in (e); N = 7,7]. [Data are means ± SEM. Mann-Whitney test; *p < 0.05. HPRT was used as a reference gene]. See also Fig. S4.
We continued our investigation of the consequences of lack of Hes3 by focusing on the expression of Microphage Migration Inhibitory Factor (MIF). Low-grade inflammation is a hallmark of type 2 diabetes mellitus. Of the inflammatory cytokines, MIF has been demonstrated to positively regulate Hes3 expression in isolated NSC cultures15. We addressed how MIF levels are altered by diet and by the lack of Hes3 (comparing WT and Hes3 null mice).
We compared MIF mRNA levels in RB and HPT of WT and Hes3 null mice in both ND and HFD (Fig. 3b,c). In ND we observed the following: Hes3 null mice express a lower amount of MIF in the RB compared to WT. In HPT, the difference did not reach statistical significance. In HFD we observed the following: MIF expression showed no significant difference between WT and Hes3 null mice in the RB, whereas in the HPT there was a non-statistically significant trend towards an increase. MIF expression level in WT mice was affected by diet; HFD reduced MIF expression in the RB. In contrast, we observed an increase in the HPT.
Whereas MIF levels were altered by diet in WT mice in both the RB and HPT, Hes3 null mice did not exhibit changes in MIF expression in the RB due to diet. They did, however, exhibit an increase in HPT, similarly to WT mice. The results suggest a complex interplay among Hes3, diet, and MIF in different brain areas and call for additional studies to address the potential reciprocal regulation between Hes3 and the inflammatory system.
The expression changes in the brain prompted us to investigate potential phenotypes that relate to brain function. We performed a set of neurological phenotypic analyses in Hes3 null and WT mice fed a ND. We did not observe significant differences between the Hes3 null and WT mice in terms of behavior (open field test), neurology (Modified SHIRPA, auditory brain stem response, rotarod test, grip strength), nociception (hot plate test), and eye functions (Scheimpflug imaging, OCT, LIB, drum). Regarding energy metabolism, indirect calorimetry showed no differences in ND, whereas NMR showed a trend towards increased fat content over time mainly in male mutants. We also found no dysmorphology phenotypes (Anatomical observation, X-ray, MicroCT scans of dissected bones), in ND. We also did not find any clear cardiovascular phenotypes in ND; awake ECG Echocardiography in ND showed a very mild increase in septum width in systole (males), a very mild reduction in heart rate and very mild alterations in QRS, ST and Qtdisp intervals (probably by chance) (Table 1; https://www.mouseclinic.de; Fig. S5a-l, and Supplementary Material-GMC Report).
The very mild phenotypes relating to behavior, neurology, and nociception pushed our focus to other potential phenotypes, for example, those that are relevant to metabolism. Using male mice, we found that in ND, Hes3 null mice behaved similarly to wild-type (WT) mice in terms of BW gain; in HFD, Hes3 null mice gained less BW, compared to WT mice (Fig. 3d). In both ND and HFD, Hes3 null mice showed lower glucose levels during the course of the ipGTT compared to WT mice, resulting in slightly lower AUC values especially for the second part of the test [AUC (30–120 mins)], which might indicate an improved glucose tolerance (HFD: Fig. 3e; ND: Supplementary Material-GMC Report). Male Hes3 null mice fed a HFD also exhibited a trend (p = 0.053) towards higher rectal and body surface temperature (Fig. 3f). [Two Way ANOVA analysis revealed a significant increase in the body temperature of WT versus KO mice when both male and female mice were analyzed together (p = 0.036; N = 14,14)] (Supplementary Material-GMC Report).
Beyond the brain, Hes3 null mice in ND also exhibited subtle alterations in the leukocyte subpopulations [increased frequency of B cells, decreased frequency of CD4 single positive T cells, increased frequency of CD4 CD8 double positive T cells, increased proportion of CD8 single positive T cells (females), and increased CD44 expression on CD4+ T cells], although no evidence for pathological effects in the immune system was observed (Table 1 and Supplementary Material-GMC Report).
Overall, the phenotypic analyses of the Hes3 null mouse strain show mild phenotypes along with an altered equilibrium of the expression of multiple other Hes/Hey genes in the brain and point towards multiple roles that may be addressed in future studies.
Discussion
Diabetes affects the brain in ways that are insufficiently understood34. Ironically, diabetes medication such as metformin also affects the brain, and we don’t know the consequences to the patient. It is important to reveal the molecular mechanisms that are affected in order to be able to predict outcome and design appropriate therapeutic interventions. A sensitive biomarker in the brain whose expression levels change in response to disease progression and metformin administration would be a valuable new tool. Such a biomarker would be even more valuable if it is already known to play roles in brain regeneration.
Here we used a mouse model of type 1 diabetes mellitus (the streptozotocin-induced β-cell damage model), a mouse model of high fat diet exhibiting systemic inflammation and insulin resistance, often used to study aspects of type 2 diabetes, and a mouse model of metformin administration to establish the transcription factor Hes3 as such a biomarker. We show that the expression of Hes3 in the brain is regulated in all three mouse models. We present a conceptual diagram of how Hes3 may be regulated by a number of parameters, including signal transduction pathways, inflammatory responses, insulin levels, age, etc. (Fig. S6).
Future work may address the mechanisms by which Hes3 is regulated in the animal models we employed, as well as, potentially in patients with type 1 and type 2 diabetes, and during metformin administration. It may also address the impact that Hes3 expression changes might have to the brain. For now, we may speculate on a number of possibilities.
First, insulin may directly regulate Hes3+ cells in the brain. Type 1 and type 2 diabetes mellitus as well as being overweight or obese are known risk factors for developing cognitive impairment and dementia35,36,37,38,39,40. In part, this risk is thought to be due to aberrant insulin actions directly to the brain. Beyond stimulating glucose metabolism, insulin (along with the related hormone Insulin-like growth factor 1, IGF-1) has multiple functions in the brain, supporting the survival of neurons and oligodendrocytes41, promoting synaptic integrity and plasticity42, and helping working memory and cognition43. At the signal transduction level, post-mortem analysis of brains from patients with Alzheimer’s disease reveals perturbed signaling downstream of the insulin receptor, including reduced insulin and IGF-1 binding to their receptors and impaired PI3K/Akt signaling44,45,46,47. Hes3 is regulated by insulin, as we previously showed in neural stem cell cultures as well as in vivo4,5,6. In fact, Hes3 is a key component of a signal transduction pathway that is involved in a variety of regeneration paradigms in different cell types in vitro and in different tissues in vivo4,5,6,11,12,14,15,21,48,49,50. It is possible, therefore, that altering circulating insulin levels may lead to sufficient changes in brain interstitial fluid levels, and that may directly affect Hes3-expressing cells. But it has proven complicated to determine how insulin levels in the interstitial fluid of the brain change when circulating insulin levels are altered51,52,53. Cerebrospinal fluid (CSF) levels have been used as surrogate measurements for brain interstitial levels. However, this may not be an accurate measurement as insulin is thought to predominantly enter the brain via the blood-brain barrier, as has been expertly reviewed previously54. In addition, elevated extracellular glucose concentrations have been suggested to be able to induce insulin production from cerebral cortical neuroglial cells52,53,54,55; how that may contribute to local Hes3 expression regulation is not yet addressed. Alternatively, insulin that reaches the cerebrospinal fluid (CSF) may also be able to induce Hes3 expression in certain brain areas adjacent to the ventricles; in support of this, in the original studies demonstrating endogenous NSC activation by insulin and other reagents, the treatments were injected into the lateral ventricles of adult rats4,5,6.
Second, oxidative and inflammatory responses may also play an important role in regulating Hes3 expression in different cell types56,57. It is important to not group all inflammatory cytokines together when addressing their effects on Hes3. Whereas many inflammatory cytokines (e.g., the interleukin family) activate the JAK-STAT signaling pathway and would thus be expected to suppress Hes3 expression, MIF has been shown to promote it via an Akt/mTOR/STAT3-Ser mechanism11,12,15,21. Therefore, the precise inflammatory responses activated in different disease models (or, at different stages of the same model) may contribute to the exact Hes3 expression patterns that we observed. Such a task will require a detailed determination of the production of multiple cytokines at different time points, and an assessment of their potential to induce or suppress Hes3 expression. A better understanding of the interaction between specific inflammatory responses and Hes3 may help inform drug discovery programs aimed at both modulating inflammation and protecting brain tissue. Future studies may also address how Hes3 expression is specifically altered by oxidative and inflammatory stress components such as oxygen radicals produced by the mitochondria of affected cells and activated, nuclear NF-kB. Cross-talk between NF-kB and JAK-STAT signaling as well as between reactive oxygen species and JAK-STAT increases the complexity by which Hes3 may be regulated58,59,60.
In addition to the disease models themselves, we hypothesized that metformin, a common medication for type 2 diabetes mellitus61,62 would also affect brain Hes3 levels. Our data show different effects of metformin on Hes3 expression in vitro and in vivo, arguing for complex mechanisms by which metformin regulates Hes3 expression. This could be due to the broad range of signaling pathways affected by metformin that include several known modulators of Hes34,63,64. Although the precise molecular mechanisms by which metformin affects cells are not fully elucidated, studies from various cell systems have identified a number of signaling pathways that are involved. In triple-negative breast cancer cell lines, metformin was shown to oppose both JAK/STAT3-Tyr activity and STAT3-Ser phosphorylation65. It is possible that, depending on which of the two branches of STAT3 is mostly affected, the outcome may either favor or oppose Hes3 expression because Hes3 is positively regulated by inducers of STAT3-Ser phosphorylation and negatively regulated by JAK kinase which leads to STAT3-Tyr phosphorylation4. In addition, metformin opposes mTOR activation, via 5′ AMP-activated protein kinase (AMPK) stimulation32,33,66,67,68, a function that should also oppose Hes3 expression (mTOR is a positive regulator of Hes3 expression4). Metformin also regulates insulin-like growth factor 1 (IGF-1) and p38 mitogen-activated protein kinases (p38 MAPK) which are signaling components with prominent roles in the self-renewal of NSCs and the regulation of Hes3 expression4,5,64. Inflammatory responses triggered by metformin (as well as by type 2 diabetes itself)69,70,71 further complicate the predictability of its effects on Hes3 expression because different inflammatory cytokines may have opposite effects on Hes3 expression (depending on whether they predominantly stimulate the JAK-STAT or Akt/mTOR pathways, for example). It will be interesting to determine whether the complex mechanisms by which metformin may regulate Hes3 contribute to the lack of consensus regarding the drug’s potential role as a therapeutic agent in neurodegenerative disease72,73. It is conceivable that patients on metformin will benefit from concomitant treatments that modulate the inflammatory response in the brain such that Hes3 levels are maintained within an appropriate range. For example, promoting MIF responses while suppressing interleukin responses may have beneficial effects on the neural stem/progenitor population, on neurons, and on cognitive function.
Our data implicate aging in the regulation of Hes3. This may not be too surprising, given that there is a general consensus that neural stem cell biomarkers decrease with age. Our observation that Hes3 expression in the brain drops with age might have important connotations in diabetes mellitus. This is in light of studies suggesting differences between children and adults with type 1 diabetes in the way that their brain responds to the disease. In an imaging study involving a large cohort of children with type 1 diabetes, it was revealed that hippocampal volumes were increased in the children with the largest number of severe hypoglycemic episodes74. This result was interpreted as a manifestation of sensitivity of the hippocampus to acute hypoglycemia. It is contrary to data obtained from studies with adult type 1 diabetes patients that revealed no changes in hippocampal volume75. Animal studies reinforce the hypothesis that postnatal age affects the way that the brain responds to hypoglycemia1,76. It will be of significant interest to determine the potential role of Hes3 in this age-related effect. It will also be valuable to determine whether the abundant, Hes3-expressing endogenous NSC/progenitor cell population in the hippocampus is affected in these children. The postnatal hippocampus is one of the few brain areas where regular neurogenesis is observed from a local pool of stem/progenitor cells77. Many putative stem/progenitor cells in the hippocampus co-express Hes3 and their number increases in vivo when pharmacological agents that induce Hes3 expression are injected into the brain17. Therefore, one could expect that the combination of a high-Hes3 young brain and elevated Hes3 expression due to diabetes mellitus could impact the stem cell-rich cytoarchitecture of the hippocampus. Given that MIF expression drops with age78,79 and is associated with longevity80, it is intriguing to speculate that age-dependent MIF expression changes may be, in part, responsible for the accompanying drop in Hes3 expression.
The age-based differences in brain Hes3 expression are particularly relevant in light of current efforts to assess new clinical potential of metformin. One study (Targeting Aging with Metformin, TAME; https://www.afar.org/natgeo/) will assess its anti-aging potential81; it may be of interest to obtain more information on how Hes3 expression is regulated in humans by age and by metformin itself. The other study (Autoimmune Diabetes Accelerator Prevention Trial, adAPT; http://adaptdiabetes.org/) involves administering metformin to healthy children who are at risk of developing type 1 diabetes as a prevention strategy; it may be beneficial to assess differences in brain Hes3 expression in detail between children and adults in order to potentially better predict unforeseen consequences.
Given the potential of Hes3 as a biomarker, it is valuable to obtain information on its potential roles. In our past work and here we have focused on measuring Hes3 expression changes in the damaged and regenerating pancreas and brain. But it is expressed in various other tissues as well, where its roles are much less known. Here we provide a first-order phenotypic analysis of Hes3 null mice. Future studies may involve the use of conditional and inducible genetic mouse models so as to obtain phenotypic information on Hes3 in particular time points and tissues. For now, the phenotypic analysis we present provides some clues to its roles.
Hes3 null mice exhibit differences in the expression of other Hes/Hey gene family members in the brain, relative to WT mice. They also show altered MIF expression as well as differences in leukocyte lineage frequencies are (albeit mildly), suggesting potential consequences in the levels of inflammatory cytokines and in chronic low-grade systemic inflammation, a risk factor in many patients with obesity82.
These differences do not seem to obviously affect brain function as phenotypic analyses on behavior, neurology, nociception, and eye function showed no significant differences between Hes3 null and WT mice. It is possible, however, that, in the context of an appropriate challenge to the mouse, phenotypic differences might be revealed. This is in accordance with our previous work showing that Hes3 null mice exhibit significantly altered responses to cuprizone-induced damage of oligodendrocytes in the brain and altered regeneration of oligodendrocytes afterwards7. It is possible that the roles of Hes3 become more evident under conditions of challenge. In fact, this is very similar to what we observed in the pancreas, in our previous work: Hes3 null mice behave very similarly to WT mice, in terms of pancreatic function under normal conditions but exhibit much greater damage and reduced regeneration following streptozotocin-induced pancreas damage11,12. Therefore, future work may search for disease paradigms where the lack of Hes3 results in clear brain phenotypes.
We report a number of mild phenotypes, some of which are observed under HFD conditions but not on ND conditions. For example, we found no differences in BW between Hes3 null and WT mice when fed a ND, but significant differences when placed on a HFD. (In a separate cohort of mice placed under a distinct HFD protocol/composition, we did not observe statistically significant BW changes: Supplementary Material-GMC Report). Future studies may investigate the reasons behind this difference (fat tissue composition, thermoregulation, composition of diet, immune/inflammatory reactions, animal housing stresses, etc.).
Conversely, at the molecular level, we observed much more pronounced effects of the lack of Hes3 in ND, compared to in HFD, regarding the expression of other members of the Hes/Hey gene family. It is possible that because in HFD Hes3 expression is reduced relative to ND, the effect of lacking Hes3 is not as pronounced. It is also possible that HFD regulates other Hes/Hey genes independently from Hes3 in a manner that overrides, in part, the genetic lack of Hes3.
Here we show that STZ-induced β-cell damage, high fat diet, and metformin administration in vivo regulate Hes3 levels in the adult mouse brain. Our data establish Hes3 as a potentially valuable biomarker for the impact of diabetes on the plasticity potential of the brain. Because Hes3 is a key component of a signal transduction pathway involved in neural stem cell biology and regeneration, it may also provide leads towards new therapeutic opportunities.
Methods
All methods were performed in accordance with the relevant guidelines and regulations.
Animals
6 week-old C57Bl6/J male mice were obtained from Janvier and they were used in accordance with the approved guidelines from the Landesdirektion Sachsen. The Hes3 null mouse line was kindly provided by R. Kageyama9. At the GMC mice are housed according to the GMC housing conditions and German laws. All tests performed at the GMC were approved by the responsible authority of the district government of Upper Bavaria, Germany.
Microscopy
Brains were dissected, fixed overnight and cryoprotected in 30% sucrose solution phosphate buffered saline at 4 °C until sinking. 16 μm brain tissue sections were prepared in the cryostat and mounted on glass slides. Immunofluorescence staining for Hes3 was performed as described previously11. Images were acquired with a Zeiss LSM780 system. Image analysis was performed using the Fiji software.
Tissue Collection
Brain tissues were dissected on ice. Depending on the experimental plan brain without cerebellum and olfactory bulb (indicated as BRAIN), brain without cerebellum, olfactory bulb and hypothalamus (indicated as Remaining Brain ‘’RB”), hypothalamus (HPT) and cerebellum (CBL) were dissected. We removed the olfactory bulbs for consistency as, sometimes, they are damaged during dissection. RB was split into two hemispheres and one was directly processed for RNA extraction. CBL was also divided in two parts and the same procedure was followed. Full HPT was used for the RNA extraction. Total RNA was isolated with the High Pure RNA isolation kit (Roche, 11828665001) and 1 μg of total RNA per sample was reverse transcribed using Promega M-MLV reverse transcriptase (Promega, M170B).
Real-Time PCR
PCR was performed with DreamTaq Green DNA Polymerase (EP0712, ThermoScientific) with addition of betaine (5M, B0300, Sigma, B0300). qPCR experiments were performed with SsoFast EvaGreen Supermix (172–5201, Biorad) in a CFX384 Real time PCR Detection System83. Primer sets and reaction protocols are in Suppl. Tables 1–3. Relative gene expression was evaluated with the ΔΔCt method upon normalization to hypoxanthine-guanine phosphoribosyltransferase (HPRT). Primer sets and reaction protocols (Gene: Seq. 5′—> 3′ FW/Seq. 5′—> 3′ REV/Method) were:
mHes1: AAGATAGCTCCCGGCATTCCAAGC/AGCGCGGCGGTCATCTGC/PCR, qPCR;
mHes3: AAAGCTGGAGAAGGCCGATA/TCCTTGCCTACGTCTCACCA/PCR;
mHes3a: GTGATCTCCAAGCCTCTGATGGAGAA/CAGCTTTCGTTTCCGTATCTGATGTGA/PCR, qPCR;
mHes3b: CCAGCAGCTTCCGAAAGATCTCCA/TCTCCAGCTTTCGTTTCCGTATCTGA/PCR, qPCR;
mHes5: CAACAGCAGCATAGAGCAGC/AGGCTTTGCTGTGTTTCAGG/PCR, qPCR;
mHes6: GGTGCAGGCCAAGCTAGAG/TGAAAGCTGCTACCCTCACG/PCR, qPCR;
mHes7: CCCAAGATGCTGAAGCCGTTGGT/AGCTTCGGGTTCCGGAGGTTCT/PCR, qPCR;
mHey1: AGGCATCATCGAGAAGCGCC/AGCTTAGCAGATCCCTGCTTCTCA/PCR, qPCR;
mHey2: TGAGAAGACTAGTGCCAACAGC/TGGGCATCAAAGTAGCCTTTA/PCR, qPCR;
mHeyL: CAGCCCTTCGCAGATGCAA/CCAATCGTCGCAATTCAGAAAG/PCR, qPCR;
mHPRT: AAGCTTGCTGGTGAAAAGGA/TTGCGCTCATCTTAGGCTTT/PCR, qPCR;
mMIF: TTAGCGGCACGAACGATCC/ACAGCAGCTTACTGTAGTTGC/qPCR.
The PCR protocol steps were (Cycling step: Temp, °C/Time/# of cycles): Initial Denaturation: 95/10 min/1; Denaturation: 95/4 min/35; Annealing: 60/30 sec/35; Extension: 72/1 min/35; Final Extension: 72/10 min/1.
The qPCR protocol steps were (Cycling step: Temp, °C/Time/# of cycles): Enzyme activation: 95/1 min/1; Denaturation: 95/5 sec/40; Annealing-Extension: 60/15 sec/40; Melt Curve: 65–95 °C in 0.5 °C increments/5 sec per step/1
Aging
We examined 17 week old mice (from here on referred to as “Young”) and 34 week old mice (from here on referred to as “Old”).
Animal models
Single High Dose STZ
8 week-old mice were injected intraperitoneally [phosphate-buffered saline (PBS) vehicle control or STZ (180 mg/kg, Sigma Aldrich, S0130)] and were euthanized 8 weeks later.
HFD
8 week old mice were fed Normal Diet (ND, 10% kcal % fat, D12450B, OpenSource Diets – Research Diets) or High Fat Diet (HFD, 60% kcal % fat, D12450B, OpenSource Diets – Research Diets) and euthanized after ~10 weeks (“Short Feeding”) or ~30 weeks (“Long Feeding”). For the HFD feedings performed at the GMC, please refer to the GMC Report in the Supplementary Materials section.
Metformin administration
Metformin was administered in the drinking water (2 g/l, Sigma, D-150959) of 8 week old mice for 8 weeks. Water was changed 2 times per week.
Metabolic Analyses
Body weight (BW): We measured the BW of the mice weekly at the same time of day. Intraperitoneal Glucose-Tolerance-Test (ipGTT): Mice were used for the glucose tolerance test after a 16–18 hours-lasting overnight food-withdrawal. In the beginning of the test, the body weight of mice was determined. For the determination of the fasting blood glucose level, the tip of the tail was scored using a sterilized scalpel blade and a small drop of blood was analyzed with the Accu-Chek Aviva glucose analyzer (Roche/Mannheim). Thereafter mice were injected intraperitoneally with 2 g of glucose/kg body weight using a 20% glucose solution, a 25-gauge needle and a 1-ml syringe. 15, 30, 60 and 120 minutes after glucose injection, additional blood samples (one drop each) were collected and used to determine blood glucose levels as described before. Repeated bleeding was induced by removing the clot from the first incision and massaging the tail of the mouse. After the experiment was finished, mice were placed in a cage with plentiful supply of water and food. Fasting BW, Fasting Blood Insulin and Fasting Blood Glucose levels: Fasting BW and fasting blood glucose were measured with Accu-Chek glucose meter (Roche, Mannheim, Germany) and blood samples were collected for fasting insulin concentration determination with an Elisa Kit (Crystal Chem) and according to the manufacturer’s instructions.
NSC cultures
Cell isolation
Fetal NSCs (fNSCs) were dissected and cultured from mouse embryos at embryonic day 13.5 (E13.5). Cells were grown in serum-free N2 medium; basic fibroblast growth factor [bFGF (233-FB-01M, R&D Systems)] was added to the cells at a concentration of 20 ng/ml daily84.
Treatments
Cells were seeded at 10,000 cells per well in a 12 well plate and treated with different concentrations of metformin (Sigma, D-150959) or exendin-4 [Ex-4 (Biotrend, BP0111)], beginning at 24 h after plating. Cells were fixed after 72 h with 4% paraformaldehyde (PFA) for 20 minutes and nuclei were stained with 4′,6-Diamidino-2 Phenylindole (DAPI).
Cell number, Proliferation
Cell proliferation was determined after 72 h by incubating the cells with 10 μM 5-ethynyl-2′-deoxyuridine (EdU) for 5 h and followed by visualization using the Click-IT EdU Alexa Fluor 594 Imaging Kit (Invitrogen C10339). Five images (using a 20x objective) from each well were acquired (3 wells per plate) with a standard Zeiss structured illumination microscope (Zeiss – Axio Observer Z1, inverted) and cells were counted using the Fiji software.
PCR/qPCR
For the time-course experiments cells were seeded at 500,000 cells per T25 flask and treatments (0 or 500 μM metformin) were initiated 24 h after plating. Cells were collected at 6, 24, 48 and 72 h and processed directly for PCR/qPCR experiments. Cell culture medium and treatments were changed daily.
Mouse phenotyping
Comprehensive phenotypical characterization of Hes3 null mice was performed at the German Mouse Clinic (GMC), Munich, Germany85. 64 mice (16 males, 16 females, 16 control males and 16 control females) were used beginning at age 9 weeks. Tests were conducted using the protocols described before86 and referenced at https://www.mouseclinic.de (Click on “VIEW RESULTS OF MUTANT LINES” or “phenomap” and search for project “Hes3_KO”). The methods described in Fig. S5 (for Behavior, neurology, and nociception phenotyping) as well as additional analyses can also be found in the Supplementary Material-GMC Report.
An additional cohort (11 males, 11 females, 11 control males and 11 control females) was fed a HFD (E15741-347 (D12492 mod.) Ssniff Spezialdiäten GmbH, Soest, Germany, containing 60 energy-% from beef tallow). Mice were subjected to the following tests beginning at age 12–13 weeks: Body composition analysis (qNMR, Minispec LF 50, Bruker, Ettlingen, Germany), 21 hours indirect calorimetry (Phenomaster, TSE Systems Gmbh, Bad Homburg, Germany), rectal body temperature, body surface temperature (thermosensor: Almemo ZA 9040, data logger: Almemo 2290-8, Ahlborn, Holzkirchen, Germany), and ipGTT after overnight food deprivation.
Heat maps
Heat maps were generated in Morpheus https://software.broadinstitute.org/morpheus/.
Statistical analyses
Data are expressed as means ± SEM. Statistical analyses were performed in Graphpad Prism (GraphPad Software, Inc., San Diego, CA). The Student’s t test, a Mann- Whitney U test, or one-way ANOVA were used and significance was set at p < 0.05. A detailed account of the statistical methods used in the phenotypic analysis of the Hes3 null mice is provided in the GMC Report in the Supplementary Materials section.
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request and in the https://www.mouseclinic.de repository (Click on “VIEW RESULTS OF MUTANT LINES” or “phenomap” and search for project “Hes3_KO”).
References
Seaquist, E. R. The final frontier: how does diabetes affect the brain? Diabetes 59, 4–5, https://doi.org/10.2337/db09-1600 (2010).
Fatt, M. et al. Metformin Acts on Two Different Molecular Pathways to Enhance Adult Neural Precursor Proliferation/Self-Renewal and Differentiation. Stem cell reports 5, 988–995, https://doi.org/10.1016/j.stemcr.2015.10.014 (2015).
Dadwal, P. et al. Activating Endogenous Neural Precursor Cells Using Metformin Leads to Neural Repair and Functional Recovery in a Model of Childhood Brain Injury. Stem cell reports 5, 166–173, https://doi.org/10.1016/j.stemcr.2015.06.011 (2015).
Androutsellis-Theotokis, A. et al. Notch signalling regulates stem cell numbers in vitro and in vivo. Nature 442, 823–826 (2006).
Androutsellis-Theotokis, A., Rueger, M. A., Mkhikian, H., Korb, E. & McKay, R. D. Signaling pathways controlling neural stem cells slow progressive brain disease. Cold Spring Harb Symp Quant Biol 73, 403–410 (2008).
Androutsellis-Theotokis, A. et al. Targeting neural precursors in the adult brain rescues injured dopamine neurons. Proc Natl Acad Sci USA 106, 13570–13575, https://doi.org/10.1073/pnas.0905125106 (2009).
Toutouna, L. et al. Hes3 expression in the adult mouse brain is regulated during demyelination and remyelination. Brain Res 1642, 124–130, https://doi.org/10.1016/j.brainres.2016.03.014 (2016).
Hirata, H., Ohtsuka, T., Bessho, Y. & Kageyama, R. Generation of structurally and functionally distinct factors from the basic helix-loop-helix gene Hes3 by alternative first exons. J Biol Chem 275, 19083–19089 (2000).
Hirata, H., Tomita, K., Bessho, Y. & Kageyama, R. Hes1 and Hes3 regulate maintenance of the isthmic organizer and development of the mid/hindbrain. Embo J 20, 4454–4466 (2001).
Imayoshi, I. & Kageyama, R. bHLH factors in self-renewal, multipotency, and fate choice of neural progenitor cells. Neuron 82, 9–23, https://doi.org/10.1016/j.neuron.2014.03.018 (2014).
Masjkur, J. et al. Hes3 is expressed in the adult pancreatic islet and regulates gene expression, cell growth, and insulin release. J Biol Chem 289, 35503–35516, https://doi.org/10.1074/jbc.M114.590687 (2014).
Masjkur, J. et al. Endocrine Pancreas Development and Regeneration: Noncanonical Ideas From Neural Stem Cell Biology. Diabetes 65, 314–330, https://doi.org/10.2337/db15-1099 (2016).
Androutsellis-Theotokis, A. et al. Angiogenic factors stimulate growth of adult neural stem cells. PLoS One 5, e9414, https://doi.org/10.1371/journal.pone.0009414 (2010).
Salewski, R. P. et al. The generation of definitive neural stem cells from PiggyBac transposon-induced pluripotent stem cells can be enhanced by induction of the NOTCH signaling pathway. Stem Cells Dev 22, 383–396 (2012).
Ohta, S. et al. Macrophage migration inhibitory factor (MIF) promotes cell survival and proliferation of neural stem/progenitor cells. J Cell Sci 125, 3210–3220 (2012).
Androutsellis-Theotokis, A., Walbridge, S., Park, D. M., Lonser, R. R. & McKay, R. D. Cholera toxin regulates a signaling pathway critical for the expansion of neural stem cell cultures from the fetal and adult rodent brains. PLoS One 5, e10841, https://doi.org/10.1371/journal.pone.0010841 (2010).
Pacioni, S. et al. Fast, potent pharmacological expansion of endogenous hes3+/sox2+ cells in the adult mouse and rat hippocampus. PLoS One 7, e51630, https://doi.org/10.1371/journal.pone.0051630 (2012).
Asrafuzzaman, M. et al. Animal models for assessing the impact of natural products on the aetiology and metabolic pathophysiology of Type 2 diabetes. Biomedicine & pharmacotherapy=Biomedecine & pharmacotherapie 89, 1242–1251, https://doi.org/10.1016/j.biopha.2017.03.010 (2017).
Kitada, M., Ogura, Y. & Koya, D. Rodent models of diabetic nephropathy: their utility and limitations. International journal of nephrology and renovascular disease 9, 279–290, https://doi.org/10.2147/ijnrd.S103784 (2016).
Kleinert, M. et al. Animal models of obesity and diabetes mellitus. Nature reviews. Endocrinology 14, 140–162, https://doi.org/10.1038/nrendo.2017.161 (2018).
Poser, S. W. et al. Concise Review: Reprogramming, Behind the Scenes: Noncanonical Neural Stem Cell Signaling Pathways Reveal New, Unseen Regulators of Tissue Plasticity With Therapeutic Implications. Stem Cells Transl Med 4, 1251–1257, https://doi.org/10.5966/sctm.2015-0105 (2015).
Al-Awar, A. et al. Experimental Diabetes Mellitus in Different Animal Models. Journal of diabetes research 2016, 9051426, https://doi.org/10.1155/2016/9051426 (2016).
Nikolakopoulou, P. et al. STAT3-Ser/Hes3 Signaling: A New Molecular Component of the Neuroendocrine System? Horm Metab Res 48, 77–82, https://doi.org/10.1055/s-0041-111699 (2016).
Harris, K., Boland, C., Meade, L. & Battise, D. Adjunctive therapy for glucose control in patients with type 1 diabetes. Diabetes, metabolic syndrome and obesity: targets and therapy 11, 159–173, https://doi.org/10.2147/dmso.S141700 (2018).
Livingstone, R., Boyle, J. G., Petrie, J. R. & Team, R. S. A new perspective on metformin therapy in type 1 diabetes. Diabetologia 60, 1594–1600, https://doi.org/10.1007/s00125-017-4364-6 (2017).
Saisho, Y. Metformin and Inflammation: Its Potential Beyond Glucose-lowering Effect. Endocrine, metabolic & immune disorders drug targets 15, 196–205 (2015).
Cameron, A. R. et al. Anti-Inflammatory Effects of Metformin Irrespective of Diabetes Status. Circ Res 119, 652–665, https://doi.org/10.1161/CIRCRESAHA.116.308445 (2016).
Evans, J. M., Donnelly, L. A., Emslie-Smith, A. M., Alessi, D. R. & Morris, A. D. Metformin and reduced risk of cancer in diabetic patients. Bmj 330, 1304–1305 (2005).
Chae, Y. K. et al. Repurposing metformin for cancer treatment: current clinical studies. Oncotarget 7, 40767–40780, https://doi.org/10.18632/oncotarget.8194 (2016).
Wang, J. et al. Metformin activates an atypical PKC-CBP pathway to promote neurogenesis and enhance spatial memory formation. Cell Stem Cell 11, 23–35, https://doi.org/10.1016/j.stem.2012.03.016 (2012).
van Harten, B., de Leeuw, F. E., Weinstein, H. C., Scheltens, P. & Biessels, G. J. Brain imaging in patients with diabetes: a systematic review. Diabetes care 29, 2539–2548, https://doi.org/10.2337/dc06-1637 (2006).
Rozengurt, E. Mechanistic target of rapamycin (mTOR): a point of convergence in the action of insulin/IGF-1 and G protein-coupled receptor agonists in pancreatic cancer cells. Front Physiol 5, 357, https://doi.org/10.3389/fphys.2014.00357 (2014).
Sinnett-Smith, J., Kisfalvi, K., Kui, R. & Rozengurt, E. Metformin inhibition of mTORC1 activation, DNA synthesis and proliferation in pancreatic cancer cells: dependence on glucose concentration and role of AMPK. Biochem Biophys Res Commun 430, 352–357, https://doi.org/10.1016/j.bbrc.2012.11.010 (2013).
Selvarajah, D. et al. Magnetic resonance neuroimaging study of brain structural differences in diabetic peripheral neuropathy. Diabetes care 37, 1681–1688, https://doi.org/10.2337/dc13-2610 (2014).
de la Monte, S. M. Insulin Resistance and Neurodegeneration: Progress Towards the Development of New Therapeutics for Alzheimer’s Disease. Drugs 77, 47–65, https://doi.org/10.1007/s40265-016-0674-0 (2017).
Pedditizi, E., Peters, R. & Beckett, N. The risk of overweight/obesity in mid-life and late life for the development of dementia: a systematic review and meta-analysis of longitudinal studies. Age and ageing 45, 14–21, https://doi.org/10.1093/ageing/afv151 (2016).
Alosco, M. L. & Gunstad, J. The negative effects of obesity and poor glycemic control on cognitive function: a proposed model for possible mechanisms. Current diabetes reports 14, 495, https://doi.org/10.1007/s11892-014-0495-z (2014).
Luchsinger, J. A. et al. Relation of diabetes to mild cognitive impairment. Archives of neurology 64, 570–575, https://doi.org/10.1001/archneur.64.4.570 (2007).
S Roriz-Filho, J. et al. (Pre)diabetes, brain aging, and cognition. Biochim Biophys Acta 1792, 432–443, https://doi.org/10.1016/j.bbadis.2008.12.003 (2009).
Fotuhi, M., Do, D. & Jack, C. Modifiable factors that alter the size of the hippocampus with ageing. Nat Rev Neurol 8, 189–202, https://doi.org/10.1038/nrneurol.2012.27 (2012).
de la Monte, S. M. & Wands, J. R. Review of insulin and insulin-like growth factor expression, signaling, and malfunction in the central nervous system: relevance to Alzheimer’s disease. Journal of Alzheimer’s disease: JAD 7, 45–61 (2005).
Chiu, S. L., Chen, C. M. & Cline, H. T. Insulin receptor signaling regulates synapse number, dendritic plasticity, and circuit function in vivo. Neuron 58, 708–719, https://doi.org/10.1016/j.neuron.2008.04.014 (2008).
de la Monte, S. M. Intranasal insulin therapy for cognitive impairment and neurodegeneration: current state of the art. Expert opinion on drug delivery 10, 1699–1709, https://doi.org/10.1517/17425247.2013.856877 (2013).
Rivera, E. J. et al. Insulin and insulin-like growth factor expression and function deteriorate with progression of Alzheimer’s disease: link to brain reductions in acetylcholine. Journal of Alzheimer’s disease: JAD 8, 247–268 (2005).
Talbot, K. et al. Demonstrated brain insulin resistance in Alzheimer’s disease patients is associated with IGF-1 resistance, IRS-1 dysregulation, and cognitive decline. J Clin Invest 122, 1316–1338, https://doi.org/10.1172/JCI59903 (2012).
Steen, E. et al. Impaired insulin and insulin-like growth factor expression and signaling mechanisms in Alzheimer’s disease–is this type 3 diabetes? Journal of Alzheimer’s disease: JAD 7, 63–80 (2005).
Moloney, A. M. et al. Defects in IGF-1 receptor, insulin receptor and IRS-1/2 in Alzheimer’s disease indicate possible resistance to IGF-1 and insulin signalling. Neurobiology of aging 31, 224–243, https://doi.org/10.1016/j.neurobiolaging.2008.04.002 (2010).
Masjkur, J. et al. A defined, controlled culture system for primary bovine chromaffin progenitors reveals novel biomarkers and modulators. Stem Cells Transl Med 3, 801–808, https://doi.org/10.5966/sctm.2013-0211 (2014).
Cassady, J. P. et al. Direct lineage conversion of adult mouse liver cells and B lymphocytes to neural stem cells. Stem cell reports 3, 948–956, https://doi.org/10.1016/j.stemcr.2014.10.001 (2014).
Park, D. M. et al. Hes3 regulates cell number in cultures from glioblastoma multiforme with stem cell characteristics. Sci Rep 3, 1095, https://doi.org/10.1038/srep01095 (2013).
Gray, M. J. et al. HIF-1alpha, STAT3, CBP/p300 and Ref-1/APE are components of a transcriptional complex that regulates Src-dependent hypoxia-induced expression of VEGF in pancreatic and prostate carcinomas. Oncogene 24, 3110–3120 (2005).
Blazquez, E., Velazquez, E., Hurtado-Carneiro, V. & Ruiz-Albusac, J. M. Insulin in the brain: its pathophysiological implications for States related with central insulin resistance, type 2 diabetes and Alzheimer’s disease. Frontiers in endocrinology 5, 161, https://doi.org/10.3389/fendo.2014.00161 (2014).
Havrankova, J., Roth, J. & Brownstein, M. J. Concentrations of insulin and insulin receptors in the brain are independent of peripheral insulin levels. Studies of obese and streptozotocin-treated rodents. J Clin Invest 64, 636–642, https://doi.org/10.1172/JCI109504 (1979).
Gray, S. M., Meijer, R. I. & Barrett, E. J. Insulin regulates brain function, but how does it get there? Diabetes 63, 3992–3997, https://doi.org/10.2337/db14-0340 (2014).
Molnar, G. et al. GABAergic neurogliaform cells represent local sources of insulin in the cerebral cortex. J Neurosci 34, 1133–1137, https://doi.org/10.1523/JNEUROSCI.4082-13.2014 (2014).
Heydemann, A. An Overview of Murine High Fat Diet as a Model for Type 2 Diabetes Mellitus. Journal of diabetes research 2016, 2902351, https://doi.org/10.1155/2016/2902351 (2016).
Kristiansen, O. P. & Mandrup-Poulsen, T. Interleukin-6 and diabetes: the good, the bad, or the indifferent? Diabetes 54(Suppl 2), S114–124 (2005).
Ahmad, S. F. et al. Regulation of TNF-alpha and NF-kappaB activation through the JAK/STAT signaling pathway downstream of histamine 4 receptor in a rat model of LPS-induced joint inflammation. Immunobiology 220, 889–898, https://doi.org/10.1016/j.imbio.2015.01.008 (2015).
Grivennikov, S. I. & Karin, M. Dangerous liaisons: STAT3 and NF-kappaB collaboration and crosstalk in cancer. Cytokine & growth factor reviews 21, 11–19, https://doi.org/10.1016/j.cytogfr.2009.11.005 (2010).
Simon, A. R., Rai, U., Fanburg, B. L. & Cochran, B. H. Activation of the JAK-STAT pathway by reactive oxygen species. The American journal of physiology 275, C1640–1652 (1998).
Wilkin, T. J. The accelerator hypothesis: weight gain as the missing link between Type I and Type II diabetes. Diabetologia 44, 914–922, https://doi.org/10.1007/s001250100548 (2001).
Inzucchi, S. E. et al. Management of hyperglycemia in type 2 diabetes: a patient-centered approach: position statement of the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD). Diabetes care 35, 1364–1379, https://doi.org/10.2337/dc12-0413 (2012).
Lei, Y. et al. Metformin targets multiple signaling pathways in cancer. Chinese journal of cancer 36, 17, https://doi.org/10.1186/s40880-017-0184-9 (2017).
Sun, J. et al. Deciphering Signaling Pathway Networks to Understand the Molecular Mechanisms of Metformin Action. PLoS computational biology 11, e1004202, https://doi.org/10.1371/journal.pcbi.1004202 (2015).
Deng, X. S. et al. Metformin targets Stat3 to inhibit cell growth and induce apoptosis in triple-negative breast cancers. Cell Cycle 11, 367–376, https://doi.org/10.4161/cc.11.2.18813 (2012).
Hawley, S. A. et al. Use of cells expressing gamma subunit variants to identify diverse mechanisms of AMPK activation. Cell Metab 11, 554–565, https://doi.org/10.1016/j.cmet.2010.04.001 (2010).
Owen, M. R., Doran, E. & Halestrap, A. P. Evidence that metformin exerts its anti-diabetic effects through inhibition of complex 1 of the mitochondrial respiratory chain. The Biochemical journal 348(Pt 3), 607–614 (2000).
Jiang, Y. et al. Metformin plays a dual role in MIN6 pancreatic beta cell function through AMPK-dependent autophagy. International journal of biological sciences 10, 268–277, https://doi.org/10.7150/ijbs.7929 (2014).
Bloom, J. & Al-Abed, Y. MIF: mood improving/inhibiting factor? J Neuroinflammation 11, 11, https://doi.org/10.1186/1742-2094-11-11 (2014).
Oliveira, W. H. et al. Effects of metformin on inflammation and short-term memory in streptozotocin-induced diabetic mice. Brain Res 1644, 149–160, https://doi.org/10.1016/j.brainres.2016.05.013 (2016).
Dandona, P. et al. Increased plasma concentration of macrophage migration inhibitory factor (MIF) and MIF mRNA in mononuclear cells in the obese and the suppressive action of metformin. J Clin Endocrinol Metab 89, 5043–5047, https://doi.org/10.1210/jc.2004-0436 (2004).
Kaneb, H. M., Sharp, P. S., Rahmani-Kondori, N. & Wells, D. J. Metformin treatment has no beneficial effect in a dose-response survival study in the SOD1(G93A) mouse model of ALS and is harmful in female mice. PLoS One 6, e24189, https://doi.org/10.1371/journal.pone.0024189 (2011).
Moreira, P. I. Metformin in the diabetic brain: friend or foe? Annals of translational medicine 2, 54, https://doi.org/10.3978/j.issn.2305-5839.2014.06.10 (2014).
Hershey, T. et al. Hippocampal volumes in youth with type 1 diabetes. Diabetes 59, 236–241, https://doi.org/10.2337/db09-1117 (2010).
Lobnig, B. M., Kromeke, O., Optenhostert-Porst, C. & Wolf, O. T. Hippocampal volume and cognitive performance in long-standing Type 1 diabetic patients without macrovascular complications. Diabetic medicine: a journal of the British Diabetic Association 23, 32–39, https://doi.org/10.1111/j.1464-5491.2005.01716.x (2006).
Ennis, K., Tran, P. V., Seaquist, E. R. & Rao, R. Postnatal age influences hypoglycemia-induced neuronal injury in the rat brain. Brain Res 1224, 119–126, https://doi.org/10.1016/j.brainres.2008.06.003 (2008).
Goncalves, J. T., Schafer, S. T. & Gage, F. H. Adult Neurogenesis in the Hippocampus: From Stem Cells to Behavior. Cell 167, 897–914, https://doi.org/10.1016/j.cell.2016.10.021 (2016).
Mathew, B. et al. Role of migratory inhibition factor in age-related susceptibility to radiation lung injury via NF-E2-related factor-2 and antioxidant regulation. American journal of respiratory cell and molecular biology 49, 269–278, https://doi.org/10.1165/rcmb.2012-0291OC (2013).
Sauler, M., Bucala, R. & Lee, P. J. Role of macrophage migration inhibitory factor in age-related lung disease. American journal of physiology. Lung cellular and molecular physiology 309, L1–10, https://doi.org/10.1152/ajplung.00339.2014 (2015).
Miller, R. A. et al. Gene expression patterns in calorically restricted mice: partial overlap with long-lived mutant mice. Mol Endocrinol 16, 2657–2666, https://doi.org/10.1210/me.2002-0142 (2002).
Barzilai, N., Crandall, J. P., Kritchevsky, S. B. & Espeland, M. A. Metformin as a Tool to Target Aging. Cell Metab 23, 1060–1065, https://doi.org/10.1016/j.cmet.2016.05.011 (2016).
Gregor, M. F. & Hotamisligil, G. S. Inflammatory mechanisms in obesity. Annual review of immunology 29, 415–445, https://doi.org/10.1146/annurev-immunol-031210-101322 (2011).
Chung, K. J. et al. A self-sustained loop of inflammation-driven inhibition of beige adipogenesis in obesity. Nature immunology 18, 654–664, https://doi.org/10.1038/ni.3728 (2017).
Poser, S. W. & Androutsellis-Theotokis, A. Growing neural stem cells from conventional and nonconventional regions of the adult rodent brain. J Vis Exp, e50880, https://doi.org/10.3791/50880 (2013).
Gailus-Durner, V. et al. Introducing the German Mouse Clinic: open access platform for standardized phenotyping. Nature methods 2, 403–404, https://doi.org/10.1038/nmeth0605-403 (2005).
Fuchs, H. et al. Mouse phenotyping. Methods 53, 120–135, https://doi.org/10.1016/j.ymeth.2010.08.006 (2011).
Acknowledgements
This work was supported by the Helmholtz Alliance ICEMED-Imaging and Curing Environmental Metabolic Diseases, through the Initiative and Network Fund of Helmholtz Association Grant 051_40001, and the Deutsche Forschungsgemeinschaft Grant SFB 655, “Cells into tissues,” Project A24, by the German Federal Ministry of Education and Research to the German Center for Diabetes Research (DZD e.V.), and to the GMC (Infrafrontier grant 01KX1012). The funding sources had no involvement in study design, collection, analysis, and interpretation of data, writing the report, and in the decision to submit the article for publication. We thank Dr. Triantafyllos Chavakis for advice and help in this project.
Author information
Authors and Affiliations
Consortia
Contributions
P.N. conceived/designed the project, analyzed/interpreted data, collected/assembled data, wrote the manuscript; C.A.-F. collected/assembled data, provided administrative support; J.R., B.R., J.A.A.-P., G.M.C. Consortium collected/assembled data, analyzed/interpreted data; E.W. conceived/designed the clinical-chemical and hematological studies; M.K. conceived/designed the energy metabolism studies; M.O. and C.S.-W. conceived/designed the allergy screen; H.F. and V.G.-D. participated in the conception of the phenotyping study; M.H.d.A. conceived and designed the phenotyping experiments; A.C., I.K., G.C., C.-X.Y., C.G.C., M.H.T., S.R.B., analyzed/interpreted data; L.T., J.M., S.W.P., L.S.M., V.T., M.T., A.W., V.A. collected/assembled data; A.A.-T. conceived/designed the project, analyzed/interpreted data, wrote the manuscript. All authors approved the manuscript for publication.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
A comprehensive list of consortium members appears at the end of the paper
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Nikolakopoulou, P., Chatzigeorgiou, A., Kourtzelis, I. et al. Streptozotocin-induced β-cell damage, high fat diet, and metformin administration regulate Hes3 expression in the adult mouse brain. Sci Rep 8, 11335 (2018). https://doi.org/10.1038/s41598-018-29434-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-29434-2
This article is cited by
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.