Abstract
Because many of the negative effects of ocean acidification on marine life may result from underlying energetic short-falls associated with increased metabolic demands, several studies have hypothesized that negative responses to high CO2 could be reduced by energy input. Although this hypothesis was supported by a recent meta-analysis, we believe that the meta-analytic calculation used was not appropriate to test the stated hypothesis. Here, we first clarify the hypothesis put forward, the crux being that the effects of increased food supply and CO2 interact statistically. We then test this hypothesis by examining the available data in a more appropriate analytical framework. Using factorial meta-analysis, we confirm that food addition has a positive effect and CO2 has a negative effect on both growth and calcification. For calcification, food addition did indeed reduce CO2 impacts. Surprisingly, however, we found that food addition actually exacerbated the effects of acidification on growth, perhaps due to increased scope upon which CO2 effects can act in food-replete situations. These interactive effects were undetectable using a multilevel meta-analytic approach. Ongoing changes in food supply and carbonate chemistry, coupled with under-described, poorly understood, and potentially surprising interactive outcomes for these two variables, suggest that the role of food should remain a priority in ocean acidification research.
Arising from: L. Ramajo et al., Sci. Rep. 6: 19374 (2016).
Similar content being viewed by others
Introduction
Ocean acidification (OA) has been recognized as one of the most significant threats to marine life1, with demonstrated negative effects across many taxa in terms of growth and calcification2. In many cases, impairment by acidification reflects an underlying energetics problem, as organisms may need to redirect energy from growth and reproduction towards acid-base regulation, especially at sites of calcification3,4. Given that sub-lethal negative effects of OA can result from this energetic trade off, a number of studies have proposed the hypothesis that negative responses to OA could be minimized in situations where energy (e.g., food) is more readily available5. If true, this has far-reaching implications for how we both understand and study OA6,7. Although the energy limitation hypothesis is ecologically relevant and physiologically plausible, many of the recent tests of the idea suffer from a lack of adequately framed and precisely stated expectations regarding the combined effects of OA and food supply, or from inappropriate statistical comparisons among different experimental groups. Perhaps as a result of these framing of expectations, the most appropriate meta-analytical techniques have not yet been employed to seek generalities across studies. Here, We (1) clarify the hypothesis put forward by a recent meta-analysis (Ramajo et al.5) and others8,9,10,11,12,13,14,15,16,17,18,19, and (2) test the hypothesis using available data in a more appropriate meta-analytical framework.
The crux of the hypothesis that a negative response to OA by calcifiers could be modified by food availability lies in the idea that increased food supply does not simply offset some of the negative consequences of ocean acidification; rather, it implies a positive interactive effect of food and OA (sensu Morris et al.20). For this type of statistical interaction, the term ‘positive’ refers to the fact that the observed response to simultaneously elevated food and CO2 is more positive, or less negative, than the response predicted from the single-factor effects by a null, non-interactive model. The proper statistical test of the food limitation hypothesis would therefore be to estimate the statistical interaction between food supply and ocean acidification where the effect of OA on performance is stronger with limited food and weaker with abundant food. The distinction between this positive interactive scenario and two alternatives (non-interactive and negative interactive scenarios) is outlined in Fig. 1. The non-interactive scenario is one in which the effect of OA does not depend on food availability (Fig. 1a, slopes are equal). Here, we use the term ‘non-interactive’ to imply a null additive model in multilevel meta-analysis and a null multiplicative model in factorial meta-analysis. In the positive interactive scenario, the response of an organism to acidification is altered by food addition such that the negative effect of OA apparent under low food conditions is reduced or disappears when food supply is high (Fig. 1b, low food slope is more negative). Finally, in the negative interactive scenario, food addition actually promotes a stronger negative response to CO2 (Fig. 1c, high food slope is more negative). This last case is not widely predicted, but thus far the only example of a significant interaction between factorially manipulated CO2 and food supply is of this type (see Cole et al.21). These three hypotheses lead to how food supply alters the effect of OA (Fig. 1d–f) and, equivalently, how OA affects an animal’s response to food supplementation (Fig. 1g–i). Using a factorial meta-analysis, we can further test if a negative effect of CO2 and a positive effect of food supply can combine in a multiplicative way (i.e. no interaction, Fig. 1j) or in an interactive way, either where the interaction on average improves (Fig. 1k) or decreases performance relative to the non-interactive null expectation (Fig. 1l).
Schematic demonstrating hypothetical responses of organisms where high CO2 has a negative effect and high food supply has a positive effect on performance. Left-hand panels show a scenario where organismal response to high CO2 is not modified by food supply, demonstrating a non-interactive effect of food and OA on performance (a). Middle and right-hand panels depict interactive effects of food and ocean acidification on performance, where organismal response to high CO2 is modified by food supply in a positive (b) or negative (c) way. The outcomes of (a–c) could be realized across many studies in a meta-analysis, where the mean effect sizes (in Ln response ratio, LnRR) of OA may remain the same, regardless of food addition (d) or differ under food supplementation (e,f). In (d), while higher food may be beneficial and OA may be detrimental, a change in food supply does not alter the magnitude of the OA effect. Similarly, the effect of food supply could have no relationship with CO2 (g) or be influenced by change in CO2 (h,i). Finally, in a factorial meta-analysis, the interaction between food supply and OA could be non-significant (a simple multiplicative outcome, in this case (j), significantly positive (k), where the interaction food and OA on average improves the performance of organisms relative to the non-interactive case (compare b to a), or significantly negative (l), where the interaction between food and OA on average decreases performance of organisms relative to the non-interactive case (compare c to a).
Ramajo et al.5 set out to explore these possibilities using 12 studies that factorially manipulated OA and food supply. However, rather than comparing OA effects at high and low food supply (i.e. slopes of lines, Fig. 1a–c), Ramajo and colleagues compared both the high food acidified treatment (solid upward-pointing triangles, Fig. 1a,b) and the low food acidified treatment (solid downward-pointing triangles, Fig. 1a–c) to a single reference treatment: high food, non-acidified (open upward-pointing triangles, Fig. 1a–c). This comparison using a common denominator is atypical in the meta-analysis literature, where, in subgroup analyses, independent effect sizes are computed within subgroups22. Because the effect size of acidification was not calculated independently for each subgroup (level of food supply) by Ramajo et al.5, the response ratios (lnRR) presented for low and intermediate food supply cannot be considered true effect sizes and thus cannot be used to compare if the effect of CO2 changes under food supply levels. Therefore, the authors inadvertently tested a substantially different hypothesis, namely that when food is abundant, increasing CO2 has a small effect, but that simultaneous exposure to OA and food deprivation has a large negative impact relative to well-fed, non-acidified controls. Because the analysis did not include any data from organisms under ambient CO2 conditions and low or intermediate food supply (open down-facing triangles, Fig. 1a–c), the analysis cannot determine if or how the response to OA changes with food supply (i.e. cannot distinguish between the interactive vs. non-interactive scenarios outlined in Fig. 1).
To clarify the relationship between OA effects and food supply, we re-examined the available literature using two different types of meta-analysis: multilevel meta-analysis (employed by Ramajo et al.5 and others23 and factorial meta-analysis20,24,25. Within each of these types of meta-analysis we used three different datasets to test the hypothesis that the response to OA is modified by food supply. We first created an updated dataset by extracting data from the eleven published studies in Ramajo et al.5 and six additional studies11,19,21,26,27,28 (see Supplementary Information, Table S1). In order to compare these results to findings in Ramajo et al.5, we next replicated their dataset twice, first including the incorrect log response ratio (LnRR) calculation along with other errors in the dataset, and secondly by correcting the LnRR calculation in order to demonstrate the impact that this miscalculation makes on the results and inferences (see Supplementary Information).
Results and Discussion
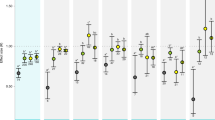
Using the updated dataset, we calculated log-response ratio (LnRR) of the effect of CO2 using response to OA/control (solid vs. open symbols, Fig. 1a,b) where control was low CO2 conditions under the same food supply level as the OA treatment. We found non-interactive effects of OA and food supply for both growth and calcification. In other words, the response to OA did not depend on food supply (calcification QM(coef) = 1.84, P = 0.17, Fig. 2a; growth QM(coef) = 1.31, P = 0.25, Fig. 2b). Furthermore, restricting this analysis to the data and methods used by Ramajo et al.5 did not alter our conclusions (calcification QM(coef) = 0.60, P = 0.44, Fig. 3b, and growth QM(coef) = 0.28, P = 0.61, Fig. S2d). With regard to the data plotted in Figs 2a and 3b, we remind readers that a statistical difference cannot be inferred between a significant result and a non-significant result29, although it is a common mistake to draw this conclusion when the confidence interval crosses zero in one case but not the other. In other words, a difference in statistics does not equate to a statistical difference.
Mean effect sizes (LnRR) for the responses of calcification (left hand panels) and growth (right hand panels) to food supply and elevated CO2. Mean responses to elevated CO2 did not depend on food supply for (a) calcification (QM(coef) = 1.84, P = 0.17) nor (b) growth (QM(coef) = 1.31, P = 0.25). Across food treatments, there was a significant negative response to high CO2 for (a) calcification (QM = 6.56, P = 0.037) but not (b) growth (QM = 1.33, P = 0.52). Response to high food was not different between elevated and ambient CO2 levels for (c) calcification (QM(coef) = 2.62, P = 0.11) or (d) growth (QM(coef) = 1.64, P = 0.20) responses, nor did response to high food supply depend on continuous CO2 for (e) calcification (QMslope = 1.72, P = 0.19, slope = 0.0001) or (f) growth (QMslope = 0.48, P = 0.49, slope = 0.00). Across OA levels, food had no effect (but trending positive) on (c) calcification (QM = 5.33, P = 0.069) and a significant positive effect on (d) growth (QM = 20.59, P < 0.0001). Across a continuous CO2 gradient, food had a significant positive effect on (f) growth (QM(intercept) = 15.74, P < 0.0001), but not (e) calcification (QM(intercept) = 3.19, P = 0.074) responses. Parameter estimates and confidence intervals from fitted REML models. Symbols represent the treatment in each study from which the LnRR was calculated (open vs. closed circles for ambient and elevated CO2, respectively). Numbers in brackets indicate the number of studies contributing to the LnRR; for (e) and (f), n = 11 and n = 15, respectively.
Factorial analysis. Overall and interactive effect sizes for CO2 and food supply for (a) calcification and (b) growth responses. (a) Here, we found that overall, CO2 had a significant negative effect on calcification (LnRR = −0.26, lower = −0.29, upper = −0.23), whereas food addition had an overall significant positive effect on calcification (LnRR = 1.07, lower = 1.03, upper = 1.11). The interaction between CO2 and food supply was significantly positive for calcification (LnRR = 0.13, lower = 0.07, upper = 0.19), meaning that OA effects were weaker (less negative) at high food. (b) For growth responses, CO2 had a small but significant negative effect (LnRR = −0.020, lower = −0.026, upper = −0.013), food had a positive effect (LnRR = 0.57, lower = 0.56, upper = 0.59). The interaction between CO2 and food supply was small but significantly negative for growth (LnRR = −0.024, lower = −0.035, upper = −0.012), meaning that OA effects were stronger (more negative) at high food. Error bars are 95% confidence intervals calculated from standard error for small sample sizes. If the error bars do not overlap zero then a significant response is inferred. Numbers in brackets indicate the number of studies contributing to the LnRR.
Across food treatments, we detected an overall significant negative response to high CO2 for calcification-related responses (QM = 6.56, P = 0.037, Fig. 2a). For calcification, the effect size of OA was positively related to the experimental change in CO2 (QM = 10.79, P = 0.0045, Fig. S1a). In contrast, overall growth responses to OA were less variable, did not differ from zero (QM = 1.33, P = 0.52, Fig. 2b), and did not change with increasing experimental change in CO2 (QM = 2.37, P = 0.30 Fig. S1b). These results concur with a recent meta-analysis showing that calcification responses to OA across many taxa were stronger than growth responses2. The non-significant growth response to OA found here suggest either that our analysis represents an insufficient sample size to detect a true effect or that the species tested were relatively resistant to acidification compared to responses reported elsewhere2.
Across CO2 levels, food supply had no effect (but trending positive) on calcification (OA: QM = 5.33, P = 0.069, Fig. 2c; continuous CO2: QM(intercept) = 3.19, P = 0.074, Fig. 2e) and a strong positive effect on growth (OA: QM = 20.59, P < 0.0001, Fig. 2d; continuous CO2: QM(intercept) = 15.74, P < 0.0001, Fig. 2f). The response to high food supply did not depend on OA manipulation or continuous CO2 level for growth (OA: QM(coef) = 1.64, P = 0.20, Fig. 2d; continuous CO2: QM = 0.48, P = 0.49, Fig. 2f) or calcification responses (OA: QM(coef) = 2.62, P = 0.11, Fig. 2c; continuous CO2: QM = 1.73, P = 0.19, Fig. 2e). These results suggest that growth may be more tightly linked to food supply than OA, while OA may be relatively more important for calcification.
As an alternative approach, we next employed a factorial meta-analysis on the same data to calculate overall effects of CO2 and food addition and their interaction. This analysis is a powerful tool when using fully factorial datasets (i.e. each experiment manipulated both OA and food supply)20,24,25. Here, we confirm that CO2 has a significant negative effect on calcification (LnRR = −0.26, lower = −0.29, upper = −0.23, Fig. 3a), as found in Fig. 2a. For growth in this analysis we were able to detect a small, significant negative effect of CO2 (LnRR = −0.020, lower = −0.026, upper = −0.013, Fig. 3), whereas we were unable to detect this effect using multilevel meta-analysis (Fig. 2b). In this analysis, we found strong significant overall effects of food on both calcification (LnRR = 1.07, lower = 1.03, upper = 1.11, Fig. 3a) and growth (LnRR = 0.57, lower = 0.56, upper = 0.59, Fig. S2b), whereas previously we found a significant effect on growth only and a positive trend for calcification (Fig. 2c–f). When analyzed factorially, we found that the interaction between OA and food supply had a small and significant positive effect on calcification (LnRR = 0.13, lower = 0.07, upper = 0.19, Fig. 3a). This result provides support for the positive interaction hypothesis that food addition can ameliorate response to CO2 (Fig. 1b). For growth, we found support for the opposite hypothesis, where the interaction between food supply and CO2 had a small but significant negative interactive effect on growth (LnRR = −0.024, lower = −0.035, upper = −0.012, Fig. 3b). This result implies that negative CO2 effects are actually more severe at high food concentrations, perhaps because high food conditions support a larger scope for the effect of CO2 to act on (Fig. 1c).
When we restricted the factorial analysis to the data used by Ramajo et al.5, we also found negative effects of CO2 and positive effects of food on both calcification and growth responses. In addition, the interactive effect of food supply and OA on calcification was of similar magnitude (LnRR = 0.10, lower = 0.043, upper = 0.15, Fig. S3b). However, using only data from Ramajo et al.5, we found that the interactive effect of CO2 and food supply on growth was slightly positive (LnRR = 0.03, lower = 0.015, upper = 0.048, Fig. S3d), which is in the opposite direction to the same analysis using more data (Fig. 3b). However, a change in sign of the interactive effect is an indication of extraction errors in the original Ramajo et al.5 dataset (Table S1), as re-analysis without extraction errors yields a non-interactive effect. Therefore, the negative interaction effect on growth is only detectable with a larger sample size.
Overall, there was no consistent tendency for the effects of OA on growth and calcification to be modified in the same way by food supply. We found that for calcification there is evidence that positive effects of food supply do mediate negative response to acidification. For growth responses, in contrast, we found a significant negative interactive effect using factorial analysis, and no response using multilevel meta-analysis. Although both statistically sound methods, factorial analysis likely has more power to accurately estimate the effect size of the interaction and is particularly useful for understanding the growing list of multiple stressor studies30,31. The findings of the multilevel analyses align more with the findings of the individual studies making up the meta-analysis, where the interaction between food and OA was non-significant in 21 of 22 cases, although, it should be noted that the single significant interactive case revealed an increased OA effect on growth at higher food rather than a decreased one21 (consistent with Fig. 1c and findings of factorial meta-analysis on growth). The number of studies considered here and by Ramajo et al.5 is relatively low, although neither sensitivity nor bias analyses indicated cause for concern (see methods for details). It is also worth noting that some of the included studies did find interactive effects for other response variables where negative responses to OA were ameliorated under high food, including metabolic rate17, aragonite crystal length15 and, frequency of normal development21, suggesting food supply could be important in mediating other physiological and developmental responses to OA beyond calcification and growth responses.
There may be physiological mechanisms, yet to be explored, for the various hypotheses described above. Mechanistically, for added food to reduce the negative impact of OA (i.e., a positive interactive outcome), either feeding rates would need to increase to compensate for higher energetic demands, or energy acquisition from ingested food would need to be more efficient. The reverse would need to be true to support the negative interactive outcome, where OA impacts are most severe at high food supply. Currently, there is little evidence to support compensatory feeding under acidification. In fact, feeding and clearance rates have been shown to decline under acidification across a wide range of taxa, both herbivorous and carnivorous, encompassing a variety of feeding appendages and behaviours32, which may help to explain the negative interactive effect we found for growth. Similarly, available evidence does not suggest that increased assimilation efficiency is likely to be common under high CO233,34,35. From a physiological standpoint, therefore, the mechanisms underlying the significant positive interactive effects on calcification described here are yet to be pinpointed. It should be noted, however, that reductions in search and handling times and increases in palatability under acidification36,37 could influence an organism’s energetic balance in such a way that OA costs are minimized by higher food availability, but most published experiments – including those in our meta-analysis – do not allow for these types of longer-term effects to emerge. Finally, it is important to recognize that food supply could mediate OA effects in a non-interactive scenario for one response variable (i.e. maximum metabolic rate), but appear interactive for another variable if the combined effects of OA and food limitation crosses some important physiological threshold (e.g., metabolic supply becomes insufficient to meet demand and the animal dies) or ecological threshold (e.g., population intrinsic growth rate, λ, drops below 1)38. To the best of our knowledge, these scenarios have not been explicitly tested.
Moving forward, we recommend that researchers take great care when framing and testing hypotheses related to the effects of food availability on responses to OA. The individual studies in this meta-analysis overwhelmingly found non-interactive effects, yet several authors use phrasing such as “food supply reduces the impacts of experimental OA”5, “failure to provide food can increase vulnerability to OA in experimental assessments”5, “food supply… can mitigate the negative impacts of future OA”12, “feeding and energy availability can mediate reductions in growth due to OA stress”18, and “Zooplanktivory ameliorates the effects of ocean acidification”8. Others have pointed out this discrepancy, for example Drenkard et al.16 comment on the misclassification of non-interactive results as interactive by Edmunds8 and underscore that, despite misinterpretation, both of their datasets confirm that energetic status does not alter calcification sensitivity to ocean acidification. The authors of the above papers may have intended to convey that the negative effects of OA at low food availability can be offset by supplying more food. We believe the language used above could equally be interpreted as a modification of the effect of OA (i.e. an interactive effect) or an offsetting of the effect of OA (i.e. a non-interactive effect). Careful and deliberate consideration of language used to describe experimental results can help guide further mechanistic experiments to enhance our understanding of ocean acidification responses.
Many of the studies cited here were driven by observations in the field where ecosystems are observed to be healthy in the presence of high food despite acidic conditions9,12,39. In the absence of a non-acidic, high food ‘control’, however, it isn’t clear if acidic conditions are truly having no effect, or if there is simply no difference between non-acidified, low food situations and acidified, high food situations. Of course, this does not rule out the broader contention that adding more food will allow organisms to maintain performance under increasing acidification; indeed, that outcome would be expected from a non-interactive scenario (compare the open downward-facing triangle to the filled upward-facing triangle in Fig. 1a). However, if this argument is applied to global change scenarios generally, it would be akin to comparing a present-day ocean with little food to a future ocean with an abundance of food. While it is certainly possible that OA can increase food availability (e.g., for some herbivores7), this will not be the case for all species, nor in all habitats, and the opposite can also be true40. Unless changes in food supply through time are known in advance, the proper comparison is the effect of OA (elevated vs. ambient CO2) at low food supply compared to the effect of OA (elevated vs. ambient CO2) at high food supply (Fig. 1a–c solid and dashed lines, respectively). Overall, our results suggest that the ultimate role of food as a modifying influence on ecological responses to ocean acidification will depend on the relative importance of calcification and growth to fitness and population, community, and ecosystem dynamics. Further, we suggest that future changes in ocean acidity could affect systems of both high and low productivity and food availability, but the ecological effects may be difficult to predict without knowledge of the ways in which acidification indirectly affects food supply.
Understanding the distinction between interactive and non-interactive effects is important for furthering our mechanistic understanding of ocean acidification. If these effects are indeed non-interactive (as suggested by multilevel meta-analysis), we can make specific predictions about the effect of OA separately from the effect food, allowing us to have somewhat more confidence in cautiously predicting OA effects for food supply conditions not directly considered in the original studies. However, the negative interactive effect on growth (found in the factorial analysis) suggests that ecological contexts in which food is super-abundant may be more susceptible to OA effects than more food-limited systems, and the converse is true in terms of calcification. The presence of mixed interactive effects prevents us from generalizing broadly from a limited set of results as the outcome of one effect cannot be predicted independently from the other and suggests that future OA impacts may vary meaningfully from one context to another.
Conclusions
Overall, we provide evidence that food supply both mitigates and worsens the negative effects of OA, depending on the response variable. Clearly, there remains much work to be done on this topic. We agree with Ramajo et al.5 that researchers should, where possible, explicitly relate food supply levels to natural conditions (e.g.9,16) and organismal requirements, as interactive effects of food supply and OA could emerge when crossing ecological thresholds. Furthermore, if and when “low food” treatment levels equate to starvation of the organism, researchers should consider that other energetic processes, such as metabolic depression, are likely to become important and this could change the response to OA. More studies are needed to determine the full extent to which responses to OA change across a wide range of food supply levels and feeding modes. The careful selection of natural field-relevant food supply levels can help identify when responses to OA are likely to cross ecological thresholds, which can generate interactive outcomes at higher levels of biological organization even when the underlying effects are non-interactive. Given that both food supply and marine carbonate chemistry are changing simultaneously through time, a more detailed understanding of the inter-relationship of their effects, both physiological and ecological, remains a research priority.
Methods
Two meta-analytic techniques were explored to test the hypotheses posed above. The first, multilevel meta-analysis, uses ln response ratios of the effect of CO2 (comparing across food levels) and the effect of food (comparing across CO2 levels). The second technique, factorial meta-analysis, is suitable for papers that employ fully-factorial experiments. The analysis used by Ramajo et al.5, specifically their calculation of effect size, was not appropriate for the stated hypothesis and, this mismatch calls their conclusion into question. We have therefore replicated results from Ramajo et al.5, using both types of meta-analysis. These methods can be found in the Supplementary Information.
Multilevel meta-analysis
We extracted data on growth and calcification responses to CO2 from 17 articles in which both food supply and CO2 were manipulated. (Figure 2) All data (68 observations for calcification measures, 144 for growth measures) were extracted from figures using WebPlotDigitizer41. The papers used in the multilevel analysis were all the published articles from Ramajo et al.5 and six additional articles found during an ISI Web of Knowledge search using similar search terms (Supplementary Information, Table S1). Here, we used the width variable from Taylor et al.26 because this is the most commonly used measure of urchin growth in both fisheries and population-level literature. Furthermore, Taylor et al.26 was only used for the LnRR effect of CO2 analysis but not for the effect of food analysis since negative and positive values cannot be compared using LnRR. We only used the aggregate ambient CO2 level described in Maier et al.19 and only one elevated CO2 level (intermediate) because, as above, LnRR cannot evaluate dissolution at the same time as calcification. We added a constant (0.5%) to all percentage growth measurements in Büscher et al.11 since values close to zero approach infinity when taking ln(0).
Meta-analytic calculations and statistics were performed in R (Version 3.3.1, R Development Core Team42), using the metafor package43. We calculated a weighted ln Response Ratio (lnRR) using OA/Control within each food supply level (escalc function, metafor package in R42,43). We constructed multilevel models with food supply as a fixed effect and aspects of non-independence as random effects using the rma.mv function in R43 and checked these models for publication bias (using a contour-enhanced funnel plot44) and sensitivity to outliers (following methods in Habbeck et al.45). We used restricted maximum likelihood (REML) approaches to test if effect size estimates were significantly different than zero and significantly different between food supply and CO2 levels. Some levels of CO2 were different between food levels within an experimental treatment, therefore we averaged between food levels to obtain a single CO2 level for each low food-high food pair to regress against the LnRR effect of food. We excluded intermediate food levels because the sample size was too small to make meaningful comparisons. The data used included several non-independent measures: common control for multiple levels of CO2, multiple variables for same response (growth responses only), and multiple independent studies within the same article. We accounted for dependent sampling errors caused by using a common control for multiple levels of CO2 by constructing a variance-covariance matrix of the effect size estimates46. We accounted for multiple variables for the same response and multiple independent studies within an article by including an unstructured random effect of response variable unit (of the form ~Unit|Paper_no) and food supply (of the form ~Food.supply|Paper_no) which allows the random effects to have different variances for each outcome while also allowing random effects to be correlated47. We plotted estimates for the mean and confidence intervals generated from the rma.mv models. For the calculated LnRR of food supply (high food/control, low food), we used data from all CO2 levels, therefore there was an unequal number of observations in each category. To account for this, we included an unstructured random effect for CO2 level (of the form ~ CO2_level|Paper_no) in addition to an unstructured random effect of response variable unit (of the form ~Unit|Paper_no). We tested all models first for an overall effect of the moderator (QM), food supply or CO2, and then tested between levels of the moderator – i.e. testing for the interaction between food and CO2 (QMcoef), e.g., High vs. Low food or Ambient vs. Elevated CO2. These tests of heterogeneity described by the moderators (QM tests) are akin to a Wald-type Chi-squared test43. When there was only a single moderator (as in Fig. 2e,f), the effect of the moderator (e.g., CO2) is a test of the slope vs. zero (QMslope) and the overall response (regardless of moderator) is a test of the intercept vs. zero (QMintercept).
Factorial Meta-analysis
For this analysis, we used the same data as the multilevel analysis (Fig. 2), with one exception: Taylor et al.26 was excluded because we cannot take the ln of negative values (as above). (Figure 3) In order to account for multiple CO2 levels per paper, we averaged effect sizes across a single paper, as factorial LnRR are not appropriate for constructing a variance-covariance matrix to account for multiple comparisons to the same CO2 control. We accounted for multiple responses per paper in this analysis in the same way. Multiple study sites within papers were considered to be independent enough to consider separately.
We calculated ln response ratios for overall and interactive effects of food limitation and high CO2, using factorial meta-analysis methods20,24,25. Overall effects incorporate the effects of the interacting factor at all levels. In this analysis, we assume effects (food supply and high CO2) are multiplicative, which is a more biologically realistic null model than simply additive (i.e. “you can’t die twice”48). We calculated overall and interactive effect sizes and sampling variance from equations Appendix B.7 to B.12 in Morris et al.20. We weighted effect sizes by inverse sampling variance using a random effects model where weights were assigned using within study variance plus between studies variance49 and corrected for the small number of studies using equation 7 in Hedges et al.50. We constructed 95% confidence intervals using equation 8 in Hedges et al.50 and inferred significance from these confidence intervals. If the interaction effect is not different than zero (CIs overlap zero), then we can infer multiplicative effects20. If otherwise, we infer significant interactive effects: if the interaction effect is positive (above zero), then the interaction between the two factors has a positive effect on performance relative to the simple multiplicative case; if negative (below zero), we infer the interaction of the two factors has a negative effect on performance relative to the multiplicative scenario20,51,52,53,54,55.
Change history
10 July 2018
A correction to this article has been published and is linked from the HTML and PDF versions of this paper. The error has been fixed in the paper.
References
Wittmann, A. C. & Pörtner, H.-O. Sensitivities of extant animal taxa to ocean acidification. Nat. Clim. Chang. 3, 995–1001 (2013).
Kroeker, K. J. et al. Impacts of ocean acidification on marine organisms: quantifying sensitivities and interaction with warming. Glob. Chang. Biol. 19, 1884–1896 (2013).
Sokolova, I. M., Frederich, M., Bagwe, R., Lannig, G. & Sukhotin, A. A. Energy homeostasis as an integrative tool for assessing limits of environmental stress tolerance in aquatic invertebrates. Mar. Environ. Res. 79, 1–15 (2012).
Stumpp, M., Trübenbach, K., Brennecke, D., Hu, M. Y. & Melzner, F. Resource allocation and extracellular acid–base status in the sea urchin Strongylocentrotus droebachiensis in response to CO2 induced seawater acidification. Aquat. Toxicol. 110–111, 194–207 (2012).
Ramajo, L. et al. Food supply confers calcifiers resistance to ocean acidification. Sci. Rep. 6, 19374 (2016).
Rossoll, D. et al. Ocean acidification-induced food quality deterioration constrains trophic transfer. Plos One 7, e34737 (2012).
Gaylord, B. et al. Ocean acidification through the lens of ecological theory. Ecology 96, 3–15 (2015).
Edmunds, P. J. Zooplanktivory ameliorates the effects of ocean acidification on the reef coral Porites spp. Limnol. Oceanogr. 56, 2402–2410 (2011).
Thomsen, J. J., Casties, I., Pansch, C., Körtzinger, A. & Melzner, F. Food availability outweighs ocean acidification effects in juvenile Mytilus edulis: laboratory and field experiments. Glob. Chang. Biol. 19, 1017–1027 (2013).
Ramajo, L. et al. Biomineralization changes with food supply confer juvenile scallops (Argopecten purpuratus) resistance to ocean acidification. Glob. Chang. Biol. 22, 2025–2037 (2015).
Büscher, J. V., Form, A. U. & Riebesell, U. Interactive effects of ocean acidification and warming on growth, fitness and survival of the cold-water coral Lophelia pertusa under different food availabilities. Front. Mar. Sci. 4, 101 (2017).
Pansch, C., Schaub, I., Havenhand, J. N. & Wahl, M. & Sciences, B. Habitat traits and food availability determine the response of marine invertebrates to ocean acidification. Glob. Chang. Biol. 20, 765–777 (2014).
Hettinger, A. et al. The influence of food supply on the response of Olympia oyster larvae to ocean acidification. Biogeosciences 10, 6629–6638 (2013).
Comeau, S., Carpenter, R. C. & Edmunds, P. J. Effects of feeding and light intensity on the response of the coral Porites rus to ocean acidification. Mar. Biol. 160, 1127–1134 (2013).
Crook, E. D., Cooper, H., Potts, D. C., Lambert, T. & Paytan, A. Impacts of food availability and pCO2 on planulation, juvenile survival, and calcification of the azooxanthellate scleractinian coral Balanophyllia elegans. Biogeosciences 10, 7599–7608 (2013).
Drenkard, E. J. et al. Calcification by juvenile corals under heterotrophy and elevated CO2. Coral Reefs 32, 727–735 (2013).
Pan, T.-C. F., Applebaum, S. L. & Manahan, D. T. Experimental ocean acidification alters the allocation of metabolic energy. Proc. Natl. Acad. Sci. 112, 4696–4701 (2015).
Towle, E. K., Enochs, I. C. & Langdon, C. Threatened Caribbean coral is able to mitigate the adverse effects of ocean acidification on calcification by increasing feeding rate. Plos One 10, 1–17 (2015).
Maier, C. et al. Effects of elevated pCO2 and feeding on net calcification and energy budget of the Mediterranean cold-water coral Madrepora oculata. J. Exp. Biol. 2, jeb.127159 (2016).
Morris, W. F. et al. Direct and indirect effects of enemies and mutualists on plant performance: a meta-analysis. Ecology 88, 1021–1029 (2007).
Cole, V. J. et al. Effects of multiple climate change stressors: ocean acidification interacts with warming, hyposalinity, and low food supply on the larvae of the brooding flat oyster Ostrea angasi. Mar. Biol. 163, 125 (2016).
Borenstein, M., Hedges, L. V., Higgins, J. P. T. & Rothstein, H. R. In Introduction to Meta-Analysis 239–242 (John Wiley and Sons Ltd., 2009).
Konstantopoulos, S. Fixed effects and variance components estimation in three-level meta-analysis. Res. Synth. Methods 2, 61–76 (2011).
Gurevitch, J., Morrison, J. A. & Hedges, L. V. The interaction between competition and predation: a meta-analysis of field experiments. Am. Nat. 155, 435–453 (2000).
LaJeunesse, M. J. On the meta-analysis of response ratios for studies with correlated and multi-group designs. Ecology 92, 2049–2055 (2011).
Taylor, J. R. et al. Physiological effects of environmental acidification in the deep-sea urchin Strongylocentrotus fragilis. Biogeosciences 11, 1413–1423 (2014).
Swezey, D. S. et al. Interactive effects of temperature, food and skeletal mineralogy mediate biological responses to ocean acidification in a widely distributed bryozoan. Proc. R. Soc. B 284, 20162349 (2017).
Hurst, T. P., Laurel, B. J., Hanneman, E., Scott, A. & Ottmar, M. L. Elevated CO2 does not exacerbate nutritional stress in larvae of a Pacific flatfish. Press 1–21, https://doi.org/10.1111/fog.12195 (2017).
Gelman, A. & Stern, H. The difference between “significant” and “not significant” is not itself statistically significant. Am. Stat. 60, 328–331 (2006).
Crain, C., Kroeker, K. & Halpern, B. S. Interactive and cumulative effects of multiple human stressors in marine systems. Ecol. Lett. 11, 1304–1315 (2008).
Piggott, J. J., Townsend, C. R. & Matthaei, C. D. Reconceptualizing synergism and antagonism among multiple stressors. Ecol. Evol. 5, 1538–1547 (2015).
Clements, J. C. Meta-analysis reveals taxon- and life stage-dependent effects of ocean acidification on marine calcifier feeding performance. bioRxiv 1–18 (2016).
Xu, X., Yang, F., Zhao, L. & Yan, X. Seawater acidification affects the physiological energetics and spawning capacity of the Manila clam Ruditapes philippinarum during gonadal maturation. Comp. Biochem. Physiol. -Part A Mol. Integr. Physiol. 196, 20–29 (2016).
Wang, Y., Li, L., Hu, M. & Lu, W. Physiological energetics of the thick shell mussel Mytilus coruscus exposed to seawater acidification and thermal stress. Sci. Total Environ. 514, 261–272 (2015).
Sui, Y. et al. Combined effects of short-term exposure to elevated CO2 and decreased O2 on the physiology and energy budget of the thick shell mussel Mytilus coruscus. Chemosphere 155, 207–216 (2016).
Duarte, C. et al. Ocean acidification induces changes in algal palatability and herbivore feeding behavior and performance. Oecologia 180, 453–462 (2016).
Cripps, G., Flynn, K. J. & Lindeque, P. K. Ocean acidification affects the phyto-zoo plankton trophic transfer efficiency. Plos One 11, e0151739 (2016).
Kroeker, K. J., Kordas, R. L. & Harley, C. D. G. Embracing interactions in ocean acidification research: Confronting multiple stressor scenarios and context dependence. Biol. Lett. 13, 1–4 (2017).
Melzner, F. et al. Food supply and seawater pCO2 impact calcification and internal shell dissolution in the blue mussel Mytilus edulis. PLoS One 6 (2011).
Polovina, J. J., Howell, E. A. & Abecassis, M. Ocean’s least productive waters are expanding. Geophys. Res. Lett. 35, 2–6 (2008).
Rohatgi, A. Web Plot Digitizer (2015).
R Development Core Team. R: a language and environment for statistical computing (2009).
Viechtbauer, W. Conducting meta-analyses in R with the metafor package. Statiscal Softw. 36, 1–48 (2010).
Peters, J. L., Sutton, A. J., Jones, D. R., Abrams, K. R. & Rushton, L. Contour-enhanced meta-analysis funnel plots help distinguish publication bias from other causes of asymmetry. J. Clin. Epidemiol. 61, 991–996 (2008).
Habeck, C. W. & Schultz, A. K. Community-level impacts of white-tailed deer on understory plants in North American forests: a meta-analysis. AoB Plants 7, plv119 (2015).
Gleser, L. J. & Olkin, I. Stochastically dependent effect sizes. The handbook of research synthesis and meta-analysis (2nd ed.), 357–376 (2009).
Berkey, C. S., Hoaglin, D. C., Antczak-Bouckoms, A., Mosteller, F. & Colditz, G. A. Meta-analysis of multiple outcomes by regression with random effects. Stat. Med. 17, 2537–2550 (1998).
Sih, A., Englund, G. & Wooster, D. Emergent impacts of multiple predators on prey. Trends Ecol. Evol. 13, 350–355 (1998).
Borenstein, M., Hedges, L. V., Higgins, J. P. T. & Rothstein, H. R. Introduction to Meta-Analysis, https://doi.org/10.1002/9780470743386 (John Wiley & Sons, 2009).
Hedges, L. V., Gurevitch, J. & Curtis, P. S. The meta-analysis of response ratios in experimental ecology. Ecology 80, 1150–1156 (1999).
Hall-Spencer, J. M. & Allen, R. The impact of CO2 emissions on ‘nuisance’ marine species. Res. Reports Biodivers. Stud. 4, 33–46 (2015).
Harley, C. D. et al. Effects of climate change on global seaweed communities. J. Phycol. 48, 1064–1078 (2012).
Kroeker, K. J., Kordas, R. L., Crim, R. N. & Singh, G. G. Meta-analysis reveals negative yet variable effects of ocean acidification on marine organisms. Ecol. Lett. 13, 1419–1434 (2010).
Lortie, C. J. & Callaway, R. M. Re-analysis of meta-analysis: Support for the stress-gradient hypothesis. J. Ecol. 94, 7–16 (2006).
Higgins, J. PT. & Green, S. Cochrane handbook for systematic reviews of interventions (Wiley, 2008).
Acknowledgements
We first thank Laura Ramajo and colleagues for their paper and ideas on this important topic. We are further grateful to these authors for supplying us with their data and for discussions regarding the data and interpretation. We thank Jenn Sunday for her thoughtful discussions and comments on early drafts. We thank Mary O’Connor and five anonymous reviewers for their helpful revisions.
Author information
Authors and Affiliations
Contributions
All authors contributed to the idea and design of the paper; N.B., J.B., K.A. extracted the data; N.B. and J.B. managed the database, analyzed the data, and produced figures; N.B. drafted the paper; all authors contributed to discussions, writing, and interpretation.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Brown, N.E.M., Bernhardt, J.R., Anderson, K.M. et al. Increased food supply mitigates ocean acidification effects on calcification but exacerbates effects on growth. Sci Rep 8, 9800 (2018). https://doi.org/10.1038/s41598-018-28012-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-28012-w
This article is cited by
-
Impacts of ocean acidification under multiple stressors on typical organisms and ecological processes
Marine Life Science & Technology (2020)
-
Adult Antarctic krill proves resilient in a simulated high CO2 ocean
Communications Biology (2018)
-
Reply to ‘Increased food supply mitigates ocean acidification effects on calcification but exacerbates effects on growth’
Scientific Reports (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.