Abstract
The peripheral sensory system is critical to regulating motor plasticity and motor recovery. Peripheral electrical stimulation (ES) can generate constant and adequate sensory input to influence the excitability of the motor cortex. The aim of this proof of concept study was to assess whether ES prior to each hand function training session for eight weeks can better improve neuromuscular control and hand function in chronic stroke individuals and change electroencephalography-electromyography (EEG-EMG) coherence, as compared to the control (sham ES). We recruited twelve subjects and randomly assigned them into ES and control groups. Both groups received 20-minute hand function training twice a week, and the ES group received 40-minute ES on the median nerve of the affected side before each training session. The control group received sham ES. EEG, EMG and Fugl-Meyer Assessment (FMA) were collected at four different time points. The corticomuscular coherence (CMC) in the ES group at fourth weeks was significantly higher (p = 0.004) as compared to the control group. The notable increment of FMA at eight weeks and follow-up was found only in the ES group. The eight-week rehabilitation program that implemented peripheral ES sessions prior to function training has a potential to improve neuromuscular control and hand function in chronic stroke individuals.
Similar content being viewed by others
Introduction
Stroke is one of the leading contributing factors to the loss of functional abilities and independence in daily life in adults1. The most common and widely observed impairment following stroke is motor impairment, which can be regarded as a loss or limitation of function in muscle control or movement2,3,4,5. Most stroke survivors later regain the ability to walk independently, but only fewer than 50% of them will have fully recovered upper extremity functions6,7. From a review focusing on motor recovery after stroke, it has been indicated that the recovery of both arm and hand function among subacute and chronic stroke survivors is limited in current neural rehabilitation settings4; therefore, additional management with activating plasticity before or during performing motor training is necessary for better motor recovery.
The fundamental principle of stroke rehabilitation is inducing brain plasticity by sensory or proprioceptive input in order to facilitate motor functions8,9. It has been demonstrated that strong sensory input can induce plastic changes in the motor cortex via direct or indirect pathways10,11,12,13,14,15,16,17. In this case, electrical stimulation (ES) that provides steady and adequate somatosensory input can be an ideal method of stimulating the motor cortex.
Recent studies using functional magnetic resonance imaging (fMRI) or transcranial magnetic stimulation (TMS) suggest that ES on peripheral nerves can increase motor-evoked potential (MEP)18,19,20, increase the active voxel count in the corresponding motor cortex13, and increase blood-oxygen-level dependent (BOLD) signals in fMRI, suggesting peripheral ES induced higher excitability and activation level of cortical neurons21. Since the expansion of the motor cortical area or increase in the excitability of neural circuits is associated with learning new motor skills22,23,24,25,26, clinicians should take advantage and assist patients with stroke on motor tasks training during this period of time. Celnik and colleagues27 found that the hand function of chronic stroke subjects improved immediately after two-hour peripheral nerve stimulation combined with functional training, and the effect lasted for one day. Based on previous studies, the ES that increases corticomuscular excitability may turn out to be an ideal intervention added prior to traditional motor training to “activate” the neural circuit, so that patients may get the most out of the training. According to a recent study that applied single session peripheral ES on post-stroke individuals, the corticomuscular coherence (CMC), which is the synchronization level between EEG and EMG, increased significantly and was accompanied by improvement in the steadiness of force output28.
To our knowledge, however, there is no study investigating the long-term effect of ES combined with functional training on both motor performance and cortical excitability. We targeted the median nerve because its distribution covered the dorsal side of index, middle, and half of ring finger and the palmar side of the first three fingers and half of the ring finger. Besides, median nerve is in charge of the flexion of the first three fingers, which combined they accounts for most of the functional tasks of hand. Therefore, the purpose of this pilot study was to preliminarily evaluate the effect of eight-week ES-combined hand functional training among chronic stroke patients based on CMC and motor performance. We followed up for four weeks after the intervention ceased and examined the lasting effect. We hypothesized that those who received intervention with ES would have better hand function and higher CMC than those who received intervention with sham ES. We also hypothesized that the effect would last for at least four weeks during our follow-up.
Methods
This current study was a single-blind (subject) randomized placebo control trial. Blocked randomization was used to ensure equal number of subjects in each group.
Subjects
Twelve chronic stroke survivors (1 female and 11 male subjects, age: 56.5 ± 9.5) were recruited for this study. All the participants were screened based on following criteria. The inclusion criteria were (1) first-ever cerebral cortical region involved chronic stroke, onset over a month, (2) able to perform active thumb flexion on the affected side with the scores of manual muscle test at least two points, and (3) at stable medical condition for intervention confirmed by a specialized physician. The exclusion criteria were (1) history of other neurological disorders, (2) cognitive impairment (Mini-Mental State Examination score < 24, MMSE)29, (3) unable to follow instructions, (4) contraindications of ES, and (5) under 20 years old. The protocol was approved by the Institutional Review Board at Taipei Veterans General Hospital, and the subjects gave their informed consent prior to participation in the experiments. All experimental procedures were performed in accordance with the Declaration of Helsinki (Clinical Registration number: NCT03277534; Date of registration: September 6, 2017).
Experimental procedures
Eligible participants were randomly assigned into the ES-intervention group and the placebo control group. Both groups received treatment twice a week for eight weeks. The ES-intervention group received 40 minutes of ES19,28,30,31, while the placebo control group received 40-minute sham ES. The evaluation time points were set at the baseline, four weeks after, at the end of the eight-week intervention and four weeks after the intervention period ended. The evaluation included real-time electroencephalography (EEG) and electroencephalography (EMG) collection for CMC analysis and a functional test.
ES parameters and functional training
The ES was delivered though 5 cm × 5 cm surface electrodes (S88 STIMULATOR, Grass technologies, RI, USA.) One of the electrodes was placed at volar side of elbow joint and the other placed 1 cm apart distally, just over the surface of median nerve. The subjects in ES group received 1 millisecond-rectangular pulse at 100 Hz with a 20-second on 20-second off cycle. The total ES intervention time is 40 minutes and the intensity was set at the highest tolerable level without pain or muscle twitch. These ES parameters have been shown to increase the cortical activities previously19,28,30,31. For the placebo control group, the surface electrodes were also placed on the aforementioned location, but no current was delivered. Subjects in the placebo control group were informed that due to the parameter settings of ES, it might not induce sensation. After 40-minute real or placebo ES, a certified physical therapist started 20-minute routine hand functional training for all subjects. They were asked to do tasks at different levels depending on their abilities, including picking up objects of different sizes and other instrumental activities of daily living tasks such as, using spoon, using scissors, writing, and so on. We documented the ES intensity for each subject. For each stimulation session, we started with the intensity that was last used and adjusted it according to the sensation of subject at the moment to ensure it was the highest tolerable sensory stimulation without pain and muscle twitch.
EEG and EMG collection
The thenar eminence of the affected side were tested. The EEG and EMG signals were collected simultaneously while the subjects performed thumb isometric flexion at 50% maximal voluntary contraction (MVC) for 20 seconds. The subject’s forearm was supported on the table in the full supinated position. The thumb was secured in a ring-shape device, which was attached to a force transducer (FT10, Grass Technologies, RI, USA). EMG signals were recorded using surface electrodes (11-mm Ag-AgCl, 3.0 cm inter electrode distance) and were placed on the affected thenar eminence, proximal to first metacarpophalangeal joint and the ground electrode on styloid process. The force and EMG signals were amplified (P511 AC amplifier, Grass Technologies, RI, USA), and sampled (sampling rate 1000 Hz, CED Power1401 MK II, Cambridge Electronic Design Limited, UK) in real time and stored in a computer for later analysis. Verbal cues and visual feedback were given to help and encourage the subjects to maintain force output at 50% MVC. The subjects were asked to perform the trial twice with 1-minute break in between to avoid fatigue. We chose the trial with more stable force output for further comparison. The 16-channel EEG (actiCAP, Brain Products GmbH, Germany) was used to collect signals from bilateral sensorimotor and primary motor area including FC3, FC1, FCz, FC2, FC4, C3, C1, Cz, C2, C4, CP3, CP1, CPz, CP2, CP4, and Pz by 10–20 system where Cz is defined as the cross of midpoint between nasion and inion and between two ears. The EEG signals were amplified, filtered (1–100 Hz, 60 Hz notch) and sampled (2000 Hz) with BrainVision V-Amp (Brain Products GmbH, Germany.)
Functional test
The Fugl-Meyer Assessment for Upper Extremity (FMA-UE)32,33 subset A to D were used for the functional test. There are 33 items in the assessment and the total score is 66 points. A higher score stands for better motor function of upper limb. Previous studies showed that FMA-UE provided high inter-rater and test-retest reliability, and is highly correlated to the Hemispheric Stroke Scale, which is a measurement of impairment34,35,36.
Data management (CMC calculation)
Both EEG and EMG signals were further analyzed offline with MATLAB (The MathWorks, USA) to calculate the CMC based on the following equation37:
Cxy(f) is the coherence between x and y in frequency domain and can be calculated via Equation (1). In this study, Pxx(f) and Pyy(f) represent the power spectral density (PSD) of EEG or EMG in frequency domain; Pxy(f) represents the cross PSD between EEG and EMG in frequency domain. The signals were analyzed within a 2048-sample epoch with 50% overlap. The length of the signal is 20 seconds and the resolution of frequency is 0.98 Hz.
Since only CMC values that exceed a theoretical threshold represent physiologically meaningful functional connections as described in an earlier study by Rosenberg37, we first calculated the theoretical threshold for each subject using the following Equation (2). In the equation, \({\rm{\alpha }}\) represents the confident interval, commonly being set at 95%, and n represents the number of epochs. Therefore, the length of the data can affect the theoretical threshold directly.
However, Rosenberg’s original equation did not take overlapping of the epoch into consideration during coherence analysis. Therefore, the equation was modified based on the overlapping of the epoch by a later study38.
In Equation (3), \(\tilde{{\rm{k}}}\) represents the number of overlapped epochs and was calculated with the following Equation (4), where k represents the number of epochs without overlapping.
Equation (4) was derived from a cumulative distribution function. To calculate \({C}_{w}(D)\), the following two equations (Equations 5 and 6) were used:
D represents the length that two signals were not overlapped and L represents the length of the epoch. In this study, we used the modified theoretical threshold to identify the physiological meaningful functional connection.
The CMC was evaluated between 0–70 Hz. However, since the motor task performed in this study was static isometric muscle contractions at 50% MVC, we focused on beta band CMC (13–30 Hz), as it is found to be more dominant during weak to moderate39, static40, and isometric41 muscle contractions. The summation of beta band CMC that exceeded the critical threshold of the electrodes in the affected hemisphere was calculated and referred to as the CMC value.
Statistical analysis
All statistical analyses were performed using SPSS Statistics 22.0 (IBM, USA). Normal distribution of the data was checked by Shapiro-Wilk test. The primary outcomes of the study were CMC and FMA-UE. Intra-group comparisons of CMC and FMA-UE scores among four time points were conducted using the Friedman test. The Wilcoxon signed-rank test was used for post hoc analysis between two different time points. Inter-group comparisons of CMC and FMA-UE scores between the two groups at each time point were conducted using Mann–Whitney U test. All data are presented as mean values ± standard error of mean (±SEM) in figures or standard deviation (±SD) in text and the significant level was set at p < 0.05. For the post-hoc analysis between timing points, the significant level with Bonferroni adjustment was set at 0.017. The intra- and inter- power and effect size r42 for all between paired comparisons of each primary dependent variable were computed as well.
Data Availability
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
Results
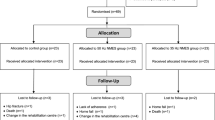
All subjects (N = 12) received the intervention twice a week for eight weeks and finished all assessment sessions including follow-up. (Figure 1) The intervention was given based on the group assignment. The ES group received 40-min sensory level ES prior to motor training and the control group received sham-ES instead. The baseline of the two groups was not different in regards to demographic and clinical characteristics data (Table 1), CMC and FMA-UE (p > 0.05). Since the sample size was small and not normal distributed (p < 0.05), the non-parametric tests were performed for the following results.
CONSORT flow chart. Fifteen stroke patients were screened for eligibility and three of them were excluded (for weak thumb flexion (manual muscle test = 0,0,1/5)). Twelve subjects were randomly allocated and all of them completed the intervention session and the follow-up assessment. The data collected from all subjects were analyzed.
Figure 2 showed the C2 CMC plot of single subject from each group. The CMC values significantly differed among the four time points (Friedman test, p = 0.01). For the post-hoc analysis, the CMC in ES group at the 4th week has a trend to be higher than baseline (p = 0.046, power = 0.940, effect size r = 0.785), 8th week (p = 0.028, power = 0.939, effect size r = 0.899), and follow-up (p = 0.028, power = 0.966, effect size r = 0.899), while no difference was found in the control group. Moreover, the CMC at the 4th week in the ES group was significantly higher (p = 0.004, power = 0.998, effect size r = 0.786) than that in control group (Fig. 3).
CMC of C2 electrode from typical subjects. CMC values before intervention (Pre), at the fourth week (4 wk), after completing 8 weeks of intervention (8 wk), and 4 weeks follow-up (f/u) for one subject in the ES group (dark line) and one subject in the control group (dashed line) were shown. The C2 electrode was chosen here because both subjects had right hemisphere stroke and C2 is the closet the right primary motor cortex hand area. The red line represents the critical threshold (0.0859) calculated by the equations described in Methods section. At the fourth week, the subject in the ES group had more area in beta band above the critical threshold, indicating the motor cortex had greater and physiologically meaningful functional connectivity with corresponding muscles.
CMC values between the ES and control groups. The data were presented as mean and SE. The CMC value in ES group at 4-wk had a trend to be higher than baseline (p = 0.046), 8-wk (p = 0.028), and follow-up (p = 0.028) while no significance difference can be found in control group. Moreover, the CMC value at 4-wk in ES group was significantly higher (p = 0.004) than that in control group. CMC: corticomuscular coherence; ES: electrical stimulation; 4 wk: 4th week; 8 wk: 8th week; f/u: follow-up. *Significance between groups.
The FMA-UE score significantly differed among the four time points (Friedman test, p = 0.035). For the post-hoc analysis, the notable increase of FMA-UE score at the 8th week and during follow-up can only be found in the ES group as compared to the baseline (p = 0.042, power = 0.473, effect size r = 0.830 and p = 0.042, power = 0.374, effect size r = 0.830 respectively). No significant difference in FMA-UE between groups was found at any time point. On the other hand, the FMA-UE score of the control group only increased slightly without any significant change between each assessment session (Fig. 4).
FMA-UE score between the ES and control groups. The data were presented as mean and SE. The FMA-UE scores in ES group at both 8-wk and follow-up had the trend to be higher compared to baseline (p = 0.042 and p = 0.042.) There was no significant change with time in control group. No statistically significance can be found between groups. FMA-UE: Fugl-Meyer Assessment for Upper Extremity; ES: electrical stimulation; 4 wk: 4th week; 8 wk: 8th week; f/u: follow-up.
We further conducted a Chi-square test with Fisher’s exact test to compare the number of subjects that improved more than 5 points or reached full score (66) between the two groups at each of the time points after treatment. This sub-analysis showed no significant difference in the proportion of people between the two groups at the 4th week (p = 0.727), the 8th week (p = 0.121), and the follow-up (p = 0.284). We further performed Pearson’s correlation analysis but no correlation between CMC increment at the fourth week and FMA-UE score at the eighth week (r = 0.234, p = 0.288).
Discussion
In this proof of concept study, we confirmed our initial hypothesis that ES stimulation using an ON/OFF paradigm induces a significant effect in CMC and a significant improvement in motor function as indexed by FMA-UE scores. There are important novel insights from these data to be discussed: (1) duration and timing of behavioral effects; (2) changes in the neural marker (CMC).
Although not achieving statistical significance, it is important to note that 66.7% (4 out of 6) of the subjects in the ES group achieved a significant improvement of at least 5 points or achieved full score (66) in FMA-UE after 8-week intervention and the follow-up assessment, whereas only 1 subject in the control group achieved significant improvement. In fact, motor recovery after stroke is found to be limited, especially for the upper extremities6,7. Duncan and colleagues43 found that the subjects who had greatest motor recovery occurred at the first 30 days after stroke onset across different severity of initial impairments. However, those with moderate to severe initial impairments experienced additional motor recovery up to 90 days post-stroke. It was indicated that the motor recovery plateau occurred at around 12 weeks or three months after stroke44,45, yet there were still around 50% stroke survivors who suffered from upper extremity impairment at different levels6. This suggests that current rehabilitation programs are less effective for chronic stroke survivors. In this study, most of our subjects were at the chronic stage of stroke; however, those in the ES group still improved in motor function.
ES applied in the periphery elicits sensory inputs to modulate the excitability of motor cortex via two ascending pathways46,47. Our results indicate that additional sensory stimuli which facilitate cortical plasticity may have assisted motor re-learning even long after the onset of stroke. Although it has been suggested that peripheral ES can enhance the effectiveness of neurorehabilitation48,49,50, to our knowledge, this is the first study to monitor long-term effects of motor training combined with peripheral ES to induce cortical plasticity and motor recovery in chronic stroke patients. We found that in the ES group, the CMC in beta band increased significantly after four weeks of intervention and the FMA-UE score improved significantly after eight weeks of intervention. This improvement maintained throughout the four-week follow-up. We discuss below the timing of these changes, i.e., first neural marker changes and then behavioral changes.
In the ES group, the beta band CMC increased significantly after four weeks of intervention, but not for the eight-week assessment or during follow-up. Previous studies found that cortical plasticity is crucial for learning new motor tasks22, and greater cortical excitability was observed during motor re-learning51,52. Increased CMC in beta band suggested that during motor re-learning, the motor cortex has stronger connectivity between the central nervous system and muscles at the periphery. As the specific motor task being re-learned, less effort or attention will be needed to perform the specific motor task52. This phenomenon was observed as decreased or back-to-baseline CMC values after eight weeks of intervention in current study. In fact, the temporal sequence of results supports this CMC indication for motor learning. When we performed the EEG-EMG recording for CMC calculation, we recorded the actual force output level of thumb flexion at the same time. Therefore, we were able to observe the performance of sustained force output at 50% MVC. The accuracy (mean difference between target and actual force output) and steadiness (coefficient of variation between target and actual force output) of thumb flexion during EEG-EMG collection was calculated and the lower values stand for better performance. We found improvement of accuracy in ES group (6.9% ± 3.8% to 2.6% ± 1.9%) and improvement of steadiness as well (8.1% ± 5.5% to 6.2% ± 2.5%) even with lower CMC. On the other hand, those in control group even showed decreased performance with 2.3% ± 1.7% to 4.7% ± 3.6% for accuracy and 7.4% ± 2.4% to 12.8% ± 18.5% for steadiness. Hence, we can conclude that the motor task became less difficult for the subjects in ES group. The difference in trend change for CMC and FMA-UE score reflects the progress of motor re-learning. At the fourth week, as the subjects were still learning the new motor task, or establishing the best connections between the motor cortex and skeletal muscles, more attention was needed to perform the specific motor task. Therefore, the CMC value increased in order to maintain such motor performance. At the eighth week, the subjects acquired better motor function and neural planning (FMA-UE score) as they perform the same motor task during EEG-EMG collection, so less attention was required. As a result, less neural demand was needed between the motor cortex and muscles53,54. Even without significant correlation, we believe changes in CMC is linked to FMA-UE score as previously observed28. However, the correlation between CMC and motor function might not be strong in stroke patients55, and could be influenced by age, severity and area of stroke.
Our findings are in accordance with other studies that used sensorial enhancement as a method to engage the sensory system to enhance the effects of motor training strategies49,56,57,58,59,60,61,62,63,64. Other studies about motor recovery after stroke that also used FME-UA as outcome measurement reveal similar improvement in the score increment56,60,61,65. Quite a few other studies66,67 focused on the Constraint-Induced Movement Therapy (CIMT) in recent years. Yoon and colleagues66 compared the effect of CIMT, CIMT with mirror therapy, and control in subacute stroke subjects. The improvement in FMA-UE score was from 35.4 to 47.0, 47.9 to 53.3, and 32.7 to 37.0 for CIMT with mirror therapy, CIMT only, and control group, respectively. Based on a review, those who received CIMT improved 7.8 points (4.2 to 11.4) on average67.
Studies that applied additional electrical stimulation also found additional gains in motor recovery, though the targets for electrical stimulation were different. For example, for repetitive transcranial magnetic stimulation (rTMS), peripheral ES, and transcranial direct current stimulation (tDCS), positive results have been reported. Blesneag and colleagues68 applied low frequency rTMS over the primary motor cortex on acute ischemic stroke survivors and found that the FMA-UE scores of the subjects increased roughly from 29.6 at baseline to 42.8 after 45 days of intervention and reached 45 during a 90-day follow-up. Another study also applied rTMS over the primary motor cortex but focused on subacute to chronic stroke survivors. The authors found that the FMA-UE score increased from 28 to 30.969. McDonnell and colleagues56 found that applying associative electrical stimulation of motor points on both hands prior to task-oriented training for three weeks showed improvement in FMA-UE score from 47.3 to 53.3. This particular study had intervention procedures similar to ours but with major differences in regard to ES parameters and the chronicity of the stroke (4.1 mon. vs. 38.5 mon.). Cha and colleagues70 applied tDCS on chronic stroke subjects (13.8 months post stroke) and found improvement of FMA-UE score from 20.5 to 48.7; this specific study revealed the greatest improvement among all, which may be due to the lower FMA-UE score at baseline. That is, the subjects had more room to improve after the intervention.
As aforementioned, a 5-point change in FMA-UE score indicates significant clinical change of motor function71. Therefore, the change of FMA-UE score in our study showed significant clinical improvement of upper extremity function and was similar to the results of other studies that provided motor training or electrical/magnetic stimulation as an approach for stroke intervention. The subjects of the ES group in this study were at more chronic phases (38.5 mon.) than other studies, but nevertheless, they still showed clinically significant improvement in the FMA-UE score. Hence, it can be assumed that chronic stroke survivors could benefit more from combined training. That is, by applying ES prior to functional training of upper extremities, we can facilitate the cortical plasticity of the corresponding region of the motor cortex and strengthen the connections between the region and the actuating muscles, so that patient’s corticomuscular pathway is “primed” for subsequent motor training72. The control group in this study showed trend of slight improvement in motor function. The results were in line with previous studies about the motor recovery in chronic stages; the most dramatic change took place within the first year after stroke and reached a plateau thereafter73.
The final point of discussion is the parameters of stimulation. There is intensive research on the optimal parameters of stimulation. In fact, it is a critical factor to determining the behavioral and neural effects of electrical stimulation74,75,76. Chen et al. showed that a variation of intensity can have a significant impact in induced neuroplasticity77; in fact, we used a protocol of ON/OFF to deliver ES that may also have been associated with significant improvements observed in this study. Another point to consider is potential home use. ES can be easily applied for home-based rehabilitation program. Therefore, the intervention in the current study could be appropriate rehabilitation at the chronic stage. The suitability at the acute phase requires future study to investigate the possible effects.
The main limitation of this pilot study was the small sample size. However, with only that limited sample size we showed clinically significant results that support our a priori hypothesis. Another limitation was that the CMC value was calculated based on one out of the two trials, however, the CMC values were stable between the trials within the same subject during the same assessment session. Thus, the findings here are important to encourage further research in this area. Future studies with more subjects and with different regimens of stimulation will be needed to confirm the results of this preliminary pilot study.
In conclusion, the results of our pilot study indicate that chronic stroke survivors who received eight weeks of ES prior to functional training can facilitate corticomuscular functional connectivity and hence improve upper limb function. Additionally, they bring important insights into the timing of neural and behavioral changes as well as support further studies applying peripheral electrical stimulation.
References
Benjamin, E. J. et al. Heart Disease and Stroke Statistics-2017 Update: A Report From the American Heart Association. Circulation 135, e146–e603 (2017).
Yan, K., Fang, J. & Shahani, B. T. Motor unit discharge behaviors in stroke patients. Muscle Nerve 21, 1502–1506 (1998).
Garland, S. J., Gray, V. L. & Knorr, S. Muscle activation patterns and postural control following stroke. Motor Control 13, 387–411 (2009).
Langhorne, P., Coupar, F. & Pollock, A. Motor recovery after stroke: a systematic review. Lancet Neurol. 8, 741–754 (2009).
Cengić, L., Vuletić, V., Karlić, M., Dikanović, M. & Demarin, V. Motor and cognitive impairment after stroke. Acta Clin. Croat. 50, 463–467 (2011).
Gowland, C. Recovery of motor function following stroke: Profile and predictors. Physiother. Can. 34, 77–84 (1982).
Nakayama, H., Jørgensen, H. S., Raaschou, H. O. & Olsen, T. S. Recovery of upper extremity function in stroke patients: the Copenhagen Stroke Study. Arch. Phys. Med. Rehabil. 75, 394–398 (1994).
Bobath, B. Observations on adult hemiplegia and suggestions for treatment. Physiotherapy 45, 279–289 (1959).
Kabat, H. & Knott, M. Proprioceptive facilitation therapy for paralysis. Physiotherapy 40, 171–176 (1954).
Charlton, C. S., Ridding, M. C., Thompson, P. D. & Miles, T. S. Prolonged peripheral nerve stimulation induces persistent changes in excitability of human motor cortex. J. Neurol. Sci. 208, 79–85 (2003).
Backes, W. H., Mess, W. H., van Kranen-Mastenbroek, V. & Reulen, J. P. Somatosensory cortex responses to median nerve stimulation: fMRI effects of current amplitude and selective attention. Clin. Neurophysiol. Off. J. Int. Fed. Clin. Neurophysiol. 111, 1738–1744 (2000).
Gizewski, E. R. et al. Cerebral activation using a MR-compatible piezoelectric actuator with adjustable vibration frequencies and in vivo wave propagation control. NeuroImage 24, 723–730 (2005).
Spiegel, J., Tintera, J., Gawehn, J., Stoeter, P. & Treede, R. D. Functional MRI of human primary somatosensory and motor cortex during median nerve stimulation. Clin. Neurophysiol. Off. J. Int. Fed. Clin. Neurophysiol. 110, 47–52 (1999).
Chipchase, L. S., Schabrun, S. M. & Hodges, P. W. Peripheral electrical stimulation to induce cortical plasticity: a systematic review of stimulus parameters. Clin. Neurophysiol. Off. J. Int. Fed. Clin. Neurophysiol. 122, 456–463 (2011).
Nudo, R. J., Plautz, E. J. & Frost, S. B. Role of adaptive plasticity in recovery of function after damage to motor cortex. Muscle Nerve 24, 1000–1019 (2001).
Wu, C. W. H. & Kaas, J. H. The effects of long-standing limb loss on anatomical reorganization of the somatosensory afferents in the brainstem and spinal cord. Somatosens. Mot. Res. 19, 153–163 (2002).
Volz, M. S. et al. Effects of sensory behavioral tasks on pain threshold and cortical excitability. PloS One 8, e52968 (2013).
Khaslavskaia, S., Ladouceur, M. & Sinkjaer, T. Increase in tibialis anterior motor cortex excitability following repetitive electrical stimulation of the common peroneal nerve. Exp. Brain Res. 145, 309–315 (2002).
Mang, C. S., Clair, J. M. & Collins, D. F. Neuromuscular electrical stimulation has a global effect on corticospinal excitability for leg muscles and a focused effect for hand muscles. Exp. Brain Res. 209, 355–363 (2011).
Ridding, M. C., Brouwer, B., Miles, T. S., Pitcher, J. B. & Thompson, P. D. Changes in muscle responses to stimulation of the motor cortex induced by peripheral nerve stimulation in human subjects. Exp. Brain Res. 131, 135–143 (2000).
Kimberley, T. J. et al. Electrical stimulation driving functional improvements and cortical changes in subjects with stroke. Exp. Brain Res. 154, 450–460 (2004).
Pavlides, C., Miyashita, E. & Asanuma, H. Projection from the sensory to the motor cortex is important in learning motor skills in the monkey. J. Neurophysiol. 70, 733–741 (1993).
Pascual-Leone, A. et al. Modulation of muscle responses evoked by transcranial magnetic stimulation during the acquisition of new fine motor skills. J. Neurophysiol. 74, 1037–1045 (1995).
Classen, J., Liepert, J., Wise, S. P., Hallett, M. & Cohen, L. G. Rapid plasticity of human cortical movement representation induced by practice. J. Neurophysiol. 79, 1117–1123 (1998).
Bütefisch, C. M. et al. Mechanisms of use-dependent plasticity in the human motor cortex. Proc. Natl. Acad. Sci. USA 97, 3661–3665 (2000).
Ziemann, U., Muellbacher, W., Hallett, M. & Cohen, L. G. Modulation of practice-dependent plasticity in human motor cortex. Brain J. Neurol. 124, 1171–1181 (2001).
Celnik, P., Hummel, F., Harris-Love, M., Wolk, R. & Cohen, L. G. Somatosensory Stimulation Enhances the Effects of Training Functional Hand Tasks in Patients With Chronic Stroke. Arch. Phys. Med. Rehabil. 88, 1369–1376 (2007).
Lai, M.-I. et al. Investigating the Effects of Peripheral Electrical Stimulation on Corticomuscular Functional Connectivity Stroke Survivors. Top. Stroke Rehabil. 23, 154–162 (2016).
Folstein, M. F., Folstein, S. E. & McHugh, P. R. Mini-mental state. A practical method for grading the cognitive state of patients for the clinician. J. Psychiatr. Res. 12, 189–198 (1975).
Mang, C. S., Lagerquist, O. & Collins, D. F. Changes in corticospinal excitability evoked by common peroneal nerve stimulation depend on stimulation frequency. Exp. Brain Res. 203, 11–20 (2010).
Mang, C. S., Bergquist, A. J., Roshko, S. M. & Collins, D. F. Loss of short-latency afferent inhibition and emergence of afferent facilitation following neuromuscular electrical stimulation. Neurosci. Lett. 529, 80–85 (2012).
Fugl-Meyer, A. R., Jääskö, L., Leyman, I., Olsson, S. & Steglind, S. The post-stroke hemiplegic patient. 1. a method for evaluation of physical performance. Scand. J. Rehabil. Med. 7, 13–31 (1975).
Berglund, K. & Fugl-Meyer, A. R. Upper extremity function in hemiplegia. A cross-validation study of two assessment methods. Scand. J. Rehabil. Med. 18, 155–157 (1986).
Gladstone, D. J., Danells, C. J. & Black, S. E. The fugl-meyer assessment of motor recovery after stroke: a critical review of its measurement properties. Neurorehabil. Neural Repair 16, 232–240 (2002).
Platz, T. et al. Reliability and validity of arm function assessment with standardized guidelines for the Fugl-Meyer Test, Action Research Arm Test and Box and Block Test: a multicentre study. Clin. Rehabil. 19, 404–411 (2005).
Hsieh, Y. et al. Responsiveness and validity of three outcome measures of motor function after stroke rehabilitation. Stroke J. Cereb. Circ. 40, 1386–1391 (2009).
Rosenberg, J. R., Amjad, A. M., Breeze, P., Brillinger, D. R. & Halliday, D. M. The Fourier approach to the identification of functional coupling between neuronal spike trains. Prog. Biophys. Mol. Biol. 53, 1–31 (1989).
Gallet, C. & Julien, C. The significance threshold for coherence when using the Welch’s periodogram method: Effect of overlapping segments. Biomed. Signal Process. Control 6, 405–409 (2011).
Witte, M., Patino, L., Andrykiewicz, A., Hepp-Reymond, M.-C. & Kristeva, R. Modulation of human corticomuscular beta-range coherence with low-level static forces. Eur. J. Neurosci. 26, 3564–3570 (2007).
Kristeva, R., Patino, L. & Omlor, W. Beta-range cortical motor spectral power and corticomuscular coherence as a mechanism for effective corticospinal interaction during steady-state motor output. NeuroImage 36, 785–792 (2007).
Gwin, J. T. & Ferris, D. P. Beta- and gamma-range human lower limb corticomuscular coherence. Front. Hum. Neurosci. 6, 258 (2012).
Rosenthal, R. Parametric measures of effect size. In The handbook of research synthesis (eds Hedges, L. V. & Valentine, J. C.) p. 231–244 (New York: Russell Sage Foundation).
Duncan, P. W., Goldstein, L. B., Matchar, D., Divine, G. W. & Feussner, J. Measurement of motor recovery after stroke. Outcome assessment and sample size requirements. Stroke J. Cereb. Circ. 23, 1084–1089 (1992).
Parker, V. M., Wade, D. T. & Langton Hewer, R. Loss of arm function after stroke: measurement, frequency, and recovery. Int. Rehabil. Med. 8, 69–73 (1986).
Olsen, T. S. Arm and leg paresis as outcome predictors in stroke rehabilitation. Stroke 21, 247–251 (1990).
Pons, T. P. & Kaas, J. H. Corticocortical connections of area 2 of somatosensory cortex in macaque monkeys: a correlative anatomical and electrophysiological study. J. Comp. Neurol. 248, 313–335 (1986).
Horne, M. K. & Tracey, D. J. The afferents and projections of the ventroposterolateral thalamus in the monkey. Exp. Brain Res. 36, 129–141 (1979).
Johansson, K., Lindgren, I., Widner, H., Wiklund, I. & Johansson, B. B. Can sensory stimulation improve the functional outcome in stroke patients? Neurology 43, 2189–2192 (1993).
Conforto, A. B., Kaelin-Lang, A. & Cohen, L. G. Increase in hand muscle strength of stroke patients after somatosensory stimulation. Ann. Neurol. 51, 122–125 (2002).
Carrico, C. et al. Nerve Stimulation Enhances Task-Oriented Training in Chronic, Severe Motor Deficit After Stroke: A Randomized Trial. Stroke J. Cereb. Circ. 47, 1879–1884 (2016).
Askim, T., Indredavik, B., Vangberg, T. & Håberg, A. Motor network changes associated with successful motor skill relearning after acute ischemic stroke: a longitudinal functional magnetic resonance imaging study. Neurorehabil. Neural Repair 23, 295–304 (2009).
Halsband, U. & Lange, R. K. Motor learning in man: a review of functional and clinical studies. J. Physiol. Paris 99, 414–424 (2006).
Feige, B., Aertsen, A. & Kristeva-Feige, R. Dynamic synchronization between multiple cortical motor areas and muscle activity in phasic voluntary movements. J. Neurophysiol. 84, 2622–2629 (2000).
Kristeva-Feige, R., Fritsch, C., Timmer, J. & Lücking, C.-H. Effects of attention and precision of exerted force on beta range EEG-EMG synchronization during a maintained motor contraction task. Clin. Neurophysiol. Off. J. Int. Fed. Clin. Neurophysiol. 113, 124–131 (2002).
Rossiter, H. E. et al. Changes in the location of cortico-muscular coherence following stroke. NeuroImage Clin. 2, 50–55 (2012).
McDonnell, M. N., Hillier, S. L., Miles, T. S., Thompson, P. D. & Ridding, M. C. Influence of Combined Afferent Stimulation and Task-Specific Training Following Stroke: A Pilot Randomized Controlled Trial. Neurorehabil. Neural Repair 21, 435–443 (2007).
Floel, A. et al. Influence of somatosensory input on motor function in patients with chronic stroke. Ann. Neurol. 56, 206–212 (2004).
Wu, C. W., Seo, H.-J. & Cohen, L. G. Influence of Electric Somatosensory Stimulation on Paretic-Hand Function in Chronic Stroke. Arch. Phys. Med. Rehabil. 87, 351–357 (2006).
Lin, Z. & Yan, T. Long-term effectiveness of neuromuscular electrical stimulation for promoting motor recovery of the upper extremity after stroke. J. Rehabil. Med. 43, 506–510 (2011).
Chan, M. K., Tong, R. K. & Chung, K. Y. Bilateral Upper Limb Training With Functional Electric Stimulation in Patients With Chronic Stroke. Neurorehabil. Neural Repair 23, 357–365 (2009).
Kowalczewski, J., Gritsenko, V., Ashworth, N., Ellaway, P. & Prochazka, A. Upper-Extremity Functional Electric Stimulation–Assisted Exercises on a Workstation in the Subacute Phase of Stroke Recovery. Arch. Phys. Med. Rehabil. 88, 833–839 (2007).
Rosewilliam, S., Malhotra, S., Roffe, C., Jones, P. & Pandyan, A. D. Can Surface Neuromuscular Electrical Stimulation of the Wrist and Hand Combined With Routine Therapy Facilitate Recovery of Arm Function in Patients With Stroke? Arch. Phys. Med. Rehabil. 93, 1715–1721.e1 (2012).
Page, S. J., Levin, L., Hermann, V., Dunning, K. & Levine, P. Longer Versus Shorter Daily Durations of Electrical Stimulation During Task-Specific Practice in Moderately Impaired Stroke. Arch. Phys. Med. Rehabil. 93, 200–206 (2012).
Lin, K.-C. et al. Effect of mirror therapy combined with somatosensory stimulation on motor recovery and daily function in stroke patients: A pilot study. J. Formos. Med. Assoc. 113, 422–428 (2014).
Kim, K., Lee, S., Kim, D., Lee, K. & Kim, Y. Effects of mirror therapy combined with motor tasks on upper extremity function and activities daily living of stroke patients. J. Phys. Ther. Sci. 28, 483–487 (2016).
Yoon, J. A. et al. Effect of constraint-induced movement therapy and mirror therapy for patients with subacute stroke. Ann. Rehabil. Med. 38, 458–466 (2014).
Shi, Y. X., Tian, J. H., Yang, K. H. & Zhao, Y. Modified constraint-induced movement therapy versus traditional rehabilitation in patients with upper-extremity dysfunction after stroke: a systematic review and meta-analysis. Arch. Phys. Med. Rehabil. 92, 972–982 (2011).
Blesneag, A. et al. Low-frequency rTMS in patients with subacute ischemic stroke: clinical evaluation of short and long-term outcomes and neurophysiological assessment of cortical excitability. J. Med. Life 8, 378–387 (2015).
Sung, W.-H. et al. Efficacy of coupling inhibitory and facilitatory repetitive transcranial magnetic stimulation to enhance motor recovery in hemiplegic stroke patients. Stroke J. Cereb. Circ. 44, 1375–1382 (2013).
Cha, H.-K., Ji, S.-G., Kim, M.-K. & Chang, J.-S. Effect of transcranial direct current stimulation of function in patients with stroke. J. Phys. Ther. Sci. 26, 363–365 (2014).
Chang, W. H., Uhm, K. E., Shin, Y.-I., Pascual-Leone, A. & Kim, Y.-H. Factors influencing the response to high-frequency repetitive transcranial magnetic stimulation in patients with subacute stroke. Restor. Neurol. Neurosci. https://doi.org/10.3233/RNN-150634 (2016).
Ackerley, S. J., Stinear, C. M., Barber, P. A. & Byblow, W. D. Priming sensorimotor cortex to enhance task-specific training after subcortical stroke. Clin. Neurophysiol. Off. J. Int. Fed. Clin. Neurophysiol. 125, 1451–1458 (2014).
Feys, H. et al. Early and Repetitive Stimulation of the Arm Can Substantially Improve the Long-Term Outcome After Stroke: A 5-Year Follow-up Study of a Randomized Trial. Stroke 35, 924–929 (2004).
Bjordal, J. M., Johnson, M. I. & Ljunggreen, A. E. Transcutaneous electrical nerve stimulation (TENS) can reduce postoperative analgesic consumption. A meta-analysis with assessment of optimal treatment parameters for postoperative pain. Eur. J. Pain Lond. Engl. 7, 181–188 (2003).
Fregni, F. Analgesia with noninvasive electrical cortical stimulation: challenges to find optimal parameters of stimulation. Anesth. Analg. 111, 1083–1085 (2010).
Zaghi, S., Acar, M., Hultgren, B., Boggio, P. S. & Fregni, F. Noninvasive brain stimulation with low-intensity electrical currents: putative mechanisms of action for direct and alternating current stimulation. Neurosci. Rev. J. Bringing Neurobiol. Neurol. Psychiatry 16, 285–307 (2010).
Chen, C.-F., Lin, Y.-T., Chen, W.-S. & Fregni, F. Contribution of Corticospinal Modulation and Total Electrical Energy for Peripheral-Nerve-Stimulation-Induced Neuroplasticity as Indexed by Additional Muscular Force. Brain Stimulat. 9, 133–140 (2016).
Acknowledgements
This study was supported in part by a grant from Ministry of Science and Technology, Taiwan (106-2917-I-010-004 to L.H.P.; 104-2314-B-010-024 to L.W.C.). The authors would like to thank Aleksandra Galus and the Writing Center at Loyola University Chicago for the editing work on this manuscript.
Author information
Authors and Affiliations
Contributions
L.H.P. co-designed and conducted the experiments, performed data analyses, and prepared the manuscript. W.W.Y. performed data analyses, and prepared the manuscript. C.L.K. and M.W.T. co-designed patient criteria and rehabilitation program, and revised the manuscript. S.H.W. and F.F. provided critical suggestions, and revised the manuscript. V.C.C. interpreted the results, provided critical suggestions, and revised the manuscript, L.W.C. designed and supervised the experiments, interpreted the results, and revised the manuscript. All authors reviewed the manuscript.
Corresponding authors
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Pan, LL.H., Yang, WW., Kao, CL. et al. Effects of 8-week sensory electrical stimulation combined with motor training on EEG-EMG coherence and motor function in individuals with stroke. Sci Rep 8, 9217 (2018). https://doi.org/10.1038/s41598-018-27553-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-27553-4
This article is cited by
-
Lesion-specific cortical activation following sensory stimulation in patients with subacute stroke
Journal of NeuroEngineering and Rehabilitation (2023)
-
Literature review of stroke assessment for upper-extremity physical function via EEG, EMG, kinematic, and kinetic measurements and their reliability
Journal of NeuroEngineering and Rehabilitation (2023)
-
Electrical stimulation of the common peroneal nerve and its effects on the relationship between corticomuscular coherence and motor control in healthy adults
BMC Neuroscience (2021)
-
Pathway-specific modulatory effects of neuromuscular electrical stimulation during pedaling in chronic stroke survivors
Journal of NeuroEngineering and Rehabilitation (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.