Abstract
Shifts in species distribution, or ‘range shifts’, are one of the most commonly documented responses to ocean warming, with important consequences for the function and structure of ecosystems, and for socio-economic activities. Understanding the genetic signatures of range shifts can help build our knowledge of the capacity of species to establish and persist in colonised areas. Here, seven microsatellite loci were used to examine the population connectivity, genetic structure and diversity of Octopus tetricus, which has extended its distribution several hundred kilometres polewards associated with the southwards extension of the warm East Australian Current along south-eastern Australia. The historical distribution and the range extension zones had significant genetic differences but levels of genetic diversity were comparable. The population in the range extension zone was sub-structured, contained relatively high levels of self-recruitment and was sourced by migrants from along the entire geographic distribution. Genetic bottlenecks and changes in population size were detected throughout the range extension axis. Persistent gene flow from throughout the historical zone and moderate genetic diversity may buffer the genetic bottlenecks and favour the range extension of O. tetricus. These characteristics may aid adaptation, establishment, and long-term persistence of the population in the range extension zone.
Similar content being viewed by others
Introduction
In response to the accelerated warming of the oceans, marine species can experience changes in their patterns of geographic distribution and abundance as they track their preferred temperatures1,2. Such range shifts occur in the form of range contractions, relocations, or extensions that have ecological and socio-economic impacts3,4, with already significant consequences on human well-being5. Most of our understanding of how genetic and evolutionary processes may relate to marine range shifts is based on terrestrial invasive or range shifting species6,7,8 and on theoretical studies9,10,11, with only a handful of empirical marine studies12,13. However, a range of different barriers influence the connectivity and genetic patterns of marine populations in comparison to terrestrial populations; these include water masses, currents, eddies, and coastlines, among other factors. As a consequence, the nature and magnitude of genetic patterns and evolutionary processes related to marine range shifts may differ from those that occur in the terrestrial realm. Therefore it is important to perform marine field-based studies that can increase our understanding of genetic patterns and evolutionary processes related to marine range shifts and help validate theoretical models.
Maintaining genetic diversity is essential if populations are to persist during range shifts associated with oceanic warming12. Selection, the reproductive exchange between individuals, dispersal capacity and the presence of barriers to dispersal, can all influence genetic diversity6,14,15 and survival at the trailing edge, at the centre of the distribution, and at the leading edge of range shifts6,10. Populations at the trailing edge may be negatively affected due to low genetic variability unless they receive genes from better adapted populations6. Populations at the centre of the distribution are likely to have more genetic variability due to gene flow from neighbouring populations that are better adapted to different conditions6,16. Genetic diversity at the leading edge of range shifts can increase provided there is gene flow from different areas of the distribution6,16, with maximum survival predicted to occur if the gene flow is from areas with similar conditions compared with the newly colonised areas10,17.
Genetic diversity can decrease due to genetic drift under scenarios of limited gene flow, genetic recombination and selection against poorly adapted genotypes6. Consecutive genetic bottlenecks or founder effects, where the size of the population is reduced or where only few individuals establish in new areas and subsequently become isolated, can result in genetic drift and inbreeding depression18,19,20. There are exceptions where invasive populations have overcome the negative impacts of the loss of genetic diversity via multiple introductions that aid the increase of genetic recombination, confer high phenotypic plasticity to the founder population, and strengthen its ability to respond to natural selection7,21,22. For instance, multiple introductions of individuals appeared to help sustain genetic diversity of the invasive long-spined sea urchin Centrostephanus rodgersii at the southern Tasman Sea off Australia’s east coast12. This area is warming up to four times faster than the global average23, partly caused by the strengthening of the warm East Australian Current (EAC) which flows polewards from the southern Coral Sea along the south-east coast of mainland Australia24. Over the past 60 years the EAC has strengthened and extended approximately 350 km further south towards the temperate east coast of Tasmania25,26.
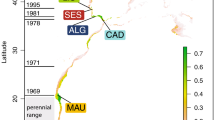
The strengthening of the EAC has been associated with the transport of several dozen marine species during their planktonic larval stage, and their subsequent polewards range shift27,28. One of these range-shifting species is the gloomy or common Sydney octopus Octopus tetricus (Gould, 1852). This commercially important octopus has a short life span (~11 months)29, high reproductive capacity30 and a planktonic paralarval phase. The paralarval phase is likely to last 35–60 days as demonstrated for the closely related O. vulgaris31,32 under laboratory conditions33,34. The historical geographic range of O. tetricus is in shallow-waters from southern Queensland to southern New South Wales (NSW)35,36,37. Octopus tetricus was detected for the first time off Victoria around the year 2000 and off north-eastern Tasmania around 2006 (Fig. 1). Presence-absence records of O. tetricus along the east coast of Australia that support the polewards range extension hypothesis include systematic marine life censuses36,38, fisheries records39, and citizen science monitoring using scientist-verified and geo-referenced photographs40. Using a cost-effective and rapid screening assessment tool based on monitoring data, the range extension of O. tetricus was classified with a “high” level of confidence41. Negative impacts on the structure and function of the Tasmanian reef community are anticipated if the size of the O. tetricus population increases in Tasmanian waters42.
Collection sites for Octopus tetricus along eastern Australia, including historical and extension zones. Numbers in parentheses indicate the approximate years when O. tetricus individuals were detected for the first time off Victoria and off north-eastern Tasmania. The pie charts indicate the percentage of individuals from each site that correspond to each cluster assigned by colour (Cluster 1 – green; Cluster 2 – blue; Cluster 3 – orange; Cluster 4 – purple; Cluster 5 – red). The right panel is the graphic representation of the percentage contribution of individuals to clusters using Discriminant Analysis of Principal Components with the genetic structure being captured by the first three principal components. The Group Ot1 is comprised of individuals from all sites. The distinct Group Ot2 is predominately comprised of individuals from Tasmania (indicated in red). The map was modified from30.
Examination of the genetic structure, level of connectivity and genetic diversity of range shifting populations will provide valuable information on the genetic signatures during range shifts and may allow forecasting of successful colonisations as a function of gene flow and genetic diversity20. Therefore, using O. tetricus as a model range extending marine species, this study aims: (1) to describe the population genetic structure of O. tetricus along eastern Australia, including extension areas; (2) to quantify the genetic diversity of the range extension area versus other population components; (3) to determine the level of gene flow between historical and range extension areas; (4) to identify the source populations that contribute to the range extension area; (5) to determine if there is evidence of genetic bottlenecks that may negatively affect the persistence of O. tetricus at the extended areas; and (6) to describe the recent evolutionary history of this range extending species.
Results
A total of 182 individuals of the common Sydney octopus, Octopus tetricus, were genotyped for seven microsatellite loci (Table 1). Approximately 85% of pairs of loci (89 out of 105 tests over sites with ≥17 samples) were in linkage equilibrium (Supplementary Tables S1, S2). Exclusion of loci involved in linkage disequilibrium comparisons did not improve exploratory analyses. Moreover, Hardy-Weinberg equilibrium was met in 90% of each of the site-locus comparisons (Supplementary Tables S3, S4); therefore, all loci were included in further analyses. There was no evidence of large-allele dropout; stuttering was suggested for loci Ovul02 and Ovul16, as indicated by the shortage of heterozygote genotypes with alleles of one repeat unit difference. Moderate levels of null alleles (false homozygotes) were found in loci Ovul01 (10%), Ovul02 (14%), Ovul05 (≤19%), Ovul16 (≤13%), and at Tasmania (<14%); however, allele frequency bias was corrected. There were some differences in the results estimated from uncorrected data compared with results estimated from corrected data for null alleles. Therefore we present and discuss results based on corrected data where any bias is rectified. Results based on uncorrected data are presented in the Supplementary information online for comparative purposes only.
Population sub-structuring
Discriminant Analysis of Principal Components (DAPC) suggested five clusters (Table 2; Supplementary Fig. S5 and Table S6): (1) green, (2) blue, (3) orange, (4) purple, and (5) red, with 40 Principal Components explaining >80% of cumulated variance. Clusters one to four were largely overlapped, i.e. comprised of a mixture of individuals from along the entire distribution and there was no clear differentiation between these clusters. Therefore, clusters one to four were pooled in the common Group Ot1 (Fig. 1, right panel). Cluster five (red) was separated from the rest of the clusters and was mostly comprised of individuals from Tasmania in the range extension zone (n = 27), but also contained a few individuals from Nambucca Heads (n = 1), Swansea (n = 1) and Merimbula (n = 1) in the historical distribution zone; this distinct group was termed Ot2 (Fig. 1, right panel). Tasmania (n = 61) was thus sub-structured with 56% (n = 34) individuals that belonged to the common Group Ot1, and with 44% (n = 27) individuals that belonged to the genetically distinct Group Ot2. DAPC analyses carried out on a range of subsets of the dataset, i.e. (a) at all sites within mainland Australia only, (b) at the historical distribution zone only, (c) at the range extension zone only, (d) Tasmania only, and (e) not including the distinct Group Ot2, showed similar population structure. The software Structure suggested only four clusters (Table 2; Supplementary Fig. S5 and Table S6), for which individual allocation was not as obvious as for DAPC. Most individuals assigned to clusters one to four in DAPC were allocated to clusters one to three by Structure, which comprise the common Group Ot1. Most individuals assigned to cluster five in DAPC were allocated to cluster four by Structure, which comprise the distinct Group Ot2 (Table 2; Supplementary Table S6 and Fig. S7).
Phylogenetic analysis
Phylogenetic analysis of the mitochondrial gene Cytochrome Oxidase subunit I (COI) demonstrated that individuals of the groups Ot1 and Ot2 correspond to O. tetricus. None of the individuals analysed were assigned to any other species that are closely related to O. tetricus, e.g. O. cf tetricus or O. vulgaris (Supplementary Fig. S8).
Genetic diversity
Allelic richness was moderate with more than seven alleles at each site; the number of private alleles and heterozygosity in the range extension zone were higher than for the historical distribution zone, and inbreeding coefficients were not significant (Table 3; Supplementary Tables S3, S4). Overall, moderate levels of genetic diversity were observed across sites and loci, and genetic diversity was similar at the range extension compared with the historical distribution zone (Table 3).
Genetic connectivity and differentiation
FST estimations provided further detail of the population structure and complemented DAPC and Structure results. There was no significant differentiation between Nambucca Heads, Swansea, and Merimbula (Table 4; Supplementary Table S9). Low but significant FST values indicated that Mallacoota was different from all other sites, except from Swansea. Tasmania was different from all sites along mainland Australia (Table 4; Supplementary Table S9). Exploratory analysis of population differentiation indicated that Ot2 Tasmanian individuals were differentiated from all other sites including the Ot1 Tasmanian individuals (FST = 0.147, P < 0.001). Thus, from the FST estimations we inferred that the common Group Ot1 was comprised of sub-group 1 (Nambucca Heads, Swansea, and Merimbula), sub-group 2 (Mallacoota), and sub-group 3 (Ot1 Tasmanian individuals). The distinct Group Ot2 was comprised of the sub-group 4 (Ot2 Tasmanian individuals). The FST analysis at the Group level confirmed that the common Group Ot1 was significantly different from the Group Ot2 (FST = 0.157, P < 0.001). The Mantel test indicated that the level of genetic differentiation among collection sites was not significantly correlated to geographic distance (P = 0.258) or year of collection (P = 0.096). Accordingly, the Spearman’s correlation analysis suggested that only two loci were significantly correlated to year of collection (Ovul01, rs = 0.231, P = 0.002; Ovul08, rs = 0.148, P = 0.047). Two loci were associated to depth of sampling (Ovul01, rs = −0.230, P = 0.002; Ovul05, rs = −0.176, P = 0.018) (Supplementary Table S10). The AMOVA detected significant percentages of genetic variation among collection sites (3.3%, d.f. = 7, SS = 30.950, VC = 0.062, P < 0.001).
Migration and self-recruitment
Migration rates were asymmetric between sites, with higher migration rates from Merimbula to the other sites (Table 5; Supplementary Table S11). Migration rates were asymmetric between the common Group Ot1 and the distinct Group Ot2, with greater migration rates from the common Group Ot1 towards the distinct Group Ot2 (0.095 ± 0.029 SD), than from the distinct Group Ot2 towards the common Group Ot1 (0.025 ± 0.008 SD). Merimbula had the highest level of self-recruitment (0.914 ± 0.024) (Table 5; Supplementary Table S11). Both groups Ot1 and Ot2 had high levels of self-recruitment with slightly higher self-recruitment in Ot1 than in Ot2 (0.975 ± 0.008 and 0.905 ± 0.029, respectively).
Genetic bottleneck analysis
Heterozygosity excess as an indication of genetic bottleneck was detected under the Stepwise Mutation Model (SMM) at all sites, except for the sign test for Nambucca Heads. Under the Two-Phase Mutation Model (TPM), heterozygosity excess was detected at Tasmania. Genetic bottleneck effect was also detected at the distinct Group Ot2 and at the Group Ot1 under the SMM and the TPM. Tests under the Infinite Allele Model (IAM) and the mode-shift test did not detect genetic bottleneck effects at any site (Table 6; Supplementary Table S12).
Population history
In the first step, scenario 4 was selected as the best population topology (Pp = 0.332) (Fig. 2, colour coding in Fig. 2 relates to the clusters identified in the population structure analysis; Supplementary Tables S13, S16). In the second step, scenario 6 had the highest cumulative posterior probability (P = 0.968) and was selected as the most accurate model of demographic history for our data. In this scenario, sub-group 4 (distinct Group Ot2) diverged from sub-group 1 (Nambucca Heads, Swansea, and Merimbula) relatively early in the history of the population (t3, 95% CI). One genetic bottleneck occurred to sub-group 4 relatively early in time (t2b), with a subsequent increase in the size of its population, from Ne = 286 (N4b) to Ne = 7,930 (N4) at t2 (95% CI). Sub-group 1 experienced a recent genetic bottleneck (t0b), with a posterior decrease in the size of its population from Ne = 8,570 (N1b) to Ne = 7,220 (N1). Sub-group 2 (Mallacoota; Ne = 4,200) diverged from sub-group 1 at t2 (95% CI), whereas sub-group 3 (Ot1 Tasmania; Ne = 5,060) diverged from the admixture of sub-groups 1 and 2 at t1 (95% CI) (Fig. 2; Supplementary Tables S17–S20). Posterior estimations of all parameters for the selected scenario were consistent when an alternative set of priors was used.
Hierarchical Approximate Bayesian Computation analysis of population history scenarios of the range extending Octopus tetricus along the east coast of Australia. In step 1, the scenario 4 was selected as the most likely population topology. In step 2, scenario 6 was selected as the most accurate model of demographic history for our data. Sub-group 1 – Nambucca Heads, Swansea, and Merimbula; Sub-group 2 – Mallacoota; Sub-group 3 – Ot1 Tasmania. The common Group Ot1 is comprised of the sub-groups 1 to 3. Sub-group 4 – distinct Group Ot2 predominately comprised of individuals from Tasmania. Ulladulla, Eden and Cape Conran were not included in this analysis due to their small sample sizes (n < 17). The colours represent different sub-groups and changes in the size of their population. Times are not to scale. Selected scenarios at each step are indicated by posterior probabilities (Pp) in bold font.
Discussion
The key findings of this study are that genetic differentiation was detected between sites present within the historical distribution and the range extension zones, and also within the range extension zone. The population was sub-structured in one common group along eastern Australia and Tasmania (Group Ot1), and a distinct group mainly comprised of individuals from Tasmania (Group Ot2). Migration rates were asymmetrical with greater migration occurring from the historical distribution zone towards sites at the range extension zone. The genetic diversity at the range extension zone was comparable to that detected at the historical distribution area. Genetic bottleneck effects were detected at sites within the historical distribution and range extension zones. Population history simulations suggest that the distinct group diverged from the common group relatively early in the history of the population.
Systematic marine life censuses36,38, fisheries records39, and citizen science monitoring40 suggest that O. tetricus has recently extended its distribution polewards into Tasmanian waters. Moreover, this range extension was classified with “high” level of confidence41 and we detected genetic signatures that are characteristic of range shifts9. The presence of the common Group Ot1 throughout the historical distribution and the range extension zones is supported by the detected gene flow between all sites and by shared haplotypes in O. tetricus between NSW and Tasmania using mitochondrial DNA32. In contrast, Ot2 individuals were common in Tasmanian waters but uncommon along the east coast of mainland Australia. This finding is consistent with the increase in size of the Group Ot2, detected by the Approximate Bayesian Computation analysis (ABC), probably from a genotype that is relatively uncommon in the historical distribution zone. The differentiation and expansion in size of the Group Ot2 was likely favored by high self-recruitment, rapid population turnover and high reproductive capacity29,30. These characteristics provide species with high evolutionary capacity43 and may facilitate long-term persistence in range extension zones44. However, the range extension of O. tetricus appears to be rather complex. A genetic break was detected in Tasmanian waters between the Group Ot2 and Ot1 Tasmanian individuals. This genetic break is beyond the scope of this study and requires further investigation to understand the mechanisms that may cause limited mixing of both groups despite coexisting in the same area, e.g. differential selection, non-random mating or short-term departures from local panmixia. In addition, the ABC simulations estimated that the Group Ot1 appeared in Tasmanian waters more recently than the Group Ot2, which diverged early in the history of the population. These signatures suggest that O. tetricus historically diverged between Tasmania and mainland Australia, and secondary contact occurred due to the recent expansion of the mainland population polewards towards the south.
Following theoretical expectations10, the ABC analysis suggested a decrease in size of the population at the equatorward sites. A relatively high level of heterozygosity suggests that Tasmania is within the leading edge of the range extension; this finding is consistent with the genetic pattern observed at the leading edge of range shifts of Quaternary trees in response to climate change6. The range shift of O. tetricus thus appears to consist in the polewards relocation of the population, with characteristics of recent and rapid demographic expansions9. Similarly, populations of C. rodgersii undergoing rapid range expansions had high levels of genetic diversity and heterozygosity at the leading edge of the range12. Constant gene flow from along the entire distribution aids sustained genetic diversity in extension areas21, which can confer phenotypic plasticity to founder individuals and the ability to respond to natural selection7,22. Accordingly, discrete morphological differences were found in male O. tetricus between the east coast of mainland Australia and Tasmania32.
The population structure and connectivity of O. tetricus is expected to be shaped by the dynamics of the EAC24,25,26, which is likely the main driver of larval transport along the east coast of Australia27,28. With an assumed planktonic duration of 35–60 days, as estimated for O. vulgaris33,34, paralarvae of O. tetricus can be transported the linear distance between Nambucca Heads and Tasmanian waters (~1,150 km) at an average seawater flow of 55 cm·s−1 24. Our findings are supported by the connectivity detected between the east coast of mainland Australia and Tasmania32. However, the use of Euclidean distances may be a poor predictor of gene flow45 because the effect of the coastline creates circuitous, turbulent, and nonlinear flow, which in addition to eddies present along the east coast of Australia may result in patchy larval dispersal46,47,48.
Collection sites of this study are exposed to a gradient of temperatures from NSW (annual average SST 20.2 ± 0.2 SE °C at −32° 31′ 15′′ and 152° 28′ 45′′) to north-eastern Tamania (annual average SST 15.2 ± 0.2 SE °C at −39° 33′ 45′′ and 148° 13′ 45′′). Such a gradient of environmental conditions also may have aided in shaping the population structure of O. tetricus49. Constant gene flow from along different environmental conditions within the historical distribution zone, and subsequent interbreeding, can assist in maintaining genetic diversity, create new gene complexes21,50,51, and buffer against any new set of environmental stressors in the range extension zone8,52. Moreover, Merimbula was the main source of migrants towards the range extension zone. This site is located at the historical polewards edge of O. tetricus range and shares more similar environmental conditions (i.e. temperature) with the range extension zone than other sites further equatorward. Such similarities may further facilitate the capacity of founder individuals to respond to selective pressure at the range extension zone17. Maintenance of genetic diversity by sufficient gene flow is therefore likely to contribute to the establishment, early success and persistence of O. tetricus in the extension zone16,21.
The polewards dispersion of larvae is projected to intensify gradually over the next decades along the east coast of Australia53, likely facilitating the population connectivity between historical and range extension zones. However, the accelerated warming along the east coast of Australia is anticipated to shorten the paralarval phase of O. tetricus. A shorter paralarval phase may reduce dispersal capacity, population connectivity54,55, and recovery of genetic diversity in the range extension area. Effects of ocean warming on reproductive seasons, frequency of paralarvae releases, swimming capabilities, paralarvae settlement windows, habitat suitability, and paralarvae mortality may also influence the connectivity of populations46. Therefore ‘seascape genetics’ studies must include oceanographic, life history, ecological and physiological data, in the context of ocean warming and changes in oceanic circulation into the examination of the population connectivity. This approach may provide better understanding of the structure, connectivity, genetic diversity, and capacity of populations to prevail in the new sections of their geographic distribution56. Octopus tetricus has a life-span of 11 months29 and allele frequencies could change between years of collection. To strengthen the robustness of our conclusions, future research should include larger samples sizes and from consecutive years within sites to assess stability of allele frequencies through time.
This study suggests that the range shift of O. tetricus is characterized by a genetically diverse range extension zone. Constant gene flow from a diversity of source areas along the entire distribution may promote relatively high genetic diversity and counteract genetic bottleneck effects at extension areas. Therefore, the genetic signatures examined in this study suggest that O. tetricus is well placed to be able to persist in its range extension zone provided that no demographic or environmental effect negatively affects the population.
Methods
This research was conducted under the permits no. A11591 and A13740 approved by the University of Tasmania Animal Ethics Committee and in accordance with the ‘Australian code of practice for the care and use of animals for scientific purposes’57.
Specimen collection
Octopus were collected from inshore waters along the NSW coast at Nambucca Heads (n = 17; −30° 38′ 46′′, 153° 0′ 12′′) and Swansea (n = 30; −33° 5′ 9′′, 151° 38′ 20′′) during February 2014; and at Ulladulla (n = 5; −35° 19′ 20′′, 150° 31′ 29′′), Merimbula (n = 29; −36° 53′ 42′′, 149° 54′ 25′′), and Eden (n = 8; −37° 4′ 18′′, 149° 54′ 33′′) during May 2013. Sites along the Victorian coastline were Mallacoota (n = 29; −37° 33′ 22′′, 149° 45′ 36′′) and Cape Conran (n = 3; −37° 48′ 49′′, 148° 43′ 37′′) with samples collected during May 2013. Samples also were collected off north-eastern Tasmania (n = 61; −39° 43′ 36′′, 148° 27′ 17′′) during April, September and December 2011. The centre of the known historical distribution included Nambucca Heads, Swansea, and Ulladulla; sites at the polewards edge of the historical distribution were Merimbula and Eden, whereas sites at the range extension zone were Mallacoota, Cape Conran, and Tasmania (Fig. 1). Octopus from Tasmania were collected on board of the FV Farquharson using black plastic shelter pots (0.3-m long × 0.1-m high × 0.1-m wide) laid on the seafloor at depths of 35–46 m. Octopus were euthanized by commercial fishers and immediately put in watery ice in an insulated container. A tissue sample was taken from the arm of every specimen and octopus carcasses were returned to the fishers. Octopus from Ulladulla were collected during diving activities whereas specimens from all other sites along the coast of mainland Australia were collected by hand while snorkelling at depths of 1–3 m. These animals were anesthetized by immersion in a 2% MgCl2 solution, a tissue sample was taken and octopus were released after recovery. All tissue samples were fixed in 95% ethanol.
DNA extraction, PCR amplification and genotyping
DNA was extracted using the high salt method58 from a total of 182 animals. Seven microsatellite primers (Table 1) identified for O. vulgaris59 were amplified in O. tetricus and found to be polymorphic. The microsatellites loci revealed a moderate level of heterozygosity (Table 3) with two loci exhibiting more than 10 alleles at each site (Supplementary Tables S3, S4). Microsatellite loci were assigned unique fluorophores (FAM, VIC, NED, PET)60 to enable fluorescent tagging of PCR products.
PCR reactions were performed59, with modifications to the annealing temperature (Ta) (Table 1). Each PCR contained 4.725 µL of double distilled H20, 6.25 µL of MyTaq Redmix (Bioline), 0.075 µL of 10 mM forward primer, 0.25 µL of 10 mM reverse primer, 0.20 µL of 5pmol/µL fluorophore labelled primer, and 1 µL (18–37 ng) of DNA. PCR conditions were modified slightly to optimize PCR products for some samples, such that 1 µL of 25 mM MgCl2 (Promega) was added in place of water. The number of cycles was reduced from 35 to 30, and the final extension was reduced from 5 to 3 min. Capillary separation of PCR products was performed by the Australian Genome Research Facility Ltd (AGRF). Genotypes were scored by eye using Geneious Pro v. 5.6.461. PCRs were repeated up to three more times for individuals with unclear or missing single-locus genotypes before being categorized as missing data and scored as 000000 (n = 25).
Genetic polymorphism
Micro-Checker v. 2.2.362 was used to search for evidence of allele dropout and stuttering for each locus. High frequencies of null alleles are commonly observed in marine invertebrate species including molluscs63,64,65. The presence of null alleles can lead to overestimation of FST in cases of low levels of gene flow and significant population differentiation. Therefore, following the Expectation Maximization algorithm66, FreeNA67 was used to estimate null allele frequencies detected as false homozygotes. Null alleles were then corrected by re-naming them as 999 (e.g. a false homozygote with allele size 257257 was corrected to 257999). This method greatly reduces the bias of FST caused regardless of the frequency of null alleles, the level of gene flow, and the number of loci67.
Population sub-structuring
The DAPC68 was used with the package ‘adegenet’69 in RStudio v. 0.99.43570 to test separately for population structure along the east coast of Australia and on a range of subsets of the dataset, i.e. (a) at all sites within mainland Australia, (b) at the historical distribution only, (c) at the range extension zone only, (d) Tasmania only, and (e) not including the distinct Group Ot2. The admixture and correlated allele frequencies models were also implemented in Structure v. 2.3.471 to examine population structure. The number of clusters (K) explored was 1–10, with 10 independent runs of 500,000 Monte Carlo Markov Chain (MCMC) replicates and a burnin length of 50,000. The Evanno method72 implemented within Structure Harvester v. 0.6.9373 was used to evaluate the results, which were graphically displayed using Distruct v. 1.174.
Phylogenetic analysis
A phylogenetic analysis was performed to corroborate that individuals of the different groups detected from population structure analyses were in fact O. tetricus and not representative of a cryptic species (Supplementary methods online: ‘Phylogenetic analysis’).
Genetic diversity
Descriptive statistics were estimated for each collection site with ≥17 samples, as well as for the groups inferred from the population structure analyses. Arlequin v. 3.5.1.375 was used to test for genotypic linkage disequilibrium among loci within sites (20,000 iterations; P < 0.05), and for departures from Hardy–Weinberg equilibrium using the score test for heterozygote deficiency with level of significance determined by the Markov chain method (1,000,000 iterations). Arlequin v. 3.5.1.375 was also used to estimate the number of alleles (NA), observed (HO) and expected (HE) heterozygosity and inbreeding coefficient (FIS). Allelic richness (AR) was rarefied to 17 samples using FSTAT76. The number of private alleles (NPA) was calculated using Convert v. 1.3177.
Genetic connectivity and differentiation
Pairwise FST was calculated using Arlequin v. 3.5.1.375 between all sites with ≥17 samples. A Mantel matrix correlation test was performed in Genepop web v. 4.278,79 to examine if genetic differentiation (FST) was explained by isolation by geographical distance (km) between collection sites (excluding Ulladulla, Eden, and Cape Conran with <17 samples). Isolation by year of collection was also tested for between collection sites, i.e. 2011 (Tasmania), 2013 (Mallacoota and Merimbula), and 2014 (Swansea and Nambucca Heads). A Spearman’s correlation analysis also was carried out to discount any effect of the sampling scheme by testing for an association between loci and year and depth of collection. In addition, AMOVA implemented in Arlequin v. 3.5.1.375 was used to test for partitioning of genetic variation among collection sites.
Migration and self-recruitment
BayesAss v. 3.0.180 was implemented to assess admixture81. Migration rates (Δm), allele frequencies (Δa), and inbreeding coefficients (Δf) were given a mixing parameter value of 1 each for the analysis at the collection sites level, and a mixing parameter value of 0.2 each for the analysis at the groups level. We used 50,000,000 iterations, a 5,000,000 burnin length, and a 1000 interval between samples. Trace output convergence was assessed using Tracer v. 1.682. Recent migration rates (i.e. within the recent 2–3 generations) were then estimated between collection sites, as well as between groups inferred from the population structure analyses.
Bottleneck analysis
Heterozygosity excess is a genetic signature of genetic bottlenecks because alleles are usually lost faster than heterozygosity during a bottleneck as the mutation-drift equilibrium is lost83. Heterozygosity excess was tested with the software Bottleneck v. 1.2.0284, using 10,000 iterations for each site with ≥17 samples, and for the groups inferred from the population structure analyses. The tests used were: (i) The “sign test”83, (ii) the 2-tailed “Wilcoxon sign rank test”85, and (iii) the “mode-shift test”86. These tests allow the detection of the heterozygosity excess due to the faster loss of alleles at low-frequency class (<0.1) than alleles in 1 or more intermediate allele frequency classes85,86. For instance, the mode-shift test detects the L-shaped distribution of allele proportions observed at mutation-drift equilibrium, and bottlenecked populations that do not present the L-shaped distribution86. Microsatellite loci may tend to evolve under a model more similar to the SMM87 than to the IAM83,85,87,88. However, the SMM, the IAM, and a model in between both models, the TPM (using default settings)89 were examined. Examination of the three mutation models allowed greater caution regarding the detection of genetic bottlenecks.
Population history
To reconstruct the history of divergence, migration and admixture events between the sub-groups identified based on the significant levels of genetic differentiation (FST), datasets of historical and/or demographic events were simulated under a total of eight different scenarios using a coalescent-based ABC algorithm implemented in DIYABC v. 2.190. The ABC approach relies on gene genealogy, demographic history and ancestral polymorphisms to explain evolutionary causes of present-day genetic variation, and thus can be used to discriminate among putative evolutionary scenarios. This approach is commonly used to distinguish the effect of contemporary recurrent gene flow from the effects of historical connectivity91. The ABC algorithm compares summary statistics of simulated datasets for each historical scenario and/or demographic events with summary statistics of the observed data. The posterior probability and distribution of parameters of each scenario are estimated and alternative scenarios can be compared.
The eight scenarios were examined in two successive steps. In the first step, five scenarios were examined: Scenarios 1 and 2 assumed successive independent divergence suggesting historical diversification; scenarios 3 and 4 assumed a single admixture event, and scenario 5 assumed two successive admixture events. We chose the scenario with the highest posterior probability (best fitted topology) from the first step. In the second step, three scenarios based on the best fitted topology selected in the first step were examined to refine changes in effective population size. These scenarios assumed a common origin of the sub-groups with successive divergence to test the hypothesis of recent colonization due to range extension. Thus, in the second step scenarios 6 and 7 considered changes in the effective number of founder specimens after bottlenecks and in the effective population size of cluster 4 at different generation times. Scenario 8 also considered changes in the effective number of founder specimens after bottlenecks and in the effective population size of other clusters (e.g. cluster 2).
To perform the ABC analysis a reference table with 3,000,000 coalescent-simulated data sets for each scenario was built, from which summary statistics parameter values were used in prior distributions (Supplementary Tables S13 and S17). Posterior probabilities were computed for each scenario applying linear discriminant analysis on summary statistics to find the best-supported scenario based on the direct approach estimates (500 selected data sets). The level of confidence in the chosen scenario was evaluated by estimating the type I and type II error rates based on simulated datasets. The marginal posterior distribution of each parameter was estimated based on the best model. The goodness of fit of the model-posterior parameter distribution combination was evaluated with the data. The scenario with the highest posterior probability (non-overlapping 95% confidence intervals) under the direct approach was then chosen as the best-supported scenario. See further details on the ABC analysis at90,92.
Data availability
The datasets generated during and/or analysed during the current study are available in the DRYAD repository, [https://doi.org/10.5061/dryad.t483s94].
References
Sunday, J. M., Bates, A. E. & Dulvy, N. K. Thermal tolerance and the global redistribution of animals. Nat. Clim. Chang. 2, 686–690, https://doi.org/10.1038/nclimate1539 (2012).
Poloczanska, E. S. et al. Global imprint of climate change on marine life. Nat. Clim. Chang. 3, 919–925, https://doi.org/10.1038/NCLIMATE1958 (2013).
Sorte, C. J. B., Williams, S. L. & Carlton, J. T. Marine range shifts and species introductions: comparative spread rates and community impacts. Glob. Ecol. Biogeogr. 19, 303–316, https://doi.org/10.1111/j.1466-8238.2009.00519.x (2010).
Madin, E. M. P. et al. Socio-economic and management implications of range-shifting species in marine systems. Glob. Environ. Change 22, 137–146, https://doi.org/10.1016/j.gloenvcha.2011.10.008 (2012).
Pecl, G. T. et al. Biodiversity redistribution under climate change: Impacts on ecosystems and human well-being. Science 355, eaai9214, https://doi.org/10.1126/science.aai9214 (2017).
Davis, M. B. & Shaw, R. G. Range shifts and adaptive responses to Quaternary climate change. Science 292, 673–679, https://doi.org/10.1126/science.292.5517.673 (2001).
Dlugosch, K. M. & Parker, I. M. Founding events in species invasions: genetic variation, adaptive evolution, and the role of multiple introductions. Mol. Ecol. 17, 431–449, https://doi.org/10.1111/j.1365-294X.2007.03538.x (2008).
Whitney, K. D. & Gabler, C. A. Rapid evolution in introduced species, ‘invasive traits’ and recipient communities: challenges for predicting invasive potential. Divers. Distrib. 14, 569–580, https://doi.org/10.1111/j.1472-4642.2008.00473.x (2008).
Excoffier, L., Foll, M. & Petit, R. J. Genetic Consequences of Range Expansions. Annu. Rev. Ecol. Evol. Syst. 40, 481–501, https://doi.org/10.1146/annurev.ecolsys.39.110707.173414 (2009).
McInerny, G. J., Turner, J. R. G., Wong, H. Y., Travis, J. M. J. & Benton, T. G. How range shifts induced by climate change affect neutral evolution. Proc. R. Soc. Lond., B, Biol. Sci. 276, 1527–1534, https://doi.org/10.1098/rspb.2008.1567 (2009).
Arenas, M., Ray, N., Currat, M. & Excoffier, L. Consequences of range contractions and range shifts on molecular diversity. Mol. Biol. Evol. 29, 207–218, https://doi.org/10.1093/molbev/msr187 (2012).
Banks, S. C. et al. Genetic structure of a recent climate change-driven range extension. Mol. Ecol. 19, 2011–2024, https://doi.org/10.1111/j.1365-294X.2010.04627.x (2010).
Potts, W. M. et al. Ocean warming, a rapid distributional shift, and the hybridization of a coastal fish species. Glob. Chang. Biol. 20, 2765–2777, https://doi.org/10.1111/gcb.12612 (2014).
Ray, N., Currat, M. & Excoffier, L. Intra-deme molecular diversity in spatially expanding populations. Mol. Biol. Evol. 20, 76–86, https://doi.org/10.1093/molbev/msg009 (2003).
Waples, R. S. & Gaggiotti, O. What is a population? An empirical evaluation of some genetic methods for identifying the number of gene pools and their degree of connectivity. Mol. Ecol. 15, 1419–1439, https://doi.org/10.1111/j.1365-294X.2006.02890.x (2006).
Drake, J. M. Heterosis, the catapult effect and establishment success of a colonizing bird. Biol. Lett. 2, 304–307, https://doi.org/10.1098/rsbl.2006.0459 (2006).
Sexton, J. P., Strauss, S. Y. & Rice, K. J. Gene flow increases fitness at the warm edge of a species’ range. Proc. Natl. Acad. Sci. USA 108, 11704–11709, https://doi.org/10.1073/pnas.1100404108 (2011).
Austerlitz, F., Jung-Muller, B., Godelle, B. & Gouyon, P. H. Evolution of coalescence times, genetic diversity and structure during colonization. Theor. Popul. Biol. 51, 148–164 (1997).
Hallatschek, O. & Nelson, D. R. Life at the front of an expanding population. Evolution 64, 193–206, https://doi.org/10.1111/j.1558-5646.2009.00809.x (2010).
Signorile, A. L. et al. Do founder size, genetic diversity and structure influence rates of expansion of North American grey squirrels in Europe? Divers. Distrib. 20, 918–930, https://doi.org/10.1111/ddi.12222 (2014).
Roman, J. & Darling, J. A. Paradox lost: genetic diversity and the success of aquatic invasions. Trends Ecol. Evol. 22, 454–464, https://doi.org/10.1016/j.tree.2007.07.002 (2007).
Kanuch, P., Berggren, A. & Cassel-Lundhagen, A. Genetic diversity of a successful colonizer: isolated populations of Metrioptera roeselii regain variation at an unusually rapid rate. Ecol. Evol. 4, 1117–1126, https://doi.org/10.1002/ece3.1005 (2014).
Hobday, A. J. & Pecl, G. T. Identification of global marine hotspots: sentinels for change and vanguards for adaptation action. Rev. Fish Biol. Fish. 24, 415–425, https://doi.org/10.1007/s11160-013-9326-6 (2014).
Ridgway, K. R. & Dunn, J. R. Mesoscale structure of the mean East Australian Current System and its relationship with topography. Prog. Oceanogr. 56, 189–222, https://doi.org/10.1016/s0079-6611(03)00004-1 (2003).
Ridgway, K. R. Long-term trend and decadal variability of the East Australian Current. Geophys. Res. Lett. 34, L13613, https://doi.org/10.1029/2007GL030393 (2007).
Hill, K. L., Rintoul, S. R., Coleman, R. & Ridgway, K. R. Wind forced low frequency variability of the East Australia Current. Geophys. Res. Lett. 35, L08602, https://doi.org/10.1029/2007gl032912 (2008).
Johnson, C. R. et al. Climate change cascades: Shifts in oceanography, species’ ranges and subtidal marine community dynamics in eastern Tasmania. J. Exp. Mar. Biol. Ecol. 400, 17–32, https://doi.org/10.1016/j.jembe.2011.02.032 (2011).
Last, P. R. et al. Long-term shifts in abundance and distribution of a temperate fish fauna: a response to climate change and fishing practices. Glob. Ecol. Biogeogr. 20, 58–72, https://doi.org/10.1111/j.1466-8238.2010.00575.x (2011).
Ramos, J. E. et al. Body size, growth and life span: implications for the polewards range shift of Octopus tetricus in south-eastern Australia. PLoS ONE 9, e103480, https://doi.org/10.1371/journal.pone.0103480 (2014).
Ramos, J. E. et al. Reproductive capacity of a marine species (Octopus tetricus) at its range extension area. Mar. Freshw. Res. 66, 999–1008, https://doi.org/10.1071/MF14126 (2015).
Guzik, M. T., Norman, M. D. & Crozier, R. H. Molecular phylogeny of the benthic shallow-water octopuses (Cephalopoda:Octopodinae). Mol. Phylogenet. Evol. 37, 235–248, https://doi.org/10.1016/j.ympev.2005.05.009 (2005).
Amor, M. D., Norman, M. D., Cameron, H. E. & Strugnell, J. M. Allopatric speciation within a cryptic species complex of Australasian octopuses. PLoS ONE 9, e98982, https://doi.org/10.1371/journal.pone.0098982 (2014).
Villanueva, R. Experimental rearing and growth of planktonic Octopus vulgaris from hatching to settlement. Can. J. Fish. Aquat. Sci. 52, 2639–2650 (1995).
Carrasco, J. F., Arronte, J. C. & Rodríguez, C. Paralarval rearing of the common octopus, Octopus vulgaris (Cuvier). Aquac. Res. 37, 1601–1605 (2006).
Edgar, G. J. Australian Marine Life: The plants And Animals Of Temperate Waters, 624 p (Reed New Holland Publishers 2000).
Norman, M. & Reid, A. A guide to Squid, Cuttlefish and Octopuses of Australasia, 96 p (CSIRO Publishing 2000).
Rowling, K., Hegarty, A. M. & Ives, M. Octopus (Octopus spp.). In: Status of Fisheries Resources in NSW 2008/2009. Industry & Investment NSW, pp. 225–227 (2010).
Edgar, G. J. & Stuart-Smith, R. D. Systematic global assessment of reef fish communities by the Reef Life Survey program. Sci. Data 1, 1–8, https://doi.org/10.1038/sdata.2014.7 (2014).
Tasmania Department of Primary Industries and Water. Scalefish Fishery Management Plan review. Octopus fishery. Tasmania Wild Fisheries Management Branch, Department of Primary Industries and Water (2009).
Range Extension Database and Mapping Project, http://www.redmap.org.au (2013).
Robinson, L. M. et al. Rapid assessment of an ocean warming hotspot reveals “high” confidence in potential species’ range extensions. Glob. Environ. Change 31, 28–37, https://doi.org/10.1016/j.gloenvcha.2014.12.003 (2015).
Marzloff, M. P. et al. Modelling marine community responses to climate-driven species redistribution to guide monitoring and adaptive ecosystem-based management. Glob. Chang. Biol. 22, 2462–2474, https://doi.org/10.1111/gcb.13285 (2016).
Bromham, L. The genome as a life-history character: why rate of molecular evolution varies between mammal species. Philos. Trans. R. Soc. Lond., B, Biol. Sci. 366, 2503–2513, https://doi.org/10.1098/rstb.2011.0014 (2011).
O’Connor, M. I., Selig, E. R., Pinsky, M. L. & Altermatt, F. Toward a conceptual synthesis for climate change responses. Glob. Ecol. Biogeogr. 21, 693–703, https://doi.org/10.1111/j.1466-8238.2011.00713.x (2012).
White, C. et al. Ocean currents help explain population genetic structure. Proc. R. Soc. Lond., B, Biol. Sci. 277, 1685–1694, https://doi.org/10.1098/rspb.2009.2214 (2010).
Siegel, D. A. et al. The stochastic nature of larval connectivity among nearshore marine populations. Proc. Natl. Acad. Sci. USA 105, 8974–8979, https://doi.org/10.1073/pnas.0802544105 (2008).
Weersing, K. & Toonen, R. J. Population genetics, larval dispersal, and connectivity in marine systems. Mar. Ecol. Prog. Ser. 393, 1–12, https://doi.org/10.3354/meps08287 (2009).
Roughan, M., Macdonald, H. S., Baird, M. E. & Glasby, T. M. Modelling coastal connectivity in a Western BoundaryCurrent: Seasonal and inter-annual variability. Deep Sea Res. Part II Top. Stud. Oceanogr. 58, 628–644, https://doi.org/10.1016/j.dsr2.2010.06.004 (2011).
Lind, C. E., Evans, B. S., Taylor, J. J. U. & Jerry, D. R. Population genetics of a marine bivalve, Pinctada maxima, throughout the Indo-Australian Archipelago shows differentiation and decreased diversity at range limits. Mol. Ecol. 16, 5193–5203, https://doi.org/10.1111/j.1365-294X.2007.03598.x (2007).
Ibrahim, K. M., Nichols, R. A. & Hewitt, G. M. Spatial patterns of genetic variation generated by different forms of dispersal during range expansion. Heredity 77, 282–291 (1996).
Verhoeven, K. J. F., Macel, M., Wolfe, L. M. & Biere, A. Population admixture, biological invasions and the balance between local adaptation and inbreeding depression. Proc. R. Soc. Lond., B, Biol. Sci. 278, 2–8, https://doi.org/10.1098/rspb.2010.1272 (2010).
Hoffmann, A. A. & Sgrò, C. M. Climate change and evolutionary adaptation. Nature 470, 479–485, https://doi.org/10.1038/nature09670 (2011).
van Gennip, S. J. et al. Going with the flow: the role of ocean circulation in global marine ecosystems under a changing climate. Glob. Chang. Biol 23, 2602–2617, https://doi.org/10.1111/gcb.13586 (2017).
O’Connor, M. I. et al. Temperature control of larval dispersal and the implications for marine ecology, evolution, and conservation. Proc. Natl. Acad. Sci. USA 104, 1266–1271, https://doi.org/10.1073/pnas.0603422104 (2007).
Munday, P. L. et al. Climate change and coral reef connectivity. Coral Reefs 28, 379–395, https://doi.org/10.1007/s00338-008-0461-9 (2009).
Selkoe, K. A. et al. Taking the chaos out of genetic patchiness: seascape genetics reveals ecological and oceanographic drivers of genetic patterns in three temperate reef species. Mol. Ecol. 19, 3708–3726 (2010).
Australian Government. Australian Code of Practice for the Care and Use of Animals for Scientific Purposes (Australian Government & National Health and Medical Research Council 2004).
Sambrook, J., Fristsch, E. F. & Maniatis, T. Molecular Cloning: ALaboratory Manual (Cold Spring Harbor Laboratory, 1989).
Zuo, Z., Zheng, X., Liu, C. & Li, Q. Development and characterization of 17 polymorphic microsatellite loci in Octopus vulgaris Cuvier, 1797. Conserv. Genet. Resour. 4, 367–369, https://doi.org/10.1007/s12686-011-9550-0 (2012).
Blacket, M. J., Robin, C., Good, R. T., Lee, S. F. & Miller, A. D. Universal primers for fluorescent labelling of PCR fragments-an efficient and cost-effective approach to genotyping by fluorescence. Mol. Ecol. Resour. 12, 456–463, https://doi.org/10.1111/j.1755-0998.2011.03104.x (2012).
Drummond, A. J. et al. GENEIOUS version 5.6.4. ed. Auckland, New Zealand: Biomatters Ltd (2012).
van Oosterhout, C., Hutchinson, W. F., Wills, D. P. M. & Shipley, P. MICRO-CHECKER: software for identifying and correcting genotyping errors in microsatellite data. Mol. Ecol. Notes 4, 535–538, https://doi.org/10.1111/j.1471-8286.2004.00684.x (2004).
Li, G., Hubert, S., Bucklin, K., Ribes, V. & Hedgecock, D. Characterization of 79 microsatellite DNA markers in the Pacific oyster Crassostrea gigas. Mol. Ecol. Notes 3, 228–232, https://doi.org/10.1046/j.1471-8286.2003.00406.x (2003).
Kaukinen, K. H., Supernault, K. J. & Miller, K. M. Enrichment of tetranucleotide microsatellite loci from invertebrate species. J. Shellfish Res. 23, 621–626 (2004).
Astanei, I., Gosling, E., Wilson, J. & Powell, E. Genetic variability and phylogeography of the invasive zebra mussel, Dreissena polymorpha (Pallas). Mol. Ecol. 14, 1655–1666, https://doi.org/10.1111/j.1365-294X.2005.02530.x (2005).
Dempster, A. P., Laird, N. M. & Rubin, D. B. Maximum likelihood from incomplete data via the EM algorithm. J. R. Stat. Soc., Series. B., Stat. Methodol. 39, 1–38 (1977).
Chapuis, M. P. & Estoup, A. Microsatellite null alleles and estimation of population differentiation. Mol. Biol. Evol. 24, 621–631, https://doi.org/10.1093/molbev/msl191 (2007).
Jombart, T., Devillard, S. & Balloux, F. Discriminant analysis of principal components: a new method for the analysis of genetically structured populations. BMC Genet. 11, 94, https://doi.org/10.1186/1471-2156-11-94 (2010).
Jombart, T. adegenet: a R package for the multivariate analysis of genetic markers. Bioinformatics 24, 1403–1405, https://doi.org/10.1093/bioinformatics/btn129 (2008).
RStudio Team. RStudio: Integrated Development for R. RStudio, Inc., Boston, MA, http://www.rstudio.com/ (2015).
Pritchard, J. K., Stephens, M. & Donnelly, P. Inference of population structure using multilocus genotype data. Genetics 155, 945–959 (2000).
Evanno, G., Regnaut, S. & Goudet, J. Detecting the number of clusters of individuals using the software STRUCTURE: a simulation study. Mol. Ecol. 14, 2611–2620, https://doi.org/10.1111/j.1365-294X.2005.02553.x (2005).
Earl, D. A. & vonHoldt, B. M. STRUCTURE HARVESTER: a website and program for visualizing STRUCTURE output and implementing the Evanno method. Conserv. Genet. Resour. 4, 359–361, https://doi.org/10.1007/s12686-011-9548-7 (2012).
Rosenberg, N. A. DISTRUCT: a program for the graphical display of population structure. Mol. Ecol. Notes 4, 137–138, https://doi.org/10.1046/j.1471-8286.2003.00566.x (2004).
Excoffier, L. & Lischer, H. E. L. Arlequin suite ver 3.5: A new series of programs to perform population genetics analyses under Linux and Windows. Mol. Ecol. Resour. 10, 564–567, https://doi.org/10.1111/j.1755-0998.2010.02847.x (2010).
Goudet, J. FSTAT (Version 1.2): A computer program to calculate F-statistics. J. Hered. 86, 485–486 (1995).
Glaubitz, J. C. CONVERT: A user-friendly program to reformat diploid genotypic data for commonly used population genetic software packages. Mol. Ecol. Notes 4, 309–310, https://doi.org/10.1111/j.1471-8286.2004.00597.x (2004).
Raymond, M. & Rousset, F. GENEPOP (Version 1.2): Population Genetics Software for Exact Tests and Ecumenicism. J. Hered. 86, 248–249 (1995).
Rousset, F. Genepop'007: a complete reimplementation of the Genepop software for Windows and Linux. Mol. Ecol. Resour. 8, 103–106, https://doi.org/10.1111/j.1471-8286.2007.01931.x (2008).
Wilson, G. A. & Rannala, B. Bayesian inference of recent migration rates using multilocus genotypes. Genetics 163, 1177–1191 (2003).
Faubet, P., Waples, R. S. & Gaggiotti, O. E. Evaluating the performance of a multilocus Bayesian method for the estimation of migration rates. Mol. Ecol. 16, 1149–1166, https://doi.org/10.1111/j.1365-294X.2006.03218.x (2007).
Rambaut, A., Drumond, A. J. & Suchard, M. Tracer. MCMC Trace Analysis Tool version 1.6.0, 2003–2013, http://www.molecularevolution.org/software/phylogenetics/tracer (2013).
Luikart, G. & Cornuet, J. M. Empirical evaluation of a test for identifying recently bottlenecked populations from allele frequency data. Conserv. Biol. 12, 228–237, https://doi.org/10.1046/j.1523-1739.1998.96388.x (1998).
Cornuet, J. M. et al. BOTTLENECK version 1.2.02, http://susanejohnston.wordpress.com/2013/09/10/bottleneck-genetic-software-stand-alone-executable/ (1999).
Luikart, G., Sherwin, W. B., Steele, B. M. & Allendorf, F. W. Usefulness of molecular markers for detecting population bottlenecks via monitoring genetic change. Mol. Ecol. 7, 963–974, https://doi.org/10.1046/j.1365-294x.1998.00414.x (1998a).
Luikart, G., Allendorf, F. W., Cornuet, J. M. & Sherwin, W. B. Distortion of allele frequency distributions provides a test for recent population bottlenecks. J. Hered. 89, 238–247, https://doi.org/10.1093/jhered/89.3.238 (1998b).
Ohta, T. & Kimura, M. A model of mutation appropriate to estimate the number of electrophoretically detectable alleles in a finite population. Genet. Res. 89, 367–370, https://doi.org/10.1017/s0016672308009531 (1973).
Kimura, M. & Crow, J. The number of alleles that can be maintained in a finite population. Genetics 49, 725–738 (1964).
Di Rienzo, A. et al. Mutational process of simple-sequence repeat loci in human populations. Proc. Natl. Acad. Sci. USA 91, 3166–3170 (1994).
Cornuet, J. M. et al. DIYABC version 2.1. A user-friendly software for inferring population history through Approximate Bayesian Computations using microsatellite, DNA sequence and SNP data. 201 p, http://www1.montpellier.inra.fr/CBGP/diyabc/ (2015).
Marko, P. B. & Hart, M. W. The complex analytical landscape of gene flow inference. Trends Ecol. Evol. 26, 448–456, https://doi.org/10.1016/j.tree.2011.05.007 (2011).
Cornuet, J. M. et al. Inferring population history with DIY ABC: a user-friendly approach to approximate Bayesian computation. Bioinformatics 24, 2713–2719, https://doi.org/10.1093/bioinformatics/btn514 (2008).
Acknowledgements
C. Silvey, M. Amor, and L. Henríquez assisted during collection of samples along the east coast of mainland Australia. K. Wood, A. Reid, and S. Geary provided samples from the east coast of mainland Australia. C. Hardy facilitated the collection of tissue at north-eastern Tasmania. E. Morgan, M. Amor, and A. Smolenski were of invaluable help during lab work and data analysis. A. Roura provided constructive comments on an early version of the manuscript. J.E. Ramos was awarded the CONACYT scholarship 308672 from the Mexican Government, a Tasmania Graduate Research scholarship from the University of Tasmania, and the 2013 Jill Landsberg Trust Fund Scholarship, which allowed the collection of samples and laboratory work. C.A. Souza was supported by the post-doctoral Fellowship ‘Science without borders’ by the Brazilian Funding Agency. G.T. Pecl was supported by an Australian Research Council Future Fellowship.
Author information
Authors and Affiliations
Contributions
All authors conceived the study. J.E.R. and J.M. Strugnell conceived and designed the experiments. J.E.R. performed the experiments. J.E.R., C.A.S. and J.M. Strugnell analysed the data. G.T.P. and J.M. Strugnell contributed reagents/materials/analysis tools. All authors wrote the paper.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ramos, J.E., Pecl, G.T., Moltschaniwskyj, N.A. et al. Population genetic signatures of a climate change driven marine range extension. Sci Rep 8, 9558 (2018). https://doi.org/10.1038/s41598-018-27351-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-27351-y
This article is cited by
-
Size-dependent thermal limits in Australian hybrid abalone: implications for productivity shifts with ocean warming
Reviews in Fish Biology and Fisheries (2024)
-
Mechanisms, detection and impacts of species redistributions under climate change
Nature Reviews Earth & Environment (2024)
-
Incorporating evolutionary based tools in cephalopod fisheries management
Reviews in Fish Biology and Fisheries (2021)
-
Against all odds: a tale of marine range expansion with maintenance of extremely high genetic diversity
Scientific Reports (2020)
-
Patterns of mitochondrial and microsatellite DNA markers describe historical and contemporary dynamics of the Humboldt squid Dosidicus gigas in the Eastern Pacific Ocean
Reviews in Fish Biology and Fisheries (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.