Abstract
The relationship between vitamin D and breast cancer is still controversial. The present meta-analysis examines the effects of the 25(OH)D, 1,25(OH)2D and vitamin D intake on breast cancer risk. For this purpose, a PubMed, Scopus and Web of Science-databases search was conducted including all papers published with the keywords “breast cancer” and “vitamin D” with at least one reported relative risk (RR) or odds ratio (OR). In total sixty eight studies published between 1998 and 2018 were analyzed. Information about type of study, hormonal receptors and menopausal status was retrieved. Pooled OR or RR were estimated by weighting individual OR/RR by the inverse of their variance Our study showed a protective effect between 25 (OH) D and breast cancer in both cohort studies (RR = 0.85, 95%CI:0.74–0.98) and case-control studies (OR = 0.65, 95%CI: 0.56–0.76). However, analyzing by menopausal status, the protective vitamin D – breast cancer association persisted only in the premenopausal group (OR = 0.67, 95%CI: 0.49–0.92) when restricting the analysis to nested case-control studies. No significant association was found for vitamin D intake or 1,25(OH)2D. Conclusion: This systematic review suggests a protective relationship between circulating vitamin D (measured as 25(OH) D) and breast cancer development in premenopausal women.
Similar content being viewed by others
Introduction
Breast cancer is an important public health problem in developed countries as it is one of the most common cancers, being the most if only the female population is considered1. The incidence is decreasing every year, which is partly due to early detection programs2.
In the last decades, cellular in vitro experiments and in vivo models have evaluated the role of vitamin D in the development of breast cancer, finding a protective anticancer role of 1,25(OH)D33. It has been demonstrated that treating breast cancer cells with 1,25(OH)D3 induces two beneficial effects: an anti-proliferative effect4 and a pro-apoptotic effect5,6. The former is linked to the suppression of growth stimulatory signals and the potentiation of growth inhibitory signals, whilst the second one is explained by the bcl-2 family proteins. The interaction between vitamin D and its receptors induces an increase in the expression of pro-apoptotic family member (bax and bak protein) and simultaneously a decrease of anti-apoptotic (bcl-2/bcl-XL)6. In addition, the breast tissue contains the 1-α-hydroxylase, allowing for the generation of the active vitamin D metabolite (1,25 dihydroxyvitamin D) from the circulating precursor (25 hydroxyvitamin D). As vitamin D receptors are found in the breast6, an autocrine role of vitamin D has been suggested7.
Despite this biological background, literature shows inconsistent results8,9,10,11,12,13,14,15,16 (Table 1). Several additional observational studies have appeared since the last meta-analysis publication (including articles until 2013). The main purpose of the present meta-analysis is to update the relationship between vitamin D exposure and breast cancer risk by adding the studies published more recently. Thus sixty-eight observational studies: thirty of these were case-control, twenty-one were nested case-control and the remaining were cohort studies.
Methods
Search strategy
Firstly, the following inclusion criteria were defined: we looked for cohort or case-control studies performed in humans, which reported, at least, one relative risk (RR) or odds ratio (OR) with confidence interval at 95%. (95% CI)
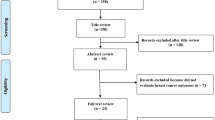
We began our search in Pub-Med, Scopus and Web of Science database using “breast cancer” and “vitamin D” as keywords, finding 2313 articles. After having read the title and abstract, 2123 articles that did not meet the above criteria were eliminated. Next, we carried out a more exhaustive and complete reading, which allowed us to reject another additional 69 articles (Fig. 1). Finally, sixty eight studies meeting our inclusion criteria were identified: fifty one case-control10,17,18,19,20,21,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51,52,53,54,55,56,57,58,59,60,61,62,63,64,65 and seventeen cohort studies65,66,67,68,69,70,71,72,73,74,75,76,77,78,79,80,81. Tables 2 and 3 summarize the main characteristics of the included articles.
Data extraction
The following step was to create a database to gather all relevant information extracted from each article: year of publication, author, journal, follow up, country, sample size, exposure levels, units of measure, data for the creation of the contingency table and RR/OR with 95% CI; as well as a section to assess the quality of the study using the STROBE scale82.
Statistical analysis
Statistical analysis was performed separately for cohort and case-control studies. In the case control studies a sensitivity analysis was also carried-out including only nested case-control studies. We performed separate analyses for any type of vitamin D exposure reported in at least three studies: 25(OH)D, dietary intake of vitamin D, 1,25(OH)2D and vitamin D supplements.
The ways that doses or levels of vitamin D were reported in each individual article were not standardized across studies (for instance, some papers reported vitamin D levels in quartiles; others in tertiles, and so on), making it difficult to extract them in an analyzable form. Therefore, in order to provide a consistent criterion of comparability, we selected the OR or RR reported for the highest category compared to the lowest one.
Regarding the type of breast cancer, we analyzed all invasive breast cancers together, and breast cancer stratified according to the cancer estrogen receptor status and woman’s menopausal status. Pooled OR or RR were estimated by weighting individual OR/RR by the inverse of their variance. OR or RR heterogeneity was measured using Q and I2 statistics83. A fixed-effect model was preferred if the Q statistic was higher than 0.1 or I2 lower than 25%, indicating no relevant heterogeneity; a random-effect model was otherwise chosen84. The presence of small-study bias was explored with Rosenthal model and with Egger test85; due to the low sensitivity of Egger test, the cut-off was set at p = 0.1. Funnel plots86 were applied to detect publication bias.
An analysis of influence was performed via the re-estimation of pooled OR/RR by removing one study at a time. Studies that, when removed, strongly changed the OR/RR would be considered as highly influential. Results are displayed as forest plots showing OR/RR and their 95% confidence intervals for each individual study and for the pooled result. Cumulative meta-analyses were carried out to deem the stability of the OR/RR estimates. In order to do that, all studies considered were arranged from oldest to neweest. Then an OR/RR estimate was obtained for the two eldest studies; another for the three eldest, and so on, adding a study each time. Results are reported as forest plots.
All the statistical analyses were carried out with the package Stata 14/SE (Stata Corporation, College Station, TX, US).
Results
Relationship between 25(OH) D and breast cancer
Twenty-nine case control studies were analyzed to study the relationship between 25 (OH) D and breast cancer10,19,20,21,22,25,27,29,30,31,32,33,34,35,38,42,44,45,46,48,49,51,55,56,58,59,60,61,62,63 obtaining a pooled OR of 0.65 (95%CI: 0.56–0.76) (Fig. 2a, Table 4). This value was calculated using the random effects model because of the high heterogeneity (I2 = 77.76%) of the fixed-effect. Although Egger test cannot rule out a small-study effect (p = 0.001), no study shows a relevant influence. The funnel plot shows asymmetry (Supplementary Fig. 1a), indicating either publication bias or heterogeneity that cannot be explained by a random-effect meta-analysis. Rosenthal model shows that 1194 negative studies would be needed to lose statistical significance. In order to further clarify the heterogeneous result, we carried out a sensitivity analysis including only nested case-control studies21,22,25,31,32,33,34,42,45,46,51,55,56,59 reaching a pooled OR = 0.92 (95%CI: 0.83–1.01) (Fig. 2b) with I2 = 15.87%, Q-based p value = 0.22 and a very symmetrical-looking funnel plot (Supplementary Fig. 1b).
Four cohort studies75,78,79,80 provided results on 25(OH)D and breast cancer relationship, from which we obtained a pooled RR of 0.85 (95% CI:0.74–0.98).
We also analyzed the relationship between 25(OH) D and breast cancer, stratifying results by hormonal receptors (ER+/ER−) and menopausal status (postmenopausal or premenopausal). Regarding hormonal receptors (Table 4), we have found only one cohort study80 and five case-control studies19,32,33,42,45. In both cases (ER+ and ER− tumors) statistical significance was not reached. With respect to menopausal status (Table 4), we obtained a protective effect in both groups: nineteen case-control studies targeted postmenopausal women18,21,28,30,34,35,36,38,41,47,49,51,55,60,81 with a pooled OR of 0.74 (95%CI: 0.59–0.93), and nine focused on premenopausal21,30,34,35,38,49,51,55,60 obtaining a pooled OR of 0.63 (95%CI: 0.49–0.80) (Fig. 3a). When the sensitivity analysis was carried out including only nested case-control studies, the protective vitamin D – breast cancer association persisted only in the premenopausal group (Fig. 3b, Supplementary Table 1). On the other hand three cohorts studies analyzed separately postmenopausal women79,81 without reaching statistical significance (OR = 1.15 (0.59–2.23)).
Relationship between 1,25(OH)2D and breast cancer
Three case-control studies25,37,39 examined the relationship between circulating 1,25(OH)2D and breast cancer; significant association was not found either in the whole analysis (pooled OR = 0.61 (0.33–1.16)) or in postmenopausal women (combined OR = 1.28 IC 95%: 0.98–1.67)36,37.
Relationship between dietary vitamin D and breast cancer
We found eight case-control studies24,38,40,50,52,53,57,64 on the relationship between dietary vitamin D and breast cancer with a pooled OR of 0.91 (95%CI: 0.72–1.17) (Table 4, Supplementary Fig. 2a). In addition, by combining five cohort studies66,68,70,71,72 we obtained a RR of 1.00 (95% CI 0.93–1.07) (Table 4, Supplementary Fig. 2b).
When stratifying by menopausal status, four case-control38,40,53,64 and five cohort studies66,73,74,76,77 assessed the risk of breast cancer in postmenopausal women. The pooled OR for case-control studies was 0.78 (95%CI: 0.68–0.90) and the pooled RR for cohort studies was 0.95 (95%CI: 0.83–1.09) (Table 4). In both analyses, Egger test rejected the possibility of small study bias (p = 0.536 in case-control studies and p = 0.68 in cohort studies). On the other hand, five case-control studies17,38,40,53,63 and three cohort studies66,73,77 targeted premenopausal women; the pooled OR was 0.65 (95%CI: 0.52–0.82) for case-control studies and the RR for cohort studies was 1.01 (95% CI: 0.86–1.18) (Table 4).
Relationship between supplements of vitamin D and breast cancer
We identified five case-control studies23,24,43,52,65 and two cohort studies67,71 that had evaluated the association between supplements of vitamin D and breast cancer risk. The pooled OR and RR were 0.78 (95% CI: 0.63–0.98) and 1.06(95% IC: 0.90–1.25) respectively (Table 4). Regarding menopausal status, Kim et al.41 published a study on five different populations of postmenopausal women; when combining all five results, we found no significant association (OR: 0.82 95%CI: 0.49–1.35).In addition, we found two case-control studies26,54 focused on premenopausal women obtaining a weak protection (pooled OR 0.89 95%CI (0.84–0.95)).
Relationship between total vitamin D intake (dietary and supplements) and breast cancer
Finally, we found two cohort studies69,71 and four case control studies23,24,38,64 on vitamin D intake (dietary plus supplements) and breast cancer risk, providing no separate results on dietary/supplemented vitamin D origin. We obtained a combined RR = 0.93 (95% CI: 0.86–1.02) for cohort studies, and a combined OR = 0.84 (95% CI: 0.68–1.05) for case-control studies. Five cohort studies69,73,74,76,77 provided results on postmenopausal women (RR = 0.94 95% CI: 0.87–1.00) and three cohort studies69,73,77 on about premenopausal women (RR = 0.90 95% CI: 0.72–1.12) (Table 4). Only two case-control studies provided results according menopausal status38,64 without being significant in both groups.
Discussion
According to our results, 25(OH)D levels were associated with smaller risk of breast cancer in both case-control and cohort studies; these results were consistent on premenopausal women for case-control studies but could not be analyzed for cohort studies. Results for the relationships between breast cancer and dietary vitamin D or between breast cancer and vitamin D supplements, however, showed a protective association only in case-control studies.
In relation to the influence of vitamin D on breast cancer development prospective (cohort and nested case-control) and case control studies tend to show discrepant results: case-control studies usually show a protective effect while prospective studies rarely find it87. This discrepancy might be the result of several factors: Firstly, it is well known that prospective studies are less prone to be affected by both information and reverse-causation bias. Secondly, several authors highlight the season when the vitamin D measurement was made as a potential limitation of case-control studies. Eliassen et al.33 in a nested case-control study found an inverse association between serum 25(OH) D levels and breast cancer limited only to summer measures. It can be assumed that people with low vitamin D levels in summer would also have low levels year-round; therefore, vitamin D levels in summer would be more adequate for analyzing vitamin D – breast cancer relationship than vitamin D levels in any other moment of the year.
When stratifying by menopausal status, our meta-analysis shows a consistent protective effect of 25(OH) D in both case-control and nested case-control studies, but only in premenopausal women. There are different explanations for the influence of menopausal status in the relationship between vitamin D and breast cancer. One of them may be related to the joint relationship between vitamin D and insulin-like growth factors (IGFs). IGF-I is a mitogenic and antiapoptotic peptide that can stimulate the proliferation of breast epithelial cells, increasing the risk of neoplastic transformation88,89. The active vitamin D metabolite is able to block the mitogenic effects of IGF-I, leading to a decrease in proliferation and an increase in apoptosis90. As there is a physiological decline of the IGF with aging91, the interaction between IGF pathways and vitamin D is likely to be stronger for premenopausal than for postmenopausal women, leading to greater risk reduction in premenopausal breast cancer73,92. Finally, high levels of vitamin D may reduce progesterone and estradiol, providing a potential mechanism for reducing breast cancer risk in young women93.
Previous meta-analyses of prospective studies showed contradictory results. Kim et al.13 (who included 24 studies, 14 of those having measured serum 25(OH)D) found a slightly stronger inverse association among premenopausal than among postmenopausal women but without significant differences, whereas in the meta-analysis of Bauer et al.8 (nine studies included) the inverse association was only observed in postmenopausal women. In our meta-analysis, new prospective studies31,33,41,56,58,59,67,78,79,80,81,94 not included in previous reviews, were added and this fact may explain the differences in the results.
Concerning hormonal receptors (ER+/ER−), the relationship with breast cancer remains controversial. On the one hand, a decreased risk in ER+ would be expected, since it seems that sensitivity to 1,25(OH)2D is generally reported as being higher in breast cancer cells that express the estrogen receptor than in those that do not93,95. It has been demonstrated that treating breast cancer cells ER+ with 1,25(OH)D3 induces a cell cycle shutdown in GO/G13,96. On the other hand, two-thirds of triple negative tumors express VDR97 and it has been demonstrated that VDR expression is inversely associated with more aggressive breast cancer98. In consonance with previous epidemiological studies32,33,42,45, our study does not reach significant differences when the analysis was performed separately in ER+ or ER− subgroups. However, other studies found a decreased risk of ER− breast cancer regarding the serum levels of 25 (OH) D18,60.
No relationship is found between the level of circulating 1,25(OH)2D and breast cancer. This result is consistent with previous studies9, while Janowsky et al.39 found an inverse association. Several authors consider that 1,25(OH)2D is not a good indicator of vitamin D status: First, 1,25(OH)2D’s half-life is only 4–6 h, whereas 25(OH)D’s half-life is 3 weeks; second, 1,25(OH)2D is influenced by many factors10, for instance, it can be elevated in patients with vitamin D deficiency as a result of hyperparathyroidism12,99; finally, as 1,25(OH)2D is metabolized by 1-α -hydroxylase in breast tissue, plasma levels may not adequately represent breast tissue levels12,100.
We do not find a relationship between vitamin D intake and breast cancer in the overall analysis. In contrast, when stratifying by menopausal status, a protective effect is observed in case-control studies in both premenopausal and postmenopausal women, whereas this association is not present in cohort studies. On the other hand, when analyzing the influence of vitamin D supplements on breast cancer risk, we find a borderline protective effect.
In the relationship between vitamin D intake (dietary and/or supplements) and breast cancer, most observational studies showed non-significant differences; only two articles17,53 found a protective association. In a previous meta-analysis13, this association was not significant for either vitamin D intake or supplements.
A probable explanation for the lack of association observed in the analysis of dietary intake or supplements compared to the 25(OH)D levels may be that the main source of vitamin D is sunlight rather than food or supplements.
In addition, the French E3N Cohort Study12 reported that high vitamin D intake is associated with lower breast cancer risk in regions with high ultraviolet solar radiance. These results suggested that the total amount of vitamin D needed to reach a protective effect on breast cancer is too high to be achieved in regions with low ultraviolet radiance. Under these circumstances, as the vitamin D intake has to be higher than the usually recommended, it could eventually lead to side effects such as hypercalcemia, constipation or muscle weakness.
Our study has some limitations; firstly each article uses different cutoff points according to serum levels of vitamin D. To analyze it we restricted our analysis to the comparison between the highest vs. lowest category of exposure. This analysis strategy does not allow for a dose-response analysis. Moreover, we carried out a sensitivity analysis excluding one study at a time, showing that no single study substantially affected the pooled RR/OR. Secondly, there is huge variability in the literature on the type of vitamin D studied, which makes it difficult to perform the analysis. In addition, levels of vitamin D depend on the season, so it would be advisable to take all samples at the same time, or at least refer to when they were collected75. Thirdly, case-control studies are more prone to methodological issues, such as recall and selection biases, which limits the strength and quality of evidence. However, about half of the case-control studies included in our meta-analysis are nested in cohort studies, which minimizes the possibility of introducing biases. Finally, breast cancer is a heterogeneous disease and it is possible that vitamin D only affects certain breast cancer subtypes. However, this aspect has been scarcely studied in primary articles, so we have not been able to analyze it in the present meta-analysis.
Despite these limitations, our study also has several strengths; first, we have gathered all the observational studies published in the last twenty years. In addition, we have focused the analysis on different types of vitamin D exposure (diet, supplements and blood-levels of 25(OH) D and 1,25(OH)2D) whereas other meta-analyses are only focused on 25(OH)D levels9,10,16,99 or vitamin D intake12. This strategy allows us to obtain a more detailed analysis of the relationship between vitamin D and breast cancer.
In conclusion, our meta-analysis supports the hypothesis that high serum levels of 25(OH) vitamin D has a protective effect on breast cancer risk in premenopausal women; we cannot draw the same conclusion regarding vitamin D intake or supplements of vitamin D since the number of studies are still limited and publication biases cannot be excluded.
References
Siegel, R. L., Miller, K. D. & Jemal, A. Cancer statistics. CA Cancer J Clin. 66, 7–30 (2016).
Habib, O. S. et al. Epidemiology of Breast Cancer among Females in Basrah. Asian Pac J Cancer Prev. 17, 91–5 (2016).
Colston, K. W. & Hansen, M. Mechanisms implicated in the growth regulatory effects of vitamin D in breast cancer. Endocr Relat Cancer. 9, 45–59 (2001).
Simboli-Campbell, M., Narvaez, C. J., Tenniswood, M. & Welsh, J. E. 1α,25(OH)2D3 induces morphological and biochemical indices of apoptosis in MCF-7 breast cancer cells. Journal of Steroid Biochemistry and Molecular Biology 58, 367–376 (1996).
Welsh, J. E. Induction of apoptosis in breast cancer cells in response to vitamin D and antiestrogens. Biochemistry and Cell Biology 72, 537–545 (1994).
James, S. Y., Mackay, A. G. & Colston, K. W. Effects on 1,25 dihydroxyvitamin D3 and its analogues on induction of apoptosis in breast cancer cells. Journal of Steroid Biochemistry and Molecular Biology 58, 395–401 (1996).
Khan, Q. J., Kimler, B. F. & Fabia, C. J. The Relationship Between Vitamin D and Breast Cancer Incidence and Natural History. Curr Oncol Rep. 12, 136–142 (2010).
Bauer, S. R., Hankinson, S. E., Bertone-Johnson, E. R. & Ding, E. L. Plasma Vitamin D Levels, Menopause, and Risk of Breast Cancer: Dose-Response Meta-Analysis of Prospective Studies. Medicine. 92, 123–131 (2013).
Chen, P. et al. Meta-analysis of vitamin D, calcium and the prevention of breast cancer. Breast Cancer Res Treat 121, 469–77 (2010).
Chen, P. et al. Higher Blood 25(OH)D Level May Reduce the Breast Cancer Risk: Evidence from a Chinese Population Based Case-Control Study and Meta-analysis of the Observational Studies. PLoS One. 8, e49312 (2013).
Gandini, S. et al. Meta-analysis of observational studies of serum 25-hydroxyvitamin D levels and colorectal, breast and prostate cancer and colorectal adenoma. Int J Cancer. 128, 1414–24 (2011).
Gissel, T., Rejnmark, L., Mosekilde, L. & Vestergaard, P. Intake of vitamin D and risk of breast cancer–a meta-analysis. J Steroid Biochem Mol Biol 111, 195–9 (2008).
Kim, Y. & Je, Y. Vitamin D intake, blood 25(OH)D levels, and breast cancer risk or mortality: a meta-analysis. Br J Cancer 110, 2772–84 (2014).
Mohr, S. B. et al. Serum 25-Hydroxyvitamin D and Prevention of Breast Cancer: Pooled Analysis. Anticancer Res 31, 2939–48 (2011).
Wang, D., Velez de-la-Paz, O. I., Zhai, J. X. & Liu, D. W. Serum 25-hydroxyvitamin D and breast cancer risk: a meta-analysis of prospective studies. Tumor Biology 34, 3509 (2013).
Yin, L. et al. Meta-analysis: Serum vitamin D and breast cancer risk. Eur J Cancer 4, 2196–2205 (2010).
Abbas, S., Linseisen, J. & Chang-Claude, J. Dietary Vitamin D and Calcium Intake and Premenopausal Breast Cancer Risk in a German Case-Control Study. Nutr Cancer. 59, 54–61 (2007).
Abbas, S. et al. Serum 25-hydroxyvitamin D and risk of post-menopausal breast cancer—results of a large case–control study. Carcinogenesis. 29, 93–99 (2008).
Abbas, S., Linseisen, J. & Chang-Claude, J. Plasma 25-hydroxyvitamin D and premenopausal breast cancer risk in a German case-control study. Int J Cancer. 124, 250–5 (2009).
Alipour, S. et al. Levels of Serum 25-Hydroxy-Vitamin D in Benign and Malignant Breast Masse. Asian Pac J Cancer Prev. 15, 129–32 (2014).
Almquist, M., Bondeson, A. G., Bondeson, L., Malm, J. & Manjer, J. Serum levels of vitamin D, PTH and calcium and breast cancer risk—a prospective nested case–control study. Int J Cancer. 127, 2159–2168 (2010).
Amir, E. et al. 25-Hydroxy vitamin-D, obesity, and associated variables as predictors of breast cancer risk and tamoxifen benefit in NSABP-P1. Breast Cancer Res Treat. 133, 1077–1088 (2012).
Anderson, L. N., Cotterchio, M., Cole, D. E. & Knight, J. A. Vitamin D-Related Genetic Variants, Interactions with Vitamin D Exposure, and Breast Cancer Risk among Caucasian Women in Ontario. Cancer Epidemiol Biomarkers Prev. 20, 1708–17 (2011).
Anderson, L. N., Cotterchio, M., Vieth, R. & Knight, J. A. Vitamin D and calcium intakes and breast cancer risk in pre- and postmenopausal women. Am J Clin Nutr 91, 1699–707 (2010).
Bertone-Johnson, E. R. et al. Plasma 25-Hydroxyvitamin D and 1,25-Dihydroxyvitamin D and Risk of Breast Cancer. Cancer Epidemiol Biomarkers Prev 14, 1991–7 (2005).
Bidgoli, S. A. & Azarshab, H. Role of Vitamin D Deficiency and Lack of Sun Exposure in the Incidence of Premenopausal Breast Cancer: a Case Control Study in Sabzevar, Iran. Asian Pac J Cancer Prev 15, 3391–6 (2014).
Bilinski, K. & Boyages, J. Association between 25-hydroxyvitamin D concentration and breast cancer risk in an Australian population: an observational case–control study. Breast Cancer Res Treat. 137, 599–607 (2013).
Chlebowski, R. T. et al. Calcium Plus Vitamin D Supplementation and the Risk of Breast Cancer. J Natl Cancer Inst. 100, 1581–91 (2008).
Colagar, A. H., Firouzjah, H. J. & Halalkho, S. Vitamin D Receptor Poly(A) Microsatellite Polymorphism and 25-Hydroxyvitamin D Serum Levels: Association with Susceptibility to Breast Cancer. J Breast Cancer. 18, 119–12 (2015).
Crew, K. D. et al. Association between Plasma 25-Hydroxyvitamin D and Breast Cancer Risk. Cancer Prev Res. 2, 589–604 (2009).
Deschasaux, M. et al. Weight Status and Alcohol Intake Modify the Association between Vitamin D and Breast Cancer Risk. J Nutr. 143, 576–85 (2016).
Eliassen, A. H. et al. Plasma 25-hydroxyvitamin D and risk of breast cancer in the Nurses’ Health Study II. Breast Cancer Res. 13, R50 (2011).
Eliassen, A. H. et al. Plasma 25-hydroxyvitamin D and risk of breast cancer in women followed over 20 years. Cancer Res. 76, 5423–30 (2016).
Engel, P. et al. Serum 25(OH) Vitamin D and Risk of Breast Cancer: A Nested Case-Control Study from the French E3N Cohort. Cancer Epidemiol Biomarkers Prev. 19, 2341–50 (2010).
Fedirko, V. et al. Serum 25-hydroxyvitamin D and risk of breast cancer: results of a large population-based case–control study in Mexican wome. Cancer Causes Control. 23, 1149–62 (2012).
Freedman, M. et al. Serum Levels of Vitamin D Metabolites and Breast Cancer Risk in the Prostate, Lung, Colorectal, and Ovarian Cancer Screening Trial. Cancer Epidemiol Biomarkers Prev. 17, 889–94 (2008).
Hiatt, R. A. et al. Prediagnostic Serum Vitamin D and Breast Cancer. J Natl Cancer Inst. 90, 461–3 (1998).
Jamshidinaeini, Y., Akbari, M. E., Abdollahi, M., Ajami, M. & Davoodi, S. H. Vitamin D Status and Risk of Breast Cancer in Iranian Women: A Case–Control Stud. J Am Coll Nutr. 35, 639–646 (2016).
Janowsky, E. C. et al. Association between low levels of 1,25-dihydroxyvitamin D and breast cancer risk. Public Health Nutr. 2, 283–29 (1999).
Kawase, T. et al. Association between vitamin D and calcium intake and breast cancer risk according to menopausal status and receptor status in Japan. Cancer Sci. 101, 1234–40 (2010).
Kim, Y. et al. Plasma 25-hydroxyvitamin D3 is associated with decreased risk of postmenopausal breast cancer in whites: a nested case-control study in the multiethnic cohort study. BMC Cancer. 17(14), 29 (2014).
Kühn, T. et al. Plasma 25-hydroxyvitamin D and the risk of breast cancer in the European Prospective Investigation into Cancer and Nutrition: A nested case–control study. Int J Cancer. 133, 1689–700 (2013).
Levi, F., Pasche, C., Lucchini, F. & La Vecchia, C. Dietary intake of selected micronutrients and breast-cancer risk. Int J Cancer. 91, 260–3 (2001).
Lowe, L. C. et al. Plasma 25-hydroxy vitamin D concentrations, vitamin D receptor genotype and breast cancer risk in a UK Caucasian population. Eur J Cancer. 41, 1164–9 (2005).
McCullough, M. L. et al. Serum 25-hydroxyvitamin D concentrations and postmenopausal breast cancer risk: a nested case control study in the Cancer Prevention Study-II Nutrition Cohort. Breast Cancer Res. 11, R64 (2009).
Mohr, S. B. et al. Serum 25-hydroxyvitamin D and breast cancer in the military: a case–control study utilizing pre-diagnostic serum. Cancer Causes Control. 24, 495–504 (2013).
Neuhouser, M. L. et al. The Influence of Health and Lifestyle Characteristics on the Relation of Serum 25-Hydroxyvitamin D With Risk of Colorectal and Breast Cancer in Postmenopausal Women. Am J Epidemio. 175, 673–84 (2012).
Oliveira-Sediyama, C. M. et al. Lifestyle and vitamin D dosage in women with breast cancer. Nutr Hosp. 33, 1179 (2016).
Park, S. et al. Serum 25-hydroxyvitamin D deficiency and increased risk of breast cancer among Korean women: a case–control study. Breast Cancer Res Treat. 152, 147–54 (2015).
Potischman, N. et al. Intake of food groups and associated micronutrients in relation to risk of early-stage breast cancer. Int J Cancer. 82, 315–21 (1999).
Rejnmark, L. et al. Reduced prediagnostic 25-hydroxyvitamin D levels in women with breast cancer: a nested case-control study. Cancer Epidemiol Biomarkers Prev. 18, 2655–60 (2009).
Rollison, D. E. et al. Vitamin D intake, vitamin D receptor polymorphisms, and breast cancer risk among women living in the southwestern U.S. Breast Cancer Res Treat. 132, 683–91 (2012).
Rossi, M. et al. Vitamin D intake and breast cancer risk: a case-control study in Italy. Ann Oncol. 20, 374–8 (2009).
Salarabadi, A., Bidgoli, S. A. & Madani, S. H. Roles of Kermanshahi Oil, Animal Fat, Dietary and Non- Dietary Vitamin D and other Nutrients in Increased Risk of Premenopausal Breast Cancer: A Case Control Study in Kermanshah, Iran. Asian Pac J Cancer Prev. 16, 7473–8 (2015).
Scarmo, S. et al. Circulating levels of 25-hydroxyvitamin D and risk of breast cancer: a nested case-control study. Breast Cancer Res. 133, 1689–700 (2013).
Shirazi, L., Almquist, M., Borgquist, S. & Manjer, J. Serum vitamin D (25OHD3) levels and the risk of different subtypes of breast cancer: A nested case-control study. Breast. 28, 184–190 (2016).
Simard, A., Vobecky, J. & Vobecky, J. S. Vitamin D deficiency and cancer of the breast: an unprovocative ecological hypothesis. Can J Public Health. 82, 300–3 (1991).
Sofi, N. Y. et al. Nutritional risk factors and status of serum 25(OH)D levels in patients with breast cancer: A case control study in India. J Steroid Biochem Mol Biol. 175, 55–59 (2016).
Wang, J., Eliassen, A. H., Spiegelman, D., Willett, W. C. & Hankinson, S. E. Plasma free 25-hydroxyvitamin D, vitamin D binding protein, and risk of breast cancer in the Nurses’ Health Study II. Cancer Causes Control. 25, 819–27 (2014).
Yao, S. et al. Pretreatment Serum Concentrations of 25-Hydroxyvitamin D and Breast Cancer Prognostic Characteristics: A Case-Control and a Case-Series Study. PLoS One 6, e17251 (2011).
Yousef, F. M. et al. Vitamin D status and breast cancer in Saudi Arabian women: case-control study. Am J Clin Nutr. 98, 105–10 (2013).
Wu, Y., Sarkissyan, M., Clayton, S., Chlebowski, R. & Vadgama, J. V. Association of Vitamin D3 Level with Breast Cancer Risk and Prognosis in African-American and HispanicWomen. Cancers. 9, 144 (2017).
Sofi, N. Y. et al. Reproductive factors, nutritional status and serum 25(OH)D levels in women with breast cancer: A case control study. Journal of Steroid Biochemistry and Molecular Biology 175, 200–204 (2018).
Lee, M. S. et al. Vitamin D Decreases Risk of Breast Cancer in Premenopausal Women of Normal Weight in Subtropical Taiwan. J Epidemiol. 21, 87–94 (2011).
Leung, H. W. C., Muo, C. H., Liu, C. F. & Chan, A. L. F. Vitamin D3 Intake Dose and Common Cancer: A Population-Based Case Control Study in a Chinese Population. Journal of Cancer 7, 2028–2034 (2016).
Abbas, S. et al. Dietary Intake of Vitamin D and Calcium and Breast Cancer Risk in the European Prospective Investigation into Cancer and Nutrition. Nutr Cancer. 65, 178–87 (2013).
Cadeau, C. et al. Interaction between current vitamin D supplementation and menopausal hormone therapy use on breast cancer risk: evidence from the E3N cohort. Am J Clin Nutr. 102, 966–73 (2015).
Edvardsen, K. et al. Vitamin D-effective solar UV radiation, dietary vitamin D and breast cancer risk. Int J Cancer. 128, 1425–1433 (2011).
Engel, P., Fagherazzi, G., Mesrine, S., Boutron-Ruault, M. C. & Clavel-Chapelon, F. Joint Effects of Dietary Vitamin D and Sun Exposure on Breast Cancer Risk: Results from the French E3N Cohort. Cancer Epidemiol Biomarkers Prev. 20, 187–98 (2010).
Frazier, A. L., Li, L., Cho, E., Willett, W. C. & Colditz, G. A. Adolescent diet and risk of breast cancer. Cancer Causes Control. 15, 73–82 (2004).
John, E. M., Schwartz, G. G., Dreon, D. M. & Koo, J. Vitamin D and Breast Cancer Risk: The NHANES I Epidemiologic Follow-up Study, 1971–1975 to 1992. Cancer Epidemiol Biomarkers Prev 8, 399–406 (1999).
Kuper, H. et al. Prospective study of solar exposure, dietary vitamin D intake, and risk of breast cancer among middle-aged women. Cancer Epidemiol Biomarkers Prev. 18, 2558–6 (2009).
Lin, J. et al. Intakes of calcium and vitamin D and breast cancer risk in women. Arch Intern Med. 167, 1050–9 (2007).
McCullough, M. L. et al. Dairy, Calcium, and Vitamin D Intake and Postmenopausal Breast Cancer Risk in the Cancer Prevention Study II Nutrition Cohort. Cancer Epidemiol Biomarkers Prev 14, 2898–904 (2005).
Ordóñez-Mena, J. M. et al. Serum 25-hydroxyvitamin d and cancer risk in older adults: results from a large German prospective cohort study. Cancer Epidemiol Biomarkers Prev. 22, 905–16 (2013).
Robien, K., Cutler, G. J. & Lazovich, D. Vitamin D intake and breast cancer risk in postmenopausal women: the Iowa Women’s Health Study. Cancer causes control. 18, 775–782 (2007).
Shin, M. H. et al. Intake of Dairy Products, Calcium, and Vitamin D and Risk of Breast Cancer. J Natl Cancer Inst 94, 1301–11 (2002).
Skaaby, T. et al. Prospective population-based study of the association between serum 25-hydroxyvitamin-D levels and the incidence of specific types of cáncer. Cancer Epidemiol Biomarkers Prev 23, 1220–9 (2014).
O’Brien, K. M., Sandler, D. P., Taylor, J. A. & Weinberg, C. R. Serum Vitamin D and Risk of Breast Cancer within Five Years. Environ Health Perspect 25, 077004, https://doi.org/10.1289/EHP943 (2017).
Palmer, J. R. et al. Predicted 25-hydroxyvitamin D in relation to incidence of breast cancer in a large cohort of African American women. Breast Cancer Research 18, 86 (2016).
Ordoñez-Mena, J. M. et al. Pre-diagnostic vitamin D concentrations and cancer risks in older individuals: an analysis of cohorts participating in the CHANCES consortium. Eur J Epidemiol. 31, 311–23 (2016).
Elma, E. V. et al. Declaración de la Iniciativa STROBE (Strengthening the Reporting of Observational studies inEpidemiology): directrices para la comunicación de estudios observacionales. Revista Española de Salud Pública 82, 144–150 (2008).
Higgins, J. P. & Thompson, S. G. Quantifying heterogeneity in a meta-analysis. Stat Med. 21, 1539–58 (2002).
DerSimonian, R. & Laird, N. Meta-analysis in clinical trials. Control Clin Trials 7, 177–188 (1986).
Egger, M., Davey, S. G., Schneider, M. & Minder, C. Bias in meta-analysis detected by a simple, graphical test. BMJ. 315, 629–34 (1997).
Light, R. J. & Pillemer, D. B. Summing up: the science of reviewing research. Cambridge: Harvard University Press. (1984).
Moukayed, M. & Grant, W. B. The roles of UVB and vitamin D in reducing risk of cancer incidence and mortality: A review of the epidemiology, clinical trials, and mechanisms. Rev Endocr Metab Disord. 18, 167–182 (2017).
Hankinson, S. E. et al. Plasma Sex Steroid Hormone Levels and Risk of Breast Cancer in Postmenopausal women. J Natl Cancer Inst. 90, 1292–1299 (1998).
Christopoulos, P. F., Msaouel, P. & Koutsilieris, M. The role of the insulin-like growth factor-1 system in breast cancer. 15, 43 (2015).
Ameri, P. et al. Interactions between vitamin D and IGF-I: from physiology to clinical practice. Clin Endocrinol (Oxf). 79, 457–63 (2013).
Gomez, M. The role of insulin-like growth factor I components in the regulation of vitamin D. Curr Pharm Biotechnol. 7, 125–32 (2006).
Chlebowski, R. T. Vitamin D and breast cancer: interpreting current evidence. Breast Cancer Res. 3, 217 (2011).
Knight, J. A., Wong, J., Blackmore, M., Raboud, J. M. & Vieth, R. Vitamin D association with estradiol and progesterone in young women. Cancer Causes Control. 21, 479 (2010).
Cadeau, C. et al. Postmenopausal breast cancer risk and interactions between body mass index, menopausal hormone therapy use, and vitamin D supplementation: Evidence from the E3N cohort. Int. J. Cancer. 139, 2193–2200 (2016).
Narvaez, C. J., Zinser, G. & Welsh, J. Functions of 1,25-dihydroxyvitamin D3 in mammary gland: from normal development to breast cancer. Steroids. 66, 301–8 (2001).
Shao, T., Klein, P. & Grossbard, M. L. Vitamin D and Breast Cancer. Oncologist. 17, 36–45 (2012).
Thakkar, A. et al. Vitamin D and androgen receptor-targeted therapy for triple-negative breast cancer. Breast Cancer Res Treat 157(1), 77–90 (2016).
Al-Azhri et al. Tumor Expression of Vitamin D Receptor and Breast Cancer Histopathological Characteristics and Prognosis. Clin Cancer Res 23(1), 97–103 (2016).
Garland, C. F. et al. Vitamin D and prevention of breast cancer: pooled analysis. J Steroid Biochem Mol Biol 103, 708–711 (2007).
Bertone-Johnson, E. R. Vitamin D and breast cancer. Ann Epidemiol 19, 462–467 (2009).
Author information
Authors and Affiliations
Contributions
N.E., T.D.S. and I.G.A. contributed substantially to the conception, design and acquisition of data. N.E. and T.D.S.: wrote the main manuscript text. N.E. and C.P. prepared figures. T.D.S., I.G.A. and J.L. contributed to the analysis and interpretation of the data. N.E. and T.D.S., I.G.A., C.P. and J.L. contributed to devising the draft of the article and all of the other authors revised it critically. All authors participated in revising the manuscript and in the final approval of the version to be published.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Estébanez, N., Gómez-Acebo, I., Palazuelos, C. et al. Vitamin D exposure and Risk of Breast Cancer: a meta-analysis. Sci Rep 8, 9039 (2018). https://doi.org/10.1038/s41598-018-27297-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-27297-1
This article is cited by
-
Non-genetic factors and breast cancer: an umbrella review of meta-analyses
BMC Cancer (2024)
-
Dual effect of vitamin D3 on breast cancer-associated fibroblasts
BMC Cancer (2024)
-
Vitamin D and potential effects on cancers: a review
Molecular Biology Reports (2024)
-
Association between Gastric Cancer and Vitamin D in Kashmiri Population: A 2 Year Case–Control Study at a Tertiary Care Hospital
Indian Journal of Surgery (2024)
-
Oral health-related quality of life in patients aged 8 to 19 years with cleft lip and palate: a systematic review and meta-analysis
BMC Oral Health (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.