Abstract
Proper cholesterol transport is crucial for the functionality of cells. In C. elegans, certain cholesterol derivatives called dafachronic acids (DAs) govern the entry into diapause. In their absence, worms form a developmentally arrested dauer larva. Thus, cholesterol transport to appropriate places for DA biosynthesis warrants the reproductive growth. Recently, we discovered a novel class of glycosphingolipids, PEGCs, required for cholesterol mobilization/transport from internal storage pools. Here, we identify other components involved in this process. We found that strains lacking polyunsaturated fatty acids (PUFAs) undergo increased dauer arrest when grown without cholesterol. This correlates with the depletion of the PUFA-derived endocannabinoids 2-arachidonoyl glycerol and anandamide. Feeding of these endocannabinoids inhibits dauer formation caused by PUFAs deficiency or impaired cholesterol trafficking (e.g. in Niemann-Pick C1 or DAF-7/TGF-β mutants). Moreover, in parallel to PEGCs, endocannabinoids abolish the arrest induced by cholesterol depletion. These findings reveal an unsuspected function of endocannabinoids in cholesterol trafficking regulation.
Similar content being viewed by others
Introduction
Cholesterol is a lipid used by virtually all eukaryotic organisms. Its primary role as a structural component of cell membranes is to regulate their fluidity, permeability and topology. However, cholesterol also serves as a precursor of a plethora of other important biomolecules, such as steroid hormones, oxysterols and bile acids. Thus, tight regulation of the intake, biosynthesis, transport and metabolism of cholesterol is crucial for cellular integrity and functionality. In cells, cholesterol needs to be delivered to various destinations where it performs its structural functions or is further metabolized. This process is known as intracellular cholesterol trafficking and its importance is underscored by an amounting number of pathologies resulting from its impairment, including atherosclerosis and many genetic disorders such as the Niemann-Pick type C (NPC) disease and the Tangier disease1,2. Therefore, the cholesterol trafficking machinery has been intensively studied and many protein mediators of vesicular and non-vesicular cholesterol transport have been described2.
C. elegans is an excellent model for studying cholesterol trafficking and metabolism because worms are unable to synthesize cholesterol and require its presence in the diet. This allows an easy manipulation of worm sterol composition by introducing variations of its levels in the diet3. Following cholesterol intake, the systemic distribution of this lipid in worms is mediated by various transport systems, including the vitellogenin lipoprotein particles4. After receptor-mediated endocytosis of cholesterol carriers, cholesterol is trafficked through the endolysosomal system and directed to other cellular compartments with the assistance of the Niemann-Pick type C1 (NPC1) homologs NCR-1 and NCR-25. The importance of this process is demonstrated by the fact that ncr-2;ncr-1 null mutants fail to produce fertile adults and instead arrest at the dauer diapause, an alternative developmental stage for survival under harsh conditions such as overcrowding and starvation5. This developmental arrest has been shown to occur as a result of a decreased production of bile acid like steroid hormones called dafachronic acids (DAs)5,6. These important hormones integrate cues from various signaling pathways, including the transforming growth factor (TGF)-β-like (defined by the TGF-β homologue DAF-7) pathway, the insulin-like pathway (involving nematode insulin receptor DAF-2) and the cyclic GMP pathway, by binding a nuclear hormone receptor (NHR) named DAF-126,7,8,9,10. In its DA-bound form, DAF-12 stimulates reproductive development, whereas in the absence of DAs it promotes dauer arrest.
Even though cholesterol is associated with cell membranes and interacts with multiple lipid species, very little is known about how lipids influence cholesterol trafficking. One of the few known examples is the positive effect of the phospholipid lysobisphosphatidic acid on the trafficking of cholesterol through the endolysosomal compartment11. Owing to the huge diversity of membrane lipids, multiple other lipid species might emerge as additional modulators of the cholesterol trafficking process. More recently, we have discovered a novel class of glycolipids, phosphoethanolamine glucosylceramides (PEGCs), that stimulates the trafficking of cholesterol in C. elegans10. PEGCs stimulate the growth of worms under conditions of cholesterol scarcity and abolish the developmental arrest of ncr-2;ncr-1 mutants. By enhancing the mobilization of cholesterol from intracellular pools, PEGCs stimulate the production of DAs, thus inhibiting the dauer promoting activity of DAF-12.
Another class of lipids, endocannabinoids, have been implicated in the regulation of dauer formation as well12. These molecules are conserved lipid mediators that regulate multiple biological processes in a variety of organisms13,14. Previous studies have shown that one class of endocannabinoids, N-acylethanolamines (NAEs), antagonize dauer formation in C. elegans. NAEs are endocannabinoids composed of a fatty acyl chain of varying length and unsaturation (polyunsaturated fatty acids, PUFAs) linked to an ethanolamine head group. It has been shown that supplementation with the NAE eicosapentanoyl ethanolamide (EPEA), was able to rescue the dauer phenotypes of daf-2 mutant lines12.
In this study, we show that the synthesis of PUFAs is important for cholesterol trafficking and, accordingly, for the reproductive development of worms. Further characterization of the bioactive PUFA-derivatives revealed that arachidonoyl-ethanolamine (AEA) and 2-O-arachidonoyl-glycerol (2-AG), the best-characterized arachidonic acid containing endocannabinoids15, are responsible for abolishing dauer larva arrest by stimulating cholesterol trafficking. We also show that endocannabinoids via mobilization of internal cholesterol pools can rescue the dauer arrest induced by PEGCs deficiency/cholesterol depletion. We propose that endocannabinoids and PEGCs act synergistically to promote growth and development by regulating cholesterol homeostasis in C. elegans.
Results
Depletion of PUFAs leads to enhanced dauer formation
In a previous study, we have shown that C. elegans interrupts reproductive development and arrests as a dauer-like larva (L2*) with incomplete molting when grown for two generations without cholesterol3. More recently, we have found that this arrest can be abolished by supplementation of PEGCs, substances belonging to a novel class of lipids that mobilizes internal pools of cholesterol10. We were interested to further investigate this process and to identify other components that might be involved in it. We reasoned that strains aberrant in cholesterol mobilization would arrest already in the first generation without externally provided sterols. Indeed, a small screening performed on a mutant strain collection (Mende, F. and Kurzchalia, T.V., unpublished data) indicated that one of them, with inactive Δ6-desaturase [fat-3(ok1126)] showed growth defects already in the first, rather than in the second generation when grown without cholesterol. Unlike the wild type strain, that produced almost 100% gravid adults in the first generation without cholesterol, fat-3 displayed a high incidence of arrested larvae (Fig. 1a) with typical dauer morphology (Supplementary Figure 1). This mutant strain bears a large deletion in the coding region of gene fat-3, and is null for FAT-3 desaturase. Animals accumulate linoleic and α-linolenic acids and are unable to produce the major C20 PUFAs found in C. elegans: arachidonic (20:4 (n-6), AA) and eicosapentanoic acid (20:5(n-3), EPA)16,17.
PUFA-deficient mutants fat-3 and daf-7;fat-3 exhibit a dauer-like or a Daf-c phenotype, respectively. (a) Unlike wild-type worms, fat-3(ok1126) mutants are incapable of synthesizing PUFAs and undergo a dauer-like arrest in the first generation when grown in cholesterol-free media. One-way analysis of variance p < 0.001. All pairwise multiple comparison procedures (Holm-Sidak method), (*) indicates statistically significant difference with fat-3/solvent, p < 0.001. Bars represent mean values and error bars represent standard errors. The number of independent experiments is n = 3 for all conditions. (b) While daf-7 worms form ~10% dauers at 20 ºC, fat-3 worms do not form dauers. In stark contrast, double mutant daf-7;fat-3 forms ~85% dauers at 20 ºC, and addition of DA suppresses dauer formation almost completely. One-way analysis of variance p < 0.001. All pairwise multiple comparison procedures (Holm-Sidak method): (*) indicates statistically significant difference with daf-7, p < 0.001. Bars represent mean values and error bars represent standard errors. The number of independent experiments is n = 5 for daf-7, and n = 13 for fat-3 and daf-7; fat-3. [DA] = 90 nM. (c) Introducing the fat-3 null-mutation into background daf-7 increases dauer formation significantly (dauers are indicated with arrowheads). Representative images are taken from at least three experiments. Scale bars, 0.5 mm. (d) daf-7;fat-3 dauers can partly bypass dauer arrest if grown in excess cholesterol (130 µM). (*) indicates statistically significant difference to control plates (13 µM cholesterol). t-test, p < 0.001. Bars represent mean values and error bars represent standard errors. The number of independent experiments is n = 14 for daf-7;fat-3 at 13 µM cholesterol, and n = 2 for daf-7;fat-3 at 130 µM cholesterol.
Next, we set out to confirm the interaction between the PUFA biosynthesis pathways and the dauer formation regulating pathways. In particular, we tested the interaction with the DAF-7/TGF-β pathway that regulates dauer development by affecting sterol trafficking and metabolism10,18,19. In our previous study we have shown that temperature-sensitive daf-7 mutants with “dauer formation constitutive” (Daf-c) phenotype are hypersensitive to cholesterol depletion and form dauer larvae in the absence of external cholesterol already at the semi-permissive growth temperature despite internally stored sterols10. We reasoned that if the biosynthesis of PUFAs is connected with this regulatory pathway, depletion of PUFAs should enhance the phenotype of daf-7. To this end, we generated a double mutant strain for fat-3(ok1126) and daf-7(e1372) and scored dauer formation. When grown at the semi-permissive growth temperature (20° C), daf-7(e1372);fat-3(ok1126) double mutants formed significantly more dauers (~85%) than the parental daf-7 strain (<10%) (Fig. 1b,c). Moreover, enhanced dauer formation in double mutants was fully abolished when the medium was supplemented with DA (Fig. 1b). These results suggest that a decrease in the synthesis of C20 PUFAs could be leading to deficiency of DAs. We speculated that depletion of PUFAs might exacerbate cholesterol deficiency due to a decrease in its delivery rate from storage sites to the locations of DA production. Therefore, we tested whether very high cholesterol availability in the growth medium was sufficient to compensate for the loss of FAT-3 in daf-7;fat-3 animals. As shown in Fig. 1d, providing a high-cholesterol diet did promote dauer recovery of daf-7;fat-3 mutants.
Arachidonic acid-containing endocannabinoids abolish dauer formation of daf-7;fat-3 and daf-7;fat-4 mutants
Then, we asked which PUFA-derived molecules could be responsible for the dauer arrest of daf-7;fat-3 double mutants. PUFAs are metabolized into many potent bioactive compounds such as eicosanoids and endocannabinoids20. The latter, shown in Fig. 2a, might be one of the most likely class of candidates because previous studies have shown that N-acylethanolamines (NAEs) antagonize dauer formation in C. elegans12. Using high-performance liquid chromatography coupled to mass spectrometry (HPLC MS/MS), we found that, in agreement with a previous report21, N2 has detectable amounts of 2O-arachidonoylglycerol (2-AG, Fig. 2a) and arachidonoyl-ethalonamine (anandamide or AEA, Fig. 2a) (Fig. 2b and Supplementary Table 1). We also determined that these arachidonic acid-containing endocannabinoids are synthesized by daf-7 (Fig. 2b). In contrast, both fat-3 and daf-7;fat-3 strains do not synthesize PUFA-derived endocannabinoids to detectable levels (Fig. 2b). Next, we tested the capability of the detected endocannabinoid molecules to suppress dauer formation of daf-7;fat-3. We found that supplementation of the media with 2-AG and AEA significantly reduced the Daf-c phenotype of daf-7;fat-3 worms (Fig. 2c). In contrast, EPEA failed to rescue the Daf-c phenotype of daf-7;fat-3 (Fig. 2a,c).Treatment with AA, which is both a precursor as well as the hydrolytic breakdown product of both 2-AG and AEA15, was unable to promote reproductive growth of daf-7;fat-3 (Fig. 2c). However, in a previous study, we have established the use of fatty acid methyl esters (FAMEs) instead of the corresponding free fatty acids due to the much higher rescue activities of the former22. FAMEs are more active presumably because membranes are more permeable for them. Indeed, addition of AA-methyl ester (methyl-AA) to daf-7;fat-3 worms significantly reduced the Daf-c phenotype (Fig. 2c). This raised the question whether methyl-AA is a precursor of 2-AG and/or AEA23 or AA has an effect by itself. In order to find out whether methyl-AA serves as a precursor of 2-AG and AEA, lipid extracts of fat-3 and daf-7;fat-3 mutants fed with methyl-AA were analyzed by LC-MS/MS. This analysis revealed that indeed these mutants were capable to synthesize both 2-AG and AEA (Supplementary Table 1) upon methyl-AA supplementation. Importantly, we found that noladin24 (Fig. 2a), a non-hydrolysable analog of 2-AG, was also able to rescue the Daf-c phenotype of daf-7;fat-3 to an extent comparable to the effect of 2-AG, demonstrating that the endocannabinoids are not merely precursors for other AA-derived compounds (Fig. 2c).
Endocannabinoids rescue the Daf-c phenotype of daf-7;fat-3 worms. (a) Chemical structures of the endocannabinoid molecules assayed. (b) Extracted ion chromatograms of HPLC- MS/MS experiments in order to detect the endocannabinoids 2-AG and AEA in lipid extracts of the following worm strains: N2, daf-7, fat-3 and daf-7;fat-3. The upper panels show the chromatograms of the analytical standards 2-AG and AEA. (c) daf-7;fat-3 dauers cannot be rescued by supplementation with EPEA. However, addition of the endocannabinoids 2-AG and AEA, and the non-hydrolysable noladin ether suppresses dauer formation significantly. Supplementation of methyl-AA also prevents daf-7;fat-3 dauer formation. Kruskal-Wallis one-way analysis of variance on ranks, p < 0.001. Multiple comparisons versus control group (Dunn’s Method). (*) indicates statistically significant difference with solvent, p < 0.05. Bars represent mean values and error bars represent standard errors. The number of independent experiments is n = 14 for solvent, n = 2 for EPEA and noladin, n = 14 for 2-AG, n = 4 for AEA and AA, and n = 8 for methyl-AA. The concentration of all additives is 50 µM.
Although AA-derived endocannabinoids rescue the dauer phenotype of daf-7;fat-3, it could be argued that this double mutant is unable to synthesize dihommo-γ-linolenic acid [20:3(n-6)] (DGLA) and eicosatetraenoic acid [20:4(n-3)], [ETA (n-3)] and that endocannabinoids derived from these C20 PUFAs could serve the same function as 2-AG or AEA. To answer this question we generated daf-7;fat-4 double mutant strain that synthesizes DGLA and ETA (n-3), but is unable to synthesize AA25 due to its deficiency in Δ5 fatty acid desaturase enzyme FAT-4. daf-7;fat-4 double mutant displays a very strong Daf-c phenotype similar to the one observed in daf-7;fat-3 (Supplementary Figure 2a). Furthermore, supplementation of the growth medium with an excess of cholesterol, DA, 2-AG and noladin ether substantially lowers dauer formation in daf-7;fat-4 (Supplementary Figures 2a and 2b). These results demonstrate the specificity of AA-derived endocannabinoids to rescue the enhanced dauer formation of daf-7 animals deprived of C20 PUFAs. Taken together, we conclude that endocannabinods containing AA residues are the active compounds promoting reproductive development.
Endocannabinoids do not substitute for DA
As shown above, both endocannabinoids and DAs are able to rescue the dauer constitutive phenotype displayed by daf-7;fat-3 (Figs 1b and 2c). This raised the possibility that the endocannabinoid compounds were merely substituting for DAs as DAF-12 ligands. To address this question, we tested whether 2-AG and AEA were able to compensate for the deficiency of DAF-9 cytochrome P450 enzyme, necessary for the production of DA from cholesterol6,26. As seen in Supplementary Figure 3, unlike DA, 2-AG and AEA failed to inhibit dauer formation in daf-9(dh6) mutants. This indicates that these endocannabinoids cannot substitute for DA. Thus, it seems likely that these endocannabinoids inhibit dauer formation by enhancing the synthesis of DAs.
Endocannabinoids rescue the arrest induced by cholesterol depletion through its mobilization
Our previous studies have demonstrated that PEGCs also enhance the synthesis of DAs by mobilization of internal pools of maternally deployed cholesterol10. Because of this activity, PEGCs are also able to abolish the developmental arrest caused by cholesterol starvation in the second generation of worms grown on sterol-depleted medium10. We asked whether endocannabinoids could similarly inhibit the arrest of cholesterol-depleted worms. Indeed, addition of 2-AG to sterol-lacking medium resulted in formation of many late stage (L4) larvae and adults in the N2 wild-type strain in the second generation, whereas the untreated worms arrested mainly as L2* larvae as published before (Fig. 3a)3.
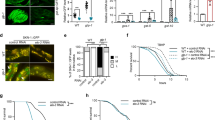
Endocannabinoids suppress dauer formation induced by cholesterol depletion. (a) Wild-type control animals grown for two generations in the absence of cholesterol arrest as L2-like larvae (upper micrograph), however, when fed with 2-AG worms form adults (lower micrograph). Representative images from at least three experiments. Scale bars, 0.25 mm. (b) When grown in the absence of cholesterol daf-7 worms form ~65% dauers in the first generation at 15 ºC. This arrest is largely suppressed by the addition of either cholesterol, or DA, or by the endocannabinoid 2-AG. (*) indicates statistically significant difference with solvent control. t-test, p = 0.002. Bars represent mean values and error bars represent standard errors. The number of independent experiments is n = 5 for solvent, and 2-AG, n = 7 for cholesterol, and n = 4 for ∆7-DA. [Cholesterol] = 13 µM. [DA] = 90 nM. [2-AG] = 50 µM. (c) 2-AG suppresses the Daf-c phenotype of ncr-2;ncr-1 at 25 ºC. (*) indicates statistically significant difference with solvent control. Kruskal-Wallis one-way analysis of variance on ranks, p < 0.001 and all pairwise multiple comparison procedures (Student-Newman-Keuls Method), p < 0.05. Bars represent mean values and error bars represent standard errors. The number of independent experiments is n = 6 for all conditions. [Cholesterol] = 13 µM. [DA] = 90 nM. [2-AG] = 50 µM. (d) faah-1 RNAi reduces the percent of dauer formation of strain ncr-2;ncr-1 at 25 ºC. (*) indicates statistically significant difference with solvent empty vector control (EV). One Way Analysis of Variance (p < 0.001) and comparison by Holm-Sidak method, p < 0.001 in both cases. Bars represent mean values and error bars represent standard errors. The number of independent experiments is n = 6 for all conditions tested. [DA] = 90 nM.
We next set out to investigate whether AEA and 2-AG have the ability to mobilize internal reserves of sterols in C. elegans. First, we made use of daf-7 mutants which are hypersensitive to cholesterol starvation. They form dauers already in the first generation even at permissive temperature (15˚C) when the external cholesterol source is missing. Although these worms receive high amounts of maternal cholesterol, they are unable to use this sterol for DA synthesis presumably due to an impairment in mobilizing sterols efficiently10. We found that upon cholesterol deprivation, 2-AG rescues the dauer arrest of daf-7 animals grown without cholesterol (Fig. 3b). Next, we investigated the interaction of endocannabinoids with the NPC1 sterol-trafficking system27. As mentioned earlier, cholesterol transport in C. elegans is partly mediated by the NPC1 homologs NCR-1 and NCR-228. Because of this, double mutant ncr-2(nr2023);ncr-1(nr2022) displays constitutive dauer formation5. This dauer formation can be rescued by supplementation with DAs5,7. As seen in Fig. 3c, 2-AG could also largely suppress ncr-2;ncr-1 dauer formation. Moreover, the Daf-c phenotype of ncr-2;ncr-1, as well as of daf-7, was diminished by RNAi against faah-1, a gene encoding a conserved fatty acid amide hydrolase (FAAH) enzyme, responsible for the breakdown of N-acylethanolamines such as AEA and EPEA in worms12 (Fig. 3d and Supplementary Figure 4).
The ability of the AA-derived endocannabinoids to rescue the development of cholesterol-depleted worms pointed out to the possibility that the endogenous production of these compounds might be enhanced in response to cholesterol withdrawal. This could be a mechanism to optimize cholesterol mobilization when the diet is not rich in sterols. In agreement with this hypothesis, we found that removing cholesterol from the growth medium resulted in a reproducible two-fold increase in 2-AG levels in N2 animals already in the first generation (Supplementary Table 2). Taken together, our data suggest that in C. elegans endocannabinoids exert their effects on post-embryonic development by enhancing the efficiency of cholesterol trafficking and the utilization of internal pools.
Endocannabinoids and PEGCs are acting through parallel pathways
Next, we examined how 2-AG and AEA are connected to recently discovered PEGCs: Can endocannabinoids rescue PEGCs deficiency? And vice versa, can PEGCs rescue endocannabinoid deficiency?
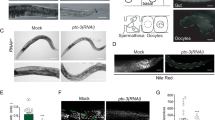
To induce PEGCs deficiency, we inhibited the synthesis of their precursors, the glucosylceramides (GlcCer). For this, two approaches have been used. Firstly, we decided to decrease C15iso and C17iso monomethyl branched-chain fatty acids (mmBCFA), needed for the synthesis of the long-chain base in C. elegans29 (Supplementary Figures 5 and 6). For this, we applied RNAi of LET-767, the major 3-ketoacyl-CoA reductase, necessary for the elongation of long-chain and branched-chain fatty acids. RNAi of LET-767 in wild type worms leads to multiple developmental phenotypes in the first generation and early larval arrest in the second generation (L1-L2)22. Indeed, RNAi against LET-767 in daf-7 mutants grown at 20 °C significantly increases the Daf-c phenotype of the strain (Fig. 4a). GC-MS analysis of the relative fatty acid profiles of control and let-767 RNAi animals confirmed that RNAi treatment leads to a strong decrease in the levels of C15iso and C17iso mmBCFAs, but not of PUFAs (Supplementary Table 3). Accordingly, as seen in Fig. 4b, supplementation with C17iso mmBCFA methyl esters rescued the let-767 RNAi-induced dauer arrest of daf-7 worms. Astoundingly, both 2-AG and AEA also rescued the dauer formation observed in daf-7 animals subjected to let-767 RNAi (Fig. 4b). To exclude the possibility that endocannabinoids act by elevating levels of mmBCFAs, i.e. by inducing (a) not yet identified alternative 3-ketoacyl-CoA reductase(s), we performed GC-MS analysis of mmBCFA in daf-7 let-767 RNAi worms in the presence of 2-AG (Supplementary Table 4). This experiment clearly shows that endocannabinoid treatment does not increase mmBCFA. In consistency with impaired cholesterol trafficking in daf-7 let-767 (RNAi), dauer formation was partly inhibited by high cholesterol levels in the medium (Fig. 4c).
PEGCs and endocannabinoids stimulate cholesterol transport by employing parallel pathways. (a) RNAi of let-767 leads to a significant increase of dauers compared to control plates in daf-7 mutant background (empty vector; from ~20% to ~80%), but has no effect on wild type (N2) dauer formation. Kruskal-Wallis one-way analysis of variance on ranks, p < 0.001, All pairwise multiple comparison procedures (Dunn’s Method), (*) indicates statistically significant difference between daf-7/empty vector and daf-7/let-767 RNAi, p < 0.05. Bars represent mean values and error bars represent standard errors. The number of independent experiments is n = 6 for N2/empty vector, and N2/let-767 RNAi, n = 27 for daf-7/empty vector, n = 33 for daf-7/let-767 RNAi, and n = 6 for daf-7/let-767 RNAi+DA. [DA] = 90 nM. (b) daf-7 on let-767 RNAi at 20 ºC forms ~80% dauers. Supplementation with either of the endocannabinoids 2-AG and AEA, or the addition of methyl-C17iso suppresses dauer formation significantly. Kruskal-Wallis one-way analysis of variance on ranks, p < 0.001. Multiple comparisons versus control group (Dunn’s Method). (*) indicates statistically significant difference with empty vector, p < 0.05. Bars represent mean values from at least three independent experiments and error bars represent standard errors. [2-AG] or [AEA] = 50 µM. [methyl-C17iso] = 100 µM. (c) daf-7 let-767 RNAi worms partly bypass dauer arrest if grown in excess cholesterol (130 µM). (*) indicates statistically significant difference with control plates (13 µM cholesterol). Mann-Whitney rank sum test p = 0.013. Bars represent mean values and error bars represent standard errors. The number of independent experiments is n=26 for daf-7 let-767 RNAi at 13 µM cholesterol, and n = 4 for daf-7 let-767 RNAi at 130 µM cholesterol. (d) Inhibition of the d17iso-glucosylceramide synthesis (and hence PEGCs) by PDMP treatment leads to an increase in dauer formation in daf-7 background, that can be largely suppressed by endocannabinoids, DA and excess cholesterol, but not by methyl-C17iso. One-way analysis of variance, p < 0.001. Multiple comparisons versus control group (Holm-Sidak method). (*) indicates statistically significant difference with PDMP treatment, p < 0.001 for all conditions except for empty vector (p = 0.002). Bars represent mean values and error bars represent standard errors. The number of independent experiments is n = 3 for all conditions. [DA] = 90 nM. [Cholesterol] = 130 µM. [2-AG] or [AEA] = 50 µM. [methyl-C17iso] = 100 µM. (e) mmPEGC-C22 is partly able to suppress dauer formation of daf-7;fat-3 at 20 ºC. One-way analysis of variance, p < 0.001. Multiple comparisons versus control group (Holm-Sidak method). (*) indicates statistically significant difference with solvent control, p < 0.001 for both conditions. Bars represent mean values and error bars represent standard errors. The number of independent experiments is n = 2 for all conditions. [DA] = 1 µM. [mmPEGC-C22] = 0.5 mM. (f) Endocannabinoids and PEGCs act in parallel to promote transport/mobilization of cholesterol from internal pools. Endocannabinoids are synthesized from their precursors DAG and NAPE most probably by DAGL and NAPE-PLD enzymatic activities. The enzyme(s) that produce(s) PEGC from d17iso-GlcCer is(are) unknown but the synthesis is dependent on NCR-1/2. Both endocannabinoids and PEGCs have the ability to mobilize cholesterol. The latter is then delivered to the sites of DAF-9 mediated DA synthesis. After systemic distribution of DA within the organism, it binds and inhibits the dauer-promoting activities of DAF-12. DAG – diacylglycerol; NAPE - N-acyl-phosphatidylethanolamine; DAGL - DAG lipase; NAPE-PLD - NAPE-specific phospholipase D.
The second approach to deplete PEGCs was to treat daf-7 with PDMP, an inhibitor of the final glucosylation step in the synthesis of GlcCer (Supplementary Figure 6). We found that DA, high cholesterol and endocannabinoids rescue daf-7 dauers generated in the presence of PDMP (Fig. 4d)10. However, as expected, PDMP treatment could not be rescued by addition of mmBCFA C17iso methyl ester because PDMP inhibits a later step of the synthesis of PEGCs. Thus, PEGCs deficiency could be rescued by supplementation with endocannabinoids.
In another set of experiments, we investigated whether PEGCs could abolish dauer formation in the endocannabinoids deficient strain daf-7;fat-3. As seen in Fig. 4e, synthetic mmPEGC-C22, the major PEGC species found in worms10, can partially rescue the dauer formation induced by the absence of endocannabinoids. Taking altogether, we propose that two unrelated lipid species, endocannabinoids and PEGCs, regulate worm cholesterol transport/mobilization through parallel pathways.
Discussion
The major finding of our study is that the endocannabinoids 2-AG and AEA regulate the transport/mobilization of cholesterol in C. elegans, which results in enhanced DA production. Several lines of evidence support this notion: endocannabinoids (i) abolish the developmental arrest caused by cholesterol depletion in wild-type and daf-7 worms, (ii) stimulate the reproductive development of daf-7 worms with diminished glucosylceramide/PEGC synthesis and (iii) rescue ncr-2;ncr-1 mutants with aberrant cholesterol transport.
Based on our data, we propose a tentative scheme of the interaction between endocannabinoids and PEGC in the control of cholesterol transport (Fig. 4f). Multiple reactions produce the precursors diacylglycerol (DAG), N-acyl-phosphatidylethanolamine (NAPE), and GlcCer required for the synthesis of 2-AG, AEA and PEGCs, respectively. The conversions of these precursors to the active compounds are carried out by not yet identified enzymes in C. elegans. 2-AG and AEA are probably produced by enzymes homologous to the mammalian DAG lipase (DAGL) and NAPE-specific phospholipase D (NAPE-PLD)30, although alternative biosynthetic pathways also exist12,20,31. The enzymatic system required for PEGCs production is completely unknown. Intriguingly, PEGCs cannot be synthesized in the absence of functional NCR-1 and NCR-2 proteins10. This suggests that in worms PEGCs mediate cholesterol transport downstream of NPC1. Endocannabinoids and PEGCs act in parallel to stimulate the mobilization/transport of cholesterol. This leads to delivery of the latter to DAF-9, production of DA and inhibition of the dauer-promoting activity of DAF-12. It should be noted that, in addition to their effect as regulators of cholesterol mobilization, endocannabinoids might also stimulate the synthesis of DA via regulation of alternative signaling pathways such as the monoaminergic neurotransmission, GPCR and MAP kinase – mediated cascades, etc.32,33. PEGCs, on the other hand, could also display pleiotropic effects due to their specific glycospingolipid structure: they might physically interact with cholesterol or affect its distribution by regulating different properties of the cellular membranes: their topology, fluidity or permeability34.
One important implication of the fact that the endocannabinoids and GlcCer/PEGCs act in parallel is that endocannabinoids might be involved in the regulation of a plethora of other known glucosylceramide-dependent processes in worms. These include the activation of the nutrient-sensing TOR signaling, the establishment of epithelial polarity in intestinal cells, foraging behavior and sensory neuron maturation29,35,36,37. Future studies will reveal the relationships between glycosphingolipids, cholesterol and endocannabinoids in the control of these processes.
Many of the physiological effects produced by endocannabinoids are not completely understood. Some of them may reflect their influence on cholesterol trafficking. In higher organisms, many aspects of the intracellular cholesterol transport are regulated by the transcription factors LXR and SREBP1. Intriguingly, LXR is one of the vertebrate homologs of DAF-12, which binds oxysterols38. This points to the interesting possibility that LXR and DAF-12 might have similar endocannabinoid-dependent activities in regard to the control of cholesterol homeostasis. On the other hand, endocannabinoids and endocannabinoid agonists are known to increase the hepatic expression of SREBP in mice39. Whether 2-AG and AEA could also operate via SREBP in worms is worth investigating.
Finally, deregulation of cholesterol and lipid homeostasis has major consequences on development and disease. Cholesterol metabolism in particular is closely interrelated with cardiovascular disease in humans. It is well known that dietary supplementation with omega-6 polyunsaturated fatty acids (PUFAs) reduces the risk of cardiovascular disease. In part, this is attributable to the observation that increased plasma levels of omega-6 PUFAs favorably affect plasma LDL-cholesterol and HDL-cholesterol levels40,41. While it is widely accepted that lipid metabolism is linked to cholesterol homeostasis38,42, exactly how PUFAs control cholesterol balance and metabolism remains largely unexplored. Hence, our findings point up the exciting scenario that the role of PUFA-derived endocannabinoids could be conserved in humans. Thus, unraveling the mechanisms by which endocannabinoids affect sterol-regulated processes in C. elegans could have important implications for the better understanding of human pathological conditions associated with impaired cholesterol homeostasis, such as atherosclerosis, non-alcoholic fatty liver disease and Niemann-Pick type C disease.
Materials and Methods
Materials
2-AG, AEA, noladin, EPEA and DL-threo-PDMP were purchased from Cayman Chemical (Ann Arbor, Michigan, USA). Cholesterol, AA, C17iso fatty acid, Dubelcco´s medium (DMEM) and antioxidant BHT were purchased from Sigma (Sigma-Aldrich, St. Louis, Missouri, USA). mmPEGC-C2210, Δ4-DA and Δ7-DA were provided by Prof. H.-J. Knölker43,44,45.
The worm strain N2 Bristol and the single mutants daf-7(1372), fat-3(ok1126), fat-4 (ok958), ncr-1;ncr-2 (JT10800) were obtained from Caenorhabditis Genetics Center (CGC). daf-9(dh6);dhEx24 was provided by Prof. A. Antebi.
Growth and maintenance of worm strains
Worms were routinely propagated on nematode growth medium (NGM) agar plates seeded with E. coli OP5046. Saturated cultures of E. coli OP50 or NA22 grown in lysogeny broth (LB) medium were concentrated 10 times and spread on NGM-agar plates. Worms were placed on the plates either as mixed stage populations or as embryos obtained by hypochlorite treatment. The temperature-sensitive Daf-c mutants were grown at 15 °C to grow in the reproductive mode and at 25 °C to arrest as dauer larvae.
RNAi by feeding
RNAi against let-767 was performed with E. coli HT115 double-stranded RNA (dsRNA)-producing strain from the Ahringer library. RNAi was performed in the first generation. Bleached embryos were left to hatch overnight in M9 at 20 °C and the resulting L1s were seeded to RNAi plates and grown at 20 °C or 15 °C according to the experiment. Similarly, faah-1 RNAi was performed in the first generation. Unlike the experiments with let-767, the worms subjected to faah-1 RNAi were seeded on the dsRNA-producing E. coli as embryos. Finally, the dauer phenotype was scored after 72 h or 144 h, depending on the assay. For biochemical analysis worms were harvested from RNAi plates after 72 h.
Dauer formation assays
In general, 80–100 L1s or embryos were transferred to NGM plates seeded with E. coli (HT115) containing RNAi against let-767, faah-1 or empty vector as a negative control. The supplementation with endocannabinoids (final concentration 50 µM), arachidonic acid (final concentration 50 µM), methyl esters from different fatty acids (final concentration 50 µM), mm-PEGC-C22 (final concentration 10 µM) and DL-threo-PDMP (final concentration 20 µM) was added to the bacteria immediately prior to seeding. The final concentrations of these compounds were calculated according to the volume of the NGM agar used for the preparation of the plates. It should be noted that the active concentrations of mm-PEGC-C22 published before10 have been calculated according to the volume of the bacterial loan seeded on the agar/agarose surface. If calculated using the latter method, the concentration of mm-PEGC-C22 used here would be 500 µM. Finally, after 72 h the percentage of dauers was scored. Δ4-DA and Δ7-DA43,44 were used alternatively in dauer rescue experiments with working concentrations that were chosen (1 µM and 90nM, respectively).
To determine dauer formation of daf-9(dh6) strain, embryos were placed on NGM plates with E. coli complemented with Δ7-DA or endocannabinoids. dhEx24 is an extrachromosomal array carrying active DAF-9 and nuclear GFP. After 3 days daf-9 null worms were identified on the basis of the absence of GFP signal and dauer formation was scored47.
Cholesterol deprivation assay
The protocol from Matyash et al.3 was carried out. Briefly, to produce sterol-depleted media, agar was replaced by agarose (Sigma-Aldrich) that was washed three times with chloroform to deplete the trace sterols in it. E. coli NA22 was grown in low-glucose, sterol-free DMEM culture medium (Sigma-Aldrich). Bacteria were rinsed with M9 before use and concentrated 20 times. Then the plates were seeded with 100 µl of bacteria and the supplements were added (or solvent carrier as negative control). 80–100 L1s were plated, incubated at 15° C for 6 days, and finally scored for percentage of dauers formed.
Lipid extraction and endocannabinoid analysis by HPLC-MS/MS
The protocol is an adaptation from Folch (1957)48. Lipid extracts were generated from approximately 100 mg of frozen worm pellets belonging to N2, daf-7, fat-3 and daf-7;fat-3 grown at 20 °C. The pellets were thawed on ice in 1.3 ml methanol and subjected to 4 min sonication on ice. After sonication, 1000 ppb of the internal standard 2-AG-d5 were added followed by 2.6 ml chloroform and 1.3 ml 0.5 M KCl/0.08 M H3PO4 to a final ratio of 1:2:1. Samples were vortexed and then sonicated in an ultrasonic water bath for 15 min. After vortexing twice for 1 min, samples were centrifuged for 10 min at 2.000 g to induce phase separation. The lower phase was collected into a clean glass tube, dried under nitrogen and re-suspended in 100 µl of acetonitrile.
2-AG and AEA were quantified from nematode samples by liquid chromatography (Ultimate 3000 RSLC Dionex, Thermo Scientic) coupled with an ESI triple quadrupole mass spectrometer (TSQ Quantum Access Max (QQQ), Thermo-Scientific). Reversed-phase HPLC column (C18 Hypersil-GOLD (50 x 2.1 mm), Thermo Scientific) was carried out employing the following step-wise gradient: 0 min (water 4:6 acetonitrile); 0.5 min (water 4:6 acetonitrile); 6.5 (water 2.5:7.5 acetonitrile); 7.5 min (water 2.5:7.5 acetonitrile); 8 min (water 4:6 acetonitrile). Column temperature was maintained at 40 °C and autosampler tray temperature was set at 10 °C. The following ionization conditions were used: ESI, positive-ion mode; drying gas (N2) temp., 300 °C; drying gas flow rate, 10 l/min; nebulizer pressure, 10 UA, and cap. voltage, 4 kV. Analyte detection was performed using NRM with the following transitions: m/z 379,2→289,2 for 2-AG; m/z 348,2→62,2 for AEA and m/z 384,2→289,2 for 2-AG-d5. Deuterated internal standard 2-AG-d5 was used for quantification, and peak-area ratios of the analyte to the internal standard were calculated as a function of the concentration ratios of the analyte to the internal standard. Sample concentrations were calculated from the calibration curve using an equally weighted linear regression (X) algorithm, where X represents concentration.
Generation of daf-7;fat-3 and daf-7;fat-4 double mutant strains
Homozygous fat-3(ok1126) male worms were crossed to daf-7(e1372) hermaphrodites. The hermaphrodites from the progeny laid eggs that at 25 °C developed into dauers or adults. The dauers were relocated to 15 °C to re-enter reproduction and their progeny was screened using PCR for homozygous daf-7(e1372);fat-3(ok1126) individuals. Using an equivalent strategy, homozygous fat-4(ok958) male worms were crossed to daf-7(e1372) hermaphrodites. The hermaphrodites from the progeny laid eggs that at 25 °C developed into dauers or adults. The dauers were relocated to 15 °C to re-enter reproduction and their progeny was screened using PCR for homozygous daf-7(e1372);fat-4(ok958) individuals.
GC-MS determination of fatty acid methyl-ester content
Lipid extraction was performed using Bligh and Dyer49 method. Briefly, the N2 and fat-3 were grown at 20 °C on E. coli OP50 for 72 h and then harvested from plates with 5–10 ml of M9 buffer. For the interference assay with let-767 RNAi, daf-7 worms were grown on E. coli OP50 with TH115 empty vector as a control, and with RNAi against let-767 at 20 °C for 72 h. Subsequently, worms were harvested as described above. Thereafter, worms were washed three times with M9 buffer to eliminate excess bacteria. The worm pellet was transferred to a glass tube and 3 ml of a chloroform:methanol 1:2 mixture was added. Dibutylhydroxytoluene (BHT) was added as an antioxidant at a final concentration of 5 µg/ml. The samples were incubated for 12 h at −20 °C. After incubation the samples were centrifuged and the supernatants were transferred to a new tube. 1 ml of KCl (1M) and 1 ml of chloroform were added to the samples, the samples were mixed by vortexing and then centrifugated for 2 min at 2000 g. Finally, the organic phases were transferred to new tubes and dried under N2 flow.
Chemical General Information
1H and 13C NMR spectra were acquired on a Bruker Avance II 300 MHz (75.13 MHz) using CDCl3 as solvent. Electrospray Ionization High-Resolution Mass Spectra (ESI-HRMS) were recorded on a Bruker MicrOTOF II with lock spray source. IR spectra were obtained using an FTIR Shimadzu spectrometer. Chemical reagents were purchased from commercial suppliers and used without further purification, unless otherwise noted. Solvents were analytical grade or were purified by standard procedures prior to use. Yields were calculated for material judged homogeneous by thin layer chromatography (TLC) and nuclear magnetic resonance (1H-NMR). All reactions were monitored by thin layer chromatography performed on silica gel 60 F254 pre-coated aluminum sheets, visualized by a 254 nm UV lamp, and stained with an ethanolic solution of 4-anisaldehyde. Column flash chromatography was performed using silica gel 60 (230–400 mesh).
2-AG-d5 preparation
For obtaining 2-AG-d5 as a deuterated internal standard for quantification assays, we followed a three-step protocol: differential protection, esterification and deprotection.
Differential protection step: In order to only protect primary alcohols, a solution of glycerol-d8 (38 mg, 0.18 mmol) in anhydrous dichloromethane (5 ml) under inert atmosphere was prepared and cooled at −78 ºC. Then, collidine (54 mg, 0.36 mmol) and tert-butyl dimethyl silyl chloride (TBDMSCl, 70 mg, 0.54 mmol) was added. The mixture reacted for 3 h at −78 ºC, then warmed to room temperature and stirred for additional 12 h. Brine was added and the solution was extracted with dichloromethane. The combined organic extracts were dried over sodium sulfate and evaporated. Products were purified by column chromatography on silica gel with increasing hexane/ethyl acetate gradients. 1-O,3-O-Bis(TBDMS) glycerol-d6 was obtained as a colorless liquid in 44% yield (55 mg). For the esterification step, a solution of 1-O,3-O-Bis(TBDMS) glycerol-d6 (10 mg, 0.03 mmol) in anhydrous dichloromethane (2 ml) under inert atmosphere was prepared and cooled at 0 ºC. Next, arachidonic acid (36 mg, 0.12 mmol), 4-dimethylaminopyridine (15 mg, 0.12 mmol) and N,N’-dicyclohexylcarbodiimide (15 mg, 0.12 mmol) were added in that order. The mixture reacted for 3 h at 0 ºC, and was then warmed to room temperature and stirred for additional 12 h. Water was added and the solution was extracted with dichloromethane. Combined organic extracts were dried over sodium sulfate and evaporated. Products were purified by column chromatography on silica gel with increasing hexane/ethyl acetate gradients. 1-O,3-O-bis(TBDMS)−2-AG-d5 was obtained as a yellowish liquid in 80% yield (15 mg). For the deprotection step, a solution of 1O,3O-Bis(TBDMS)−2-AG-d5 (15 mg, 0.025 mmol) in tetrahydrofuran (2 ml) was prepared. Then, a 1M solution of tetrabutylammonium fluoride (150 ml, 0.15 mmol) was added cautiously dropwise. Once the reaction was complete (1 h), water was added and the solution was extracted with dichloromethane. Combined organic extracts were dried over sodium sulfate and evaporated. A quantitative amount of product 2-AG-d5 (10 mg, quantitative) was obtained as a yellowish liquid.
Methyl- ester preparation
For the derivatization of fatty acids prior to GC-MS analysis, 0.5 ml of anhydrous sodium methoxide 0.5 M were added to the lipid samples and then incubated 30 min at room temperature. Then 1 ml of HCl (2 N) was added for neutralization of the reaction. The fatty acid methyl esters were extracted with hexane and finally resuspended in 500 ml of hexane.
For obtaining the AA and C17iso methyl-esters used for supplementation assays we followed a protocol based on Walker et al.50. Briefly, a solution of diazomethane in diethyl ether was added cautiously dropwise to a stirred solution of the appropriate fatty acid (50 mg, 0.16 mmol) in dichloromethane (5 ml) until the yellow color persisted. The excess of the reagent was vented under a nitrogen purge. The solvent was evaporated and the product dried under vacuum to provide the methyl ester as a colorless oil (54 mg, quantitative) that was used without further purification.
Gas chromatography coupled to mass spectrometry (GC-MS)
The methyl ester analysis was performed in a Shimadzu GC-2010 Plus equipment. The column used was a Supelco WAX-10 (Sigma Aldrich) 100% polyethyleneglycol. The helium flux was 1 ml/min and the heating program was 180 °C (0 min to 32 min) and then a gradient increasing 3 °C/min from 180 °C to 240 °C. The split was 1/30 and the ionization voltage was 70 eV with an ionic range from 50 to 600 Da. The ion specters were registered as relative abundance in function of mass/charge (m/z). Peak assignation was done using the mixture of standards of fatty acid methyl esters AGs Supelco (Sigma Aldrich).
References
Ikonen, E. Cellular cholesterol trafficking and compartmentalization. Nat. Rev. Mol. Cell Biol. 9, 125–138 (2008).
Iaea, D. B. & Maxfield, F. R. Cholesterol trafficking and distribution. Essays Biochem. 57, 43–55 (2015).
Matyash, V. et al. Sterol-derived hormone(s) controls entry into diapause in Caenorhabditis elegans by consecutive activation of DAF-12 and DAF-16. PLoS Biol. 2, e280 (2004).
Matyash, V. et al. Distribution and transport of cholesterol in Caenorhabditis elegans. Mol. Biol. Cell 12, 1725–36 (2001).
Li, J., Brown, G., Ailion, M., Lee, S. & Thomas, J. H. NCR-1 and NCR-2, the C. elegans homologs of the human Niemann-Pick type C1 disease protein, function upstream of DAF-9 in the dauer formation pathways. Development 131, 5741–5752 (2004).
Motola, D. L. et al. Identification of ligands for DAF-12 that govern dauer formation and reproduction in C. elegans. Cell 124, 1209–23 (2006).
Fielenbach, N. & Antebi, A. C. elegans dauer formation and the molecular basis of plasticity. Genes Dev. 22, 2149–65 (2008).
Antebi, A., Yeh, W. H., Tait, D., Hedgecock, E. M. & Riddle, D. L. daf-12 encodes a nuclear receptor that regulates the dauer diapause and developmental age in C. elegans. Genes Dev. 14, 1512–1527 (2000).
Antebi, A., Culotti, J. G. & Hedgecock, E. M. daf-12 regulates developmental age and the dauer alternative in Caenorhabditis elegans. Development 125, 1191–1205 (1998).
Boland, S. et al. Phosphorylated glycosphingolipids essential for cholesterol mobilization in Caenorhabditis elegans. Nat. Chem. Biol. 13, 647–654 (2017).
Kobayashi, T. et al. Late endosomal membranes rich in lysobisphosphatidic acid regulate cholesterol transport. Nat. Cell Biol. 1, 113–118 (1999).
Lucanic, M. et al. N-acylethanolamine signalling mediates the effect of diet on lifespan in Caenorhabditis elegans. Nature 473, 226–9 (2011).
Skaper, S. D. & Di Marzo, V. Endocannabinoids in nervous system health and disease: the big picture in a nutshell. Philos. Trans. R. Soc. Lond. B. Biol. Sci. 367, 3193–200 (2012).
McPartland, J. M., Matias, Marzo, I., Di, V. & Glass M. Evolutionary origins of the endocannabinoid system. Gene 370, 64–74 (2006).
Iannotti, F. A., Di Marzo, V. & Petrosino, S. Endocannabinoids and endocannabinoid-related mediators: Targets, metabolism and role in neurological disorders. Prog. Lipid Res. 62, 107–128 (2016).
Watts, J. L. & Browse, J. Genetic dissection of polyunsaturated fatty acid synthesis in Caenorhabditis elegans. Proc. Natl. Acad. Sci. USA 99, 5854–9 (2002).
Watts, J. L., Phillips, E., Griffing, K. R. & Browse, J. Developmental Defects in Caenorhabditis elegans fat-3 Mutants. 589, 581–589 (2003).
Ren, P. et al. Control of C. elegans larval development by neuronal expression of a TGF-beta homolog. Science 274, 1389–91 (1996).
Liu, T., Zimmerman, K. K. & Patterson, G. I. Regulation of signaling genes by TGF-beta during entry into dauer diapause in C. elegans. BMC Dev. Biol. 4, 11 (2004).
Vrablik, T. L. & Watts, J. L. Polyunsaturated fatty acid derived signaling in reproduction and development: insights from Caenorhabditis elegans and Drosophila melanogaster. Mol. Reprod. Dev. 80, 244–59 (2013).
Lehtonen, M. et al. Determination of endocannabinoids in nematodes and human brain tissue by liquid chromatography electrospray ionization tandem mass spectrometry. J. Chromatogr. B 879, 677–694 (2011).
Entchev, E. V. et al. LET-767 is required for the production of branched chain and long chain fatty acids in Caenorhabditis elegans. J. Biol. Chem. 283, 17550–60 (2008).
Silvestri, C. & Di Marzo, V. The endocannabinoid system in energy homeostasis and the etiopathology of metabolic disorders. Cell Metab. 17, 475–490 (2013).
Hanus, L. et al. 2-arachidonyl glyceryl ether, an endogenous agonist of the cannabinoid CB1 receptor. Proc. Natl. Acad. Sci. USA 98, 3662–5 (2001).
Watts, J. L. & Ristow, M. Lipid and carbohydrate metabolism in Caenorhabditis elegans. Genetics 207, 413–446 (2017).
Gerisch, B. et al. A bile acid-like steroid modulates Caenorhabditis elegans lifespan through nuclear receptor signaling. Proc. Natl. Acad. Sci. USA 104, 5014–9 (2007).
Rosenbaum, A. I. & Maxfield, F. R. Niemann-Pick type C disease: Molecular mechanisms and potential therapeutic approaches. J. Neurochem. 116, 789–795 (2011).
Sym, M., Basson, M. & Johnson, C. A model for Niemann – Pick type C disease in the nematode Caenorhabditis elegans. Curr. Biol. 8, 527–530 (2000).
Zhu, H., Shen, H., Sewell, A. K., Kniazeva, M. & Han, M. A novel sphingolipid-TORC1 pathway critically promotes postembryonic development in Caenorhabditis elegans. Elife 2013, 1–19 (2013).
Harrison, N. et al. Characterization of N-Acyl Phospholipase-D Isoforms in the Nematode Caenorhabditis elegans. PLoS One 2, 1–21 (2014).
Lin, Y.-C. Y.-H. et al. Diacylglycerol lipase regulates lifespan and oxidative stress response by inversely modulating TOR signaling in Drosophila and C. elegans. Aging Cell 13, 755–64 (2014).
Pastuhov, S. I., Matsumoto, K. & Hisamoto, N. Endocannabinoid signaling regulates regenerative axon navigation in Caenorhabditis elegans via the GPCRs NPR-19 and NPR-32. Genes to Cells 21, 696–705 (2016).
Oakes, M., Jing Law, W., Clark, T., Bamber, B. & Komuniecki, R. Cannabinoids activate monoaminergic signaling to modulate key C. elegans behaviors. J. Neurosci. https://doi.org/10.1523/JNEUROSCI.3151-16.2017 (2017).
Hannich, J. T., Umebayashi, K. & Riezman, H. Distribution and Functions of Sterols and Sphingolipids. Cold Spring Harb. Perspect. Biol. 3, 1–14 (2011).
Zhang, H. et al. Apicobasal domain identities of expanding tubular membranes depend on glycosphingolipid biosynthesis. Nat. Cell Biol. 13, 1189–1201 (2011).
Kniazeva, M., Crawford, Q. T., Seiber, M., Wang, C. Y. & Han, M. Monomethyl branched-chain fatty acids play an essential role in Caenorhabditis elegans development. PLoS Biol. 2 (2004).
Kniazeva, M., Zhu, H., Sewell, A. K. & Han, M. A Lipid-TORC1 Pathway Promotes Neuronal Development and Foraging Behavior under Both Fed and Fasted Conditions in C. elegans. Dev. Cell 33, 260–271 (2015).
Hong, C. & Tontonoz, P. Liver X receptors in lipid metabolism: opportunities for drug discovery. Nat. Rev. Drug Discov. 13, 433–444 (2014).
Osei-hyiaman, D. et al. Endocannabinoid activation at hepatic CB 1 receptors stimulates fatty acid synthesis and contributes to diet-induced obesity. J. Clin. Invistigation 115, 1298–1305 (2005).
Harris, W. S. et al. Omega-6 fatty acids and risk for cardiovascular disease: A science advisory from the American Heart Association nutrition subcommittee of the council on nutrition, physical activity, and metabolism; council on cardiovascular nursing; and council on epidem. Circulation 119, 902–907 (2009).
Katan, M. B. B. Omega-6 polyunsaturated fatty acids and coronary heart disease. Am. J. Clin. Nutr. 89, 1283 (2009).
Lee, S. D. & Tontonoz, P. Liver X receptors at the intersection of lipid metabolism and atherogenesis. Atherosclerosis 242, 29–36 (2015).
Martin, R. et al. Synthesis and biological activity of the (25R)-cholesten-26-oic acids-ligands for the hormonal receptor DAF-12 in Caenorhabditis elegans. Org. Biomol. Chem. 7, 909–920 (2009).
Martin, R., Däbritz, F., Entchev, E. V., Kurzchalia, T. V. & Knölker, H.-J. Stereoselective synthesis of the hormonally active (25S)-D7-dafachronic acid, (25S)-D4-dafachronic acid and (25S)-Cholestenoic Acid. Org. Biomol. Chem. 6, 4293–4295 (2008).
Saini, R. et al. Stereoselective synthesis and hormonal activity of novel dafachronic acids and naturally occurring steroids isolated from corals. Org. Biol. Chem. 10, 4159–4163 (2012).
Brenner, S. The Genetics of Caenorhabditis elegans. Genetics 77, 71–94 (1974).
Gerisch, B., Weitzel, C., Kober-eisermann, C., Rottiers, V. & Antebi, A. A Hormonal Signaling Pathway Influencing C. elegans Metabolism, Reproductive Development, and Life Span. Dev. Cell 1, 841–851 (2001).
Folch, J., Lees, M. & Sloane Stanley, G. H. A Simple method for the Isolation and Purification of Total Lipids from Animal Tissues. 55, 999–1033 (1987).
Bligh, E. G. & Dyer, W. J. A Rapid Method of Total Lipid Extraction and Purification. Can. J. Biochem. Physiol. 37, 911–917 (1959).
Walker, M., Roberts, D. & Dumbroff, E. B. Convenient apparatus for methylating small samples with diazomethane. J. Chromatogr. 241, 390–391 (1982).
Acknowledgements
Supported by grants (PICTs 2155 and 3693) from the Agencia Nacional de Promoción Científica y Técnica (ANPCYT), a Humboldt Research Award from the Alexander von Humboldt Foundation and a grant Fundación Bunge y Born-Max Planck. C.G., G.P., and E.O.J.P. are fellows from Fundación Bunge y Born, ANPCYT and CONICET respectively. We thank the anonymous reviewer for many insightful comments and suggestions. We thank Fanny Mende for sharing unpublished data. We thank Prof. T. Keith Blackwell’s lab for the faah-1 RNAi construct. We are thankful to Prof. A. Antebi for the worm strains and to WormBase. Some strains were also obtained from the CGC, which is funded by NIH Office of Research Infrastructure Programs (P40 OD010440).
Author information
Authors and Affiliations
Contributions
C.G., G.M.P. and S.P. performed phenotypic and biochemical analysis; C.G. conducted microscopy; G.M.P. performed mass spectrometry; S.B. contributed analytical tools; U.S. and H.-J.K. contributed synthetic dafachronic acids and mmPEGC-C22; G.R.L. and E.O.J.P. contributed synthetic deuterated endocannabinoids; S.G.A., G.M.P. and C.G. performed GCMS; C.G., G.M.P., S.P., T.V.K. and D.d.M. wrote the paper.
Corresponding authors
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Galles, C., Prez, G.M., Penkov, S. et al. Endocannabinoids in Caenorhabditis elegans are essential for the mobilization of cholesterol from internal reserves. Sci Rep 8, 6398 (2018). https://doi.org/10.1038/s41598-018-24925-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-24925-8
This article is cited by
-
Cannabidiol and Tetrahydrocannabinol Antinociceptive Activity is Mediated by Distinct Receptors in Caenorhabditis elegans
Neurochemical Research (2024)
-
Cannabinoids and healthy ageing: the potential for extending healthspan and lifespan in preclinical models with an emphasis on Caenorhabditis elegans
GeroScience (2024)
-
Anandamide Modulates Thermal Avoidance in Caenorhabditis elegans Through Vanilloid and Cannabinoid Receptor Interplay
Neurochemical Research (2024)
-
A metabolic switch regulates the transition between growth and diapause in C. elegans
BMC Biology (2020)
-
An endocannabinoid catabolic enzyme FAAH and its paralogs in an early land plant reveal evolutionary and functional relationship with eukaryotic orthologs
Scientific Reports (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.