Abstract
Primary Toxoplasma gondii infection in pregnant women may result in abortion, stillbirth, or lifelong disabilities of the unborn child. One of the main transmission routes to humans is consumption of raw or undercooked meat containing T. gondii tissue cysts. We aim to determine and compare the regional distribution of T. gondii seroprevalence in pregnant women and meat-producing livestock in China through a systematic literature review. A total of 272 eligible publications were identified from Medline, Scopus, Embase and China National Knowledge Infrastructure. Apparent and true seroprevalence were analysed by region using a novel Bayesian hierarchical model that allowed incorporating sensitivity and specificity of the applied serological assays. The true seroprevalence of T. gondii in pregnant women was 5.0% or less in seven regions of China. The median of the regional true seroprevalences in pigs (24%) was significantly higher than in cattle (9.5%), but it was not significantly higher than in chickens (20%) and small ruminants (20%). This study represents the first use of a Bayesian hierarchical model to obtain regional true seroprevalence. These results, in combination with meat consumption data, can be used to better understand the contribution of meat-producing animals to human T. gondii infection in China.
Similar content being viewed by others
Introduction
Toxoplasma gondii is an obligate intracellular protozoan parasite that causes toxoplasmosis. The parasite is widely distributed in the world and can infect a wide range of warm-blooded animals, including humans, pets and livestock. In the general population, T. gondii infection can remain asymptomatic, cause lymphadenopathy and flu-like symptoms, or lead to eye disease, most frequently chorioretinitis, while in immune-compromised patients, it can be fatal1. Pregnant women constitute a specific risk group: if primary infection is acquired during pregnancy, this may lead to abortion, stillbirth and neurological disorders in the unborn child2. Congenital and acquired toxoplasmosis caused more than 20 million new cases worldwide in 2010, resulting in an estimated global disease burden of 1.68 million (95% UI 1.24–2.45 million) disability-adjusted life years (DALYs), of which 829,000 DALYs (95% UI 561,000–1.26 million) were estimated to be foodborne3. In a global multicriteria based ranking (considering public health, animal health, microbial ecology, agribusiness and trade, and socio-economic impact) T. gondii ranked fourth out of 24 foodborne parasites4.
The main routes of postnatal infection for humans are consumption of raw or undercooked meat containing tissue cysts and food or water contaminated with sporulated oocysts shed by the primary infected definite hosts, felines1. As intermediate hosts of T. gondii, meat-producing animals serve as one of the main sources of human infections5. It was estimated that 30–63% of infections in pregnant women from six large European cities was attributed to meat6. As the effectiveness of treatment is unclear7,8, prevention of infection is so far the most important strategy but relies on knowledge of the relative attribution of different transmission routes. Results from an extensive literature review showed that there is a positive relationship between detection of antibodies to T. gondii and presence of this parasite in pigs, chickens and small ruminants, but not in cattle and horses9. Thus, with the exception of cattle and horses the seroprevalence and geographical distribution of the T. gondii infection in different meat-producing animals gives an indication of the risk of human infection via consumption of undercooked meat and is useful for developing health education material for pregnant women and other risk groups. In China, the seroprevalence of T. gondii in two national surveys conducted in 1988–1992 and 2001–2004 has increased from 5.2% to 7.9%10. The geographical distribution of the T. gondii seroprevalence in the general population and meat-producing animals is not reported systematically and most of the studies were published in Chinese which are not easily accessible for the international scientific community. Apart from the two national surveys, studies concerning T. gondii infection in the general population were hardly available, thus we decided to use data from pregnant women as a proxy for the general population.
In epidemiological studies true prevalence (TP) rather than apparent prevalence (AP) is the parameter of interest, and requires information on test sensitivity (Se) and specificity (Sp). A variety of serological assays have been developed for the detection of T. gondii-specific immunoglobulins11 and enzyme-linked immunosorbent assay (ELISA), modified agglutination test (MAT), and indirect hemagglutination antibody test (IHA) are commonly used in China. Unfortunately, estimates of Se and Sp are often lacking or evaluated in the absence of appropriate reference tests or on samples that are not relevant for the target population. Bayesian modelling is therefore increasingly used for veterinary epidemiological studies to infer true prevalence while taking into account the uncertainty of Se and Sp12,13,14,15,16. Furthermore, the Bayesian framework allows explicitly modelling complex hierarchical structures, such as studies nested within regions, which in turn are nested within a country. This has the advantageous side-effect that regions in which few or no studies were performed, can “borrow strength” from the remaining regions, and data gaps can be imputed14,17. To our knowledge, however, both applications i.e., true prevalence estimation and hierarchical modelling with data imputation has never been combined in a single model.
The present study estimates the seroprevalence of T. gondii in pregnant women and in the main meat-producing animals (i.e., pig, cattle, sheep, goat, chicken, duck, goose and donkey) from different regions of China and analyses the potential link between the seroprevalence in humans and livestock. Data on apparent seroprevalence were collected by systematically reviewing international and Chinese bibliographic databases. A Bayesian hierarchical model that allowed incorporating the sensitivity and specificity of the applied serological assays was used to estimate true prevalence by region and impute possible data gaps.
Results
Characteristics of eligible studies
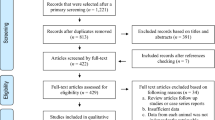
The selection process of published papers for pregnant women and livestock is summarized in a Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) flow diagram as shown in Fig. 1. A total of 72 eligible studies which reported the seroprevalence of IgG antibodies to T. gondii for pregnant women were collected from 21 provinces of China; no eligible studies were conducted in Chongqing, Fujian, Hainan, Jiangxi, Neimenggu, Ningxia, Qinghai, Tianjin, Yunnan provinces or the Tibet Autonomous Region (Xizang). The included pregnant women were at different weeks of their pregnancy, and the information of mean age was available from 31 studies, and ranged from 25 to 32 years old.
The literature review identified 200 publications for livestock from 30 provinces. None of the included studies were performed in Tianjin province. A total number of 240 studies were published in these 200 publications. The studies for pregnant women from 21 provinces and for pigs, chickens, cattle and small ruminants from the 30 provinces were merged to seven regions of China (Fig. 2). The entire list of 272 selected studies can be found as Supplementary File S1.
Seven regions from 31 provinces of mainland China. Thirty-one provincial-level divisions of mainland China were grouped as Central, East, North, Northeast, Northwest, South and Southwest China. Geographic data for mainland China was obtained from “Global Administrative Areas” (http://www.gadm.org/home). Map was created using R version 3.3.1 (https://www.r-project.org/).
Seroprevalence of pregnant women
In pregnant women, a small variation of non-modelled apparent prevalence (obtained by summing the data per region) was observed between different regions of China. The non-modelled AP ranged from 2.1% to 11% in all seven regions (Table 1). The apparent seroprevalence estimates from the hierarchical model were similar to the results of non-modelled AP. The highest mean AP from the hierarchical model was identified in Northeast (5.7%, 95% PPI (posterior prediction interval) [2.9–11%]), and the lowest in Northwest China (3.1%, 95% PPI [1.2–5.4%]) (Table 2).
Extracted information of Se and Sp from the commercial kits used in pregnant women in the selected studies and the parameters of beta distribution used in the Bayesian hierarchical model are shown in Table 3. A total number of 49 out of 72 studies used commercial kits with unknown test characteristics. The mean posterior seroprevalences in Chinese pregnant women and the mean posterior estimates for serological test characteristics are shown in Tables 4 and 5. The Se and Sp adjusted seroprevalences in Chinese pregnant women ranged from 2.4% to 5.0% among different regions. Trace plots and the potential scale reduction factors showed good convergences for the parameters in all models. The codes for the Bayesian hierarchical model can be found as Supplementary File S2.
Seroprevalence of livestock
A wide variation of non-modelled apparent seroprevalence was observed in different animal species (Table 1). The non-modelled AP of T. gondii in ducks, goose and donkeys ranged from 9.4–27%, 1.7–21% and 0–24% respectively. However, since only few studies were eligible from ducks (5), geese (5), and donkeys (5) through the literature review, these animal species were not included in meta-analysis and correlation analysis with pregnant women. Among the other four animal species, the non-modelled AP of T. gondii infection was higher in pigs and chickens than in small ruminants and cattle (Table 1). The highest non-modelled AP was found in pigs (53%) in Southwest China and the lowest in cattle (5.8%) in Southwest China. No eligible studies in cattle and small ruminants were conducted in the Southern region of China.
The results of T. gondii apparent seroprevalence in different animals from the hierarchical models were similar to the results of non-modelled AP and they are shown in Table 2. The T. gondii modelled AP in small ruminants and cattle in South China were imputed as 9.6% (95% PPI, 2.2–25%) and 11% (95% PPI, 3.6–25%).
Extracted information of Se and Sp from the commercial kits used in livestock in the selected studies and the parameters of beta distribution used in the Bayesian hierarchical model are shown in Table 3. Among the 240 records, the numbers of records using kit 1–4 were 116, 9, 8 and 26 respectively, 48 studies used ELISA kits with unknown Se and Sp, 33 studies used IHA kits with unknown Se and Sp. The mean posterior estimates of the regional true seroprevalence in pigs (20–33%), chickens (11–24%), small ruminants (15–24%) and cattle (6.8–12%) are summarized in Table 4. Wide posterior probability intervals for the sensitivities and specificities were found in some of the included kits used for animals (Table 5). The Kruskal-Wallis test indicated that the median of the regional true seroprevalences in different species was significantly different from each other (p < 0.01). The post-hoc analysis results showed that the median of the regional true seroprevalences in pigs (24%) was significantly higher than in cattle (9.5%) (p < 0.05) and it was not significantly higher than in chickens (20%) and small ruminants (20%). Trace plots and the potential scale reduction factors showed good convergences for the parameters in all models.
The results of Spearman’s rank correlation coefficient showed that there is no strong relationship between the estimated true seroprevalence in pregnant women and the estimated true seroprevalence in any of the livestock species from the corresponding seven regions of China (Table 6). However, the true seroprevalence in chicken was found strongly correlated with the true seroprevalence in small ruminant (Spearman correlation = 0.86, p = 0.01).
Discussion
T. gondii causes a high disease burden in humans and is a source of economic losses to livestock industries18,19. The increased seroprevalence in T. gondii in the Chinese human population together with a notable increase in number of immune-compromised patients suffering cancer and HIV make this opportunistic parasitic disease an important public health challenge to China10,20. The aims of our study were to determine the seroprevalence of T. gondii in pregnant women and in main meat-producing animals from different regions of China and to examine the potential link between the seroprevalence in humans and livestock.
A total of 72 publications on pregnant women and 200 publications on livestock were included in the dataset. The selected eligible studies for pregnant women covered all seven regions of China. However, data gaps were identified for cattle and small ruminant from Southern China and limited eligible studies were found in ducks, goose and donkeys. These data gaps called for the use of a hierarchical model in which the fitted national (logit-transformed) seroprevalence and the fitted between-region variance could be used to estimate the seroprevalence for regions where no data were available. In all of the eligible publications, seroprevalence data were reported without correction for test sensitivity and specificity. Correctly determining disease status largely depends on the sensitivity and specificity of the serological test. The chance of having a false result can be considerable and the prevalence of test positives may deviate from prevalence of truly infected individuals21. For that reason, data on test characteristics were additionally collected from literature. In our study, most of the selected studies dealing with the T. gondii IgG seroprevalence in pregnant women and many of the studies on livestock used kits with unknown Se and Sp. Moreover, different values of Se and Sp were reported in literature for the same kit to detect T. gondii antibodies in animals. This variation may be due to different characteristics of the reference population (e.g. different levels of exposure to additional pathogens or other biological confounders) and the sampling strategies used in the validation procedures22. Thus, it is inappropriate to assume that the test characteristics are constant over populations23. Therefore a Bayesian approach to estimate the true seroprevalence of T. gondii infection was used. In this approach, prior information about test characteristics was incorporated into the analysis as random variables described using probability distributions in the modelling process. They are therefore not fixed, constant values over different populations. Because insufficient convergence occurred from the initial runs of the model, we used truncated beta distributions for the priors of Se and Sp (Table 3) assuming that the values of Se and Sp of used kits were at least larger than 0.1. After truncation of the distribution of priors, the models for both pregnant women and livestock showed good convergence. In general, the Bayesian hierarchical model estimates for T. gondii true seroprevalence in pregnant women and livestock were similar to the non-modelled apparent seroprevalence but with wider 95% posterior probability intervals. In our opinion, the seroprevalences based on the Bayesian hierarchical model provide the most appropriate estimates of seroprevalence, as regional variation and information on test characteristics are taken into account. The uncertainty present due to the use of insufficiently validated diagnostic kits remains unnoticed when apparent prevalences are reported, but is reflected in the wide posterior probability intervals from the Bayesian hierarchical model. Moreover, the Bayesian hierarchical model allowed us to impute seroprevalence estimates for regions that lacked data.
The true seroprevalence from the Bayesian hierarchical model in Chinese pregnant women obtained from this study ranged from 2.4% to 5.0% in all seven regions of China. Even though the results were low compared to some countries24, this also means that the majority of pregnant women in China are susceptible to a primary infection and their babies to congenital toxoplasmosis. The true seroprevalence was also lower than the overall seroprevalence in Chinese cancer patients reported from a systematic review (21%)25. This is however not a surprise, as cancer patients are generally older than pregnant women and thus have experienced more time at risk of infection.
The results of true seroprevalence for livestock showed that T. gondii infection is wide spread in meat-producing animals in different regions of China. The median of the true seroprevalences from seven regions of China in pigs (24%) was significantly higher than in cattle (9.5%), but it was not significantly higher than in chickens (20%) and small ruminants (20%). In addition, results showed that the regional variation of true seropevalences in chickens (from 11% to 24%) was strongly correlated with variation in small ruminants (Spearman correlation = 0.86, p = 0.01). Chickens are considered good indicators of T. gondii contamination in the environment, as they become infected mostly by ingesting T. gondii oocysts-contaminated soil26. Therefore, regional variability may indicate variation in environmental contamination with oocysts. In China, sheep is one of the major grazing livestock and they are grazing rotationally by following a predetermined range and routine in pastures27. The outdoor access of these animals means that, similarly to chickens, environmental contamination with oocysts is also major risk of infection small ruminants, which may explain the correlation between regional variations of seroprevalence in these species. However, seroprevalence is also strongly associated with farming system with the seroprevalence of T. gondii infection higher in outdoor farming systems than in indoor farming systems28,29,30. In addition, the age of animals, number of cats present in the farm, and feed source are considered as important risk factors associated with T. gondii seroprevalence31,32. Farming system and exposure to risk factors are likely to also vary by region, and may be correlated for the different species (e.g. in regions with a lot of backyard poultry, backyard farming may also be more common for small ruminants). Therefore, to better understand regional variability in seroprevalence it is important to collect and analyse data taking into account exposure to potential risk factors that are related to T. gondii infection in future studies. This type of information was lacking from most of the publications included in the review.
With the exception of cattle and horses, the risk of human T. gondii infection via undercooked meat likely increases with a higher seroprevalence in the animal species. Nonetheless, no strong regional relationship between the true seroprevalence in livestock and pregnant women was found. This may be due to the limited data points in our analysis (7 regions). More importantly, the total meat consumption volume and species-specific preparation habits play crucial roles for the relative attribution of different meat-producing animals on a population level33. In China, pork is the dominant type of meat consumed by the whole population, except for some religious groups, followed by poultry, beef and lamb, the average annual consumption volumes in 2015 were 20.1, 8.4, 1.6 and 1.2 kg respectively34. The highest seroprevalence and total amount of consumption make pork a good potential source of human infections. Additionally, people living in Yunnan, Guizhou and Sichuan provinces eat raw or undercooked pork and beef in their tradition, and the seroprevalence in the ethnic groups from these provinces were found higher than the general population35. In our study, the true seroprevalence in pregnant women from the same region was found to be one of the highest 4.8% (1.7–14%). Due to the diversification of food sources and preparation habits among different regions and ethnic groups, the risk of human infection can vary accordingly. To determine the risk of humans to become infected via the different meat-producing animals, information on prevalence in livestock and meat consumption needs to be combined in a quantitative risk assessment36.
In conclusion, the seroprevalence and geographical distribution of T. gondii infection in pregnant women and meat-producing animals in China were systematically reviewed and summarized. The results obtained from Bayesian hierarchical models showed that T. gondii seroprevalence ranged from 2.4% to 5.0% in pregnant women, pigs (20–33%) and chickens (11–24%) had higher true seroprevalence than small ruminants (15–24%) and cattle (6.8–12%). Studies to better evaluate the performance of kits are needed to get more accurate estimates of T. gondii seroprevalence in humans and animals. More detailed insight in the geographical distribution of T. gondii prevalence in humans and livestock animals can be helpful for making effective intervention strategies to reduce the burden of this disease in the Chinese population. However, the risk of meatborne T. gondii infection in humans is not only depending on the prevalence in meat-producing animals but also on consumed volumes and food preparation habits. Therefore the results obtained from this study should be used to determine meatborne toxoplasmosis risk by using a quantitative microbial risk assessment of T. gondii infection in China.
Methods
Literature review and data sources
Relevant studies on the seroprevalence of T. gondii infection in pregnant women and farm animals in the mainland of China were searched through the Ovid Medline, Scopus, Embase and China National Knowledge Infrastructure (CNKI) electronic databases for English and Chinese publications. Cochrane guidelines and European Food Safety Authority (EFSA) guidance for carrying out systematic reviews were followed for identifying eligible studies37,38, and the PRISMA guidelines was followed for reporting39. The key elements of this review question were: population (pregnant women and meat-producing animals in China) and outcome (T. gondii seroprevalence). The literature search on Medline, Scopus, Embase was last updated on 20/10/2017, and last updated on CNKI on 18/05/2016.
Search strategy and study selection
All population-based studies published in the last 16 years (2000–2016) that reported the seroprevalence of anti-T. gondii antibodies in Chinese pregnant women or at least one of the animal species of interest were considered for inclusion. There was no restriction on language and the sample size of the study. The combination of “Toxoplasma”, “gondii”, “toxoplasmosis”, “China”, and “Chinese” were used as search terms in Medline, Scopus and Embase, the combination of “T. gondii”, “toxoplasmosis”, “pig”, “cattle”, “sheep” and “goat”, “chicken”, “duck”, “goose” and “donkey” in Chinese were searched in CNKI. One reviewer (HD), fluent in English and Chinese, read the titles and abstracts of all publications retrieved from the electronic databases and excluded those that clearly did not meet the aforementioned selection criteria. In the next stage, all potential eligible studies were retrieved in full-text and checked by the same reviewer.
Several exclusion criteria were used to select eligible studies: (a) Descriptive studies, reviews, case reports, editorials or letters to the editors without original data, individual animal diagnosis and treatments (epidemiological cross-sectional studies were preferred). (b) Studies not representative for our target population. (c) Animal species not intended for meat consumption (e.g. animals for research purpose). (d) Studies limited to experimental infection with T. gondii (rather than natural infection). (e) The outcomes of the studies (T. gondii infection) were not confirmed by a serological assay. (f) Information about the total number of seropositive samples and sample size were not available. (g) Studies not conducted in the mainland of China. (h) Duplicated data.
Information from all relevant studies was extracted and coded into a Microsoft Excel datasheet. Variables extracted included author information, year of publication, animal species, period of data collection, location of the study, diagnostic method, seroprevalence or number of seropositive cases and sample size. If a publication contained multiple studies (e.g. different livestock species or regions), data were extracted separately. For pregnant women, many studies additionally reported the prevalence of IgM antibodies, but only data on IgG seroprevalence of T. gondii were included in the analysis.
Data analysis
Hierarchical meta-analysis model
The 31 provinces of mainland China were categorized into seven regions, including central, east, north, northeast, northwest, south and southwest of China in our study. Apparent seroprevalence in pregnant women and livestock from different regions were summarized using a three-level hierarchical meta-analysis model. First, at the individual study level, the number of seropositive samples (\({x}_{i}\)) out of total number of tested samples (\({n}_{i}\)) in every single study \(i\) was assumed to follow a binomial distribution, equation (1). The logit-transformed seroprevalence (\(A{P}_{i}\)) of every individual study, conducted in a certain region \(j\), was assumed to arise from a normal distribution with a region-specific mean seroprevalence (\({\theta }_{j}\)) and within-region variance (\({\sigma }_{w}^{2})\), equation (2). Second, at the regional level, every specific regional seroprevalence was assumed to arise from a normal distribution with the mean of national level seroprevalence (\({\theta }_{0}\)) and between-region variance (\({\sigma }_{b}^{2}\)), equation (3).
After fitting this hierarchical random effects model to the available data, seroprevalence values for regions with no data were imputed based on the resulting posterior predictive distributions. In other words, we represented missing seroprevalence data by distributions based on the fitted mean and variance parameters. For regions where no data were available, the (logit-transformed) seroprevalence was imputed as multiple random draws from a normal distribution with mean equal to the fitted national intercept \({\theta }_{0}\) and variance equal to the fitted between-region variance (thus imputing the seroprevalence as that of a “random” region, with the uncertainty interval describing the variability between regions). The equation was:
True seroprevalence estimation
All studies included in the analysis reported apparent seroprevalence instead of true seroprevalence. Therefore, publications about the Se and Sp for each commercial kit used in our selected studies were searched and reviewed. To take the uncertainty of Se and Sp into account, the information was then used by fitting beta distributed priors. The beta distribution is defined on the interval 0–1 and it is very flexible, therefore often used for model probabilities in Bayesian analyses. For those commercial kits which had more than one reported Se and Sp, the numbers of positive sample and tested sample obtained from validation studies were fitted to a random effect meta-analysis similar to the previous hierarchical meta-analysis model but without the multilevel structure. For those commercial kits which had no information at all, they were all categorized as unknown kit. To fit the random effect meta-analysis, data from all the known kits which were as the same type of assay as the unknown kits were used. The parameters of beta distribution were then found by using “fitdist” function in the R package “fitdistrplus” with the generated data from the random effect meta-analysis. As initial runs of the Bayesian hierarchical model showed insufficient convergence, beta distributions for the priors of Se and Sp were truncated (Table 3) assuming that the values of Se and Sp of used kits were at least larger than 0.1. The characteristics of the diagnostic tests (\({Se}\) and \({Sp}\)) were taken into account in equation (6), where \({{AP}}_{i}\) is the apparent seroprevalence found in the record and \(T{P}_{i}\) is the true seroprevalence. The equations for the Bayesian hierarchical model were:
Statistical analyses were performed using R version 3.3.140. The model was implemented in a Bayesian framework, using independent normal distributed (0, 100000) priors for all \({\theta }_{j}\) and \({\theta }_{0}\); a uniform distributed (0, 10) prior for \({\sigma }_{w}^{2}\); and a Folded-t (1) distributed prior for \({\sigma }_{b}^{2}\), as suggested by Gelman41. Models were constructed using “rjags” package and data were fitted using Markov Chain Monte Carlo sampling techniques. The models were implemented in R with the first 10,000 iterations as burn-in and 10,000 iterations as posterior inference. The convergence was checked by visual inspection of density and trace plots, as well as the multivariate potential scale reduction factors (or Brooks-Gelman-Rubin diagnostic). Approximate convergence was diagnosed if the upper confidence limit of the potential scale reduction factor was close to one.
The unit of analysis was set as a single study. Separate analysis was performed for pregnant women and each animal species. The estimates of posterior mean from different regions of China together with the 95% posterior probability intervals, defined as the distribution’s 2.5th and 97.5th percentile, were generated from the analysis. A Kruskal-Wallis test was used to compare the true seroprevalences in different species. If the Kruskal-Wallis test showed a significant result then a post-hoc analysis will be performed using Dunn’s test and p-values were adjusted by “Holm” method for multiple comparisons. The relationship between seroprevalence in pregnant women and meat-producing animals in different regions of China was estimated by checking Spearman’s rank correlation coefficient.
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Tenter, A. M., Heckeroth, A. R. & Weiss, L. M. Toxoplasma gondii: from animals to humans. Int. J. Parasitol. 30, 1217–1258 (2000).
Torgerson, P. R. & Mastroiacovo, P. The global burden of congenital toxoplasmosis: a systematic review. Bull. World Health Organ. 91, 501–508 (2013).
Torgerson, P. R. et al. World Health Organization estimates of the global and regional disease burden of 11 foodborne parasitic diseases, 2010: a data synthesis. PLoS Med. 12, e1001920 (2015).
FAO/WHO. Multicriteria-based ranking for risk management of food-borne parasites. http://www.fao.org/publications/card/en/c/ee07c6ae-b86c-4d5f-915c-94c93ded7d9e/ (2014).
Tenter, A. M. Toxoplasma gondii in animals used for human consumption. Mem. Inst. Oswaldo Cruz. 104, 364–369 (2009).
Cook, A. J. et al. Sources of toxoplasma infection in pregnant women: European multicentre case-control study. BMJ 321, 142–147 (2000).
Gilbert, R. E. et al. Effect of prenatal treatment on mother to child transmission of Toxoplasma gondii: retrospective cohort study of 554 mother-child pairs in Lyon, France. Int. J. Epidemiol. 30, 1303–1308 (2001).
Montoya, J. G. & Liesenfeld, O. Toxoplasmosis. Lancet 363, 1965–1976 (2004).
Opsteegh, M. et al. Relationship between seroprevalence in the main livestock species and presence of Toxoplasma gondii in meat (GP/EFSA/BIOHAZ/2013/01) An extensive literature review. Final report. EFSA supporting publication 2016:EN-996; https://www.efsa.europa.eu/en/supporting/pub/996e (2016).
Ji, Z., Xue, S., Ji, Y. & Zhi, B. A national survey on current status of the important parasitic diseases in human population. Chin. J. Parasitol. Parasit. Dis. 23, 332–340 (2005).
Liu, Q., Wang, Z. D., Huang, S. Y. & Zhu, X. Q. Diagnosis of toxoplasmosis and typing of Toxoplasma gondii. Parasit. Vectors 8, 292 (2015).
McAloon, C. G. et al. Bayesian estimation of prevalence of paratuberculosis in dairy herds enrolled in a voluntary Johne’s Disease Control Programme in Ireland. Prev. Vet. Med. 128, 95–100 (2016).
Beauvais, W., Orynbayev, M. & Guitian, J. Empirical Bayes estimation of farm prevalence adjusting for multistage sampling and uncertainty in test performance: a Brucella cross-sectional serostudy in southern Kazakhstan. Epidemiol. Infect. 9, 1–9 (2016).
McDonald, S. A. et al. Data-driven methods for imputing national-level incidence in global burden of disease studies. Bull. World Health Organ. 93, 228–236 (2015).
Hanson, T., Johnson, W. O. & Gardner, I. A. Hierarchical models for estimatingherd prevalence and test accuracy in the absence of a gold standard. J. Agric. Biol. Environ. Stat. 8, 223–239 (2003).
Branscum, A. J., Gardner, I. A. & Johnson, W. O. Bayesian modeling of animal- and herd-level prevalences. Prev. Vet. Med. 66, 101–112 (2004).
Devleesschauwer, B. et al. Methodological framework for World Health Organization estimates of the global burden of foodborne disease. PloS One 10, e0142498 (2015).
Torgerson, P. R. et al. The global burden of foodborne parasitic diseases: an update. Trends Parasitol. 30, 20–26 (2014).
Cenci-Goga, B. T., Rossitto, P. V., Sechi, P., McCrindle, C. M. & Cullor, J. S. Toxoplasma in animals, food, and humans: an old parasite of new concern. Foodborne Pathog. Dis. 8, 751–762 (2011).
Yang, G. et al. Rapid health transition in China, 1990–2010: findings from the Global Burden of Disease Study 2010. Lancet 381, 1987–2015 (2013).
Speybroeck, N., Devleesschauwer, B., Joseph, L. & Berkvens, D. Misclassification errors in prevalence estimation: Bayesian handling with care. Int. J. Public Health 58, 791–795 (2013).
Greiner, M. & Gardner, I. A. Epidemiologic issues in the validation of veterinary diagnostic tests. Prev. Vet. Med. 45, 3–22 (2000).
Berkvens, D., Speybroeck, N., Praet, N., Adel, A. & Lesaffre, E. Estimating disease prevalence in a Bayesian framework using probabilistic constraints. Epidemiology 17, 145–153 (2006).
Pappas, G., Roussos, N. & Falagas, M. E. Toxoplasmosis snapshots: global status of Toxoplasma gondii seroprevalence and implications for pregnancy and congenital toxoplasmosis. Int. J. Parasitol. 39, 1385–1394 (2009).
Jiang, C., Li, Z., Chen, P. & Chen, L. The seroprevalence of Toxoplasma gondii in Chinese population with cancer: A systematic review and meta-analysis. Medicine 94, e2274 (2015).
Ruiz, A. & Frenkel, J. K. Intermediate and transport hosts of Toxoplasma gondii in Costa Rica. Am. J. Trop. Med. Hyg. 29, 1161–1166 (1980).
Hu, Z. & Zhang, D. The pasture/forage resource profile for China. http://www.fao.org/ag/agp/agpc/doc/counprof/PDF%20files/China.pdf (2006).
van der Giessen, J., Fonville, M., Bouwknegt, M., Langelaar, M. & Vollema, A. Seroprevalence of Trichinella spiralis and Toxoplasma gondii in pigs from different housing systems in the Netherlands. Vet. Parasitol. 148, 371–374 (2007).
Maksimov, P. et al. Serological survey and risk factors for Toxoplasma gondii in domestic ducks and geese in Lower Saxony, Germany. Vet. Parasitol. 182, 140–149 (2011).
Guo, M. et al. A systematic meta-analysis of Toxoplasma gondii prevalence in food animals in the United States. Foodborne Pathog. Dis. 13, 109–118 (2016).
Deng, H. et al. Risk factors related to Toxoplasma gondii seroprevalence in indoor-housed Dutch dairy goats. Prev. Vet. Med. 124, 45–51 (2016).
Guo, M. et al. Prevalence and risk factors for Toxoplasma gondii infection in meat animals and meat products destined for human consumption. J. Food Prot. 78, 457–476 (2015).
Jones, J. L. et al. Risk factors for Toxoplasma gondii infection in the United States. Clin. Infect. Dis. 49, 878–884 (2009).
National Bureau of Statistics of China. Statistical yearbook s. http://www.stats.gov.cn/tjsj/ndsj/2016/indexeh.htm (2016).
Zhou, P. et al. Toxoplasma gondii infection in humans in China. Parasit. Vectors 4, 165 (2011).
Opsteegh, M., Prickaerts, S., Frankena, K. & Evers, E. G. A quantitative microbial risk assessment for meatborne Toxoplasma gondii infection in The Netherlands. Int. J. Food Microbiol. 150, 103–114 (2011).
EFSA. Application of systematic review methodology to food and feed safety assessments to support decision making. EFSA Journal 8, 1637 (2010).
Higgins, J. P. T., & Green S. Cochrane handbook for systematic reviews of interventions. www.handbook.cochrane.org (2011).
Moher, D. et al. Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015 statement. Syst. Rev. 4, 1 (2015).
R: A Language and Environment for Statistical Computing (R Foundation for Statistical Computing, Vienna, Austria, 2006).
Gelman, A. Prior distributions for variance parameters in hierarchical models. Bayesian Anal. 1, 515–533 (2006).
Calderaro, A. et al. Evaluation of Toxoplasma gondii immunoglobulin G (IgG) and IgM assays incorporating the newVidia analyzer system. Clin. Vaccine Immunol. 15, 1076–1079 (2008).
He, Y., Jiang, S., Ma, X. & Qiu, Q. Evaluation of Sixteen kinds of kits inner and abroad available for detecting antibodies of Toxoplasma gondii. Science of Travel Medicine 14, 43–45 (2008).
Trinity Biotech. Captia™ Toxoplasma gondii IgG. http://documents.trinitybiotech.com/product%20documents/2325100-29%20EN.pdf (2015).
Trinity Biotech. Captia™ Toxoplasma gondii IgM. http://documents.trinitybiotech.com/product%20documents/2325160-29%20EN.pdf (2015).
Duan, J. & Zhuang, S. Compare the results of Chinese and imported kits for detection of Toxoplasma gondii IgG antibodies. Chin. J. Zoonoses 19, 127 (2003).
Wang, W., Xu, S. & Xia, J. Evaluation of one ELISA kit for detection of Toxoplasma gondii IgG antibodies. Central China Medical Journal 31, 229–231 (2007).
Yu, E. & Gan, S. Re-evaluation of several test kits for detecting Toxoplasma gondii antibodies. Chin. J. Zoonoses 17, 5–6 (2001).
Jiang, S., Zhang, S., Pan, C., He, Y. & Wei, M. Evaluation of five commercial available kits for detecting antibodies of Toxoplasma gondii. Chin. J. Zoonoses 19, 97–99 (2003).
Yang, Y., Chen, Y., Wei, S. & Song, R. Efficiency of three methods for detecting Toxoplasma IgG antibody. Chin. J. Schisto. Control 26, 109–110 (2014).
Luo, C., You, J., Liu, J. & Huang, C. Comparison of ELISA and IHA for detecting serum antibody against Toxoplasma gondii in Swine. China Animal Health 14, 31–34 (2012).
Liu, S. et al. Comparison of several methods for detection of IgG against Toxoplasma gondii. Chinese J. Vet. Med. 48, 72–74 (2012).
Acknowledgements
This study was carried out as part of the China Exchange Project funded by the Royal Netherlands Academy of Arts and Sciences (530-5CDP21) and the Memorandum of Understanding programme funded by the Ministry of Health, Welfare and Sports in the Netherlands. The authors wish to acknowledge the help of Rob van Spronsen with literature searching and Peter Teunis for useful discussions in the preparation of this paper (both at National Institute for Public Health and the Environment).
Author information
Authors and Affiliations
Contributions
M.O., v.d.G.J., and H.D. conceived and designed the review, and critically revised the manuscript. H.D. and B.D. drafted the manuscript and performed the data analysis. M.L., J.L. and Y.W. allowed access to Chinese data and critically revised the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Deng, H., Devleesschauwer, B., Liu, M. et al. Seroprevalence of Toxoplasma gondii in pregnant women and livestock in the mainland of China: a systematic review and hierarchical meta-analysis. Sci Rep 8, 6218 (2018). https://doi.org/10.1038/s41598-018-24361-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-24361-8
This article is cited by
-
Genetic characterization of Toxoplasma gondii in meat-producing animals in Iran
Parasites & Vectors (2022)
-
A serological survey of Toxoplasma gondii in Polish pigs from organic farms, other housing systems and in pigs of different age groups
Acta Veterinaria Scandinavica (2022)
-
Prevalence of Toxoplasma gondii infection in chickens in China during 1993–2021: a systematic review and meta-analysis
Parasitology Research (2022)
-
One severe case of congenital toxoplasmosis in China with good response to azithromycin
BMC Infectious Diseases (2021)
-
Spatial distribution of Toxoplasma gondii in cows and associated risk factors
Tropical Animal Health and Production (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.