Abstract
In temperate climates, transmission of West Nile virus (WNV) is detectable rarely during the coldest months (late fall through early spring), yet the virus has reappeared consistently during the next warm season. Several mechanisms may contribute to WNV persistence through winter, including bird-to-bird transmission among highly viremic species. Here we consider whether, under realistic scenarios supported by field and laboratory evidence, a winter bird community could sustain WNV through the winter in the absence of mosquitoes. With this purpose we constructed a deterministic model for a community of susceptible birds consisting of communally roosting crows, raptors and other birds. We simulated WNV introduction and subsequent transmission dynamics during the winter under realistic initial conditions and model parameterizations, including plausible contact rates for roosting crows. Model results were used to determine whether the bird community could yield realistic outbreaks that would result in WNV infectious individuals at the end of the winter, which would set up the potential for onward horizontal transmission into summer. Our findings strongly suggest that winter crow roosts could allow for WNV persistence through the winter, and our model results provide synthesis to explain inconclusive results from field studies on WNV overwintering in crow roosts.
Similar content being viewed by others
Introduction
West Nile virus (WNV; family Flaviviridae, genus Flavivirus) was introduced into New York in 19991 and spread rapidly across the continent, reaching California in 20032. The virus is maintained in transmission cycles between ornithophilic mosquitoes in the genus Culex and various passerine birds, with tangential transmission occurring when infectious mosquitoes bite humans3,4,5,6,7,8,9,10. The virus has caused over 40,000 human disease cases in the U.S., and more than 1,900 deaths11, and these numbers are underestimates12.
The intensity of WNV transmission is strongly seasonal due in part to influences of temperature. Warmer weather accelerates transmission by reducing the time for mosquito development and increasing rates of mosquito biting and viral replication13,14,15,16,17,18. Colder temperatures limit the reproduction of Culex mosquitoes and, along with shortening daylength, can induce some species to enter reproductive diapause19,20,21,22,23. As a consequence, in geographic locations with temperate climates such as California, mosquito-mediated arbovirus transmission declines to barely detectable levels during the winter season13,24,25,26.
Therefore, in bird communities exposed to extended cold seasons, it would be expected that WNV could fade out in the absence of other transmission mechanisms; however, the virus consistently reappears during the next warm season. WNV overwintering mechanisms supported by field or laboratory findings include vertical transmission to mosquitoes in winter rest, continued vector-bird transmission through the winter at low rates, direct transmission between avian hosts (predation scavenging and others pathways such as fecal-oral transmission) or recrudescence of viremia in chronically infected birds19,20,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44. These mechanisms are not mutually exclusive and overwintering is almost certain to involve several of them. Of these possibilities, bird-to-bird transmission dynamics in winter avian communities remain poorly understood, and models are needed as a holistic framework to be reconciled with laboratory and field findings.
Within a winter bird community, American Crows (Corvus brachyrhynchos; hereafter “crows”) have the potential to play a significant role in WNV overwintering because this species is a highly competent host for WNV39,40,45,46,47,48,49,50,51,52. In winter, crows spend nights in communal roosts of thousands of birds flocking together29,53,54. Individuals living in large aggregations are likely to be in close proximity, therefore, crows are likely to have higher contact rates compared to other avian species55,56. Direct WNV transmission between crows has been demonstrated in the laboratory39,40, infected crows can shed the virus from the oral cavity39, and their feces can have high titers of WNV50. Field studies have reported that crows within a communal roost are frequently stained with feces of other crows and they exhibit preening behavior that could subject them to oral infection29, infected birds used the same roosts as healthy birds29 and WNV-positive dead crows have been recovered during cold periods when mosquitoes are not blood feeding27,29,57.
Consequently, in this study we used modeling to assess whether a realistic winter avian community consisting of crows, raptors and other birds can sustain WNV through the winter under plausible bird-to-bird transmission parameters in the absence of mosquitoes, while showing disease dynamics consistent with data from previous studies. We hypothesized that crow-to-crow transmission is the primary maintenance mechanism for WNV infection through the cold season, therefore, we initially identified the range of values for the crow-to-crow transmission parameter that yielded the largest fraction of realistic WNV outbreaks that resulted in viremic birds at the end of winter. We assessed the relevance of transmission among communally roosting crows and alternative WNV transmission pathways between birds (predation and scavenging), and finally, we evaluated the necessary conditions supporting realistic outbreaks and persistence of the virus through the winter across relevant parameter space. We discuss the plausibility of the selected crow-to-crow WNV transmission parameter values in nature, the coherence of model results with previous field studies, the relevance of other potential bird-to-bird transmission pathways for WNV overwintering, and the conditions supporting WNV overwintering in the bird community.
Methods
Study Population
Our study population consisted of the winter bird community of the University of California, Davis (UC Davis) (38.539975 N, 121.752187 W, Yolo County, California). The main campus has an area of ~7 km2 and it contains a well-documented crow roost with around 10,000 birds between November and March29. A previous study of this roost showed that crows are frequently stained with feces of other crows, crows exhibit preening behavior that could subject them to oral infection, WNV-infected and healthy crows are present in the roost, WNV-positive dead crows have been recovered during the cold season, and Culex mosquitoes are at very low abundance during this period27,57. The population of crows and other bird species are estimated yearly during a winter bird survey conducted during the third or fourth week of January by the UC Davis Museum of Wildlife and Fish Biology. The survey takes place in a single 24-hour period (midnight to midnight) in which over 40 walking observers systematically traverse all UC Davis campus. In this study, we used the mean counts from 2009–2014 annual censuses.
Dynamical model
We constructed a deterministic, continuous-time model of WNV transmission within the study population. Bird species were classified into 3 types: (1) crows, which are competent hosts for WNV, have high contact rates, and scavenge on raptors and other birds; (2) raptors, which are competent hosts for WNV and prey on other birds, including crows; and (3) other birds, which could be targets of scavenging or predation by crows or raptors, respectively, but could not become infected. Crows and raptors were divided into 7 compartments: susceptible (S; birds that are not infected with WNV), exposed (E; birds that have been infected with WNV but are not yet infectious), acutely viremic (I1; birds that are viremic and infectious if contact occurs with competent S birds via predation, scavenging, or fecal shedding), fecal shedders (I2; birds that survive WNV viremia; however, they remain infectious to S birds through fecal-oral transmission as they continue shedding virus through feces), chronically infected (I3; birds that survived WNV viremia and they stop shedding WNV through feces, but maintain WNV in their organs and can be infectious if preyed upon), recovered (R; birds that clear WNV infection completely so that they are not infectious and remain permanently immune to new WNV infection) and dead (D; crows that died while in any other compartment and raptors that died while in the S, E, and R compartments). Dead raptors had an extra compartment: infectious dead (DI; raptors that die while in the I1, I2 and I3 compartments and are infectious if S crows scavenge them). The group of other birds were divided in 2 compartments: S and D, as we assumed that in absence of mosquitoes, they cannot get infected with WNV through another mechanism. Births were not modeled because the late fall and winter study period did not overlap the breeding season58.
The system of ordinary differential equations was as follows; for crows:
for raptors:
and for other birds:
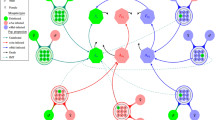
The subscripts C, R and O for the compartments refers to crows, raptors and other birds, respectively. A full summary of model parameters is shown in Table 1, while bird states, parameters, and interactions are summarized in Fig. 1.
Model summary. (a) Bird compartments and parameters determining compartment transitions. The green, light blue and orange boxes correspond to O, C and R compartments, respectively. Black arrows show movement of C, R and O among compartments, while red arrows depict routes of WNV transmission. (b) Interactions among crows in the roost. (c) Predation of raptors on crows and other birds. (d) Scavenging of crows on carcasses of raptors and other birds. In (b), (c) and (d) the red arrows shows interactions that may involve WNV transmission. Blue arrow shows interactions not involving WNV transmission. Credits: Crows roosting in 1b: Diego Montecino-Latorre; crow flying in 1c: Emilian Robert Vicol and Bob Comix (http://www.supercoloring.com/silhouettes/crows; published under a CCBY SA license); raptor flying in 1c: (https://www.vecteezy.com/vector-art/94660-free-eagle-silhouette-vector); other bird in bottom right of 1c: Russell Murphy (http://animalsclipart.com/small-bird-silhouette); other bird below the blue arrow in 1c: Matthew Townsend and Bob Comix (http://www.supercoloring.com/silhouettes/mockingbird; published under a CCBY SA license); other bird in the bottom left of 1c and dead other bird in 1d: Wanda Butler (http://animalsclipart.com/bird-silhouette); crow scavenging in 1d: https://openclipart.org/detail/259888/raven-silhouette-2); and dead raptor in 1d: Natalia Duque.
Susceptible crows (SC) became EC after an infectious contact with IC1 and IC239,40 at daily per capita transmission rate βCC, and/or after scavenging upon DIR at ɑCB*nC*pCB daily rate39,40,59,60,61, where ɑCB is the daily per capita rate of crows scavenging upon bird carrion, nC is the number of crows that scavenge upon a single raptor or other bird carcass, and pCB is the WNV transmission probability from WNV-infected bird carrion to scavenging SC. We parameterized predator-prey infectious disease transmission as recommended in past research62. The rate at which crows scavenged upon DIR was proportional to their availability with respect to other carcasses (DR and DO). We assumed that crows did not feed upon dead conspecifics because this behavior has not been observed in the study population. The EC became IC1 at rate εC39,46,50,51,63, and died due to WNV with a probability ρC at the end of the acutely viremic period. Most IC1 died following WNV viremia39,40,45,46,47,48,49,50,51,52; however, surviving individuals cleared the viremia at rate γC1 and moved to IC2. The IC2 remained infectious as they continued shedding WNV through feces39,50 and left this compartment at rate γC2 moving to one of 2 compartments: IC3 or RC with probabilities λC and 1-λC, respectively41,42,44, where λC is the probability of becoming chronically infected. The IC3 individuals retained WNV in their organs39,51,63, and could transmit the disease if fed upon by susceptible raptors (SR, see below), while RC cleared WNV infection and retained immunity for life42,64,65. Finally, IC3 cleared the chronic infection at rate γC3 and moved to RC41,42,44. All crows moved to DC according to the proportion of the expected mortality rate of crows, μC, not explained by raptor predation: μC- αRC, where αRC is the predation rate of raptors upon crows and equals μC* predRC. Here, predRC is the proportion of μC explained by raptor predation over crows. DC decomposed and were removed from the system at rate γC66,67.
The SR moved to ER when preying upon the IC1, IC2 and IC3 fractions of all live crows, NC39,59. This predation happened at αRC*pRC rate, where pRC is the WNV transmission probability from infected crows to raptors. We parameterized predator-prey infectious disease transmission as previously mentioned62. We assumed all raptors could prey on other birds because WNV-infected raptors continue to feed59. Individuals in the ER compartment became acutely viremic, IR1, at rate εR39,59. The WNV-induced mortality at the end of the viremia for the raptor species present in our study area, ρR, is lower than ρC39,59,60,68. Similar to crows, raptors surviving IR1 move to IR2, at rate γR139,59, while IR2 recovered from shedding at rate γR239,59 and moved into one of the two compartments, IR3 or RR, with probabilities λR and 1-λR, respectively41,42,44,59, where λR is the probability that raptors become chronically infected. Raptors in IR3 cleared the infection and moved into the RR compartment at rate γR341,42,44. All raptors died at rate μR, and those that died in non-infectious compartments SR, ER and RR moved to DR. Raptors that died in the IR1, IR2 and IR3 compartments became infectious after death (DIR) to scavenging SC. We assumed DIR remained infectious until decomposed or consumed39,69. Finally, DR and DIR rot and were removed from the system at rate τR66,67, and also by scavenging crows at rate ɑCB*nC. Individuals in the IC1 compartment did not scavenge due behavioral changes as result of WNV illness39.
Other birds remained uninfected (SO) and died at rate μO. Dead other birds that remained in the system, DO, arose at rate μO - ɑRO and included those that died from causes other than raptor predation. Here, αRO is the predation rate of raptors upon other birds and equals μO* predRO, where predRO is the proportion of μO explained by raptor predation over other birds. DO decomposed and were removed from the system at rate τO66,67, and also by crows scavenging upon them at rate ɑCB*nC.
Crow-to-crow transmission in the roost was modeled as density-dependent, implying that the transmission rate among crows increased linearly with the number of crows per unit area70. We also assumed that crow-to-crow transmission occurred exclusively when roosting, that the area used by the UC Davis bird community and the crow roost remained constant over the time period simulated1114, that the bird community is closed during winter after the initial introduction of WNV, and that the course of the disease in birds infected through bird-to-bird transmission follows that of mosquito-bird transmission39.
Simulations
We simulated the introduction of EC into a completely susceptible study population on November 1 (time 0), which represents the most permissive scenario for WNV transmission at that time of year. After introduction, we ran the model for 151 days ending March 31, which was considered late enough for mosquito-bird transmission cycles to take over as the primary mechanism of viral amplification into spring and summer and because crows stop roosting communally by this time29.
In order to account for uncertainty about model parameters, we constructed 300 parameter sets by Latin Hypercube Sampling (LHS) from the ranges defined in Table 1 (except βCC, see next paragraph) using the ‘lhs’ package71 in R72, with κ i ~Unif(κ imin , κ imax ) where κ is the vector of 26 parameters in the model. We used 300 parameter sets for our deterministic simulations following recommendations for LHS of 10 samples per parameter assessed73.
Finding the crow-to-crow daily WNV transmission rate (βCC) range causing the largest proportion of realistic WNV outbreaks
We used iterative sampling of the parameter space to determine the range for βCC that would result in a high probability of WNV persistence through the winter whilst remaining realistic. Specifically, we searched the βCC range for values that maximized the number of 300 simulations causing: a) at least 15 infected birds at the end of the winter, which we assumed to be a number large enough to avoid stochastic fadeout of WNV during onward bird-mosquito amplification into the warmer season, b) at least 67% of the original crow population living at the end of the winter, because it is not expected to lose more than one third of these birds after WNV initial introduction in a completely susceptible crow population74, c) less than 95% of the original crow population as this is the median crow population at the end of the winter when no WNV is introduced in our model, and d) less than 200 dead birds on any particular day of the study period because larger die-offs at the spatial scale of this study have not been observed and would have been unlikely to occur unnoticed. Therefore, we focused on the following three outcomes of interest (hereafter ‘OoI’): the number of crows at the end of winter, the number of infectious birds at the end of the winter, and the maximum number of dead birds at any given day during the simulation period.
We defined an initial βCC range of [2.14*10−9–2] infectious contacts per crow per day. The lower bound for this range was chosen because it matches the avian influenza transmission parameter estimated for waterfowl75, which we expected to be lower than the contact rate for gregarious crows. The upper bound of 2 was chosen because it is closer to the fecal-oral transmission rate for Campylobacter jejuni in chicken flocks (2.4, bacterial but fecal-oral transmission) previously estimated76. We regarded βCC >2 as unlikely to occur in free-ranging populations of roosting crows.
The initial βCC range was partitioned in ranges defined by minβCC, maxβCC and its quartiles q1βCC = [minβCC,…,Q1βCC], q2βCC = (Q1βCC, Q2βCC], q3βCC = (Q2βCC, Q3βCC] and q4βCC = (Q3βCC, maxβCC]. With each range we generated by LHS the 300 sets of unique parameter values. We ran the model with each parameter set, recording the proportion of simulations that were within our bounds for realism for the three OoI, which corresponded to ‘realistic simulations.’ We selected the qj βCC that yielded the greatest proportion of realistic simulations, and then divided this selected qj βCC again as previously explained. We continued this selection process, subdividing selected ranges and running sets of 300 simulations at each step until we found the values that caused the largest proportion of realistic simulations in a range.
Relevance of transmission among communally roosting crows and alternative WNV transmission pathways between birds such as predation and scavenging
Once the βCC range was found, we conducted a global sensitivity analysis as recommended77 with the purpose to find those significant parameters for which the OoI were sensitive. We used the results from 300 LHS draws to estimate the partial rank correlation coefficient (PRCC) of each parameter in the model with respect to each OoI. We tested the null hypothesis that there was no correlation between each parameter and the OoI. Monotonicity of the relationship between κ i and the number of crows, the number of infectious birds at the end of the winter, and the maximum number of dead birds at any given day was assessed graphically. In consequence, a separate set of parameters with significant PRCCs for at least one OoI was identified (denoted as ϴ; therefore, ϴ U ϴ’ = κ, where κ is the complete set of 26 parameters in the model).
Conditions supporting realistic outbreaks and infectious birds at the end of the winter across the parameter space
We explored the OoI across the space of selected βCC range and the parameters in ϴ, by partitioning their ranges as explained previously. Using LHS we obtained 100 unique values for βCC as well as for each parameter in ϴ from the corresponding qj βCC and qj ϴ k ranges, while the remaining significant parameters in ϴ, and the non-significant parameters in ϴ’ were assigned the mean value of their original range (Table 1). We ran the model with each set of parameters and calculated the proportion of simulations that fulfilled our thresholds for realism. From these results we evaluated the conditions across the parameter space supporting WNV overwintering in the avian community in the absence of vectors.
Data availability
Data generated and analyzed during the current study are available in the figshare repository78.
Results
Study Population
The bird community in this study initially consisted of 9,952 crows, 112 raptors, and 12,409 other birds.
Finding the crow-to-crow daily WNV transmission rate (βCC) range causing the largest proportion of realistic WNV outbreaks
The broadest a priori ranges of parameters resulted in no realistic outbreaks. For these simulations, a median of 13 (range: 0–54) infectious birds and 439 (range: 0–876) total living crows remained at the end of the winter, while the median peak daily number of dead birds was 2,921 (range: 1,970–3,985). These simulations resulted in very rapid spread of WNV through the crow population, causing extremely high mortality and exhausting most susceptibles before the study period was over. However, after 20 cycles of parameter selection (Supplementary Information 1), we identified a plausible range for βCC = (2.91*10–5, 3.05*10–5]. This range yielded medians of 24 and 7,753 for infectious birds and living crows at the end of winter, 49 as the median peak daily number of dead birds, and 35% of the 300 simulations met our criteria for realistic outbreaks (Table 2).
Simulated trajectories over the study period for SC (susceptible crows), the sum of IC1, IC2 and IC3 (infectious crows), RC, SR, the sum of of IR1, IR2 and IR3 (infectious raptors) and the sum of DC, DR, DIR and DO (dead birds) are shown in Fig. 2. The realistic simulations, in general, showed slow decline of susceptible crows and a smooth increase in the number of infectious crows over the study period, reaching a median number of 7,238 and 37 of these individuals, respectively, at the end of the winter. The median number of RC (immune crows) by March 31st in successful simulations was 60. The total number of infectious birds consisted primarily of crows. Consequently, the number of dead birds during the study period followed the number of infected crows closely. The median daily number of dead birds for successful simulations was 43. Furthermore, in these realistic simulations the average daily proportion of IC1 and IC2 (WNV fecal shedders), and IC3 (visceral WNV, chronically infected birds) in the roost was 0.005 and 0.0005, respectively. Conversely, simulations that turned out to be unrealistic generally led to early, rapid rises in infected crows and rapid depletion of susceptible crows. The dynamics of raptor infections differed little between realistic and unrealistic scenarios.
Time series for the number of susceptible, infectious, and recovered crows, susceptible and infectious raptors, and dead birds for each simulation based on random draws from the final selected ranges of βCC and other parameters. Lines represent individual simulations that were either realistic (colored) or unrealistic (gray) based on our defined criteria.
Relevance of transmission among communally roosting crows and alternative WNV transmission pathways between birds such as predation and scavenging
The global sensitivity analysis showed that the three OoI were sensitive to (1) the daily per capita WNV crow-to-crow transmission rate (βCC), (2) the daily per capita rate at which IC1 clear WNV viremia (γC1), and (3) the probability that IC1 die due to WNV infection at the end of the acute viremic period (ρC). Moreover, the initial number of WNV-exposed crows introduced at the start of the winter (EC) affected the number of living crows at the end of the study period and the maximum number of dead birds. Finally, the number of infectious birds at the end of the winter was sensitive to the daily per capita rate at which EC become acutely viremic (εC), whilst the number of living crows at the end of the study period was sensitive to the daily per capita mortality rate of crows (μC), and the peak daily number of dead birds was sensitive to the daily per capita rate of DC elimination through decomposition (τC). Therefore, the set of parameters to which our OoI were sensitive were defined as ϴ = {βCC, EC, εC, γC1, ρC, τC, μC}. The corresponding estimates of PRCC and the 95% CI are shown in Table 3.
Conditions supporting realistic outbreaks and infectious birds at the end of the winter across the parameter space
The proportion of realistic simulations varied little across the ranges of βCC and the ranges of most of the significant parameters, ϴ (Fig. 3). The proportion of realistic simulations was maximized at >75% for the middle quartiles of γC1, corresponding to a moderate infectious period in acutely viremic crows that was neither too long nor too short. When the recovery rate of acutely viremic crows was moderately low (q2), outbreaks most outbreaks were realistic across values of βCC, but for high recovery rates (q4), the opposite was true, yielding no realistic outbreaks (Fig. 3).
Discussion
We constructed a dynamical model to simulate a bird community consisting of crows, raptors and other birds in order to evaluate whether WNV could persist through the winter under plausible bird-to-bird transmission parameters while causing realistic outbreaks in the absence of mosquito-borne transmission. If WNV is introduced into a completely susceptible bird community at the beginning of winter, our results demonstrate that where they exist, large crow roosts are expected to dominate the dynamics of WNV during the cold season and that plausibly low transmission rates within the roost could enable WNV persistence through the winter. Viremic crows at the end of winter could then initiate horizontal bird-mosquito transmission in the spring when the weather warms and mosquitoes become active.
Our simulations introduce WNV into a completely susceptible communal crow roost at the start of winter. This is a reasonable initial condition because the immune fraction is expected to be low due to the birth pulse of new susceptible crows each spring and death of most infected crows before becoming immune. Because of fecal WNV shedding in highly viremic crows, fecal-oral transmission due to fecal stain and preening behavior is likely the primary crow-to-crow transmission pathway29,50, and for this reason we included WNV viremic and non-viremic fecal shedder crows (IC1 and IC2),
To our knowledge, estimates of bird-to-bird per-capita transmission rate have not been published for WNV in free-ranging birds, but our estimated range for the crow-to-crow daily transmission rate βCC = (2.91*10–5, 3.05*10–5], which implies 1 WNV transmission event every ~3 days in the initial roost, is within the range of previously published estimates of daily transmission rates for infectious diseases in captive birds and non-roosting wild birds. For example, our range for βCC was lower than the estimated daily per capita transmission rate for WNV in experimental caged crows79, for avian influenza in high-density enclosures of poultry80,81,82 and for primarily fecal-oral transmitted bacterial pathogens, such as Campylobacter C. jejuni, C. coli Salmonella enterica serovar Enteritidis, and Salmonella sp. in poultry as well76,83,84,85,86. In the case of non-communally roosting free-ranging aquatic birds, the estimated daily per capita transmission rate for avian influenza was lower than our selected βCC range75 which would be expected.
The number of total infectious birds in the community was heavily driven by crows, and all of the parameters to which our OoI were sensitive to were related to infection dynamics in crows. The duration of fecal shedding following the acute viremia and the duration of the chronic infection had little effect, probably due to the reduced number of crows reaching that stage, as most infected crows die after the acute viremic period39,40,45,46,47,48,49,50,51,52. The trajectory of crows in realistic simulations is consistent with results from previous studies. For example, the rarity of seropositive crows is attributable in part to high mortality rates following infection, and one serosurvey did not find seropositive crows during the cold season (February and March 2002) after the initial introduction of WNV in 200148. In our successful simulations the average daily fraction of RC (immune crows) was 0.003. Under this average seroprevalence, sampling zero seropositive crows if collecting blood from 1 to 152 individuals (the last number is the total N reported by the authors of the serosurvey) has a probability between 0.63 to 1. Other published studies have also found low percentages of seropositive crows during the cold season57,87.
Moreover, a previous field study of the same roost that we simulated collected 909 WNV-negative fecal samples without finding a WNV-positive sample during the cold season29. The authors reported that the probability of such a result, assuming WNV prevalence of 0.023 and independence of observations over time, was <1 * 10−7. In our realistic simulations the average daily proportion of crows shedding WNV in feces (IC1 and IC2) at the roost was 0.005. If we apply this shedding prevalence and assume that: (1) crows defecate once daily at the roost, (2) 88 fecal samples are collected in a single day (the reported median number of samples collected by month during the field study), and (3) samples collected in a single day are independent, the binomial probability of all samples negative is 0.64, which is more consistent with the previous field results29. Further, the authors of this same field study also reported 12 WNV positive crow carcasses out of the 32 that were collected under the roost (prevalence of 0.375). In our model, we did not track infectious crow carcasses, however, simulations without any WNV-infected bird introduced caused an average daily number of 7.49 dead crows, while realistic simulations under the selected βCC for WNV transmission yielded 43.03 average daily dead crows. This means that on average ~83% of crow carcasses present in a single day are positive to WNV during winter, which results in a probability >0.999 to find at least one WNV-positive carcass if 10 dead crows are collected in a single day. Moreover, if the 32 carcasses were collected in one day, the probability of finding at least 12 positives is also >0.999. Another study in winter crow roosts was also more successful in finding WNV-infected specimens when testing crow carcasses compared to feces57. Finally, because only a small fraction of total dead birds are observed88, the average number of dead birds, mainly crows, per day in successful simulations is consistent with observations during WNV outbreaks in initially naive bird communities89,90,91.
Other bird-to-bird transmission pathways, including scavenging of crows upon other birds and predation by raptors, were not relevant for WNV dynamics or the persistence of the virus in the bird community during the winter. These phenomena may be due to the relatively small number of raptors within the avian community studied, which limited the overall rate of contact between raptors and crows. These results are consistent with previous model-based findings that transmission between crows via close contact could have a considerable impact on WNV establishment when the density of ornithophilic mosquitoes is low and that during such periods, scavenging and the effects of other birds in the community are not relevant for determining the WNV basic reproductive number (R0)79. Furthermore, current data show that many infected raptors do not develop WNV infection in the organs or shed the virus through their feces39,59. This would further diminish the importance of this group of birds for WNV dynamics.
The realism of outbreaks was remarkably dependent on the value of the infectious period of acutely viremic crows. Infectious periods that were too short (3.0–3.3 days) or too long (4.3–5.0 days) resulted in very few or not realistic outbreak trajectories over the winter, whereas moderate acute infectious periods of 3.3–4.3 days were much more realistic, specially in the range 3.7–4.3 days.
Future modelling work should consider the effects of stochasticity and different population sizes of the crows, raptors, and other birds within the community. Our model assumes that crow density declines in proportion to the total number of crows in the roost, but future studies should consider whether crow mortality also results in changes to aggregation and roost structure that may affect density. Models should also account for the fact that crows have variable home range sizes92 and that infected crows may roost with susceptible crows less frequently during illness associated with acute viremia93. Potential effects of temperature on crow behavior and resulting differences in contact rates should also be considered. Evaluating these additional aspects will determine the generalizability of our results to other epidemiological settings and also to study the role of crows in WNV spread and maintenance across space and time.
Conclusions
Our results strongly support plausible scenarios by which birds can sustain WNV through the winter via bird-to-bird transmission, specifically due to a limited but persistent number of new WNV crow-to-crow transmission events. In nature, these transmission events would be driven mainly by WNV fecal shedding by infectious crows, fecal staining of susceptible individuals, and posterior preening. The values for the daily per capita WNV crow-to-crow transmission rate are sufficiently small to be plausible when density-dependent transmission is assumed. Moreover, these values are between estimates for pathogen transmission parameter in poultry and in non-roosting wild birds. The realistic simulations generate consistently low numbers of infectious and recovered crows over the study period and a higher number of dead crows in the system compared to periods when WNV is absent. These characteristics are consistent with a high WNV detection probability in crow carcasses found under roosts during winter, a low probability of WNV detection in feces during the same period, and low seroprevalence in the population. Our findings add to previous research on the importance of crow roosts for WNV overwintering and WNV dynamics in crows and improve our understanding on how WNV persists in temperate climates when cold temperatures preclude viral replication and diminish vector blood-feeding activity.
References
Lanciotti, R. S. et al. Origin of the West Nile virus responsible for an outbreak of encephalitis in the northeastern United States. Science 286, 2333–2337 (1999).
Reisen, W. et al. West Nile virus in California. Emerg. Infect. Dis. 10, 1369–1378 (2004).
Molaei, G., Andreadis, T. G., Armstrong, P. M., Anderson, J. F. & Vossbrinck, C. R. Host feeding patterns of Culex mosquitoes and West Nile virus transmission, northeastern United States. Emerg. Infect. Dis. 12, 468–474 (2006).
Campbell, G. L., Marfin, A. A., Lanciotti, R. S. & Gubler, D. J. West Nile virus. Lancet Infect. Dis. 2, 519–529 (2002).
Marra, P. P., Griffing, S. M. & McLean, R. G. West Nile virus and wildlife health. Emerg. Infect. Dis. 9, 898–899 (2003).
Kilpatrick, A. M. et al. West Nile virus risk assessment and the bridge vector paradigm. Emerg. Infect. Dis. 11, 425–429 (2005).
Kilpatrick, A. M., Daszak, P., Jones, M. J., Marra, P. P. & Kramer, L. D. Host heterogeneity dominates West Nile virus transmission. Proc. Biol. Sci. 273, 2327–2333 (2006).
Molaei, G. et al. Vector-host interactions governing epidemiology of West Nile virus in Southern California. Am. J. Trop. Med. Hyg. 83, 1269–1282 (2010).
Hamer, G. L. et al. Culex pipiens (Diptera: Culicidae): a bridge vector of West Nile virus to humans. J. Med. Entomol. 45, 125–128 (2008).
Simpson, J. E. et al. Vector host-feeding preferences drive transmission of multi-host pathogens: West Nile virus as a model system. Proc. Biol. Sci. 279, 925–933 (2012).
Center For Disease Control and Prevention. West Nile virus. West Nile virus - Center for Disease Control and Prevention Available at: https://www.cdc.gov/westnile/index.html. (Accessed: 13th August 2017) (2015).
Petersen, L. R. & Hayes, E. B. Westward ho?—The spread of West Nile virus. N. Engl. J. Med. 351, 2257–2259 (2004).
Reisen, W. K., Fang, Y. & Martinez, V. M. Effects of temperature on the transmission of West Nile virus by Culex tarsalis (Diptera: Culicidae). J. Med. Entomol. 43, 309–317 (2006).
Hartley, D. M. et al. Effects of temperature on emergence and seasonality of West Nile virus in California. Am. J. Trop. Med. Hyg. 86, 884–894 (2012).
Ruiz, M. O. et al. Local impact of temperature and precipitation on West Nile virus infection in Culex species mosquitoes in northeast Illinois, USA. Parasit. Vectors 3, 19 (2010).
Kilpatrick, A. M., Meola, M. A., Moudy, R. M. & Kramer, L. D. Temperature, viral genetics, and the transmission of West Nile virus by Culex pipiens mosquitoes. PLoS Pathog. 4, e1000092 (2008).
Ciota, A. T., Matacchiero, A. C., Kilpatrick, A. M. & Kramer, L. D. The effect of temperature on life history traits of Culex mosquitoes. J. Med. Entomol. 51, 55–62 (2014).
Reisen, W. K. Effect of temperature on Culex tarsalis (Diptera: Culicidae) from the Coachella and San Joaquin Valleys of California. J. Med. Entomol. 32, 636–645 (1995).
Nelms, B. M., Macedo, P. A., Kothera, L., Savage, H. M. & Reisen, W. K. Overwintering biology of Culex (Diptera: Culicidae) mosquitoes in the Sacramento Valley of California. J. Med. Entomol. 50, 773–790 (2013).
Nelms, B. M. et al. Phenotypic variation among Culex pipiens complex (Diptera: Culicidae) populations from the Sacramento Valley, California: Horizontal and vertical transmission of West Nile virus, diapause potential, autogeny, and host selection. Am. J. Trop. Med. Hyg. 89, 1168–1178 (2013).
Spielman, A. & Wong, J. Environmental control of ovarian diapause in Culex pipiens. Ann. Entomol. Soc. Am. 66, 905–907 (1973).
Denlinger, D. L. & Armbruster, P. A. Mosquito diapause. Annu. Rev. Entomol. 59, 73–93 (2014).
Eldridge, B. F. The effect of temperature and photoperiod on blood-feeding and ovarian development in mosquitoes of the Culex pipiens complex. Am. J. Trop. Med. Hyg. 17, 133–140 (1968).
Bellamy, R. E. & Reeves, W. C. The winter biology of Culex tarsalis (Diptera: Culicidae) in Kern County, California. Ann. Entomol. Soc. Am. 56, 314–323 (1963).
Reeves, W. C. & Others. Overwintering of arboviruses. Epidemiology and control of mosquito-borne arboviruses in California 1943–1987, 357–382 (1990).
Dohm, D. J., Sardelis, M. R. & Turell, M. J. Experimental vertical transmission of West Nile virus by Culex pipiens (Diptera: Culicidae). J. Med. Entomol. 39, 640–644 (2002).
Reisen, W. K. et al. Overwintering of West Nile virus in Southern California. J. Med. Entomol. 43, 344–355 (2006).
Nasci, R. S. et al. West Nile virus in overwintering Culex mosquitoes, New York City, 2000. Emerg. Infect. Dis. 7, 742–744 (2001).
Hinton, M. G., Reisen, W. K., Wheeler, S. S. & Townsend, A. K. West Nile virus activity in a winter roost of American crows (Corvus brachyrhynchos): is bird-to-bird transmission important in persistence and amplification? J. Med. Entomol. 52, 683–692 (2015).
Goddard, L. B., Roth, A. E., Reisen, W. K. & Scott, T. W. Vertical transmission of West Nile virus by three California Culex (Diptera: Culicidae) species. J. Med. Entomol. 40, 743–746 (2003).
Fechter-Leggett, E., Nelms, B. M., Barker, C. M. & Reisen, W. K. West Nile virus cluster analysis and vertical transmission in Culex pipiens complex mosquitoes in Sacramento and Yolo Counties, California, 2011. J. Vector Ecol. 37, 442–449 (2012).
Anderson, J. F. & Main, A. J. Importance of vertical and horizontal transmission of West Nile virus by Culex pipiens in the Northeastern United States. J. Infect. Dis. 194, 1577–1579 (2006).
Nelms, B. M. et al. Experimental and natural vertical transmission of West Nile virus by California Culex (Diptera: Culicidae) mosquitoes. J. Med. Entomol. 50, 371–378 (2013).
Reisen, W. K. et al. Chronic infections of West Nile virus detected in California dead birds. Vector Borne Zoonotic Dis. 13, 401–405 (2013).
Garmendia, A. E. & Van Kruiningen, H. J. Recovery and identification of West Nile virus from a hawk in winter. Journal of Clinical (2000).
Banet-Noach, C., Simanov, L. & Malkinson, M. Direct (non-vector) transmission of West Nile virus in geese. Avian Pathol. 32, 489–494 (2003).
Austin, R. J., Whiting, T. L., Anderson, R. A. & Drebot, M. A. An outbreak of West Nile virus-associated disease in domestic geese (Anser anser domesticus) upon initial introduction to a geographic region, with evidence of bird to bird transmission. Can. Vet. J. 45, 117–123 (2004).
Ip, H. S. et al. West nile virus transmission in winter: the 2013 Great Salt Lake bald eagle and eared grebes mortality event. PLoS Curr. 6 (2014).
Komar, N. et al. Experimental infection of North American birds with the New York 1999 strain of West Nile virus. Emerg. Infect. Dis. 9, 311–322 (2003).
McLean, R. G. et al. West Nile virus transmission and ecology in birds. Ann. N. Y. Acad. Sci. 951, 54–57 (2001).
Nemeth, N. et al. Persistent West Nile virus infection in the house sparrow (Passer domesticus). Arch. Virol. 154, 783–789 (2009).
Wheeler, S. S., Vineyard, M. P., Woods, L. W. & Reisen, W. K. Dynamics of West Nile virus persistence in House Sparrows (Passer domesticus). PLoS Negl. Trop. Dis. 6, e1860 (2012).
Miller, B. R. et al. First field evidence for natural vertical transmission of West Nile virus in Culex univittatus complex mosquitoes from Rift Valley Province, Kenya. Am. J. Trop. Med. Hyg. 62, 240–246 (2000).
Wheeler, S. S. et al. Detection of persistent West Nile virus RNA in experimentally and naturally infected avian hosts. Am. J. Trop. Med. Hyg. 87, 559–564 (2012).
McLean, R. G. West Nile Virus: Impact on crow populations in the United States. In 21st Vertebrate Pest Conference (eds. Timm, R. M. & Gorenzel, W. P.) 180–184 (University of California-Davis, 2004).
Brault, A. C. et al. Differential virulence of West Nile strains for American crows. Emerg. Infect. Dis. 10, 2161–2168 (2004).
Reed, L. M. et al. Declining mortality in American crow (Corvus brachyrhynchos) following natural West Nile virus infection. Avian Dis. 53, 458–461 (2009).
Yaremych, S. A. et al. West Nile virus and high death rate in American crows. Emerg. Infect. Dis. 10, 709–711 (2004).
Taylor, R. M., Work, T. H., Hurlbut, H. S. & Rizk, F. A study of the ecology of West Nile virus in Egypt. Am. J. Trop. Med. Hyg. 5, 579–620 (1956).
Kipp, A. M. et al. West Nile virus quantification in feces of experimentally infected American and fish crows. Am. J. Trop. Med. Hyg. 75, 688–690 (2006).
Nemeth, N. M. et al. Clinical and pathologic responses of American crows (Corvus brachyrhynchos) and fish crows (C. ossifragus) to experimental West Nile virus infection. Vet. Pathol. 48, 1061–1074 (2011).
Caffrey, C., Weston, T. J. & Shauna, C. R. Smith. High mortality among marked crows subsequent to the arrival of West Nile virus. Wildl. Soc. Bull. 31, 870–872 (2003).
Caccamise, D. F. & Reed, L. M. Jerzy Romanowski & Stouffer, P. C. Roosting behavior and group territoriality in American crows. Auk 114, 628–637 (1997).
Gorenzel, W. P. & Salmon, T. P. Characteristics of American crow urban roosts in California. J. Wildl. Manage. 59, 638–645 (1995).
Altizer, S. et al. Social organization and parasite risk in mammals: integrating theory and empirical studies. Annu. Rev. Ecol. Evol. Syst. 34, 517–547 (2003).
Cross, P. C. et al. Wildlife population structure and parasite transmission: implications for disease management. In Management of Disease in Wild Mammals 9–29 (Springer Japan, 2009).
Dawson, J. R. et al. Crow deaths caused by West Nile virus during winter. Emerg. Infect. Dis. 13, 1912–1914 (2007).
Cornell Laboratory of Ornithology. The Birds of North America Online. The birds of North America online (2015).
Nemeth, N., Gould, D., Bowen, R. & Komar, N. Natural and experimental West Nile virus infection in five raptor species. J. Wildl. Dis. 42, 1–13 (2006).
Nemeth, N. M. et al. Clinical evaluation and outcomes of naturally acquired West Nile virus infection in raptors. J. Zoo Wildl. Med. 40, 51–63 (2009).
Madge, S. Crows and jays. (A&C Black, 2010).
Han, L., Ma, Z. & Hethcote, H. W. Four predator prey models with infectious diseases. Math. Comput. Model. 34, 849–858 (2001).
Weingartl, H. M., Neufeld, J. L., Copps, J. & Marszal, P. Experimental West Nile virus infection in blue jays (Cyanocitta cristata) and crows (Corvus brachyrhynchos). Vet. Pathol. 41, 362–370 (2004).
Nemeth, N. M., Oesterle, P. T. & Bowen, R. A. Humoral immunity to West Nile virus is long-lasting and protective in the house sparrow (Passer domesticus). Am. J. Trop. Med. Hyg. 80, 864–869 (2009).
Wilcox, B. R., Yabsley, M. J., Ellis, A. E., Stallknecht, D. E. & Gibbs, S. E. J. West Nile virus antibody prevalence in American crows (Corvus brachyrhynchos) and fish crows (Corvus ossifragus) in Georgia, USA. Avian Dis. 51, 125–128 (2007).
Ward, M. R., Stallknecht, D. E., Willis, J., Conroy, M. J. & Davidson, W. R. Wild bird mortality and West Nile virus surveillance: biases associated with detection, reporting, and carcass persistence. J. Wildl. Dis. 42, 92–106 (2006).
Osborn, R. G., Higgins, K. F., Usgaard, R. E., Dieter, C. D. & Neiger, R. D. Bird mortality asssociated with wind turbines at the Buffalo Ridge Wind Resource Area, Minnesota. Am. Midl. Nat. 143, 41–52 (2000).
Gancz, A. Y. et al. West Nile virus outbreak in North American owls, Ontario, 2002. Emerg. Infect. Dis. 10, 2135–2142 (2004).
Panella, N. A. et al. Rapid West Nile virus antigen detection. Emerg. Infect. Dis. 11, 1633–1635 (2005).
Begon, M. et al. A clarification of transmission terms in host-microparasite models: numbers, densities and areas. Epidemiol. Infect. 129, 147–153 (2002).
Carnell, R., Carnell, M. R. & R Unit, S. Package ‘lhs’. (2016).
R Core Team. R: A language and environment for statistical computing. Vienna, Austria; 2017. http://www.R-project.org (2017).
Matala, A. Sample size requirement for Monte Carlo simulations using Latin hypercube sampling. (Department of Engineering Physics and Mathematics, Systems AnalysisLaboratory, Helsinki University of Technology, 2008).
LaDeau, S. L., Kilpatrick, A. M. & Marra, P. P. West Nile virus emergence and large-scale declines of North American bird populations. Nature 447, 710–713 (2007).
Vaidya, N. K. & Wahl, L. M. Avian influenza dynamics under periodic environmental conditions. SIAM J. Appl. Math. 75, 443–467 (2015).
van Gerwe, T. et al. Quantifying transmission of Campylobacter jejuni in commercial broiler flocks. Appl. Environ. Microbiol. 75, 625–628 (2009).
Marino, S., Hogue, I. B., Ray, C. J. & Kirschner, D. E. A methodology for performing global uncertainty and sensitivity analysis in systems biology. J. Theor. Biol. 254, 178–196 (2008).
Montecino-Latorre, D. F. & Barker, C. M. Model results.: Overwintering of West Nile virus in a bird community with a communal crow roost. https://doi.org/10.6084/m9.figshare.6099572.v1
Hartemink, N. A., Davis, S. A., Reiter, P., Hubálek, Z. & Heesterbeek, J. A. P. Importance of bird-to-bird transmission for the establishment of West Nile virus. Vector Borne Zoonotic Dis. 7, 575–584 (2007).
Gonzales, J. L., van der Goot, J. A., Stegeman, J. A., Elbers, A. R. W. & Koch, G. Transmission between chickens of an H7N1 Low Pathogenic avian influenza virus isolated during the epidemic of 1999 in Italy. Vet. Microbiol. 152, 187–190 (2011).
van der Goot, J. A., de Jong, M. C. M., Koch, G. & Van Boven, M. Comparison of the transmission characteristics of low and high pathogenicity avian influenza A virus (H5N2). Epidemiol. Infect. 131, 1003–1013 (2003).
Saenz, R. A. et al. Quantifying transmission of highly pathogenic and low pathogenicity H7N1 avian influenza in turkeys. PLoS One 7, e45059 (2012).
Conlan, A. J. K. et al. Transmission and dose–response experiments for social animals: a reappraisal of the colonization biology of Campylobacter jejuni in chickens. J. R. Soc. Interface rsif20110125 (2011).
Van Gerwe, T. J. W. M. et al. Quantifying transmission of Campylobacter spp. among broilers. Appl. Environ. Microbiol. 71, 5765–5770 (2005).
Thomas, M. E. et al. Quantification of horizontal transmission of Salmonella enterica serovar Enteritidis bacteria in pair-housed groups of laying hens. Appl. Environ. Microbiol. 75, 6361–6366 (2009).
Heres, L., Urlings, H. A. P., Wagenaar, J. A. & de Jong, M. C. M. Transmission of Salmonella between broiler chickens fed with fermented liquid feed. Epidemiol. Infect. 132, 107–116 (2004).
Ringia, A. M. et al. Antibody prevalence of West Nile virus in birds, Illinois, 2002. Emerg. Infect. Dis. 10, 1120–1124 (2004).
Caffrey, C., Smith, S. C. R. & Weston, T. J. West Nile virus devastates an America crow population. Condor 107, 128–132 (2005).
Mostashari, F., Kulldorff, M., Hartman, J. J., Miller, J. R. & Kulasekera, V. Dead bird clusters as an early warning system for West Nile virus activity. Emerg. Infect. Dis. 9, 641–646 (2003).
Eidson, M. et al. Dead bird surveillance as an early warning system for West Nile virus. Emerg. Infect. Dis. 7, 631–635 (2001).
Kulasekera, V. L. et al. West Nile virus infection in mosquitoes, birds, horses, and humans, Staten Island, New York, 2000. Emerg. Infect. Dis. 7, 722–725 (2001).
Yaremych, S. A., Novak, R. J., Raim, A. J., Mankin, P. C. & Warner, R. E. Home range and habitat use by American crows in relation to transmission of West Nile virus. Wilson Bull. 116, 232–239 (2004).
Ward, M. P., Raim, A., Yaremych-Hamer, S., Lampman, R. & Novak, R. J. Does the roosting behavior of birds affect transmission dynamics of West Nile virus? Am. J. Trop. Med. Hyg. 75, 350–355 (2006).
Sherrod, S. K. Diets of North American falconiformes. Raptor Res 12, 49–121 (1978).
Campbell, R. W., Manuwal, D. A. & Harestad, A. S. Food habits of the common barn-owl in British Columbia. Can. J. Zool. 65, 578–586 (1987).
Otteni, L. C., Bolen, E. G. & Cottam, C. Predator-prey relationships and reproduction of the barn owl in Southern Texas. Wilson Bull. 84, 434–448 (1972).
Glasgow, L. L. The barn owl. Bull. Am. Assoc. Hist. Nurs. (1962).
Marti, C. D. A long-term study of food-niche dynamics in the common barn-owl: comparisons within and between populations. Can. J. Zool. 66, 1803–1812 (1988).
Cromrich, L. A., Holt, D. W. & Leasure, S. M. Trophic niche of North American great horned owls. J. Raptor Res. 36, 58–65 (2002).
Verbeek, N. A. & Caffrey, C. American crow (Corvus brachyrhynchos). The Birds of North America Available at: https://birdsna.org/Species-Account/bna/species/amecro/demography#lifespan (Accessed: November 10, 2015) (2002).
Henny, C. J. An Analysis of the Population Dynamics of Selected Avian Species with Special References to Changes During the Modern Pesticide Era. (United States Department of the Interior, 1972).
Keran, D. The incidence of man-caused and natural mortalities to raptors. Raptor Research 15, 108–112 (1981).
Rohner, C. The numerical response of great horned owls to the snowshoe hare cycle: consequences of non-territorial floaters’ on demography. J. Anim. Ecol. 359–370 (1996).
Bildstein, K. L., MacWhirter, R. B., Poole, A. & Gill, F. Northern harrier (Circus cyaneus). Birds North America Available at: https://birdsna.org/Species-Account/bna/species/norhar/introduction. (Accessed: 21st March 2016) (1996).
Koenig, W. D. & Stacey, P. B. Acorn woodpeckers: group-living and food storage under contrasting ecological conditions. Cooperative breeding in birds. Cambridge University Press, Cambridge, UK 415–453 (1990).
DeSante, D. F., Kaschube, D. R. & Saracco, J. F. Vital rates of North American landbirds. The Institute for Bird Populations. Available at: www.VitalRatesOfNorthAmericanLandbirds.org. (Accessed: 3rd May 2016) (2015).
Stewart, P. A. Annual survival rate of yellow-rumped warblers. North American Bird Bander 13, 106 (1988).
Schroeder, D. A. Breeding biology and population limitation in the black phoebe (Sayornis nigricans) in Southern coastal California. (University of California, Santa Barbara, 1985).
Searcy, W. A. & Yasukawa, K. Sexual size dimorphism and survival of male and female blackbirds (Icteridae). Auk 98, 457–465 (1981).
Scott, D. M. & Ankney, C. D. Fecundity of the brown-headed cowbird in Southern Ontario. Auk 97, 677–683 (1980).
Raitt, R. J. & Genelly, R. E. Dynamics of a population of California quail. J. Wildl. Manage. 28, 127–141 (1964).
Sandercock, B. K. & Jaramillo, A. Annual survival rates of wintering sparrows: assessing demographic consequences of migration. Auk 119, 149–165 (2002).
Ricklefs, R. E., Tsunekage, T. & Shea, R. E. Annual adult survival in several new world passerine birds based on age ratios in museum collections - Springer. J. Ornithol. 152, (481–495 (2011).
Badyaev, A. V., Hill, G. E., Stoehr, A. M., Nolan, P. M. & McGraw, K. J. The evolution of sexual size dimorphism in the house finch. II. Population divergence in relation to local selection. Evolution 54, 2134–2144 (2000).
Badyaev, A. V. & Martin, T. E. Sexual dimorphism in relation to current selection in the house finch. Evolution 54, 987–997 (2000).
Badyaev, A. V., Young, R. L., Oh, K. P. & Addison, C. Evolution on a local scale: developmental, functional, and genetic bases of divergence in bill form and associated changes in song structure between adjacent habitats. Evolution 62, 1951–1964 (2008).
Summers-Smith, J. D. The Sparrows: a study of the genus. Passer. T & AD Poyser, Staffordshier, England (1988).
McGillivray, W. B. & Murphy, E. C. Sexual differences in longevity of house sparrows at Calgary, Alberta. Wilson Bull. 96, 456–458 (1984).
Clark, M. E. & Martin, T. E. Modeling tradeoffs in avian life history traits and consequences for population growth. Ecol. Modell. 209, 110–120 (2007).
Dyer, M. I., Pinowski, J. & Pinowska, B. 3. Population dynamics. Granivorous birds in ecosystems: their evolution, populations, energetics, adaptations, impact and control 12, 53 (2012).
Wheelwright, N. T., Schultz, C. B. & Hodum, P. J. Polygyny and male parental care in Savannah sparrows: effects on female fitness. Behav. Ecol. Sociobiol. 31, 279–289 (1992).
Johnston, R. F. Population structure in salt marsh song sparrows: Part I. Environment and annual cycle. Condor 58, 24–44 (1956).
Sogge, M. K. & Van Riper, C. Breeding biology and population dynamics of the San Miguel Island Song Sparrow (Melospiza melodia micronyx). (Cooperative National Park Resources Studies Unit, 1988).
Linsdale, J. M. Survival in birds banded at the hastings reservation. Condor 51, 88–96 (1949).
Gardali, T. & Nur, N. Site-specific survival of black-headed grosbeaks and spotted towhees at four sites within the Sacramento Valley, California. Wilson J. Ornithol. 118, 178–186 (2006).
Keyser, A. J., Keyser, M. T. & Promislow, D. E. L. Life-history variation and demography in western bluebirds (Sialia mexicana) in Oregon. Auk 121, 118–133 (2004).
Flux, J. E. C. & Flux, M. M. Population dynamics and age structure of starlings (Sturnus Vulgaris) in New Zealand. N. Z. J. Ecol. 4, 65–72 (1981).
Arnold, T. W. & Clark, R. G. Survival and philopatry of female dabbling ducks in Southcentral Saskatchewan. J. Wildl. Manage. 60, 560–568 (1996).
Smith, G. W. & Reynolds, R. E. Hunting and mallard survival, 1979–88. J. Wildl. Manage. 56, 306–316 (1992).
Otis, D. L., Schulz, J. H. & Scott, D. P. Mourning dove (Zenaida macroura) harvest and population parameters derived from a national banding study. US Department of Interior, Fish and Wildlife Service, Biological Technical Publication FWS. (BTP-R3010-2008, Washington, DC, USA, 2008).
Wiebe, K. L. A review of adult survival rates in woodpeckers. Ann. Zool. Fennici 43, 112–117 (2006).
Michel, N., DeSante, D. F., Kaschube, D. R. & Nott, M. P. The monitoring avian productivity and survivorship (MAPS) program annual reports, 1989–2003. NBII/MAPS Avian Demographics Query Interface (2006).
Murton, R. K., Thearle, R. J. P. & Thompson, J. Ecological studies of the feral pigeon Columba livia var. I. Population, breeding biology and methods of control. J. Appl. Ecol. 9, 835–874 (1972).
Atwood, J. L., Elpers, M. J. & Collins, C. T. Survival of breeders in Santa Cruz island and mainland California Scrub Jay populations. Condor 92, 783–788 (1990).
Caffrey, C. Feeding rates and individual contributions to feeding at nests in cooperatively breeding Western American crows. Auk 116, 836–841 (1999).
Acknowledgements
We thank Andrew Engilis, Jr., Curator of the Museum of Wildlife and Fish Biology at the University of California - Davis for supporting the data on winter bird censuses that are conducted in this campus. This work was funded by a grant to CMB from the Center for Equine Health at the School of Veterinary Medicine, University of California, Davis.
Author information
Authors and Affiliations
Contributions
D.M.L. conceived and constructed the model, generated and analyzed the data, and drafted the manuscript; C.M.B. conceived the study, constructed the model, supported data analysis and helped draft the manuscript. Both authors gave final approval for publication.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Montecino-Latorre, D., Barker, C.M. Overwintering of West Nile virus in a bird community with a communal crow roost. Sci Rep 8, 6088 (2018). https://doi.org/10.1038/s41598-018-24133-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-24133-4
This article is cited by
-
The wintering ecology of the Rook Corvus frugilegus in Northern Kyushu, Japan
Urban Ecosystems (2022)
-
West Nile virus transmission and human infection risk in Veneto (Italy): a modelling analysis
Scientific Reports (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.