Abstract
Here we show that a commercial blocking reagent (G2) based on modified eukaryotic DNA significantly improved DNA extraction efficiency. We subjected G2 to an inter-laboratory testing, where DNA was extracted from the same clay subsoil using the same batch of kits. The inter-laboratory extraction campaign revealed large variation among the participating laboratories, but the reagent increased the number of PCR-amplified16S rRNA genes recovered from biomass naturally present in the soils by one log unit. An extensive sequencing approach demonstrated that the blocking reagent was free of contaminating DNA, and may therefore also be used in metagenomics studies that require direct sequencing.
Similar content being viewed by others
Introduction
Modern microbial ecology studies are often based on extraction of community nucleic acids, followed by molecular analyses of the recovered DNA or RNA. The extreme complexity and variation of the chemical and physical properties of some sample materials such as soils makes it impossible or at least very challenging to standardize nucleic acid extraction procedures. Consequently, numerous different in-house and commercial protocols have been developed and made available for this purpose. In a review, Orgiazzi et al.1 described the need for a standard across laboratories, and discussed the current ISO standards within this context. Because the ISO standard2 is inefficient for extraction of fungal DNA3, Orgiazzi et al.1 recommended a modified protocol; however, this suggestion revealed that the idea of standardization faces obvious problems. Based on these observations we conclude that no single protocol works optimally for all soils and suggest a new modified method targeted for low organic high clay soil.
Clay particles can tightly adsorb organic as well as inorganic phosphorous compounds4. Because of the phosphate rich backbone of nucleic acids, these molecules tend to stick to clay sorption sites. Thus, due to their large surface area free clay particles are particularly problematic when extracting the nucleic acids. Essentially, nucleic acids released from lysed cells can be immobilized on particulate adsorption sites before the extraction procedure is completed5. Since adsorption is rarely taken into consideration during protocol development, this potential confounder may have a significant influence on the efficiency of DNA extractions from complex matrices, such as soil.
Adsorption of nucleic acids in soil is primarily associated with the inorganic fraction, and especially with minerals such as montmorillonite and kaolinite, which are present at high levels in clay4,5,6,7. Adsorption in soil increases if the organic fraction is removed, which suggests that the organic fraction has little adsorption potential towards nucleic acids; actually, components of the organic fraction might coat potential nucleic acid binding sites, thereby preventing nucleic acids from binding8,9. DNA molecules bound to clay minerals are protected against nucleases and can persist in the environment for a long period of time6. Therefore, with regards to extraction of nucleic acids from soil, mineral adsorption introduces two sources of biases: (1) loss of target nucleic acid released upon lysis of microbial cells due to the adsorption, and (2) co-extraction of remaining “ancient” nucleic acids bound in the clay fraction10. In protocols for direct DNA and RNA extraction from environmental samples, one parameter of success has been extraction yield, which could be problematic if the aim is to study viable present day organisms. To minimize such contamination by soil-bound nucleic acid, one solution would be to extract and recover cells from soil prior to lysis and DNA extraction although other biases like differential cell lysis may be introduced11,12.
Additions of blocking reagents based on skim milk powder, tRNA, or DNA improve DNA extraction efficiency from clay-rich soils9,13,14,15. However, addition of foreign reagents based on biological materials very likely introduces contaminating nucleic acid residues, which may obscure or bias downstream molecular analyses of target nucleic acids. Previously, we developed a blocking reagent (G2) based on short fragments of modified double-stranded DNA, ensuring minimum co-extraction of DNA residues and preventing the added DNA residues from serving as templates for PCR, hybridization, or sequencing4. Used in connection with a custom phenol/chloroform-based protocol, the G2 blocking reagent can increase extraction of DNA and mRNA from clay rich groundwater sediments more than 10,000-fold16.
The aim of this study was to test the blocking reagent in combination with commercially available DNA extraction kits used for soil in a multiple-lab ring experiment. As the G2 compound is derived from short nucleic acids, we tested whether it contained any contaminating DNA by direct deep sequencing. To test the performance of the blocking reagent in combination with the commonly used MOBIOTM PowerLyzer PowerSoil DNA extraction kit, we performed an inter-laboratory comparison test among 11 different laboratories. All groups received three samples of the same clay sediment and extracted DNA using the same kit, with or without inclusion of the blocking reagent.
Materials and Methods
Sampling of soil samples
The clay subsoil used for the multi-laboratory comparison of the extraction protocol was collected at depths between 110 and 130 cm in January 2012 in Kolding, Jutland, Denmark, using handheld drill equipment (Eijkelkamp, Giesbeek, The Netherlands). The intact clay subsoil was kept at 4 °C for 2 months, at which time six replicated 0.25 g portions was weighed out in 2 ml Eppendorf tubes and immediately freeze-dried prior to shipment at ambient temperature to the participating laboratories. The high clay inorganic soil was not homogenized prior to dividing in 0.25 g portions.
DNA extraction protocol
DNA was extracted from the 0.25 g soil samples by participating laboratories using the protocol provided with the PowerLyzer PowerSoil DNA extraction kit (MOBIO, Carlsbad, CA, USA). From each sample, three replicate samples were subjected to bead-beating using regular 0.1 mm glass beads, and three other replicates from the same sample were subjected to bead-beating using the same 0.1 mm glass beads but pre-coated with lyophilized G2 DNA/RNA enhancer (Ampliqon A/S, Odense, Denmark). The G2 is released from the glass-beads upon mixture with the lysing buffer from the DNA extraction kit and become fully sorbed to the clay within one minute (data not shown).
Quantitative PCR protocol
The quantitative PCR (q-PCR) reactions were performed in one lab on all samples. Standards with known numbers of 16 S rRNA genes were produced by extraction of DNA from 100 μl of 10-fold dilutions of E. coli cells washed in 0.015 M phosphate buffer (pH 7.4) using the MOBIO kit. Quantitative PCR reactions were performed in triplicate on all DNA samples using the following setup: 10 µl SsoFast™ EvaGreen® Supermix (BIO-RAD, Hercules, CA, USA), 3.4 µl PCR-grade water (MOBIO, Carlsbad, CA, USA), 400 nM (final concentration) of each primer (341 F: CCTACGGGAGGCAGCAG and 518 R: ATTACCGCGGCTGCTGG)17, and 5 µl of 10X diluted template DNA, all in a 20 µl volume. All qPCR preparations were performed on the epMotion 5070 pipetting robot (Eppendorf, Hamburg, Germany) in a high-pressure clean room. qPCR was performed on the CFX96 Touch™ Real-Time PCR Detection System (BIO-RAD) under the following conditions: initial denaturation at 95 °C for 2 minutes; 50 cycles of denaturation at 95 °C for 30 seconds, annealing at 60 °C for 30 seconds, elongation at 72 °C for 45 seconds, and (to prevent quantification of possible primer-dimers) fluorescence measurement at 82 °C for 10 seconds; followed by a final elongation step at 72 °C for 6 minutes.
DNA loss during extraction protocol and quality control of G2 beads
Escherichia coli was grown to near late log phase, and 100,000 dpm 3H-thymidine (Sigma-Aldrich, Copenhagen, Denmark) was added in late log phase, while the culture was allowed to continue growing and incorporating the 3H-thymidine (the late addition maximizes the amount of 3H that is incorporated into DNA). Twenty-five µl of the culture was added to 250 mg of soil and immediately three replicate DNA extractions were performed using the regular PowerLyzer PowerSoil kit (MOBIO, Carlsbad, CA, USA), and three using G2 modified bead tubes (Ampliqon, Odense, Denmark) but otherwise following the exact same protocol. One aliquot of the 3H labeled culture and one 10% (vol/vol) aliquot of the DNA extraction were withdrawn at four steps in the protocol (MOBIO): (1) after step 7 (lysis); (2) after step 11 (first inhibitor removal precipitation); (3) after step 14 (second inhibitor removal precipitation); and, finally, (4) after step 23 (final step). The 3H signal in the samples was determined by liquid scientilation by combining 0.1 ml of the supernatant with 4 ml of scintillation fluid (Wallac Scintillation Products, Turku, Finland) followed by a 10 mins counting in a liquid scintillation counter (Wallac 1409). Subsequent quality control of G2 beads was performed by following the protocol described above and measuring radioactivity after step 7.
HiSeq sequencing of potential DNA contamination from G2
Cupriavidus pinatubonensis JMP134 was inoculated in Luria–Bertania broth (Alpha BioScience, Baltimore, MD, USA) and incubated with shaking for 24 hours at 28 °C. DNA was extracted from the 2 ml culture both with and without the addition of the G2 compound using the PowerLyzer PowerSoil DNA Isolation Kit (MOBIO). A single-end Illumina HiSeq sequencing library was prepared using a modified version of the NEBNext ® DNA library Prep Master Mix Set kit (New England BioLabs, MA, USA). Briefly, 20 µl DNA was mixed in a PCR tube with 2.4 µl NEBNext 10X Repair Reaction Buffer and 1.25 µl NEBNext End Repair Enzyme which was incubated for 30 minutes at 30 °C. The reaction was purified on a MinElute column (Qiagen, Hilden, Germany) and eluted in 18 µl EB buffer at 37 °C for 15 minutes. In a new PCR tube, the following were added to 17 µl purified DNA: 5 µl Quick Ligation 5X buffer, 0.5 µl of 25 µM stock DNA adaptors for Illumina HiSeq, and 2.5 µl Quick T4 DNA ligase. The reaction was incubated for 15 minutes at 20 °C, then purified on a MinElute column and eluted in 22 µl EB buffer at 37 °C for 15 minutes. In a new PCR tube, the 22 µl of purified DNA was mixed with 2.5 µl NEBNext Adapter Fill-in Reaction Buffer and 1.5 µl Bst DNA polymerase. The reaction was incubated for 20 minutes at 65 °C and subsequently heat inactivated at 80 °C for 20 minutes. The resulting DNA library was sequenced on the Illumina HiSeq platform.
Applying the BWA MEM algorithm with default settings18, reads were mapped to the genome of Salmo salar (RefSeq accession# GCF_000233375.1), as well as to the genome of Cupriavidus pinatubonensis JMP134 (RefSeq accession# GCF_000203875.1). Mapping results were explored and the best hit (in case a read mapped to both reference genomes) was determined using Samtools view.
Statistical analyses
We log-transformed data prior to analysis and then used a mixed ANOVA-model in SAS Enterprise Guide (Ver. 6.1), with addition of G2 as a fixed effect and laboratory as a random effect.
Importance
Nucleic acid extractions from low-biomass samples are often problematic because (1) the samples contain low levels of DNA and RNA in the first place, and (2) the sample matrix may contain high levels of surfaces with the capacity to adsorp DNA or RNA released from microorganisms following lysis. The DNA/RNA enhancing substance “G2” significantly increased DNA extraction from a low biomass clay subsoil in a multi-laboratory trial and can hence circumvent this difficulty.
Disclosure
Jacob Bælum and Carsten Suhr Jacobsen are inventors of G2, and according to Danish law the patent was issue by their former employer, The Geological Survey of Denmark and Greenland (GEUS). GEUS later sold the patent to Ampliqon A/S (Odense, Denmark), which produces G2 today. The inventors (JB and CSJ) receive a percentage of the net sale of G2.
Results and Discussion
Quantitative-PCR on DNA from inter-laboratory test
Extraction of soil DNA of sufficient quality for applications in molecular biology was pioneered by the Tiedje laboratory11, and the protocol by Griffiths et al.19 has over the years been the leading protocol for co-extracting DNA and RNA from soil samples. The Griffith protocol, however, has serious drawbacks when extracting DNA and RNA from mineral soils that are high in clay and low in organic material; e.g. subsurface soils or groundwater sediment, likely because the clay particles are highly adsorptive9. Addition of various nucleic acids or proteins (e.g., salmon sperm DNA, skim milk powder, or tRNA) can overcome problems related to DNA/RNA-adsorbing surfaces9,14,15,20.
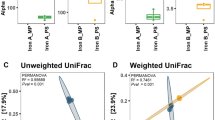
The inter-laboratory test of the effect of G2 on recovery of DNA from the same high-clay soil using qPCR revealed that G2 consistently increased the 16 S rRNA gene copy number when used in connection with the DNA extraction protocol. Yield increased on an average 7.5 fold and16S rRNA gene copy numbers were significantly (F = 48.8; P < 0.0001) higher in G2-supplemented DNA extractions.
The result from this inter-laboratory comparison (Fig. 1) also revealed large differences between the participating laboratories, especially in terms of overall yield of DNA. Today, many molecular microbial ecology laboratories use commercial kits for DNA and RNA extractions for environmental matrices due to their ease of use and expected reproducibility. However, we found that DNA yields differed enormously across participating laboratories, even when they were using the same kit. Using exactly the same soil and exactly the same standard kit (no G2 addition), one laboratory were able to extract around 107 copies of 16 S rRNA genes per gram of soil, whereas others laboratories only obtained fewer than 106 copies. Two main reasons for this high discrepancy can be mentioned – one is that clearly some variation is expected in biomass between the triplicate soil samples, but also some difference in individual skills working with the kit exist, i.e. some laboratories have never tried the kit before and others are using it as a standard DNA extraction method. However, the reason is beyond the scope of this paper, but certainly merits further attention.
DNA contamination from G2
Addition of a eukaryotic DNA or RNA into a DNA-RNA extraction protocol is only feasible if the additional nucleic acid does not interfere with the purpose of the study, e.g., if the PCR target is a specific bacterial gene, like a respiratory gene16, a herbicide degradation gene21, or 16 S rRNA genes9. In all of these cases, salmon sperm DNA did not lead to false positive results because gene-specific qPCR analysis targeting prokaryotic DNA was used. In theory the addition of DNA or RNA as blocking reagents could be eliminated, if the nucleic acid extraction protocol was gene-specific, using a complementary DNA strand bound to a magnetic particle as hybridization probe fishing out the gene of interest at high stringency conditions22.
Metagenomic and metatranscriptomic studies are increasingly becoming part of the standard tool box for many studies within soil microbial ecology. In this context, contaminating DNA is unacceptable. Analysis of 82 and 53 million HiSeq reads of C. pinatubonensis JMP134 DNA with or without addition of G2 showed in both cases that the same amount of reads had the best match to the standard genome. The reads not mapping to C. pinatubonensis had similar mapping ratios obtained in the presence and absence of G2, which indicates that no traces of the G2 origin DNA were present in the final DNA extract. Thus, our in-depth sequencing showed no traces of DNA left in the G2 compound. Hence, to our knowledge, this is the only commercially available blocking reagent that can be used to increase yield from low-biomass samples without introducing contaminating DNA.
Loss of radioactivity during DNA extraction protocol
Batch-tests of G2 beads using thymidine-labeled E. coli added to the subsurface clay soil showed that about one-third of the radioactivity was lost during the initial steps of the lysis process (MOBIO kit protocol step 7) in the presence of G2, whereas two-thirds of the radioactivity was lost if no G2 was added to the soil (Fig. 2).
Radioactivity measured at different steps in the MOBIO PowerLyzer PowerSoil DNA extraction procedure. Radioactivity was measured after step 7: supernatant of bead-beated soil, 11: supernatant after removing non-DNA organic and inorganic material including humic substances, cell debris and proteins, 14: supernatant after second removal of non-DNA organic and inorganic material, and 23: final DNA elution.
Moreover, the protocol steps 11–14 decreased the recovered radioactivity to about half of what was recovered at step 7; finally, at the end of the protocol (step 23), the amount of recovered radioactivity did not differ significantly from the amount recovered at step 14. Furthermore, the ratio between radioactivity recovered with and without addition of G2 remained constant throughout the extraction protocol, i.e., about twice as much radioactivity was recovered in the presence of G2, suggesting that also protein yield is increased by the presence of G2. For soil nucleic acid analyses, the increased presence of such proteins would not be a problem due to the nucleic acid purification procedures applied.
References
Orgiazzi, A., Dunbar, M. B., Panagos, P., de Groot, G. & Lemanceau, P. Soil biodiversity and DNA barcodes: opportunities and challenges. Soil Biol. Biochem. 80, 244–250 (2015).
Petric, I. et al. Interlaboratory evaluation of the ISO standard 11063 “Soil quality e method to directly extract DNA from soil samples”. J. Microbiol. Meth. 84, 454–460 (2011).
Terrat, S. et al. Meta-barcoded evaluation of the ISO standard 11063 DNA extraction procedure to characterize soil bacterial and fungal community diversity and composition. Microb. Biotech. 8, 131–142 (2015).
Cai, P. et al. Effects of lowmolecular- weight organic ligands and phosphate on DNA adsorption by soil colloids and minerals. Colloids and Surfaces B. Biointerfaces 54, 53–59 (2007).
Bælum, J. & Jacobsen, C. S. Improvement of low biomass soil DNA/RNA extraction yield and quality (WO/2010/146026) (2010).
Romanowski, G., Lorenz, M. G. & Wackernagel, W. Adsorption of plasmid DNA to mineral surfaces and protection against DNase I. Appl. Environ. Microbiol. 57, 1057–1061 (1991).
Pietramellara, G., Franchi, M., Gallori, E. & Nannipieri, P. Effect of molecular characteristics of DNA on its adsorption and binding on homoionic montmorillonite and kaolinite. Biol. Fert. Soils 33, 402–409 (2001).
Cai, P., Huang, Q., Jiang, D., Rong, X. & Liang, W. Microcalorimetric studies on the adsorption of DNA by soil colloidal particles. Coll. Surfa. B: Biointerfaces 49, 49–54 (2006).
Pauline, M. M. et al. Improving Griffith’s protocol for co-extraction of microbial DNA and RNA in adsorptive soils. Soil Biol.Biochem. 63, 37–49 (2013).
Chamier, B., Lorenz, M. G. & Wackernagel, W. Natural transformation of Acinetobacter calcoaceticus by plasmid DNA adsorbed on sand and groundwater aquifer material. Appl. Environ. Microbiol. 59, 1662–1667 (1993).
Holben, W. E., Jansson, J. K., Chelm, B. K. & Tiedje, J. M. DNA probe method for the detection of specific microorganisms in the soil bacterial community. Appl. Environ. Microbiol. 54, 703–711 (1988).
Holmsgaard, P. E. et al. Bias in bacterial diversity as a result of Nycondenz extraction from bulk soil. Soil Biol. Biochem. 43, 2152–2159 (2011).
Frostegård, A. et al. Quantification of bias related to the extraction of DNA directly from soils. Appl. Environ. Microbiol. 65, 5409–5420 (1999).
Takada-Hoshino, Y. & Matsumoto, N. An improved DNA extraction method using skim milk from soils that strongly adsorbDNA. Microb. Environ. 19, 13–19 (2004).
Hoshino, Y. T. & Matsumoto, N. Skim milk drastically improves the efficacy of DNA extraction from andisol, a volcanic ash soil. Japan Agri. Reas. Q. 39, 247–252 (2005).
Bælum, J. et al. A conceptual model linking functional gene expression and reductive dechlorination rates of chlorinated ethenes in clay rich groundwater sediment. Water Res. 47, 2467–2478 (2013).
Muyzer, G., de Wall, E. C. & Uitterlinden, A. G. Profiling of complex microbial populations by denaturing gradient gel electrophoresis analysis of polymerase chain reaction-amplified genes coding for 16S rRNA. Appl Environ. Microbiol. 59, 695–700 (1993).
Li, H. & Durbin, R. Fast and accurate long-read alignment with Burrows-Wheeler Transform. Bioinformatics, Epub. [PMID: 20080505] (2010).
Griffiths, R. I., Whiteley, A. S., O’Donnell, A. G. & Bailey, M. J. Rapid method for coextraction of DNA and RNA from natural environments for analysis of ribosomal DNA- and rRNA-based microbial community composition. Appl. Environ. Microbiol. 66, 5488–5491 (2000).
Ikda, S. et al. Evaluation of the effects of different additives in improving the DNA extraction yield and quality from andosol. Microb. & Environ. 23, 159–166 (2008).
Nicolaisen, M. H., Bælum, J., Jacobsen, C. S. & Sørensen, J. Transcription dynamics of the functional tfdA gene during MCPA herbicide degradation by Cupriavidus necator AEO106 (pRO101) in agricultural soil. Environ. Microbiol. 10, 571–579 (2008).
Jacobsen, C. S. Microscale detection of specific bacterial DNA in soil with a magnetic capture-hybridization and PCR amplification assay. Appl. Environ. Microbiol. 61, 3347–3352.
Acknowledgements
This work was supported by the Danish National Research Foundation (CENPERM DNRF100). We would like to acknowledge The Danish National High-Throughput DNA Sequencing Center for sequencing the samples and laboratory technician Karin Vestberg for assistance with making Illumina HiSeq libraries.
Author information
Authors and Affiliations
Contributions
The experiments were planned by Carsten Suhr Jacobsen, Jacob Bælum and Tue Kjærgaard Nielsen. The preparation of the ringtest DNA extraction was performed by Carsten Suhr Jacobsen and Pia Bach Jakobsen. The ringtest DNA extraction was done by Jan Kjølhede Vester, Peter Stougaard, Jeppe Lund Nielsen, Jana Voriskova, Anne Winding, Petr Baldrian, Binbin Liu, Åsa Frostegård, Dorthe Pedersen, Alexander Tøsdak Tveit, Mette Marianne Svenning, Christoph C. Tebbe, Lise Øvreås, Steven J. Blazewicz, Valerie Hubablek, Stefan Bertilsson, S. Craig Cary, Tue Kjærgaard Nielsen and Pia Bach Jakobsen. All other laboratory experiments were carried out by Tue Kjærgaard Nielsen and Pia Bach Jakobsen. Statistical analysis was carried out by Flemming Ekelund. Bioinformatic analysis was carried out by Tue Kjærgaard Nielsen and Jacob Bælum. The paper was drafted by Carsten Suhr Jacobsen and all authors have read and commented on the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Jacobsen, C.S., Nielsen, T.K., Vester, J.K. et al. Inter-laboratory testing of the effect of DNA blocking reagent G2 on DNA extraction from low-biomass clay samples. Sci Rep 8, 5711 (2018). https://doi.org/10.1038/s41598-018-24082-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-24082-y
This article is cited by
-
Determining an effective sampling method for eDNA metabarcoding: a case study for fish biodiversity monitoring in a small, natural river
Limnology (2021)
-
High throughput quantification of the functional genes associated with RDX biodegradation using the SmartChip real-time PCR system
Applied Microbiology and Biotechnology (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.