Abstract
E-cadherin (CDH1) is a putative tumor suppressor gene implicated in breast carcinogenesis. Yet, whether risk factors or survival differ by E-cadherin tumor expression is unclear. We evaluated E-cadherin tumor immunohistochemistry expression using tissue microarrays of 5,933 female invasive breast cancers from 12 studies from the Breast Cancer Consortium. H-scores were calculated and case-case odds ratios (OR) and 95% confidence intervals (CIs) were estimated using logistic regression. Survival analyses were performed using Cox regression models. All analyses were stratified by estrogen receptor (ER) status and histologic subtype. E-cadherin low cases (N = 1191, 20%) were more frequently of lobular histology, low grade, >2 cm, and HER2-negative. Loss of E-cadherin expression (score < 100) was associated with menopausal hormone use among ER-positive tumors (ever compared to never users, OR = 1.24, 95% CI = 0.97–1.59), which was stronger when we evaluated complete loss of E-cadherin (i.e. H-score = 0), OR = 1.57, 95% CI = 1.06–2.33. Breast cancer specific mortality was unrelated to E-cadherin expression in multivariable models. E-cadherin low expression is associated with lobular histology, tumor characteristics and menopausal hormone use, with no evidence of an association with breast cancer specific survival. These data support loss of E-cadherin expression as an important marker of tumor subtypes.
Similar content being viewed by others
Introduction
The E-cadherin protein (encoded by the CDH1 gene) is normally expressed in breast epithelial tissue and functions as a critical part of epithelial cell adhesion and epithelial-to-mesenchymal transition (EMT)1,2,3. Due to the frequent loss or inactivation of E-cadherin that is evident in epithelial cell cancers, E-cadherin is thought to have tumor-suppressor properties where loss is associated with carcinogenesis and invasion4,5.
Loss of E-cadherin expression is commonly used to confirm lobular histology that comprise 10–15% of all breast cancers6,7,8, which have also been noted to more frequently express hormone receptors [estrogen receptor (ER) and progesterone receptor (PR)] than E-cadherin high tumors9. Recent molecular profiling analysis of lobular compared with ductal cancers show E-cadherin mutation and loss to be a defining feature of lobular breast cancers, and suggest it to be a distinct molecular subtype of breast cancers10. Although many studies have identified heterogeneity in risk factor associations based on breast tumor subtypes defined by hormone receptor status (e.g. ER-positive vs. ER-negative) or histology (lobular vs. ductal)11,12,13,14,15,16,17,18,19,20,21,22, limited data have examined whether E-cadherin may define important subgroups of tumors with distinct etiologies23.
Data also suggest that loss of E-cadherin expression may be associated with malignant progression, metastasis, and reduced survival in breast cancer patients24,25,26,27,28; however, most of these studies were based on small numbers and not all analyses were stratified by ER-status, a known important prognostic and predictive marker.
Evidence on whether E-cadherin may be an important marker of etiologic heterogeneity and survival differences across the spectrum of breast tumor subtypes including ER status and histology is limited. In 1984, Prentice et al.29 introduced the concept of using case-case studies for identifying disease risk factors. In a case-case study design, information is obtained only from cases of a particular disease–in our study, breast cancer–and is used as a tool to assess etiologic heterogeneity. In this study, we were interested in determining whether known risk/protective factors for breast cancer differed by E-cadherin expression, in order to provide new insights into possible mechanisms for E-cadherin loss in breast carcinogenesis similar to analyses previously done for ER, PR and HER2 markers23,30,31. Using immunohistochemical (IHC) data for E-cadherin performed centrally using tumor tissue microarrays (TMAs), we performed a large pooled analysis of 12 studies participating in the Breast Cancer Association Consortium (BCAC), and examined whether established breast cancer risk factor associations and survival differed by low vs. high E-cadherin tumor tissue expression, stratified by ER status and histology.
Material and Methods
Study Population
Descriptions of the 12 breast cancer studies participating in the Breast Cancer Association Consortium (BCAC) included in this analysis are detailed in Supplemental Table 1. Case-case analyses were restricted to 5,933 European women from 12 breast cancer studies with invasive breast cancer who provided data on age at diagnosis and had evaluable E-cadherin tumor tissue staining results (See section on E-cadherin tumour tissue measurements). Study participants were recruited under protocols approved by the Institutional Review Board at each institution, and all subjects provided informed consent or did not opt-out, depending on national regulations. All methods were performed in accordance with the relevant guidelines and regulations and a list of ethical approval committees are listed at the end of this manuscript.
Risk factor information
The 12 participating studies provided information on one or more of the following risk factors for breast cancer: family history of breast cancer in first-degree relatives, reproductive factors including age at menarche, parity, age at first full-term birth, oral contraceptive (OC) use among women ≤50 years of age, menopausal hormonal use, type of ever menopausal hormone used, and anthropometric measures including body mass index (BMI), and height. As a proxy for menopausal status, we used age ≤50 and >50 as a proxy for pre- and postmenopausal status respectively, since not all studies captured menopausal status.
E-cadherin tumor tissue measurements
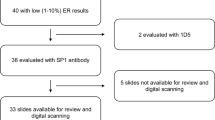
Routinely prepared formalin-fixed paraffin-embedded (FFPE) blocks of invasive breast tumors were used to construct TMA blocks at each study center. One-hundred and forty-two TMA slides with tumor samples from 6,010 individual patients were prepared for E-cadherin staining (ranging from 1–2 cores per patient). TMA’s from all participating studies were stained centrally in the Experimental Pathology Laboratory at the National Cancer Institute (NCI) to allow for consistency across sites and avoid any potential batch effect that may arise due to systematic variation in staining procedures. We recognized the study is unable to control for pre-analytic variables in tissue fixation and processing. However, to address this, the Experimental Pathology Lab at NCI carefully re-titrated the IHC assay to provide a stable assay across all samples.
IHC staining was performed on a Benchmark ULTRA autostainer (Ventana Medical Systems, Tuscon, AZ). TMA sections were deparaffinized with zylene and graded alcohols; antigen retrieval was mediated with citrate buffer pH 9 (Dako) for 20 minutes in a pressure cooker. Primary mouse monoclonal antibody, anti-E-cadherin (clone NCH-38, 1:500; Dako, Carpinteria, CA) was applied at room temperature for 2 hours. The antigen-antibody complex was detected using Envision + (Dako) and DAB was applied for 20 minutes. Slides were counterstained with hematoxylin, dehydrated and coverslipped. Slides were imaged with a Hamamatsu Nanozoomer (Bridgewater, NJ), at 20× magnification and cataloged using the SlidePath Digital Image Hub (Leica Biosystems, Wetzlar, Germany).
As our primary interest was in investigating clinically relevant expression of E-cadherin expression, we used the H-scoring system as has been proposed and evaluated in previous publications23,24,25. Two cytotechnologists assessed digital images of TMA spots using the SlidePath Digital Image Hub, blinded to any clinical data. Manual readings of each TMA spot recorded the quality of the image (unsatisfactory, limited or satisfactory), percentage of cells positively stained for E-cadherin (0, 1, 5, 10, 20, 30, 40, 50, 60, 70, 80, 90 or 100%) and the intensity of staining (0 = negative, 1 = weak, 2 = intermediate, and 3 = strong). Reproducibility of IHC scoring was assessed based on evaluation of 200 images, by the two cytotechnologist and a pathologist (M.E.S.); inter- and intra-observer agreement was excellent (weighted kappa ≥90%; P < 0.001). A summary E-cadherin score was calculated using the product of % positive tumor cells and intensity (range of 0–300)23,24,25. For patients with multiple spots, the maximum E-cadherin score across the spots was calculated for analysis. The median and interquartile range for the E-cadherin score did not vary substantially across the 12 studies (Supplementary Table 2). Tumors having a score of <100 were classified as E-cadherin low and those with a score ≥100 as E-cadherin high. Representative images are shown in Supplementary Figure 1. This cut-point was informed by the known relationship between E-cadherin expression and lobular histology supported by evidence in the literature23,32,33. Further for sensitivity analysis, we also evaluated a more stringent cut-point defining E-cadherin loss with a score = 0 (i.e. no expression of E-cadherin).
Assessment of other tumor markers
Assessment of the tumor markers ER, PR and human epidermal growth factor receptor 2 (HER2), and the definition of positive expression of the tumor markers varied across studies. For the majority of cases (N = 1891, 32%), ER status were primarily extracted from medical records, 15% (N = 908) had ER obtained from IHC staining of whole sections and 28% (N = 1685) had ER obtained from IHC staining of TMAs. Previous publications from participating groups in the current study show good concordances between marker status from medical records and standardized measurements from TMA analyses34,35,36.
Statistical analysis
Case-case analyses
As we performed all staining at the NCI to minimize batch effects, a pooled analysis was conducted using data from all 12 studies. We performed case-case analyses to assess whether there was heterogeneity in risk factor associations by E-cadherin breast tumor expression. We used logistic regression models to estimate case-case odds ratios (OR) and 95% confidence intervals (CIs) where E-cadherin low vs. E-cadherin high expression was the outcome and risk factors the explanatory variables. The ORs were interpreted as the risk factor associations of E-cadherin low disease compared to E-cadherin high disease. For each risk factor, the category that has been shown to be associated with the lowest overall breast cancer risk in the literature was selected as the reference category. Thus, the case-case OR >1 can be interpreted to mean that the risk factor examined in the analysis is more strongly associated with E-cadherin low tumors than with E-cadherin high tumors (ORE-cadherin low vs. control > ORE-cadherin high vs. control). Heterogeneity by E-cadherin subtype were tested using global F test37. Because E-cadherin expression may vary by age and study site23,30,31 all models were adjusted for age (in 10-year categories) and study site. Given that ER status is an important marker of etiologic heterogeneity38, we stratified all analyses by ER status (ER+, ER−). Among ER-positive tumors, we also evaluated associations after stratification by histology (lobular, ductal/mixed); this was not done among ER-negative tumor due to small numbers. To assess the variation in results by study for risk factors that showed evidence of a differential association by E-cadherin expression, we fitted study*risk factor interaction terms in the models to estimate p-heterogeneity by study using the likelihood ratio test; P < 0.20 was considered suggestive evidence of between-study heterogeneity37. In sensitivity analysis, we also assessed associations with risk factors using a more stringent definition of E-cadherin loss, where loss of E-cadherin was defined as those cases with a score of 0.
Survival analysis
For survival analysis, we further excluded patients with distant metastases at diagnosis of the primary tumor (N = 63) and those who were missing vital status (N = 174). In total, 5,696 invasive breast cancer cases from 12 BCAC studies were included in the survival analysis. A total of 1,085 deaths were observed within 10 years of diagnosis, 671 due to cancer. We calculated the survival time for each case as the difference between the date of diagnosis and the date of death or censoring. Analyses were left censored for time to study entry to allow for inclusion of prevalent cases. End of follow-up was defined as the date of death, date of last follow-up or 10 years, whichever came first. Hazard ratios (HR) and 95% CIs for all-cause mortality and breast cancer-specific mortality were estimated using Cox regression models, using study site as a stratifying factor. Multivariable Cox models were adjusted for potential confounders: age at diagnosis (in 10-year categories), tumor grade (well/moderately differentiated, poorly differentiated, or unknown), tumor size (≤2, >2 cm, or unknown), node status (positive, negative, or unknown), HER2 status (positive, negative, or unknown), and histology (ductal/mixed, lobular, other, or unknown). To assess whether the associations vary by tumor characteristics, we also estimated HRs and 95% CI by ER status (positive, negative, unknown), HER2 status (positive, negative, unknown), and, among ER-positive tumors, histology (lobular, ductal/mixed, other/unknown).
All statistical tests were two-sided with 5% type-I error. All pooled analyses were performed using the SAS software version 9.3 (SAS Institute, Inc, Cary, NC).
Results
Study and tumor characteristics by E-cadherin expression
The median age at breast cancer diagnosis was 52 years with some variation by study. E-cadherin low expression by study ranged from 10% to 31% (Supplementary Table 2).
Table 1 presents the distribution of clinicopathologic features by level of E-cadherin tumor tissue expression (low/high). E-cadherin low tumors were more likely to be lobular, well/moderately differentiated (low grade), larger in size (>2 cm), and HER2-negative compared to E-cadherin high tumors (P ≤ 0.005; Table 1). These associations were generally consistent across studies (Supplementary Table 3).
Case-case analyses of risk factor associations with E-cadherin tissue expression among ER-positive tumors overall and stratified by histology
Table 2 presents risk factor associations for ER-positive breast cancers overall and stratified by histology. Among ER-positive cases, compared with E-cadherin high tumors E-cadherin low tumors were marginally associated with ever use of menopausal hormones compared with never users (OR = 1.24, 95% CI = 0.97–1.59, P-het = 0.08). No consistent associations were observed for E-cadherin status by age at menarche, number of live births, age at live birth, or anthropometric measurements (BMI and height; Supplementary Table 4).
Among women with ER-positive tumors, we observed a difference by E-cadherin status for number of live births; women who had 1-birth were less likely to have E-cadherin low expression than women with 2 or more births (OR = 0.74, 95% CI = 0.58–0.95, Table 2); however no trend was present, based on the result of nulliparous women. This relationship was driven by the ductal/mixed tumors while in contrast, for lobular tumors nulliparous women had more frequent loss of E-cadherin compared to those with two or more live births. Other breast cancer risk factors examined, family history of breast cancer, age at menarche, age at menopause, age at first birth, OC and menopausal hormone use, did not exhibit heterogeneity by E-cadherin expression.
Among ER-positive breast cancers of ductal histology, no breast cancer risk factors examined exhibited heterogeneity in their associations by E-cadherin expression (Table 2 and Supplementary Table 4). Analyses using a score of 0 to define E-cadherin loss are presented in Supplemental Table 5–6. In these sensitivity analysis, we observed a stronger relationship with E-cadherin loss with ER expression (Supplemental Table 5), and analysis by risk factors (Supplemental Table 6) showed ever use of menopausal hormones more likely to have E-cadherin loss (defined as score = 0) compared to never users among ER-positive tumors (OR = 1.57, 95% CI = 1.06–2.33, p = 0.02), other factors did not show significant differences.
Case-case analyses of risk factor associations with E-cadherin tissue expression among ER-negative tumors
We did not find differences by E-cadherin expression among ER-negative breast cancers (Table 3), although the result for OC use was marginal. Cases that reported ever use of OC’s were more likely to be E-cadherin low compared with E-cadherin high tumors (OR = 1.97, 95% CI = 0.96–4.06, P-het = 0.06). No significant associations for E-cadherin status were observed for anthropometric measurements including BMI and height (Supplementary Table 4). There were too few cases to evaluate with this more stringent cut-point of 0 to define E-cadherin loss for ER-negative cases.
E-cadherin expression and survival by tumor subtypes
The mean follow-up time was 9.6 years and results for all-cause and breast cancer specific survival are presented in Table 4. E-cadherin expression showed no significant associations with survival in multivariable models overall, or in any of the tumor subtypes.
Discussion
In our study of nearly 6,000 breast cancer patients, with centrally stained and scored TMA slides, our analyses demonstrated E-cadherin loss was significantly associated with lobular histology consistent with previous work. We found limited evidence of heterogeneity of E-cadherin loss, except for menopausal hormone use, to vary by risk factors or with 10-year breast cancer specific survival within tumor subtypes.
Analysis by tumor characteristics and E-cadherin loss showed significant associations with lobular histology, low grade, larger tumor size and lack of HER2 staining, consistent with previous studies39. Lobular breast cancers feature noncohesive cells that are individually dispersed or arranged in a single file pattern, a phenotype that has been attributed to dysregulation of cell-cell adhesion, primarily by loss of E-cadherin protein expression40,41. Lobular breast cancers because of their single file pattern tend to be harder to detect in screening and hence, are larger when diagnosed39, consistent with our data showing E-cadherin loss associated with larger tumor size.
We also observed a relationship between low E-cadherin expression and ever use of menopausal hormones among ER-positive tumors, which was more pronounced when we used a more stringent cut-point of E-cadherin loss (with a score of 0). Numerous studies have shown that menopausal hormone use, particularly combined estrogen-progestin therapy, to be more strongly associated with lobular tumors than with ductal tumors and that reduced use of menopausal hormones is associated with a declining incidence rate of lobular cancers at the population level11,12,13,15,16,17,19,20,21,22,42,43. Further, we also observed among ER-negative breast cancers use of OCs compared to never users to be almost twice as likely to have E-cadherin loss, although not statistically significant. Given that findings suggesting that the relationship between menopausal hormones or OC and breast cancer risk are strongly influenced by recent exposure, it is possible that a true association in our data was attenuated by our reliance on ever as opposed to current use. Given prior epidemiologic studies and in vitro data showing that estrogen may lower E-cadherin expression, the observed relationship with OC or menopausal hormone use may be plausible44.
From our analysis of breast cancer risk factors among ER-positive tumors we observed that women who had one birth were less frequently E-cadherin low compared to women who had two or more live births. We saw an opposite relationship among lobular tumors where nulliparous women had more frequent loss of E-cadherin compared to those with two or more live births. Whether E-cadherin loss in tumors is related to reproductive characteristics requires larger datasets. With regards to genetic factors, mutation profiling studies targeted at CDH1 suggest mutations to be rare and unlikely to explain loss (33/507 based on TCGA data)45,46.
We did not observe associations between E-cadherin and breast cancer specific survival in multivariable models as reported in previous studies24,25,26,27,28. Our data, based on the largest analysis of its kind, do not support E-cadherin as an important marker of survival in breast cancer patients.
Strengths of our study include the use of large, pooled analysis, centrally stained and scored E-cadherin data which allowed for the reduction of any systematic bias that may have been introduced across participating studies. Limitations of this study include limited power for analysis of tumor subgroups due to smaller numbers especially for survival analysis. Although staining of E-cadherin was performed centrally, we had lower than expected percentages of E-cadherin associated lobular breast cancers, which may reflect variation in calling of histologic subtypes, but could also indicate the need for molecular profiling methods or more detailed image analysis studies on the compartment of where E-cadherin is stained needed, for defining E-cadherin loss47,48. In fact, the percentage of E-cadherin low lobular carcinomas varied by study, which could reflect tissue factors that influenced staining, variability in classification of cancers as lobular including potential sampling issues if TMA’s did not capture fully lobular morphology, or factors related to populations and relative frequency of risk exposures.
In summary, this study provides limited evidence for heterogeneity in risk factor associations or for differences in survival by E-cadherin tumor tissue expression. Our data are consistent with molecular profiling studies showing distinctive expression of genes associated with E-cadherin signaling among ER-positive ductal and lobular carcinomas10,49. Evaluating genetic susceptibility markers and E-cadherin loss, where data suggest that genetic susceptibility factors may influence loss of E-cadherin expression, might provide new insights on pathways of E-cadherin loss consistent with histology analysis23,50. Future studies using comprehensive molecular subtyping data, including histology, hormone markers and mRNA, might provide new insights on common and distinct molecular pathways of E-cadherin loss as well as tumor heterogeneity51.
Declarations
Ethics approval and consent to participate
Study participants were recruited under protocols approved by the Institutional Review Board at each institution, and all subjects provided informed consent or did not opt-out, depending on national regulations.
Amsterdam Breast Cancer Study (ABCS), Netherlands Leiden University Medical Center (LUMC) Commissie Medische Ethiek and Protocol Toetsingscommissie van het Nederlands Kanker Instituut/Antoni van Leeuwenhoek Ziekenhuis; Spanish National Cancer Centre Breast Cancer Study (CNIO-BCS) Spain Hospital Universitario La Paz Comite Etico de Investigacion Clinica; ESTHER Breast Cancer Study (ESTHER) GermanyRuprecht-Karls-Universitat Medizinische Fakultat Heidelberg Ethikkommission; Helsinki Breast Cancer Study (HEBCS) Finland Helsingin ja uudenmaan sairaanhoitopiiri (Helsinki University Central Hospital Ethics Committee); Kuopio Breast Cancer Project (KBCP) Finland Pohjois-Savon Sairraanhoitopiirin Kuntayhtyma Tutkimuseettinen Toimikunta; Kathleen Cuningham Foundation Consortium for Familial Breast Cancer/Australian Ovarian Cancer Study (kConFab/AOCS) Australia kConFab: The Queenland Institute of Medical Research Human Research Ethics Committee (QIMR-HREC) AOCS: Peter MacCallum Cancer Centre Ethics Committee; Mayo Clinic Breast Cancer StudY (MCBCS) USA Mayo Clinic IRB; Leiden University Medical Centre Breast Cancer Study (ORIGO) Netherlands Medical Ethical Committee and Board of Directors of the Leiden University Medical Center (LUMC); NCI Polish Breast Cancer Study (PBCS) Poland National Institute of Health (NIH) IRB; Prospective Study of Outcomes in Sporadic Versus Hereditary Breast Cancer (POSH) UK South West Multi-centre Research Ethics Committee; Rotterdam Breast Cancer Study (RBCS) Netherlands Medische Ethische Toetsings Commissie Erasmus Medisch Centrum; UK Breakthrough Generations Study (UKBGS) UK South East Multi-Centre Research Ethics Committee.
Availability of data and materials
The datasets generated and/or analyzed during the current study are not publicly available due to privacy and ethical approvals but are available from the corresponding author on reasonable request.
References
Frixen, U. H. et al. E-cadherin-mediated cell-cell adhesion prevents invasiveness of human carcinoma cells. J Cell Biol 113, 173–185 (1991).
Meiners, S., Brinkmann, V., Naundorf, H. & Birchmeier, W. Role of morphogenetic factors in metastasis of mammary carcinoma cells. Oncogene 16, 9–20, https://doi.org/10.1038/sj.onc.1201486 (1998).
Perl, A. K., Wilgenbus, P., Dahl, U., Semb, H. & Christofori, G. A causal role for E-cadherin in the transition from adenoma to carcinoma. Nature 392, 190–193, https://doi.org/10.1038/32433 (1998).
Berx, G. & Van Roy, F. The E-cadherin/catenin complex: an important gatekeeper in breast cancer tumorigenesis and malignant progression. Breast cancer research: BCR 3, 289–293 (2001).
Cheng, C. W. et al. Mechanisms of inactivation of E-cadherin in breast carcinoma: modification of the two-hit hypothesis of tumor suppressor gene. Oncogene 20, 3814–3823, https://doi.org/10.1038/sj.onc.1204505 (2001).
Acs, G., Lawton, T. J., Rebbeck, T. R., LiVolsi, V. A. & Zhang, P. J. Differential expression of E-cadherin in lobular and ductal neoplasms of the breast and its biologic and diagnostic implications. Am J Clin Pathol 115, 85–98, https://doi.org/10.1309/FDHX-L92R-BATQ-2GE0 (2001).
Gamallo, C. et al. Correlation of E-cadherin expression with differentiation grade and histological type in breast carcinoma. Am J Pathol 142, 987–993 (1993).
Rakha, E. A. et al. Clinical and biological significance of E-cadherin protein expression in invasive lobular carcinoma of the breast. Am J Surg Pathol 34, 1472–1479, https://doi.org/10.1097/PAS.0b013e3181f01916 (2010).
Wasif, N., Maggard, M. A., Ko, C. Y. & Giuliano, A. E. Invasive lobular vs. ductal breast cancer: a stage-matched comparison of outcomes. Ann Surg Oncol 17, 1862–1869, https://doi.org/10.1245/s10434-010-0953-z (2010).
Ciriello, G. et al. Comprehensive Molecular Portraits of Invasive Lobular Breast. Cancer. Cell 163, 506–519, https://doi.org/10.1016/j.cell.2015.09.033 (2015).
Gapstur, S. M., Morrow, M. & Sellers, T. A. Hormone replacement therapy and risk of breast cancer with a favorable histology: results of the Iowa Women’s Health Study. JAMA 281, 2091–2097 (1999).
Li, C. I., Malone, K. E. & Daling, J. R. Differences in breast cancer hormone receptor status and histology by race and ethnicity among women 50 years of age and older. Cancer Epidemiol Biomarkers Prev 11, 601–607 (2002).
Garcia-Closas, M. et al. Established breast cancer risk factors by clinically important tumour characteristics. British journal of cancer 95, 123–129, https://doi.org6603207 (2006).
Nyante, S. J. et al. Risk factors for specific histopathological types of postmenopausal breast cancer in the NIH-AARP Diet and Health Study. American journal of epidemiology 178, 359–371, https://doi.org/10.1093/aje/kws471 (2013).
Li, C. I. et al. The relationship between alcohol use and risk of breast cancer by histology and hormone receptor status among women 65-79 years of age. Cancer Epidemiol Biomarkers Prev 12, 1061–1066 (2003).
Newcomb, P. A. et al. Postmenopausal estrogen and progestin use in relation to breast cancer risk. Cancer Epidemiol Biomarkers Prev 11, 593–600 (2002).
Newcomer, L. M. et al. Postmenopausal hormone therapy and risk of breast cancer by histologic type (United States). Cancer Causes Control 14, 225–233 (2003).
Newcomer, L. M., Newcomb, P. A., Trentham-Dietz, A., Longnecker, M. P. & Greenberg, E. R. Oral contraceptive use and risk of breast cancer by histologic type. International journal of cancer 106, 961–964, https://doi.org/10.1002/ijc.11307 (2003).
Chen, W. Y. et al. Use of postmenopausal hormones, alcohol, and risk for invasive breast cancer. Ann Intern Med 137, 798–804 (2002).
Li, C. I., Stanford, J. L. & Daling, J. R. Anthropometric variables in relation to risk of breast cancer in middle-aged women. Int J Epidemiol 29, 208–213 (2000).
Kotsopoulos, J. et al. Risk factors for ductal and lobular breast cancer: results from the nurses’ health study. Breast cancer research: BCR 12, R106, https://doi.org/10.1186/bcr2790 (2010).
Daling, J. R. et al. Relation of regimens of combined hormone replacement therapy to lobular, ductal, and other histologic types of breast carcinoma. Cancer 95, 2455–2464, https://doi.org/10.1002/cncr.10984 (2002).
Horne, H. N. et al. Breast cancer susceptibility risk associations and heterogeneity by E-cadherin tumor tissue expression. Breast cancer research and treatment 143, 181–187, https://doi.org/10.1007/s10549-013-2771-z (2014).
Younis, L. K., El Sakka, H. & Haque, I. The Prognostic Value of E-cadherin Expression in Breast Cancer. Int J Health Sci (Qassim) 1, 43–51 (2007).
Rakha, E. A. et al. E-cadherin expression in invasive non-lobular carcinoma of the breast and its prognostic significance. Histopathology 46, 685–693, https://doi.org/10.1111/j.1365-2559.2005.02156.x (2005).
Liu, J. et al. CDH1 promoter methylation correlates with decreased gene expression and poor prognosis in patients with breast cancer. Oncol Lett 11, 2635–2643, https://doi.org/10.3892/ol.2016.4274 (2016).
Nass, S. J. et al. Aberrant methylation of the estrogen receptor and E-cadherin 5′ CpG islands increases with malignant progression in human breast cancer. Cancer research 60, 4346–4348 (2000).
Ricciardi, G. R. et al. Androgen Receptor (AR), E-Cadherin, and Ki-67 as Emerging Targets and Novel Prognostic Markers in Triple-Negative Breast Cancer (TNBC) Patients. PLoS One 10, e0128368, https://doi.org/10.1371/journal.pone.0128368 (2015).
Prentice, R. L., Vollmer, W. M. & Kalbfleisch, J. D. On the use of case series to identify disease risk factors. Biometrics 40, 445–458 (1984).
Martinez, M. E., Cruz, G. I., Brewster, A. M., Bondy, M. L. & Thompson, P. A. What can we learn about disease etiology from case-case analyses? Lessons from breast cancer. Cancer epidemiology, biomarkers & prevention: a publication of the American Association for Cancer Research, cosponsored by the American Society of Preventive Oncology 19, 2710–2714, https://doi.org/10.1158/1055-9965.EPI-10-0742 (2010).
Yang, X. R. et al. Associations of breast cancer risk factors with tumor subtypes: a pooled analysis from the Breast Cancer Association Consortium studies. J Natl Cancer Inst 103, 250–263, https://doi.org/10.1093/jnci/djq526 (2011).
Szasz, A. M. et al. Identification of a claudin-4 and E-cadherin score to predict prognosis in breast cancer. Cancer Sci 102, 2248–2254, https://doi.org/10.1111/j.1349-7006.2011.02085.x (2011).
Guler, G. et al. Stem cell-related markers in primary breast cancers and associated metastatic lesions. Mod Pathol 25, 949–955, https://doi.org/10.1038/modpathol.2012.37 (2012).
Bolton, K. L. et al. Assessment of automated image analysis of breast cancer tissue microarrays for epidemiologic studies. Cancer Epidemiol Biomarkers Prev 19, 992–999, 1055-9965.EPI-09-1023 (2010).
Sherman, M. E. et al. Variation in breast cancer hormone receptor and HER2 levels by etiologic factors: a population-based analysis. International journal of cancer 121, 1079–1085, https://doi.org/10.1002/ijc.22812 (2007).
Collins, L. C., Marotti, J. D., Baer, H. J. & Tamimi, R. M. Comparison of estrogen receptor results from pathology reports with results from central laboratory testing. J Natl Cancer Inst 100, 218–221, https://doi.orgdjm270 (2008).
Wang, R., Lagakos, S. W., Ware, J. H., Hunter, D. J. & Drazen, J. M. Statistics in medicine–reporting of subgroup analyses in clinical trials. The New England journal of medicine 357, 2189–2194, https://doi.org/10.1056/NEJMsr077003 (2007).
Jatoi, I., Chen, B. E., Anderson, W. F. & Rosenberg, P. S. Breast cancer mortality trends in the United States according to estrogen receptor status and age at diagnosis. Journal of clinical oncology: official journal of the American Society of Clinical Oncology 25, 1683–1690, https://doi.org/10.1200/JCO.2006.09.2106 (2007).
Pestalozzi, B. C. et al. Distinct clinical and prognostic features of infiltrating lobular carcinoma of the breast: combined results of 15 International Breast Cancer Study Group clinical trials. Journal of clinical oncology: official journal of the American Society of Clinical Oncology 26, 3006–3014, https://doi.org/10.1200/JCO.2007.14.9336 (2008).
McCart Reed, A. E., Kutasovic, J. R., Lakhani, S. R. & Simpson, P. T. Invasive lobular carcinoma of the breast: morphology, biomarkers and ‘omics. Breast cancer research: BCR 17, 12, https://doi.org/10.1186/s13058-015-0519-x (2015).
Morrogh, M. et al. Cadherin-catenin complex dissociation in lobular neoplasia of the breast. Breast cancer research and treatment 132, 641–652, https://doi.org/10.1007/s10549-011-1860-0 (2012).
Rosenberg, L. U. et al. Menopausal hormone therapy and other breast cancer risk factors in relation to the risk of different histological subtypes of breast cancer: a case-control study. Breast cancer research: BCR 8, R11, https://doi.org/10.1186/bcr1378 (2006).
Phipps, A. I., Li, C. I., Kerlikowske, K., Barlow, W. E. & Buist, D. S. Risk factors for ductal, lobular, and mixed ductal-lobular breast cancer in a screening population. Cancer Epidemiol Biomarkers Prev 19, 1643–1654, https://doi.org/10.1158/1055-9965.EPI-10-0188 (2010).
Oesterreich, S. et al. Estrogen-mediated down-regulation of E-cadherin in breast cancer cells. Cancer research 63, 5203–5208 (2003).
Cancer Genome Atlas, N. Comprehensive molecular portraits of human breast tumours. Nature 490, 61–70, https://doi.org/10.1038/nature11412 (2012).
Schrader, K. A. et al. Germline mutations in CDH1 are infrequent in women with early-onset or familial lobular breast cancers. Journal of medical genetics 48, 64–68, https://doi.org/10.1136/jmg.2010.079814 (2011).
Canas-Marques, R. & Schnitt, S. J. E-cadherin immunohistochemistry in breast pathology: uses and pitfalls. Histopathology 68, 57–69, https://doi.org/10.1111/his.12869 (2016).
Sarrio, D. et al. Cytoplasmic localization of p120ctn and E-cadherin loss characterize lobular breast carcinoma from preinvasive to metastatic lesions. Oncogene 23, 3272–3283, https://doi.org/10.1038/sj.onc.1207439 (2004).
Arthur, L. M. et al. Molecular changes in lobular breast cancers in response to endocrine therapy. Cancer research 74, 5371–5376, https://doi.org/10.1158/0008-5472.CAN-14-0620 (2014).
Sawyer, E. et al. Genetic predisposition to in situ and invasive lobular carcinoma of the breast. PLoS genetics 10, e1004285, https://doi.org/10.1371/journal.pgen.1004285 (2014).
Li, J. et al. Identification of high-quality cancer prognostic markers and metastasis network modules. Nature communications 1, 34, https://doi.org/10.1038/ncomms1033 (2010).
Acknowledgements
BCAC: We thank all the individuals who took part in these studies and all the researchers, clinicians, technicians and administrative staff who have enabled this work to be carried out. ABCS: Blood bank Sanquin, The Netherlands. CNIO-BCS: Guillermo Pita, Charo Alonso, Nuria Álvarez, Pilar Zamora, Primitiva Menendez, the Human Genotyping-CEGEN Unit (CNIO). ESTHER: Hartwig Ziegler, Sonja Wolf, Volker Hermann, Christa Stegmaier, Katja Butterbach. HEBCS: Sofia Khan, Johanna Kiiski, Kristiina Aittomäki, Rainer Fagerholm, KBCP: Eija Myöhänen, Helena Kemiläinen. kConFab/AOCS: We wish to thank Heather Thorne, Eveline Niedermayr, all the kConFab research nurses and staff, the heads and staff of the Family Cancer Clinics, and the Clinical Follow Up Study (which has received funding from the NHMRC, the National Breast Cancer Foundation, Cancer Australia, and the National Institute of Health (USA)) for their contributions to this resource, and the many families who contribute to kConFab. Kathleen Cuningham Foundation Consortium for research into Familial Breast cancer (kConFab) investigtaors: Morteza Aghmesheh, David Amor, Lesley Andrews, Yoland Antill, Shane Armitage, Leanne Arnold, Rosemary Balleine, Agnes Bankier, Patti Bastick, Jonathan Beesley, John Beilby, Barbara Bennett, Ian Bennett, Geoffrey Berry, Anneke Blackburn, Michael Bogwitz, Meagan Brennan, Melissa Brown, Michael Buckley, Matthew Burgess, Jo Burke, Phyllis Butow, Keith Byron, David Callen, Ian Campbell, Deepa Chauhan, Manisha Chauhan, Georgia Chenevix-Trench, Alice Christian, Christine Clarke, Paul Cohen, Alison Colley, Ashley Crook, James Cui, Bronwyn Culling, Margaret Cummings, Sarah-Jane Dawson, Anna deFazio, Martin Delatycki, Rebecca Dickson, Joanne Dixon, Alexander Dobrovic, Tracy Dudding, Ted Edkins, Stacey Edwards, Maurice Eisenbruch, Gelareh Farshid, Susan Fawcett, Andrew Fellows, Georgina Fenton, Michael Field, Frank Firgaira, James Flanagan, Jean Fleming, Peter Fong, John Forbes, Stephen Fox, Juliet French, Michael Friedlander, Clara Gaff, Mac Gardner, Mike Gattas, Peter George, Graham Giles, Grantley Gill, Jack Goldblatt, Sian Greening, Scott Grist, Eric Haan, Kate Hardie, Marion Harris, Stewart Hart, Nick Hayward, Sue Healey, Louise Heiniger, John Hopper, Evelyn Humphrey, Clare Hunt, Paul James, Mark Jenkins, Alison Jones, Rick Kefford, Alexa Kidd, Belinda Kiely, Judy Kirk, Jessica Koehler, James Kollias, Serguei Kovalenko, Sunil Lakhani, Amanda Leaming, Jennifer Leary, Jacqueline Lim, Geoff Lindeman, Lara Lipton, Liz Lobb, Graham Mann, Deborah Marsh, Sue Anne McLachlan, Bettina Meiser, Cliff Meldrum, Roger Milne, Gillian Mitchell, Beth Newman, Eveline Niedermayr, Sophie Nightingale, Shona O’Connell, Imelda O’Loughlin, Richard Osborne, Nick Pachter, Briony Patterson, Lester Peters, Kelly Phillips, Melanie Price, Lynne Purser, Tony Reeve, Jeanne Reeve, Robert Richards, Edwina Rickard, Bridget Robinson, Barney Rudzki, Mona Saleh, Elizabeth Salisbury, Joe Sambrook, Christobel Saunders, Jodi Saunus, Robyn Sayer, Elizabeth Scott, Rodney Scott, Clare Scott, Ram Seshadri, Adrienne Sexton, Raghwa Sharma, Andrew Shelling, Peter Simpson, Melissa Southey, Amanda Spurdle, Graeme Suthers, Pamela Sykes, Margaret Tassell, Donna Taylor, Jessica Taylor, Benjamin Thierry, Susan Thomas, Ella Thompson, Heather Thorne, Sharron Townshend, Alison Trainer, Lan Tran, Kathy Tucker, Janet Tyler, Jane Visvader, Logan Walker, Ian Walpole, Robin Ward, Paul Waring, Bev Warner, Graham Warren, Rachael Williams, Judy Wilson, Ingrid Winship, Kathy Wu, Mary Ann Young. ORIGO: We thank E. Krol-Warmerdam, and J. Blom for patient accrual, administering questionnaires, and managing clinical information. The LUMC survival data were retrieved from the Leiden hospital-based cancer registry system (ONCDOC) with the help of Dr. J. Molenaar. PBCS: Louise Brinton, Neonila Szeszenia-Dabrowska, Beata Peplonska, Witold Zatonski, Jolanta Lisssowska, Pei Chao, Michael Stagner. POSH: The ethical approval for the POSH study is MREC /00/6/69, UKCRN ID: 1137. We thank staff in the Experimental Cancer Medicine Centre (ECMC) supported Faculty of Medicine Tissue Bank and the Faculty of Medicine DNA Banking resource. RBCS Petra Bos, Jannet Blom, Ellen Crepin, Elisabeth Huijskens, Anja Kromwijk-Nieuwlaat, Annette Heemskerk, Renée Foekens and Anita Trapman – Jansen.and the Erasmus MC Family Cancer Clinic. UKBGS: We thank Breast Cancer Now and the Institute of Cancer Research for support and funding of the Breakthrough Generations Study, and the study participants, study staff, and the doctors, nurses and other health care providers and health information sources who have contributed to the study. We acknowledge NHS funding to the Royal Marsden/ICR NIHR Biomedical Research Centre. BCAC is funded by Cancer Research UK [C1287/A16563, C1287/A10118], the European Union’s Horizon 2020 Research and Innovation Programme (grant numbers 634935 and 633784 for BRIDGES and B-CAST respectively), and by the European Community’s Seventh Framework Programme under grant agreement number 223175 (grant number HEALTH-F2-2009-223175) (COGS). The ABCS study was supported by the Dutch Cancer Society [grants NKI 2007-3839; 2009 4363]. The CNIO-BCS was supported by the Instituto de Salud Carlos III, the Red Temática de Investigación Cooperativa en Cáncer and grants from the Asociación Española Contra el Cáncer and the Fondo de Investigación Sanitario (PI11/00923 and PI12/00070). The ESTHER study was supported by a grant from the Baden Württemberg Ministry of Science, Research and Arts. Additional cases were recruited in the context of the VERDI study, which was supported by a grant from the German Cancer Aid (Deutsche Krebshilfe). The HEBCS was financially supported by the Helsinki University Central Hospital Research Fund, Academy of Finland (266528), the Finnish Cancer Society, The Nordic Cancer Union and the Sigrid Juselius Foundation. The KBCP was financially supported by the special Government Funding (EVO) of Kuopio University Hospital grants, Cancer Fund of North Savo, the Finnish Cancer Organizations, and by the strategic funding of the University of Eastern Finland. kConFab is supported by a grant from the National Breast Cancer Foundation, and previously by the National Health and Medical Research Council (NHMRC), the Queensland Cancer Fund, the Cancer Councils of New South Wales, Victoria, Tasmania and South Australia, and the Cancer Foundation of Western Australia. Financial support for the AOCS was provided by the United States Army Medical Research and Materiel Command [DAMD17-01-1-0729], Cancer Council Victoria, Queensland Cancer Fund, Cancer Council New South Wales, Cancer Council South Australia, The Cancer Foundation of Western Australia, Cancer Council Tasmania and the National Health and Medical Research Council of Australia (NHMRC; 400413, 400281, 199600). G.C.T. and P.W. are supported by the NHMRC. RB was a Cancer Institute NSW Clinical Research Fellow. The MCBCS was supported by the NIH grants CA192393, CA116167, CA176785 an NIH Specialized Program of Research Excellence (SPORE) in Breast Cancer [CA116201], and the Breast Cancer Research Foundation and a generous gift from the David F. and Margaret T. Grohne Family Foundation. K.J.R. was supported by a training grant under the CTSA Grant Program Numbers UL1 TR000135 and KL2TR000136-09 from the National Center for Advancing Translational Sciences (NCATS) of the NIH. The contents of this paper are solely the responsibility of the authors and do not necessarily represent the official view of NIH. The ORIGO study was supported by the Dutch Cancer Society (RUL 1997-1505) and the Biobanking and Biomolecular Resources Research Infrastructure (BBMRI-NL CP16). The PBCS was funded by Intramural Research Funds of the National Cancer Institute, Department of Health and Human Services, USA. The POSH study is funded by Cancer Research UK (grants C1275/A11699, C1275/C22524, C1275/A19187, C1275/A15956 and Breast Cancer Campaign 2010PR62, 2013PR044. The RBCS was funded by the Dutch Cancer Society (DDHK 2004-3124, DDHK 2009-4318). The UKBGS is funded by Breast Cancer Now and the Institute of Cancer Research (ICR), London. ICR acknowledges NHS funding to the NIHR Biomedical Research Centre.
Author information
Authors and Affiliations
Contributions
Conceived and designed the experiments: H.N.H., H.O., M.E.S., S.H.H., M.G.C., J.D.F. Analyzed the data: H.N.H., H.O., M.P., S.H.H., J.D.F. Contributed reagents/materials/analysis tools: H.N.H., H.O., M.E.S., M.P., S.H.H., M.K.S., R.L.M., D.H., J.B., C.B., M.K.B., H.B., J.C.C., R.C., F.J.C., K.C., P.D., D.F.E., D.M.E., U.E., J.M.H., P.H., B.H., M.J.H., M.J., R.K., A.M., J.W.M.M., T.A.M., H.N., J.E.O., N.O., J.I.A.P., P.D.P.P., K.J.R., K.U.S., M.J.S., C.S., R.S., V.T.H.B.M.S., A.J.S., M.T., A.S.T., A.M.T., T.A.E.M.T., M.A.T., C.J.V.A., C.H.M.V.D., F.F.V.L., L.J.V.V., M.G.C., J.D.F. Drafted paper: H.N.H., H.O., M.E.S., M.P., S.H.H., M.K.S., R.L.M., D.H., J.B., C.B., M.K.B., H.B., J.C.C., R.C., F.J.C., K.C., P.D., D.F.E., D.M.E., U.E., J.M.H., P.H., B.H., M.J.H., M.J., R.K., A.M., J.W.M.M., T.A.M., H.N., J.E.O., N.O., J.I.A.P., P.D.P.P., K.J.R., K.U.S., M.J.S., C.S., R.S., V.T.H.B.M.S., A.J.S., M.T., A.S.T., A.M.T., T.A.E.M.T., M.A.T., C.J.V.A., C.H.M.V.D., F.F.V.L., L.J.V.V., M.G.C., J.D.F. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Horne, H.N., Oh, H., Sherman, M.E. et al. E-cadherin breast tumor expression, risk factors and survival: Pooled analysis of 5,933 cases from 12 studies in the Breast Cancer Association Consortium. Sci Rep 8, 6574 (2018). https://doi.org/10.1038/s41598-018-23733-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-23733-4
This article is cited by
-
Computational quantification and characterization of independently evolving cellular subpopulations within tumors is critical to inhibit anti-cancer therapy resistance
Genome Medicine (2022)
-
E-cadherin expressions on bladder and its association with cancer progressivity: a retrospective cohort study
African Journal of Urology (2022)
-
MicroRNA-135 inhibits initiation of epithelial-mesenchymal transition in breast cancer by targeting ZNF217 and promoting m6A modification of NANOG
Oncogene (2022)
-
Radiobiological effects of wound fluid on breast cancer cell lines and human-derived tumor spheroids in 2D and microfluidic culture
Scientific Reports (2022)
-
Long non-coding RNAs as the critical regulators of doxorubicin resistance in tumor cells
Cellular & Molecular Biology Letters (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.