Abstract
This study aimed to investigate the association of nonalcoholic fatty liver disease (NAFLD) and its severity with the decline in kidney function in patients with chronic kidney disease (CKD). We conducted a cohort study of 1,525 CKD patients who underwent repeated health check-up examinations from January 2003 through December 2013. NAFLD was diagnosed by ultrasonography and its severity was assessed by the NAFLD fibrosis score. At baseline, the prevalence of NAFLD was 40.9%, and the mean estimated glomerular filtration rate (eGFR) was 59.1 ml/min/1.73 m2. The average follow-up was 6.5 years. The age- and sex-adjusted decline in eGFR was greater in patients with NAFLD (−0.79% per year, 95% CI −1.31%, −0.27%) compared to those without it (0.30%, 95% CI −0.14%, 0.76%; p = 0.002). In multivariable adjusted models, the average difference in annual percent change in decline in eGFR comparing patients with NAFLD to those without NAFLD was −1.06% (−1.73%, −0.38%; p = 0.002). The decline in eGFR associated with NAFLD was greater in patients with higher NAFLD fibrosis score, in those with proteinuria or with low eGFR at baseline ( <45 ml/min/1.73 m2), and in those who were smokers and hypertensive. Therefore, NAFLD is independently associated with CKD progression.
Similar content being viewed by others
Introduction
Chronic kidney disease (CKD) is associated with high morbidity and mortality, and is rapidly becoming a major burden to the global health care system1,2. The global prevalence of CKD was estimated at 10.4–13.4%3. End-stage renal disease (ESRD), the last stage of CKD requiring renal replacement therapy, is rapidly increasing worldwide and expected to further increase in the next decade4. There is a substantial interest in identifying novel and potentially reversible causes of progression from early stages of CKD to ESRD.
Nonalcoholic fatty liver disease (NAFLD), characterized by excessive hepatic fat accumulation without significant alcohol intake, use of medications causing fatty liver, or other traditional causes of fatty liver, is the most common cause of chronic liver disease worldwide, especially in developed countries5,6. The global prevalence of NAFLD was estimated at 25.2%7. Furthermore, the consequences of NAFLD extend beyond the liver8. NAFLD is associated with increased risk of type 2 diabetes and cardiovascular disease9,10,11, and with increased prevalence and incidence of CKD12,13,14. No study, however, has evaluated the role of NAFLD on the decline in kidney function among patients who already have reduced kidney function (CKD stage 3 or more). NAFLD is frequently accompanied by multiple traditional risk factors for CKD progression including obesity and diabetes, but evidence supporting the independent role of NAFLD on CKD progression is still lacking.
We thus investigated the association of NAFLD and its severity with the longitudinal decline in kidney function in a cohort of CKD patients, adjusting for traditional risk factors for CKD.
Results
The mean (standard deviation, SD) age of study patients was 60.8 (11.3) years, and the prevalence of NAFLD at baseline was 40.9% (Table 1). Compared to patients without NAFLD, those with NAFLD were more likely to be young, male, smokers, and had a higher level of body mass index (BMI), total cholesterol, and other metabolic risk factors. The mean estimated glomerular filtration rate (eGFR) at baseline was 59.1 ml/min/1.73 m2 (26.2% patients had proteinuria ≥2+ on urinalysis). While eGFR at baseline was higher in patients with NAFLD compared to those without NAFLD at baseline (61.8 vs. 57.3 ml/min/1.73 m2, p < 0.001), patients with NAFLD had higher proportion of proteinuria than those without NAFLD (30.2 vs. 23.5%, p = 0.004).
The average duration of follow-up was 6.5 years (maximum 13.6 years; average number of visits per participant 6.3). During follow-up, the age- and sex-adjusted decline in eGFR was greater in patients with NAFLD (−0.79% per year, 95% CI −1.31%, −0.27%) compared to those without it (0.30%, 95% CI −0.14%, 0.76%; p = 0.002).
After adjustment for age, sex, and year of visit, the average difference in annual percent change of eGFR between patients with NAFLD and those without NAFLD was −1.09% (−1.89%, −0.27%; p = 0.009; Table 2). The results did not materially change after adjusting for confounders or potential metabolic mediators. The multivariable adjusted average difference in annual percent changes in eGFR comparing patients with low NAFLD fibrosis score (NFS) ( < −1.455) or intermediate to high NFS ( ≥ −1.455) and those without NAFLD were 0.01% (−0.74%, 0.99%) and −2.12% (−2.93%, −1.31%), respectively (Table 2).
The association between NAFLD and CKD progression was also observed in patients with only eGFR < 60 ml/min/1.73 m2, with only proteinuria, or with both at baseline (Table 3). The association between NAFLD and the decline in eGFR was consistent across most subgroups (Fig. 1), although the decline in eGFR associated with NAFLD appeared to be stronger among patients who were current smokers (p for interaction < 0.01), hypertensive (p for interaction = 0.02), and those with lower eGFR at baseline (p for interaction < 0.01).
Average difference in annual percent change in estimated glomerular filtration rate (eGFR) in predefined subgroups of CKD patients by baseline nonalcoholic fatty liver disease (NAFLD) status (n = 1,326). *Expected annual percent changes were derived from linear mixed effects models for log-transformed eGFR adjusted for baseline age, sex, year of visit, baseline smoking status (never, former, current, and missing), alcohol (none and moderate), body mass index, hypertension, diabetes, hyperlipidemia, systolic blood pressure, hemoglobin A1c, LDL cholesterol, and triglycerides (loge-transformed).
Among patients with eGFR < 45 ml/min/1.73 m2 at baseline (Supplementary Table S1), the multivariable adjusted average difference in annual percent change of eGFR between patients with NAFLD and those without NAFLD was −5.61% (−11.43%, 0.59%; p = 0.075; Table 4).
Discussion
In this large longitudinal study, NAFLD was an independent risk factor associated with the progression of CKD. The association was stronger in patients with more advanced NAFLD, as indicated by a higher fibrosis score, in those with lower eGFR, in those with proteinuria, and in patients who were current smokers, and hypertensive. Furthermore, the association did not appear to be mediated by traditional risk factors associated with NAFLD. To our knowledge, this is the first report to implicate NAFLD as an important risk factor for CKD progression. Further research needs to consider the clinical impact of screening for and managing NAFLD in CKD patients.
The control of CKD progression is still an unresolved issue4,15. Few specific treatments delay the progression of CKD, especially in patients with advanced CKD, so that identifying risk factors or mediators other than diabetes or hypertension is critically important. NAFLD is a common comorbidity in CKD patients. Although several studies have evaluated the association of NAFLD and CKD, most of them were cross-sectional and did not address the role of NAFLD in CKD progression16,17,18. Similarly, several studies have identified an association between NAFLD and proteinuria16,17,18,19,20,21, but a longitudinal association of NAFLD with kidney function decline in CKD patients had not been evaluated.
Several mechanisms may explain a role of NAFLD in CKD progression. NAFLD is associated with the activation of the nuclear factor-κB and the C-Jun-N-terminal kinase pathways, which enhance transcription of several proinflammatory genes that amplify systemic chronic inflammation and insulin resistance, respectively22,23,24,25,26. These pathways may facilitate CKD progression by augmenting intrarenal immunologic inflammatory responses in CKD, especially in glomerulonephritis where the main pathogenic mechanism is immunologic reaction27. The final common pathologic features in CKD progression are glomerulosclerosis and tubular atrophy accompanied by interstitial fibrosis28,29,30.Pro-inflammatory milieu in NAFLD may accelerate the development of these pathological features in patients with CKD even in patients in whom the initial pathogenic mechanism was not immunologic.
In our study, the association of NAFLD with kidney function decline was stronger in current smokers, and in those who were hypertensive or who had baseline eGFR < 45 ml/min/1.73 m2. This finding suggests specific subgroups may benefit most from screening, prevention and management of NAFLD. The importance of smoking cessation and hypertension control for attenuating CKD progression has been documented in many studies and emphasized in the Kidney Disease Outcomes Quality Initiative guidelines31,32,33,34. The interplay between NAFLD and traditional risk factors of CKD, however, has not been fully elucidated35,36,37,38. Further studies should focus on the interaction between NAFLD and traditional risk factors for CKD progression.
Several limitations need to be considered in the interpretation of our findings. First, we did not have information on the primary cause of CKD. However, we had detailed clinical and laboratory information on comorbidities including diabetes and hypertension, medications, anthropometry, and blood chemistry that allowed for adjusting for multiple confounders and evaluating potential mediators. Second, proteinuria was not measured using a quantitative assay. Thus, we excluded trace or 1 + protein on urinalysis from the proteinuria subgroup because of the possibility of false positives. Third, NAFLD was measured by ultrasound after exclusion of secondary causes for fatty liver. Although the accuracy of ultrasound for establishing the presence of fatty liver is high, it is subject to measurement error6,39. Similarly, we used eGFR as an outcome, which has inherent limitations derived from using creatinine as a filtration marker. Finally, this study was based on Korean patients in health screening exams, and may not be generalizable to other settings or to other ethnicities.
Screening for NAFLD in CKD patients is not routinely performed and there is no specific guideline for screening NAFLD in CKD patients34. Our data suggest that routine screening for NAFLD needs to be contemplated among CKD patients, although additional studies are required to determine the approach and the clinical management of NAFLD in CKD patients. Most treatment strategies for NAFLD involve lifestyle modification such as weight reduction, hypocaloric diet, increase physical activity, smoking cessation, and treatment of associated metabolic risk factors (diabetes, hypertension, and dyslipidemia)5. Future studies will also need to evaluate whether NAFLD resolution can reduce the risk of CKD progression independently of traditional risk factors40.
In summary, NAFLD was associated with declining kidney function in CKD patients independently of established risk factors, and the association was stronger in patients with advanced NAFLD, and in those with proteinuria or with eGFR lower than 45 ml/min/1.73 m2. Our findings suggest that NAFLD may contribute to CKD progression and may help identify patients with a higher risk of rapid progression. CKD patients may benefit from screening and management of NAFLD, which warrants prospective validation.
Methods
Study population
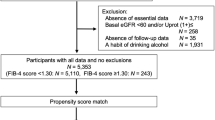
We conducted a retrospective cohort analysis of men and women 18 years of age or older who underwent a comprehensive health screening examination at the Samsung Medical Center Health Promotion Center in Seoul, South Korea, from January 2003 to December 2013 (Fig. 2). Since our objective was to evaluate the longitudinal association between NAFLD and change of eGFR among patients with CKD, the analysis was restricted to subjects who had CKD (eGFR < 60 ml/min/1.73 m2 or proteinuria ≥2 + on urinalysis), and underwent an abdominal ultrasound (US) at baseline, and had at least 1 additional follow up for serum creatinine (n = 1,843). We then excluded patients who had any of the following conditions at baseline: history of cancer (n = 34), history of liver cirrhosis, positive hepatitis B surface antigen, or hepatitis C virus antibodies (n = 70), alcohol intake ≥ 30 g/day in men or ≥20 g/day in women (n = 85), or had received a kidney transplant or started dialysis within 1 year after the baseline examination (n = 13). We also excluded patients who had missing information on alcohol intake (n = 113), NFS (n = 3), or less than 6 months follow up (n = 6). Since study patients could have more than one exclusion criteria, the final sample size was 1,525 (1,065 men and 460 women). The Institutional Review Board of the Samsung Medical Center approved this study and waived the requirement for informed consent as we used only de-identified data routinely collected during health screening visits. All methods were performed in accordance with the relevant guidelines and regulations.
Data collection
At each visit, demographic characteristics, smoking status, alcohol consumption, medical history, and medication use were collected through standardized, self-administered questionnaires. Smoking status was categorized into never, former, or current smoker. Alcohol consumption was categorized into none or moderate (<30 g/day in men and <20 g/day in women). Height, weight, and sitting blood pressure were measured by trained nurses. BMI was calculated as weight in kilograms divided by height in meters squared.
Serum creatinine was measured with the kinetic alkaline picrate method (Jaffe method) using automated chemistry analyzers Hitachi 7600 (Hitachi, Tokyo, Japan) from 2003 to 2009 and Modular DP (Roche, Basel, Switzerland) from 2009 to 2013. Serum glucose was measured by the hexokinase/glucose-6-phosphate dehydrogenase method. Hemoglobin A1c (HbA1c) was measured by high performance liquid chromatography. Aspartate aminotransferase (AST), alanine aminotransferase (ALT), and gamma-glutamyltransferase (GGT) were measured following the International Federation of Clinical Chemistry method. Urine protein was measured semi-quantitatively by urine dipstick using urine chemistry analyzers, Cliniteck Atlas (Siemens, Munich, Germany) or Urisys 2400 (Roche). The Department of Laboratory Medicine and Genetics at Samsung Medical Center has participated in several proficiency testing programs operated by the Korean Association of Quality Assurance for Clinical Laboratory, the Asian Network of Clinical Laboratory Standardization and Harmonization, and the College of American Pathologists.
Abdominal US
Abdominal US imaging was performed using LogiQ E9 (GE Healthcare, Milwaukee, Wisconsin, USA), iU22 xMatrix (Philips Medical Systems, Cleveland, Ohio, USA) or ACUSON Sequoia 512 (Siemens, Issaquah, Washington, USA) equipments by experienced radiologists unaware of the study aims. Images were captured in a standard fashion with the patient in the supine position with the right arm raised above the head. An US diagnosis of fatty liver was made based on standard criteria, including parenchymal brightness, liver-to-kidney contrast, deep beam attenuation and bright vessel walls41,42. Since we had already excluded patients with excessive alcohol use (≥30 g/day for men and ≥20 g/day for women) as well as other identifiable causes of fatty liver at baseline as described in the exclusion criteria, fatty liver was considered NAFLD.
Among individuals with NAFLD, we used the NFS to assess severity of fibrosis. The NFS was calculated as −1.675 + 0.037 × age (years) + 0.094 × BMI (kg/m2) + 1.13 × impaired fasting glucose/diabetes (yes = 1, no = 0) + 0.99 × AST/ALT ratio − 0.013 × platelet count (×109/l) − 0.66 × albumin (g/dl)43. Based on NFS, patients with NAFLD were classified as high-intermediate (NFS ≥ −1.455) and low probability (NFS < −1.455) of advanced fibrosis.
Estimated glomerular filtration rate and proteinuria
EGFR was calculated from serum creatinine using the CKD epidemiology collaboration (CKD-EPI) equation44: for women with serum creatinine ≤ 0.7, eGFR was calculated as 144 × (serum creatinine/0.7)−0.329× (0.993)age; for women with serum creatinine >0.7, eGFR was calculated as 144 × (serum creatinine/0.7)−1.209 × (0.993)age; for men with serum creatinine ≤ 0.9, eGFR was calculated as 141 × (serum creatinine/0.9)−0.411 × (0.993)age; and for men with serum creatinine > 0.9, eGFR was calculated as 141 × (serum creatinine/0.9)−1.209 × (0.993)age. Significant proteinuria was defined as protein ≥2+ on urinalysis. CKD was defined as eGFR < 60 ml/min/1.73 m2 or protein ≥2+ on urinalysis at the baseline examination. The degree of CKD progression was evaluated as the average annual percent change in eGFR from baseline eGFR. For the severity of CKD, patients were further divided into two groups using a cut-off value of eGFR ≥45 ml/min/1.73 m2 vs. <45 ml/min/1.73 m2 (dividing G3a and G3b) at baseline.
Statistical analysis
We compared serial changes in eGFR among CKD patients with or without NAFLD at baseline using linear mixed models for longitudinal data with random intercepts and random slopes45. To account for correlations in eGFR from repeated measurements over time in the same participant, the main analyses consisted of two-level linear mixed models for longitudinal data. We modeled linear trajectories in eGFR with time at the first level, and variations in eGFR trajectories across participants at the second level. We used loge-transformed (eGFR) as the outcome and we estimated the average difference in annual percent change in eGFR (with 95% confidence intervals [CI]) comparing patients with NAFLD to those without NAFLD at baseline. The 95% CIs were obtained from linear mixed models with different intersecting linear trends during follow-up time, interactions between linear time and baseline NAFLD status, and random variations in linear time among participants.
We used three models with increasing degrees of adjustment to account for potential confounding factors at baseline. Model 1 adjusted for age, sex, and year of visit. Model 2 further adjusted for smoking (never, former, current, and missing), alcohol consumption (none and moderate), and BMI. In addition, to evaluate potential mediation of the association between NAFLD and eGFR changes by metabolic factors, model 3 further adjusted for hypertension, diabetes, hyperlipidemia, systolic blood pressure, HbA1c, low-density lipoprotein (LDL) cholesterol, and triglycerides.
We conducted sensitivity analyses depending on CKD criteria at baseline: eGFR < 60 ml/min/1.73 m2 only, proteinuria only, and both. In addition, we performed stratified analyses to evaluate if the association of NAFLD with CKD progression differed in pre-specified subgroups defined by age (<60 vs. ≥ 60 years), sex, smoking (never or former vs. current), alcohol drinking (none vs. moderate), obese (defined as BMI ≥ 25 kg/m2), hypertension (defined as systolic blood pressure ≥ 140 mmHg, diastolic blood pressure ≥ 90 mmHg, or the use of antihypertensive medication), diabetes (defined as fasting serum glucose ≥ 126 mg/dl, hemoglobin A1c ≥ 6.5%, or the use of antidiabetic medication), hyperlipidemia (defined as high-density lipoprotein (HDL)-cholesterol < 40 mg/dl in men or < 50 mg/dl in women, triglycerides ≥ 150 mg/dl, or the use of lipid-lowering medication), or baseline eGFR (<45 vs. ≥ 45 ml/min/1.73 m2). All reported P values were two-sided and the significance level was set at 0.05. Statistical analyses were performed using Stata version 14 (Stata Corp., College Station, Texas).
Change history
21 May 2021
A Correction to this paper has been published: https://doi.org/10.1038/s41598-021-90150-5
References
Jha, V. et al. Chronic kidney disease: global dimension and perspectives. Lancet 382, 260–272, https://doi.org/10.1016/S0140-6736(13)60687-X (2013).
Eckardt, K. U. et al. Evolving importance of kidney disease: from subspecialty to global health burden. Lancet 382, 158–169, https://doi.org/10.1016/S0140-6736(13)60439-0 (2013).
Coresh, J. Update on the Burden of CKD. J Am Soc Nephrol 28, 1020–1022, https://doi.org/10.1681/ASN.2016121374 (2017).
Liyanage, T. et al. Worldwide access to treatment for end-stage kidney disease: a systematic review. Lancet 385, 1975–1982, https://doi.org/10.1016/S0140-6736(14)61601-9 (2015).
European Association for the Study of the Liver, European Association for the Study of Diabetes & European Association for the Study of Obesity. EASL-EASD-EASO Clinical Practice Guidelines for the management of non-alcoholic fatty liver disease. J Hepatol 64, 1388-1402, https://doi.org/10.1016/j.jhep.2015.11.004 (2016).
Kim, J. M. et al. Predicting Hepatic Steatosis in Living Liver Donors via Noninvasive Methods. Medicine (Baltimore) 95, e2718, https://doi.org/10.1097/MD.0000000000002718 (2016).
Younossi, Z. M. et al. Global epidemiology of nonalcoholic fatty liver disease-Meta-analytic assessment of prevalence, incidence, and outcomes. Hepatology 64, 73–84, https://doi.org/10.1002/hep.28431 (2016).
Byrne, C. D. & Targher, G. NAFLD: a multisystem disease. J Hepatol 62, S47–64, https://doi.org/10.1016/j.jhep.2014.12.012 (2015).
Loomba, R. & Sanyal, A. J. The global NAFLD epidemic. Nature reviews. Gastroenterology & hepatology 10, 686–690, https://doi.org/10.1038/nrgastro.2013.171 (2013).
Anstee, Q. M., Targher, G. & Day, C. P. Progression of NAFLD to diabetes mellitus, cardiovascular disease or cirrhosis. Nature reviews. Gastroenterology & hepatology 10, 330–344, https://doi.org/10.1038/nrgastro.2013.41 (2013).
Targher, G., Day, C. P. & Bonora, E. Risk of cardiovascular disease in patients with nonalcoholic fatty liver disease. The New England journal of medicine 363, 1341–1350, https://doi.org/10.1056/NEJMra0912063 (2010).
Targher, G., Chonchol, M., Zoppini, G., Abaterusso, C. & Bonora, E. Risk of chronic kidney disease in patients with non-alcoholic fatty liver disease: is there a link? J Hepatol 54, 1020–1029, https://doi.org/10.1016/j.jhep.2010.11.007 (2011).
Armstrong, M. J., Adams, L. A., Canbay, A. & Syn, W. K. Extrahepatic complications of nonalcoholic fatty liver disease. Hepatology 59, 1174–1197, https://doi.org/10.1002/hep.26717 (2014).
Sinn, D. H. et al. Development of chronic kidney disease in patients with non-alcoholic fatty liver disease: A cohort study. J Hepatol, https://doi.org/10.1016/j.jhep.2017.08.024 (2017).
Levey, A. S. & Coresh, J. Chronic kidney disease. Lancet 379, 165–180, https://doi.org/10.1016/S0140-6736(11)60178-5 (2012).
Targher, G. et al. Non-alcoholic fatty liver disease is independently associated with an increased prevalence of chronic kidney disease and proliferative/laser-treated retinopathy in type 2 diabetic patients. Diabetologia 51, 444–450, https://doi.org/10.1007/s00125-007-0897-4 (2008).
Sirota, J. C., McFann, K., Targher, G., Chonchol, M. & Jalal, D. I. Association between nonalcoholic liver disease and chronic kidney disease: an ultrasound analysis from NHANES 1988-1994. American journal of nephrology 36, 466–471, https://doi.org/10.1159/000343885000343885 (2012).
Targher, G. et al. Relationship between kidney function and liver histology in subjects with nonalcoholic steatohepatitis. Clinical journal of the American Society of Nephrology: CJASN 5, 2166–2171, https://doi.org/10.2215/CJN.05050610 (2010).
Hwang, S. T. et al. Impact of non-alcoholic fatty liver disease on microalbuminuria in patients with prediabetes and diabetes. Internal medicine journal 40, 437–442, https://doi.org/10.1111/j.1445-5994.2009.01979.x (2010).
Yasui, K. et al. Nonalcoholic steatohepatitis and increased risk of chronic kidney disease. Metabolism: clinical and experimental 60, 735–739, https://doi.org/10.1016/j.metabol.2010.07.022 (2011).
Targher, G. et al. Nonalcoholic fatty liver disease is independently associated with an increased incidence of chronic kidney disease in patients with type 1 diabetes. Diabetes care 37, 1729–1736, https://doi.org/10.2337/dc13-2704 (2014).
Badman, M. K. & Flier, J. S. The adipocyte as an active participant in energy balance and metabolism. Gastroenterology 132, 2103–2115, https://doi.org/10.1053/j.gastro.2007.03.058 (2007).
Shoelson, S. E., Herrero, L. & Naaz, A. Obesity, inflammation, and insulin resistance. Gastroenterology 132, 2169–2180, https://doi.org/10.1053/j.gastro.2007.03.059 (2007).
Byrne, C. D. Dorothy Hodgkin Lecture 2012: non-alcoholic fatty liver disease, insulin resistance and ectopic fat: a new problem in diabetes management. Diabetic medicine: a journal of the British Diabetic Association 29, 1098–1107, https://doi.org/10.1111/j.1464-5491.2012.03732.x (2012).
Sabio, G. et al. A stress signaling pathway in adipose tissue regulates hepatic insulin resistance. Science 322, 1539–1543, https://doi.org/10.1126/science.1160794 (2008).
Tilg, H. & Moschen, A. R. Evolution of inflammation in nonalcoholic fatty liver disease: the multiple parallel hits hypothesis. Hepatology 52, 1836–1846, https://doi.org/10.1002/hep.24001 (2010).
Anders, H. J., Jayne, D. R. & Rovin, B. H. Hurdles to the introduction of new therapies for immune-mediated kidney diseases. Nature reviews. Nephrology 12, 205–216, https://doi.org/10.1038/nrneph.2015.206 (2016).
Kok, H. M., Falke, L. L., Goldschmeding, R. & Nguyen, T. Q. Targeting CTGF, EGF and PDGF pathways to prevent progression of kidney disease. Nature reviews. Nephrology 10, 700–711, https://doi.org/10.1038/nrneph.2014.184 (2014).
Kriz, W. & Lemley, K. V. A potential role for mechanical forces in the detachment of podocytes and the progression of CKD. J Am Soc Nephrol 26, 258–269, https://doi.org/10.1681/ASN.2014030278 (2015).
Schelling, J. R. Tubular atrophy in the pathogenesis of chronic kidney disease progression. Pediatric nephrology 31, 693–706, https://doi.org/10.1007/s00467-015-3169-4 (2016).
Jain, G. & Jaimes, E. A. Nicotine signaling and progression of chronic kidney disease in smokers. Biochemical pharmacology 86, 1215–1223, https://doi.org/10.1016/j.bcp.2013.07.014 (2013).
Palit, S. et al. Association of BP with Death, Cardiovascular Events, and Progression to Chronic Dialysis in Patients with Advanced Kidney Disease. Clinical journal of the American Society of Nephrology: CJASN 10, 934–940, https://doi.org/10.2215/CJN.08620814 (2015).
Lash, J. P. et al. Chronic Renal Insufficiency Cohort (CRIC) Study: baseline characteristics and associations with kidney function. Clinical journal of the American Society of Nephrology: CJASN 4, 1302–1311, https://doi.org/10.2215/CJN.00070109 (2009).
Summary of Recommendation Statements. Kidney international supplements 3, 5–14, https://doi.org/10.1038/kisup.2012.77 (2013).
Kurella, M., Lo, J. C. & Chertow, G. M. Metabolic syndrome and the risk for chronic kidney disease among nondiabetic adults. J Am Soc Nephrol 16, 2134–2140, https://doi.org/10.1681/ASN.2005010106 (2005).
Weiner, D. E. et al. The relationship between nontraditional risk factors and outcomes in individuals with stage 3 to 4 CKD. American journal of kidney diseases: the official journal of the National Kidney Foundation 51, 212–223, https://doi.org/10.1053/j.ajkd.2007.10.035 (2008).
Kendrick, J. & Chonchol, M. B. Nontraditional risk factors for cardiovascular disease in patients with chronic kidney disease. Nature clinical practice. Nephrology 4, 672–681, https://doi.org/10.1038/ncpneph0954 (2008).
Kronenberg, F. Emerging risk factors and markers of chronic kidney disease progression. Nature reviews. Nephrology 5, 677–689, https://doi.org/10.1038/nrneph.2009.173 (2009).
Hernaez, R. et al. Diagnostic accuracy and reliability of ultrasonography for the detection of fatty liver: a meta-analysis. Hepatology 54, 1082–1090, https://doi.org/10.1002/hep.24452 (2011).
Musso, G. et al. Fatty Liver and Chronic Kidney Disease: Novel Mechanistic Insights and Therapeutic Opportunities. Diabetes care 39, 1830–1845, https://doi.org/10.2337/dc15-1182 (2016).
Saverymuttu, S. H., Joseph, A. E. & Maxwell, J. D. Ultrasound scanning in the detection of hepatic fibrosis and steatosis. British medical journal 292, 13–15 (1986).
Mathiesen, U. L. et al. Increased liver echogenicity at ultrasound examination reflects degree of steatosis but not of fibrosis in asymptomatic patients with mild/moderate abnormalities of liver transaminases. Digestive and liver disease: official journal of the Italian Society of Gastroenterology and the Italian Association for the Study of the Liver 34, 516–522, https://doi.org/10.1016/S1590-8658(02)80111-6 (2002).
Angulo, P. et al. The NAFLD fibrosis score: a noninvasive system that identifies liver fibrosis in patients with NAFLD. Hepatology 45, 846–854, https://doi.org/10.1002/hep.21496 (2007).
Levey, A. S. et al. A new equation to estimate glomerular filtration rate. Annals of internal medicine 150, 604–612, https://doi.org/10.7326/0003-4819-150-9-200905050-00006 (2009).
Fitzmaurice, G. M., Laird, N. M. & Ware, J. H. Applied Longitudinal Analysis. 2nd edn (Wiley, 2011).
Author information
Authors and Affiliations
Contributions
Jang H.R. and Gwak G.Y. designed the study. Kang D., Gu S. and Cho S.J. collected the data. Kang D., Gu S., Guallar E. and Cho J. performed the data analysis. Jang H.R., Kang D., Sinn D.H., Lee J.E., Huh W., Paik S.W., Ryu S., Chang Y., Shafi T., Lazo M., Gullar E., Cho J. and Gwak G.Y. wrote the final report. All authors contributed to critical revision of the final report. Cho J. and Gwak G.Y. are guarantors. All authors had full access to all of the data and can take responsibility for the integrity of the data and the accuracy of the data analysis. All authors approved the final version of the manuscript.
Corresponding authors
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original online version of this Article was revised: The original version of this Article contained a repeated error in Affiliations 1,2,3,4 and 5 where “Kangbuk Samsung Hospital” was incorrectly given as the Organisational name.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Jang, H.R., Kang, D., Sinn, D.H. et al. Nonalcoholic fatty liver disease accelerates kidney function decline in patients with chronic kidney disease: a cohort study. Sci Rep 8, 4718 (2018). https://doi.org/10.1038/s41598-018-23014-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-23014-0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.