Abstract
While saline soils account for 6.5% of the total land area globally, it comprises about 70% of the area in northwestern China. Microbiota in these saline soils are particularly important because they are critical to maintaining ecosystem services. However, little is known about the microbial diversity and community composition in saline soils. To investigate the distribution patterns and edaphic determinants of bacterial communities in saline soils, we collected soil samples across the hypersaline Ebinur Lake shoreline in northwestern China and assessed soil bacterial communities using bar-coded pyrosequencing. Bacterial communities were diverse, and the dominant phyla (>5% of all sequences) across all soil samples were Gammaproteobacteria, Actinobacteria, Firmicutes, Alphaproteobacteria, Bacteroidetes and Betaproteobacteria. These dominant phyla made a significant (P < 0.05) contribution to community structure variations between soils. Halomonas, Smithella, Pseudomonas and Comamonas were the indicator taxa across the salinity gradient. Bacterial community composition showed significant (P < 0.05) correlations with salt content and soil pH. Indeed, bacterial phylotype richness and phylogenetic diversity were also higher in soils with middle-level salt rates, and were significantly (P < 0.05) correlated with salt content and soil pH. Overall, our results show that both salinity and pH are the determinants of bacterial communities in saline soils in northwest China.
Similar content being viewed by others
Introduction
Over 800 million hectares of land throughout the world are affected by salt1. Taking China as an example, northwestern China accounts for 71% of the total area of China, with 70% of the area in northwestern China comprising saline soil2. Saline soils are considered harsh habitats for life, but such habitats harbor active and diverse microbial communities3,4,5. It is well known that soil microorganisms are critical to maintaining ecosystem services due to their contributions to soil formation, organic matter decomposition, biogeochemical cycling and plant nutrition6,7,8,9. Studies in hypersaline ecosystems have characterized some enzymes and novel organisms with enhanced potential for biotechnological applications5. Thus, knowledge of microbial diversity in saline soil is indispensable to a comprehensive insight into the biology of extreme ecosystems.
In the past few years, reports are available on soil microbial diversity and composition from various terrestrial habitats, yielding useful insights and highlighting the importance of contemporary soil factors such as soil moisture10, soil trophic status11,12,13 and soil pH14,15,16,17,18. However, most of these studies focused on the microbiology of non-saline ecosystems. Only a few studies assessed the microbiology of saline environments, with the majority of them focusing on aquatic communities19,20,21,22 and/or sediments23,24,25,26, and far fewer attempting to characterize saline soils5,27. Thus, there is a dearth of knowledge about the structure and ecology of microbial communities in saline soils.
A recent global survey of microbial communities from natural environments found that saline soil harbors diverse bacterial communities, and that salinity was the major factor in shaping bacterial composition28. However, nearly half of the samples used in the study were collected from non-saline soils or different soil types. Similarly, another study reported that in soils and sediments from a hypersaline lake located in southern Texas, microbial communities were mainly driven by water content and phosphorus, rather than salt5. However, samples used in this study were sampled along a wide transect that spanned vegetated areas, upland areas, exposed lakebed sediments and waterlogged locations, which represent different habitats including terrestrial, intermediate and aquatic. Specifically, water influences oxygen concentrations, thus it might have stronger impacts on bacterial community structures in sediments and waterlogged locations than that of terrestrial soils. In addition, vegetated communities may result in very different microbial distribution patterns compared to upland areas. These considerations raise pertinent question about the relative influence of soil salt content as compared to other soil attributes in structuring soil microbiota in the same saline habitat. To unravel the importance of these potential drivers, our study focused on bacterial communities in the saline soil from a vegetated shoreline.
The Ebinur Lake region located in the center of an oasis, is the most representative saline ecosystem in the arid zone of China. This region has a vast area of dry lakebeds covered with halophytic vegetation that plays a key role in maintaining the regional ecological balance. It thus provides a natural laboratory for studying bacterial diversity and composition on a regional scale in a saline ecosystem. Using next-generation sequencing method, this study examined the bacterial communities in pristine soils from ten representative halophytic vegetated sites around the Ebinur Lake. Specifically, our study aimed: (i): to reveal the bacterial diversity and community composition in saline soils around the Ebinur Lake; (ii) to determine the key factors shaping bacterial distribution patterns in these soils and quantify their relative contribution to community variation. We hypothesized that salinity rather than other soil properties is more important in shaping bacterial community structure in the saline soil under halophytic vegetation.
Results
Soil physicochemical properties
The general physicochemical characteristics of soil samples represent the large pairwise geographical distance across 120 km (Supplementary Table S1). Soil salinity significantly (P < 0.05) varied from 1.77 to 8.74% (m/m) across the soil samples where the EC showed corresponding values from 4.44 to 10.83. The soil samples also differed significantly (P < 0.05) from each other in terms of pH (ranging from 7.94 to 8.62), organic matter (OM) (ranging from 3.32 to 9.84 g kg−1) or TN (ranging from 0.28 to 0.85 g kg−1). There was a significant (P < 0.05) variation in available nutrients, which ranged from 2.81 to 11.60 mg kg−1 for available phosphorous (AP), and from 192.11 to 453.11 mg kg−1 for available potassium (AK). In addition, other nutrients such as NH4+–N (ammonium nitrogen) and NO3−–N (nitrate nitrogen) significantly (P < 0.05) varied from 4.87 to 12.05 mg kg−1 and 2.98 to 9.61 mg kg−1, respectively.
Distribution of taxa and phylotypes
In total, 298667 quality 16S rRNA gene sequences were yielded from all 30 soil samples, and 4500–20415 sequences were obtained per sample (mean 9955). Of these sequences, when grouped at the 97% similarity threshold, there were 4389 different phylotypes over all samples, with a mean of 701 phylotypes per sample. The dominant groups (relative abundance >5%) across all soil samples were Gammaproteobacteria (23.55%), Actinobacteria (16.74%), Firmicutes (9.96%), Alphaproteobacteria (9.06%), Bacteroidetes (7.49%) and Betaproteobacteria (7.27%), which together accounted for more than 74.07% of the total bacterial sequences (Fig. 1). In addition, groups of Acidobacteria, Chloroflexi, Cyanobacteria, Deltaproteobacteria, Gemmatimonadetes, Planctomycetes and Saccharibacteria were present in most soils at low abundance (relative abundance >0.1%) (Fig. 1), and 20 other rarer phyla were identified (Supplementary Table S2).
The most abundant group of Gammaproteobacteria (r = 0.714, P < 0.001) were negatively correlated with salt content, while Betaproteobacteria (r = 0.835, P < 0.001) and Firmicutes (r = 0.813, P < 0.001) were significantly positively correlated with salt content (Supplementary Table S3). Interestingly, these three group (Gammaproteobacteria, Betaproteobacteria and Firmicutes) had the same trends towards correlating positively or negatively with soil pH. The relative abundance of Actinobacteria exhibited no correlation with salt content and pH, but it was significantly negatively correlated with OM (r = −0.476, P < 0.001), NH4+–N (r = −0.385, P < 0.05) and AK (r = −0.365, P < 0.05). Another dominant class, Alphaproteobacteria, had no significant correlations with any edaphic soil properties (Supplementary Table S3).
Bacterial community structure
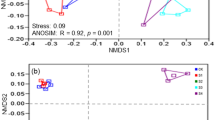
The difference or similarity of bacterial community structures across all soil samples was illustrated by using non-metric multidimensional scaling (NMDS) plots of UniFrac community distances (Fig. 2). The NMDS plot indicated that the bacterial structure was well divided and strongly influenced by salt content (Mantel text, r = 0.712, P < 0.01) and soil pH (Mantel text, r = 0.602, P < 0.01) (Table 1). Based on the NMDS dissimilarity matrix, the bacterial communities were roughly clustered into three groups based on the NMDS dissimilarity matrix (see Supplementary Fig. S2). Group I consisted of two soils with salt content between 1.5–2.0% (Low content); Group II consisted of six soils with salt content between 3.5–5.5% (Middle content); and Group III contained two soils with salt content between 7.5–8.5% (High content). Together, the results indicated bacterial community structures were significantly different across salt content.
Bacterial community compositional structure in saline soils as indicated by non-metric multidimensional scaling (NMDS) plots of weighted pairwise UniFrac community distances (A), and the Bray-Curtis dissimilarity of bacterial community between sites (B). Sites have been color-coded to salt gradients.
The SIMPER analysis was conducted to explore bacterial phyla that contributed the most to the community dissimilarity between the samples. The relative abundance of microbes and contribution of bacterial phyla between soils were analyzed separately (Table 2). Bacterial communities in the soils with a salt concentration between 3.5 and 4.5% and a salt concentration between 4.5 and 5.5% were most similar, but were different from those observed in the soils with a salt concentration of 1.5–2.0% and the soils with a salt concentration of 7.5–8.5%. The dominant phyla such as Gammaproteobacteria, Actinobacteria, Firmicutes and Betaproteobacteria made a greater contribution than other phyla to the community structure variations between soils. While the relative abundance of Alphaproteobacteria was greater than that of Betaproteobacteria, they made a smaller contribution to the community differences (Table 2, Supplementary Table S2).
Indicator taxa
Taxonomic assignment of indicator species across the salt gradient has been presented in Table 3. OTU988 was the most abundant indicator taxon (relative abundance >10%) in the soils with salt concentration between 1.5 and 2.0%, and it was closely related to the genus Halomonas. Similarly, OTU2607 was the most abundant indicator taxon in the soils with salt concentration between 3.5 and 4.5% and it was related to the genus Smithella. At the next salinity level (4.5 and 5.5%), the most abundant indicator taxon was OTU3196, which was closely related to the genus Pseudomonas. OTU1657, closely related to the genus Comamonas, was the most abundant (relative abundance >20%) indicator taxon in soils with salt concentration between 7.5 and 8.5%.
Bacterial diversity
The diversity of the bacterial communities was highly variable in terms of both phylotype richness and phylogenetic diversity across the sample set (Figs 3 and 4). Each site contained 300–700 unique phylotypes within the 4500 randomly selected sequences per soil. Phylotype richness and phylogenetic diversity were generally higher in soils with salt concentration between 3.5 and 5.5%, while the lowest phylotype richness and phylogenetic diversity was found in soil samples with low salt content (Group I, salt concentration was between 1.5 and 2.5%) (Fig. 3). Regardless of the diversity metric employed, the bacterial diversity was best expressed by quadratic equations and it was strongly correlated with soil salt (R2 = 0.781, P < 0.001 for phylogenetic diversity; R2 = 0.793, P < 0.001 for phylotype richness) and soil pH (R2 = 0.548, P < 0.001 for phylogenetic diversity; R2 = 0.583, P < 0.001 for phylotype richness) (Fig. 4). Together, these results strongly suggest that local soil salt and pH play an important role in determining the soil bacterial composition and diversity among sites across Ebinur Lake.
Edaphic drivers of bacterial community structure
Bacterial communities were sharply varied with salt content gradient, and the Mantel test showed there was a significant correlation between bacterial communities and salt concentration (r = 0.712, P < 0.001). Bacterial community composition was also significantly correlated (P < 0.001) with soil pH, EC, NH4+–N, AK, NO3−–N, OM and TN (Table 1). Of these variables, soil pH showed the highest correlation with the bacterial community composition (r = 0.602, P < 0.001). Based on the results of the Mantel test, the soil parameters significantly correlated with bacterial community structures were selected to perform canonical correspondence analysis (CCA). The CCA plots of bacterial community structure clearly exhibited that salt content and pH were the two longest vectors along CCA1 explaining 29.98% of variation of communities (Fig. 5). Thus, salt content and pH were the two important drivers controlling the community composition, which was then followed by other soil properties.
Discussion
This study showed that bacterial communities in the saline soils under halophytic vegetation around Ebinur Lake are distinct and considerably different from those described in hypersaline sediments. For example, previous work found low G + C Gram-positive bacteria (a subgroup of Firmicutes), Alphaproteobacteria or Deltaproteobacteria was the predominant group in many saline sediments4,21,23,24, but Gammaproteobacteria was the dominant phylum in the saline soils in the current study. Additionally, Acidobacteria was major group in the hypersaline soils and sediments across La Sal del Rey lake in southern Texas5, but this phylum was less abundant across the saline soil around Ebinur Lake. Interestingly, the relative abundance of Chloroflexi, Gemmatimonadetes and Planctomycetes at sites with mid-level salt concentration was nearly 5%, which was similar to the abundances of those phyla in agricultural black soils29. However, the abundances of those phyla in low salt sites or high salt sites was nearly 1%, which was similar to values of those phyla in pristine soils14,15. These results suggest that bacterial community composition might be strongly governed by saline environmental factors around Ebinur Lake. In addition, the deep sequencing performed here was helpful for detection of bacterial taxa not described previously in saline ecosystems. These taxa generally occurred in low abundance and included members of the Armatimonadetes, Fibrobacteres, Microgenomates and Candidate division OP3, suggesting the enhanced sampling depth allowed the profiling of microbial communities in greater detail. It should be noted that in this study, 4500 sequences were randomly selected per sample to calculate distances between samples, lesser than studies on non-saline soils30. However, the coverage ranged from 89.56% (site D) to 99.82% (site I), indicating that the sequencing depth was adequate for the purpose of this study. This was also congruent with previous studies investigating bacterial community structure in saline soil (vegetated area, 1430 sequences per sample5) and in saline sediments (4000 sequences per sample17), which had similar sequencing depth yet obtained high coverage. It is possible that salinity resulted in lower bacterial abundance in these harsh environments.
While the effect of salt concentration on microbial community structure is a pertinent issue for soil ecological studies, the effect is still unclear due to contrasting evidences5,26. For example, significant correlation was observed between bacterial communities and salt concentrations in sediments globally26, whereas salt was not a major factor shaping bacterial structures in hypersaline sediments and soils5. Thus, there is still an active debate about the relative contribution of salt to bacterial community dissimilarity in soils. Here, we used a phylogenetic approach to compare more subtle differences among the soil samples. Consequently, we found that the bacterial community compositions, phylotype richness and phylogenetic diversity were primarily correlated with salt. The results support our hypothesis and further emphasize the notion that soil salt plays a key role in shaping the bacterial community structure in saline soils.
Indicator taxa varied among saline soils; close relatives for many OTUs could be identified, yet some remained unidentified. Some indicator species were well known, for example, the genus Pseudomonas containing hundreds of described species with potential for biotechnological applications. Similarly, members of the genus Halomonas have been isolated from saline environments (https://www.ezbiocloud.net/taxonomy)31. In contrast, we know little about the indicator taxon related to genera Smithella and Comamonas (https://www.ezbiocloud.net/taxonomy)31. Previous studies showed that indicators in fungal communities are regulated by resource type and availability32,33, and thus, the variation of indicator species in this study suggests that bacterial communities might also be regulated by other soil characters beside salt content.
Bacterial taxonomic richness and phylogenetic diversity both exhibited a nonlinear pattern with increasing soil salinity in the present study. Such observation was beyond our expectations because high osmotic pressure would bring adverse effect to most bacteria. Previous work found that microbial community richness and evenness decreased along increasing waterlogged and salt-rich soils and sediments, however, the highest bacterial diversity and richness observed in the mid-level salt concertation5. Similar negative correlations between salinity and bacterial abundances have also been reported in sediments from Pearl River to coastal South China Sea, while groups such as Thaumarchaeota increased with salinity26. In the present study, bacterial taxonomic richness and phylogenetic diversity enriched at intermediate salt rates. Moreover, the correlation between dominant phyla and salt was similar to the correlation between these phyla and pH or nutrient content. These results suggest that the saline environment is a complex system, salinity plays a critical role in structuring bacterial communities and selecting stress tolerant groups. Although bacterial communities occurring along our study transect were mainly determined by their tolerance or affinity for salt, other soil parameters such as soil pH or nutrient content might have also contributed to the bacterial community compositions in saline soils.
The importance of soil pH for bacterial community structure is widely regarded. Recent studies demonstrated pH as the driver of soil bacterial communities across North and South America14,34,35, in the Arctic15, in alkaline lake sediments across the Tibetan Plateau17, across British soils36, in Changbai Mountain soils18 and in the black soils of northeast China29. Our multiple analyses also revealed that pH had highly significant correlations with bacterial diversity and it was the main factor shaping bacterial community composition, phylotype richness and phylogenetic diversity. Typically, salt content and pH have collinearity in alkaline soils (salt mainly consisted of carbonates and pH ranges from 9.5 to 10.5). In our study, the salt primarily consisted of chlorides or/and sulphates and there was no clear relationship between salt content and pH. Taken together, these findings imply that pH is an equally important driver of bacterial community structure as salinity.
The distribution patterns of some specific bacterial phyla across pH gradient differed from their pH responses observed in previous studies. For example, the relative abundance of Acidobacteria and Bacteroidetes has often been observed to increase towards lower pH in various soils14,15,16,18, but the relative abundance of these two phyla decreased with a decreasing of pH in the current study. Furthermore, the relative abundance of Alphaproteobacteria was significantly correlated with soil pH in other studies15,18, but these trends were not observed in our study. Additionally, Gammaproteobacteria was positively correlated with soil pH in black soils29, but this phylum was negatively correlated with higher soil pH in this study. These findings indicate that the impact of soil pH on the distribution of bacterial groups in saline soils differs from other soils. However, as stated by Rousk et al., we still do not know whether soil pH itself is the factor directly structuring bacterial communities or if it is indirectly integrated with many environmental factors16. Thus, the importance of soil pH in determining bacterial communities does not completely nullify the effect of other local features in driving the individual bacterial distribution.
Strong correlations between microbial community and nutrients such as soil carbon or nitrogen contents and phosphorus contents, as well as other characters like soil EC, were found in studies across various scales. For example, soil organic carbon and TN significantly affected the relative abundance of different dominant or subdominant phyla from a wide range of ecosystems15,37,38,39. Furthermore, soil EC was a dominant factor that drove the bacterial community composition in secondary saline soils40. Indeed, we found that soil EC, OM, AK, TN, NH4+–N and NO3−–N had significant correlations with relative abundances of five dominant taxa. In particular, to the best of our knowledge, the present study is the first report to show a significant relationship between bacterial phyla and AK. We speculate that K+ ions are critical to some bacteria (e.g. Betaproteobacteria, Firmicutes) as proton replacements to cope with high external Na+. Interestingly, several bacterial groups such as Deltaproteobacteria, Chloroflexi, Gemmatimonadetes and Planctomycetes showed negative correlations with OM or NH4+–N, whereas Cyanobacteria was positively correlated with OM and TN in this study (Supplementary Table S3), and those relationships had not been previously discerned. These results suggest that nutrients that could influence the competition between species become more important for particular bacteria at local scales in salt-rich systems. We infer that the relationships between individual bacterial groups and these soil nutrient factors are likely contributing to the overall soil bacterial community structure.
Given the pivotal role of soil moisture in structuring microbial communities in various soils10,29, an influence of moisture on bacterial communities was also expected in these saline soils. However, there was no significant correlation between soil moisture and bacterial diversity. Since the saline soils were sampled from vegetated shoreline, we speculate that the intensity of soil moisture gradient was not strong enough to affect bacterial communities.
It is noteworthy that soil bacterial community structure may not be directly influenced by vegetation type at a landscape scale41, but vegetation may regulate bacterial communities by altering the soil physical environment and by modifying the quantity and quality of substrate supply (e.g. C or N source)42,43. In this study, although we sampled the soil from vegetation in which at least one of the three halophytic species was common, vegetation type might have indirectly affected bacterial distribution through altering the soil C and N status. Similarly, the growth and/or biomass of the vegetation might also be important factors influencing bacterial communities. Given the limited inference space of our study, it is clear that we still have much to learn about bacterial communities and their relationships with edaphic factors in these saline soils, and to determine if our results are consistent in similar saline environments. For example, influence of soil salinity might be more important at a larger scale than at local scale, and as a result the bacterial diversity might exhibit monotonic decreasing trend with salt content. Moreover, soils in sample sites I and J in our study were covered by gravel and soil depth was less than 5 cm (at 0–15 cm depth). These sites are located in a pass between two mountain peaks (Altau and Maili), which is a well-known wind gap in China. Strong wind can cause serious soil erosion affecting the diversity and abundance of soil organisms44. Thus, it is possible that soil erosion caused by wind might be an important factor leading to reduced diversity in these two sites (Fig. 3). Further work is necessary to explore the consistency of such observations.
Overall, we demonstrated bacterial community characteristics in the saline soils under halophytic vegetation around Ebinur Lake. We showed that Gammaproteobacteria and Actinobacteria were the dominant phyla in this saline environment. Bacterial communities were determined by both salt content and pH, and the effect of salt content was similar to that of the soil pH. Other environmental factors might also have promoted unique bacterial communities in the saline soil. To our knowledge, this is the first study to reveal bacterial phyla were significantly related to AK. This study presented an overview of bacterial communities at the local scale in saline soils, however, it is clear that we have only just scratched the surface. As sequencing technologies continue to advance, future studies will employ latest methods such as Single Molecule Real Time sequencing as well as culture-based approaches to reveal patterns of microbial communities and putative drivers in saline soils.
Materials and Methods
Soil sampling
Ebinur Lake (43°380–45°520 N; 79°530–85°020E) is the largest Salt Lake in Xinjiang, China. The dry salt lakebed and the surrounding region become the salt accumulation centers to form unique salt deserts where the lacustrine facies consist of abundant salt deposits. Briefly, the halophytic vegetation mainly exists in the west shoreline, and the vegetation included Halocnemum strobilaceum, Haloxylon ammodendron, Tamarix spp., Kalidium foliatum, Halostachys caspica and Salicornia europaea. Sparse patches of bulrush (Phragmites communis) were also present. Due to strong evaporation, an ice-like layer of salt was formed on the surface of the soil at some points with high salt content.
Soil samples were collected from north to south across a wide area around Ebinur Lake from 24 to 26 September 2015 (see Supplementary Fig. S1). Soils were collected at ten sites in total. At each site, three 100 m2 areas were randomly selected. At each area, soils (0–15 cm) were collected at ten sampling points using a sterile blade and mixed to obtain one composite sample. This resulted in 30 soil samples in total. Soils were sampled at similar locations below heath halophytic vegetation in which at least one of the following plant species was common: Halocnemum strobilaceum, Salicornia europaea or Halostachys caspica. Solid salt crusts were removed before sampling. The composited soil samples were sieved through a 2-mm mesh to remove the plant residues and stones. A portion of each soil sample was put into a 50 mL centrifuge tube, and transferred to laboratory by using ice-box. The tubes were stored at −80 °C until soil DNA extraction. The remaining soils were tested physicochemical properties.
Soil physicochemical property determination
Soil soluble salt content was determined after drying residue; soil moisture was detected after oven drying at 105 °C for 24 hours; available phosphorus was measured by using spectrophotometer (UVmini-1240, Shimadzu, Japan); available potassium was determined by using atomic absorption spectrum analyzer (240-AA, Agilent, USA)45. Soil electrical conductivity (EC) was detected by using electrolytic conductivity meter (soil:water = 1:5). Soil pH was measured by using pH meter after shaking the soil/water (1:5, w/v) suspension for 30 min. Soil total carbon (TC) and total nitrogen (TN) were measured by CN Analyzer (Vario Max CN, Elementar, Germany). Mineral N was determined by using Skalar SAN plus Segmented Flow Analyzer (Skalar Analytic B.V., De Breda, the Netherlands). Ion concentrations were detected by using inductively coupled plasma mass spectrometry (ICP-MS) (ELAN 9000/DRC-e, PerkinElmer, USA).
Barcoded pyrosequencing
Bacterial 16S rRNA gene targeted at the hypervariable V1–V3 region was amplified by PCR using a 10-nucleotide barcoded forward primer 27 F (5′-AGA GTT TGA TCC TGG CTC AG-3′) and the reverse primer 533 R (5′-TTA CCG CGG CTG CTG GCA C-3′) for high-throughput 454 GS-FLX pyrosequencing29. PCR reactions were performed in a 25 μL mixture containing 1 μL of each primer at 10 μ mol L−1, 2 μL template DNA (20 ng μL−1) and 22 μL Platinum PCR SuperMix (Invitrogen, Shanghai, China). The following thermal program was used for amplification: 94 °C for 4 min, followed by 26 cycles of 94 °C for 30 s, 50 °C for 45 s and 72 °C for 1 min, followed by a final extension at 72 °C for 7 min. An equal amount of the PCR product from each sample was run on a Roche FLX 454 pyrosequencing machine at Personalbio Technology Co., Ltd., Shanghai, China.
Processing the pyrosequencing data
The raw FASTQ data were processed using QIIME Pipeline Version 1.8.0 (http://qiime.org/tutorials/tutorial.html)46. The paired reads were joined with FLASH (fast length adjustment of short reads) software47. Briefly, only sequences >200 bp in length, with an average quality score >25 and without ambiguous base calls were reserved in the subsequent analysis. The trimmed sequences were chimera-detected and removed using the Uchime algorithm48. The high-quality sequences were clustered through the UPARSE pipeline (http://drive5.com/uparse/) at a 97% similarity level to generate operational taxonomic units. A representative sequence from each phylotype was aligned using the Python Nearest Alignment Space Termination (PyNAST) tool46,49 with a relaxed neighbor-joining tree built using FastTree50. The taxonomy of each phylotype was determined using the Ribosomal Database Project (RDP) classifier with a confidence threshold of 0.80 (http://rdp.cme.msu.edu/)51. To correct for survey effort, we used a randomly selected subset of 4500 bacterial sequences per sample for calculating both α-diversity and β-diversity analyses to compare the differences and similarities between samples. All sequences have been deposited in the GenBank short-read archive (Sequence Read Archive; accession no. SRX2730198).
Statistical analysis
The diversity indices of Faith’s index of phylogenetic diversity (Faith’s PD)52 and Chao 1 index53 were used to compare soil bacterial α-diversity. Spearman’s correlation coefficient was used to reveal the possible correlations between the abundances of different taxonomic levels of bacteria, diversity and soil characteristics, which were calculated using the IBM Statistical Product and Service Solutions (SPSS) Statistics package for Windows (version 18.0, NY, USA). Pairwise UniFrac distances calculated for the total community analyses were visualized using non-metric multidimensional scaling (NMDS) plots as implemented in PRIMER v654. The Cluster analysis of the bacterial communities based on the NMDS dissimilarity matrix was applied using the ‘picante’ and ‘vegan’ packages in the R environment. Indicator species were determined by the Dufrene-Legendre indicator species analysis method to identify OTUs that are specifically associated with the different salinity regimes33,55. SIMFER analysis was performed to determine the respective contribution of bacterial communities to between-treatment variations. Mantel tests, SIMFER and CCA analysis were performed using the ‘vegan’ package in the R environment (R v.3.3.1)56.
References
Rozema, J. & Flowers, T. Crops for a salinized world. Science 322, 1478–80 (2008).
Fan, Z. & Ma, Y. J. Salinized soils and their improvement and utilization in west China. Arid Zone Res. 18, 1–6 (2001).
Caton, T. M. et al. Halotolerant aerobic heterotrophic bacteria from the great salt plains of Oklahoma. Microb. Ecol. 48, 449–462 (2004).
Ventosa, A., Mellado, E., Sanchez-Porrro, C. & Marquez, M.C. Halophilic and halotolerant micro-organisms from soils in Microbiology of Extreme Soils (eds Dion, P. & Nautiyal, C. S.) 87–115 (Springer-Verlag: Berlin, 2008).
Hollister, E. B. et al. Shifts in microbial community structure along an ecological gradient of hypersaline soils and sediments. ISME J. 4, 829–838 (2010).
Chaparro, J. M., Sheflin, A. M., Manter, D. K. & Vivanco, J. M. Manipulating the soil microbiome to increase soil health and plant fertility. Biol. Fert. Soils 48, 489–499 (2012).
Ferris, H. & Tuomisto, H. Unearthing the role of biological diversity in soil health. Soil Biol. Biochem. 85, 101–109 (2015).
van der Heijden, M. G. A., de Bruin, S., Luckerhoff, L., van Logtestijn, R. S. P. & Schlaeppi, K. A widespread plant-fungal-bacterial symbiosis promotes plant biodiversity, plant nutrition and seedling recruitment. ISME J. 10, 389–399 (2016).
Graham, E. B. et al. Microbes as engines of ecosystem function: when does community structure enhance predictions of ecosystem processes? Front. Microbiol. 7, 214 (2016).
Banerjee, S. et al. Legacy effects of soil moisture on microbial community structure and N2O emissions. Soil Biol. Biochem. 95, 40–50 (2016).
Hansel, C. M., Fendorf, S., Jardine, P. M. & Francis, C. A. Changes in bacterial and archaeal community structure and functional diversity along a geochemically variable soil profile. Appl. Environ. Microb. 74, 1620–1633 (2008).
Sun, R., Guo, X., Wang, D. & Chu, H. Effects of long-term application of chemical and organic fertilizers on the abundance of microbial communities involved in the nitrogen cycle. Appl. Soil Ecol. 95, 171–178 (2015).
Banerjee, S. et al. Network analysis reveals functional redundancy and keystone taxa amongst bacterial and fungal communities during organic matter decomposition in an arable soil. Soil Biol. Biochem. 97, 188–198 (2016).
Lauber, C. L., Hamady, M., Knight, R. & Fierer, N. Pyrosequencing-based assessment of soil pH as a predictor of soil bacterial community structure at the continental scale. Appl. Environ. Microb. 75, 5111–5120 (2009).
Chu, H. et al. Soil bacterial diversity in the Arctic is not fundamentally different from that found in other biomes. Environ. Microbiol. 12, 2998–3006 (2010).
Rousk, J. et al. Soil bacterial and fungal communities across a pH gradient in an arable soil. ISME J. 4, 1340–1351 (2010).
Xiong, J. et al. Geographic distance and pH drive bacterial distribution in alkaline lake sediments across Tibetan Plateau. Environ. Microbiol. 14, 2457–2466 (2012).
Shen, C. et al. Soil pH drives the spatial distribution of bacterial communities along elevation on Changbai Mountain. Soil Biol. Biochem. 57, 204–211 (2013).
Humayoun, S. B., Bano, N. & Hollibaugh, J. T. Depth distribution of microbial diversity in Mono lake, a meromictic soda lake in California. Appl. Environ. Microb. 69, 1030–1042 (2003).
Demergasso, C. et al. Distribution of prokaryotic genetic diversity in athalassohaline lakes of the Atacama Desert, Northern Chile. FEMS Microbiol. Ecol. 48, 57–69 (2004).
Jiang, H. et al. Microbial diversity in water and sediment of lake Chaka, an athalassohaline lake in Northwestern China. Appl. Environ. Microb. 72, 3832–3845 (2006).
Mutlu, M. B. et al. Prokaryotic diversity in Tuz lake, a hypersaline environment in inland Turkey. FEMS Microbiol. Ecol. 65, 474–483 (2008).
Dong, H. L. et al. Microbial diversity in sediments of saline Qinghai lake, China: linking geochemical controls to microbial ecology. Microb. Ecol. 51, 65–82 (2006).
Jiang, H. et al. Microbial response to salinity change in Lake Chaka, a hypersaline lake on Tibetan plateau. Environ. Microbiol. 9, 2603–2621 (2007).
Mesbah, N. M., Abou-El-Ela, S. H. & Wiegel, J. Novel and unexpected prokaryotic diversity in water and sediments of the alkaline, hypersaline lakes of the Wadi An Natrun, Egypt. Microb. Ecol. 54, 598–617 (2007).
Xie, W., Zhang, C., Zhou, X. & Wang, P. Salinity-dominated change in community structure and ecological function of Archaea from the lower Pearl River to coastal South China Sea. Appl. Microbiol. Biot. 98, 7971–7982 (2014).
David, A. W., Papke, R. T. & Doolittle, W. F. Archaeal diversity along a soil salinity gradient prone to disturbance. Environ. Microbiol. 7, 1655–1666 (2005).
Lozupone, C. A. & Knight, R. Global patterns in bacterial diversity. P. Natl. Acad. Sci. USA 104, 11436–11440 (2007).
Liu, J. et al. High throughput sequencing analysis of biogeographical distribution of bacterial communities in the black soils of northeast China. Soil Biol. Biochem. 70, 113–122 (2014).
Thompson, L. R. et al. A communal catalogue reveals Earth’s multiscale microbial diversity. Nature. https://doi.org/10.1038/nature24621 (2017).
Kim, O. S. et al. Introducing EzTaxon-e: a prokaryotic 16S rRNA gene sequence data- base with phylotypes that represent uncultured species. Int. J. Syst. Evol. Microbiol. 62, 716–721 (2012).
Moll, J. et al. Resource type and availability regulate fungal communities along arable soil profiles. Microb. Ecol. 70, 390–399 (2015).
Sun, R. et al. Fungal community composition in soils subjected to long-term chemical fertilization is most influenced by the type of organic matter. Environ. Microbiol. 18, 5137–5150 (2016).
Fierer, N. & Jackson, R. B. The diversity and biogeography of soil bacterial communities. P. Natl. Acad. Sci. USA 103, 626–631 (2006).
Banerjee, S. et al. Determinants of bacterial communities in Canadian agroforestry systems. Environ. Microbiol. 18, 1805–1816 (2016).
Griffiths, R. I. et al. The bacterial biogeography of British soils. Environ. Microbiol. 13, 1642–1654 (2011).
Fierer, N., Bradford, M. A. & Jackson, R. B. Toward an ecological classification of soil bacteria. Ecology 88, 1354–1364 (2007).
Ramirez, K. S., Lauber, C. L., Knight, R., Bradford, M. A. & Fierer, N. Consistent effects of N fertilization on soil bacterial communities in contrasting systems. Ecology. 91, 3463–3470 (2010).
Zeng, J. et al. Nitrogen fertilization directly affects soil bacterial diversity and indirectly affects bacterial community composition. Soil Biol. Biochem. 92, 41–49 (2016).
Shen, W. et al. Bacterial community composition is shaped by soil secondary salinization and acidification brought on by high nitrogen fertilization rates. Appl. Soil Ecol. 108, 76–83 (2016).
Nielsen, U. N., Osler, G. H. R., Campbell, C. D., Burslem, D. F. R. P. & van der Wal, R. The influence of vegetation type, soil properties and precipitation on the composition of soil mite and microbial communities at the landscape scale. J. Biogeog. 37, 1317–1328 (2010).
Wallenstein, M. D., McMahon, S. & Schimel, J. Bacterial and fungal community structure in Arctic tundra tussock and shrub soils. FEMS Microbiol. Ecol. 59, 428–435 (2007).
Chu, H., Neufeld, J. D., Walker, V. K. & Grogan, P. The influence of vegetation type on the dominant soil bacteria, archaea, and fungi in a low Arctic tundra landscape. Soil Sci. Soc. Am. J. 75, 1756–1765 (2011).
Pimentel, D. & Kounang, N. Ecology of soil erosion in ecosystems. Ecosystems 1, 416–426 (1998).
Bao, S.D. Soil agro-chemistrical analysis. 201–400 (China Agriculture Press: Beijing, 2008).
Caporaso, J. G. et al. QIIME allows analysis of high-throughput community sequencing data. Nat. Methods 7, 335–336 (2010).
Magoč, T. & Salzberg, S. L. FLASH: fast length adjustment of short reads to improve genome assemblies. Bioinformatics 27, 2957–2963 (2011).
Edgar, R. C., Haas, B. J., Clemente, J. C., Quince, C. & Knight, R. UCHIME improves sensitivity and speed of chimera detection. Bioinformatics 27, 2194–2200 (2011).
DeSantis, T. Z. et al. NAST, a multiple sequence alignment server for comparative analysis of 16S rRNA genes. Nucleic Acids Res. 34, 394–399 (2006).
Price, M. N., Dehal, P. S. & Arkin, A. P. FastTree: computing large minimum evolution trees with profiles instead of a distance matrix. Mol. Biol. Evol. 26, 1641–1650 (2009).
Cole, J. R. et al. The Ribosomal Database Project RDP-II: sequences and tools for high-throughput rRNA analysis. Nucleic Acids Res. 33, D294–D296 (2005).
Faith, D. P. Conservation evaluation and phylogenetic diversity. Biol. Conserv. 61, 1–10 (1992).
Chao, A. Non-parametric estimation of the number of classes in a population. Scand. J. Stat. 11, 265–270 (1984).
Clarke, K. R. & Warwick, R. M. A further biodiversity index applicable to species lists: variation in taxonomic distinctness. Mar. Ecol. Prog. Ser. 216, 265–278 (2001).
Dufrene, M. & Legendre, P. Species assemblages and indicator species: the need for a flexible asymmetrical approach. Ecol. Monogr. 67, 345–366 (1997).
R Development Core Team. R: a Language and Environment for Statistical Computing. R Foundation for Statistical Computing, Vienna, Austria (2006).
Acknowledgements
This work was supported by the Foundation of Chinese Academy of Sciences (Nos. 2015-XBQN-B-16, KFJ-SW-STS-141 and TSS-2015-014-FW-3-1) and the Foundation of Science & Technology Department of Xinjiang Uygur Autonomous Region (No. QN2015BS017), and by the National Natural Science Foundation of China (Nos 41671251 and 31300432) and National Key R&D Plan (2016YFC0501400). We thank the editor and two reviewers for their constructive suggestions and insightful comments.
Author information
Authors and Affiliations
Contributions
C.Y.-Tian conceived and designed the experiments. S. Zhao and J.-J. Liu wrote the paper. C.Y.-Tian, J.-J. Liu and S Banerjee revised the manuscript. N. Zhou and Z.Y. Zhao conducted the experiments. K. Zhang performed data analysis.
Corresponding authors
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zhao, S., Liu, JJ., Banerjee, S. et al. Soil pH is equally important as salinity in shaping bacterial communities in saline soils under halophytic vegetation. Sci Rep 8, 4550 (2018). https://doi.org/10.1038/s41598-018-22788-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-22788-7
This article is cited by
-
Response of the metabolic activity and taxonomic composition of bacterial communities to mosaically varying soil salinity and alkalinity
Scientific Reports (2024)
-
Low and Moderate Rice-Sodic Soils Affect Bacterial Diversity and Their Functions
Journal of Soil Science and Plant Nutrition (2024)
-
Exploring the application of signaling compounds and soil amendments to modulate plant–microbe interactions for improved plant salinity tolerance
Plant and Soil (2024)
-
Epichloë endophytes alter the soil microbial communities and soil metabolite profile after their host decomposition under salt stress
Plant and Soil (2024)
-
Leaf stoichiometry of Leontopodium lentopodioides at high altitudes on the northeastern Qinghai-Tibetan Plateau, China
Journal of Arid Land (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.







