Abstract
The oxygen (18O/16O) isotope analysis of hair is commonly applied to reconstruct an individual’s residence history. However, region-of-origin as determined from oxygen isotope values (δ18O) alone is often spatially indistinct. Adding additional geochemical recorders can refine region-of-origin estimates. In this capacity, strontium (87Sr/86Sr) isotope analysis has attracted increased interest. While 87Sr/86Sr reflects the influences of local geology, 87Sr/86Sr of hair includes both external environmental signals as well as the internal dietary indicators. To better understand the impact of these contributions to the spatial signal encoded within 87Sr/86Sr of hair, human hair was collected from three locations within Salt Lake City, Utah along with the donor’s sex. The 87Sr/86Sr and δ18O of hair and local tap water were measured. There were no significant relationships between sex and either δ18O or 87Sr/86Sr of hair, nor between collection location and the δ18O of hair. However, we found significant associations between collection location and 87Sr/86Sr of hair. These findings suggest that interactions with local water may be an important source of Sr to human hair and that the 87Sr/86Sr of hair may have the capacity to record differences in 87Sr/86Sr of tap waters on small spatial scales.
Similar content being viewed by others
Introduction
The stable oxygen (18O/16O) isotope values (δ18O) of human tissues can be utilized to identify and reconstruct an individual’s region-of-residence or origin as δ18O of human tissue relates largely to the δ18O of an individual’s drinking water, which varies with geography1. Numerous studies have applied the δ18O of human tissues to determine the likely origin of modern, historic, and prehistoric humans2,3,4,5,6,7,8,9. However, predictions of origin using δ18O can be geographically broad10,11. Similar to δ18O, variations in the strontium (Sr) isotope ratios (87Sr/86Sr) relate to geography and the analysis of 87Sr/86Sr in human tissues has been utilized to address many historical6,12,13,14 and prehistorical human provenance questions14,15,16,17,18. With 87Sr/86Sr, the age and geology of the underlying bedrock control the 87Sr/86Sr in soils, waters, plants, and animals19,20,21, which are incorporated into human tissues22. Thus, combining the independent δ18O and 87Sr/86Sr systems may allow for greater resolution in human provenance predictions.
Measurements of both δ18O and 87Sr/86Sr in tooth enamel have been successfully applied in several modern23,24 and prehistorical applications25 to identify the probable locations where an individual spent their early life. The measurement of δ18O and 87Sr/86Sr in hydroxylapatite tissues from humans is straightforward with tooth enamel having an average of 544 μg g−1 Sr26. Where tooth enamel represents early periods in an individual’s life, the isotope values of hair keratin represent much more recent intervals of time. Thus, the combined measurement of δ18O and 87Sr/86Sr in hair is emerging as a very attractive approach to understand an individual’s very recent life history27. This combination of isotopes has many potential applications, especially recognizing known variations in tap waters that exist within and among metropolitan regions28,29,30. However, while the δ18O systematics in keratin is relatively well understood and commonly applied1, the Sr isotopic system in human hair is not22.
The various means of Sr incorporation into internal human tissues are ingestion of food and beverages in addition to inhalation of aerosols and particles31,32; however, Sr in hair – an external tissue – represents a complex mixture of both these endogenous and exogenous sources of Sr33. Endogenous sources of Sr to hair originate from the body’s Sr pools within blood and bones31,32, while exogenous sources represent external environmental influences from aerosols, particulates, and environmental waters27,33,34. The overwhelming importance of exogenous contributions is evidenced by elevated Sr concentrations in the cortex of the hair35,36 as well as continual increases in Sr concentration from the proximal to distal terminus of the hair27,37. Chemical separation and isolation of the exterior and interior Sr pools demonstrated that these inputs may impart unique 87Sr/86Sr ratios and bias the overall 87Sr/86Sr value34. Distinguishing the impact of these two potentially conflicting Sr sources on the overall 87Sr/86Sr value is required to fully utilize 87Sr/86Sr of hair for the reconstruction of region-of-residence and remains a fundamental question in the field.
To begin to distinguish the importance of endogenous and exogenous Sr to hair and create a better understanding of how these sources of Sr relate to the spatial signal encoded within 87Sr/86Sr of hair, we collected and analyzed the 87Sr/86Sr and δ18O of human hair from three locations within a single city (Salt Lake City, Utah, USA). In addition to 87Sr/86Sr and δ18O, the sex of the hair donors were also gathered, as well as, the 87Sr/86Sr and δ18O of tap waters collected from six sites near hair collection locations. Our aim was to explore the relationships of 87Sr/86Sr and δ18O of hair at the city-scale and to begin to develop a better understanding of the geospatial signal encoded in the 87Sr/86Sr of hair.
Results
Oxygen isotope values of hair
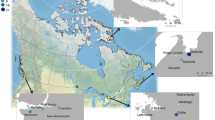
Sixty-seven hair samples were obtained from students from three public schools (Fig. 1). We analyzed thirty-one hair samples for δ18O and found hair samples ranged in δ18O from 6.7 to 12.8‰ with a mean δ18O of 9.9 ± 0.8‰ (1σ, n = 31). The mean δ18O of hair samples from females was 9.8 ± 1.4‰ (1σ, n = 28), while the mean δ18O of hair samples from males was 10.3 ± 0.4‰ (1σ, n = 3) (Fig. 2). While our dataset was weighed toward female participants, the δ18O of hair from females was not significantly different than δ18O of hair from males (Student’s t-test, p = 0.5370). When grouped by collection location, we found the mean δ18O of hair samples from individuals at Schools X, Y, and Z were 9.7 ± 1.8‰ (1σ, n = 10), 10.0 ± 1.5‰ (1σ, n = 7), and 10.0 ± 0.9‰ (1σ, n = 14), respectively (Fig. 2). Here, the δ18O of hair from individual schools were not significantly different from one another (one-way ANOVA, p = 0.8438).
Location map showing the collection locations of hair and tap water. Salt Lake City is highlighted. School locations are shown with flagged symbols and tap water collection locations are shown with points. Point shading indicates groupings with group A sites shown in grey, group B sites shown in black, and the group C site shown in white. Figure produced using ArcGIS 10.4 (ESRI; Redlands, CA, USA).
Oxygen isotope values of water
Water samples were collected from taps in six public buildings four times a year for four years (Fig. 1). The mean δ18O of collected tap water was −16.3 ± 0.2‰ (1σ, n = 66) with a range from −15.3 to −16.8‰ (Table 1). Following Ehleringer et al.1, the δ18O of drinking water predicted from measured δ18O of hair ranged between −12.5‰ to −17.0‰ with a mean δ18O of −14.8 ± 3.7‰ (1σ, n = 31) (Fig. 3). Here, we found the measured δ18O of tap water was not significantly different than δ18O of drinking water predicted from hair (Welch’s t-test, p = 0.0707).
Strontium abundance of hair
Sixty-seven hair samples were analyzed for Sr elemental abundance [Sr]. We found [Sr] ranged from 0.08–27.5 μg g−1, while the mean [Sr] of hair samples from female and male participants was 7.8 ± 5.0 μg g−1 (1σ, n = 60) and 3.7 ± 3.5 μg g−1 (1σ, n = 7), respectively. [Sr] of hair samples from female participants was significantly higher than [Sr] of hair from males (Welch’s t-test, p = 0.0003). The mean [Sr] of hair samples from students at Schools X, Y, and Z were 7.4 ± 4.7 μg g−1 (1σ, n = 11), 8.1 ± 4.8 μg g−1 (1σ, n = 29), and 6.7 ± 4.0 μg g−1 (1σ, n = 27), respectively. We did not find any significant differences in [Sr] of hair samples from collection locations (one-way ANOVA, p = 0.5510).
Strontium isotope ratios of hair
Forty-one hair samples were measured for 87Sr/86Sr and ranged from 0.70910 to 0.71509 (Fig. 4). The mean 87Sr/86Sr of hair samples from female and male participants was 0.71215 ± 0.00193 (1σ, n = 37) and 0.71150 ± 0.00139 (1σ, n = 4), respectively. Here, we observed the 87Sr/86Sr of hair from female and male students were not significantly different (Student’s t-test, p = 0.5210). The mean 87Sr/86Sr of hair samples from students at Schools X, Y, and Z were 0.70965 ± 0.00028 (1σ, n = 3), 0.71342 ± 0.00152 (1σ, n = 18), and 0.71125 ± 0.00142 (1σ, n = 20), respectively. These differences in 87Sr/86Sr of hair samples at individual schools were significantly different from one another (one-way ANOVA, p < 0.0001).
Strontium isotope ratios of water
Sixty-one tap water samples from six collection sites were analyzed for 87Sr/86Sr; they ranged from 0.70906 to 0.71399 (Table 1). Tap water samples were grouped based on proximity to hair sampling locations into Site Groups. The mean 87Sr/86Sr of tap water samples from Sites 1, 2, and 3 (Site Group A), Sites 4 and 5 (Site Group B), and Site 6 (Site Group C) were 0.71028 ± 0.00028 (1σ, n = 29), 0.71192 ± 0.00181 (1σ, n = 20), and 0.71014 ± 0.00060 (1σ, n = 12), respectively (Fig. 4). We found the mean 87Sr/86Sr of Site Groups were significantly different from one another (one-way ANOVA, p < 0.0001).
Discussion
Oxygen isotope analysis of human hair is becoming a relatively common tool in determining the region-of-origin of modern humans3,5,38. Here, we observed the δ18O of hair from individuals to be indistinguishable at the sites sampled in Salt Lake City, Utah, likely because the origins of urban tap waters share a common montane origin29,39. These data are consistent with previous reports of the δ18O of hair from humans and animals living in or near this region1,27. Previously, Ehleringer, et al.1 found that the average δ18O of human hair from individuals living in Utah was 9.7 ± 1.5‰ (1σ, n = 12). When compared to our dataset, we found no significant difference between our data and these previous data (Student’s t-test, p = 0.6478). The δ18O of hair has been linked to the δ18O of drinking water in humans40,41,42 and to assess the relationship between individuals and their drinking water, we translated the δ18O of hair to δ18O of drinking water following Ehleringer, et al.1. Consistent with expectations, the δ18O of drinking water predicted from measured δ18O of hair was not significantly different than measured δ18O of tap water. This is in agreement with previous studies that indicate the δ18O of an individual’s hair largely relates to the δ18O of their drinking water, and thus the environment and community in that they reside3,43,44,45. Further, it is not unexpected that there were no differences in the δ18O of hair from these individuals (Figs 2 and 3), given the relatively small range of δ18O of tap water within the regions where these individuals were residing.
Studies of the δ18O of tap water have described spatial and temporal dynamics within single metropolitan areas, including in those around Salt Lake City28,29,30. These dynamics may confound the application of δ18O of human hair as a geographic predictor, particularly within regions that utilize transported water46,47. Within the Salt Lake metropolitan area, Jameel et al.29 showed that different regions within the study area utilized different management strategies to supply culinary water, resulting in subtle, but unique δ18O values of tap water across the area. During the three studied seasons, Jameel et al.29 found the δ18O of tap water delivered to Salt Lake City had a relatively small range of isotope values (i.e., −16.5 to −14.0‰), consistent with our findings. This was not unexpected as all waters delivered to Salt Lake City ultimately originated from cold season, high elevation precipitation29,39. However, the utility supplying Salt Lake City utilized both surficial and groundwater sources depending on season and supply level48. While surficial and groundwater sources would have similar δ18O values given their common source, other geochemical tracers likely vary due to water-rock interactions and other processing. If there are underlying geographic controls of the supply of groundwater to an urban region (e.g., proximity to groundwater wells, etc.), then additional geochemical or isotopic tracers may be useful to uncover intra-city scale variations in water supply and possibly chemical signatures within the individuals residing within these regions.
Strontium in human tissue is generally considered a conservative tracer of geographic origin22, however, in human hair the [Sr] and 87Sr/86Sr are controlled by both diet and environmental contamination33,34. The endogenous and exogenous Sr pools in human hair may have unique or quite similar 87Sr/86Sr depending on the Sr source and either of these pools may or may not preferentially influence the final Sr isotope value34,49. Multiple methods have been developed to potentially isolate endogenous and exogenous Sr for dietary and provenancing studies in humans, yet no consensus has been reached on the preferred methodology34,36,50,51,52,53,54,55,56. Regardless of these known uncertainties, the 87Sr/86Sr of human and animal hair have been shown to trace movement histories27,37,57. However, it remains unclear if the geographic signal within 87Sr/86Sr in hair relates to the endogenous or exogenous Sr signature, or some combination of both.
Here, we found the range of [Sr] in hair was in good agreement with previous studies of hair from students58,59. We observed [Sr] of hair from female participants was significantly higher than [Sr] of hair from males. This too was consistent with other studies that have found that [Sr] in hair a key element to differentiate females and males58. In previous studies, [Sr] has been linked to sex, with females most often having greater [Sr] than males58,59,60,61,62,63. While the reason(s) behind this pattern remains unclear, it has been suggested that differences in physiology, biochemistry, diet, and/or activity levels may be the causes of these patterns31,33,64. [Sr] is known to increase along the length of hair from proximal to distal portions due to the incorporation of exogenous strontium on and/or into the hair cuticle27,35,65. Given that our dataset is weighed towards female participants and females are more likely to wear their hair longer than males, these differences may be related to female participants’ hair incorporating exogenous strontium for extended periods of time, and thereby increasing the [Sr].
While we observed a difference between [Sr] of hair from females and males, we did not find any significant differences in [Sr] of hair samples from collection locations within Salt Lake City. Given the small geographic area from that the individuals resided and our dataset bias towards female participants, it may not be unexpected that there were no differences in [Sr] of hair samples between the three collection locations.
In contrast to [Sr] and δ18O values of hair, we observed an unexpectedly wide range in the 87Sr/86Sr of hair. Here, we found the 87Sr/86Sr of hair from individuals residing in Salt Lake City varied by more than 0.005. This variation was larger than any previously reported 87Sr/86Sr from hair specimens from individuals residing in the same city or geographic region34,37. In a previous study using hair specimens collected from a single salon in Taylorsville, Utah located less than 10 km south of Salt Lake City (Fig. 1), Tipple and colleagues34 found the 87Sr/86Sr of hair ranged from 0.70909 to 0.71469 with an average of 0.71203 ± 0.00140 (1σ, n = 22)34. Our data were not statistically different than these previous data from Taylorsville, Utah (Student’s t-test; p = 0.9050). Given the constrained geographic area with identical climate and underlying geology, these variations point towards an additional control for the wide range of 87Sr/86Sr of hair observed within this single metropolitan area.
There remains no consensus regarding the dominant geospatial control on the 87Sr/86Sr of hair27,34,37,57. Several studies have assessed the relationship between the 87Sr/86Sr of modern hair and residence with some studies suggesting that the endogenous signal to hair is most important for geospatial relationships57, while others found compelling relationships with exogenous sources, such as water, atmospheric dust, etc.27,37. Unraveling the significant contributions of Sr to hair for geospatial reconstructions is becoming more important in both modern cold case66 and archeological67,68 contexts.
We found the overall range of 87Sr/86Sr of hair and tap water was similar at the locations sampled in Salt Lake City (Fig. 4). Yet, the 87Sr/86Sr of hair from participants varied systematically by collection location within the city (Fig. 4). We noted that participants from School Y had higher 87Sr/86Sr ratios and a much larger range than the other two collection locations. Previously, Vautour and colleagues37 noted that four individuals that traveled from Paris or St. Benoit sur Loire, France to Montreal, Canada showed a transition in 87Sr/86Sr along a length of hair corresponding to the change in location. They found that the 87Sr/86Sr of hair from these individuals converged towards an average value with some slight differences between the four individuals and used this finding as evidence that the Sr in hair is not only controlled by endogenous inputs, but exogenous contributions as well. Furthermore, they noted that the 87Sr/86Sr of hair was similar, but distinct from the 87Sr/86Sr of local tap water and argued that this was due to reworking and buffering of the Sr isotopic signal from Sr incorporated within bone37. Similarly, Font and associates57 argued that the 87Sr/86Sr of hair from two individuals that traveled from Kanpur, India to Amsterdam, The Netherlands slowly reached isotopic equilibrium with the new location due to recycling and incorporation of Sr from bone32. While both dietary Sr and remobilization of bone Sr likely contribute to overall 87Sr/86Sr of hair, the impact of the 87Sr/86Sr of water on hair may not have been fully appreciated in these previous studies. Here, we analyzed sixty-one water samples in a single municipality and found an astonishingly wide range of 87Sr/86Sr ratios. These previous studies analyzed five or fewer tap waters from each of the locations that the individuals resided, and thus, may have not captured all the possible 87Sr/86Sr variability that could exist. Here, we found spatial relationships between the 87Sr/86Sr of hair and tap water from the neighborhoods study participants resided in, suggesting interactions with local tap waters may contribute to the 87Sr/86Sr of hair. While the specific mechanism Sr uptake remains unclear, it may take place through imbibing local tap waters or when the hair is wetted during bathing or showering. While this study cannot specifically distinguish between the incorporation of endogenous or exogenous Sr through interactions with water, previous research has shown that significant amounts of Sr can be added to hair after it exits the scalp and is exposed to the environment33. Thus, we hypothesize that interactions with bathing water may be an important contributor of Sr to hair and that small spatial variations in the 87Sr/86Sr of water are possibly recorded in the 87Sr/86Sr of hair. Geospatial modeling of bioavailable 87Sr/86Sr for human provenancing remains nascent19,20,21,69,70,71 and our results suggest that these modeling exercises should consider additional factors or layers for applications towards modern humans72, including the 87Sr/86Sr of tap waters and the processes by which communities acquire and transport their water resources. Nonetheless, additional controlled dietary studies that consider the 87Sr/86Sr of food, beverages, and external Sr sources will be required to test this hypothesis and establish a mechanism for Sr incorporation from bathing water.
Methods
Ethics statement
The Institutional Review Board (IRB) of the University of Utah approved this research program under protocol number [00035524]. Specifically, all sampling and analytical methods used were in accordance with these IRB regulations. Informed consent was obtained from all subjects or from their legal guardians in accordance with and maintained under IRB regulations.
Hair and water samples
Sixty-seven hair samples were obtained from students from three public schools following Valenzuela, et al.73 (Fig. 1). Schools X and Y are public schools that pull students from defined areas near the school, while School Z is a public magnet school that has students from throughout the public school district. All study participants lived in Salt Lake City, Utah. Participants were assigned a randomized sample identifier and all personal and identifying information was stored according to IRB. Hair samples were placed in paper coin envelopes and labeled with sample identifier. Hair samples were returned to laboratory and stored. In addition to hair samples, participants provided their sex, along with any specific dietary or health information.
Water samples were collected from taps in six public buildings four times a year for four years (summer, 2012 to summer, 2015). Water collection sites were located within a few blocks of hair collection locations (Fig. 1). The plumbing was flushed prior to sample collection by running the cold-water tap for 10 s. Water samples were collected in 4-ml glass vials and the cap seal was with Parafilm®. Water samples were returned to laboratory and stored in 4 °C refrigerator prior to analysis.
Hair cleaning
Hair samples were cleaned with 2:1 chloroform:methanol (v/v). Samples were enclosed in filter paper and completely submerged in the solvents. Samples were agitated using a shaker plate and after 5 min the supernatant was decanted and discarded. This process was repeated a total of three times. Chloroform (OmniSolv®, EMD; Darmstadt, Germany) and methanol (OmniSolv®, EMD; Darmstadt, Germany) used in hair cleaning were HPLC grade. Cleaned hair samples were allowed to dry at room temperature for 72 hrs within a laminar flow hood. After dried, cleaned hair samples were ground in a ball mill and stored in ashed glass vials until further analysis-specific preparation procedures.
Oxygen isotope analysis of hair
Hair samples equilibrated with ambient laboratory atmospheric water vapor for 48 hrs alongside in-house keratin reference materials (RMs) following Chau, et al.27. Hair samples (~150 mg), as well as RMs, were weighed in 3.5 × 5 mm silver capsules (Costech Analytical Technologies, Inc.; Valencia, California, USA). Weighed samples and RMs were stored under vacuum for 7 days before being analyzed. The stable oxygen isotope values of keratin were determined with a continuous flow isotope ratio mass spectrometer (IRMS) (MAT 253, Thermo Finnigan; Bremen, Germany), housed at IsoForensics, Inc. in Salt Lake City, Utah. Samples were introduced to the IRMS via a zero-blank autosampler attached to a high temperature conversion elemental analyzer (TC/EA, ThermoFinnigan; Breman, Germany). Samples were analyzed alongside sets of natural keratin RM for primary quality assurance and secondary quality control (QA/QC). Keratin RMs were previously calibrated to Vienna Standard Mean Ocean Water (VSMOW)-Standard Light Antarctic Precipitation (SLAP) international isotope scale. Sets included two primary QA RMs for slope/intercept normalization and a secondary QC RMs to insure suitable calibration. Primary keratin QA RMs were DS and ORX (δ18O = 6.02‰ and 25.09‰, respectively), while POW was used for QC (long-term mean δ18O = 12.44‰, 1σ = 0.54‰, n = 335). A minimum of four sets of RMs was analyzed along side samples. All δ18O values of hair are expressed relative to VSMOW. All light stable isotope values are reported in parts per thousand (‰) and in delta notation:
where R represents the 18O/16O abundance ratio, and R samp and R std are the ratios in the sample and standard, respectively.
Oxygen isotope analysis of water
Water samples were analyzed using cavity ring-down water isotope spectroscopy (L1102-I, Picarro; Sunnyvale, California). Each sample was analyzed four times (four consecutive replicate injections) alongside a set of three liquid water laboratory RMs, (ZE = −0.2‰, EV = −10.2‰, DI = −16.5‰ for δ18O values) that had previously been calibrated to the VSMOW-SLAP international isotope scale. Two QA RMs were used for data normalization (ZE and DI) and a QC RM for quality control (EV). Analytical precision of the QA RM was ± 0.3‰ for δ18O values. All δ18O values of water are expressed relative to VSMOW.
Digestion for Sr abundance and isotope analysis
Hair samples were digested using an Ethos EZ® microwave digestion system (Milestone, Inc.; Shelton, Connecticut, USA). Approximately 50 mg of hair was weighed into a Teflon® digestion microvessel. Two milliliters of concentrated ultrapure concentrated HNO3 (Aristar® ULTRA, BDH Chemical; Darmstadt, Germany) were added to the microvessel containing the hair and the microvessel was then sealed, submerged in 10 ml of milli-Q water and 50 ml H2O2 (30% v/v), and then placed within an outer vessel. The outer vessel was then placed in the digester carousel. Two certified reference materials (TORT-2, National Research Council, Ottawa, Canada; Human Hair No. 13, National Institute for Environmental Studies, Tsukuba, Japan) and a method blank of reagents were digested along with the hair samples. The microwave program used for hair digestion was 13.3 °C/min ramp to 200 °C, followed by an isothermal at 200 °C for 15 min with a 60 min cool down to room temperature. The microwave was operated at full power (1500 W) for all heating cycles. Once cooled to room temperature, the hair digests were transferred to acid-leached 2-ml snap-cap centrifuge tubes. A 100-ml aliquot of the primary hair digest was transferred to a 15-ml tube and the volume was brought to 10 ml with ultrapure water. The ultrapure water used for sample cleaning and acid dilutions was from a Milli-Q Academic A10® system (EMD Millipore; Billerica, Massachusetts, USA) with a resistivity >18 MΩ. A standard solution containing 10 ppb In was added to each sample as an internal concentration standard.
Strontium abundance analysis
All strontium elemental abundances were measured via inductively coupled plasma quadrupole-mass spectrometry (ICP-MS) (Agilent 7500ce, Agilent Technologies; Santa Clara, California, USA) at the Department of Geology & Geophysics at the University of Utah, Salt Lake City, Utah. A double-pass spray chamber with perfluoroalcoxy fluorocarbon (PFA) nebulizer (0.1 mL/min), a quartz torch, and nickel cones were used. A calibration solution containing Sr was prepared gravimetrically using a single-element standard (Inorganic Ventures, Inc.; Christiansburg, Virginia, USA). Standard reference solution T-205 (USGS; Reston, Virginia, USA) was measured as an external calibration standard at least five times within each analytical run. The long-term reproducibility for T-205 and differences relative to the accepted values indicated that the Sr concentrations were accurate within 10%. TORT-2 has a certified Sr concentration of 45.2 ± 1.9 mg g−1 (1σ) and the measured Sr concentration of TORT-2 was 41.2 ± 4.4 mg g−1 (1σ, n = 6).
Strontium isotope analysis
All strontium isotope measurements were made using a Neptune Plus multi-collector ICP-MS (Thermo Fisher Scientific; Bremen, Germany) housed in the Department of Geology & Geophysics at the University of Utah, Salt Lake City, Utah, USA. Digests were introduced using an online Sr purification method following Tipple, et al.34 for hair samples and Chesson, et al.74 for water samples. This online system automates the purification of Sr by utilizing a peristaltic pump, a pair of 6-way valves, an in-line separation column, and a SC-2 DX autosampler with a FAST2 valve block (Elemental Scientific; Omaha, Nebraska, USA). The in-line separation column was packed with crown ether Sr resin (Eichrom Technologies; Lisle, Illinois, USA). Variable speed settings on the peristaltic pump allowed samples to be rapidly loaded into the purification column where Sr was trapped while all other elements were rinsed away; the column flow was then reversed and purified Sr was eluted into the spray chamber. A timing solution containing 66 ppb Sr was analyzed daily to insure proper chromatography and to assess column chemistry and efficiency. The instrument was operated at an RF power of 1200 W with nickel sampling and skimmer cones (1.1 mm and 0.8 mm apertures, respectively) and was optimized daily for signal intensity and stability. Cool, auxiliary and sample gas flow rates were 16 L/min, 0.85 L/min, and 0.91 L/min, respectively. The instrument was tuned for sensitivity daily with a solution containing 20 ppb Sr. For 87Sr/86Sr analysis, a static multi-collector routine was used that consisted of 1 block of 170 cycles with an integration time of 1.032 sec per cycle for an individual analysis. Each analysis was followed by a blank to monitor the efficiency of the crown ether Sr resin column. Sr isotope ratios of samples and references were blank- and interference-corrected and then normalized for instrumental mass discrimination using a defined 86Sr/88Sr of 0.1194.
Samples were analyzed for 87Sr/86Sr alongside sets of reference materials and blanks. Reference sets included one primary QA reference for normalization and a keratin QC references to insure measurement reproducibility. The primary reference material was SRM® 987 (0.71034 ± 0.00026 [95% CI]; National Institute of Standards and Technology; Gaithersburg, Maryland, USA), while Human Hair No. 13 was used for QC. Samples and SRM® 987 were analyzed at 5:1. The measured 87Sr/86Sr of SRM 987 and Human Hair No. 13 was 0.71030 ± 0.00004 (1σ, n = 92) and 0.70827 ± 0.00004 (1σ, n = 6), respectively.
Statistical analysis
Statistical analysis was completed using JMP Pro 13® (SAS Institute Inc.; Cary, NC, USA). Normality of the distributions were tested with the Shapiro-Wilk test. If the distributions were normal, then the Student’s t-test was used to compare means at α = 0.005. If the distributions were not normally distributed, then the Welch’s t-test was used at differences of α = 0.005. One-way ANOVA test was used to assess differences between groups at α = 0.005.
Mapping
Mapping and original figure creation was conducted using ArcGIS 10.4 (ESRI; Redlands, CA, USA).
Tap water sources
The culinary water delivered to Salt Lake City originates from mountain streams, surface water reservoirs, and groundwater wells and springs48 with each source having a distinct 87Sr/86Sr value (Supplemental Data Table S1). Groundwater in the Salt Lake Valley has more a radiogenic 87Sr/86Sr than surface water sources (Supplemental Data Table S1). School Y is located near several groundwater wells with groundwater being most likely utilized in the autumn and winter (personal communication, M. Hubbard-Rice, Salt Lake City Public Utilities).
References
Ehleringer, J. R. et al. Hydrogen and oxygen isotope ratios in human hair are related to geography. Proceedings of the National Academy of Science 105, 2788–2793 (2008).
White, C. D., Spence, M. W., Stuart-Williams, H. L. Q. & Schwarcz, H. P. Oxygen isotopes and the identification of geographical origins: The valley of Oaxaca versus the valley of Mexico. Journal of Archaeological Science 25, 643–655 (1998).
Remien, C. H. et al. Deconvolution of isotope signals from bundles of multiple hairs. Oecologia 175, 781–789, https://doi.org/10.1007/s00442-014-2945-3 (2014).
Bartelink, E. J. et al. In New Perspectives in Forensic Human Skeletal Identification (eds Krista E. Latham, Eric J. Bartelink, & Michael Finnegan) Ch. 15, 175–184 (Academic Press, 2018).
Chesson, L. A., Tipple, B. J., Youman, L. V., O’Brien, M. A. & Harmon, M. M. In New Perspectives in Forensic Human Skeletal Identification (eds Krista E. Latham, Eric J. Bartelink, & Michael Finnegan) Ch. 14, 157–173 (Academic Press, 2018).
Lamb, A. L., Evans, J. E., Buckley, R. & Appleby, J. Multi-isotope analysis demonstrates significant lifestyle changes in King Richard III. Journal of Archaeological Science 50, 559–565, https://doi.org/10.1016/j.jas.2014.06.021 (2014).
Meier-Augenstein, W. & Fraser, I. Forensic isotope analysis leads to identification of a mutilated murder victim. Science and Justice 48, 153–159 (2008).
Schwarcz, H. P. Tracing unidentified skeletons using stable isotopes. Forensic Magazine 4, 28–31 (2007).
Lehn, C., Rossmann, A. & Graw, M. Provenancing of unidentified corpses by stable isotope techniques - presentation of case studies. Science and Justice 55, 72–88, https://doi.org/10.1016/j.scijus.2014.10.006 (2015).
Bowen, G. J., Ehleringer, J. R., Chesson, L. A., Stange, E. & Cerling, T. E. Stable isotope ratios of tap water in the contiguous United States. Water Resources Research 43, 1–12 (2007).
Bowen, G. J. & Revenaugh, J. Interpolating the isotopic composition of modern meteoric precipitation. Water Resources Research 39, 1299 (2003).
Price, T. D., Manzanilla, L. & Middleton, W. D. Immigration and the ancient city of Teotihuacan in Mexico: a study using strontium isotope ratios in human bone and teeth. Journal of Archaeological Science 27, 903–913, https://doi.org/10.1006/jasc.1999.0504 (2000).
Price, T. D., Frei, K. M., Dobat, A. S., Lynnerup, N. & Bennike, P. Who was in Harold Bluetooth’s army? Strontium isotope investigation of the cemetery at the Viking Age fortress at Trelleborg, Denmark. Antiquity 85, 476–489 (2011).
Beard, B. L. & Johnson, C. M. Strontium isotope composition of skeletal material can determine the birth place and geographic mobility of humans and animals. Journal of Forensic Sciences 45, 1049–1061 (2000).
Bentley, R. A. Strontium isotopes from the Earth to the archaeological skeleton: A review. Journal of Archaeological Method and Theory 13, 135–187 (2006).
Bentley, R. A. et al. Prehistoric migration in Europe: Strontium isotope analysis of Early Neolithic skeletons. Current Anthropology 43, 799–804 (2002).
Bentley, R. A., Price, T. D. & Stephan, E. Determining the ‘local’ 87Sr/86Sr range for archaeological skeletons: a case study from Neolithic Europe. Journal of Archaeological Science 31, 365–375 (2004).
Price, T. D., Burton, J. H. & Bentley, R. A. The characterization of biologically available strontium isotope ratios for the study of prehistoric migration. Archaeometry 44, 117–135 (2002).
Bataille, C. P., Laffoon, J. & Bowen, G. J. Mapping multiple source effects on the strontium isotopic signatures of ecosystems from the circum-Caribbean region. Ecosphere 3, art118, https://doi.org/10.1890/es12-00155.1 (2012).
Kootker, L. M., van Lanen, R. J., Kars, H. & Davies, G. R. Strontium isoscapes in The Netherlands. Spatial variations in 87Sr/ 86Sr as a proxy for palaeomobility. Journal of Archaeological Science: Reports 6, 1–13, https://doi.org/10.1016/j.jasrep.2016.01.015 (2016).
Evans, J. A., Montgomery, J. & Wildman, G. Isotope domain mapping of 87Sr/86Sr biosphere variation on the Isle of Skye, Scotland. Journal of the Geological Society, London 166, 617–631 (2009).
Coelho, I., Castanheira, I., Bordado, J. M., Donard, O. & Silva, J. A. L. Recent developments and trends in the application of strontium and its isotopes in biological related fields. TrAC Trends in Analytical Chemistry 90, 45–61, https://doi.org/10.1016/j.trac.2017.02.005 (2017).
Font, L., Jonker, G., van Aalderen, P. A., Schiltmans, E. R. & Davies, G. R. Provenancing of unidentified World War II caualties: Application of strontium and oxygen isotope analysis in tooth enamel. Science and Justice 55, 10–17 (2015).
Font, L., van der Peijl, G., van Leuwen, C., van Wetten, I. & Davies, G. R. Identification of the geographical place of origin of an unidentified individual by multi-isotope analysis. Science and Justice 55, 34–42 (2015).
Laffoon, J. E. et al. Investigating human geographic origins using dual-isotope (87Sr/86Sr, δ18O) assignment approaches. PLoS One 12, e0172562, https://doi.org/10.1371/journal.pone.0172562 (2017).
Valentine, B. et al. Evidence for patterns of selective urban migration in the Greater Indus Valley (2600-1900 BC): A lead and strontium isotope mortuary analysis. PLoS One 10, e0123103, https://doi.org/10.1371/journal.pone.0123103 (2015).
Chau, T. H. et al. Reconstruction of travel history using coupled δ18O and 87Sr/86Sr measurements of hair. Rapid Communications in Mass Spectrometry 31, 583–589 (2017).
Tipple, B. J. et al. Stable hydrogen and oxygen isotopes of tap water reveal structure of the San Francisco Bay Area’s water systems and adjustments during a major drought. Water Research 119, 212–224 (2017).
Jameel, Y. et al. Tap water isotope ratios reflect urban water system structure and dynamics across a semi-arid metropolitan area. Water Resources Research 52, 1–20, https://doi.org/10.1002/2016WR019104 (2016).
Ueda, M. & Bell, L. S. A City-wide investigation of the isotopic distribution and source of tap waters for forensic human geolocation ground-truthing. Journal of Forensic Science, 1–13, https://doi.org/10.1111/1556-4029.13319 (2016).
Agency for Toxic Substances and Disease Registry. Toxicological profile for strontium. 445 (U.S. Department of Health and Human Services, Atlanta, Georgia, USA, 2004).
Pors Nielsen, S. The biological role of strontium. Bone 35, 583–588, https://doi.org/10.1016/j.bone.2004.04.026 (2004).
Kempson, I. M. & Lombi, E. Hair analysis as a biomonitor for toxicology, disease and health status. Chemical Society Review 40, 3915–3940 (2011).
Tipple, B. J., Chau, T., Chesson, L. A., Fernandez, D. P. & Ehleringer, J. R. Isolation of strontium pools and isotope ratios in modern human hair. Analytica Chimica Acta 798, 64–73, https://doi.org/10.1016/j.aca.2013.08.054 (2013).
Kempson, I. M. & Skinner, W. M. ToF-SIMS analysis of elemental distributions in human hair. Science of the Total Environment 338, 213–227 (2005).
Hu, L. Spatial concentration of trace elements in keratin Master of Science thesis, University of Utah, (2015).
Vautour, G., Poirier, A. & Widory, D. Tracking mobility using human hair: What can we learn from lead and strontium isotopes? Science & Justice 55, 63–71, https://doi.org/10.1016/j.scijus.2014.10.001 (2014).
Bartelink, E. J., Mackinnon, A. T., Prince-Buitenhuys, J. R., Tipple, B. J. & Chesson, L. A. In Handbook of Missing Persons (eds Stephen J. Morewitz & Caroline Sturdy Colls) Ch. 29, 443–462 (Springer, 2016).
Ehleringer, J. R., Barnette, J. E., Jameel, Y., Tipple, B. J. & Bowen, G. J. Urban water - a new frontier in isotope hydrology. Isotopes in Environmental and Health Studies, 1–10, https://doi.org/10.1080/10256016.2016.1171217 (2016).
Sharp, Z., Atudorei, V., Panarello, H., Fernandez, J. & Douthitt, C. B. Hydrogen isotope systematics of hair: archeological and forensics applications. Journal of Archaeological Science 30, 1709–1716 (2008).
Mant, M., Nagel, A. & Prowse, T. Investigating residential history using stable hydrogen and oxygen isotopes of human hair and drinking water. Journal of Forensic Science 61, 884–991, https://doi.org/10.1111/1556-4029.13066 (2016).
Thompson, A. H. et al. Stable isotope analysis of modern human hair collected from Asia (China, India, Mongolia, and Pakistan). Am J Phys Anthropol 141, 440–451, https://doi.org/10.1002/ajpa.21162 (2010).
Valenzuela, L. O., Chesson, L. A., O’Grady, S. P., Cerling, T. E. & Ehleringer, J. R. Spatial distributions of carbon, nitrogen and sulfur isotope ratios in human hair across the central United States. Rapid Communications in Mass Spectrometry 25, 861–868 (2011).
Thompson, A. H., Wilson, A. S. & Ehleringer, J. R. In Treatise on Geochemistry Vol. 23 Archaeology and Anthropology (ed Thure E. Cerling) 371-393 (Elsevier, 2014).
O’Grady, S. P. et al. Hydrogen and oxygen isotope ratios in body water and hair: modeling isotope dynamics in nonhuman primates. American Journal of Primatology 74, 651–660, https://doi.org/10.1002/ajp.22019 (2012).
Kennedy, C. D., Bowen, G. J. & Ehleringer, J. R. Temporal variation of oxygen isotope ratios (δ18O) in drinking water: Implications for specifying location of origin with human scalp hair. Forensic Science International 208, 156–166 (2011).
Good, S. P. et al. Patterns of local and nonlocal water resource use across the western U.S. determined via stable isotope intercomparisons. Water Resources Research 50, 8034–8049, https://doi.org/10.1002/2014wr015884 (2014).
Briefer, L. In Salt Lake County Watershed Symposium (Salt Lake City Department of Public Utilities, Utah Cultural Celebration Center, 2012).
von Holstein, I. C. C., Font, L., Peacock, E. E., Collins, M. J. & Davies, G. R. An assessment of procedures to remove exogenous Sr before 87Sr/86Sr analysis of wet archaeological wool textiles. Journal of Archaeological Science 53, 84–93, https://doi.org/10.1016/j.jas.2014.10.006 (2015).
Gellein, K. et al. Trace element profiles in single strands of human hair determined by HR-ICP-MS. Biological Trace Element Research 123, 250–260 (2008).
Morton, J., Carolan, V. A. & Gardiner, P. H. E. Removal of exogenously bound elements from human hair by various washing procedures and determination by inductively coupled plasma mass spectrometry. Analytica Chimica Acta 455, 23–34 (2002).
Kempson, I. M. & Skinner, W. M. A comparison of washing methods for hair mineral analysis: Internal versus external effects. Biological Trace Element Research 150, 10–14 (2012).
Bencko, V. Use of human hair as a biomarker in the assessment of exposure to pollutants in occupational and environmental settings. Toxicology 101, 29–39 (1995).
Dombovari, J. & Papp, L. Comparison of sample preparation methods for elemental analysis of human hair. Microchemical Journal 59, 187-193, doi:MJ981573 (1998).
Razagui, I. B.-A. A comparative evaluation of three washing procedures for minimizing exogenous trace element contamination in fetal scalp hair of various obstetric outcomes. Biological Trace Element Research 123, 47–57 (2008).
Rodushkin, I. & Axelsson, M. D. Application of double focusing sector field ICP-MS for multielemental characterization of human hair and nails. Part I. Analytical methodology. The Science of the Total Environment 250, 83–100 (2000).
Font, L., van der Peijl, G., van Wetten, I., Vroon, P. & van der Wagt, B. Strontium and lead isotope ratios in human hair: investigating a potential tool for determining recent human geographical movements. Journal of Analytical Atomic Spectrometry 27, 719–732 (2012).
Tamburo, E., Varrica, D. & Dongarra, G. Gender as a key factor in trace metal and metalloid content of human scalp hair. A multi-site study. Science of the Total Environment 573, 996–1002, https://doi.org/10.1016/j.scitotenv.2016.08.178 (2016).
Senofonte, O., Violante, N. & Caroli, S. Assessment of reference values for elements in human hair of urban schoolboys. Journal of Trace Elements in Medicine and Biology 14, 6–13 (2000).
Chojnacka, K., Michalak, I., Zielińska, A., Górecka, H. & Górecki, H. Inter-relationship between elements in human hair: The effect of gender. Ecotoxicology and Environmental Safety 73, 2022–2028, https://doi.org/10.1016/j.ecoenv.2010.09.004 (2010).
Rodushkin, I. & Axelsson, M. D. Application of double focusing sector field ICP-MS for multielemental characterization of human hair and nails. Part II. A study of the inhabitants of northern Sweden. The Science of the Total Environment 262, 21–36 (2000).
Zaichick, S. & Zaichick, V. The scalp hair as a monitor for trace elements in biomonitoring of atmospheric pollution. International Journal of Environment and Health 5, 106–124 (2011).
Zaichick, S. & Zaichick, V. The effect of age and gender on 37 chemical element contents in scalp hair of healthy humans. Biological Trace Element Research 134, 41–54 (2010).
Vahter, M., Akesson, A., Liden, C., Ceccatelli, S. & Berglund, M. Gender differences in the disposition and toxicity of metals. Environmental Research 104, 85–95, https://doi.org/10.1016/j.envres.2006.08.003 (2007).
Kempson, I. M., Skinner, W. M. & Kirkbride, K. P. Advanced Analysis of Metal Distributions in Human Hair. Environmental Science and Technology 40, 3423–3428 (2006).
Allen, E. & Crimaldi, L. In Boston Globe (Boston, MA, 2015).
Frei, K. M. et al. Tracing the dynamic life story of a Bronze Age Female. Scientific Reports 5, 10431, https://doi.org/10.1038/srep10431 (2015).
Frei, K. M. et al. A matter of months: High precision migration chronology of a Bronze Age female. PLoS One 12, e0178834, https://doi.org/10.1371/journal.pone.0178834 (2017).
Bataille, C. P. & Bowen, G. J. Mapping 87Sr/86Sr variations in bedrock and water for large scale provenance studies. Chemical Geology 304-305, 39–52 (2012).
Willmes, M. et al. Mapping of bioavailable strontium isotope ratios in France for archaeological provenance studies. Applied Geochemistry 90, 75–86, https://doi.org/10.1016/j.apgeochem.2017.12.025 (2018).
Frei, K. M. & Frei, R. The geographic distribution of strontium isotopes in Danish surface waters – A base for provenance studies in archaeology, hydrology and agriculture. Applied Geochemistry 26, 326–340 (2011).
Evans, J. A., Montgomery, J., Wildman, G. & Boulton, N. Spatial variations in biosphere 87Sr/86Sr in Britain. Journal of the Geological Society 167, 1–4, https://doi.org/10.1144/0016-76492009-090 (2010).
Valenzuela, L. O. et al. Evaluation of childhood nutrition by dietary survey and stable isotope analyses of hair and breath. American Journal of Human Biology, e23103, https://doi.org/10.1002/ajhb.23103 (2018).
Chesson, L. A. et al. Strontium isotope ratios of tap water from the coterminous USA. Ecosphere 3, Article 67 (2012).
Acknowledgements
This project was supported by the National Institute of Justice, Office of Justice Programs, U.S. Department of Justice (Award No. 2011-DN-BX-K544) awarded to B.J.T. and IsoForensics, Inc. The samples were collected as part of an Innovative Research Grant [Activity 18751 “Monitoring Childhood Nutrition Using Stable Isotopes in Hair”] to L.O.V. through the National Children’s Study program managed at the University of Utah. The authors are indebted to the participants of the National Children’s Study for their involvement in this study. The authors thank Thure Cerling, Lesley Chesson, Edna Ehleringer, Marian Hubbard-Rice, Laurie Moyer-Mileur, Larry Madden, and Shannon O’Grady for their support and input. Further, the authors recognize Lindsey Enright, Diego Fernandez, and Glen Mackey for their help in the stable isotope analysis, trace element analysis, and strontium isotope analysis. B.J.T. is grateful to IsoForensics, Inc. for financial management of this award. All samples were obtained and maintained under University of Utah IRB [00035524]. The opinions, findings, and conclusions or recommendations expressed in this manuscript are those of the authors and do not reflect those of the Department of Justice.
Author information
Authors and Affiliations
Contributions
Conceived and designed the experiments: B.J.T., L.O.V., and J.R.E. Performed the experiments: B.J.T. and L.O.V. Analyzed the data: B.J.T. Wrote the paper: B.J.T., L.O.V., and J.R.E.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Tipple, B.J., Valenzuela, L.O. & Ehleringer, J.R. Strontium isotope ratios of human hair record intra-city variations in tap water source. Sci Rep 8, 3334 (2018). https://doi.org/10.1038/s41598-018-21359-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-21359-0
This article is cited by
-
Sr–Pb isotope differences in pre- and post-burial human bone, teeth, and hair keratin: implications for isotope forensics
International Journal of Legal Medicine (2024)
-
A part per trillion isotope ratio analysis of 90Sr/88Sr using energy-filtered thermal ionization mass spectrometry
Scientific Reports (2022)
-
Revisited digestion methods for trace element analysis in human hair
Journal of Analytical Science and Technology (2020)
-
Longitudinal and transverse variation of trace element concentrations in elephant and giraffe hair: implication for endogenous and exogenous contributions
Environmental Monitoring and Assessment (2018)
-
Applying the principles of isotope analysis in plant and animal ecology to forensic science in the Americas
Oecologia (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.