Abstract
Rensch’s rule, stating that sexual size dimorphism (SSD) becomes more evident and male-biased with increasing body size, has been well supported for taxa that exhibit male-biased SSD. Bats, primarily having female-biased SSD, have so far been tested for whether SSD allometry conforms to Rensch’s rule in only three studies. However, these studies did not consider phylogeny, and thus the mechanisms underlying SSD variations in bats remain unclear. Thus, the present study reviewed published and original data, including body size, baculum size, and habitat types in 45 bats of the family Rhinolophidae to determine whether horseshoe bats follow Rensch’s rule using a phylogenetic comparative framework. We also investigated the potential effect of postcopulatory sexual selection and habitat type on SSD. Our findings indicated that Rensch’s rule did not apply to Rhinolophidae, suggesting that SSD did not significantly vary with increasing size. This pattern may be attributable interactions between weak sexual selection to male body size and strong fecundity selection for on female body size. The degree of SSD among horseshoe bats may be attributed to a phylogenetic effect rather than to the intersexual competition for food or to baculum length. Interestingly, we observed that species in open habitats exhibited greater SSD than those in dense forests, suggesting that habitat types may be associated with variations in SSD in horseshoe bats.
Similar content being viewed by others
Introduction
Animal body size is an important trait under evolutionary scrutiny1. For small mammals such as rodents and bats, body size plays a pivotal role in shaping variations in reproductive success2,3. Body size dimorphism between sexes is ubiquitous in nature and extensively varies even among closely related groups. Rensch (1950, 1960) was the first to observe a common pattern of interspecific covariance between body size and sexual size dimorphism (SSD)4. Rensch’s rule predicts that when males are larger than females, SSD increases with body size, but when females are larger than males, SSD decreases in larger species. Rensch’s rule implies that male body size has a faster evolutionary rate than females5,6. Rensch’s rule was originally formulated at the interspecific level, and most publications on SSD present broad interspecific comparisons. For example, there is strong support for Rensch’s rule in all mammals and birds7. Rensch’s rule has been confirmed at the macroecological level by observations in various animal taxa including insects8,9, fish10, reptiles11, birds12,13, and primates14. In general, SSD allometry consistent with Rensch’s rule occurs most frequently in animals for which males are larger than females. Previous studies on SSD in taxa that display female-biased SSD provide mixed support for Rensch’s rule15,16,17,18.
Most mammals exhibit SSD and is mainly male-biased. Bats, however, primarily display female-biased SSD19. Previous studies have shown that various species of Vespertilionidae, Rhinolophidae, and Pteropodidae have female-biased SSD with respect to body mass and skeletal measures (e.g., forearm length), but these studies have mostly focused on SSD differences both within and between species20,21,22,23. Little is known about whether bats follow Rensch’s rule at the intraspecific or interspecific level and the causes for different SSDs in the context of a phylogenetic comparative framework. To date, three studies have tested Rensch’s rule in bats. One study confirmed that the pattern of SSD in Myotis bats did not conform to Rensch’s rule24. The second study indicated that variations in SSD among greater horseshoe bat (Rhinolophus ferrumequinum) populations is consistent with Rensch’s rule25. The third study showed that variations in body size in 10 bat species (2 from Vespertilionidae and 6 from Phyllostomidae) apparently do not conform to Rensch’s rule at intraspecific and interspecific levels26. Therefore, additional studies validating Rensch’s rule in bats (a unique mammal group), especially after controlling for phylogenetic associations, are warranted.
Biologists generally explain SSD in terms of sexual selection27,28,29. SSD is often used as an indicator of the intensity of precopulatory sexual selection30,31. Meanwhile, sexual selection is to drive diversity of baculum form32,33. Large baculum and testes are expected to confer an advantage in sperm competition and reproductive success34. Baculum size (adjusted for body size) has often been used as a proxy of the intensity of postcopulatory sexual selection. Beacuse large testes/bacula and increased spermatogenesis entail high production cost and is hypothesized to be associated with a trade-off between pre- and postcopulatory traits35. For example, a trade-off (negative correlation) between precopulatory (SSD) and postcopulatory (relative testes mass/baculum length) traits was observed in 14 species of pinnipeds36 and in 17 male cetaceans35.
In addition to sexual selection, habitat type is also an important ecological factor that influences SSD. Species living in different habitats may need to deal with variations in environmental conditions (e.g., food availability, competitors and predators), any of which could be associated with SSD variations among species37. For example, the existence of habitat-specific sexual dimorphism has been reported in Anolis spp.38. Moreover, males in 138 turtle species are proportionally smaller in more aquatic habitats and larger in terrestrial habitats39.
Rhinolophidae is a suitable clade for studying body size patterns change and associated mechanisms as it is the second most speciose bat species (77 species listed in40). Linear forearm length ranges from 30 cm in adult Rhinolophus subbadius to 75 cm long in R. luctus, and this genus exhibits predominantly female-biased size dimorphism, an uncommon pattern in other mammalian taxa. Additionally, they are ecologically diverse, inhabit different habitats across temperate and tropical regions of the Old World41,42. Although horseshoe bats all fly close to the substrate and vegetation irrespective of habitat type or their body size43, some species prefer relatively open habitats for foraging such as arid areas, fynbos (a small belt of natural shrubland or heathland vegetation), and savanna woodlands, whereas others occupy ecosystems with dense vegetation (e.g., forests). For example, R. mehelyi always forages in woodlands and avoids open spaces, suggesting that this species prefers cluttered spaces44. In the present study, we analysed a comprehensive dataset of male and female body mass and forearm length estimates for 45 species of horseshoe bats using a unifying comparative phylogenetic framework (Tables 1 and 2; Fig. 1). Our aims were: (1) to test whether patterns of interspecific variations in SSD in horseshoe bats conform to Rensch’s rule, (2) to test the hypothesis that habitat types fuel variations in SSD among horseshoe bats, and (3) to test the additional alternative hypothesis that differences in SSD are the result of sexual selection by assessing the relationship between baculum size and SSD or are simply a reflection of phylogenetic constraint.
Results
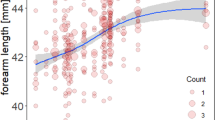
The taxa exhibited varying degrees and directions of size dimorphism. In 37 out of 45 species of horseshoe bats, females showed longer forearms than males. In 27 out of 34 species of horseshoe bats with body mass data, females were larger in twenty-seven of them (see Table 2 for details). On average, SSD in Rhinolophdae is female-biased. The reduced major axis regression of log10 (male size) on log10 (female size) showed that the allometric slopes did not significantly differ from 1. After correcting for phylogeny, the slopes still did not significantly differ from 1 (Table 3; Figs 2 and 3). After excluding species with male-biased SSD and fewer than five individuals of each sex, we obtained the same results, i.e., an allometric slope that did not differ from 1, and SSD following an isometric pattern.
Phylogenetic analyses
SSD in body mass and body mass of both sexes were significantly influenced by phylogeny (SSD: λ = 0.999, p < 0.001; K = 0.764, P = 0.028; male body mass: λ = 0.999, P = 0.002; K = 0.901, P = 0.01; female body mass: λ = 0.999, P = 0.0005; K = 0.976, P = 0.004), whereas both female and male forearm length showed a weaker phylogenetic signal (male forearm length: λ = 0.494, P = 0.309; K = 0.629, P = 0.083, female forearm length: λ = 0.455, P = 0.316; K = 0.612, P = 0.097), suggesting that related species were not statistically more likely to have similar forearm length than would be expected.
Tracing the evolution of body size and SSD on a pruned ML phylogeny unequivocally optimized median size (male forearm length = 48.37 mm; female forearm length = 48.88 mm), and female-biased SSD (SD = 1.009) was observed as ancestral in Rhinolophidae (Fig. 4). Size evolution was then inferred to have proceeded through repeated increases and decreases, and SSDs were variable among species (Fig. 4).
The influence of habitat type on the evolution of SSD
SSD significantly differed between open and closed habitat lineages when phylogeny was not considered (ANOVA: F 1 ,25 = 11.47, P = 0.002). Species inhabiting open habitats showed greater mean dimorphism than those linked to forest habitats (Fig. 5a). Additionally, female forearm length was greater than that in males in open habitats compared to those in forests (Fig. 5b). After using independent contrasts to control for phylogenetic inertia in these data, SSD in forearm length did not correlate with habitat types (phyaov: F 1 ,25 = 11.47, P = 0.15).
The influence of baculum size on the evolution of SSD
Linear regression demonstrated that male body size is not associated with baculum length (forearm length: R2 = 0.09, F1,19 = 1.904, P = 0.184; body mass: R2 = 0.10, F1,14 = 1.539, P = 0.235). After using independent contrasts to control for phylogenetic inertia in these data, male forearm length was positively associated with baculum length (pgls: F1,17 = 6.263, R2 = 0.269, P = 0.005). Additionally, our results showed that SSD is positively associated with baculum length (R2 = 0.291, F1,18 = 7.382, P = 0.014). However, when controlling for the effect of male body size, SSD was not significantly associated with baculum length (male forearm length × baculum length: R2 = 0.323, F3,16 = 0.0919, P = 0.649). Thus, our study found no clear relationship between baculum size and dimorphism.
Discussion
In this study, we found that the slopes of the regression of log10 (male size) on log10 (female size) were not significantly greater than one regardless of traits of body size and data sets, suggesting no evidence to support Rensch’s rule among horseshoe bats (Table 3). Moreover, our results indicated that species occupying open habitats had greater mean dimorphism compared to those in forest habitats, suggesting that habitat type may drive the evolution of SSD among horseshoe bats. Additionally, SSD was not significantly associated with baculum length when controlling for the effect of male body size, suggesting that baculum length may not be linked with the degree of SSD among horseshoe bats.
As small mammals, horseshoe bats have relatively subtle size differences between males and females. Our results indicated that SSD patterns in horseshoe bats do not conform to Rensch’s rule irrespective of SSD measurements (body mass vs. forearm length) and computation method (conventional vs. phylogenetically informed). This study joins other gruops that question the veracity of Rensch’s rule15,45,46,47, especially in taxa with female-biased SSD. Similarly, our results agree with previous work on SSD in bats, as one recent study showed that Myotis does not follow Rensch’s rule among species either24. Another study including seven phyllostomid and two Myotis bat species also reported similar results26. However, variations in SSD among populations within R. ferrumequinum conform to Rensch’s rule25. These studies show inconsistencies in SSD variations in horseshoe bats between interspecific and intraspecific comparative analysis. Additionally, the present study observed that the most recent common ancestor in Rhinolophidae exhibited medium-sized forearm length (males = 48.37 mm; females = 48.88 mm, Fig. 4), similar to that of a previous study (forearm length: 50.16)48, and also matched well with the size observed in many median-size horseshoe bats42.
Several factors may explain the lack of conformity to Rensch’s rule and female-biased SSD in horseshoe bats. First, the limited extent of SSD in this group reduces statistical power to detect any existing trend. Second, because horseshoe bats possess extensive variability with respect to mating systems (e.g., polygamy in R. ferrumequinum49 vs monogamy in R. luctus50), sexual selection for male size may be nondirectional. Third, sexual selection may act more on echolocation call frequencies in males than body size in horseshoe bats, suggesting weak sexual selection to body size in males48,51. Meanwhile, in bats, fecundity selection might confer advantages to larger female body size, such as reduced proportionate fetal or newborn load52, increased stomach capacity for food, reproductive success, and reduced relative cost of milk production22,53. In this case, interactions between weak sexual selection in males and strong fecundity selection in females are likely to determine female-biased SSD in horseshoe bats, and thus may explain the observed nonconformity to Rensch’s rule because stronger sexual selection in males in conjunction with weaker selection in females is considered a major determinant of Rensch’s rule (sexual selection is for increased male size in species with male-biased SSD when that trait results in greater mating success; sexual selection is for reduced male size in taxa with female-biased SSD)12.
Although habitat has been suggested to influence the evolution of SSD, this hypothesis has rarely been demonstrated. In this study, we found that species in open habitats (e.g., fynbos, savanna woodland, and arid) exhibit more extensive SSD than those inhabiting highly dense forests (Fig. 5a). The role of SSD in reducing competition between sexes is often assessed in investigations that aim to identify habitat-related factors that fuel SSD evolution. The niche theory predicts that SSD should be smaller when the range of available resources is smaller. The large SSD may be selected for by intraspecific competition when different sizes deemed most effective among various resource types54. A few studies have validated the assumption that SSD reduces intersexual competition38. Dechmann et al.55 did not observe any difference between male and female diets in the common noctule (Nyctalus noctula). In horseshoe bats, echolocation call frequencies of females in most species are only slighter higher than that of males, and such small differences seem inappropriate to differentiate prey size. In this case, sex differences in body size may not reduce competition between the sexes for food resources in both open and forest habitats.
Selective pressures might influence SSD as it relates to structural habitat38. Optimal feeding models predict that the distribution of optimal body size may depend on forage strategy56, i.e., active searchers display unimodal plots, whereas sit-and-wait predators have bimodal plots. Thus, SSD should be more distinct among the latter. This prediction has been validated in greater Antillean Anolis lizards38. Two foraging styles, aerial hawking and flycatching, are often used by horseshoe bats to hunt for insects57,58. A previous study showed that horseshoe bats perform flycatching (a sit-and-wait strategy) more than aerial hawking to save energy when there are relatively fewer insects59. Tree density and diversity in open habitats (e.g., fynbos, savanna woodland, and arid in Africa) are lower than in tropical moist forest biomes (e.g., Southeast Asia and southern China)60. Because diversity and richness of plants are useful proxies for insect abundance61, the present study presumed that insect richness in open habitats would be lower than that in thick forests. Thus, in open habitats, flycatching would be mainly employed to hunt prey and would be associated with greater SSD, whereas those inhabiting dense forests may mainly be active searchers and have a lower degree of SSD.
In this study, females in open habitats had larger forearms than males compared to that in forests (Fig. 5b), which in turn may lead to bigger SSD in open habitats. In the horseshoe bat, sexual segregation is common after mating in the spring. Females and their offspring often constitute a maternal colony58. In open habitats, the distribution of food resources is relatively disperse. In this case, the intensity of competition for food among females may be relatively small, which may increase the number of larger females in open habitats compared to the forest. Moreover, females in open habitats have to spend more time foraging compared to their forest counterparts because of food dispersion49,57. To save energy, they evolved larger bodies to reduce the surface-to-volume ratio for heat conduction. Interestingly, SSD did not correlate significantly with habitat types after using independent contrasts to control for phylogenetic inertia, suggesting that differences in SSD among horseshoe bats may be attributed to their evolutionary history (common ancestor) rather than to adaptation to different habitat types. However, we should be cautious because only two types of habitats were studied here, and this may suggest that closely related species may have the same habitat types. This would hence magnify the effect of phylogeny during analysis. Thus, further experimental examination will help to clarify the relationship between SSD and habitat types in bats.
No clear relationship between baculum size and SSD was observed in the present study, although SSD appeared to be positively associated with baculum length. However, when controlling for the effect of male body size, SSD was not significantly associated with relative baculum length. Similar to other related studies in mammals62,63, we found no evidence of a trade-off between precopulatory (SSD) and postcopulatory (baculum length) traits in horseshoe bats. In fact, a few studies suggest a theoretical trade-off between pre- and postcopulatory traits across a phylogeny, perhaps due to complications of interspecific comparisons. The correlation between postcopulatory traits (testes mass and/or baculum length) and SSD exsits only in polygamous species for which males engage in competition to monopolize access to multiple females64. This is unlikely to be the case for all horseshoe bats, as polygamy is not a universal trait of all horseshoe bat species (e.g., monogamous R. luctus). Furthermore, bats are the only mammals that truly fly, thus traits in bats favored by sexual selective pressure may be differ from those of other mammals. For example, female greater sac-winged bats (Saccopteryx bilineata) favor small and symmetric males for mating65. Moreover, female in R. mehelyi preferentially select males with high frequency echolocation calls for mating51. Additionally, bat baculum length may not to be associated with sexual selection intensity according to comparative analysis66. These studies indicated that sexual selection pressure on male bats may preferentially act on smaller body size and higher echolocation call frequencies rather than larger body size or baculum size. Thus, these possibilities may obscure a clear prediction of the trade-off hypothesis on precopulatory versus postcopulatory investment, as well as suggest that sexual selection may not be associated with variations in SSD among horseshoe bats. Future studies should thus aim to generate direct evidence for the relationship between sexual selection and SSD in future studies.
Some studies have suggested that phylogenetic constraints are mainly responsible for the degree of sexual dimorphism67, and the findings of the present study support this theory. We found a significant phylogenetic signal with regard to body mass of the two sexes and SSD, suggesting that closely related species should exhibit highly in SSD than more distantly related ones because they share more recent ancestors. Additionally, repeated increases and decreases in SSD among horseshoe bats were noted during evolution.
Conclusion
In summary, we found that SSD variations among species within the Rhinolophidae do not conform to Rensch’s rule. These results are similar to the findings of previous studies done involving Vespertilionidae and Phyllostomatidae, suggesting that Rensch’s rule may not be applicable to all bat species within a family. Despite extensive efforts in identifying the causal mechanisms of SSD evolution, only a few studies have investigated the impact of ecological factors. Our results indicate that evolutionary changes in the degree of SSD among horseshoe bats may undergo phylogenetic constraints, and that the evolotuin of SSD may be closely linked with habitat types rather than sexual selection. A critical limitation of this study is that the results relating to SSD and habitat types should be interpreted with caution because habitat types of horseshoe bats have to be classified as either open or forest in the absence of specific habitat use information in each species. Future studies should focus the relationship between the degree of SSD and habitat use in sympatric horseshoe bats.
Materials and Methods
Taxa Sampling
Morphometric data (body mass, forearm length, baculum length, and baculum width) were collected from published and our unpublished data (Table 1). We carefully analyzed the literature to collect individual data. For our own data, every individual was only measured once. Data from 45 species of horseshoe bats were collected in this study. Twenty species had exact coordinate information about the sample sites (Table 2). Ten species collected in southern and central Africa from the literatures only had the distribution range for the sample sites (Table 2). For the other 15 species, since their data was collected from a area including several neighboring sites, exact coordinate information and sample size for every site was not available in the literatures (Table 2). In this case, all individual data from different sites of a species was pooled to calculate mean value for subsequent analyses. Although the presence and level of SSD may depend on the sample sites and/or populations in some phyllostomid bats26, this was not this case in horseshoe bats for two reasons. First, SSD was observed for most horseshoe bats in this study (see Table 2 for details). Second, in our previous study25, we analyzed SSD of 23 populations of R. ferrumequinum with a wide range of distributions, and found that SSD was consistently female-biased and not statistically significantly different among along a latitudinal cline, suggesting environmental conditions may not influence in SSD variation at intraspecific level. These two facts implied that SSD in horseshoe bats may be only slightly different among sites or populations. Additionally, although individuals of some species were captured at different times, this did not influence our results because only adult data was collected in this study. In this case, it may be rational to pool data of a species from different sites or times to increase sample size, especially for interspecific comparative analysis.
SSD for each species was calculated using the Lovich–Gibbons index (Lovich and Gibbons 1992)68, as proposed by Smith (1999)69. The formulas of Lovich–Gibbons index was displayed as follows:
To compare the relative effects of sample size reduction and the direction of SSD, four datasets were analyzed: (1) a full dataset for which taxonomic inclusion is maximized (45 species for forearm length; 34 species for body mass); (2) a reduced dataset only with taxa with body size measurements from at least 5 individuals of each sex (33 species for forearm length; 27 species for body mass); (3) a reduced dataset with taxa with female-biased SSD (37 species for forearm length; 27 species for body mass); (4) a reduced dataset for species with female-biased SSD and body size measurements from at least 5 individuals of each sex (28 species for forearm length; 17 species for body mass).
Additionally, we collected and analyzed information about habitat types based on the published literature and our own data (Table 2). In this study, we did not consider the bats’ roosting habitats (caves, mines, buildings, and trees). Thus, habitat type was classified based on nighttime activity and foraging behavior. Horseshoe bats all fly close to the substrate and vegetation regardless of habitat type or their body size43, but habitat preferences have been observed in some species (e.g., R. mehelyi in woodland44; R. megaphyllus in woodland70, and R. hipposideros in linear landscape elements like hedgerows or highly structured open landscapes71). Here habitat type was classified as open (relative open and less cluttered habitats than forest) or forest (highly cluttered habitats), two types in which Rhinolophdae species can be found. The habitats of 14 bat species (R. capensis, R. denti, R. simulator, R. swinnyi, R. euryale, R. arcuatus, R. clivosus, R. darlingi, R. ferrumequinum, R. fumigatus, R. hildebrandtii, R. hipposideros, R. blasii, and R. landeri) were relatively open. These included fynbos, arid areas, savanna woodland, hedgerows, riparian forest, pastures, hedges, and ditches. Most of the 14 species are distributed in Africa and thus occupy relatively open ecosystems such as Savanna72. The closed habitats including different types of forest, such as evergreen forest, deciduous forest, rainforests, secondary forest, bamboo forest, and so forth, were home to all other species except for R. euryotis, R. eloquens, R. xinzhongguoensis, R. yunanensis, R. pumilus, R. shortridgei, and R. rouxii (Table 2). Most of these species were mainly distributed in southeastern Asia and southern China, and so occupy ecosystems with relatively dense vegetation (e.g., forests)42. Moreover, many common species (e.g. R. affinis, R. pearsonii, R. macrotis, R. rex, R. huanus, R. lepidus, R. osgoodi, R. pusillus, R. sinicus, R. thomasi, and R. luctus), mainly distributed in China, have been observed to frequently forage in forest by acoustic monitoring (personal observation during fieldworks by Tinglei Jiang and Xiaobin Huang). In a previous study73, habitat types have been classified as forest or other (habitat other than forest, e.g., savanna, arid, woodland, and fynbos) to assess the contribution of habitat types to echolocation frequency by Bayes Discrete analysis. Differences in wing parameters in bats can lead to various degrees of flexibility in using open space or clutter habitat at both the intraspecific and interspecific levels74,75. So far, forage habitat use of many horseshoe bats is still unknown. In light of facts, we here considered it appropriate to classify the habitat types of horseshoe bats as open or forested, because moderately cluttered intermediate habitats (between forest and open) were difficult to define for horseshoe bats in the absence of accurate data regarding habitat use.
We also collected male baculum length and width in horseshoe bats based on the published literature and our own data (Table 2). We obtained data about male baculum length and width from 20 horseshoe bats in this study. Although the mating system is very important to explain the evolution of SSD, we did not collected this data because little is known about it.
Statistical Analysis
All variables were log10-transformed, and we performed tests of normality using the Kolmogorov–Smirnov test with Lilliefors correction. The results showed that all variables met assumptions of a normal distribution (P > 0.05, Table 1). We calculated the allometric slope as the reduced major axis regression of log10 (male size) on log10 (female size) and tested whether the slope was significantly different from 1. Some authors suggested placing males to the x-axis when SSD is assessed based upon a log/log plot of the size of one sex against the size of the other sex8,76, but the other authors have preferred to assign females on the x-axis25,77. Thus, there is still no convention to assign of the sexes on the x- and y-axe78. Here we place female size on this x-axis when we estimated SSD from a log/log plot by the reduced major axis regression of log10 (male size) on log10 (female size). In this case, the slope >1 indicated allometry consistent with Rensch’s rule. We used the Smart R package79 for these analyses. A general linear regression model was used to examine the relationship between SSD and baculum size using an identity link function and a gaussian error structure, and ANOVA was used for testing the effect of habitat types on SSD.
Phylogeny
Although nuclear introns may exceed mitochondrial DNA in interspecific phylogenetic reconstruction, mitochondrial DNA remains a very useful marker for studying phenotypic evolution because the mtDNA phylogeny can quickly and cheaply provides a global overview of the phylogenetic relationships48,80. Moreover, mtDNA sequences on online databases (e.g., GenBank) were more complete than nuclear markers in both within and between species. In this study, we first checked the GenBank and found that a large number of cytb gene sequences rather than the other molecular markers were already available for a large proportion of horseshoe bats. For these reasons, we here used cytb gene sequence data to reconstruct the phylogeny. Two closely related species (Hipposideros armiger and H. cineraceus) were used to root the tree. We obtained cytb sequences from Genbank (see Table 2 for accession numbers). Sequences were aligned with ClustalW81. After visual inspection, they were imported into jModelTest 0.1.182 to calculate the best-fit model of nucleotide substitution for the cytb gene according to Akaike information criterion (AIC). The most complex general-time-reversible model (GTR + I + Γ) was chosen as the best substitution model for this gene. Maximum likelihood (ML) tree reconstruction was conducted in PAUP* 4.0 and RaxML. We then used the R-package ape83 to prune species for which we had no morphological or ecological data (analyses including forearm length: N = 38, Fig. 1a; analyses involving body mass: N = 32, Fig. 1b; analyses involving baculum length: N = 19; analyses including habitat type: N = 30).
Ancestral Size Reconstruction
We reconstructed ancestral states of continuous characters (male and female size, and SSD) on the ML tree pruned for outgroups and species without forearm length data using parsimony analyses in Mesquite version 2.75. We sought to understand evolutionary changes of characters rather than the probability of particular ancestral states on the phylogeny.
Phylogenetic Comparative Analyses
We also performed phylogenetic reduced major axis regressions84 using the phyl.RMA function in the phytol package to estimate phylogenetically informed allometric slopes for 38 species that had cytb sequence information.
We measured the strength of the phylogenetic signal in our continuous variables (male and female forearm length, male and female body mass, SSD, baculum size) by estimating Pagel’s λ85,86 and Blomberg’s K87 using the phytools package84. In addition, we tested whether estimates of these two metrics of phylogenetic signal were significantly different from 0 (no phylogenetic signal).
Then we used phylogenetic generalized least squares (PGLS)86,88 to test for a relationship between 1) male and female body mass, 2) SSD in body mass and male or female body mass, 3) SSD and the baculum length. Finally, since SSD in forearm length was normally distributed, we used phylogenetic analyses of variance (ANOVA) to assess whether SSD was influenced by habitat types (open habitats, closed habitats). Phylanovas (10,000 iterations) were conducted using the geiger package89. In these analysis, SSD was calculated using the Lovich–Gibbons index based on body mass and forearm length. All statistical analyses were carried out in R90.
Ethics Statement
Our work did not cause any physical injuries to bats. All research involving animals was carried out in accordance with the relevant laws for experiments involving vertebrates of the People’s Republic of China, and approved by the National Animal.
Research Authority in Northeast Normal University, China ((Permit Number: NENU-W-2008–108).
Data availability
The datasets generated and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Peters, R. H. The ecological implications of body size. Vol. 2 (Cambridge University Press, 1986).
Yoccoz, N. G. & Mesnager, S. Are alpine bank voles larger and more sexually dimorphic because adults survive better? Oikos, 85–98%@ 0030–1299 (1998).
Ward, H. L., Ransome, R. D., Jones, G. & Rossiter, S. J. Determinants and patterns of reproductive success in the Greater Horseshoe bat during a population recovery. PloS one 9, e87199 (2014).
Rensch, B. & Rensch, B. Evolution above the species level. Vol. 21 (Columbia University Press, 1959).
Fairbairn, D. J. & Preziosi, R. F. Sexual selection and the evolution of allometry for sexual size dimorphism in the water strider, Aquarius remigis. American Naturalist 101–118 (1994).
Blanckenhorn, W. U. Behavioral causes and consequences of sexual size dimorphism. Ethology 111, 977–1016 (2005).
Fairbairn, D. J., Blanckenhorn, W. U. & Székely, T. Sex, size, and gender roles: evolutionary studies of sexual size dimorphism. (Oxford University Press Oxford, 2007).
Abouheif, E. & Fairbairn, D. J. A comparative analysis of allometry for sexual size dimorphism: assessing Rensch’s rule. American Naturalist 540–562 (1997).
Serrano-Meneses, M. A., Córdoba-Aguilar, A., Azpilicueta-Amorín, M., González-Soriano, E. & Székely, T. Sexual selection, sexual size dimorphism and Rensch’ rule in Odonata. Journal of evolutionary biology 21, 1259–1273 (2008).
Young, K. A. Life–history variation and allometry for sexual size dimorphism in Pacific salmon and trout. Proceedings of the Royal Society of London B: Biological Sciences 272, 167–172 (2005).
Berry, J. F. & Shine, R. Sexual size dimorphism and sexual selection in turtles (Order Testudines). Oecologia 44, 185–191 (1980).
Székely, T., Freckleton, R. P. & Reynolds, J. D. Sexual selection explains Rensch’s rule of size dimorphism in shorebirds. Proceedings of the National academy of Sciences of the United States of America 101, 12224–12227 (2004).
Colwell, R. K. Rensch’ rule crosses the line: convergent allometry of sexual size dimorphism in hummingbirds and flower mites. The American Naturalist 156, 495–510 (2000).
Smith, R. J. & Cheverud, J. M. Scaling of sexual dimorphism in body mass: a phylogenetic analysis of Rensch’s rule in primates. International Journal of Primatology 23, 1095–1135 (2002).
Tubaro, P. L. & Bertelli, S. Female-biased sexual size dimorphism in tinamous: a comparative test fails to support Rensch’s rule. Biological Journal of the Linnean Society 80, 519–527 (2003).
Blanckenhorn, W. U. et al. Proximate causes of Rensch’s rule: does sexual size dimorphism in arthropods result from sex differences in development time? The American Naturalist 169, 245–257 (2007).
Liao, W. B., Zeng, Y., Zhou, C. Q. & Jehle, R. Sexual size dimorphism in anurans fails to obey Rensch’s rule. Frontiers in zoology 10, 10 (2013).
Stuart-Fox, D. A test of Rensch’s rule in dwarf chameleons (Bradypodion spp.), a group with female-biased sexual size dimorphism. Evolutionary Ecology 23, 425–433 (2009).
Lindenfors, P., Gittleman, J. L. & Jones, K. E. Sexual size dimorphism in mammals. Sex, size and gender roles: evolutionary studies of sexual size dimorphism (eds Fairbairn, D. J., Blanckenhorn, W. U. & Szekely, T.), 19–26 (2007).
Dietz, C., Dietz, I. & Siemers, B. M. Wing measurement variations in the five European horseshoe bat species (Chiroptera: Rhinolophidae). Journal of Mammalogy 87, 1241–1251 (2006).
Lisón, F., Haz, A., González-Revelles, C. & Calvo, J. F. Sexual size dimorphism in greater mouse-eared bat Myotis myotis (Chiroptera: Vespertilionidae) from a Mediterranean region. Acta Zoologica 95, 137–143 (2014).
Myers, P. Sexual dimorphism in size of vespertilionid bats. The American Naturalist 112, 701–711 (1978).
Storz, J. F. et al. Clinal variation in body size and sexual dimorphism in an Indian fruit bat, Cynopterus sphinx (Chiroptera: Pteropodidae). Biological Journal of the Linnean Society 72, 17–31 (2001).
Stevens, R. D. & Platt, R. N. Patterns of secondary sexual size dimorphism in New World Myotis and a test of Rensch’s rule. Journal of Mammalogy gyv120 (2015).
Wu, H. et al. A test of Rensch’s rule in greater horseshoe bat (Rhinolophus ferrumequinum) with female-biased sexual size dimorphism. PloS one 9, e86085 (2014).
Ulian, C. M. V. & Rossi, M. N. Intraspecific variation in body size and sexual size dimorphism, and a test of Rensch’s rule in bats. Acta Zoologica 98, 37–386 (2017).
Trivers, R. Parental investment and sexual selection (1972).
Darwin, C. The Descent of Man, and Selection in Relation to Sex: Reprinted from the Second English Edition, Revised and Augmented (Burt, 1874).
Ghiselin, M. T. The economy of nature and the evolution of sex. (University of California Press, 1974).
Clutton-Brock, T. H., Harvey, P. H. & Rudder, B. Sexual dimorphism, socionomic sex ratio and body weight in primates. Nature 269, 797–800 (1977).
Lindenfors, P., Gittleman, J. L. & Jones, K. E. Sexual size dimorphism in mammals. Sex, size and gender roles: evolutionary studies of sexual size dimorphism, 16–26 (2007).
Simmons, L. W. & Firman, R. C. Experimental evidence for the evolution of the Mammalian baculum by sexual selection. Evolution 68, 276–283 (2014).
Hosken, D. J. & Stockley, P. Sexual selection and genital evolution. Trends in Ecology & Evolution 19, 87–93 (2004).
Stockley, P. et al. Baculum morphology predicts reproductive success of male house mice under sexual selection. BMC biology 11, 66 (2013).
Dines, J. P. et al. A trade‐off between precopulatory and postcopulatory trait investment in male cetaceans. Evolution 69, 1560–1572 (2015).
Fitzpatrick, J. L. & Simmons, L. W. Male contest competition and the coevolution of weaponry and testes in pinnipeds. Evolution 66, 3595–3604 (2012).
Selander, R. K. Sexual Dimorphism and Differential Niche Utilization in Birds. Condor 68, 113–151 (1966).
Butler, M. A., Schoener, T. W. & Losos, J. B. The relationship between sexual size dimorphism and habitat use in Greater Antillean anolis lizards. Evolution 54, 259–272 (2000).
Ceballos, C. P., Adams, D. C., Iverson, J. B. & Valenzuela, N. Phylogenetic patterns of sexual size dimorphism in Turtles and their implications for Rensch’s rule. Evolutionary Biology 40, 1–15 (2013).
Simmons, N. B. Order Chiroptera. In: Wilson, D. E. and D. M. Reeder (eds), Mammal Species of the World, 3rd ed., (Smithsonian Institution Press, 2005).
Nowak, M. R. & Paradiso, L. J. Walker’s Mammals of the World. (Johns Hopkins University, 1983).
Csorba, G., Ujhelyi, P. & Thomas, N. Horseshoe bats of the world:(Chiroptera: Rhinolophidae). (Alana books, 2003).
Jacobs, D. S., Barclay, R. M. & Walker, M. H. The allometry of echolocation call frequencies of insectivorous bats: why do some species deviate from the pattern? Oecologia 152, 583–594 (2007).
Salsamendi, E. et al. Foraging Ecology in Mehely’s Horseshoe Bats: Influence of Habitat Structure and Water Availability. Acta Chiropterologica 14, 121–132 (2016).
Matějů, J. & Kratochvíl, L. Sexual size dimorphism in ground squirrels (Rodentia: Sciuridae: Marmotini) does not correlate with body size and sociality. Frontiers in zoology 10, 1–10 (2013).
Liao, W. B. Evolution of Sexual Size Dimorphism in a Frog Obeys the Inverse of Rensch’s Rule. Evolutionary Biology 40, 1–7 (2012).
Hirst, A. G. & Kiørboe, T. Macroevolutionary patterns of sexual size dimorphism in copepods. Proceedings of the Royal Society of London B: Biological Sciences 281, 20140739 (2014).
Dool, S. E. et al. Nuclear introns outperform mitochondrial DNA in inter-specific phylogenetic reconstruction: Lessons from horseshoe bats (Rhinolophidae: Chiroptera). Molecular Phylogenetics & Evolution 97, 196–212 (2016).
Rossiter, S. J., Ransome, R. D., Faulkes, C. G., Dawson, D. A. & Jones, G. Long-term paternity skew and the opportunity for selection in a mammal with reversed sexual size dimorphism. Molecular Ecology 15, 3035–3043 (2006).
Crichton, E. G. & Krutzsch, P. H. Reproductive biology of bats. (Academic Press, 2000).
Puechmaille, S. J. et al. Female mate choice can drive the evolution of high frequency echolocation in bats: A case study with Rhinolophus mehelyi. PLoS ONE 9, e103452 (2014).
de Camargo, N. F. & de Oliveira, H. F. M. Sexual Dimorphism in Sturnira lilium (Chiroptera, Phyllostomidae): Can Pregnancy and Pup Carrying Be Responsible for Differences in Wing Shape? PLoS ONE 7, e49734 (2012).
Williams, D. F. & Findley, J. S. Sexual size dimorphism in vespertilionid bats. American Midland Naturalist 102, 113–126 (1979).
Schoener, T. W. In Biology of the Reptilia Vol. 4 (eds C. Gans & D. Tinkle) Ch. Competition and the niche, 35–136 (Academic Press, 1977).
Dechmann, D. K. N. et al. Tracking post-hibernation behavior and early migration does not reveal the expected sex-differences in a “Female-Migrating” bat. PLoS ONE 9, e114810 (2014).
Schoener, T. W. Models of Optimal Size for Solitary Predators. American Naturalist 103, 277–313 (1969).
Neuweiler, G. & Covey, E. The biology of bats. (Oxford University Press, 2000).
Kunz, T. H. & Fenton, M. B. Bat ecology. (University of Chicago Press, 2003).
Neuweiler, G. et al. Foraging Behaviour and Echolocation in the Rufous Horseshoe Bat (Rhinolophus rouxi) of Sri Lanka. Behavioral Ecology and Sociobiology 20, 53–67 (1987).
Crowther, T. W. et al. Mapping tree density at a global scale. Nature 525, 201–205 (2015).
Dinnage, R., Cadotte, M. W., Haddad, N. M., Crutsinger, G. M. & Tilman, D. Diversity of plant evolutionary lineages promotes arthropod diversity. Ecology Letters 15, 1308–1317 (2012).
Ferrandiz-Rovira, M., Lemaître, J.-F., Lardy, S., López, B. C. & Cohas, A. Do pre-and post-copulatory sexually selected traits covary in large herbivores? BMC evolutionary biology 14, 1 (2014).
Gage, M. J., Parker, G. A., Nylin, S. & Wiklund, C. Sexual selection and speciation in mammals, butterflies and spiders. Proceedings of the Royal Society of London B: Biological Sciences 269, 2309–2316 (2002).
Fitzpatrick, J. L., Almbro, M., Gonzalez‐Voyer, A., Kolm, N. & Simmons, L. W. Male contest competition and the coevolution of weaponry and testes in pinnipeds. Evolution 66, 3595–3604 (2012).
Voigt, C. C., Heckel, G. & Mayer, F. Sexual selection favours small and symmetric males in the polygynous greater sac-winged bat Saccopteryx bilineata (Emballonuridae, Chiroptera). Behavioral Ecology and Sociobiology 57, 457–464 (2005).
Hosken, D., Jones, K., Chipperfield, K. & Dixson, A. Is the bat os penis sexually selected? Behavioral Ecology and Sociobiology 50, 450–460 (2001).
Cheverud, J. M., Dow, M. M. & Leutenegger, W. The quantitative assessment of phylogenetic constraints in comparative analyses: sexual dimorphism in body weight among primates. Evolution 39, 1335–1351 (1985).
Lovich, J. E. & Gibbons, J. W. A review of techniques for quantifying sexual size dimorphism. Growth, development, and aging: GDA 56, 269 (1992).
Smith, R. J. Statistics of sexual size dimorphism. Journal of Human Evolution 36, 423–459 (1999).
Pavey, C. R. Habitat use by the eastern horseshoe bat, Rhinolophus megaphyllus, in a fragmented woodland mosaic. Wildlife Research 25, 489–498 (1998).
Jones, G. & Rayner, J. M. V. Foraging behavior and echolocation of wild horseshoe bats Rhinolophus ferrumequinum and R. hipposideros (Chiroptera, Rhinolophidae). Behavioral Ecology & Sociobiology 25, 183–191 (1989).
Monadjem, A., Taylor, P. J., Cotterill, W. & Schoeman, M. Bats of southern and central Africa: a biogeographic and taxonomic synthesis. (Wits University Press Johannesburg, 2010).
Stoffberg, S., Jacobs, D. S. & Matthee, C. A. The divergence of echolocation frequency in horseshoe bats: moth hearing, body size or habitat? Journal of Mammalian Evolution 18, 117–129 (2011).
Jacobs, D. S. Intraspecific variation in wingspan and echolocation call flexibility might explain the use of different habitats by the insectivorous bat, Miniopterus schreibersii (Vespertilionidae: Miniopterinae). Acta Chiropterologica 1, 93–103 (1999).
Lee, Y. F. et al. Ecomorphology, differentiated habitat use, and nocturnal activities of Rhinolophus and Hipposideros species in East Asian tropical forests. Zoology 115, 22–29 (2012).
Webb, T. J. & Freckleton, R. P. Only half right: species with female-biased sexual size dimorphism consistently break Rensch’s rule. PLoS One 2, e897 (2007).
Björklund, M. A phylogenetic interpretation of sexual dimorphism in body size and ornament in relation to mating system in birds. Journal of Evolutionary Biology 3, 171–183 (1990).
Fairbairn, D. J. Allometry for sexual size dimorphism: pattern and process in the coevolution of body size in males and females. Annual Review of Ecology and Systematics 28,659–687 (1997).
Warton, D. I., Duursma, R. A., Falster, D. S. & Taskinen, s smatr 3 - an R package for estimation and inference about allometric lines. Methods in Ecology and Evolution 3, 257–259 (2012).
Luo, B. et al. Social call divergence in bats: a comparative analysis. Behavioral Ecology 28, 533–540 (2017).
Thompson, J. D., Higgins, D. G. & Gibson, T. J. CLUSTAL W: Improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Research 22, 4673–4680 (1994).
Posada, D. & Crandall, K. A. Modeltest: testing the model of DNA substitution. Bioinformatics 14, 817–818 (1998).
Paradis, E. et al. Package ‘ape’. Analyses of phylogenetics and evolution, version 2, 4–1 (2015).
Revell, L. J. phytools: an R package for phylogenetic comparative biology (and other things). Methods in Ecology and Evolution 3, 217–223 (2012).
Pagel, M. Inferring evolutionary processes from phylogenies. Zoologica Scripta 26, 331–348 (1997).
Pagel, M. Inferring the historical patterns of biological evolution. Nature 401, 877–884 (1999).
Blomberg, S. P., Garland, T. & Ives, A. R. Testing for phylogenetic signal in comparative data: behavioral traits are more labile. Evolution 57, 717–745 (2003).
Freckleton, R. P., Harvey, P. H. & Pagel, M. Phylogenetic analysis and comparative data: a test and review of evidence. The American Naturalist 160, 712–726 (2002).
Harmon, L. J., Weir, J. T., Brock, C. D., Glor, R. E. & Challenger, W. GEIGER: investigating evolutionary radiations. Bioinformatics 24, 129–131 (2008).
Team, R. C. R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing, Vienna, Austria (2013).
Kock, D., Csorba, G. & Howell, K. M. Rhinolophus maendeleo n. sp. from Tanzania, a horseshoe bat noteworthy for its systematics and biogeography (Mammalia, Chiroptera, Rhinolophidae). Senckenbergiana biologica 80, 233–240 (2000).
Goiti, U., Garin, I., Almenar, D., Salsamendi, E. & Aihartza, J. Foraging by Mediterranean horseshoe bats (Rhinolophus euryale) in relation to prey distribution and edge habitat. Journal of Mammalogy 89, 493–502 (2008).
Dietz, C., Helversen, O. V. & Nill, D. Bats of Britain, Europe & Northwest Africa. (A&C Black, 2009).
Hill, J. A record of Rhinolophus arcuatus (Peters, 1871)(Chiroptera: Rhinolophidae) from Sulawesi. Mammalia 52, 588–589 (1988).
Ith, S. et al. A taxonomic review of Rhinolophus coelophyllus Peters 1867 and R. shameli Tate 1943 (Chiroptera: Rhinolophidae) in continental Southeast Asia. Acta Chiropterologica 13, 41–59 (2011).
Robson, S. K., Inkster, T. E. & Krockenberger, A. K. Bats of the YUS Conservation Area, Papua New Guinea: Result 5 Task 3.2. (2012).
Lanza, B. Notizie sull’osso peniale dei chirotteri europei e su alcuni casi di parallelismo morfologico. Monitore Zoologico Italiano 67, 3–14 (1959).
Zhou, Z.-M. et al. A new species from southwestern China in the Afro-Palearctic lineage of the horseshoe bats (Rhinolophus). Journal of Mammalogy 90, 57–73 (2009).
Benda, P. On a small collection of bats (Chiroptera) from western Sabah (North Borneo, East Malaysia). Vespertilio 13, 45–76 (2010).
Matveev, V. A. Checklist of Cambodian bats (Chiroptera), with new records and remarks on taxonomy. Russian Journal of Theriology 4, 43 (2005).
Bates, P. J. et al. A review of Rhinolophus (Chiroptera: Rhinolophidae) from Myanmar, including three species new to the country. Acta Chiropterologica 6, 23–48 (2004).
Soisook, P. et al. A taxonomic review of Rhinolophus stheno and R. malayanus (Chiroptera: Rhinolophidae) from continental Southeast Asia: an evaluation of echolocation call frequency in discriminating between cryptic species. Acta Chiropterologica 10, 221–242 (2008).
Esselstyn, J. A., Widmann, P. & Heaney, L. R. The mammals of Palawan island, Philippines. Proceedings of the Biological Society of Washington 117, 271–302 (2004).
Borissenko, A. V. K. Bats of Vietnam and adjacent territoriesan identification manua l (2003).
Yoshino, H. et al. Geographical variation in echolocation call and body size of the Okinawan least horseshoe bat, Rhinolophus pumilus (Mammalia: Rhinolophidae), on Okinawa-jima Island, Ryukyu Archipelago, Japan. Zoological science 23, 661–667 (2006).
Pearch, M., Khin, M., Pjj, B., Tin Nwe, K. M. S. & Si Si Hla, B. First record of bats (Chiroptera) from Rakhine State, Myanmar (Burma). Natural History Bulletin of the Siam Society 51, 241–259 (2003).
Bates, P. J. J. & Harrison, D. L. Bats of the Indian subcontinent. (ETI, University of Amsterdam, 2000).
Acknowledgements
We thank the students of Jilin Provincal Key Laboratory of Animal Resource Conservation and Utilization from Northeast Normal University for assistance with data collection in the field. This research was supported by the National Natural Science Foundation of China (Grant Nos 31700331, 31470457, 31670390, 31270414) and the Fundamental Research Funds for the Central Universities (No. 2412016KJ045). We thank LetPub for its linguistic assistance during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
H.W. designed the study, collected the data, implemented most genetic methodology, and wrote the manuscript. T.J. assisted with study design, sample collection, and gave important comments on multiple versions of the manuscript. X.H. assisted with sample collection. J.F. assisted with study design, and provided laboratory space and some funding.
Corresponding authors
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Wu, H., Jiang, T., Huang, X. et al. Patterns of sexual size dimorphism in horseshoe bats: Testing Rensch’s rule and potential causes. Sci Rep 8, 2616 (2018). https://doi.org/10.1038/s41598-018-21077-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-21077-7
This article is cited by
-
Sexual dimorphism in toothed whales (Odontoceti) follows Rensch’s rule
Mammalian Biology (2022)
-
Coexistence of two sympatric cryptic bat species in French Guiana: insights from genetic, acoustic and ecological data
BMC Evolutionary Biology (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.