Abstract
The cerebellum can influence the responsiveness of the primary motor cortex (M1) to undergo spike timing-dependent plastic changes through a complex mechanism involving multiple relays in the cerebello-thalamo-cortical pathway. Previous TMS studies showed that cerebellar cortex excitation can block the increase in M1 excitability induced by a paired-associative stimulation (PAS), while cerebellar cortex inhibition would enhance it. Since cerebellum is known to be affected in many types of dystonia, this bidirectional modulation was assessed in 22 patients with cervical dystonia and 23 healthy controls. Exactly opposite effects were found in patients: cerebellar inhibition suppressed the effects of PAS, while cerebellar excitation enhanced them. Another experiment comparing healthy subjects maintaining the head straight with subjects maintaining the head turned as the patients found that turning the head is enough to invert the cerebellar modulation of M1 plasticity. A third control experiment in healthy subjects showed that proprioceptive perturbation of the sterno-cleido-mastoid muscle had the same effects as turning the head. We discuss these finding in the light of the recent model of a mesencephalic head integrator. We also suggest that abnormal cerebellar processing of the neck proprioceptive information drives dysfunctions of the integrator in cervical dystonia.
Similar content being viewed by others
Introduction
Primary dystonia is in all likelihood a developmental disorder of sensorimotor circuits, involving both the cortico-striato-pallido-thalamo-cortical and cerebello-thalamo-cortical pathways1. Primary cervical dystonia (CD) is a focal dystonia characterized by involuntary posturing of the head in any of the three axes.
The two pathophysiological features of dystonia, i.e. the abnormal propensity of the motor cortex to develop plasticity and the abnormal somatosensory processing are considered as part of a common endophenotype favoring dystonia yet not directly involved in generating abnormal dystonic movements. Indeed, the cortical plasticity of the motor cortex per se was abnormal within the hand representation in writer’s cramp2,3 and in CD4,5, irrespective of whether the hand representation mapped a dystonic or non-dystonic body part. Abnormalities of somatosensory processing were noted in the non-affected body parts of patients with unilateral dystonia, in patients with CD and blepharospasm, and also in their unaffected relatives6,7,8,9,10. In keeping with the reasoning that such abnormalities are endophenotypic markers but not directly linked to symptoms, deep brain stimulation of the globus pallidus internus in CD alleviates only the dystonic symptoms, but not the somatosensory processing in patients11.
When the dynamic relation between the cerebellum and M1 was explored either with double-pulses12 or with two consecutive repetitive transcranial magnetic stimulations13 in patients with writer’s cramp (one over the cerebellum followed by one over M1), no effective output was found from the cerebellum to M1 for the dystonic hand representation. This also seemed to play a direct role in generating dystonic symptoms13.
We reasoned that if the defective cerebellar modulation of M1 plasticity participates in the hand cramping in focal hand dystonia, the same modulation might be spared in cervical dystonia patients whose hands are normal. We investigated how modulation of the cerebellar output influences the plasticity of the hand representation in the motor cortex in cervical dystonia patients.
The cortical plasticity was investigated using a rapid, excitatory paired associative stimulation (PAS) protocol14,15, and the cerebellar output was modulated by decreasing or increasing cerebellar cortex excitability by means of repetitive transcranial magnetic stimulation (continuous (cTBS) or intermittent (iTBS) theta-burst protocols)16. Since the head of every patient was turned spontaneously throughout the experiment, the healthy controls were recorded either with the heads straight or voluntarily turned. To further disentangle the role of neck sensory inputs during head turn, we also recorded the healthy volunteers with the head straight during vibration of a neck rotator muscle.
Results
Cerebellar control in normal straight head versus dystonic position
We compared two groups of subjects (one of patients with cervical dystonia (CD) manifesting as a right-sided torticollis and one of age- and gender-matched healthy controls, Table 1) in three different conditions (Fig. 1A) described before16,17: continuous theta-burst stimulation over the right cerebellum (cTBSCB), intermittent theta-burst stimulation over the right cerebellum (iTBSCB), and sham stimulation of the cerebellum (ShamCB), all three followed by a rapid paired-associative stimulation (PAS). Throughout these recordings, the CD did not constrain the position of their head, letting it rest in the dystonic position, while controls maintained the head in a neutral, straight position.
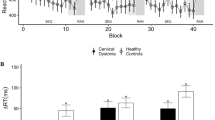
Experimental design and the comparison between healthy controls and patients with cervical dystonia. (A) Cortical excitability was quantified with the amplitude of motor potentials evoked with TMS pulses from the primary motor cortex, before and 10, 20, and 30 minutes after the repetitive stimulation. The repetitive stimulation consisted in real or sham stimulation of the cerebellum followed by PAS over M1 and median nerve. (B) The effect of the combined cerebellar and cortical stimulation in healthy controls and cervical dystonia patients are opposite. The effects are evident both when the groups are analyzed as a whole or split in responder and non-responders to PAS. cTBSCB: continuous theta-burst stimulation over the right cerebellar hemisphere; iTBSCB: intermittent theta-burst stimulation over the right cerebellar hemisphere; MEP: motor evoked potential; PAS: paired-associative stimulation; T0-T30: time points before the stimulation and at 10, 20, 30 minutes post-stimulation.
The resting and active motor thresholds, and the size of the motor-evoked potentials (MEP) before any intervention did not differ between the two groups (Table 2). When the subjects were divided in only two groups, CD patients and controls (Fig. 1B), the ANOVA did not reveal any effect of individual factors (GROUP: CD and controls; INTERVENTION: ShamCB → PAS, cTBSCB → PAS, and iTBSCB → PAS; TIME: T10, T20, T30), but one significant interaction (rmANOVA: GROUP: F1,42 = 0.01, P = 0.93; INTERVENTION: F2,42 = 1.2, P = 0.32; TIME: F2,42 = 0.1, P = 0.82; INTERVENTION * GROUP: F2,84 = 12.9, P < 0.0001). This suggests that the way in which the groups differ in their response to each intervention is more complex than what can be captured by this direct comparison.
The basic response to PAS is known to be variable in the general population18,19,20. Following the previously validated approach to enhance the description accuracy of plastic phenomena19,21, we separated the subjects in responders and non-responders to PAS.
Responders to ShamCB → PAS
The effects of the 3 interventions differed (Fig. 1B) between CD patients and the controls (rmANOVA: GROUP: F1,24 = 1.1 P = 0.3; INTERVENTION: F2,48 = 1.7, P = 0.02; TIME: F2,48 = 0.1, P = 0.9; INTERVENTION * GROUP: F2,50 = 58.7, P = 0.0006).
The post-hoc comparison of the INTERVENTION * GROUP interaction showed that the CD patients and the controls respond differently to each intervention. Unpaired t-test for ShamCB → PAS revealed that the overall PAS effect in responder patients was significantly higher (CD vs controls: 1.53 ± 0.08 vs 1.33 ± 0.06, P = 0.049). This is in line with the previous reports that when a significant PAS effect is measured, it is stronger in dystonic patients4,22. Unpaired t-test for cTBSCB → PAS (CD vs controls: 1.02 ± 0.08 vs 1.44 ± 0.07, P = 0.0003) and iTBSCB → PAS (CD vs controls: 1.48 ± 0.1 vs 0.99 ± 0.05, P = 0.0001) revealed exact opposite responses in the two groups.
Non-responders to ShamCB → PAS
The effects of the 3 interventions differed (Fig. 1B) between the CD patients and the controls, despite their “apparent” non-responsiveness to PAS (rmANOVA: GROUP: F1,17 = 1.2, P = 0.3; INTERVENTION: F2,34 = 4.4, P = 0.02; TIME: F2,34 = 0.4, P = 0.7; INTERVENTION * GROUP: F2,34 = 5.7, P = 0.007; INTERVENTION * TIME: F4,68 = 3.1, P = 0.02).
The post-hoc analysis of the INTERVENTION * GROUP interaction showed that the CD patients and the controls respond differently to each intervention. Unpaired t-test for ShamCB → PAS revealed that the overall PAS effect was similarly low in both groups (CD vs controls: 0.97 ± 0.03 vs 0.88 ± 0.04, P = 0.1). This was expected, since both sub-groups were non-responders to PAS according to the selection criteria. Unpaired t-test for cTBSCB → PAS (CD vs controls: 0.98 ± 0.06 vs 1.73 ± 0.2, P = 0.0008) and iTBSCB → PAS (CD vs controls: 1.10 ± 0.07 vs 0.90 ± 0.06, P = 0.03) revealed exact opposite responses in the two groups. It is noteworthy that cTBSCB can restore the PAS responsiveness in non-responder healthy controls, as previously described19.
The post-hoc analysis of the INTERVENTION * TIME interaction did not reveal any significant effect of TIME during any intervention (rmANOVA with TIME as repeated factor for ShamCB → PAS: F2,18 = 0.5, P = 0.64; cTBSCB → PAS: F2,18 = 3.3, P = 0.051; iTBSCB → PAS: F2,18 = 1.9, P = 0.17), but a significant effect of INTERVENTION at the 2nd and 3rd time points, which was expected because the rTMS after-effects need ~10–15 min to reach their maximum level when they are not suppressed (paired t-tests at T10: Sham vs iTBSCB P = 0.35, Sham vs cTBSCB P = 0.17, iTBSCB vs cTBSCB P = 0.43; at T20: Sham vs iTBSCB P = 0.70, Sham vs cTBSCB P = 0.02, iTBSCB versus cTBSCB P = 0.02; at T30: Sham vs iTBSCB P = 0.20, Sham vs cTBSCB P = 0.02, iTBSCB versus cTBSCB P = 0.07).
Cerebellar control in normal straight head versus normal, voluntary turned head position
In order to determine whether the newly identified differences in cerebellar modulation of motor cortex plasticity were specific to cervical dystonia or normally observed during head turning, we designed an additional experiment involving only PAS-responder, healthy subjects.
Two groups of 12 healthy subjects matched for age and gender underwent the same three interventions as in the previous experiment with the only difference being the different head-trunk alignment: one group kept the head in a neutral position facing forward throughout the stimulation and recording periods, while the other group maintained the head turned towards the right shoulder throughout the stimulation and recording periods.
The resting and active motor thresholds, and the MEP amplitudes before any intervention did not differ between the two groups (Table 2).
The effects of the 3 interventions differed according to the head position (POSITION: F1,22 = 0.5, P = 0.5; INTERVENTION: F2,44 = 0.5, P = 0.6; TIME: F2,44 = 4.7, P = 0.01; INTERVENTION*POSITION: F2,44 = 10.5, P = 0.0002; INTERVENTION * TIME * POSITION: F4,88 = 2.5, P = 0.051).
The post-hoc analysis of the INTERVENTION * POSITION interaction showed that the two head positions induce opposite results (Fig. 2), similar to what was observed between CD patients and controls. Unpaired t-test revealed that the overall effects of the ShamCB → PAS did not differ between subjects maintaining the head turned and subjects maintaining the head straight (P = 0.67). Active cerebellar conditioning however, differed according to the head position for both iTBSCB → PAS (P = 0.004) and cTBSCB → PAS (P < 0.0001). The group maintaining the head straight reproduced the previous findings16, i.e. cTBSCB significantly enhanced, while iTBSCB prevented any plastic effect of PAS (paired t-tests ShamCB → PAS vs cTBSCB → PAS P = 0.003, ShamCB → PAS vs iTBSCB → PAS P = 0.005, iTBSCB → PAS vs cTBSCB → PAS P < 0.0001). In the group of subjects maintaining the head turned, the cerebellar effects were inverted, just as in the CD patients: cTBSCB suppressed the PAS effects instead of enhancing it, while iTBSCB only appears to enhance the PAS effects, though not significantly because of the high variability of the small cohort (paired t-test: ShamCB → PAS vs cTBSCB → PAS P = 0.006, ShamCB → PAS vs iTBSCB → PAS P = 0.2, iTBSCB → PAS vs cTBSCB → PAS P = 0.003).
Cerebellar stimulation effects on motor cortex plasticity in healthy volunteers. Continuous theta-burst stimulation of the cerebellum enhances the PAS effect in responsive healthy subjects with the head straight, but blocks it when the head is turned. Intermittent theta-burst of the cerebellum has the exact opposite effect: it blocks the PAS effect in responsive healthy subjects with the head straight, but enhances it when the head is turned.
Cerebellar control in normal straight head position with and without vibration of neck muscles
In order to assess the role of neck proprioception in the inversion of cerebellar modulation of motor cortex plasticity, we have performed a control experiment on a small group of six healthy volunteers. They underwent vibration of the left sterno-cleido-mastoid (SCM) muscle while maintaining the head in the straight, neutral position. This was supposed to induce the illusion of a head rotation towards the contralateral side. In this control recording, we measured only the effect of cTBSCB → PAS (as the paradigm with the biggest change). The results were compared to the cTBSCB → PAS data recorded in age- and gender-matched healthy subjects from each of the responder groups in the experiment above (i.e., six maintaining the head straight and six maintaining the head turned).
The cTBSCB enhanced the responsiveness to PAS in the group with the head in the midline, but decreased it to the same extent in the other two groups (Fig. 3), the one maintaining the head turned and the one with the head in the midline but receiving SCM vibration (GROUP: F2,15 = 4.4, P = 0.03, TIME: F2,30 = 2.8, P = 0.07; GROUP*TIME: F4,30 = 0.9, P = 0.5; post-hoc unpaired t-tests: head straight vs vibration P = 0.001, head straight vs head turned P < 0.0001, vibration vs head turned P = 0.78).
Control experiment on healthy volunteers. Continuous theta-burst stimulation applied over the right cerebellum enhanced the effect of PAS in responsive healthy subjects with the head kept straight, but blocked it in subjects with the head turned or undergoing vibration of the left sterno-cleido-mastroid muscle.
Discussion
These results demonstrate for the first time that proprioceptive feedback from the neck plays a crucial role in the way in which the cerebellum can influence the plasticity of the primary motor cortex. We investigated the cerebellar influence over the M1 spike time-dependent plasticity in cervical dystonia patients with the head in a spontaneous dystonic position and age-matched healthy controls maintaining their heads either straight or voluntarily rotated to mimic the position of a right rotational torticollis. In healthy subjects maintaining their head straight in a neutral position, cTBS over the lateral cerebellum enhanced the contralateral M1 responsiveness to paired associative stimulation, while iTBS over the lateral cerebellum blocked it entirely, confirming previous reports16,21. When taking into account the individual responsiveness to PAS alone, these effects resulted more nuanced: in responders, cTBSCB did not enhance the PAS effect significantly above the baseline response to PAS, while in non-responders, cTBSCB did enhance it. The opposite was true for the iTBSCB: in responders, it effectively blocked the baseline response to PAS, while in non-responders, no significant effect could be observed since the PAS response was absent by default. Inversely, in CD patients, healthy controls maintaining the head rotated, and healthy controls maintaining the head straight during vibration of SCM, cTBSCB and iTBSCB had completely opposite effects on PAS: cTBSCB blocked PAS effects and iTBSCB enhanced them. The same nuances related to the responder/non-responder classification could be observed in CD patients, suggesting that the mirroring phenomenon is wide-reaching. This observation alone suggests that CD is a separate entity, in which the cerebellar output reaches and influences the motor circuits, unlike focal hand dystonia in which cerebellar output is virtually absent12,13.
The cerebellum is known to integrate multiple reference frames for motion by supporting a vestibular-somatosensory convergence23,24. However, most of the previous animal studies have concentrated on the multimodal integration of head-on-body and head-in-space with or without simultaneous whole-body rotation during an actual movement of the head. We have performed all recordings with the subjects maintaining a stationary position of the head either in their subjective “straight ahead” or in a voluntarily vertical rotation. This allowed us to exclude any confounding vestibular or fluctuating proprioceptive inputs. Changing the position of the head alone inverted the cerebellar effect on M1 plasticity but not the M1 plasticity per se, in all cases in which the head was rotated voluntarily, involuntarily, or illusorily.
The cerebellar TBS likely affects only the cerebellar cortex25 and particularly the horizontally orientated parallel fibers in the more superficial cerebellar molecular layer projecting on the Purkinje cells26,27. These parallel fiber-Purkinje cell synapses can undergo both postsynaptically expressed long-term depression or long-term potentiation, depending on whether or not the climbing fiber input is coactivated during tetanization, in which case the climbing fiber activity acts as a polarity switch28. Moreover, this differentiated plastic effect seems to coexist for different processes29,30. Considering that the molecular layer is where most of the multimodal inputs converge within the cerebellum31,32, we can infer that the neck proprioception alone might play a key role in the potentiation and the responsiveness of the parallel fiber-Purkinje cell synapses. A change in the responsiveness of these synapses might thus explain how the effects of cTBSCB and iTBSCB are inverted when the head is turned.
In order to minimize confounding factors, we have recruited only right-handed subjects and patients with predominantly right torticollis, and asked the healthy controls to maintain head rotation towards right. Despite mechanical differences between involuntary head rotations of the CD patients (manifested as complex movement, i.e. torticollis / laterocollis / anterocollis / retrocollis) and simple voluntary head rotation of the healthy controls, the direction and extent of the cerebellar priming of PAS effects were similar. This is in contrast with recordings in writer’s cramp patients, in whom both types of cerebellar stimulation did not influence PAS effects13,33.
In 2002, Klier and collaborators introduced the notion of a head and eye neural integrator34. They demonstrated in macaques that inactivation and stimulation of interstitial nucleus of Cajal (INC) cause head postures and oscillations resembling cervical dystonia: (1) left INC inactivation with muscimol resulted in right laterocollis and left torticollis, while right INC inactivation caused left laterocollis and right torticollis; (2) electrical excitation of INC resulted in head position changes exactly opposite of what were found with inactivation. This led to the idea that the postural symptoms of cervical dystonia in humans could be explained by severely imbalanced activity within such an integrator, either from intrinsic dysfunction or from an imbalance of inputs, or a combination of these two34,35. Shaikh and colleagues have further expanded this concept demonstrating in humans the existence of a leaky head neural integrator in the midbrain36,37. The authors brought compelling arguments that malfunctions in any of the inputs to this head neural integrator can lead to abnormalities resembling CD. This concept is in agreement with and represents a solid convergence point for most classical and recent models pointing to dysfunctions in the cerebellum, basal ganglia, or neck proprioception. Therefore, the new model also emphasizes that dystonia is a clinical syndrome but with heterogeneity in the underlying biological causes.
Our results show that, in healthy subjects, proprioceptive input from the neck can substantially change the way in which cerebellar output influences M1 plasticity. Both artificially triggering this input by vibration and physiologically triggering it by head rotation leads to inhibitory stimulation of the cerebellar cortex to prevent PAS plasticity in M1. The fact that cerebellar cTBS had the same effect in CD patients in their own neutral head position suggests that the cerebello-thalamo-cortical communication might exist in CD, unlike in focal hand dystonia, but “locked” in abnormal processing of proprioceptive inputs. This is in line with the finding that external visuo-motor and force-field adaptations, both involving cerebellum, are normal in CD patients38, but abnormal when estimation of one’s own body-schema is involved39,40. It is not clear though whether the changes of the parallel fibers-Purkinje cells synapses are intrinsic to the cerebellum (and thus part of the pathophysiology of the disease) or brought about by secondary inputs (from neck proprioceptors and/or basal ganglia via the pontine nuclei). This leaves open the possibility that cerebellar stimulations employed here (both cTBS and iTBS) elicit different plastic changes from those elicited in healthy volunteers.
For a long time, basal ganglia were considered as the main structure generating dystonic symptoms. Indeed, asymmetric local field potentials in the pallidum were recently reported, suggesting an asymmetry in pallidal outflow41,42. Given that the cerebellum and basal ganglia are strongly interconnected43,44 and that dysfunction in one can entrain dysfunction in the other45, a new question arises: is this asymmetric outflow part of the abnormal input only towards the mesencephalic neural integrator of the head or is it the result of a constantly abnormal outflow from the cerebellum towards the basal ganglia? In either case, a palliative treatment approach such as DBS targeting the GPi seems to give positive results in CD46.
Neck proprioception plays a crucial role in both limb coordination and representation of the body-scheme47,48. In healthy subjects, neck muscle vibration induces disparity between subjective perception and objective position of the body midline36,49,50,51. Subjects with chronic neck pain show consistent impairment of joint position sense52. Patients with cervical dystonia display significant kinematic abnormalities in non-dystonic segments, which can be corrected with botulinum toxin injections in the neck muscles39. In our study, vibration of the sternocleidomastoid muscle inverted the effect of the cerebellar cTBS on PAS in the same way head turning did, i.e. cancelling the PAS effect instead of amplifying it. The small but fast vibration cycles almost selectively induce a one-to-one train of action potentials in the primary endings of the large-diameter group Ia afferent fibers53,54, which would eventually induce the illusion of active contraction of the respective muscle55 and the subsequent rotational illusion of the head towards right in our study56. It is known that neck proprioception is relayed to the cerebellar anterior vermis in cats57,58, but the influence over the cerebellar output towards M1 hand area suggests that its weight throughout the cerebellum might be wider than anticipated. It is worth noting that CD patients had the same inversion of the cerebellar effects on M1 plasticity at small head rotation angles (TWSTRS score for rotation: 1.5 ± 1.2) as the healthy controls had at large rotations. This suggests that the cerebellar circuits in CD might be hyper-excitable, over-responding to proprioceptive information that could be dismissed as noise in healthy subjects.
Repetitive TMS (both inhibitory and excitatory) over cerebellum, M1, or premotor cortex was reported to improve dystonic symptoms in CD22,59,60,61,62, albeit not always33. Considering the apparently intact cerebello-cortical communication and the wide range of simulations resulting in positive clinical effect, it is possible that the non-invasive stimulations are acting through breaking a self-enforcing and/or self-sustaining chain of abnormal integration of proprioceptive feedback. It remains now to determine whether this cycle concerns only the priming of motor plasticity or other processes as well, like other aspects of the motor control, sensory computations, or even cognitive processes.
In summary, these data are compatible with the novel conceptual framework for the pathogenesis of cervical dystonia based on an abnormal dialogue between the mesencephalic head neural integrator and its inputs. Among these inputs, proprioception, the cerebellum, and basal ganglia were cited as key sources of feedback1,36,40,63. Our results suggest that a likely abnormal integration of the neck proprioceptive information at the cerebellar level subsequently drives an abnormal functioning of the integrator, which creates a head turn and feeds a vicious circle during the manifestation of the dystonic phenomenon.
This framework suggests that novel therapies for cervical dystonia such as chronic electrical stimulation of the cerebellum might be only palliative, while modulation of proprioception using vibration or electrocutaneous stimulation devices might be more suitable.
Methods
Procedures
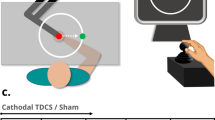
The subjects were seated comfortably in an armchair, with both hands resting symmetrically on a pillow in their lap. The motor “hot-spot” for Abductor pollicis brevis (APB) muscle was identified in the left motor cortex and marked on the default brain reconstruction with the help of an MRI-based neuronavigation system (eXimia 2.2.0, Nextim Ltd in the French lab; BrainSight 2, Rogue Resolutions in the Indian lab). It allowed us to maintain the placement and tilt of the stimulator coil throughout each session and from one session to the next in the same subject. MEPs were recorded in the right APB with disposable Ag/AgCl surface electrodes in a muscle belly–tendon montage. Responses were amplified (1000x) and filtered (100–3000 Hz) with a Digitimer D360 amplifier (Digitimer Ltd, Welwyn Garden City, UK), then digitally transformed with 10 kHz sampling rate (CED Power 1401MkII, CED Ltd., Cambridge, UK) and stored offline for analysis (Signal 4.02, CED Ltd., Cambridge, UK). The EMG activity was continuously monitored to ensure muscle relaxation. Trials contaminated by EMG activity anywhere within 500 ms around each MEP were discarded from the offline analysis.
Each experiment started by calculating the APB resting (RMT) and active (AMT) motor thresholds according to the standard procedure64. This was done using both the Magstim 200 and the SuperRapid2 magnetic stimulators (Magstim Company, Whitland, Wales, UK).
Cerebellar stimulation
Right cerebellar lobule VIII was chosen as target, since it is part of the sensorimotor network65 and readily accessible to TMS. Previous studies16,25,66 verified that this target identified on individual MRI scans corresponds to a surface landmark localized ~2 cm lower and 4 cm lateral to the inion. Continuous or intermittent theta-burst stimulations67 were delivered over the target with a 70 mm figure-of-eight coil held with the handle pointing upwards (so that the induced current had caudal-to-rostral orientation) and connected to a SuperRapid2 magnetic stimulator. For the sham stimulation (delivered with a cTBS pattern), the coil was moved vertically 5 cm below the cerebellar target25. For both active and sham stimulations, 600 pulses were delivered at 80% AMTrapidstim.
Paired associative stimulation
We used a low intensity, high frequency PAS14. The electrical pulses were delivered over the right median nerve at the wrist at 2.5x the sensory threshold or just below the motor threshold, whichever was lower. Each electrical pulse was followed 25 ms later by a magnetic pulse delivered over the APB’s hotspot in the left M1 at 90% AMTrapidstim. Six hundred pairs of stimuli were delivered at 5 Hz through a 70 mm figure-of-eight coil connected to a SuperRapid2 magnetic stimulator.
Cortico-spinal excitability assessment
Effects of PAS intervention on the cortico-spinal excitability were assessed with single-pulse TMS delivered at 130% RMTmagstim200 at 0.2 Hz, through a 70-mm figure of-eight coil connected to a Magstim200 stimulator. The direction of the induced current was posterior-to-anterior. Fifteen MEPs were averaged prior to the intervention (T0), and at 10 min (T10), 20 min (T20), and 30 min (T30) after the end of the PAS.
Vibration of the sternocleidomastoid muscle
A DC motor with an eccentric on the shaft (Brüel & Kjær, Nærum, Denmark) produced 7 N peak-to-peak vibrations at 80 Hz, with the amplitude < 6 mm. The vibrator was applied on the belly of the left sterno-cleido-mastoid muscle, 1 cm above its distal insertion on the clavicle. The vibrator was kept at the same spot throughout the experiment. After the session, subjects were asked specifically about head tilt or rotation illusions: nobody reported explicit sensations of head turning or tilting in response to the vibration. This is similar to the previous reports in which dorsal neck vibration rarely produced illusions of head movement56.
Experimental design
All participants to this study gave their written informed consent prior to the study. The experimental procedures were in accord with the Declaration of Helsinki and approved by the following Ethics Committees: IEC at SCTIMST (India), CPP Ile-de-France VI (France), and.
Experiment 1
Twenty-three healthy subjects and 22 CD patients were recruited from 3 centers, all of them attended three sessions. In all three sessions, 5Hz-PAS was used to induce plasticity in the dominant (left) M1. PAS was preceded by right cerebellar stimulation consisting of the following three, randomized interventions: (i) cerebellar cortex excitation [cerebellar-intermittent theta burst stimulation (iTBSCB → PAS)], (ii) cerebellar cortex inhibition [cerebellar-continuous theta burst stimulation (cTBSCB → PAS)], or (iii) sham stimulation of the cerebellum (ShamCB → PAS). The three sessions were separated by intervals of at least 1 week. Five Hz PAS was applied 5 min after the end of cerebellar conditioning (Fig. 1A). Dystonia severity was assessed from videos recorded at the beginning of the first session. The video protocol was designed to score the Toronto Western Spasmodic Torticollis Rating Scale (TWSTRS) and was used by all three centers. All videos from the three centers were rated offline by the same movement disorders specialist (H.C.).
Experiment 2
Experiment 1 revealed difference between the group of CD patients and the control group. Experiment 2 was designed to answer the question whether such differences are related to neck muscles activation in the CD group while neck muscles were at rest in the healthy subjects. Two groups of 12 healthy subjects each were compared (Group 1: mean age: 39.9 ± 10.2 years, range 30–66, 8 women; Group 2: mean age 39.7 ± 11.3 years, range 24–64, 8 women). Subjects of each group underwent the same three interventions as in experiment 1, the only difference being the different head-trunk alignments. Subjects of Group 1 looked forward throughout the stimulation and the recording periods in each session. Subjects of Group 2 maintained a voluntary head turn towards the right shoulder (angle 40–50°) throughout the stimulation and the recording periods in each session.
Control experiment
This experiment was designed to assess, the role of neck afferents versus change in gaze direction or vestibular stimulation during voluntary head turn on the cerebellar priming. An additional group of 6 healthy volunteers was enrolled. These subjects were tested with cTBSCB → PAS, while they received a vibration of the left sterno-cleido-mastoid muscle and maintained the head in the midline. We chose to use cTBSCB → PAS as the effect of the head turn was more significant on cTBSCB → PAS-induced effects than on iTBSCB → PAS-induced effects (see Results). We then compared the effects of cTBSCB → PAS in this group (n = 6, mean age 35.2 ± 10.9 years, range 24–56) with those obtained in 2 subgroups of 6 subjects matched one by one for age and sex and extracted from group 1 of experiment 2 i.e tested with the head on the midline (n = 6, mean age = 34.2 ± 8.5, range 25–50) and from group 2 of experiment 2 i.e while maintaining a head turn (n = 6, mean age = 35.3 ± 8.9, range 24–50).
The datasets generated during the current study are available from the corresponding author on reasonable request.
References
Shakkottai, V. G. et al. Current Opinions and Areas of Consensus on the Role of the Cerebellum in Dystonia. Cerebellum 16, 577–594, https://doi.org/10.1007/s12311-016-0825-6 (2017).
Quartarone, A. et al. Abnormal associative plasticity of the human motor cortex in writer’s cramp. Brain: a journal of neurology 126, 2586–2596, https://doi.org/10.1093/brain/awg273 (2003).
Weise, D. et al. The two sides of associative plasticity in writer’s cramp. Brain: a journal of neurology 129, 2709–2721, https://doi.org/10.1093/brain/awl221 (2006).
Quartarone, A. et al. Abnormal plasticity of sensorimotor circuits extends beyond the affected body part in focal dystonia. J Neurol Neurosurg Psychiatry 79, 985–990, https://doi.org/10.1136/jnnp.2007.121632 (2008).
Weise, D., Schramm, A., Beck, M., Reiners, K. & Classen, J. Loss of topographic specificity of LTD-like plasticity is a trait marker in focal dystonia. Neurobiology of disease 42, 171–176, https://doi.org/10.1016/j.nbd.2010.11.009 (2011).
Bradley, D. et al. Temporal discrimination thresholds in adult-onset primary torsion dystonia: an analysis by task type and by dystonia phenotype. Journal of neurology 259, 77–82, https://doi.org/10.1007/s00415-011-6125-7 (2012).
Kimmich, O. et al. Temporal discrimination, a cervical dystonia endophenotype: penetrance and functional correlates. Movement disorders: official journal of the Movement Disorder Society 29, 804–811, https://doi.org/10.1002/mds.25822 (2014).
Scontrini, A. et al. Somatosensory temporal discrimination tested in patients receiving botulinum toxin injection for cervical dystonia. Movement disorders: official journal of the Movement Disorder Society 26, 742–746, https://doi.org/10.1002/mds.23447 (2011).
Antelmi, E. et al. Neurophysiological correlates of abnormal somatosensory temporal discrimination in dystonia. Movement disorders: official journal of the Movement Disorder Society 32, 141–148, https://doi.org/10.1002/mds.26804 (2017).
Tinazzi, M., Fiorio, M., Bertolasi, L. & Aglioti, S. M. Timing of tactile and visuo-tactile events is impaired in patients with cervical dystonia. Journal of neurology 251, 85–90, https://doi.org/10.1007/s00415-004-0282-x (2004).
Sadnicka, A. et al. Pallidal stimulation for cervical dystonia does not correct abnormal temporal discrimination. Movement disorders: official journal of the Movement Disorder Society 28, 1874–1877, https://doi.org/10.1002/mds.25581 (2013).
Brighina, F. et al. Effects of cerebellar TMS on motor cortex of patients with focal dystonia: a preliminary report. Experimental brain research 192, 651–656, https://doi.org/10.1007/s00221-008-1572-9 (2009).
Hubsch, C. et al. Defective cerebellar control of cortical plasticity in writer’s cramp. Brain: a journal of neurology 136, 2050–2062, https://doi.org/10.1093/brain/awt147 (2013).
Quartarone, A. et al. Rapid-rate paired associative stimulation of the median nerve and motor cortex can produce long-lasting changes in motor cortical excitability in humans. The Journal of physiology 575, 657–670, https://doi.org/10.1113/jphysiol.2006.114025 (2006).
Stefan, K., Kunesch, E., Cohen, L. G., Benecke, R. & Classen, J. Induction of plasticity in the human motor cortex by paired associative stimulation. Brain: a journal of neurology 123(Pt 3), 572–584 (2000).
Popa, T. et al. Cerebellar processing of sensory inputs primes motor cortex plasticity. Cerebral cortex 23, 305–314, https://doi.org/10.1093/cercor/bhs016 (2013).
Kishore, A. et al. Cerebellar sensory processing alterations impact motor cortical plasticity in Parkinson’s disease: clues from dyskinetic patients. Cerebral cortex 24, 2055–2067, https://doi.org/10.1093/cercor/bht058 (2014).
Fratello, F. et al. Modulation of corticospinal excitability by paired associative stimulation: reproducibility of effects and intraindividual reliability. Clinical neurophysiology: official journal of the International Federation of Clinical Neurophysiology 117, 2667–2674, https://doi.org/10.1016/j.clinph.2006.07.315 (2006).
Kishore, A. et al. Motor cortex plasticity can indicate vulnerability to motor fluctuation and high L-DOPA need in drug-naive Parkinson’s disease. Parkinsonism Relat Disord 35, 55–62, https://doi.org/10.1016/j.parkreldis.2016.12.005 (2017).
Muller-Dahlhaus, J. F., Orekhov, Y., Liu, Y. & Ziemann, U. Interindividual variability and age-dependency of motor cortical plasticity induced by paired associative stimulation. Experimental brain research 187, 467–475, https://doi.org/10.1007/s00221-008-1319-7 (2008).
Kishore, A. et al. Age-related decline in the responsiveness of motor cortex to plastic forces reverses with levodopa or cerebellar stimulation. Neurobiology of aging 35, 2541–2551, https://doi.org/10.1016/j.neurobiolaging.2014.05.004 (2014).
Koch, G. et al. Effects of two weeks of cerebellar theta burst stimulation in cervical dystonia patients. Brain stimulation 7, 564–572, https://doi.org/10.1016/j.brs.2014.05.002 (2014).
Manzoni, D., Pompeiano, O. & Andre, P. Convergence of directional vestibular and neck signals on cerebellar purkinje cells. Pflugers Arch 435, 617–630, https://doi.org/10.1007/s004240050562 (1998).
Shaikh, A. G., Meng, H. & Angelaki, D. E. Multiple reference frames for motion in the primate cerebellum. The Journal of neuroscience: the official journal of the Society for Neuroscience 24, 4491–4497, https://doi.org/10.1523/JNEUROSCI.0109-04.2004 (2004).
Popa, T., Russo, M. & Meunier, S. Long-lasting inhibition of cerebellar output. Brain stimulation 3, 161–169, https://doi.org/10.1016/j.brs.2009.10.001 (2010).
Mancic, B. et al. Transcranial theta-burst stimulation alters GLT-1 and vGluT1 expression in rat cerebellar cortex. Neurochem Int 100, 120–127, https://doi.org/10.1016/j.neuint.2016.09.009 (2016).
Lee, S. A., Oh, B. M., Kim, S. J. & Paik, N. J. The molecular evidence of neural plasticity induced by cerebellar repetitive transcranial magnetic stimulation in the rat brain: a preliminary report. Neuroscience letters 575, 47–52, https://doi.org/10.1016/j.neulet.2014.05.029 (2014).
Coesmans, M., Weber, J. T., De Zeeuw, C. I. & Hansel, C. Bidirectional parallel fiber plasticity in the cerebellum under climbing fiber control. Neuron 44, 691–700, https://doi.org/10.1016/j.neuron.2004.10.031 (2004).
Boyden, E. S., Katoh, A. & Raymond, J. L. Cerebellum-dependent learning: the role of multiple plasticity mechanisms. Annual review of neuroscience 27, 581–609, https://doi.org/10.1146/annurev.neuro.27.070203.144238 (2004).
Gao, Z., van Beugen, B. J. & De Zeeuw, C. I. Distributed synergistic plasticity and cerebellar learning. Nature reviews. Neuroscience 13, 619–635, https://doi.org/10.1038/nrn3312 (2012).
Chabrol, F. P., Arenz, A., Wiechert, M. T., Margrie, T. W. & DiGregorio, D. A. Synaptic diversity enables temporal coding of coincident multisensory inputs in single neurons. Nature neuroscience 18, 718–727, https://doi.org/10.1038/nn.3974 (2015).
Ishikawa, T., Shimuta, M. & Hausser, M. Multimodal sensory integration in single cerebellar granule cells in vivo. Elife 4, https://doi.org/10.7554/eLife.12916 (2015).
Bologna, M. et al. Effects of cerebellar theta-burst stimulation on arm and neck movement kinematics in patients with focal dystonia. Clinical neurophysiology: official journal of the International Federation of Clinical Neurophysiology 127, 3472–3479, https://doi.org/10.1016/j.clinph.2016.09.008 (2016).
Klier, E. M., Wang, H., Constantin, A. G. & Crawford, J. D. Midbrain control of three-dimensional head orientation. Science 295, 1314–1316, https://doi.org/10.1126/science.1067300 (2002).
Farshadmanesh, F., Klier, E. M., Chang, P., Wang, H. & Crawford, J. D. Three-dimensional eye-head coordination after injection of muscimol into the interstitial nucleus of Cajal (INC). Journal of neurophysiology 97, 2322–2338, https://doi.org/10.1152/jn.00752.2006 (2007).
Shaikh, A. G., Wong, A. L., Zee, D. S. & Jinnah, H. A. Keeping your head on target. The Journal of neuroscience: the official journal of the Society for Neuroscience 33, 11281–11295, https://doi.org/10.1523/JNEUROSCI.3415-12.2013 (2013).
Shaikh, A. G., Zee, D. S., Crawford, J. D. & Jinnah, H. A. Cervical dystonia: a neural integrator disorder. Brain 139, 2590–2599, https://doi.org/10.1093/brain/aww141 (2016).
Sadnicka, A. et al. Normal motor adaptation in cervical dystonia: a fundamental cerebellar computation is intact. Cerebellum 13, 558–567, https://doi.org/10.1007/s12311-014-0569-0 (2014).
Pelosin, E., Bove, M., Marinelli, L., Abbruzzese, G. & Ghilardi, M. F. Cervical dystonia affects aimed movements of nondystonic segments. Movement disorders: official journal of the Movement Disorder Society 24, 1955–1961, https://doi.org/10.1002/mds.22693 (2009).
Anastasopoulos, D., Maurer, C. & Mergner, T. Interactions between voluntary head control and neck proprioceptive reflexes in cervical dystonia. Parkinsonism & related disorders 20, 1165–1170, https://doi.org/10.1016/j.parkreldis.2014.08.008 (2014).
Lee, J. R. & Kiss, Z. H. Interhemispheric difference of pallidal local field potential activity in cervical dystonia. Journal of neurology, neurosurgery, and psychiatry 85, 306–310, https://doi.org/10.1136/jnnp-2013-305476 (2014).
Moll, C. K. et al. Asymmetric pallidal neuronal activity in patients with cervical dystonia. Frontiers in systems neuroscience 8, 15, https://doi.org/10.3389/fnsys.2014.00015 (2014).
Bostan, A. C., Dum, R. P. & Strick, P. L. The basal ganglia communicate with the cerebellum. Proceedings of the National Academy of Sciences of the United States of America 107, 8452–8456, https://doi.org/10.1073/pnas.1000496107 (2010).
Hoshi, E., Tremblay, L., Feger, J., Carras, P. L. & Strick, P. L. The cerebellum communicates with the basal ganglia. Nature neuroscience 8, 1491–1493, https://doi.org/10.1038/nn1544 (2005).
Kishore, A. & Popa, T. Cerebellum in levodopa-induced dyskinesias: the unusual suspect in the motor network. Frontiers in neurology 5, 157, https://doi.org/10.3389/fneur.2014.00157 (2014).
Kiss, Z. H. et al. The Canadian multicentre study of deep brain stimulation for cervical dystonia. Brain: a journal of neurology 130, 2879–2886, https://doi.org/10.1093/brain/awm229 (2007).
Armstrong, B., McNair, P. & Taylor, D. Head and neck position sense. Sports Med 38, 101–117 (2008).
Burns, J. K. & Blohm, G. Multi-sensory weights depend on contextual noise in reference frame transformations. Frontiers in human neuroscience 4, 221, https://doi.org/10.3389/fnhum.2010.00221 (2010).
Biguer, B., Donaldson, I. M., Hein, A. & Jeannerod, M. Neck muscle vibration modifies the representation of visual motion and direction in man. Brain: a journal of neurology 111(Pt 6), 1405–1424 (1988).
Bove, M., Diverio, M., Pozzo, T. & Schieppati, M. Neck muscle vibration disrupts steering of locomotion. J Appl Physiol (1985) 91, 581–588 (2001).
Ceyte, H., Cian, C., Nougier, V., Olivier, I. & Roux, A. Effects of neck muscles vibration on the perception of the head and trunk midline position. Experimental brain research 170, 136–140, https://doi.org/10.1007/s00221-006-0389-7 (2006).
de Vries, J. et al. Joint position sense error in people with neck pain: A systematic review. Manual therapy 20, 736–744, https://doi.org/10.1016/j.math.2015.04.015 (2015).
Burke, D., Hagbarth, K. E., Lofstedt, L. & Wallin, B. G. The responses of human muscle spindle endings to vibration of non-contracting muscles. The Journal of physiology 261, 673–693 (1976).
Roll, J. P. & Vedel, J. P. Kinaesthetic role of muscle afferents in man, studied by tendon vibration and microneurography. Experimental brain research 47, 177–190 (1982).
Goodwin, G. M., McCloskey, D. I. & Matthews, P. B. The contribution of muscle afferents to kinaesthesia shown by vibration induced illusions of movement and by the effects of paralysing joint afferents. Brain: a journal of neurology 95, 705–748 (1972).
Taylor, J. L. & McCloskey, D. I. Illusions of head and visual target displacement induced by vibration of neck muscles. Brain: a journal of neurology 114(Pt 2), 755–759 (1991).
Manzoni, D., Andre, P. & Pompeiano, O. Proprioceptive neck influences modify the information about tilt direction coded by the cerebellar anterior vermis. Acta Otolaryngol 124, 475–480 (2004).
Pompeiano, O., Andre, P. & Manzoni, D. Spatiotemporal response properties of cerebellar Purkinje cells to animal displacement: a population analysis. Neuroscience 81, 609–626 (1997).
Hoffland, B. S. et al. Cerebellum-dependent associative learning deficits in primary dystonia are normalized by rTMS and practice. The European journal of neuroscience 38, 2166–2171, https://doi.org/10.1111/ejn.12186 (2013).
Pirio Richardson, S., Tinaz, S. & Chen, R. Repetitive transcranial magnetic stimulation in cervical dystonia: effect of site and repetition in a randomized pilot trial. PloS one 10, e0124937, https://doi.org/10.1371/journal.pone.0124937 (2015).
Zittel, S., Helmich, R. C., Demiralay, C., Munchau, A. & Baumer, T. Normalization of sensorimotor integration by repetitive transcranial magnetic stimulation in cervical dystonia. Journal of neurology 262, 1883–1889, https://doi.org/10.1007/s00415-015-7789-1 (2015).
Bradnam, L. V., McDonnell, M. N. & Ridding, M. C. Cerebellar Intermittent Theta-Burst Stimulation and Motor Control Training in Individuals with Cervical Dystonia. Brain Sci 6, https://doi.org/10.3390/brainsci6040056 (2016).
Prudente, C. N., Hess, E. J. & Jinnah, H. A. Dystonia as a network disorder: what is the role of the cerebellum? Neuroscience 260, 23–35, https://doi.org/10.1016/j.neuroscience.2013.11.062 (2014).
Rossini, P. M. et al. Non-invasive electrical and magnetic stimulation of the brain, spinal cord and roots: basic principles and procedures for routine clinical application. Report of an IFCN committee. Electroencephalography and clinical neurophysiology 91, 79–92 (1994).
Stoodley, C. J. & Schmahmann, J. D. Functional topography in the human cerebellum: a meta-analysis of neuroimaging studies. NeuroImage 44, 489–501, https://doi.org/10.1016/j.neuroimage.2008.08.039 (2009).
Hardwick, R. M., Lesage, E. & Miall, R. C. Cerebellar transcranial magnetic stimulation: the role of coil geometry and tissue depth. Brain stimulation 7, 643–649, https://doi.org/10.1016/j.brs.2014.04.009 (2014).
Huang, Y. Z., Edwards, M. J., Rounis, E., Bhatia, K. P. & Rothwell, J. C. Theta burst stimulation of the human motor cortex. Neuron 45, 201–206, https://doi.org/10.1016/j.neuron.2004.12.033 (2005).
Acknowledgements
We thank the Center for Clinical Investigations (CIC) Pitié-Salpêtrière N° 9503 and the platform “CENIR–PANAM: physiology and analysis of movement core facility” of CR-ICM for their invaluable support with the experiments. This work was supported by the Dystonia Coalition, part of the National Institute of Health (NIH) Rare Diseases Clinical Research Network. Funding and/or programmatic support for this project has been provided through grant NS065701 from the National Institute of Health (NIH) Office of Rare Diseases Research and the National Institute of Neurological Disorders and Stroke. The views expressed in written materials or publications do not necessarily reflect the official policies of the Department of Health and Human Services; nor does mention by trade names, commercial practices, or organizations imply endorsement by the U.S. Government. This research was conducted within the framework of an “Institut National de la Santé et de la Recherche Médicale (INSERM)–Indian Council of Medical Research (ICMR)” collaborative project. INSERM supported the research through grant #C10–01 and ICMR through “Indo-INSERM/Neurol/21/2010-NCD-I”. T.P. was supported by Fondation Motrice and the “Investissements d’avenir” program ANR-10-IAIHU-06. C.H. was the recipient of a scholarship from Fondation Groupama pour la Santé. S.M. was the beneficiary of an INSERM/APHP Contrat d’interface. A.R. was the recipient of a scholarship from AMADYS. The research leading to these results has received funding from the program “Investissements d’avenir” ANR-10-IAIHU-06 and project AMEDYST ANR-16-CE37-0003.
Author information
Authors and Affiliations
Contributions
Study concept and design: A.K., S.M., T.P., E.R.; Data collection: C.H., P.J., S.K., R.M., S.P., T.P., A.R.; Analysis and interpretation of the data: A.K., C.H., P.J., S.M., T.P.; Writing: A.K., S.M., T.P.; Revising: C.H., P.J., S.K., S.P., A.R., E.R., M.R. All authors approved the final version of the manuscript and agree to be accountable for all aspects of the work.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Popa, T., Hubsch, C., James, P. et al. Abnormal cerebellar processing of the neck proprioceptive information drives dysfunctions in cervical dystonia. Sci Rep 8, 2263 (2018). https://doi.org/10.1038/s41598-018-20510-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-20510-1
This article is cited by
-
A gait-based paradigm to investigate central body representation in cervical dystonia patients
Neurological Sciences (2023)
-
Consensus Paper: Novel Directions and Next Steps of Non-invasive Brain Stimulation of the Cerebellum in Health and Disease
The Cerebellum (2021)
-
Contemporary clinical neurophysiology applications in dystonia
Journal of Neural Transmission (2021)
-
Altered sensory system activity and connectivity patterns in adductor spasmodic dysphonia
Scientific Reports (2020)
-
Abnormal microscale neuronal connectivity triggered by a proprioceptive stimulus in dystonia
Scientific Reports (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.