Abstract
We investigated age-related changes in in vivo and in vitro functions and gene expression of the bladder of male and female mice. Mature and aged (12 and 27–30 month old) C57BL/6 mice of both sexes were used. Frequency volume, conscious free-moving cystometry and detrusor contractile and relaxant properties in in vitro organ bath were evaluated. mRNA expression level of muscarinic, purinergic, and β-adrenergic receptors and gene expression changes by cDNA microarray analysis of the bladder were determined. Cystometry demonstrated storage and voiding dysfunctions with ageing in both sexes. Detrusor strips from aged mice showed weaker contractile responses particularly in the cholinergic component and weaker relaxant responses to isoproterenol. These age-related impairments were generally severer in males. mRNA expression of bladder tissue was decreased for M3 muscarinic receptors in aged males and β2-adrenoceptors in aged females. cDNA microarray analysis results, albeit substantial sex difference, indicated “cell-to-cell signaling and interaction” as the most common feature of age-related gene expression. In summary, aged mice demonstrated voiding and storage dysfunctions resembling to detrusor hyperactivity with impaired contractility (DHIC), which were more pronounced in males. Genomic changes associated with aging may contribute to the age-related bladder functional deterioration in mice.
Similar content being viewed by others
Introduction
The rapid aging of population led by declining birth rate and mortality is a major global demographic trend1. Accordingly, the prevalence of aging-related diseases has been increasing2. In the urological field, lower urinary tract symptom (LUTS) and associated urodynamic changes, detrusor hyperactivity with impaired contractility (DHIC) in particular, is a common feature observed among the elderly3,4,5. However, these age-related changes have significant inter-individual variability and sex-related disparity3,6. The age-related changes or differences should be derived from genetic or epigenetic pathophysiology, which has not been fully investigated3,7,8,9. Previous human studies revealed decreased mRNA expression of M3 muscarinic receptor and impaired cholinergic neurotransmission with compensatory purinergic transmission in the detrusor smooth muscle of aged subjects10,11.
Several animal studies using rodents so far have explored the age-related changes in the bladder function12,13,14,15,16; in vitro intrinsic contractile property and contractile responses to cholinergic stimulation of detrusor smooth muscle were impaired13,16; cystometry in aged rats showed increased residual volume despite maintained maximum pressure12,14. However, most animal studies used only males or females, with a limited or different set of experiments (for example with or without anesthesia, etc.). Consequently, age-related changes in the bladder function showed inconsistent results even among the studies using the same type of animals, and are unknown for sex-based difference.
In the present study, we investigated age-related changes and differences of bladder functions in vivo and in vitro and gene expression in the male and female mouse bladder.
Results
Body weight, bladder weight, blood glucose, and sex hormones
Body weight in aged mice (AGED: 27–30-month old) was significantly lower compared to mature mice (MAT: 12-month old) in males alone. Bladder weight was counted for all mice excepting the mice used for cystometry (CMG) measurements (MAT: n = 13 and AGED: n = 9 in males, MAT: n = 13 and AGED: n = 14 in females). Only in males, both the actual and adjusted (by body weight) bladder weights of AGED mice, were significantly higher than those of MAT mice. Blood glucose level was not different between the two groups in either sex (Table 1A). Serum sex hormone data were obtained from 5 MAT and 4 AGED males and 5 MAT and 7 AGED females. Testosterone level was undetectable (less than 0.03 ng/ml) in 3 out of 4 AGED male mice and markedly reduced in the remaining one mouse (0.05 ng/ml) compared with MAT male mice (1.63 ± 0.92 (0.11–3.96) ng/ml). In contrast, estradiol level in female mice was not significantly different between MAT and AGED mice (Table 1).
Frequency/volume (FV) measurements
Compared with MAT mice, AGED mice showed significantly increased total voided volume in both sexes, higher voiding frequency and increased water intake in males, and lower mean flow rate in females. There were no significant differences in voided volume per micturition and food intake between the two groups in either sex (7–10 mice per group) (Table 1).
Conscious free-moving CMG measurements
We had allocated 7 males and 6 females of each group for CMG but evaluated 6 MAT and 5 AGED mice of both sexes. Two male AGED mice were dead before measurements. The CMG recordings of 1 male MAT mouse and 1 female AGED mouse were excluded, because reproducible tracings were not obtained. Compared with MAT mice, AGED mice showed multiple significant changes; (1) increased frequency of non-voiding contractions (NVCs) per minute in both sexes, (2) shorter intercontraction interval, smaller bladder capacity, and smaller voided volume in males, (3) lower mean flow rate in both sexes, and (4) lower maximum pressure in males (Table 2 and Fig. 1).
In vitro functional analysis of detrusor strips
Contractile responses
In both sexes, the contractile responses to high K+ was weaker in AGED mice compared with MAT mice (assayed in 14 to 19 detrusor strips and 9 to 11 mice per group) (Fig. 2A). The Emax of the contractile responses to carbachol (CCh) in AGED mice was also significantly lower in both sexes (5 to 7 detrusor strips and 5 to 7 mice per group) (Fig. 2B). In contrast, the pEC50 values for the responses to CCh (male: 6.07 vs. 5.62, female: 5.97 vs. 5.76) and the responses to adenosine triphosphate (ATP) showed no significant age-related differences in either sex (4 or 5 detrusor strips and 4 or 5 mice per group) (Fig. 2B and C). The amplitude of contractions induced by electric field stimulation (EFS) was significantly lower in AGED mice in both sexes, except at 2 Hz in females (5 to 7 detrusor strips and 5 to 7 mice per group). However, after atropine administration, there were no significant differences between the two groups in either sex (Fig. 2D).
Contractile and relaxant responses of detrusor strips. Contractile responses to high K+ (A), CCh (B), ATP (C), and EFS (D) and relaxant responses to isoproterenol (E). MAT: mature (12-month old), AGED: aged (27–30-month old). Values are expressed as mean ± SEM. *p < 0.05, **p < 0.01, ***p < 0.001, significantly different from MAT of the same sex (A,C,D) unpaired t-test, (B,E) f-test of nonlinear regression).
Relaxant responses
Compared with MAT mice, the maximum relaxant response to isoproterenol (ISO) was significantly weaker in AGED mice in both sexes, although its pEC50 value (male: 7.86 vs. 7.64, female: 7.94 vs. 7.61) was not significantly different between groups in either sex (Fig. 2E).
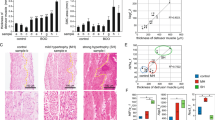
Histological examinations
In males, collagen deposition in the detrusor layer tended to increase in AGED mice, but there were no significant differences in either sex (7 or 8 bladder specimens per group, male: p = 0.077, Fig. 3).
Representative histological images of bladder specimens taken from each group of mice, with Masson’s trichrome staining. MAT: mature (12-month old), AGED: aged (27–30-month old) Scale bar = 200 µm (A). Collagen deposition rate in the detrusor layer (B). No significant differences between MAT and AGED were found in either sex (unpaired t-test).
Real-time reverse transcription polymerase chain reaction (RT-PCR)
AGED bladder showed significantly lower M3 and P2X1 receptor expression in males and β2-adrenoceptor expression in females (MAT: n = 11 and AGED: n = 4 in males, MAT: n = 5 and AGED: n = 7 in females, Fig. 4). No significant differences were found in receptor expression of M1, M2, β1, or β3 in either sex (Fig. 4).
cDNA microarray analyses
Total RNA from 4 bladders in each group were investigated. In AGED mice, 469 genes in males, 392 in females, and 8 in both sexes were significantly upregulated, whereas 544 genes in males, 75 in females, and 37 in both sexes were significantly downregulated. Of the top 5 upregulated genes (Tables 3), 4 genes were not identified as ontology terms, with Kcnj5 as an exception. In contrast, the top 5 downregulated genes were all associated with “response to stimuli” and “extracellular region” as per gene ontology analysis. When gene functions were classified into three categories (diseases and disorders, molecular and cellular functions, and physiological system development and function), the top 5 functions that significantly changed with aging in each category are summarized in Table 4. The genes that commonly changed with aging in both sexes were associated with “cell-to-cell signaling and interaction.”
Discussion
The in vivo study results indicated that aged mice had storage and voiding dysfunctions, which would resemble to DHIC, and such dysfunctions were more pronounced in males. More specifically CMG showed an increased number of NVCs and lower mean flow rate in aged mice of both sexes, with related parameters more significantly impaired in males. These results were compatible with bladder dysfunctions commonly observed among the elderly3,8,9. Less obvious age-related changes in FV parameters would be explained by polyuria in aged mice, which complicated the evaluation of the bladder function by FV.
In vitro organ bath studies using isolated detrusor strips demonstrated decreased contractile responses to high K+ with aging in both sexes, suggesting impaired intrinsic contractile property of detrusor smooth muscle. The finding is consistent with previous reports using male mice and female guinea pigs13,17. Our contractile response studies also revealed lower atropine sensitive component in EFS-induced contractions and lower responses to muscarinic stimulation with aging in both sexes. In contrast, there is no significant change in purinergic component in EFS-induced contractions and contractile responses to purinergic stimulation. These results are in line with the previous findings demonstrating age-related impairment of contractile responses to both carbachol and EFS (muscarinic but not purinergic component) in the male rat and human bladders10,14,16. We have confirmed this age-related impairment in females as well. Thus, the age-related changes of bladder contractility would be attributable to impairment of cholinergic contractile properties of detrusor smooth muscle with aging in addition to impaired intrinsic contractile property. Our experiments also showed that the relaxant responses to ISO were attenuated in aged mice of both sexes, which was consistent with findings in the rat bladder18,19. We demonstrated, for the first time to our knowledge, using real-time RT-PCR that the expression of the M3 receptors in males and the β2-adrenoceptors in females was downregulated in aged mice. The results would support the functional changes demonstrated in the in vitro organ bath studies. Our results also suggest sex differences in age-related mRNA expression changes of muscarinic, purinergic, and β-adrenergic receptors of the bladder. In contrast, downregulated P2X1 receptor expression in aged male mice, which is consistent with observations in male human detrusor without bladder outlet obstruction10,20, was not reflected in in vitro contractile experiments. We did not fully address this discrepancy, which may be due to differences between genomic and functional contributions in the isolated bladder strips. Taken together, in vivo and in vitro studies, gene expression studies as well, indicate that both contractile and relaxant responses of detrusor smooth muscle were impaired with aging in both sexes. These observations in both in vitro and in vivo experiments suggest that our aged mice model would reproduce the aged human bladder condition.
The microarray analysis of the bladder revealed “cell-to-cell signaling and interaction” as the most common age-related changes in both sexes. Such genomic changes may be responsible for functional and morphological changes in the bladder such as collagen deposition, particularly in males. This should be addressed in more detail using our current findings as a basis for future studies. For example, determining the genes responsible for age-related bladder dysfunction would bring useful information for understanding and clinical management of age-related bladder dysfunction. Regarding sex disparity, male mice showed more severe aging-related functional deterioration than females, as per the CMG measurements, in vitro organ bath studies, and histological evaluations. A possible reason would be alteration in the sex hormone status with aging21. Serum testosterone levels were remarkably lower in AGED male mice compared with those in MAT male mice, whereas there was no significant difference in serum estradiol levels between AGED and MAT female mice. Keast et al. suggested that androgens are essential for the maturation and maintenance of the structure of pelvic autonomic neurons and pelvic ganglion neurons that supply the bladder of the male Wistar rat22.
Our study has some limitations. First, we investigated age-related changes only at two points of life. Further studies using other age groups can describe chronological changes of the bladder function more precisely, disclosing the time-dependent maturation and deterioration of the bladder. Second, we have not examined the changes of the properties of neuronal tissue like as dorsal root ganglia. Considering the relevance of neuronal control on bladder function, gene expression changes of the neuronal tissue may disclose more precisely age-related bladder dysfunction. Third, the mechanisms of age-related deterioration of bladder contractile or relaxant properties are to be disclosed. Further studies including evaluating the age-related differences of voltage gated calcium channels or potential intracellular pathways are needed. Forth, the implication of age-related genomic changes remained to be speculative. Studies using genetically engineered mice would contribute to elucidating the role of genes responsible for the functional changes observed. Finally, we have not examined pharmacological responses to therapeutic agents. The age-related changes in responsiveness, if any, should be examined further for improving the management of lower urinary tract dysfunctions according to sex or age.
In conclusion, aged mice demonstrated voiding and storage dysfunctions resembling to DHIC, which were more pronounced in males. Genomic changes associated with aging may contribute to the age-related functional deterioration in mice.
Methods
Animals and experimental groups
C57BL/6 male and female mice were divided into two groups: MAT (male: N = 20, female: N = 19) and AGED (male: N = 16, female: N = 20). Mice were maintained under standard laboratory conditions, with a 12:12 h light (9:00 a.m. to 9:00 p.m.) and dark (9:00 p.m. to 9:00 a.m.) cycle and free access to food pellets and tap water. Experimental protocols were approved by the Animal Ethics Committee of the University of Tokyo Graduate School of Medicine, Tokyo, Japan and were in line with NIH guidelines for the care and use of experimental animals. Mice were obtained from the Tokyo Metropolitan Institute of Gerontology, and after a 1-week adaptation period, they were used for measurements.
FV measurements
Each mouse was separately placed, conscious and without restraint, in a MCM/TOA-UF001–006 metabolic cage (Mitsubishi Chemical Medience, Tokyo, Japan). This metabolic cage can pass urine and feces separately owing to a specially designed net, facilitating precise measurement of the voided urine volume16,23. After a 24-h adaptation, the voided volume, voiding frequency, duration of each micturition, and water intake volume were recorded with a PowerLab data acquisition system (AD Instruments, Sydney, Australia) continuously for 24 h starting from 9:00 am, with a 12:12 h light (9:00 a.m. to 9:00 p.m.) and dark (9:00 p.m. to 9:00 a.m.) cycle. Mean flow rate of each voiding was calculated by dividing voided volume by duration of micturition. The mice had free access to water and food during recordings.
Conscious free-moving CMG measurements
In separate mice from other experiments, under isoflurane anesthesia, a polyethylene catheter (Clay-Adams PE–10, Parsippany, NJ) was implanted into the bladder through the dome and subcutaneously tunneled and exteriorized at the back of the neck. Five days after the surgery, conscious CMG measurements were performed without any restraint in the metabolic cage (MCM/TOA-UF001-006). The intravesical catheter was connected via a three-way stopcock to a pressure transducer (DX-100, Nihon Kohden, Tokyo, Japan) and a syringe pump (KDS 200, Muromachi Kikai Co. Ltd., Tokyo, Japan) for saline instillation at 15 µl/min. Intravesical pressure and voided volume were measured using PowerLab (AD Instruments). After stable and reproducible recordings were obtained, micturition cycles during 1 h were averaged and the following cystometric parameters were analyzed: basal pressure (minimum bladder pressure), threshold pressure (bladder pressure at the onset of micturition), maximum pressure (maximum bladder pressure during micturition), voided volume, intercontraction interval (duration between two micturitions), bladder capacity (instilled saline volume), residual volume (bladder capacity − voided volume), mean flow rate (voided volume divided by duration of micturition), voiding efficiency (voided volume divided by bladder capacity × 100%), bladder compliance [bladder capacity divided by (threshold pressure − basal pressure)], and number and amplitude of NVCs24. NVCs were defined as bladder contractions without micturition, with amplitudes of more than 5 cmH2O25. Mice using CMG measurements were euthanatized after measurements, and the bladder tissues of them were not used for other in vitro experiments, because catheter implantation may change the property of the bladder tissue.
Blood glucose and sex hormone measurements
At approximately 1:00 pm, mice were restrained and blood glucose levels were measured in the tail vein using a disposable glucose test sensor (Glutest, Sanwa Kagaku Kenkyusho Co. Ltd, Tokyo, Japan). Mice were then anesthetized with an intraperitoneal injection of pentobarbital sodium (30 mg/kg). The peritoneal cavity was opened after central abdominal incision, and whole blood (approximately 0.4 ml) was harvested from the inferior vena cava. The blood sample was transferred to a test tube and left at room temperature for 30 min and at 4 °C for 16 h; subsequently, the sample was centrifuged at 1200 g for 25 min to separate the serum. The collected serum was stored frozen (−20 °C) until measurements. Serum testosterone levels for male by an electrochemiluminescence assay and estradiol levels for female mice by a chemiluminescence assay were analyzed by SRL, Inc. (Tokyo, Japan).
In vitro studies for functional analysis using detrusor strips
This investigation was performed as described in our previous reports16,26. After mice were sacrificed by an overdose of pentobarbital sodium, the bladder bodies were harvested, and longitudinal full-thickness bladder strips were transferred to 5 ml organ baths. After a 2-h equilibration period with a stable tension of 10 mN for contractile and 5 mN for relaxant experiments, investigations were initiated. The strips were first exposed to a high K+ (62 mM KCl) Krebs solution. After 3 times washout, contractile responses to particular stimuli were examined in separate specimens, including 1: Cch (10−8 M to 10−3 M), 2: ATP (10−6 M to 10−2 M), and 3: EFS (pulse width: 0.8 ms, 50 V; pulse duration: 5 s; and stimulation interval: 1 m at 2, 5, 10, and 20 Hz). Cumulative application of Cch and ATP were given after a previous peak contraction without washout. After baseline measurements, EFS was repeated after 10−6 M atropine administration, purinoceptor desensitization by repeated 10−5 M α, β-Methylene-ATP (mATP) administrations, and finally, 10−6 M tetrodotoxin (TTX) administration. Each drug application interval was 2 minutes. In separate specimens, relaxant responses to ISO (10−10 M to 10−4 M) were evaluated under a mechanical 5 mN tension with 5 minutes interval. Papaverine (10−3 M) was applied 5 minutes after the maximal dose of ISO as a referent drug to induce relaxation. The contractile responses to CCh and EFS and the relaxant response to ISO were evaluated using detrusor strips in the same mice and the contractile responses to ATP were evaluated using the strips in separate mice. The amplitude was measured in all in vitro contractile and relaxant experiments.
Histological examination
Isolated bladder body specimens were fixed in 4% paraformaldehyde-phosphate buffered saline, embedded in paraffin, and cut into 3-μm sections. Masson’s trichrome staining was performed to analyze fibrosis in the detrusor muscle layer. Collagen deposition was determined in 3 randomly selected sections27. Collagen deposition rate was defined as the collagen content (blue areas) divided by the muscle area × 100% in each section16. Images were analyzed using Adobe software and Image J (http://rsb.info.nih.gov/ij/)16,28.
Real-time RT-PCR
The bladder body dissected out were immediately placed in RNAlater RNA Stabilization Reagent (QIAGEN, Venlo, The Netherlands). Total RNA extracted from tissues using the miRNeasy Mini Kit (QIAGEN) was reverse transcribed to cDNA using the ReverTra Ace qPCR RT Master Mix (TOYOBO, Osaka, Japan) according to the manufacturer’s procedure. For relative quantification of mRNA expression, real-time PCR was performed using the THUNDERBIRD SYBR qPCR Mix (TOYOBO) and gene-specific primers on the StepOnePlus Real-Time PCR System (Thermo Fisher Scientific, Waltham, MA, USA). The data was normalized to Gapdh. Primer sequences were as shown in Supplementary table.
cDNA microarray analyses
Total RNA from the bladder (200 ng) extracted for real-time RT-PCR was amplified and labeled with Cy3 using a Low Input Quick Amp Labeling Kit (Agilent Technologies, Santa Clara, CA, USA) according to the manufacturer’s instructions. The labeled cRNA was hybridized to the Agilent SurePrint G3 Mouse GE 8 × 60 K Microarray according to the manufacturer’s procedure. After hybridization, the microarrays were scanned on a DNA Microarray Scanner with Scan Control software. Raw data were collected from processed images using Feature Extraction software (Agilent). Gene expression analysis was performed with the Subio platform (Subio, Kagoshima, Japan). These data were compared using t-tests between MAT and AGED groups, with p < 0.05 considered statistically significant. The remaining probes were selected using the criterion of at least a 2-fold change. The gene list was analyzed using Ingenuity pathway analysis software (QIAGEN). The microarray data discussed in this publication have been deposited in NCBI’s Gene Expression Omnibus (GEO) and are accessible through the GEO Series accession number GSE100219 (https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE100219).
Drugs
CCh, ATP, atropine, and TTX were purchased from Wako Chemical, Tokyo, Japan. ISO and mATP were purchased from Sigma-Aldrich (St. Louis, MO, USA). Papaverine was purchased from Cayman Chemical, Ann Arbor, MI, USA. Krebs solution comprised 118 mM NaCl, 4.7 mM KCl, 2.5 mM CaCl2, 25.0 mM NaHCO3, 1.2 mM KH2PO4, and 11 mM glucose (pH 7.4).
Statistical Analysis
All data are expressed as the mean ± SEM. pEC50 and Emax of the CCh- and ISO- responses were analyzed by f-test of nonlinear regression, and other results were analyzed using unpaired t-test between MAT and AGED groups of the same sex. Statistical significance was considered at p values < 0.05.
References
Ezeh, A. C., Bongaarts, J. & Mberu, B. Global population trends and policy options. Lancet 380, 142–148 (2012).
Ory, M. G., Wyman, J. F. & Yu, L. Psychosocial factors in urinary incontinence. Clinics in geriatric medicine 2, 657–671 (1986).
Irwin, D. E. et al. Population-based survey of urinary incontinence, overactive bladder, and other lower urinary tract symptoms in five countries: results of the EPIC study. European urology 50, 1306–1314; discussion 1314–1305, (2006).
Resnick, N. M. & Yalla, S. V. Detrusor hyperactivity with impaired contractile function. An unrecognized but common cause of incontinence in elderly patients. JAMA 257, 3076–3081 (1987).
Dubeau, C. E. The aging lower urinary tract. The Journal of urology 175, S11–15 (2006).
Ameda, K., Sullivan, M. P., Bae, R. J. & Yalla, S. V. Urodynamic characterization of nonobstructive voiding dysfunction in symptomatic elderly men. The Journal of urology 162, 142–146 (1999).
Abarbanel, J. & Marcus, E. L. Impaired detrusor contractility in community-dwelling elderly presenting with lower urinary tract symptoms. Urology 69, 436–440 (2007).
Al-Ghazo, M. A. et al. Urodynamic detrusor overactivity in patients with overactive bladder symptoms. International neurourology journal 15, 48–54 (2011).
Madersbacher, S. et al. The aging lower urinary tract: a comparative urodynamic study of men and women. Urology 51, 206–212 (1998).
Yoshida, M. et al. Age-related changes in cholinergic and purinergic neurotransmission in human isolated bladder smooth muscles. Experimental gerontology 36, 99–109 (2001).
Mansfield, K. J. et al. Muscarinic receptor subtypes in human bladder detrusor and mucosa, studied by radioligand binding and quantitative competitive RT-PCR: changes in ageing. British journal of pharmacology 144, 1089–1099 (2005).
Ito, H. et al. Preventive Effects of Long-Term Caloric Restriction on Aging Related In Vivo Bladder Dysfunction and Molecular Biological Changes in the Bladder and Dorsal Root Ganglia in Rats. The Journal of urology 196, 1575–1583 (2016).
Gomez-Pinilla, P. J., Pozo, M. J. & Camello, P. J. Aging differentially modifies agonist-evoked mouse detrusor contraction and calcium signals. Age (Dordr) 33, 81–88 (2011).
Zhao, W. et al. Impaired bladder function in aging male rats. The Journal of urology 184, 378–385 (2010).
Lluel, P. et al. Functional and morphological modifications of the urinary bladder in aging female rats. American journal of physiology. Regulatory, integrative and comparative physiology 278, R964–972 (2000).
Ito, H. et al. Long-term caloric restriction in rats may prevent age related impairment of in vitro bladder function. The Journal of urology 193, 2123–2130 (2015).
Gomez-Pinilla, P. J. et al. Melatonin restores impaired contractility in aged guinea pig urinary bladder. Journal of pineal research 44, 416–425 (2008).
Nishimoto, T., Latifpour, J., Wheeler, M. A., Yoshida, M. & Weiss, R. M. Age-dependent alterations in beta-adrenergic responsiveness of rat detrusor smooth muscle. The Journal of urology 153, 1701–1705 (1995).
Frazier, E. P., Schneider, T. & Michel, M. C. Effects of gender, age and hypertension on beta-adrenergic receptor function in rat urinary bladder. Naunyn-Schmiedeberg’s archives of pharmacology 373, 300–309 (2006).
Chua, W. C.-N. et al. Age-related changes of P2X1 receptor mRNA in the bladder detrusor from men with and without bladder outlet obstruction. Experimental gerontology 42, 686–692 (2007).
Pradidarcheep, W. Lower urinary tract symptoms and its potential relation with late-onset hypogonadism. The aging male: the official journal of the International Society for the Study of the Aging Male 11, 51–55 (2008).
Keast, J. R. & Saunders, R. J. Testosterone has potent, selective effects on the morphology of pelvic autonomic neurons which control the bladder, lower bowel and internal reproductive organs of the male rat. Neuroscience 85, 543–556 (1998).
Aizawa, N., Homma, Y. & Igawa, Y. Influence of High Fat Diet Feeding for 20 Weeks on Lower Urinary Tract Function in Mice. Lower urinary tract symptoms 5, 101–108 (2013).
Andersson, K. E., Soler, R. & Fullhase, C. Rodent models for urodynamic investigation. Neurourology and urodynamics 30, 636–646 (2011).
Cornelissen, L. L., Misajet, B., Brooks, D. P. & Hicks, A. Influence of genetic background and gender on bladder function in the mouse. Autonomic Neuroscience 140, 53–58 (2008).
Igawa, Y. et al. Changes in the Function and Expression of T-Type and N-Type Calcium Channels in the Rat Bladder after Bladder Outlet Obstruction. The Journal of urology (2013).
Sawada, N. et al. Protective effect of a beta3-adrenoceptor agonist on bladder function in a rat model of chronic bladder ischemia. European urology 64, 664–671 (2013).
Taylor, J. A. et al. Null mutation in macrophage migration inhibitory factor prevents muscle cell loss and fibrosis in partial bladder outlet obstruction. Am J Physiol Renal Physiol 291, F1343–1353 (2006).
Acknowledgements
The present study has been supported by a Grant-in-Aid for Scientific Research (YI; Grant no. 40159588, NA; Grant no. 80595257) from the Ministry of Education, Culture, Sport, Science and Technology of the Japanese Government and a Grant from Japanese Society of Geriatric Urology 2013 (HI).
Author information
Authors and Affiliations
Contributions
J.K. and H.I. designed and performed the majority of the experiments, analysed data and wrote this manuscript. Y.F. performed real time R.T.-P.C.R. T.K. performed cDNA microarray analysis. N.A. and Y.I. supervised the study and edited this manuscript. H.H., M.I., and Y.H. discussed the data.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kamei, J., Ito, H., Aizawa, N. et al. Age-related changes in function and gene expression of the male and female mouse bladder. Sci Rep 8, 2089 (2018). https://doi.org/10.1038/s41598-018-20406-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-20406-0
This article is cited by
-
Change of Detrusor Contractility in Patients with and without Bladder Outlet Obstruction at Ten or More Years of follow-up
Scientific Reports (2019)
-
Molecular Characteristics of Underactive Bladder
Current Bladder Dysfunction Reports (2019)
-
Attenuated lipopolysaccharide-induced inflammatory bladder hypersensitivity in mice deficient of transient receptor potential ankilin1
Scientific Reports (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.