Abstract
Guanylate Binding Proteins (GBPs) are a group of cytokine-inducible large guanosine triphosphatase. Previous studies have shown high expression of GBP1 in circulating monocytes of premenopausal subjects was correlated to extremely low peak bone mass, which is considered as an important determinant of osteoporosis. However, whether GBPs play a role in regulation of osteogenesis of mesenchymal stromal cells (MSCs) remains largely unknown. In the present study, we found that mRNA expression of GBP1 was highest among all the GBPs, and it was dramatically downregulated during osteogenic differentiation of human MSCs derived from bone marrow (hBM-MSCs). While siRNA-mediated knockdown of GBP1 promoted osteogenesis, overexpression of GBP1 suppressed osteogenesis of hBM-MSCs. Furthermore, we found GBP1 is required for expression of indoleamine 2,3 dioxygenase (IDO), Interleukin 6 (IL-6) and IL-8 induced by treatment with Interferon-γ (IFN-γ). Depletion of GBP1 rescued the inhibited osteogenesis induced by IFN-γ treatment, at least in part. Collectively, our findings indicate GBP1 inhibits osteogenic differentiation of MSCs, and inhibition of GBP1 expression may prevent development of osteoporosis and facilitate MSC-based bone regeneration.
Similar content being viewed by others
Introduction
Osteoporosis is a skeletal disorder affecting 9 million people worldwide, which is characterized by compromised bone strength with an increase in risk of fracture1,2. The skeleton is a dynamic organ where bone mass is controlled by a delicate balance between bone resorption by osteoclasts and bone formation by osteoblasts3. Mesenchymal stromal cells (MSCs) are a heterogeneous population of multipotent cells that can give rise to osteoblasts, chondrocytes and adipocytes4,5,6. As the progenitors of osteoblasts, mesenchymal stromal cells (MSCs), the osteoblastic differentiation of MSCs are critical for the maintenance of bone mass and stem cell-based bone repair. Several factors, such as parathyroid hormone (PTH) and bone morphogenetic proteins (BMPs), have been used for osteoporosis treatment and bone regeneration, respectively7,8. However, the molecular mechanisms governing osteoblastic differentiation of MSCs is still not fully understood. In addition, a lot of effort has been made to identify risk genes for susceptibility to osteoporosis that may facilitate prevention of osteoporosis9,10. For instance, human guanylate binding protein 1 (hGBP1) was found to be up-regulated in circulating monocytes of premenopausal subjects with extremely low peak bone mass (PBM), which is considered as an important determinant of osteoporosis11,12.
The GBPs are a subfamily of cytokine-induced dynamin superfamily of large guanosine triphosphatase (GTPases)13,14,15. GBPs are capable of binding to agarose-immobilized guanosine triphosphate (GTP), guanosine monophosphate (GMP), and guanosine diphosphate (GMP), and hydrolyzing GTP to both GDP and GMP. To date, 7 hGBPs (hGBP-1 through -7) have been designated within a cluster on chromosome 1in human genome, while 11 mGBPs have been designated in 2 clusters on chromosome 3 and 5 in murine genome, respectively16. hGBP-1 through -5 and all the mGBPs can be induced by interferon-gamma(IFN-γ). Thus, GBPs have been extensively used as markers of interferon responsiveness. Furthermore, accumulating evidence has revealed the critical function of the GBPs in various processes17. For instance, hGBP1 has been shown to mediate the antibacterial and antiviral activities of IFN-γ. mGbp2, the putative murine homologue of hGBP1, is required for lysis of pathogen-containing vacuoles in bone marrow-derived macrophages (BMMs). mGBP2−/− BMMs exhibited reduced levels of cell death, cytokine secretion and caspase release18. In addition, Ostler et al. reported that hGBP1 co-localized with actin and regulated remodeling of the fibrous actin structure in IFN-γ-treated Hela cells19.
In regard to MSCs, previous study has shown that hGBP1 can be induced by IFN-γ, and play an important role in the immunity of hMSCs against Toxoplasma gondii20. IFN-γ was also found to inhibit osteogenic differentiation of MSCs21. Furthermore, a recent study has revealed that mGbp2 was downregulated during osteogenic differentiation of mouse MC3T3 cells, an osteoblast precursor cell line22. However, the role of GBPs in osteogenic differentiation of MSCs remains largely unknown. In the present study, we evaluated the expression levels of hGBPs during osteogenic differentiation of MSCs derived from human bone marrow (hBM-MSCs), and investigated the changes in osteogenic differentiation potential of BMSCs in response to knockdown and overexpression of hGBP1, respectively. Finally, we found hGBP1 was induced by IFN-γ treatment, and was required for the upregulation of the target genes induced by IFN-γ in hBM-MSCs.
Methods and Materials
Cell Culture
Primary hBM-MSCs were purchased from Rooster Bio (Frederick, MD, USA). The cells were maintained in Dulbecco’s Modified Eagle Medium (DMEM), supplemented with 15% heat-inactivated FBS and 100 U/ml of K-Penicillin G and100 mg/ml of Streptomycin sulfate at 37°C in a humidified atmosphere of 5% CO2 (all from Invitrogen, Carlsbad, CA, USA). The cells used in this study were within passages 4–10. 10 ng/ml recombinant human IFN-γ (R&D systems, Minneapolis, MN, USA) was used to treat hBM-MSCs as indicated. To induce osteogenic differentiation of hBM-MSCs, osteogenic stimulus (OS) containing 100 μM ascorbic acid, 10 mM β-glycerophosphate, and 10 nM dexamethasone (all from Sigma-Aldrich, St. Louis, MO, USA) was added into the medium.
Transfection
All siRNAs were purchased from Invitrogen, including a scrambled-siRNA (siScr) as a control, and siRNA targeting hGBP1 (siGBP1). Transfection was performed using Lipofectamine RNAiMAX reagent (Invitrogen) according to the manufacturer’s instructions. For overexpression of GBP1, retroviruses expressing human GBP1 gene were purchased from Fulengen Inc. (Guangzhou, China). hBM-MSCs were infected with in the presence of polybrene (Sigma) for 24 hours. hBM-MSCs transfected with empty vector were used as control.72 hours post-transfection, 500 µg/ml G418 was added into the medium for 3 days, and the selected cells were expanded in growth medium.
Characterization of Osteoblastic Phenotypes
Alkaline phosphatase (ALP) staining and Alizarin Red S (ARS) staining were performed after several days of osteogenic induction as previously described23,24. For the quantitative measurement of ALP activity, 10 μL cell protein extraction was incubated with 50 μL ALP stabilizing buffer (Sigma) and 50 μL ALP yellow (pNPP) liquid substrate (Sigma) for 20 min at 37 °C. The absorbance was then measured at 405 nm.
RNA Extraction and Quantitative Reverse Transcription-PCR (RT-qPCR)
Total RNA was isolated using the TRIzol reagent (Invitrogen) according to manufacturer’s instructions. Then, complementary DNA was synthesized with 1 ug aliquots of total RNA using PrimeScript RT Reagent Kit (TAKARA, Otsu, Shiga, Japan). QPCR was performed using SYBR Premix Ex Taq kit (TAKARA).The primer sequences used for this analysis were as following: 5′-GGAGCGAGATCCCTCCAAAAT-3′ (forward) and 5′ -GGCTGTTGTCATACTTCTCATGG-3′ (reverse) for GAPDH; 5′-GAAGTGCTAGAAGCCAGTGC-3′ (forward) and 5′-CCACCACCATAGGCTGTGTA-3′ (reverse) for GBP1; 5′-CTATCTGCAATTACGCAGCCT -3′ (forward) and 5′ -TGTTCTGGCTTCTTGGGATGA-3′ (reverse) for GBP2; 5′ - ATTCCCTGAAGCTAACGCAAG -3′ (forward) and 5′-GGGCAGATCGAAGACAAAACATT-3′ (reverse) for GBP3; 5′-ATGGGTGAGAGAACTCTTCACG-3′ (forward) and 5′- TGCGGTATAGCCCTACAATGG -3′ (reverse) for GBP4; 5′-CCATGTGCCTCATCGAGAACT-3′ (forward) and 5′-ACAGGTTGCGTAATGGCAGAC-3′ (reverse) for GBP5; 5′-AACCATCTGGCAGGACAGAAT-3′ (forward) and 5′-TCACCCTTTTCCACATCGCC -3′ (reverse) for GBP6; 5′-GTGGAGCGACTCCTTGTCTG-3′ (forward) and 5′-GTGGGGAATCTCACTTGCTGG -3′ (reverse) for GBP7; 5′ - GAGGGCCAAGACGAAGACATC-3′ (forward) and 5′ - CAGATCACGTCATCGCACAAC -3′ (reverse) for COL1A1; 5′ - CACTGGAGCCAATGCAGAAGA-3′ (forward) and 5′-TGGTGGGGTTGTAGGTTCAAA-3′ (reverse) for IBSP; 5′-GCCGCTGTAACCTCTTCGG -3′ (forward) and 5′-GTCTTCGGCCAATCTGGCTTT -3′ (reverse) for SPP1; 5′-ACTCACCTCTTCAGAACGAATTG -3′ (forward) and 5′-CCATCTTTGGAAGGTTCAGGTTG -3′ (reverse) for IL6; 5′- CTTTCAGAGACAGCAGAGCAC-3′ (forward) and 5′-ACTGTGAGGTAAGATGGTGGC-3′ (reverse) for IL8; 5′-GCCAGCTTCGAGAAAGAGTTG-3′ (forward) and 5′-ATCCCAGAACTAGACGTGCA -3′ (reverse) for IDO.
Western Blot
Western blot was performed as previous described25. Briefly, the cells were lysed for 30 min in RIPA buffer (Santa Cruz Biotechnology, Paso Robles, CA, USA), and centrifuged at 18,000 g for 15 min at 4 °C. 30 μg aliquots of the lysates were separated on a 10% sodiumdodecyl sulfate-polyacrylamidegel. The resolved proteins were then transferred onto nitrocellulose membrane (Bio-Rad, Hercules, California, USA). The membrane was subsequently incubated with primary antibodies followed by a horseradish peroxidase (HRP)-conjugated secondary antibody (Boster, Wuhan, China). Protein bands were detected using an enhanced chemiluminescence western blotting detection kit (Thermo, Canoga Park, CA, USA). Antibodies for western blot were purchased from the following suppliers: rabbit polyclonal anti-human GBP1 antibody (Invitrogen), rabbit polyclonal anti-human IDO1 antibody (Invitrogen).
Statistical Analysis
Data was shown as mean ± SD from three independent experiments. Statistical analysis was performed using Graph Pad Prism 6 software. Student’s t-test and one-way analysis of variance (ANOVA) were performed for single comparisons and multiple comparisons, respectively. The statistical significance of differences among IFN-γ treatments were assessed using two-way ANOVAs with Holm-Sidak post-hoc test. *p < 0.05; **p < 0.01; ***p < 0.001.
Results
GBP1 is downregulated during osteogenic differentiation of hBM-MSCs
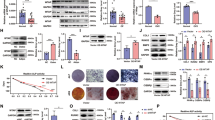
To investigate the role of GBPs in regulation of osteogenic differentiation of MSCs, we first screened the expression profiles of hGBPs in hBM-MSCs by RT-qPCR. As shown in Fig. 1A, GBP1 was of highest expression level in all the 7 GBPs, and the expression of GBP -4 to -7 was also detected. However, the expression of GBP6 and GBP7 was not detected by RT-qPCR (Fig. 1A). We next examined the expression of GBP1 to GBP5 during the osteogenic differentiation of hBM-MSCs. Notably, expression of GBP1 was dramatically downregulated upon osteogenic stimulus (OS) in hBM-MSCs (Fig. 1B). While GBP2 expression was upregulated after 7 days of osteogenic differentiation, expression of GBP3, GBP4, and GBP5 seemed to be unchanged during osteogenic differentiation of hBM-MSCs (Fig. 1C to F).
Expression profiling of GBPs in hBM-MSCs and during osteogenic differentiation of hBM-MSCs. (A) The mRNA expression levels of GBP-1through -7 in hBM-MSCs. (B–F) The mRNA expression levels of GBP1 (B), GBP2 (C), GBP3 (D), GBP4 (E), GBP5 (F) in response to osteogenic stimulus (OS) in hBM-MSCs at 0, 1, 4, 7 days. Asterisks indicate a significant difference compared to the baseline. *p < 0.05; **p < 0.01; ***p < 0.001.
Depletion of GBP1 enhanced osteogenic differentiation of hBM-MSCs
To investigate whether GBP1 inhibits osteogenic differentiation of MSCs, we used specific siRNA to knockdown the expression of GBP1 in hBM-MSCs, and the knockdown efficiency was assessed by RT-PCR and western blot (Fig. 2A). Interestingly, we found that depletion of GBP1 significantly enhanced ALP activity in hBM-MSCs after 7 days of osteogenic induction (Fig. 2B,C). We further assessed the extracellular matrix (ECM) mineralization by ARS staining after 3 weeks of osteogenic induction. As shown in Fig. 2D and E, the ECM mineralization was also enhanced by depletion of GBP1. Furthermore, the expression of osteogenesis markers, such as COL1A1 (Collagen, type I, alpha 1), IBSP (Integrin binding sialoprotein), and SPP1 (secreted phosphoprotein 1), was elevated by depletion of GBP1 (Fig. 2F).
siRNA-mediated depletion of Cdo1 enhances osteogenic differentiation of hBM-MSCs. (A) The knockdown efficiency of siRNA targeting GBP1 compared to scrambled siRNA (siScr) was confirmed by RT-PCR. (B) Knockdown of GBP1 enhanced the ALP staining after 7 days of osteogenic induction in hBM-MSCs. (C) Knockdown of GBP1 enhanced the ALP activity after 7 days of osteogenic induction as determined by quantitative ALP activity assay. (D) Knockdown of GBP1 enhanced mineralization of extracellularmatrix (ECM) after 3 weeks of osteogenic induction. (E) Quantification of Alizarin Red S (ARS) staining in D. (F) Knockdown of GBP1 promoted expression levels of COL1A1 (Collagen, type I, alpha 1), IBSP (Integrin binding sialoprotein), and SPP1 (secreted phosphoprotein 1), as determined by RT-PCR. *p < 0.05; **p < 0.01; ***p < 0.001.
Overexpression of GBP1 inhibited osteogenic differentiation of hBM-MSCs
Next, hBM-MSCs were stably transduced with retroviruses expressing hGBP1, and the overexpression efficiency was assessed by RT-PCR and western blot (Fig. 3A,B). After osteogenic induction, we found overexpression of GBP1 inhibited ALP activity, as determined by ALP staining and quantitative ALP activity assay (Fig. 3C,D). The ECM mineralization was also significantly reduced by overexpression of GBP1 in hBM-MSCs (Fig. 3E,F). Consistently, the expression of COL1A1, IBSP, and SPP1 are also suppressed in hBM-MSCs after 7 days of osteogenic induction (Fig. 3G). Taken together, these findings demonstrated that GBP1 suppressed osteogenic differentiation of hBM-MSCs.
Ectopic overexpression of GBP1 suppresses osteogenic differentiation of hBM-MSCs. (A,B) Overexpression of GBP1 in hBM-MSCs was confirmed by RT-PCR (A) and western blot (B). Original images of western blots were presented in Supplementary Figure S1. (C) Overexpression of GBP1 inhibited the ALP staining after 7 days of osteogenic induction. (D) Overexpression of GBP1 impaired the ALP activity after 7 days of osteogenic induction. (E) Overexpression of GBP1 reduced mineralization post-3 weeks of osteogenic induction. (F) Quantification of ARS staining in (E). (G) Overexpression of Cdo1 inhibited mRNA expression levels of COL1A1, IBSP, and SPP1. *p < 0.05; **p < 0.01; ***p < 0.001.
GBP1 is required for IFN-γ-induced processing of IDO, IL-6 and IL-8
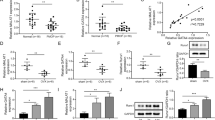
Previous study has reported that IFN-γ treatment induced expression of GBPs, and suppressed osteogenic differentiation of human MSCs from bone marrow via activation of indoleamine 2,3 dioxygenase (IDO)20,21. Consistently, we found GBP1 was upregulated in response to treatment with IFN-γ at 10 ng/ml (Fig. 4A,B). As revealed by RT-PCR and western blot assay, we found knockdown of GBP1 significantly inhibited expression of IDO in the absence or presence of IFN-γ (Fig. 5C,D). Furthermore, knockdown of GBP1 also suppressed expression of Interleukin 6 (IL-6) and IL-8 induced by IFN-γ treatment (Fig. 5E,F), indicating GBP1 may play an important role in IFN-γ signaling, and GBP1 may inhibit osteogenesis of MSCs by regulating IFN-γ signaling.
GBP1 is required for IFN-γ-induced processing of IDO, IL-6 and IL-8. (A,B) Expression of GBP1 was dramatically upregulated by treatment with IFN-γ at 10 ng/ml, as determined by RT-PCR (A) and western blot (B). Original images of western blots were presented in Supplementary Figure S2A. (C) Knockdown of GBP1 impaired the mRNA expression of indoleamine 2,3 dioxygenase (IDO) induced by treatment with IFN-γ (#p < 0.05 by two-way ANOVA with Holm-Sidak post-hoc test). (D) Knockdown of GBP1 impaired the protein expression of indoleamine 2,3 dioxygenase (IDO) induced by treatment with IFN-γ. Original images of western blots were presented in Supplementary Figure S2B. (E, F) Knockdown of GBP1 impaired the mRNA expression of Interleukin 6 (IL-6) (E) and IL-8 (F) induced by IFN-γ treatment (##p < 0.01 by two-way ANOVA with Holm-Sidak post-hoc test). *p < 0.05; **p < 0.01; ***p < 0.001.
Depletion of GBP1 partially rescued the inhibited osteogenesis by IFN-γ treatment in hBM-MSCs. (A,B) Knockdown of GBP1 partially restored the inhibited ALP activity by IFN-γ treatment, as determined by ALP staining (A) and quantitative ALP activity assay (B). (C) Knockdown of GBP1 partially rescued the reduced ECM mineralization by IFN-γ treatment. (D) Quantification of ARS staining in (D). *p < 0.05; **p < 0.01; ***p < 0.001.
Depletion of GBP1 partially rescued the inhibited osteogenesis by IFN-γ treatment in hBM-MSCs
To investigate whether the induction of GBP1 is required for the inhibitory effect of IFN-γ on osteogenesis, hBM-MSCs were transduced with siRNA targeting GBP1 for 24 h before treatment with IFN-γ. Interestingly, we found knockdown of GBP1 rescued the inhibited ALP activity of hBM-MSCs by IFN-γ treatment, at least in part (Fig. 4A,B). In addition, knockdown of GBP1 also partially restored the reduced mineralization of hBM-MSCs after 3 weeks of osteogenic induction (Fig. 4C,D). Collectively, our findings suggest IFN-γ-inducible GBP1 inhibits osteogenic differentiation of human MSCs.
Discussion
Understanding of molecular mechanisms that regulate osteogenic differentiation is critical for treatment of osteoporosis and MSC-based bone repair. In this study, we found that GBP1 showed highest expression in all the GBPs in hBM-MSCs, and it was downregulated during osteogenic differentiation. While siRNA-mediated depletion of GBP1 promoted osteogenic differentiation, ectopic overexpression of GBP1 reduced osteogenic differentiation of hBM-MSCs. Furthermore, GBP1 is required for the expression of IDO, IL-6, and IL-8 induced by IFN-γ treatment, and knockdown of GBP1 rescued the inhibited osteogenic potential by IFN-γ treatment, at least in part.
To our knowledge, this is the first time that GBP1 has been found to inhibit osteogenesis of human MSCs. GBPs were initially identified as IFN-γ-responsive genes that mediate the antibacterial and antiviral activities of IFN-γ. In mice, mGBPs accumulate around pathogen-containing vacuoles (PV) and are important in restricting intracellular pathogens and immune activation, such as Toxoplasma26. However, recent study has also reported that hGBP1 restricted Toxoplasma gondii, but did not localize to pathogen vacuoles, revealing different mechanisms between hGBPs and mGBPs27. Furthermore, compared to other hGBPs, the crystal structure of human GBP-1 has revealed its different biochemical properties16,28. In this study, we also different expression patterns of hGBPs in hBM-MSCs exposed to osteogenic stimulus. For instance, GBP1 was dramatically downregulation, but GBP2 was upregulated during osteogenic differentiation of hBM-MSCs. More efforts are still needed to investigate whether the other GBPs play a role in regulation of lineage specification of MSCs.
Our results also revealed that GBP1 is required for IFN-γ signaling in hBM-MSCs. Previous studies have highlighted the role of GBPs in inflammasome activation and innate immune response29,30. And deletion of Gbp1, Gbp2, Gbp3, and Gbp5 in macrophages often resulted in impaired immune response, respectively, indicating an important role of GBPs in inflammatory signaling29,31,32. In addition, several studies have shown that GBP1 can also mediate IFN responses that are not directly associated with host defense against pathogens, such as inhibition of matrix metalloproteinase expression33, inhibition of proliferation in endothelial cells34. Consistently, we found knockdown of GBP1 significantly suppressed expression of IDO, IL-6 and IL-8 induced by treatment with IFN-γ in this study. However, knockdown of GBP1 did not fully rescue the inhibited osteogenesis of hBM-MSCs by IFN-γ treatment. It may be because of the redundancy among the GBPs, since all GBP-1 through -5 can be induced by IFN-γ treatment in hMSCs20. Finally, one of the limitations of this study is that how GBP1 regulate IFN-γ signaling in hBM-MSCs remain largely unknown. Future studies may focus on the mechanisms by which GBPs mediate IFN-γ signaling.
In conclusion, we have found GBP1 was downregulated during osteogenic differentiation of hBM-MSCs. While knockdown of GBP1 promoted osteogenesis, overexpression of GBP1 suppressed osteogenesis of hBM-MSCs. Furthermore, we found GBP1 is required for IFN-γ signaling, and depletion of GBP1 rescued the inhibited osteogenesis induced by IFN-γ treatment, at least in part. Together with previous study, high expression of GBP1 in monocytes and MSCs may be associated with the risk to osteoporosis, and inhibition of GBP1 expression may prevent development of osteoporosis and facilitate MSC-based bone regeneration.
References
Prevention, N.I.H.C.D.P.o.O., Diagnosis & and, T. Osteoporosis prevention, diagnosis, and therapy. JAMA 285, 785–795 (2001).
Liu, W. et al. GDF11 decreases bone mass by stimulating osteoclastogenesis and inhibiting osteoblast differentiation. Nat Commun 7, 12794 (2016).
Cowin, S. C. & Hegedus, D. H. Bone remodeling I: theory of adaptive elasticity. Journal of Elasticity 6, 313–326 (1976).
Bianco, P., Robey, P. G. & Simmons, P. J. Mesenchymal stem cells: revisiting history, concepts, and assays. Cell stem cell 2, 313–319 (2008).
Deng, P., Chen, Q.-M., Hong, C. & Wang, C.-Y. Histone methyltransferases and demethylases: regulators in balancing osteogenic and adipogenic differentiation of mesenchymal stem cells. International Journal of Oral Science 7, 197–204 (2015).
Deng, P. et al. Cysteine dioxygenase type 1 promotes adipogenesis via interaction with peroxisome proliferator-activated receptor gamma. Biochem Biophys Res Commun 458, 123–127 (2015).
Kulkarni, N. H. et al. Effects of parathyroid hormone on Wnt signaling pathway in bone. Journal of Cellular Biochemistry 95, 1178–1190 (2005).
Geiger, M., Li, R. H. & Friess, W. Collagen sponges for bone regeneration with rhBMP-2. Advanced Drug Delivery Reviews 55, 1613–1629 (2003).
Ralston, S. H. & de Crombrugghe, B. Genetic regulation of bone mass and susceptibility to osteoporosis. Genes & Development 20, 2492–2506 (2006).
Richards, J. B., Zheng, H.-F. & Spector, T. D. Genetics of osteoporosis from genome-wide association studies: advances and challenges. Nat Rev Genet 13, 576–588 (2012).
Lei, S. F. et al. An in vivo genome wide gene expression study of circulating monocytes suggested GBP1, STAT1 and CXCL10 as novel risk genes for the differentiation of peak bone mass. Bone 44, 1010–1014 (2009).
Bonjour, J.-P., Chevalley, T., Ferrari, S. & Rizzoli, R. The importance and relevance of peak bone mass in the prevalence of osteoporosis. Salud Pública de México 51, s5–s17 (2009).
Vestal, D. J. & Jeyaratnam, J. A. The guanylate-binding proteins: emerging insights into the biochemical properties and functions of this family of large interferon-induced guanosine triphosphatase. Journal of interferon & cytokine research: the official journal of the International Society for Interferon and Cytokine Research 31, 89–97 (2011).
Maureen, A., Olszewski, J. G. & Dr. Deborah, J. Vestal. In Silico Genomic Analysis of the Human and Murine Guanylate-Binding Protein (GBP) Gene Clusters. Journal of Interferon & Cytokine Research. 26, 15 (2006).
Prakash, B., Praefcke, G. J. K., Renault, L., Wittinghofer, A. & Herrmann, C. Structure of human guanylate-binding protein 1 representing a unique class of GTP-binding proteins. Nature 403, 567–571 (2000).
Kresse, A. et al. Analyses of murine GBP homology clusters based on in silico, in vitro and in vivo studies. BMC genomics 9, 158 (2008).
Kim, B. H. et al. Interferon-induced guanylate-binding proteins in inflammasome activation and host defense. Nature immunology 17, 481–489 (2016).
Meunier, E. et al. Caspase-11 activation requires lysis of pathogen-containing vacuoles by IFN-induced GTPases. Nature 509, 366–370 (2014).
Ostler, N. et al. Gamma interferon-induced guanylate binding protein 1 is a novel actin cytoskeleton remodeling factor. Molecular and cellular biology 34, 196–209 (2014).
Qin, A. et al. Guanylate-binding protein 1 (GBP1) contributes to the immunity of human mesenchymal stromal cells against Toxoplasma gondii. Proceedings of the National Academy of Sciences 114, 1365–1370 (2017).
Croitoru-Lamoury, J. et al. Interferon-gamma regulates the proliferation and differentiation of mesenchymal stem cells via activation of indoleamine 2,3 dioxygenase (IDO). PLoS One 6, e14698 (2011).
Zamurovic, N. Transcriptional program of osteoblast differentiation: coordinated activation of Notch, Wnt, and TGF-[beta] signaling pathways in MC3T3 cell line. (Doctoral dissertation, University_of_Basel). (2005).
Zhang, S. et al. A Novel Nanosilver/Nanosilica Hydrogel for Bone Regeneration in Infected Bone Defects. ACS Applied Materials & Interfaces 8, 13242–13250 (2016).
Yuan, Q. et al. Increased osteopontin contributes to inhibition of bone mineralization in FGF23-deficient mice. Journal of bone and mineral research: the official journal of the American Society for Bone and Mineral Research 29, 693–704 (2014).
Yuan, Q. et al. Fgf23/Klotho signaling is not essential for the phosphaturic and anabolic functions of PTH. Journal of bone and mineral research: the official journal of the American Society for Bone and Mineral Research 26, 2026–2035 (2011).
Degrandi, D. et al. Extensive Characterization of IFN-Induced GTPases mGBP1 to mGBP10 Involved in Host Defense. The Journal of Immunology 179, 7729–7740 (2007).
Johnston, A. C. et al. Human GBP1 does not localize to pathogen vacuoles but restricts Toxoplasma gondii. Cellular Microbiology 18, 1056–1064 (2016).
Prakash, B., Renault, L., Praefcke, G. J. K., Herrmann, C. & Wittinghofer, A. Triphosphate structure of guanylate-binding protein 1 and implications for nucleotide binding and GTPase mechanism. The EMBO Journal 19, 4555–4564 (2000).
Kim, B.-H. et al. IFN-induced Guanylate Binding Proteins in Inflammasome Activation and Host Defense. Nature immunology 17, 481–489 (2016).
Vanaja, S., Rathinam, V. K. & Fitzgerald, K. A. Mechanisms of inflammasome activation: recent advances and novel insights. Trends in cell biology 25, 308–315 (2015).
Shenoy, A. R. et al. GBP5 Promotes NLRP3 Inflammasome Assembly and Immunity in Mammals. Science 336, 481–485 (2012).
Pilla, D. M. et al. Guanylate binding proteins promote caspase-11-dependent pyroptosis in response to cytoplasmic LPS. Proceedings of the National Academy of Sciences 111, 6046–6051 (2014).
Guenzi, E. et al. The guanylate binding protein-1 GTPase controls the invasive and angiogenic capability of endothelial cells through inhibition of MMP-1 expression. The EMBO Journal 22, 3772–3782 (2003).
Guenzi, E. et al. The helical domain of GBP-1 mediates the inhibition of endothelial cell proliferation by inflammatory cytokines. The EMBO Journal 20, 5568–5577 (2001).
Acknowledgements
This project Nis supported by Chongqing Research Program of Basic Research and Frontier Technology (no. cstc2015shmszx10008).
Author information
Authors and Affiliations
Contributions
J.P. and B.S. conceived of this study. B.S., M.Z. and H.Y. performed experiments and analyzed data. J.P., M.Z. and H.Y. wrote and revised the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Bai, S., Mu, Z., Huang, Y. et al. Guanylate Binding Protein 1 Inhibits Osteogenic Differentiation of Human Mesenchymal Stromal Cells Derived from Bone Marrow. Sci Rep 8, 1048 (2018). https://doi.org/10.1038/s41598-018-19401-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-19401-2
This article is cited by
-
The effect of TLR3 priming conditions on MSC immunosuppressive properties
Stem Cell Research & Therapy (2023)
-
Osteoclast fusion and bone loss are restricted by interferon inducible guanylate binding proteins
Nature Communications (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.