Abstract
There is growing recognition that the risk of attention-deficit hyperactivity disorder (ADHD) in children may be influenced by micronutrient deficiencies, including iron. We conducted this meta-analysis to examine the association between ADHD and iron levels/iron deficiency (ID). We searched for the databases of the PubMed, ScienceDirect, Cochrane CENTRAL, and ClinicalTrials.gov up to August 9th, 2017. Primary outcomes were differences in peripheral iron levels in children with ADHD versus healthy controls (HCs) and the severity of ADHD symptoms in children with/without ID (Hedges’ g) and the pooled adjusted odds ratio (OR) of the association between ADHD and ID. Overall, seventeen articles met the inclusion criteria. Peripheral serum ferritin levels were significantly lower in ADHD children (children with ADHD = 1560, HCs = 4691, Hedges’ g = −0.246, p = 0.013), but no significant difference in serum iron or transferrin levels. In addition, the severity of ADHD was significantly higher in the children with ID than those without ID (with ID = 79, without ID = 76, Hedges’ g = 0.888, p = 0.002), and there was a significant association between ADHD and ID (OR = 1.636, p = 0.031). Our results suggest that ADHD is associated with lower serum ferritin levels and ID. Future longitudinal studies are required to confirm these associations and to elucidate potential mechanisms.
Similar content being viewed by others
Introduction
Attention-deficit hyperactivity disorder (ADHD) is a common neurodevelopmental condition with an estimated global prevalence of 7.2%1. ADHD can cause considerable disability and impaired quality of life2. Furthermore, children with ADHD also suffer from a high prevalence of co-occurring medical and psychiatric conditions such as depressive disorder, anxiety disorder, restless leg syndrome, enuresis, and serious stomach problems3.
ADHD is currently thought to be a predominantly heritable condition, with genetic polymorphisms of dopamine receptors and dopamine transporters playing an important role4. While the pathophysiology of ADHD is still not entirely understood, dysregulated dopaminergic neuronal development in the prefrontal cortex and subcortical regions is believed to play a role based on research derived from neuroimaging studies and the effects of pharmacotherapeutic treatment5. Recently, the potential effect of environmental (non-genetic) factors has attracted attention in the etiology of ADHD6. Environmental factors are potentially modifiable and may represent preventable targets for these disorders, in addition to potentially providing novel therapies for certain aspects of ADHD, such as iron supplementation7. Among environmental risk factors, nutritional factors including certain micronutrient deficiencies have been increasingly implicated as possible risk factors for ADHD8.
Iron helps in homeostasis of the hemoglobin structure, antioxidants, genetic repair, and in particular central nervous system (CNS) function9. Iron is stored safely in the body via binding to ferritin, which prevents the degrading effects of free iron. Iron is very important for early brain development and is required for normal myelination and neurotransmitter function. Evidence has shown that impaired iron absorption and subsequent iron deficiency (ID) in both the prenatal and postnatal periods may impair neurodevelopment, with long-lasting and possibly irreversible consequences10. In addition, ID may result in myelinating and/or synaptogenesis dysregulation11. Moreover, ID may be associated with impaired monoamine synthesis, resulting in dysregulated monoamine signal transduction10,12. Adequate iron intake and peripheral iron levels may therefore be important factors modifying the onset of ADHD or ameliorating the severity of ADHD13.
To date, three meta-analyses have evaluated peripheral iron levels, either in forms of iron or ferritin, in children with ADHD14,15,16. While helpful, two of these meta-analyses only considered peripheral ferritin levels. This limitation may have resulted in confounding effects, since peripheral ferritin levels can be affected by various factors including infection, inflammation, and oxidative stress and, therefore, may not represent the whole picture of iron storage in clinical practice17. In the most recent meta-analysis, Wang et al. updated the evidence of the relationship between iron and ADHD, and revealed significantly lower serum ferritin levels in children with ADHD than in controls (standardized mean difference (SMD) = −0.40, 95% CI = −0.66 to −0.14) but not serum iron (SMD = −0.026, 95% CI = −0.29 to 0.24)16. Based on this insignificant result of serum iron in ADHD and controls, the relationship between iron and ADHD still remained unclear. Furthermore, that recent meta-analysis did not provide information about other parameters of iron status such as transferrin level16. Furthermore, no previous study has investigated the relationship between the diagnosis of ID and the risk or severity of ADHD. Although the use of medications for ADHD has been well-established, certain side effects such as poor appetite may still limit their use18. Several studies have investigated the use of iron supplements for patients with ADHD with mixed results, however none of these studies specifically targeted patients with a diagnosis of ID13,19. In addition, two previous meta-analyses found lower ferritin level in patients with ADHD, however it is unclear whether such lower ferritin levels in patients with ADHD reaches a clinical diagnosis of ID14,15. Therefore, investigating the relationship between a diagnosis of ID and the risk and severity of ADHD may help to identify potential treatments such as iron supplementation.
The aim of the current study was to conduct an updated systematic review and meta-analysis considering differences in peripheral iron levels and all parameters of iron status between children with ADHD and healthy (asymptomatic) controls (HCs). In addition, we also aimed to explore the association between a diagnosis of ID and the risk and severity of ADHD.
Results
Study selection
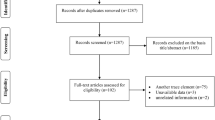
Figure 1 summarizes the details of the search results. In brief, a total of 46 studies entered the full-text review stage. Twenty-nine articles were excluded for various reasons including a lack of controls, non-clinical trials, not comparing iron between ADHD/controls, or review articles (see Supplementary Table 2). A list of excluded articles is presented in Supplementary Table 2. In total, twenty-two articles met the inclusion criteria. We were unable to conduct meta-analysis for four of the recruited studies, because we did not have enough studies (n < 3) for the outcome measurements such as hair iron level, food iron intake level, or plasma/blood iron level in children with ADHD versus controls, or the prevalence of ADHD in children with and without ID20,21,22,23. Finally, we recruited seventeen studies in our meta-analysis9,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39, and the details are summarized in Table 1.
Characteristics and methodological quality of the included studies
Of the included studies, thirteen provided information about serum ferritin levels, six about serum iron levels, two about serum transferrin levels, three about the severity of ADHD symptoms in patients with ID compared to controls, and three provided data of adjusted odds ratios (ORs) with regards the association between ADHD and ID.
We were unable to conduct meta-analysis for the prevalence of ADHD in children with and without ID because we did not have enough studies (n < 3)9. Rather, we chose differences in the severity of ADHD symptoms in children with and without ID to indicate the presence of ADHD.
Study quality appraisal
Regarding the methodological quality of the included studies, the average modified Newcastle-Ottawa Scale (NOS) score40 for case-control trials in subgroup meta-analysis of comparisons of the peripheral iron levels in children with and without ADHD was 6.82 (high quality) with a standard deviation (SD) of 1.1741. The NOS score for cross-sectional trials in subgroup meta-analysis of comparisons of the peripheral iron levels in children with and without ADHD was 4.67 (high quality) (SD = 0.58)41. In addition, the average modified NOS scores for cohort trials and cross-sectional trials in subgroup meta-analysis of comparisons of the severity of ADHD symptoms in children with and without iron deficiency were 5.00 (low quality) (SD = 1.41)40 and 5.75 (high quality) (SD = 2.50)41, respectively (Supplementary Table 3).
Meta-analysis of peripheral iron levels in the children with ADHD versus controls: peripheral ferritin
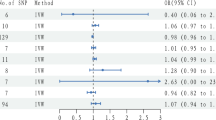
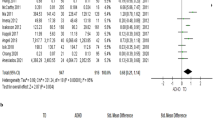
Overall, the meta-analysis results demonstrated that the peripheral serum ferritin level in the children with ADHD (n = 1560, mean age = 10.1, mean female proportion = 36.6%) was significantly lower than in the children without ADHD (n = 4691, mean age = 10.4, mean female proportion = 21.9%)25,26,28,29,32,33,34,35,36,37,38,39,42 (k = 19, Hedges’ g = −0.246, 95% confidence interval (CI) = −0.440 to −0.051, p = 0.013) (Fig. 2A). There was evidence of high heterogeneity (Q value = 105.528, df = 18, I2 = 82.94%, p < 0.001) but not publication bias according to Egger’s regression test (t = 0.816, df = 17, p = 0.426). The significance of the results did not change after sensitivity analysis with the removal of one study. However, meta-analysis could not be performed on plasma ferritin or peripheral blood ferritin levels because there were fewer than three datasets.
Forest plots showing effect sizes (Hedges’ g) and 95% confidence intervals (CIs) from individual studies and pooled results of all included studies comparing (A) serum ferritin, (B) serum iron, and (C) serum transferrin levels in children with and without ADHD; (D) Forest plot showing effect sizes (Hedges’ g) and 95% CIs from individual studies and pooled results comparing the severity of ADHD symptoms in children with and without iron deficiency (ID); (E) Forest plot pooling the adjusted odds ratio (OR) comparing the association between ADHD and ID. Figure 2(A) serum ferritin levels (p = 0.013) but not (B) serum iron (p = 0.569) or (C) serum transferrin (p = 0.096) levels were significantly lower in the children with ADHD compared to those without ADHD. Figure 2(D) The severity of ADHD symptoms was significantly greater in the children with ID than in those without ID (p < 0.001). Figure 2(E) indicated a significantly high association between ADHD and ID based on the pooled adjusted OR (p = 0.031).
Meta-regression analysis
The ESs of differences in serum ferritin levels between the children with ADHD and controls had significantly positive association between mean age (slope = 0.187, k = 17, p = 0.025) but not significantly moderated by proportion of females (p = 0.061), percentage of combined subtypes of ADHD (p = 0.550), percentage of the inattention subtype of ADHD (p = 0.860), percentage of the hyperactivity/impulsivity subtype of ADHD (p = 0.884), cognition (in forms of mean IQ) (p = 0.574), sample size of the group (p = 0.740), and geographical latitude of where the study was conducted (p = 0.690) (Supplementary Table 4).
Subgroup meta-analysis
To identify potential sources of heterogeneity, we focused on the studies recruiting children who did not receive medications. Three studies with four datasets were included34,38,42, and the results of meta-analysis showed that there was no significant difference in serum ferritin level between the children with ADHD and the controls (k = 4, Hedges’ g = −0.287, 95% CI = −0.812 to 0.237, p = 0.283).
Meta-analysis of peripheral iron levels in the children with ADHD versus controls: peripheral iron
Meta-analysis demonstrated that the serum iron level in children with ADHD (n = 941, mean age = 11.1, mean female proportion = 45.4%) was not significantly different from the children without ADHD (n = 847, mean age = 11.2, mean female proportion = 48.0%)26,27,35,36,38,42 (k = 9, Hedges’ g = −0.060, 95% CI = −0.268 to 0.147, p = 0.569) (Fig. 2B). There was evidence of high heterogeneity (Q value = 24.271, df = 8, I2 = 67.04%, p = 0.002) but no significant publication bias according to the funnel plot (Supplement Fig. 2). After removing the study by Chen (2004) via sensitivity testing, the pooled data suggested that there was a significantly lower serum iron level in the children with ADHD compared to the controls (Hedges’ g = −0.186, 95% CI = −0.323 to −0.048, p = 0.008)27.
Meta-regression analysis
The ESs of differences in serum iron levels between the children with ADHD and controls were not significantly moderated by mean age (p = 0.827), proportion of females (p = 0.958), sample size of the ADHD group (p = 0.388), and geographical latitude of where the study was conducted (p = 0.361) (Supplementary Table 4).
Meta-analysis of peripheral iron levels in the children with ADHD versus controls: peripheral transferrin
Meta-analysis demonstrated that there was no significant difference in serum transferrin level between the children with (n = 89, mean age = 8.2, mean female proportion = 36.3%) and without ADHD (n = 90, mean age = 8.2, mean female proportion = 38.1%)35,36 (k = 3, Hedges’ g = −0.324, 95% CI = −0.704 to 0.057, p = 0.096) (Fig. 2C). There was no evidence of significant heterogeneity (Q value = 3.147, df = 2, I2 = 36.440%, p = 0.207) or publication bias according to the funnel plot (Supplementary Figure 2). In the sensitivity test, the insignificant meta-analysis result became significant, and the children with ADHD had a significantly lower level of serum transferrin than those without ADHD after removing the subjects who received methylphenidate in the study by Menegassi, M. (2010) (Hedges’ g = −0.494, 95% CI = −0.828 to −0.160, p = 0.004)36. We could not perform meta-analysis on plasma transferrin or peripheral blood transferrin levels because there were fewer than three datasets.
Meta-regression analysis
Meta-regression and subgroup meta-analysis could not be performed as fewer than the minimal number of datasets were available.
Meta-analysis of the severity of ADHD in the children with ID versus controls
The severity of ADHD symptoms was significantly higher in the children with ID than in those without ID24,30,31 (79 children with ID, 76 children without ID, total mean age = 4.6, total mean female proportion = 38.7%) (k = 3; Hedges’ g = 0.888, 95% CI = 0.327 to 1.450, p = 0.002) (Fig. 2D) without evidence of heterogeneity (Q value = 3.763, df = 2, I2 = 46.850%, p = 0.152) or publication bias via inspection of the funnel plot (Supplementary Figure 2). The sensitivity test revealed that the significant meta-analysis result became insignificant after removing the studies either by Doom, J. R. (2015) (Hedges’ g = 0.777, 95% CI = −0.024 to 1.579, p = 0.057)30 or Fuglestad, A. J. (2013) (Hedges’ g = 0.742, 95% CI = −0.036 to 1.520, p = 0.062)31.
Meta-regression analysis
Meta-regression and subgroup meta-analysis could not be performed as fewer than the minimal number of datasets were available.
Meta-analysis of the association between ADHD and ID: the pooled adjusted OR
The meta-analysis of the pooled adjusted OR for the association between ADHD and ID revealed a significant association between ADHD and ID9,26,39 (k = 4; OR = 1.636, 95% CI = 1.047 to 2.557, p = 0.031) (Fig. 2E) with significant evidence of heterogeneity (Q value = 20.861, df = 3, I2 = 85.619%, p < 0.001) and publication bias via inspection of the funnel plot (Supplementary Figure 2). The adjusted ESs with publication bias through Duval and Tweedie’s trim and fill test were insignificant (OR = 1.217, 95% CI = 0.812 to 1.824). Furthermore, the sensitivity test revealed that the significant meta-analysis result became insignificant after removing the study by Bener, A. (2014) (OR = 1.364, 95% CI = 0.928 to 2.005, p = 0.114)26, the female subjects in the study by Chen, M. H. (2013) (OR = 1.556, 95% CI = 0.928 to 2.609, p = 0.093)9, or the male subjects in the study by Chen, M. H. (2013) (OR = 1.732, 95% CI = 0.855 to 3.506, p = 0.127)9. Meta-regression and subgroup meta-analysis could not be performed as fewer than the minimal number of datasets were available.
Discussion
The main results of our review and meta-analysis suggest that serum ferritin levels, but not iron or transferrin levels, are significantly lower in children diagnosed with ADHD than in those without ADHD. However, when removal of one potential confounding study, the result of meta-analysis of serum iron changed into significantly lower serum iron level in the children with ADHD compared to the controls. Children with ID were also more likely to have ADHD and have more severe ADHD symptoms than those without ID.
Our results are in general agreement with three previous meta-analyses14,15,16 (Table 2). However, our findings not simply confirm the same results in previous reports but also added further information upon current scientific knowledge, such as a higher odds of ADHD and higher severity of ADHD symptoms in the patients with ID and significantly lower serum iron level in the children with ADHD compared to the controls in specific post-hoc meta-analysis, such as sensitivity testing. Specifically, we found that the ORs and symptom severity of ADHD were higher in the patients with ID. Together with a lower ferritin level, this result may suggest an association between a lower serum iron level and ADHD symptomatology. Moreover, although both our meta-analysis and most recent meta-analysis by Wang et al. showed no significant difference in serum iron levels between ADHD and control groups16, sensitivity test was not done in the study by Wang et al. After we re-investigated the potential sources of insignificant results through sensitivity test (Table 2), we found significantly lower serum iron levels in the children with ADHD compared to those without ADHD after removing the study by Chen et al.27. In the study by Chen and colleagues (2004), we identified several significantly different baseline variables between the ADHD and control groups, including higher food iron intake and higher food vitamin C intake in the ADHD group. Vitamin C is known to enhance iron absorption from food43. Knowing that human iron nutrients primarily come from food intake, the children with ADHD included in the study by Chen who had significantly higher iron and vitamin C intake would be expected to show elevated blood iron levels. Such information about food intake was not provided by the other studies included in our subgroup meta-analysis of serum iron levels26,35,36,38. Therefore, it was reasonable to exclude data from the study by Chen et al.27 when interpreting the results of subgroup meta-analysis of serum iron levels. Following exclusion from this study, the results of subgroup meta-analysis of serum iron levels revealed a significantly lower serum iron level in the children with ADHD than in those without ADHD (Hedges’ g = −0.186, 95% CI = −0.323 to −0.048, p = 0.008). This provides further evidence of a shortage of body iron stores in children diagnosed with ADHD. Taken together, our findings provide tentative evidence that deficient iron storage in children with ADHD may be involved in the pathophysiology of the condition. However, future longitudinal research is required to confirm/refute this tentative hypothesis.
The relationship between ID and ADHD may be explained by several possible pathophysiological mechanisms. First, low peripheral iron levels, indicating insufficient iron storage, may dysregulate dopaminergic neurons, which may play a prominent role in the pathoetiology of ADHD5,44,45. In brief, iron aids in dopamine synthesis by acting as a co-factor for tyrosine hydroxylase, which is a rate-limiting enzyme for the conversion of hydroxylation of tyrosine to L-DOPA, a precursor of dopamine46. Iron deficiency may therefore result in disruption of dopamine activity, as shown in several animal studies12,47,48. This dysregulation of dopaminergic neurons may further result in multiple frontal dysfunctions that mimic the symptoms of ADHD49. In addition, ADHD has been found to be more prevalent in patients with restless leg syndrome (RLS) (27.62% with a diagnosis of ADHD), and iron deficiency and dopamine system dysregulation has also been reported to play an important role in RLS47,48,50. Therefore, iron deficiency with resulting brain dopamine dysfunction may be a common pathway for the pathophysiology of both disorders. In addition to the dopamine theory, lower ferritin levels may provide indirect evidence of elevated oxidative stress17, and heightened oxidative stress has also been reported in patients with ADHD51. This increased oxidative stress burden may disturb neurodevelopmental trajectories and gene functions potentially predisposing to the onset of ADHD51. However, given the observational nature of our data, the precise mechanisms and directionality of any relationships we observed cannot be verified.
Our meta-analysis focused on peripheral iron levels rather than brain iron levels. The extent to which peripheral and brain iron levels are correlated remains unclear. Cortese and colleagues provided some evidence that brain iron levels in the bilateral thalamus were significantly lower in children with ADHD compared to healthy controls28. However, another MRI study by Adisetiyo et al. failed to find an association between brain iron levels and serum ferritin levels42. Therefore, further studies are required to explore the relationship between brain and serum iron levels.
In the main result of our meta-analysis, high heterogeneity had been detected through the results. To address the potential source of heterogeneity, we arranged subgroup meta-analysis and meta-regression to investigate it. In part of subgroup analysis, if we only included the studies that recruited children who did not take medications, the difference in ferritin level between the ADHD and control groups became non-significant. Although, no obvious interactions have been reported between most drugs and ferritin level, D’Amato suggested that methylphenidate may cause a poor appetite and possibly less iron intake in children with ADHD52. However, only four of the nineteen datasets recruited drug-free participants, and most of the other studies did not provide much information about the medications prescribed to their participants. Therefore, further studies are warranted including drug-free participants only to further investigate the relationship between methylphenidate and ferritin level in children with ADHD. In addition to the potentially confounding effect by medication, the different food intake pattern would also contribute impact on the iron storage. For example, Lane and the colleague (2014) provided evidences about the enhancing effect on iron absorption by vitamin C intake43. Studies recruited subjects with high vitamin C intake or iron supplementation would have confounded results compared to others27.
Finally, in part of meta-regression, we tried to address the potential impact of some clinical variables on the peripheral iron levels. The differences in serum ferritin levels between the children with ADHD and the controls were only significantly moderated by mean age but not by other factors including proportion of females, subtype of ADHD, cognition (in forms of mean IQ), sample size of the ADHD group, and geographical latitude of where the study was conducted. We are uncertain about the significance of the finding that the difference in ferritin level in ADHD and control groups became larger with age because there were no previous reports or studies addressed this issue. However, previous studies showed that the reference range of ferritin level became wider and higher with age in pediatric population53. Therefore, further studies investigating ferritin level and ADHD symptoms may need to take this factor into account, before analyzing their study results. On the other hand, our meta-regression did not show significant associations between the prevalence of ADHD in children with ID and mean age, mean proportion of females, and geographic latitude of where the study was conducted.
Limitations
There are several limitations to the current study. First, the total number of included studies was relatively small, and therefore there was a risk of type I and type II errors. Second, our main targets focused on peripheral samples of iron status parameters rather than CNS parameters because few studies provided such information28,42. Third, we could not fully exclude the potential confounding effect of food iron intake on peripheral iron status because only a few studies provided such information27. In addition, we could not completely rule out the potential confounding effect of the ability of different types of assay to detect iron due to the inconsistent reporting of those data across the included studies. Fourth, we lacked comparisons of changes in iron status in the children with ADHD in long-term follow-up because there were few cohort studies7,39. Fifth, we could not perform subgroup meta-analysis of plasma ferritin, iron, or transferrin due to the limited number of datasets available in the studies. Finally, we could not perform meta-regression of peripheral iron levels and attention because of the limited data available.
Conclusions
The results of our meta-analysis suggest that children diagnosed with ADHD have lower serum ferritin levels compared to those without ADHD. We also observed that the children with ID were more likely to have ADHD and to suffer from more severe ADHD symptoms compared to those without ID. We therefore suggest that further studies are warranted to explore the benefits of iron supplementation in children with ADHD with ID, in particular those with more severe ADHD symptoms. However, given the cross-sectional nature of most of the available studies, further longitudinal and cohort studies are required to thoroughly evaluate the relationships between iron status and ADHD symptoms, and to elucidate the potential pathophysiological mechanisms. In addition, further studies may be needed to investigate the relationship between methylphenidate and ferritin level to exclude any potential effects of methylphenidate on oral iron intake.
Methods and Materials
The current study followed the Meta-analysis Of Observational Studies in Epidemiology (MOOSE) guidelines54 (see Supplementary Table 1 and Supplementary Figure 1). The meta-analysis followed our previous defined but unpublished protocol and was approved by the Institutional Review Board of Tri-Service General Hospital (TSGHIRB: B-105-12).
Eligibility criteria
The inclusion criteria were: (a) observational studies, including a cohort or cross-sectional study design, comparing all peripheral iron status parameters including iron, ferritin, and transferrin levels in children with ADHD (confirmed by either a structured or non-structured diagnostic interview) and controls; and (b) clinical human studies. We excluded preclinical studies, review articles, meeting abstracts, or peer-reviewed original articles not conducted in humans.
Search strategy and study selection
Two independent authors conducted an electronic systematic literature search from inception to August 9th, 2017 across PubMed, ScienceDirect, Cochrane CENTRAL, and ClinicalTrials.gov databases. We used the following keywords: “(iron OR ferritin OR ferrous) AND (Attention deficit hyperactivity disorder OR ADHD)”. To expand our eligible list, we consulted the reference lists of the included articles and recent reviews14,55,56.
At the eligibility stage, two authors (YS Cheng and PT Tseng) screened the titles and abstracts of all results to assess potential eligibility. The authors then reviewed the full-text articles which were deemed potentially eligible. A final list of included studies was determined and any inconsistencies were resolved by consensus or via thorough discussion with a third reviewer (PY Lin).
Primary outcomes
We set the primary outcomes as the difference in peripheral iron levels (including iron, ferritin, or transferrin) between children with ADHD and controls, and the OR of ADHD or severity of ADHD symptoms between children with or without ID. If the data of interest were not available in the articles, we contacted the authors twice over a month to request the data.
Data extraction
Two independent authors (YS Cheng and PT Tseng) extracted data using a predetermined list of variables of interest, which included: prevalence/incidence rates of ADHD, prevalence rates of iron deficiency, peripheral iron levels, amount of food iron intake, mean age, gender distribution in the form of the percentage of females, body mass index, percentage of ADHD subtypes, cognitive performance in the form of IQ, parental tobacco smoking/alcohol consumption, ethnicity (including African, Caucasian, Asian, and Hispanic), parental history of ADHD, geographical latitude of where the study was conducted, and the type of assay used to detect the iron level.
Assessment of study quality
We used the Newcastle-Ottawa Scale for cohort studies and case control studies. For cross-sectional studies, we used a modified version of the Newcastle-Ottawa Scale for observational studies to assess the quality of the included studies. This modified version of the Newcastle-Ottawa Scale score for observational studies ranges from zero to six, and a score greater than three was classified as a high-quality study. For case control studies, the Newcastle-Ottawa Scale score ranges from zero to ten, with a score greater than five being classified as a high-quality study41. For cohort studies, the Newcastle-Ottawa Scale score ranges from zero to nine, with a score of six or more being classified as a high-quality study40.
Statistical analysis
The current study was conducted in two parts. First, we analyzed the data considering iron in relation to the children with ADHD compared to the controls. Second, we analyzed the data about the severity of ADHD symptoms or OR of ADHD in the children with ID compared to the controls. To control for the potential confounding effects of clinical variables, we performed further meta-analysis based on the pooled adjusted OR from the recruited studies. In brief, we extracted the adjusted OR with regards to the association of ADHD in the children with ID from the recruited studies to calculate the pooled adjusted OR of the association between ADHD and ID.
Based on the presumed heterogeneity of background and population among the recruited studies, we conducted the meta-analyses with a random effects model rather than a fixed effects model57. In brief, random-effects modeling is more stringent than fixed-effects modeling and incorporates a between-study variance in the calculations58. For continuous outcomes (i.e. differences in iron levels between the children with ADHD and the controls), we calculated Hedges’ g and 95% CI. For dichotomous outcomes (i.e. differences in the prevalence of ID between the children with ADHD and the controls) we calculated the OR and 95% CI.
Heterogeneity, publication bias, and sensitivity test
Heterogeneity was assessed using the Cochran Q test59. The I2 statistic should be interpreted as the proportion of heterogeneity a study estimates that is due to heterogeneity60. For publication bias, we used direct inspection of funnel plots for fewer than 10 datasets61 and Egger’s regression test for 10 or more datasets62. We also used the Duval and Tweedie’s trim-and-fill procedure to adjust the ESs when publication bias was evident63. We also used a sensitivity test with one study removed to investigate any potential outliers present in the recruited studies64.
Subgroup meta-analysis and meta-regression analysis
To discover any potential sources of heterogeneity, subgroup analyses were performed to explore potential interactions between clinical variables and peripheral iron status parameters in the children with ADHD compared to the controls. We only performed subgroup analysis whenever data from three independent datasets were available65. The main clinical target for subgrouping included sample sources, subjects who were or were not drug free, and the situation when blood was drawn. When data were available for a moderator in more than five studies, we performed unrestricted maximum likelihood random-effects meta-regression to explore any potential source of heterogeneity. The moderators of interest included mean age, female proportion, body mass index, percentage of ADHD subtypes, mean IQ, percentage of parental tobacco smoking/alcohol consumption, percentage of each ethnicity, percentage of parental history of ADHD, sample size of the disease groups, and geographical latitude of where the study was conducted.
The meta-analyses were conducted using Comprehensive Meta-Analysis software, version 3 (Biostat, Englewood, NJ).
Data availability
The datasets generated and analyzed during the current study are available from the corresponding author on reasonable request.
References
Thomas, R., Sanders, S., Doust, J., Beller, E. & Glasziou, P. Prevalence of attention-deficit/hyperactivity disorder: a systematic review and meta-analysis. Pediatrics 135, e994–1001, https://doi.org/10.1542/peds.2014-3482 (2015).
Thapar, A. & Cooper, M. Attention deficit hyperactivity disorder. Lancet 387, 1240–1250, https://doi.org/10.1016/S0140-6736(15)00238-X (2016).
Jameson, N. D. et al. Medical Comorbidity of Attention-Deficit/Hyperactivity Disorder in US Adolescents. J Child Neurol 31, 1282–1289, https://doi.org/10.1177/0883073816653782 (2016).
Faraone, S. V. et al. Molecular genetics of attention-deficit/hyperactivity disorder. Biol Psychiatry 57, 1313–1323, https://doi.org/10.1016/j.biopsych.2004.11.024 (2005).
Biederman, J. Attention-deficit/hyperactivity disorder: a selective overview. Biol Psychiatry 57, 1215–1220, https://doi.org/10.1016/j.biopsych.2004.10.020 (2005).
Polanska, K., Jurewicz, J. & Hanke, W. Exposure to environmental and lifestyle factors and attention-deficit / hyperactivity disorder in children - a review of epidemiological studies. Int J Occup Med Environ Health 25, 330–355, https://doi.org/10.2478/S13382-012-0048-0 (2012).
Konofal, E. et al. Effects of iron supplementation on attention deficit hyperactivity disorder in children. Pediatr Neurol 38, 20–26, https://doi.org/10.1016/j.pediatrneurol.2007.08.014 (2008).
Verlaet, A. A., Noriega, D. B., Hermans, N. & Savelkoul, H. F. Nutrition, immunological mechanisms and dietary immunomodulation in ADHD. Eur Child Adolesc Psychiatry 23, 519–529, https://doi.org/10.1007/s00787-014-0522-2 (2014).
Chen, M. H. et al. Association between psychiatric disorders and iron deficiency anemia among children and adolescents: a nationwide population-based study. BMC Psychiatry 13, 161, https://doi.org/10.1186/1471-244X-13-161 (2013).
Doom, J. R. & Georgieff, M. K. Striking while the iron is hot: Understanding the biological and neurodevelopmental effects of iron deficiency to optimize intervention in early childhood. Curr Pediatr Rep 2, 291–298, https://doi.org/10.1007/s40124-014-0058-4 (2014).
Bakoyiannis, I. et al. An explanation of the pathophysiology of adverse neurodevelopmental outcomes in iron deficiency. Rev Neurosci 26, 479–488, https://doi.org/10.1515/revneuro-2015-0012 (2015).
Hyacinthe, C. et al. Blood withdrawal affects iron store dynamics in primates with consequences on monoaminergic system function. Neuroscience 290, 621–635, https://doi.org/10.1016/j.neuroscience.2015.01.057 (2015).
Sever, Y., Ashkenazi, A., Tyano, S. & Weizman, A. Iron treatment in children with attention deficit hyperactivity disorder. A preliminary report. Neuropsychobiology 35, 178–180 (1997).
Scassellati, C., Bonvicini, C., Faraone, S. V. & Gennarelli, M. Biomarkers and attention-deficit/hyperactivity disorder: a systematic review and meta-analyses. J Am Acad Child Adolesc Psychiatry 51, 1003–1019 e1020, https://doi.org/10.1016/j.jaac.2012.08.015 (2012).
Tan, L. N., Wei, H. Y., Zhang, Y. D., Lu, A. L. & Li, Y. Relationship between serum ferritin levels and susceptibility to attention deficit hyperactivity disorder in children: a Meta analysis. Zhongguo Dang Dai Er Ke Za Zhi 13, 722–724 (2011).
Wang, Y., Huang, L., Zhang, L., Qu, Y. & Mu, D. Iron Status in Attention-Deficit/Hyperactivity Disorder: A Systematic Review and Meta-Analysis. PLoS One 12, e0169145, https://doi.org/10.1371/journal.pone.0169145 (2017).
Finazzi, D. & Arosio, P. Biology of ferritin in mammals: an update on iron storage, oxidative damage and neurodegeneration. Arch Toxicol 88, 1787–1802, https://doi.org/10.1007/s00204-014-1329-0 (2014).
Sharma, A. & Couture, J. A review of the pathophysiology, etiology, and treatment of attention-deficit hyperactivity disorder (ADHD). Ann Pharmacother 48, 209–225, https://doi.org/10.1177/1060028013510699 (2014).
Konofal, E., Cortese, S., Lecendreux, M., Arnulf, I. & Mouren, M. C. Effectiveness of iron supplementation in a young child with attention-deficit/hyperactivity disorder. Pediatrics 116, e732–734, https://doi.org/10.1542/peds.2005-0715 (2005).
Al-Ayadhi, L. Y. Heavy metals and trace elements in hair samples of autistic children in central Saudi Arabia. Neurosciences (Riyadh) 10, 213–218 (2005).
Antalis, C. J. et al. Omega-3 fatty acid status in attention-deficit/hyperactivity disorder. Prostaglandins Leukot Essent Fatty Acids 75, 299–308, https://doi.org/10.1016/j.plefa.2006.07.004 (2006).
Dura-Trave, T. & Gallinas-Victoriano, F. Caloric and nutrient intake in children with attention deficit hyperactivity disorder treated with extended-release methylphenidate: analysis of a cross-sectional nutrition survey. JRSM Open 5, 2042533313517690, https://doi.org/10.1177/2042533313517690 (2014).
Mahmoud, M. M., El-Mazary, A. A., Maher, R. M. & Saber, M. M. Zinc, ferritin, magnesium and copper in a group of Egyptian children with attention deficit hyperactivity disorder. Ital J Pediatr 37, 60, https://doi.org/10.1186/1824-7288-37-60 (2011).
Abou-Khadra, M. K., Amin, O. R., Shaker, O. G. & Rabah, T. M. Parent-reported sleep problems, symptom ratings, and serum ferritin levels in children with attention-deficit/hyperactivity disorder: a case control study. BMC Pediatr 13, 217, https://doi.org/10.1186/1471-2431-13-217 (2013).
Bala, K. A. et al. Hormone disorder and vitamin deficiency in attention deficit hyperactivity disorder (ADHD) and autism spectrum disorders (ASDs). J Pediatr Endocrinol Metab 29, 1077–1082, https://doi.org/10.1515/jpem-2015-0473 (2016).
Bener, A., Kamal, M., Bener, H. & Bhugra, D. Higher prevalence of iron deficiency as strong predictor of attention deficit hyperactivity disorder in children. Ann Med Health Sci Res 4, S291–297, https://doi.org/10.4103/2141-9248.141974 (2014).
Chen, J. R., Hsu, S. F., Hsu, C. D., Hwang, L. H. & Yang, S. C. Dietary patterns and blood fatty acid composition in children with attention-deficit hyperactivity disorder in Taiwan. J Nutr Biochem 15, 467–472, https://doi.org/10.1016/j.jnutbio.2004.01.008 (2004).
Cortese, S. et al. Brain iron levels in attention-deficit/hyperactivity disorder: a pilot MRI study. World J Biol Psychiatry 13, 223–231, https://doi.org/10.3109/15622975.2011.570376 (2012).
Donfrancesco, R. et al. Iron and ADHD: time to move beyond serum ferritin levels. J Atten Disord 17, 347–357, https://doi.org/10.1177/1087054711430712 (2013).
Doom, J. R., Georgieff, M. K. & Gunnar, M. R. Institutional care and iron deficiency increase ADHD symptomology and lower IQ 2.5-5 years post-adoption. Dev Sci 18, 484–494, https://doi.org/10.1111/desc.12223 (2015).
Fuglestad, A. J. et al. Iron deficiency after arrival is associated with general cognitive and behavioral impairment in post-institutionalized children adopted from Eastern Europe. Matern Child Health J 17, 1080–1087, https://doi.org/10.1007/s10995-012-1090-z (2013).
Juneja, M., Jain, R., Singh, V. & Mallika, V. Iron deficiency in Indian children with attention deficit hyperactivity disorder. Indian Pediatr 47, 955–958 (2010).
Konofal, E. et al. Impact of restless legs syndrome and iron deficiency on attention-deficit/hyperactivity disorder in children. Sleep Med 8, 711–715, https://doi.org/10.1016/j.sleep.2007.04.022 (2007).
Konofal, E., Lecendreux, M., Arnulf, I. & Mouren, M. C. Iron deficiency in children with attention-deficit/hyperactivity disorder. Arch Pediatr Adolesc Med 158, 1113–1115, https://doi.org/10.1001/archpedi.158.12.1113 (2004).
Kwon, H. J. et al. Transferrin in korean children with attention deficit hyperactivity disorder. Psychiatry Investig 8, 366–371, https://doi.org/10.4306/pi.2011.8.4.366 (2011).
Menegassi, M. et al. Food intake and serum levels of iron in children and adolescents with attention-deficit/hyperactivity disorder. Rev Bras Psiquiatr 32, 132–138 (2010).
Millichap, J. G., Yee, M. M. & Davidson, S. I. Serum ferritin in children with attention-deficit hyperactivity disorder. Pediatr Neurol 34, 200–203, https://doi.org/10.1016/j.pediatrneurol.2005.09.001 (2006).
Percinel, I., Yazici, K. U. & Ustundag, B. Iron Deficiency Parameters in Children and Adolescents with Attention-Deficit/Hyperactivity Disorder. Child Psychiatry Hum Dev 47, 259–269, https://doi.org/10.1007/s10578-015-0562-y (2016).
Romanos, M. et al. No cross-sectional and longitudinal association of ferritin and symptoms of attention-deficit-/hyperactivity disorder in a large population-based sample of children: results from the GINIplus and LISAplus studies. Atten Defic Hyperact Disord 5, 313–320, https://doi.org/10.1007/s12402-013-0108-8 (2013).
Stang, A. Critical evaluation of the Newcastle-Ottawa scale for the assessment of the quality of nonrandomized studies in meta-analyses. Eur J Epidemiol 25, 603–605, https://doi.org/10.1007/s10654-010-9491-z (2010).
Juni, P., Witschi, A., Bloch, R. & Egger, M. The hazards of scoring the quality of clinical trials for meta-analysis. JAMA 282, 1054–1060 (1999).
Lane, D. J. & Richardson, D. R. The active role of vitamin C in mammalian iron metabolism: much more than just enhanced iron absorption! Free Radic Biol Med 75, 69–83, https://doi.org/10.1016/j.freeradbiomed.2014.07.007 (2014).
Beard, J. L., Chen, Q., Connor, J. & Jones, B. C. Altered monamine metabolism in caudate-putamen of iron-deficient rats. Pharmacol Biochem Behav 48, 621–624 (1994).
Burhans, M. S. et al. Iron deficiency: differential effects on monoamine transporters. Nutr Neurosci 8, 31–38, https://doi.org/10.1080/10284150500047070 (2005).
Daubner, S. C., Le, T. & Wang, S. Tyrosine hydroxylase and regulation of dopamine synthesis. Arch Biochem Biophys 508, 1–12, https://doi.org/10.1016/j.abb.2010.12.017 (2011).
Earley, C. J. et al. Altered brain iron homeostasis and dopaminergic function in Restless Legs Syndrome (Willis-Ekbom Disease). Sleep Med 15, 1288–1301, https://doi.org/10.1016/j.sleep.2014.05.009 (2014).
Youdim, M. B., Ben-Shachar, D., Ashkenazi, R. & Yehuda, S. Brain iron and dopamine receptor function. Adv Biochem Psychopharmacol 37, 309–321 (1983).
Ranganath, A. & Jacob, S. N. Doping the Mind: Dopaminergic Modulation of Prefrontal Cortical Cognition. Neuroscientist 22, 593–603, https://doi.org/10.1177/1073858415602850 (2016).
Ghorayeb, I., Gamas, A., Mazurie, Z. & Mayo, W. Attention-Deficit Hyperactivity and Obsessive-Compulsive Symptoms in Adult Patients With Primary Restless Legs Syndrome: Different Phenotypes of the Same Disease? Behav Sleep Med 1–8, https://doi.org/10.1080/15402002.2017.1326919 (2017).
Kul, M. et al. Evaluation of Oxidative Metabolism in Child and Adolescent Patients with Attention Deficit Hyperactivity Disorder. Psychiatry Investig 12, 361–366, https://doi.org/10.4306/pi.2015.12.3.361 (2015).
Adisetiyo, V. et al. Multimodal MR imaging of brain iron in attention deficit hyperactivity disorder: a noninvasive biomarker that responds to psychostimulant treatment? Radiology 272, 524–532, https://doi.org/10.1148/radiol.14140047 (2014).
D’Amato, T. J. Is iron deficiency causative of attention-deficit/hyperactivity disorder? Arch Pediatr Adolesc Med 159, 788; author reply 788, https://doi.org/10.1001/archpedi.159.8.788-a (2005).
Zacharski, L. R., Ornstein, D. L., Woloshin, S. & Schwartz, L. M. Association of age, sex, and race with body iron stores in adults: analysis of NHANES III data. Am Heart J 140, 98–104, https://doi.org/10.1067/mhj.2000.106646 (2000).
Stroup, D. F. et al. Meta-analysis of observational studies in epidemiology: a proposal for reporting. Meta-analysis Of Observational Studies in Epidemiology (MOOSE) group. JAMA 283, 2008–2012 (2000).
Adisetiyo, V. & Helpern, J. A. Brain iron: a promising noninvasive biomarker of attention-deficit/hyperactivity disorder that warrants further investigation. Biomark Med 9, 403–406, https://doi.org/10.2217/bmm.15.9 (2015).
Yui, K., Imataka, G., Nakamura, H., Ohara, N. & Naito, Y. Eicosanoids Derived From Arachidonic Acid and Their Family Prostaglandins and Cyclooxygenase in Psychiatric Disorders. Curr Neuropharmacol 13, 776–785 (2015).
Higgins, J. P., Thompson, S. G. & Spiegelhalter, D. J. A re-evaluation of random-effects meta-analysis. J R Stat Soc Ser A Stat Soc 172, 137–159, https://doi.org/10.1111/j.1467-985X.2008.00552.x (2009).
Borenstein, M., Hedges, L. V., Higgins, J. P. & Rothstein, H. R. A basic introduction to fixed-effect and random-effects models for meta-analysis. Res Synth Methods 1, 97–111, https://doi.org/10.1002/jrsm.12 (2010).
Higgins, J. P. & Thompson, S. G. Quantifying heterogeneity in a meta-analysis. Stat Med 21, 1539–1558, https://doi.org/10.1002/sim.1186 (2002).
Borenstein, M., Higgins, J. P., Hedges, L. V. & Rothstein, H. R. Basics of meta-analysis: I2 is not an absolute measure of heterogeneity. Res Synth Methods 8, 5–18, https://doi.org/10.1002/jrsm.1230 (2017).
Higgins, J. P. & Green, S. In Cochrane Handbook for Systematic Reviews of Interventions (eds Higgins, J. P. & Green, S.). (Cochrane Library, 2011).
Egger, M., Davey Smith, G., Schneider, M. & Minder, C. Bias in meta-analysis detected by a simple, graphical test. BMJ 315, 629–634 (1997).
Duval, S. & Tweedie, R. Trim and fill: A simple funnel-plot-based method of testing and adjusting for publication bias in meta-analysis. Biometrics 56, 455–463 (2000).
Tobias, A. Assessing the influence of a single study in meta-analysis. STATA technical bulletin 47, 15–17 (1999).
Davey, J., Turner, R. M., Clarke, M. J. & Higgins, J. P. Characteristics of meta-analyses and their component studies in the Cochrane Database of Systematic Reviews: a cross-sectional, descriptive analysis. BMC Med Res Methodol 11, 160, https://doi.org/10.1186/1471-2288-11-160 (2011).
Acknowledgements
We thank the following authors during our requesting for provision of additional data for our meta-analysis: Doom J.R., Heinrich T.J., Konofal E., and Millichap J.G. The study of this article received no research funding.
Author information
Authors and Affiliations
Contributions
P.T.T. and Y.S.C. generated the research topic, did the literature search, and prepared the first manuscript. P.T.T. and Y.W.C. ran the statistical tests. D.J.L, T.Y.C., W.C.Y., C.H.T., C.S.C., W.C.Y., H.Y.L., and C.K.W. conceived the study concept and contributed important parts in data interpretation. P.T.T., C.F.Y. and P.Y.L. analyzed and interpreted the data. B.S., P.W., and A.F.C. critically read the manuscript and made vital suggestions in revision. All authors reviewed and approved the final manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Tseng, PT., Cheng, YS., Yen, CF. et al. Peripheral iron levels in children with attention-deficit hyperactivity disorder: a systematic review and meta-analysis. Sci Rep 8, 788 (2018). https://doi.org/10.1038/s41598-017-19096-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-19096-x
This article is cited by
-
Nutrients patterns and attention deficit hyperactivity disorder among Egyptian children: a sibling and community matched case-control study
European Journal of Clinical Nutrition (2024)
-
A holistic review of recent advances in nano-based drug delivery systems for the treatment of triple-negative breast cancer (TNBC)
Journal of Nanoparticle Research (2024)
-
Maternal iron status during pregnancy and attention deficit/hyperactivity disorder symptoms in 7-year-old children: a prospective cohort study
Scientific Reports (2022)
-
Environmental Metal Exposure, Neurodevelopment, and the Role of Iron Status: a Review
Current Environmental Health Reports (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.