Abstract
The interrelationships of the four classes of Myriapoda have been an unresolved question in arthropod phylogenetics and an example of conflict between morphology and molecules. Morphology and development provide compelling support for Diplopoda (millipedes) and Pauropoda being closest relatives, and moderate support for Symphyla being more closely related to the diplopod-pauropod group than any of them are to Chilopoda (centipedes). In contrast, several molecular datasets have contradicted the Diplopoda–Pauropoda grouping (named Dignatha), often recovering a Symphyla–Pauropoda group (named Edafopoda). Here we present the first transcriptomic data including a pauropod and both families of symphylans, allowing myriapod interrelationships to be inferred from phylogenomic data from representatives of all main lineages. Phylogenomic analyses consistently recovered Dignatha with strong support. Taxon removal experiments identified outgroup choice as a critical factor affecting myriapod interrelationships. Diversification of millipedes in the Ordovician and centipedes in the Silurian closely approximates fossil evidence whereas the deeper nodes of the myriapod tree date to various depths in the Cambrian-Early Ordovician, roughly coinciding with recent estimates of terrestrialisation in other arthropod lineages, including hexapods and arachnids.
Similar content being viewed by others
Introduction
The evolutionary interrelationships between and within major arthropod groups were subject to much instability in the early years of molecular phylogenetics. Some hypotheses to emerge from that era – such as crustacean paraphyly with respect to Hexapoda (including insects) – have stood the test of time, whereas others have fallen by the wayside. Controversial results were exposed to be artefacts of insufficient amounts of data, flawed analytical methods, or systematic error. In recent years, phylogenomic approaches drawing on vastly expanded gene and taxon coverage, combined with improved analytical approaches, have seen stable, well supported molecular hypotheses being recovered1,2,3,4,5, and these have eliminated several instances of incongruence with morphological trees that were introduced in earlier molecular studies.
Transcriptome-based phylogenies drawing on hundreds or thousands of orthologues have assisted with phylogenetic analyses for the major groups of millipedes6,7 and centipedes7,8 but relationships between the four main myriapod groups have not been as rigorously tested (Fig. 1). A particular limitation is that only Sanger-sequenced data are available for pauropods—a group that lies at the crux of a molecular and morphological conflict within Myriapoda—probably due to their small size, difficulty in finding them, and cryptic behaviour. Analyses based on 62 nuclear protein coding genes9,10 underpin a formal taxonomic proposal that Pauropoda is most closely related to Symphyla, a putative clade named Edafopoda11. This grouping, also recovered using nuclear ribosomal genes12 and mitochondrial genomes13, is highly unexpected from the perspective of morphology because an alternative grouping of Pauropoda and Diplopoda has been widely accepted for more than a century14,15,16. This hypothesis is named Dignatha (=Collifera), referring to the mandibles and first maxillae being the only functional mouthparts, with the postmaxillary segment being limbless, parts of it forming a tergite called the collum and not incorporated into the head. Other shared morphological characters include a first maxilla coalesced with a sternal intermaxillary plate, the vas deferens opening to the tips of conical penes between the second trunk leg pair, and the spiracles opening to a tracheal pouch that functions as an apodeme. Early post-embryonic development unites Dignatha based on a motionless pupoid stage immediately after hatching, followed by a hexapodous first free-living stage. The Dignatha hypothesis has also been supported by a few smaller molecular data sets17, but it has been contradicted by Edafopoda in the analysis of larger data sets [e.g., ref.11].
The four main groups of myriapods. (A) Otostigmus (Parotostigmus) pococki (Northern Range, Trinidad, Trinidad and Tobago) (Chilopoda, Scolopendromorpha). (B) Hanseniella sp. (South Island, New Zealand) (Symphyla, Scutigerellidae). (C) Pauropus huxleyi (Massachusetts, USA) (Pauropoda, Tetramerocerata). and (D) Platydesmus sp. (La Selva, Costa Rica) (Diplopoda, Platydesmida). Pictures by Gonzalo Giribet.
In order to investigate the conflicting support for Dignatha versus Edafopoda – and therefore to shed light on the backbone of the Myriapoda Tree of Life – we present the first transcriptomic data set including a pauropod and both families of symphylans. The new data are evaluated in a phylogenomic context specifically designed to test these hypotheses. Furthermore, we expand on previous efforts to date the myriapod phylogenetic tree by coding a morphological character set for the same set of species as sampled transcriptomically as well as key fossil species for their preserved morphological characters in order to estimate the age of diversification within Myriapoda, particularly with reference to the likely timing of terrestrialisation.
Results and Discussion
Pauropoda is the sister group to Diplopoda
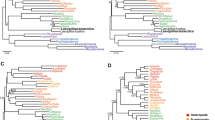
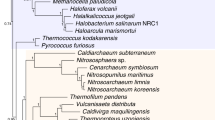
All the analyses (with the exception of two, in which Pauropoda was attracted to the outgroups, see below) recovered Pauropoda as the sister group to Diplopoda with high support (Fig. 2a,b). Notably, support for Dignatha is strong when the most complete taxonomic sampling of non-myriapod outgroups is used (Fig. 2a). Given the unanimity of support for Dignatha/Collifera in morphological studies, this stable, well-supported result reconciles classical morphological studies with molecules. In analyses based on more intensively sampled or closely related outgroups (discussed below), the sister group of Dignatha is Symphyla, together forming the traditional clade Progoneata. The only two analyses not recovering Dignatha (both maximum likelihood analyses not accounting for among-site rate heterogeneity and including the most distant outgroups) positioned Pauropoda at the base of the ingroup due to a long branch attraction artefact (LBA) (Fig. 2c,d). In fact, one of them even recovered Myriapoda as non-monophyletic (Fig. 2d), with the pauropod spuriously clustering within Pancrustacea, highlighting the potential of LBA in this data set. Edafopoda was not recovered in either of these two analyses, as symphylans fall as the sister group of Diplopoda + Chilopoda in both cases (although without strong support in one of the analyses; Fig. 2c). The attraction of symphylans and pauropods as Edafopoda (the only hypothesis exclusively based on molecular information) is therefore probably due to artefacts during phylogenetic reconstruction, as discussed below.
(A) Phylogenetic hypothesis of myriapod interrelationships (PhyloBayes, matrix 1). (B). DensiTree visualization of the four most congruent analyses (PhyloBayes, matrices 2 and 3; PhyML, matrix 3). (C,D). Main conflicting alternative hypotheses (C) PhyML, matrix 2; (D) PhyML, matrix 1. (E) Phylogenetic hypothesis of Myriapoda based on 187 morphological characters coded for both extant and extinct species (see Methods for further details); strict consensus of 105 trees of 257 steps; fossil taxa are identified with a dagger symbol. Black circles in nodes represent high support (>0.95 posterior probability, >90% bootstrap support). CHE: Chelicerata. PAN: Pancrustacea. CHI: Chilopoda. SYM: Symphyla. PAU: Pauropoda. DIP: Diplopoda. Colour codes for each clade are maintained in all figures.
Outgroup selection impacts on myriapod phylogeny
Despite Dignatha being recovered in most of our analyses, a result not often found in prior molecular studies, the interrelationships between it and the other two main groups of myriapods varied across analyses, being sensitive to outgroup choice. In the PhyloBayes analyses, matrices 1 and 3 recovered Progoneata, whereas in matrix 2 (in which only the more distantly allied chelicerates were selected as outgroups) symphylans appeared as sister group of the other Myriapoda instead of centipedes, as in the previous cases (Fig. 2a,b). The latter clade formed by these three groups was also recovered in the ML analyses (with or without strong support, though; Fig. 2c,d). This is not the first study in which this result was obtained: in Miyazawa et al.18 symphylans were likewise recovered as sister group to all other myriapods, followed by pauropods as sister group to millipedes plus centipedes. However, that study was based on just three Sanger sequenced genes, and conflicts with other well resolved nodes in our phylogeny (e.g., Dignatha). The present study and Fernández et al.7 suggest that outgroup selection is a major factor affecting phylogenetic reconstruction in myriapods. In addition, the latter study found that the most complete matrices were enriched in ribosomal proteins, and both factors strongly compromised the estimated relationships within the ingroup. In the present study, biases from ribosomal proteins were minimized by using a different orthology inference procedure, which ensures that only single copy genes are selected. In spite of this, it remains the case that the selection of only distant outgroups (chelicerates in this case) yields interrelationships of the myriapod classes that are less congruent with morphology than when closer and more comprehensively sampled outgroups are included. This study also highlights the importance of accounting for site-specific heterogeneity (through the CAT-GTR model of PhyloBayes) at least when taxon sampling is not dense for some of the groups, as even when only closer outgroups are included the long-branched pauropod is attracted to the equally long-branched Pancrustacea. The inclusion of more pauropods may alleviate this effect.
The timing of myriapod diversification
Diversification of Chilopoda (i.e., the basal split in the crown group) is dated to the Early Silurian (Fig. 3), not much earlier than the oldest fossil chilopods in the Late Silurian, these already being representatives of the chilopod crown group. Diversification of Diplopoda dates to the Middle Ordovician (autocorrelated rates)–earliest Silurian (uncorrelated rates). Though this is considerably older than the first millipede body fossils (from the Wenlock Series of the Silurian), it closely approximates the age of trace fossils that have been attributed to Diplopoda and especially compared to locomotion in Penicillata19,20. In contrast, deeper nodes associated with the divergences between myriapod classes are substantially older than available fossil data. No plausible total-group myriapod body fossils are known from the Cambrian, but as in previous studies dating Myriapoda3,17, some deep splits are estimated to be of Cambrian age. Diversification of Dignatha is inferred to date to the latest Cambrian-Early Ordovician, Progoneata to the mid-late Cambrian, and Myriapoda to the early-middle Cambrian (auto- and uncorrelated rates, respectively). The shared terrestrial adaptations of all extant myriapods (e.g., tracheae, Malpighian tubules, uniramous trunk limbs) suggest that the common ancestor of each of these estimated Cambrian nodes was terrestrial, coinciding (although being slightly younger) with estimates of terrestrialisation for other arthropod lineages, including arachnids and hexapods3,5. Although the trace fossil record is consistent with amphibious arthropods by the mid Cambrian21,22, and some such traces are potentially made by stem-group myriapods, current molecular estimates for early or middle Cambrian crown-group myriapods, earlier than the expected terrestrial flora, continue to pose an unanswered question in arthropod terrestrialisation.
Towards a fully-resolved Myriapoda Tree of Life
The branching pattern of the four main groups of myriapods has been one of the unresolved questions in arthropod phylogenetics, together with the interrelationships of the chelicerate orders and the exact sequence of crustaceans that led to the origins of hexapods. With the advent of phylogenomic methods, myriapod phylogeny has attracted attention during the last few years, with several studies devoted to shedding light on the interrelationships of the millipede6 and centipede8 orders, and more recently expanding taxon sampling to include most centipede families, most millipede orders and a couple of symphylans7. The different analyses of large data matrices combined in all these studies (as well as the current one) have allowed us to discern the main artefacts affecting phylogenetic reconstruction in this group of arthropods. The tree, its deep nodes congruent with traditional hypotheses based on morphology and development, can now be seen as a well-resolved backbone phylogeny with only a handful of untested placements, including the unsampled pauropod order Hexamerocerata, and the unexplored position of the diplopod orders Siphoniulida, Siphonophorida and Siphonocryptida. Some cases of incongruence between morphological and molecular data remain at shallower nodes, such as the interrelationships of the three orders of pentazonian millipedes23, and the position of the centipede orders Craterostigmomorpha and Lithobiomorpha relative to each other and to Scolopendromorpha + Geophilomorpha7.
Methods
Sample collection and molecular techniques
Fourteen species representing the four major groups of myriapods (Chilopoda, Diplopoda, Pauropoda and Symphyla) were included in this study. Building upon previous work6,7, our sampling was designed to maximize representation of all groups, including all orders of centipedes, both families of symphylans, the main clades of millipedes, and pauropods. New sequence data were generated from organisms targeted for their instability or lack of representation in prior analyses: a pauropod (Pauropus huxleyi) and a symphylan from the family Scolopendrellidae (Scutigerellidae was already represented in earlier studies). Information on sampling localities and accession numbers in the Sequence Read Archive database for each transcriptome can be found in Table 1. The remaining 12 myriapods from Brewer and Bond6 and our own published data7 were available from the Sequence Read Archive (SRA). The following taxa were included as outgroups: a crustacean (Daphnia pulex), two hexapods (Drosophila melanogaster, Folsomia candida), and three chelicerates (Limulus polyphemus, Liphistius malayanus and Centruroides vittatus). The new sequenced cDNA libraries are accessioned in SRA (Table 1). Tissue preservation and RNA sequencing are as described in Fernández et al.8. All molecular data included in this study were sequenced with the Illumina HiSeq 2500 platform.
Phylogenomic analyses
Single copy genes in arthropods were identified in our data sets with BUSCO v1.124 based on hidden Markov model profiles. The homologous genes detected were screened to identify multiple hits (i.e., paralogues). Only one homologue per BUSCO single copy gene was selected in each case, assuming that they were single copy in our samples as well, and therefore orthologs. The genes were parsed from each sample and combined into individual files (i.e., one file per gene) with custom python scripts. Alignment, trimming and concatenation were done as in Fernández et al.7. As the selection of outgroups may be critical in resolving myriapod relationships we constructed three matrices with different outgroup composition: matrix 1 (300 genes, 49,576 amino acids), including all outgroups (i.e., chelicerates, hexapods and crustaceans); matrix 2 (same as matrix 1, only with chelicerate outgroups); and matrix 3 (299 genes, 61,611 amino acids, only with pancrustacean outgroups). All matrices are provided as Suppl. Mat. In all cases, we selected a high level of gene occupancy to ensure the selection of a relatively large amount of genomic information while minimizing missing data and computational burden (75% gene occupancy in matrices 1 and 2 and 88% in matrix 3). Bayesian analyses were conducted with PhyloBayes MPI 1.7a25 selecting the site-heterogeneous CAT-GTR model of amino acid substitution26. Two independent Markov chain Monte Carlo (MCMC) chains were run for 5000–10,000 cycles. The initial 25% of trees sampled in each MCMC run prior to convergence (judged when maximum bipartition discrepancies across chains were <0.1) were discarded as burn-in. Convergence of chains was assessed both at the level of the bipartition frequencies (with the command bpcomp) and the summary variables displayed in the trace files (with the command tracecomp). We considered that convergence was achieved when (i) the maximum difference of the frequency of all the bipartitions observed in the chains was <0.1, and (ii) when the maximum discrepancy observed for each column of the trace file was <0.1 and the minimum effective size of 100. A 50% majority-rule consensus tree was then computed from the remaining trees. In order to further test for the effect of heterotachy and heterogeneous substitution rates, the matrices were also analysed in PhyML v.3.0.3 implementing the integrated length (IL) approach27,28. In this analysis, the starting tree was set to the optimal parsimony tree and the FreeRate model29 was selected. Congruence between the different topologies was visualized with DensiTree v2.2.530.
Molecular Dating
Divergence times for myriapods were estimated through molecular dating, constrained by the position of critical fossils using a morphological data set. Six Palaeozoic and Mesozoic myriapod fossils (three centipedes and three millipedes; Table 2) were included in our morphological data set of 187 characters. One fossil diplopod used for coding, Cowiedesmus eroticopodus, has since been redated as Early Devonian rather than mid Silurian31; another Silurian diplopod, Casiogrammus ichthyeros, replaces it as the earliest minimum age for crown-group Diplopoda and is coded as well.We also included three fossil outgroups: the crustacean Rehbachiella kinnekullensis, the scorpion Proscorpius osborni, and the collembolan Rhyniella praecursor. The matrix is available as Morphobank project P2762 (http://morphobank.org/permalink/P2762) and is provided as Supp. Mat. Multistate characters were scored as non-additive except for characters 57, 68, 81, 95 and 102, which were additive. The morphological data set was analysed under parsimony with TNT32. Traditional heuristic searches with 10,000 stepwise addition sequences resulted in 105 trees of 257 steps. Consistency Index 0.84, Retention Index 0.83 (Fig. 2e). No shorter trees were found using New Technology search strategies in TNT. Absolute dates follow the International Chronostratigraphic Chart v 2015/01. Justifications for age assignments of the fossils (Table 2) follow Wolfe et al.33. Divergence dates were estimated using the Bayesian relaxed molecular clock approach as implemented in PhyloBayes v.3.325. Both an auto-correlated and uncorrelated relaxed clock model were applied to our dataset. The calibration constraints specified above were used with soft bounds34 under a birth-death prior in PhyloBayes. Two independent MCMC chains were run for 5000–7,000 cycles, sampling posterior rates and dates every 10 cycles. The initial 25% were discarded as burn-in. Posterior estimates of divergence dates were then computed from the remaining samples of each chain.
References
Rota-Stabelli, O. et al. A congruent solution to arthropod phylogeny: phylogenomics, microRNAs and morphology support monophyletic Mandibulata. Proc. R. Soc. B. Biol. Sci. 278, 298–306 (2011).
Rota-Stabelli, O., Daley, A. C. & Pisani, D. Molecular timetrees reveal a Cambrian colonization of land and a new scenario for ecdysozoan evolution. Curr. Biol. 23, 392–398 (2013).
Lozano-Fernandez, J. et al. Molecular palaeobiological exploration of arthropod terrestrialisation. Philos. Trans. Roy. Soc. B. Biol. Sci. 371, 20150133 (2016).
Misof, B. et al. Phylogenomics resolves the timing and pattern of insect evolution. Science 346, 763–767 (2014).
Schwentner, M., Combosch, D. J., Nelson, J. P. & Giribet, G. A phylogenomic solution to the origin of insects by resolving crustacean-hexapod relationships. Curr. Biol. 27, 1818–1824 (2017).
Brewer, M. S. & Bond, J. E. Ordinal-level phylogenomics of the arthropod class Diplopoda (millipedes) based on an analysis of 221 nuclear protein-coding loci generated using next-generation sequence analyses. PLoS One 8, e79935 (2013).
Fernández, R., Edgecombe, G. D. & Giribet, G. Exploring phylogenetic relationships within Myriapoda and the effects of matrix composition and occupancy on phylogenomic reconstruction. Syst. Biol. 65, 871–889 (2016).
Fernández, R. et al. Evaluating topological conflict in centipede phylogeny using transcriptomic data sets. Mol. Biol. Evol. 31, 1500–1513 (2014).
Regier, J. C. et al. Arthropod relationships revealed by phylogenomic analysis of nuclear protein-coding sequences. Nature 463, 1079–1083 (2010).
Regier, J. C. & Zwick, A. Sources of signal in 62 protein-coding nuclear genes for higher-level phylogenetics of arthropods. PLoS One 6, e23408 (2011).
Zwick, A., Regier, J. C. & Zwickl, D. J. Resolving discrepancy between nucleotides and amino acids in deep-level arthropod phylogenomics: Differentiating serine codons in 21-amino-acid models. PLoS One 7, e47450 (2012).
Gai, Y.-H., Song, D.-X., Sun, H.-Y. & Zhou, K.-Y. Myriapod monophyly and relationships among myriapod classes based on nearly complete 28S and 18S rDNA sequences. Zool. Sci. 23, 1101–1108 (2006).
Dong, Y. et al. The complete mitochondrial genome of Pauropus longiramus (Myriapoda: Pauropoda): Implications on early diversification of the myriapods revealed from comparative analysis. Gene 505, 57–65 (2012).
Boudreaux, H. B. Phylogeny of the arthropod classes. In Arthropod phylogeny, with special reference to insects, pp. 82–122. Malabar, Florida: Robert E. Krieger Publishing Company (1987).
Pocock, R. I. On the classification of the tracheate Arthropoda. Zool. Anz. 16, 271–275 (1983).
Tiegs, O. W. The development and affinities of the Pauropoda, based on a study of Pauropus silvaticus. Quarterly. Journal of Microscopical Sciences 88, 275–336 (1947).
Rehm, P., Meusemann, K., Borner, J., Misof, B. & Burmester, T. Phylogenetic position of Myriapoda revealed by 454 transcriptome sequencing. Mol. Phylogenet. Evol. 77, 25–33 (2014).
Miyazawa, H., Ueda, C., Yahata, K. & Su, Z. H. Molecular phylogeny of Myriapoda provides insights into evolutionary patterns of the mode in post-embryonic development. Sci. Rep. 4, 4127 (2014).
Johnson, E. W., Briggs, D. E. G., Suthren, R. J., Wright, J. L. & Tunnicliff, S. P. Non-marine arthropod traces from the subaerial Ordovician Borrowdale Volcanic Group, English Lake District. Geological Magazine 131, 395–406 (2014).
Wilson, H. M. Juliformian millipedes from the Lower Devonian of Euramerica: implications for the timing of millipede cladogenesis in the Paleozoic. J. Paleontol. 80, 638–649 (2006).
MacNaughton, R. B. et al. First steps on land: Arthropod trackways in Cambrian-Ordovician Eolian sandstone, southeastern Ontario, Canada. Geology 30, 391–394 (2002).
Collette, J. H., Gass, K. C. & Hagadorn, J. W. Protichnites eremita unshelled? Experimental model-based neoichnology and new evidence for a euthycarcinoid affinity for this ichnospecies. J. Paleontol. 86, 442–454 (2012).
Blanke, A. & Wesener, T. Revival of forgotten characters and modern imaging techniques help to produce a robust phylogeny of the Diplopoda (Arthropoda, Myriapoda). Arthropod Struct. Dev. 43, 63–75 (2014).
Simão, F. A., Waterhouse, R. M., Ioannidis, P., Kriventseva, E. V. & Zdobnov, E. M. BUSCO: assessing genome assembly and annotation completeness with single-copy orthologs. Bioinformatics 31, 3210–3212 (2015).
Lartillot, N., Rodrigue, N., Stubbs, D. & Richer, J. PhyloBayes MPI: Phylogenetic reconstruction with infinite mixtures of profiles in a parallel environment. Syst. Biol. 62, 611–615 (2013).
Lartillot, N. & Philippe, H. A Bayesian mixture model for across-site heterogeneities in the amino-acid replacement process. Mol. Biol. Evol. 21, 1095–1109 (2004).
Guindon, S. & Gascuel, O. A simple, fast, and accurate algorithm to estimate large phylogenies by maximum likelihood. Syst. Biol. 52, 696–704 (2003).
Guindon, S. From trajectories to averages: an improved description of the heterogeneity of substitution rates along lineages. Syst. Biol. 62, 22–34 (2013).
Soubrier, J. et al. The influence of rate heterogeneity among sites on the time dependence of molecular rates. Mol. Biol. Evol. 29, 3345–3358 (2012).
Bouckaert, R. R. & Heled, J. DensiTree 2: Seeing trees through the forest. bioRxiv (2014)
Suarez, S. E., Brookfield, M. E., Catlos, E. J. & Stöckli, D. F. A U-Pb zircon age constraint on the oldest-recorded air-breathing land animal. PLoS One 12, e0179262 (2017).
Goloboff, P. A. & Catalano, S. A. TNT version 1.5, including a full implementation of phylogenetic morphometrics. Cladistics 32, 221–238 (2016).
Wolfe, J. M., Daley, A. C., Legg, D. A. & Edgecombe, G. D. Fossil calibrations for the arthropod Tree of Life. Earth-Sci. Rev. 160, 43–110 (2016).
Yang, Z. H. & Rannala, B. Bayesian estimation of species divergence times under a molecular clock using multiple fossil calibrations with soft bounds. Mol. Biol. Evol. 23, 212–226 (2006).
Chipman, A. D. et al. The first myriapod genome sequence reveals conservative arthropod gene content and genome organisation in the centipede Strigamia maritima. PLoS Biol. 12, e1002005 (2014).
Stoev, P. et al. Eupolybothrus cavernicolus Komerički & Stoev sp. n. (Chilopoda: Lithobiomorpha: Lithobiidae): the first eukaryotic species description combining transcriptomic. DNA barcoding and micro-CT imaging data. Biodiversity data journal 1, e1013 (2013).
Sharma, P. P. et al. Phylogenomic interrogation of Arachnida reveals systemic conflicts in phylogenetic signal. Mol. Biol. Evol. 31, 2963–2984 (2014).
Faddeeva-Vakhrusheva, A. et al. Coping with living in the soil: the genome of the parthenogenetic springtail Folsomia candida. BMC Genomics 18, 493 (2017).
Acknowledgements
Miquel Arnedo, Ligia Benavides, Brian Colby and Julia Cosgrove assisted with pauropod collection. This study was mainly supported by internal funds from the Museum of Comparative Zoology, Harvard University and by NSF grant DEB-1457539 to GG, which funded RF. Three reviewers provided comments that help to clarify some aspects of this article.
Author information
Authors and Affiliations
Contributions
All authors designed the study. R.F. and G.G. collected the samples. R.F. executed molecular work and data analysis. G.D.E. coded the morphological data. All authors wrote the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Fernández, R., Edgecombe, G.D. & Giribet, G. Phylogenomics illuminates the backbone of the Myriapoda Tree of Life and reconciles morphological and molecular phylogenies. Sci Rep 8, 83 (2018). https://doi.org/10.1038/s41598-017-18562-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-18562-w
This article is cited by
-
Alkaloid chemistry in pill-millipedes: Defensive secretion in two species of Typhloglomeris Verhoeff, 1898 (Diplopoda, Glomerida, Glomeridellidae)
Chemoecology (2024)
-
De novo metatranscriptomic exploration of gene function in the millipede holobiont
Scientific Reports (2022)
-
Molecular phylogenies map to biogeography better than morphological ones
Communications Biology (2022)
-
Mimopidae is the sister group to all other scolopendromorph centipedes (Chilopoda, Scolopendromorpha): a phylotranscriptomic approach
Organisms Diversity & Evolution (2021)
-
Four myriapod relatives – but who are sisters? No end to debates on relationships among the four major myriapod subgroups
BMC Evolutionary Biology (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.