Abstract
Mosquito surveillance and control are at the heart of efforts to eliminate malaria, however, there remain significant gaps in our understanding of mosquito behaviour that impede innovation. We hypothesised that a combination of human-associated stimuli could be used to attract and kill malaria vectors more successfully than individual stimuli, and at least as well as a real human. To test this in the field, we quantified Anopheles responses to olfactory, visual and thermal stimuli in Burkina Faso using a simple adhesive trap. Traps baited with human odour plus high contrast visual stimuli caught more Anopheles than traps with odour alone, showing that despite their nocturnal habit, malaria vectors make use of visual cues in host-seeking. The best performing traps, however, combined odour and visual stimuli with a thermal signature in the range equivalent to human body temperature. When tested against a human landing catch during peak mosquito abundance, this “host decoy” trap caught nearly ten times the number of Anopheles mosquitoes caught by a human collector. Exploiting the behavioural responses of mosquitoes to the entire suite of host stimuli promises to revolutionise vector surveillance and provide new paradigms in disease control.
Similar content being viewed by others
Introduction
Malaria vector control through long-lasting insecticide-treated bed nets (LLINs) and indoor residual spraying (IRS) has reduced malaria case incidence by 41% in the last 15 years1. The incredible success of these technologically simple interventions is based upon our knowledge of the primary African malaria vectors’ indoor feeding and resting behaviours. Yet, beyond these indoor-based interventions, only a limited range of quantified mosquito behaviours are exploited in mosquito surveillance and control methods.
Mosquito vector behaviour research has often been reductive and focussed too narrowly on responses to host odours, specifically their activation by and attraction towards sources of carbon dioxide and human-derived volatile odours2. For a natural host, the olfactory stimuli that mediate attraction are not produced in isolation, but rather with other host-associated stimuli that influence behaviour, particularly in the short-range attraction and landing responses that precede blood-feeding3. Recent laboratory studies have shown that both visual4 and thermal5 stimuli enhance close-range orientation and landing by Anopheles gambiae complex mosquitoes when these are combined with olfactory stimuli from a human. Exploitation of these non-chemical stimuli in vector monitoring and control has been neglected, impeding our ability to develop new evidence-based means of sampling and killing host-seeking mosquitoes.
This gap in our knowledge of mosquito behaviour inhibits disease control innovation and is particularly worrying as there is growing concern that we may be reaching the limits of indoor-based control measures6. Sixty malaria-endemic countries report insecticide resistance in at least one primary vector species1 and behavioural changes following the introduction of indoor control methods appear to be increasing the proportion of biting that occurs outdoors and before people go to sleep under their bed nets7,8,9 Furthermore, indoor control measures do not affect other important malaria vectors that bite and shelter predominantly outdoors10,11. Quantifying and subsequently reducing this outdoor malaria transmission is critical for eventual disease elimination.
Devising vector research and intervention tools that can be used outdoors is, therefore, of paramount importance if the momentum against malaria mosquitoes is to be maintained12,13. Where current sampling and control measures are particularly effective they are based on intercepting mosquitoes in the course of their natural blood-seeking behaviour. For example, the Human Landing Catch (HLC) is commonly considered to be the ‘gold standard’ method for sampling human-biting vectors14 as mosquitoes are caught once they orient towards and land on a real human host. New outdoor approaches underpinned by a comprehensive integration of the stimuli that are known to result in specific vector behaviours that lead to outdoor feeding are likely to be the most successful15,16. Accordingly, the first step toward monitoring and ultimately controlling outdoor-biting mosquitoes is to identify the sensory cues that mosquitoes use to locate a human host outdoors and quantify the relative contributions of these stimuli to host-seeking.
This study is the first to devise and test a mosquito trap that exploits the full range of visual, olfactory and thermal stimuli involved in the natural host-seeking behaviour of mosquitoes. The trap design allows these stimuli to be presented singly or in combination to determine the relative importance of each in enhancing or supressing the overall catch. Our aims were to (i) establish whether laboratory-observed synergies between visual, thermal and olfactory stimuli also occur in outdoor-biting populations of An. gambiae complex mosquitoes, (ii) determine if high-contrast visual and thermal stimuli, simulating a warm human body, can increase the catch of host-seeking mosquitoes when combined with olfactory stimuli, and (iii) evaluate whether a host “decoy trap”, incorporating these three stimuli in imitation of a host, could intercept wild mosquitoes outdoors at least as well as a real human bait.
Methods
Study setting and experimental design
All experiments were conducted in Vallée de Kou in south-west Burkina Faso (11° 41′ N, 04° 44′ W), where the predominant malaria vector is An. coluzzii, found widely throughout xeric West African savannas17. In a series of three experiments, we compared the catches from three designs of human odour-baited decoy traps that incorporated visual and/or heat stimuli and the design with the highest catch was then tested against the standard HLC14. All collections were made outdoors using a Latin square experimental design of traps × sites × nights. Traps were placed at least 50 m apart to limit competition between them. Collections were made between 20:00 h and 06:00 h in Experiment 1, and between 18:00 h and 06:00 h in Experiments 2 and 3. The study protocol was approved by the Comité d’Ethique Institutionnel pour la Recherche en Sciences de la Santé: A014-2014-2014/CE-CM and A17-2016/CEIRES. All experiments were performed in accordance with relevant guidelines and regulations, and informed consent was obtained from all field collectors involved in HLC.
Experiment 1: Effect of visual stimuli
We aimed to determine first whether the addition of visual stimuli enhanced mosquito attraction to a source of host odour, and second if mosquitoes could be diverted to land on the visual stimuli. To test this, two types of adhesive trap were constructed, differing in their contrast with the shade and pattern of the surrounding soil and vegetation. The low contrast trap was constructed from a cylindrical metal wire frame (45 cm high, 38 cm diameter) wrapped in Fics Film (Barrettine, UK), a commercially available insect adhesive trap material. Fics Film is a colourless, transparent plastic sheet coated with an adhesive and thus produced a near-transparent trap when wrapped around the cylindrical wire frame. The high contrast trap was identical, but with the addition of a sheet of matte black card (240 gsm), which was inserted behind the adhesive sheet to create a solid block of high visual contrast and uniform pattern. Mosquitoes were caught as they landed on the adhesive surface of the traps.
Both traps were tested either with or without odour from a single person sleeping protected in a tent. This odour was drawn from the tent by a fan (0.38 amp, 12 V, maximum airflow ~ 2000L/min) powered by a 6 V battery, along a tube (10 m length, 25 cm diameter) and vented approximately 10–15 cm from the base of the traps. Experiments were conducted over 6 nights (October 2011). The high contrast trap baited with host odour caught the most mosquitoes (Fig. 1a).
Mean nightly catches, ± s.e.m., of An. gambiae during development and evaluation of behaviour-based trap. (a) Experiment 1: Effect of high visual contrast compared to low visual contrast (n = 6). (b) Experiment 2: Effect of heating high visual contrast trap to 35 ± 5 °C versus ambient temperature (n = 9). (c) Performance of high visual contrast heated trap, the “Host Decoy Trap”, compared to Human Landing Catch method (n = 17). All traps baited with whole human odour. ***Indicate difference significant at α < 0.001.
Experiment 2: Effect of thermal stimuli
Thermal stimuli have been shown experimentally to increase close-range flight activity and landing behaviour in laboratory colonies of An. gambiae complex mosquitoes5. We hypothesised that the addition of thermal stimuli to the adhesive surface of the high contrast trap used in Experiment 1 would increase landing rates and thus overall catch. Therefore, the high contrast trap was modified by heating the trap to a relatively constant surface temperature of 35 ± 5 °C for the duration of nightly collections, simulating the surface temperature of a human body and matching the temperature range in which odour-stimulated An. gambiae complex mosquitoes land18. This was achieved by replacing the trap’s wire frame with a cylindrical metal container (40 cm high, 46 cm diameter) in which 30 l water could be contained and heated using a 1000 W immersion element. The surface temperature was measured periodically with an infrared spot thermometer. The container was wrapped in plain black nylon fabric to maintain high visual contrast. This heated trap was tested against an identical un-heated (i.e. ambient temperature) version to determine what effect, if any, the addition of thermal stimuli had on overall mosquito catch. Experiments were conducted over nine nights (May-June 2015). The heated trap baited with host odour caught the most mosquitoes (Fig. 1b).
Experiment 3: Comparison of Host Decoy Trap versus Human Landing Catch
We proceeded to test the trap model with the highest catch out of those tested in Experiments 1 and 2 (the heated, high contrast odour-baited trap) against a standard outdoor HLC. We called this device the Host Decoy Trap (HDT), as it included the three stimuli most associated with human hosts: olfactory, visual and thermal (Fig. 2). We aimed to establish whether the HDT’s combination of host-associated stimuli would be sufficient to catch the same number of mosquitoes as landed on a real human presenting these same stimuli naturally. The two collection methods were compared across three seasonal periods to investigate the performance of each during different mosquito population densities: in the rainy season (July 2015, 17 trapping nights; Fig. 1c), in the early dry season (November 2015, 14 trapping nights) and in the late dry season (May to June 2015, 9 trapping nights).
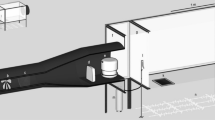
The Host Decoy Trap (a) Field assistants provide human odour while sleeping protected in a tent overnight. Fan draws air from tent along pipe, releasing odour ~10 cm from the base of the trap. Netting covers end of pipe to prevent mosquitoes entering tent. Trap kept at human body temperature (35 ± 5 °C). Un-patterned dark cloth covers trap to increase visual contrast. Clear adhesive plastic sheet wraps around trap to catch landing mosquitoes. Tent is 10 m from trap to focus mosquito search for visual host cues near trap rather than the tent. (b) Schematic, showing flow of air containing whole human odour (blue dashed line) from tent to surrounding Host Decoy Trap.
Species identification
Results presented refer to female mosquitoes, which were identified to genus. Anopheline mosquitoes were removed using Romax Glue Solvent (Barrettine, UK) and identified by morphology to species/species complex level19, with the exception of catches collected during the rainy season in Experiment 3, which were exceptionally large (>500/night). In this case, all anophelines from six complete collections of both Host Decoy Trap (9,000 out of 28,875 anophelines) and Human Landing Catch (1,020 out of 2,987 anophelines), equal to 32% of total anopheline catch in July, were identified morphologically and proportions calculated to estimate the overall number of each species (An. gambiae sensu lato and An. pharoensis). All specimens collected from the HLC samples and >99% of those from HDT could be identified morphologically to species. A subset of 419 An. gambiae s.l. from all seasons and both trap types were identified to species level by PCR20; 99.5% were found to be An. coluzzii, with only one An. arabiensis and one An. gambiae sensu stricto in Host Decoy Trap catches from collections in the late dry season.
Statistical analysis
All analyses were performed in R version 3.3.221. Generalized linear models (GLM; R package MASS 22), with trap set as a predictor and a negative binomial error structure and log link, were used to determine whether differences in mosquito catch size between trapping methods were significant. Differences in abundance between trapping methods were determined with Tukey contrasts (R package multcomp 23).
Results
Experiment 1: Effect of visual stimuli
Results from this experiment challenge the assumption that visual cues are not important for nocturnally-active mosquitoes24, as we found significantly more An. gambiae landed on the odour-baited adhesive trap with high visual contrast compared to the low contrast trap (GLM; χ2 = 15.6; P = 0.013; Fig. 1a). Furthermore, Culex and Mansonia mosquitoes were also caught in significantly higher numbers on the visually-conspicuous high contrast trap (Table 1), and other vectors, An. pharoensis and An. coustani, were also caught in consistently but not significantly greater numbers. This consistent trend suggests that visual cues are used by nocturnally-active mosquitoes across a range of genera to locate potential sources of host blood.
In the absence of human odour, no An. gambiae were caught on either trap type. No other mosquitoes were found on the unbaited high contrast trap and only one culicine mosquito was caught on the unbaited low contrast trap. This result confirms numerous field and laboratory studies which indicate that olfactory stimuli are essential components of attraction for host-seeking An. gambiae and other hematophagous mosquitoes3. Without odour, mosquitoes did not readily land on any trap, confirming that human odour is an essential element of an attractive trap. However, it also demonstrates that although the insects are nocturnally active and must contend with low light levels, wild host-seeking An. gambiae orient towards high contrast visual stimuli, but only when stimulated by host-associated odour. Visual and olfactory stimuli thus act synergistically to attract mosquitoes. This accords with laboratory findings that An. coluzzii orient towards visually conspicuous targets, but only when concurrently exposed to whole human odour4.
Experiment 2: Effect of thermal stimuli
We proceeded with the high contrast trap to test the effect of another cue associated with warm-blooded vertebrates: heat. The trap combining olfactory and visual stimuli with heat caught significantly more An. gambiae than the trap with only olfactory and visual stimuli (GLM; z = 4.7, P = 0.3 × 10−7; Fig. 1b), corresponding to a 6.5 fold increase in total catch caused by warming the trap to body temperature (Table 2). Heated traps also caught significantly more of both An. pharoensis (GLM; z = 3.0, P = 0.0144) and Culex mosquitoes (GLM; z = 4.3, P = 0.5 × 10−7), and whilst not significantly higher (GLM; z = 2.6, P = 0.1), over three times the number of Mansonia mosquitoes were also caught on heated traps. This combination of olfactory, visual and thermal stimuli resulted in the best trap – the Host Decoy Trap (HDT) – for collecting a range of outdoor-biting mosquitoes, and, crucially, malaria vectors.
Experiment 3: Comparison of Host Decoy Trap versus Human Landing Catch
Having shown that outdoor-biting mosquitoes from a range of genera could be attracted towards and induced to land upon an adhesive trapping device in the greatest numbers by combining three known host-associated stimuli, we then tested the HDT against the “gold standard” collection method of HLC, thereby comparing the trapping performance of a real and imitation human host. Table 3 provides a summary of all mosquitoes caught across the three seasons during which this experiment was conducted. Over 40 trapping nights, a total of 26,357 An. gambiae were collected outdoor by HDTs, compared to 3,083 by outdoor HLCs (Table 3). In collections made in the rainy season during the peak of the An. gambiae population, HDTs caught a nightly average of 1,498.2 An. gambiae, compared to 156.8 by HLC (Fig. 1c; GLM; z = 14.0, P < 2 × 10−16). Overall, the HDT caught nearly 10 × more An. gambiae than HLC. The catch of An. pharoensis (~10 × ), Culex (~5 × ) and Mansonia (~5.4 × ) were also significantly greater in HDTs than HLCs (Table 3).
Due to the exponential changes in mosquito density with season, statistical analyses were conducted on log data (Fig. 3). During the early dry seasons, the HDT continued to catch consistently more Anopheline, Culex and Mansonia species than HLC, with total catches varying between 2.2 and 3.5 times greater, depending on the genera. Mean catches of the vectors An. gambiae, An. pharoensis and An. coustani, which was only present during early dry season catches, and Culex and Mansonia species were all significantly higher in the HDT than in the HLC. In the late dry season, when all mosquito populations are typically at their lowest due to the paucity of available breeding sites, there was no significant difference between HDT and HLC catches of all mosquito species and genera, although the HDT continued to catch consistently more mosquitoes than the HLC.
Mean ln catch per night, ± s.e.m., of mosquitoes from Host Decoy Trap (HDT) and Human Landing Catch (HLC) across three seasons. The two methods were tested during the rainy (n = 17), early dry (n = 14) and late dry (n = 9) seasons. Significance levels from negative binomial GLM. ***Indicate difference significant at α < 0.001; NS are statistically non-significant differences.
Discussion
We show though sequential behavioural studies how the natural host-seeking behaviour of mosquitoes can be attributed to specific stimuli and ultimately exploited to trap and kill malaria vectors. By systematically testing mosquito landing behaviour in response to artificial versions of human features we demonstrate the synergistic relationship between olfactory, visual and thermal stimuli in mediating vector-host contact. Landing by vector species was significantly increased when all three of these stimuli were presented together, as they presumably share the attributes of a potential blood-meal source. During the periods of greatest abundance of An. gambiae, nearly ten-fold more were caught by the HDT than the HLC, in spite of being baited with only three simplified versions of human-host stimuli. Clearly, these behavioural cues have not yet been fully exploited as features in vector surveillance and control approaches.
New paradigms in this field must be informed by a comprehensive understanding of vector behaviour in the field. Mosquito attraction to carbon dioxide and volatile skin odours from host animals is the basis for most monitoring traps, employing either whole human odours25,26, or synthetic blends27,28. We substantially improved total catches for both anopheline and culicine mosquitoes in odour-baited traps by introducing additional visual stimuli, corroborating laboratory work showing that An. gambiae are responsive to high contrast objects, even in low light levels equivalent to starlight (~0.001 W m−2 4,29. Nocturnal mosquitoes, including An. gambiae, have evolved morphological and neural modifications to typical Dipteran eyes that maximise their light-gathering potential. Their hemispheric lenses and conical-shaped fused rhabdoms more than double the angle over which each ommatidium captures light compared to diurnally-active mosquito species, making them extremely sensitive to light, albeit at the expense of resolution30. It seems likely that, as with tsetse fly species31, the size and/or orientation of visual targets may also affect mosquito orientation and landing behaviours. Development of visually optimized vector research methods should be prioritised, as simple changes to the appearance of existing tools could increase their visibility and attractiveness to target mosquito species.
Olfactory and visual cues are thought to operate at distances many metres from the host32. However, subsequent close-range orientation and landing behaviours are likely to be mediated by other host stimuli, such as heat and convection currents, that are detected within 1 m of the host33. The anopheline proboscis and antenna have been shown to detect thermal stimuli and mediate orientation towards and contact with heat sources over a range of <0.5 m34, and increased landing rates have been observed on heated versus unheated pads5. In the field, we found that landing was significantly increased on thermally baited traps, even though the temperature of the HDT was only a few degrees warmer than typical nightly temperatures. As a close-range attractant, the careful placement of warm components could, therefore, play a crucial role in trap design by spatially concentrating mosquitoes onto or within close proximity to the collecting or killing element of the device.
In our comparison of the HDT against the standard HLC, where mosquitoes are caught as they land and attempt to feed on the bare legs of a real human, we found that the catches from the HDT always exceeded those from HLC, irrespective of season or mosquito genera. The HLC is intended to directly measure vector-host contact, but it exposes collectors to mosquito-borne disease and results can vary according to the skill and alertness of the collector35. To overcome these constraints, methods are needed that mimic the sensory cues that attract mosquitoes to humans and sample those mosquitoes in a standardised way. This is particularly important where the feeding behaviour of outdoor-biting vectors may be contributing to malaria transmission risk. Recent efforts to sample An. gambiae outdoors using a volunteer’s foot inside a box of charged wires gave promising results26, although mosquitoes electrocuted on approach to a host may not necessarily have ultimately landed and fed. Alternative methods of outdoor sampling use tents or netting and tend to rely on mosquitoes exhibiting some form of entry behaviour36,37, and may not therefore sample mosquitoes engaging in outdoor-biting behaviour. Cues from the HDT, on the other hand, stimulate the final stages of host-seeking behaviour and capture mosquitoes as they land on the trap and so may reflect a more realistic representation of vector-host contact outdoors.
In the present study we only analysed responses to human-baited traps. The HDT could be adapted to use alternative host animals as odour sources, allowing direct comparisons to be made in the host-biting preferences of zoophilic malaria vectors, such as An. arabiensis, that have been shown to feed on both humans and cattle38,39. Such studies would also be valuable in the analysis of the zoonotic malaria, Plasmodium knowlesi, which has a wildlife reservoir in long- and pig-tailed macaque monkeys40, as well as zoonoses caused by mosquito-borne arboviruses, including Japanese encephalitis virus, where pigs are amplifying hosts41.
It is assumed that an HLC conducted properly provides the most reliable estimate of the human biting rate. Yet, in the present study, the HDT caught nearly ten times more An. gambiae than a human. The larger-than-expected numbers of malaria vector mosquitoes has several important implications. First, it is possible that when mosquito populations are at their highest even well-trained and experienced technicians may be unable to keep up with collecting the overwhelming number of mosquitoes alighting on them, especially on areas other than the legs, which are not routinely scanned for alighting mosquitoes. This would fit with our observations of some HLC collections, and implies that HLCs may underestimate human-biting rates35. An alternative explanation is that the simplified and concentrated artificial versions of host-associated characteristics presented in the HDT could act as a supernormal stimulus, where exaggerated host cues attract more mosquitoes than a real human. The HDT could therefore help to provide vector bionomics data where existing methods of mosquito surveillance may not be sensitive enough to detect very low density outdoor biting vector populations.
Given the large numbers of An. gambiae and other mosquito species caught by the HDT, the basic principles used in its design could also be modified to create a vector control tool. The largest gap in the vector control portfolio is a scalable intervention that can target outdoor biting mosquito populations42. We have demonstrated that a trap with a combination of behaviourally relevant stimuli can attract and kill a wide range of mosquitoes, including the primary malaria vector An. gambiae, outdoors. By optimising the principles used in the HDT and improving our understanding of outdoor biting behaviour, there is potential to develop a means of delivering lethal doses of insecticides or other killing agents on targets designed to lure and kill mosquitoes. This approach has worked exceedingly well for control of tsetse fly; clinical incidence of tsetse-borne human trypanosomiasis was reduced by ten-fold following the introduction of attractive insecticidal “tiny-targets” in Guinea43. A similar approach could provide a complement to interventions that seek to control anopheline malaria vectors outside the home.
References
Bhatt, S. et al. The effect of malaria control on Plasmodium falciparum in Africa between 2000 and 2015. Nature. 526, 207–211 (2015).
Zwiebel, L. J. & Takken, W. Olfactory regulation of mosquito-host interactions. Insect Biochem. Mol. Biol. 34, 645–652 (2004).
Gibson, G. & Torr, S. J. Visual and olfactory responses of haematophagous Diptera to host stimuli. Med. Vet. Entomol. 13, 2–23 (1999).
Hawkes, F. & Gibson, G. Seeing is believing: the nocturnal malarial mosquito Anopheles coluzzii responds to visual host-cues when odour indicates a host is nearby. Parasit. Vectors. 9, 320 (2016).
Spitzen, J. et al. A 3D analysis of flight behaviour of Anopheles gambiae sensu stricto malaria mosquitoes in response to human odour and heat. PLoS One. 8, 5 (2013).
Sokhna, C., Ndiath, M. O. & Rogier, C. The changes in mosquito vector behaviour and the emerging resistance to insecticides will challenge the decline of malaria. Clin. Microbiol. Infect. 19, 902–907 (2013).
Reddy, M. R. et al. Outdoor host seeking behaviour of Anopheles gambiae mosquitoes following initiation of malaria vector control on Bioko Island, Equatorial Guinea. Malar. J. 10, 184 (2011).
Russell, T. L. et al. Increased proportions of outdoor feeding among residual malaria vector populations following increased use of insecticide-treated nets in rural Tanzania. Malar. J. 10, 80 (2011).
Moiroux, N. et al. Changes in Anopheles funestus biting behaviour following universal coverage of long-lasting insecticidal nets in Benin. J. Infect. Dis. 206, 1622–1629 (2012).
McCann, R. S. et al. Reemergence of Anopheles funestus as a vector of Plasmodium falciparum in western Kenya after long-term implementation of insecticide-treated bed nets. Am. J. Trop. Med. Hyg. 90, 597–604 (2014).
Kenea, O. et al. Human-biting activities of Anopheles species in south-central Ethiopia. Parasit. Vectors. 9, 527 (2016).
Gatton, M. L. et al. The importance of mosquito behavioural adaptations to malaria control in Africa. Evolution. 67, 1218–1230 (2013).
Russell, T. L., Beebe, N. W., Cooper, R. D., Lobo, N. F. & Burkot, T. R. Successful malaria elimination strategies require interventions that target changing vector behaviours. Malar. J. 12, 56 (2013).
World Health Organization. WHO Manual On Practical Entomology In Malaria Part II: Methods And Techniques (World Health Organization, 1975).
Killeen, G. F. et al. Measuring, manipulating and exploiting behaviours of adult mosquitoes to optimise malaria vector control impact. BMJ Glob. Health. 2, e000212 (2017).
Torr, S. J. & Vale, G. A. Know your foe: lessons from the analysis of tsetse fly behaviour. Trends Parasitol. 31, 95–99 (2015).
Fossog, B. T. et al. Habitat segregation and ecological character displacement in cryptic African malaria mosquitoes. Evol. Appl. 8, 326–345 (2015).
Healy, T. P. & Copland, M. J. W. Activation of Anopheles gambiae mosquitoes by carbon dioxide and human breath. Med. Vet. Entomol. 9, 331–6 (1995).
Gillies, M.T. & de Meillon, B. The Anophelinae Of Africa South Of The Sahara (Ethiopian Zoogeographical Region). Publications of the South African Institute for Medical Research, no. 54 (1968).
Santolamazza, F. et al. Insertion polymorphisms of SINE200 retrotransposons within speciation islands of Anopheles gambiae molecular forms. Malar. J. 7, 163 (2008).
R Core Team. R: A language and environment for statistical computing (Vienna, Austria, 2015).
Venables, W. N. & Ripley, B. D. Modern Applied Statistics With S, Fourth Edition. (Springer, 2002).
Torsten, H., Bretz, F. & Westfall, P. Simultaneous inferences in general parametric models. Biom. J. 50, 346–63 (2008).
Gillies, M. T. & Wilkes, T. J. Responses of host-seeking Mansonia and Anopheles mosquitoes (Diptera: Culicidae) in West Africa to visual features of a target. J. Med. Entomol. 19, 68–71 (1982).
Mathenge, E. M. et al. Comparative performance of the Mbita trap, CDC light trap and the human landing catch in the sampling of Anopheles arabiensis, An. funestus and culicine species in a rice irrigation in western Kenya. Malar. J. 4, 7 (2005).
Govella, N. J. et al. An improved mosquito electrocuting trap that safely reproduces epidemiologically relevant metrics of mosquito human-feeding behaviours as determined by human landing catch. Malar. J. 15, 465 (2016).
Okumu, F. O., Madumla, E. P., John, A. N., Lwetoijera, D. W. & Sumaye, R. D. Attracting, trapping and killing disease-transmitting mosquitoes using odor-baited stations – the Ifakara Odor-Baited Stations. Parasit. Vectors. 3, 12 (2010).
Hiscox, A. et al. Development and optimisation of the Suna trap as a tool for mosquito monitoring and control. Malar. J. 13, 257 (2014).
Gibson, G. A behavioural test of the sensitivity of a nocturnal mosquito, Anopheles gambiae, to dim white, red and infra-red light. Physiol. Entomol. 20, 224–228 (1995).
Land, M. F., Gibson, G. & Horwood, J. Mosquito eye design: conical rhabdoms are matched to wide aperture lenses. Proc. Roy. Soc. B. 264, 1183–1187 (1997).
Vale, G. A. Visual responses of tsetse flies (Diptera: Glossinidae) to odour-baited targets. Bull. Entomol. Res. 83, 277–289 (1993).
Gillies, M. T. & Wilkes, T. J. The range of attraction of single baits for some West African mosquitoes. Bull. Entomol. Res. 60, 225–235 (1970).
Cardé, R.T. & Gibson, G. Host finding by female mosquitoes: Mechanisms of orientation to host odours and other cues in Olfaction in Vector-Host Interactions: Ecology and Control of Vector-Borne Diseases Volume 2 (eds Takken, W. & Knols, B.J.G.) 115–142 (Wageningen Academic Publishers, 2010).
Maekawa, E. et al. The role of proboscis of the malaria vector Anopheles stephensi in host-seeking behaviour. Parasit. Vectors. 4, 10 (2011).
Achee, N. L., Youngblood, L., Bangs, M. J., Lavery, J. V. & James, S. Considerations for the use of human participants in vector biology research: a tool for investigators and regulators. Vector Borne Zoonotic Dis. 15, 89–102 (2015).
Wong, J. et al. Standardizing operational vector sampling techniques for measuring malaria transmission intensity: evaluation of six mosquito collection methods in western Kenya. Malar. J. 12, 143 (2013).
Tangena, J. A. A., Thammavong, P., Hiscox, A., Lindsay, S. W. & Brey, P. T. The Human-Baited Double Net Trap: An alternative to human landing catches for collecting outdoor biting mosquitoes in Lao PDR. PLoS ONE. 10, 9 (2015).
Torr, S. J., Della Torre, A., Calzetta, M., Costantini, C. & Vale, G. A. Towards a fuller understanding of mosquito behaviour: use of electrocuting grids to compare the odour-orientated responses of Anopheles arabiensis and An. quadriannulatus in the field. Med. Vet. Entomol. 22, 93–108 (2008).
James, S., Takken, W., Collins, F. H. & Gottlieb, M. Needs for monitoring mosquito transmission of malaria in a pre-elimination world. Am. J. Trop. Med. Hyg. 90, 6–10 (2014).
Hawkes, F. et al. Evaluation of electric nets as means to sample mosquito vectors host-seeking on humans and primates. Parasit. Vectors. 10, 338 (2017).
Lindahl, J. F. et al. Circulation of Japanese Encephalitis Virus in pigs and mosquito vectors within Can Tho City, Vietnam. PLoS Negl. Trop. Dis. 7(4), e2153 (2013).
Hemingway, J. et al. Tools and strategies for malaria control and elimination: what do we need to achieve a grand convergence in malaria? PLoS Biol. 14(3), e1002380 (2016).
Courtin, F. et al. Reducing human-tsetse contact significantly enhances the efficacy of sleeping sickness active screening campaigns: a promising result in the context of elimination. PLoS Negl. Trop. Dis. 9(8), e0003727 (2015).
Acknowledgements
This study was supported by the University of Greenwich Research and Enterprise fund. We thank Steve Belmain, Martin Donnelly, Mandela Fernandez-Grandon, Richard Hopkins, Mike Lehane and Glyn Vale for discussion and comments on the manuscript. We thank Jess Lingley and Emily Rippon for technical laboratory support and Stephen Young for statistics advice.
Author information
Authors and Affiliations
Contributions
F.M.H. and G.G. conceived the study; F.M.H., G.G. and S.J.T. designed the study; F.M.H., R.K.D. and S.P.S. performed experiments; F.M.H. analysed the data; F.M.H., G.G. and S.J.T. wrote the manuscript; all authors read, commented on and approved the manuscript in its final form.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Hawkes, F.M., Dabiré, R.K., Sawadogo, S.P. et al. Exploiting Anopheles responses to thermal, odour and visual stimuli to improve surveillance and control of malaria. Sci Rep 7, 17283 (2017). https://doi.org/10.1038/s41598-017-17632-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-17632-3
This article is cited by
-
The behaviour of adult Anopheles gambiae, sub-Saharan Africa’s principal malaria vector, and its relevance to malaria control: a review
Malaria Journal (2024)
-
Field comparison of broad-spectrum white LED-baited traps with narrow-spectrum green LED-baited traps in the capture of Anopheles mosquitoes (Diptera: Culicidae)
Parasitology Research (2024)
-
Evaluation of the solar-powered Silver Bullet 2.1 (Lumin 8) light trap for sampling malaria vectors in western Kenya
Malaria Journal (2023)
-
A randomized, double-blind placebo-control study assessing the protective efficacy of an odour-based ‘push–pull’ malaria vector control strategy in reducing human-vector contact
Scientific Reports (2023)
-
A statistical calibration tool for methods used to sample outdoor-biting mosquitoes
Parasites & Vectors (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.