Abstract
Skeletal fluorosis resulting from high fluoride level in drinking water is a major public health problem. The present study evaluated the association between exposures to drinking water fluoride and skeletal fluorosis in 5 villages of Poldasht County, Iran. All the data and information on the prevalence of bone diseases were obtained from the Health Record Department, Poldasht Health Centre. To obtain the odds ratio of bone disease problem in different risk factors, when considering the cluster effect of rural area, logistic regression in a multilevel model was used. Results showed that skeletal fluorosis of people who live in areas with high fluoride concentration is 18.1% higher than that of individuals who live in areas with low fluoride concentration. Skeletal fluorosis (54.5%) was observed in the age group of 71 years and above, and was more commonly found in females than males. According to Unadjusted, individuals who consume ≤3 unit milk and dairy products per week have almost the same level of bone diseases as compared to those that consume more than 3 units. This study indicated that, skeletal fluorosis is a general health problem in these rural areas because the results revealed that high percentage of the studied population had symptoms of skeletal fluorosis.
Similar content being viewed by others
Introduction
Fluoride is one of the anions that endanger human health at concentrations lower and higher than the standard, and this is one of the main problems in most parts of the world. About 200 million people from 25 countries are exposed to high concentrations of fluoride from groundwater sources1,2. Fluoride is an element from the halogen group and its average concentration in the Earth’s crust is 0.3 kg, while its background concentration in the atmosphere is 3 ng per m2. Fluoride enters into water sources from natural resources, as well as industries where the mineral fluoride is used as raw material for extraction of aluminum, mining, pottery, making of ceramics, bricks, and manure which are the artificial and important contamination sources of fluoride3,4,5,6. The relationship between fluoride and human health was first discussed in the late nineteenth century when chemists observed different concentrations of fluorine in the tissues, bones and teeth of human7,8. Fluoride is one of the essential micro-elements for animals and humans and its consumption at a reasonable range protects teeth against microbial attack, especially in childhood; however, exposure to excessive concentration of fluoride can damage skeletal tissue (bones and teeth). However, fluoride minimum nutritional requirement cannot be exactly determined but the risk of adverse effects on the skeleton can be observed at concentrations higher than 6 mg/l per day9,10,11,12,13,14,15,16. Fluoride is absorbed by the human body through food, drinking water, toothpaste, mouthwash products and air, but air cannot be a major source for absorption of this element; however, the gastrointestinal tract is one of the main routes through which fluoride is absorbed and food is the most important source. Fluoride can be found in every type of food, but some foods like curly kale, endive and fish have higher fluoride content than others. Among the listed sources for fluoride absorption, drinking water is the main one in most societies2,3. Fluoride is variable in water sources and mainly depends on the type of stone and soil which water flows from. Many epidemiological studies have shown that absorption of fluoride in drinking water in the long term leads to adverse effects on the human skeletal tissue. Therefore, the World Health Organization (WHO) has considered the minimal concentration of 1.5 mg/L2 17,18. Worldwide, the concentration of fluoride in drinking water and its relation to bone and skeletal diseases have been investigated in some restricted areas19,20,21,22. Thus, the aim of the present study was to investigate the prevalence of skeletal diseases among people in two municipalities that had different standards with regards to control of the optimal fluoride concentration in water consumed by the population.
Materials and Methods
Study areas
Poldasht county is located in North West of Azerbaijan province, Iran with coordinates (UTM) X = 446625 to 513055 to the east and Y = 4344280 to 4402863 to the north. Poldasht meteorological station showed that in a long-term, the average rainfall is equal to 131.5 mm. The city also has borderline in the West and North with Turkey. Two study areas (five villages) were selected in Poldasht County with almost the same socioeconomic status and dietary habits but different natural concentrations of F in drinking water. Two villages had a high level of F (Sari su, Konikor, Ag otlogh) and two had low level of F (Hasan kandi and Shiblu).
Determination of the water fluoride concentration
Four drinking water wells in the area were selected. A total of 60 samples were obtained over three consecutive years, 2013 to 2015. The water samples were collected from ground water wells in sterile plastic 2-L container, then transported to the Laboratory for Water and Sewage, Poldasht. Fluoride concentration of the samples was determined using SPADNS method according to the Standard. The samples of the ground water were analyzed for physico-chemical parameters: total hardness (TH), chloride, nitrate, electrical conductivity, total alkalinity (TA), total dissolved solids (TDS), bicarbonate (HCO3−) and fluoride, using standard analytical methods23.
Data collection
All data and information on prevalence of bone diseases were obtained from the Health Record Department, Poldasht Health Centre on the areas investigated. Based on this information given as health cases or questionnaire sheets, 445 persons in the high F area and 470 in the low F area, were identified.
Skeletal fluorosis was assessed using three simple diagnostic tests:
-
1- Touching the toes without bending the knees
-
2- Touching the chest with the chin; and stretching
-
3- The arms placed sideways and folding them to touch the back of the head24.
If there is pain or stiffness in the backbone, hip and joints; neck; shoulder joint and backbone, respectively, these three exercises cannot be performed.
Statistical method
To present the data, mean, standard deviation, median and range were used. To compare the case and control groups, Chi-square and Mann-Whitney tests were used. To obtain the odds ratio of bone disease problem in different risk factors, when considering the cluster effect of rural area, logistic regression in a multilevel model was used. In the last step, the simultaneous effect of all risk factors on bone disease was evaluated by multiple multilevel logistic regression. All statistical analyses were performed by STATA (version 14). P-value less than 0.05 was considered to be statistically significant.
Results
Physico-chemical characteristics
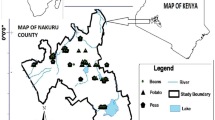
Data presented here shows the monitoring of physical and chemical characteristics including pH, EC, TDS, HCO3, CO3,SO4, Cl, NO3 and F in Poldasht County, West Azerbaijan, Iran. Figure 1 shows the study area and the sampling points. A summary of water quality characteristics and correlation of the parameters with fluoride are presented in Tables 1 and 2, respectively. Fluoride ion varied from 0.68 to 10.30 mg/L. This is given in Tables 1 and 2. Minimum (0.68 mg/L) and maximum (10.3 mg/L) concentrations of F were observed in Hasan kandi and Agh otlogh villages, respectively. Also, according to the data, 45% of villagers are in the desirable range and in 55 of them, the amount of fluorine was more than the standard level.
Location of the study areas in Poldasht City, West Azerbaijan, Iran36.
Correlation of fluoride concentration with other physicochemical parameters
In order to study the possible relationship between fluoride concentration and other physicochemical parameters studied, correlation studies were performed. There was a positive correlation between fluoride level and HCO3 (r = 0.646, P = 0.001) as compared to other physicochemical parameters studied. Other parameters such as PH, EC, TDS, ALK, chloride, nitrate and sulfate had a weak correlation with fluoride (Table 3).
Comparison of various epidemiological factors influencing the skeletal fluorosis
21.1% of people who live in areas with high fluoride concentrations have bone diseases, which means among the 445 people who live in this region, 94 have skeletal fluorosis (94/445), and 3% of those who live in areas with low fluoride concentration, have skeletal fluorosis were apparent (Fig. 2), which means among the470 people who live in this area, 14 have skeletal fluorosis (14/470). Therefore, it can be said that skeletal fluorosis problem of people who live in areas with high fluoride concentration is 18.1% higher than that of individuals who live in areas with low fluoride concentration. Additionally, the age groups are presented in Table 3. In the case of consumption of milk and dairy, 11.2% of the people who live in these areas (both high and low fluoride) and consume milk and dairy products less than 3 units per week, have skeletal fluorosis and 12.6% of those who consume 3 units and more have skeletal fluorosis (Table 4).
According to the 2 Unadjusted* table, people who live in area with low concentration of fluoride have skeletal fluorosis which is 0.12 times higher than people who live in area with high concentration of fluoride. According to the Adjusted **, people who live in area with low concentrations of fluoride have skeletal fluorosis which is 0.11 times higher than that of individuals who live in area with low concentrations of fluoride. The age group of 41–50 years has skeletal fluorosis 2.49 times higher than that of 40 years range, while people who are in the age range of 51–60 have skeletal fluorosis 1.54 times more than people in 41–50 age range. In the case of milk and dairy products, according to Unadjusted*, people who consume ≤3 units milk and dairy products per week have almost the same level of skeletal fluorosis as compared to those who consume 3 units and more. Table 3 also shows that men (Unadjusted*) who live in the high fluoride concentrations area have skeletal fluorosis 11.59 times higher than those who live in low fluoride area, and based on the Adjusted**, men who live in area with high concentrations of fluoride have skeletal fluorosis 6.049 times higher than men who live in low fluoride concentration area, while for women, according to Unadjusted*, those who live in area with high concentrations of fluoride have skeletal fluorosis 8.47 times higher than women who live in area with low concentrations of fluoride, and based on Adjusted**, they have skeletal fluorosis which is 8.727 times higher than that of women who live in area with low concentrations of fluoride (Tables 5, 6).
Discussion and Conclusion
This study revealed that F concentration in drinking water is in the range of 0.22 to 10.33 ppm, minimum and maximum, respectively. Fluoride concentration values of water sources in Poldasht County are higher than the permissible limit (0.5 to 1.5 mg/L) according to WHO guideline25 (Table 1). In a series of studies on the various effects of fluoride concentration in drinking water on human tissue, it has been well observed and documented that the toxic effects of fluoride on skeletal system are chronic and occurs over time. Skeletal fluorosis may be displayed by increase in the bone density in adults using X-ray26,27 (Fig. 2). The results of this study showed that 21.1% of people who live in areas with high fluoride concentrations have skeletal fluorosis, which means, among the 445 people who live in this region, 94 have skeletal fluorosis (94/445) and 3% of those who live in areas with low fluoride concentration have skeletal fluorosis, meaning that among the 470 people who live in this area, 14 have skeletal fluorosis (14/470) (Table 1). Choubisa showed at 3.2 ppm, 3.7 ppm, and 4.0 ppm water F concentration in villages in India, the highest prevalence of skeletal fluorosis was 39.2, 32.8, and 36.6%, respectively. In the present study, the prevalence of skeletal fluorosis is lower than that reported by this reaserch28. Regarding the prevalence of skeletal fluorosis in the two municipalities (3 and 21.4%), these values are within the variation of 31to 80% and 35 to 60% reported for fluoridated communities in the State of São Paulo and United State, respectively29,30. Skeletal fluorosis (54.5%) in the age group of 71 years & above was more commonly found in females than males, but the results of a study conducted by Asawa et al. on Association of Temporomandibular Joint Signs & Symptoms with Dental Fluorosis & Skeletal Manifestations in Endemic Fluoride Areas of Dungarpur District, Rajasthan, India, are not compatible with those of the present study because Skeletal Fluorosis (61.1%) in the age group of 55–64 years was more commonly found in males than females31. Sushella and colleagues in their research found that development of skeletal fluorosis can be due to the consumption of fluoride together with other factors such as low calcium, alkalinity of water, and also, lack of calcium and vitamin C32. The present study demonstrates there was significant difference between females and males in the prevalence of skeletal fluorosis (15 and 8.6%) respectively. Watanabe et al. showed that the prevalence of skeletal fluorosis was significantly higher for males in a moderately polluted area, but not in a severely polluted area. These results are not consistent with our findings. This matter clearly requires further study33. A survey by ChubiSa et al. showed that the prevalence of skeletal fluorosis in people who had inappropriate regime increased by 6.61%34. Furthermore, study of Karthikeyan et al. on Contribution of Fluoride in Water and Food to the Prevalence of Fluorosis in Areas of Tamil Nadu in South India showed a significant correlation between high fluoride concentration in drinking water and enhanced incidence of skeletal fluorosis in 5 fluorine-rich areas, native to Tamilnad35.
Conclusion and Recommendation
Our research is important in that it reports, for the first time in Iran, evidence of fluoride toxicities in people living in the Poldasht County in west Azerbaijan, Iran. The present study demonstrates a significant relationship between the fluoride concentrations in the water and the prevalence of skeletal fluorosis in an endemic fluorosis area. The provision of defluoridated drinking water, and health education aimed at abating fluorosis in people is highly desirable in this village area of Poldasht County in west Azerbaijan, Iran. Government and relevant organizations such as the Department of Energy in the preparation of suitable drinking water in the home and community levels by providing internal filters with affordable costs and the Ministry of Health by raising awareness and appropriate interventions in nutrition, will play important roles in dealing with the problem of fluorosis.
References
Edmunds, M & Smedley, P. L. Fluoride in natural waters Essentials of medical geology. Elsevier Academic Press, Burlington, 2005; 301–29.
WHO. Fluoride in drinking-water: background document for development of WHO Guidelines for drinking-water quality. WHO/SDE/WSH/03.04.96, English only. Geneva.Availablefrom: http://www.who.int/water_sanitation_health/dwq/chemicals/fluoride.pdf. (2004).
Mesdaghinia, A. R., Vaghefi, K. A., Montazeri, A., Mohebbi, M. R. & Saeedi, R. Monitoring of Fluoride in Groundwater Resources of Iran. Bull Environ Contam Toxicol. 84, 432–7 (2010).
Faraji, H. et al. Correlation between fluoride in drinking Water and its levels in breast milk in Golestan Province, Northern Iran. Iran J Public Health. 43, 1664–68 (2014).
Maleki, A. et al. Influence of selected anions on fluoride removal electrocoagulation/electroflotation. Fluoride. 48, 37–47 (2015).
Mohammadi, A. A. et al. Temporal and spatial variation of chemical parameter concentration in drinking water resources of Bandar-e Gaz City using Geographic Information System. Desalination and Water Treatment. 68, 170–176 (2017).
Kanduti, D., Sterbenk, P. & Artnik, B. fluoride: a review of use and effects on health. Mater Sociomed. 28, 133–137 (2016).
Karimzade, S., Aghaei, M. & Mahvi, A. H. Investigation of intelligence quotient in 9–12-year-old children exposed to high and low-drinking water fluoride in west Azerbaijan province, Iran. Fluoride. 47, 9–14 (2014).
Mahvi, A. H., Ghanbarian, M., Ghanbarian, M., Khosravi, A. & Ghanbarian, M. Determination of fluoride concentration in powdered milk in Iran 2010. Fluoride Journal. 107, 1077–1079 (2012).
Aghaei, M. et al. hypertension and fluoride in drinking water: case study from west Azerbaijan, Iran. Research report Fluoride. 48, 252–258 (2015).
Amouei, A. I. et al. Fluoride concentration in potable groundwater in rural areas of Khaf city, Razavi Khorasan Province, Northeastern Iran. Int J Occup Environ Med 3, 201–3 (2012).
Shamsollahi, H. R., Zolghadr, Z., Mahvi, A. H., Hosseini, S. S. & Mossavic, S. N. The Effect of temperature, water hardness, and exposure time on Fluoride toxicity in the Aquatic Environment. Fluoride. 48, 338–44 (2015).
Mohammadi, A. A., Yousefi, M. & Mahvi, A. H. Fluoride concentration level in rural area in poldasht city and daily fluoride intake based on drinking water consumption with temperature. Data in Brief. 13, 312–315 (2017).
Poureslami, H. R., Khazaeli, P., Faryabi, A. & Mahvi, A. H. Fluoride levels and dental fluorosis in deciduous teeth of students residing in Koohbanan, Iran, a city with high-fluoride water and food. Fluoride. 46, 224–9 (2013).
Amouei, A. I. et al. Physical and Chemical Quality Assessment of Potable Groundwater in Rural Areas of Khaf, Iran. World Appl Sci J. 18, 693–7 (2012).
Aghaei, M., Derakhshani, R., Raoof, M., Dehghani, M. & Mahvi, A. H. Effect of Fluoride in Drinking Water on Birth height and weight: An Ecological study in Kerman Province, Zarand County, iran. Fluoride. 48, 160–8 (2015).
Shanthi, M., Thimma Reddy, B. V. & Shivani, K. Health Impact to Different Concentrations of Fluoride in Drinking Water of South India. Int J Sci Stud. 2, 2–5 (2014).
Phipps, K. R., Orwoll, E. S. & Bevan, L. The Association Between Water-borne Fluoride and Bone Mineral Density in Older Adults. J Dent Res. 77, 1739–1748 (1998).
Choubisa, S. L., Modasiya, V., Bahura, C. K. & Sheikh, Z. Toxicity of Fluoride in Cattle of The Indian Thar Desert. Rajasthan, India, Research report Fluoride. 45, 371–376 (2012).
Izuora, K. et al. Skeletal Fluorosis from Brewed Tea. J Clin Endocrinol Metab. 96, 2318–2324 (2011).
Kakumanu, N. & Rao, S. D. Skeletal Fluorosis due to Excessive tea drinking. New England Journal of Medicine. 368, 1140 (2013).
Grobler, S. R., Louw, A. J., Chikte U. M. E, Rossouw, R. J. & Van W Kotze, T. J. The Relationships between Two Different Drinking Water Fluoride Levels, Dental Fluorosis and Bone Mineral Density of Children. The Open Dentistry Journal. 3, 48–75 (2009).
Eaton ADScaEWR. Standard Methods for the Examination of water waster: AWWA; (2005).
Susheela, A. K. editor. Fluorosis: An easily preventable disease through practice of interventions. In: Doctor’s Handbook. 1st Ed. New Delhi: Ministry of Health and Family Welfare (GOI) and WHO India Country Office; 2005. p. 1–21.
World Health Organization. Nitrate and nitrite in drinking-water: Background document for development of WHO Guidelines for Drinking-water Quality. 2011. Available from: http://www.who.int/water_sanitation_health/dwq/ chemicals/nitratenitrite2ndadd.pdf.
Sun, L. et al. An assessment of the relationship between excess fluoride intake from drinking water and essential hypertension in adults residing in fluoride endemic areas. Sci Total Environ. 443, 864–869 (2013).
Liu, H. et al. Assessment of relationship on excess fluoride intake from drinking water and carotid atherosclerosis development in adults in fluoride endemic areas, China. International Journal of Hygiene and Environmental Health. 217, 413–20 (2014).
Choubisa, S. L. Endemic fluorosis in Southern Rajasthan, India. Fluoride. 34(1), 61–70 (2001).
Catani, D. B., Hugo, F. N., Cypriano, S., Sousa, M. L. & Cury, J. A. Relationship between fluoride levels in the public water supply and dental fluorosis. Rev Saúde Pública. 41, 1–7 (2007).
Clark, D. C., Hann, H. J., Williamson, M. F. & Berkowitz, J. Influence of exposure to various fluoride technologies on the prevalence of dental. Community Dent Oral Epidemiol. 22, 461–4 (1994).
Asawa, K. et al. Association of Temporomandibular Joint Signs & Symptoms with Dental Fluorosis & Skeletal Manifestations in Endemic Fluoride Areas of Dungarpur District, Rajasthan, India. Journal of Clinical and Diagnostic Research. 9, 18–21 (2015).
Susheela, A. K., Kumar, A., Bhatnagar, M. & Bahadur, R. Prevalence of Endemic Fluorosis with Gastrointestinal Manifestations in People Living in some North- Indian Villages. Fluoride. 26, 97–104 (1993).
Choubisa, S. L., Choubisa, L. & Choubisa, D. Osteo-Dental Fluorosis In Relation To Nutritional Status, Living Habits, And Occupation In Rural Tribal Areas Of Rajasthan, India. Fluoride. 42, 210–15 (2009).
Watanabe, T. et al. Skeletal fluorosis from indoor burning of coal in Southwestern China. Fluoride. 33(3), 135–9 (2000).
Karthikeyan, G., Pius, A. & Apparao, B. V. Contribution of Fluoride in Water and Food to the Prevalence of Fluorosis in Areas of Tamil Nadu in South India. Fluoride. 29, 151–55 (1996).
Iran Mapping Organization. Technical service management, http://tsm.ncc.org.ir/HomePage.aspx?TabID=6925&Site=tsm.ncc.org&Lang=fa-IR (accessed 28 June 2017).
Acknowledgements
The authors thank authorities of Tehran University of Medical Sciences and Poldasht Health Centre for their comprehensive support in this study.
Author information
Authors and Affiliations
Contributions
A.H.M. provided the idea for this work and designed. M.Y. and M.J. data gathering, prepared Tables 1–2 and Figure 2. M.Y. performed statistical data analysis and prepared Tables 3–5. A.H.M. and M.A.A. Wrote the main manuscript text, discussed the scientific idea and reviewed the manuscript. A.A. Mohammadi generated the Figure 1. All authors participated in writing the manuscript. All authors reviewed the manuscript. All authors contributed to the scientific discussion. These authors jointly supervised this work.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Mohammadi, A.A., Yousefi, M., Yaseri, M. et al. Skeletal fluorosis in relation to drinking water in rural areas of West Azerbaijan, Iran. Sci Rep 7, 17300 (2017). https://doi.org/10.1038/s41598-017-17328-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-17328-8
This article is cited by
-
Severe magnitude of dental and skeletal fluorosis and its impact on society and environment in a part of Manbhum-Singhbhum Plateau, India
BMC Public Health (2024)
-
Kinetics of fluoride after brushing with the no-rinse method
BMC Oral Health (2024)
-
A comparison of conventional sodium fluoride varnish and nano-sodium fluoride varnish regarding enamel microhardness of deciduous teeth: an in-vitro study
European Archives of Paediatric Dentistry (2024)
-
Synthesis of copper oxide (CuO) nanoparticles for the efficient removal of fluoride from an aqueous solution
Journal of Radioanalytical and Nuclear Chemistry (2024)
-
Underlying Mechanism of Fluoride Inhibits Colonic Gland Cells Proliferation by Inducing an Inflammation Response
Biological Trace Element Research (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.