Abstract
Fluctuating dental asymmetry (FDA) is a tool to measure developmental stability that could be increased in gonosomal aneuploidies. The aim of this study was to quantify FDA in individuals with Down syndrome (DS). The study group comprised 40 individuals with DS, and a control group matched for age and sex was created. The target teeth were the maxillary central incisors (11,21), maxillary lateral incisors (12,22), maxillary canines (13,23), and maxillary first molars (16,26). Dental morphometric variables measured on CBCT images included tooth length, crown height, root length, mesio-distal diameter, crown-to-root ratio, vestibular-palatine diameter, mid mesio-distal diameter, mid buccal-palatal diameter, maximum buccal-palatal diameter, and cervical circumference. The FA2 fluctuating asymmetry index (Palmer and Strobeck, 1986) was applied. Some discrepancies in crown-to-root ratios and root length asymmetry were significantly lower in the DS individuals than in controls. Combining the crown-to-root ratio of tooth 11 versus 21, tooth 12 versus 22, and tooth 13 versus 23, we developed a predictive model with a discriminatory power between DS and controls of 0.983. Some dental morphometric variables may actually be more stable in DS individuals than in the general population. This offers a new perspective on the relationship between canalization, fluctuating asymmetry, and aneuploidy.
Similar content being viewed by others
Introduction
The concept of canalization refers to a phenomenon whereby the development of phenotypic traits is buffered against environmental influences, so that such traits produce a highly predictable genetically determined endpoint1. The canalization capacity of an organism is called developmental stability 2 and its measurement is based on small variations of antimeric traits at random with respect to side (right-left). This biological asymmetry of morphological traits is called fluctuating asymmetry 3.
It has been suggested that canalization is reduced, and fluctuating asymmetry thus augmented, in disorders of developmental origin, and is detectable in most if not all of the gonosomal aneuploidies4,5. The most common live-born human aneuploidy is trisomy 21, which causes Down syndrome (DS). The term amplified developmental instability was coined around 50 years ago to describe the generalized genetic imbalance that trisomy 21 causes in developmental homeostasis6. In DS, increased fluctuating asymmetry has been reported in skeletal anomalies7, dermatoglyphics8, facial dysmorphology9, and palatal dimensions10.
Application of the concept of fluctuating asymmetry to teeth has enabled small, randomly distributed morphometric differences to be identified between the teeth of contralateral arches; this is called fluctuating dental asymmetry 11. Few details of the genetic and environmental factors implicated in fluctuating dental asymmetry are yet known12, with the exception of chromosomal abnormalities and some single gene substitutions13,14. Articles published in the 1970s and 80s showed that individuals with DS had significantly greater tooth crown asymmetry than controls13,15,16. One of the drawbacks of those studies was that only the crown dimensions were evaluated. It has been stated that the study of fluctuating asymmetry requires the selection of traits with a low vulnerability to wear, as this would otherwise complicate the interpretation of asymmetry variation17, and tooth crown wear is paradoxically particularly common and severe in DS due to attrition and erosion18. A further limitation common to those studies was that other variables that could affect tooth morphometrics and asymmetry, such as sexual dimorphism19 or age20, were not taken into account.
In 2014, we published an article in which we analyzed tooth dimensions not previously studied in individuals with DS—such as root length and cervical circumference—using cone beam computed tomography (CBCT) images21. We found significant differences in crown height and crown-to-root ratio between the maxillary right and left canines, and in maximum buccal-palatal diameter between the maxillary right and left first molars21,22. In that study, the results were analyzed using additive mixed models23, which enabled us to include the smooth effect of age, the fixed effects of sex and teeth, and the random effect of patient. The main drawback of that study was that asymmetry of the dental morphometric variables was measured as the absolute value of the difference between right and left, while the most useful descriptor of fluctuating asymmetry is variance3,24. The aim of the present study has been to reassess fluctuant dental asymmetry in a series of individuals with DS, evaluated objectively without taking into account the absolute size of the teeth and, therefore, without the effect of scale.
Material and Methods
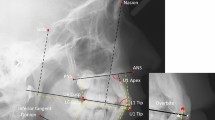
The characteristics of the study group and the methodology used to obtain the CBCT image are described in detail in our previous article21. Briefly, the study group was formed of 40 white individuals with DS (25 males and 15 females; mean age, 18.8 ± 7.3 years [range, 9–43 years]). The control group comprised 40 healthy, age- and sex-matched individuals without DS (25 males and 15 females; mean age, 19.5 ± 7.2 years [range, 10–43 years]). The CBCT images were obtained using an I-CAT® scanner (Imaging Sciences International, Hatfield, PA, USA), were reconstructed with I-CAT VISION® software (Imaging Sciences International), and were exported using the DICOM (Digital Imaging Communication in Medicine) format to a MacBook 27 personal computer (Mac OsX 10.6, Apple, Inc., Cupertino, USA). Measurements were performed using the open-source OsiriX medical image processing software (Pixmeo, Geneva, Switzerland; www.osirixviewer.com). For the analysis of tooth morphometry, the CBCT images were oriented using multiplanar reconstruction and a modification of the method described by Sherrard et al.25 was applied. The target teeth of the study were the maxillary central incisors, the maxillary lateral incisors, the maxillary canines, and the palatal root of the maxillary first molars. Overall tooth length, crown height, root length, and mesio-distal diameter were measured in the coronal plane. The crown-to-root ratio was defined as the ratio of the crown height to the root length. Vestibular-palatine diameter, mid mesio-distal diameter, mid buccal-palatal diameter, maximum buccal-palatal diameter, and cervical circumference were measured in the axial plane (Fig. 1). The interobserver and intraobserver reliability of this measurement system has been demonstrated previously21.
Measurement of some relevant dental dimensions: overall tooth length (TL), crown height (CH), root length (RL), mesio-distal diameter (MD), vestibular-palatine diameter (VP), and cervical circumference (for further details see reference Abeleira et al.21).
All the statistical analyses were carried out with the statistical software R, version 2.12.0. (R Development Core Team, Vienna, Austria), using the following packages: “fBasics” to calculate the basic statistics of each of the variables analyzed; and “mgcv” to fit additive mixed models to determine dimensional symmetry between the central incisors, between the lateral incisors, between the canines, and between the first molars. Additive mixed models are extensions of linear mixed models and enable us to include random effects in addition to the usual fixed effects26. In this study, we considered the following additive mixed models, which included the smooth effect of age, the fixed effects of gender and teeth, and the random effect of patient: [Tooth measurement = β0 + f (Age) + Gender + Teeth + random (Patient) + ε], where f (.) refers to unspecified smooth functions, producing separate effects of age in each group.
Hypothesis testing was performed using a variety of methods (t test, Wilcoxon, Kruskal-Wallis) to determine whether significant differences in tooth dimensions existed between the study group (DS) and the control group.
At the individual level, we used the FA2 fluctuating asymmetry index described by Palmer and Strobeck [3,27]). In this index, the |Right - Left| difference is divided by the mean, given as (R + L)/2. This fluctuating asymmetry index corrects for trait size effects by expressing deviations from symmetry as a proportion of trait size17. The FA2 results underwent further hypothesis testing, mainly using the Kruskal-Wallis test as some data did not have a normal distribution, to determine whether significant differences in the degree of dental asymmetry were present between the DS group and the control group.
A generalized additive model was developed to evaluate the discriminatory power of the dental morphometric variables, processed using the FA2 index, to classify a given individual as DS or non-syndromic control.
These radiological studies were performed in accordance with the radiation protection principles of As Low As Reasonably Achievable (ALARA) and following the guidelines of the SEDENTEXCT Guideline Development Panel. Radiation Protection No. 172: Cone Beam CT for Dental and Maxillofacial Radiology. Evidence Based Guidelines 2012 (www.sedentexct.eu).”
Ethical approval
The study was approved by the Institutional Review Board of the University of Santiago de Compostela (USC), Spain.
Informed consent
All the images used in this study belonged to the historical archive of the Radiology Unit of the Faculty of Medicine and Dentistry of the University of Santiago de Compostela in Spain. No specific informed consent was required as all participants or, as appropriate, their legal representatives had signed an informed consent to authorize the use of images for teaching or research purposes.
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
Results
The absolute values of the dental morphometric variables evaluated in the DS group and the control group are shown in Table 1. The Kruskal-Wallis test revealed statistically significant differences between individuals with DS and controls in 69 of the 74 dental morphometric variables evaluated (Table 2). With the exception of the crown-to-root ratio of teeth 11, 21, and 16, the root length of tooth 22, and the crown height of tooth 26, all values were significantly lower in the individuals with DS.
Table 3 list the differences in morphometric variables between contralateral teeth in the DS group and in the control group, after application of the FA2 index3,27. The Kruskal-Wallis test revealed differences in the crown-to-root ratios of tooth 11 versus 21, 12 versus 22, and 13 versus 23, with significantly lower values in the DS individuals than in controls. In addition, root length asymmetry of tooth 13 versus 23 was significantly smaller in the DS group than in the control group. In contrast, the differences in the crown-to-root ratio and the cervical circumference of tooth 16 versus 26 were greater in the DS group than in the controls (Table 4).
Combining the crown-to-root ratios of tooth 11 versus 21, 12 versus 22, and 13 versus 23, we developed a predictive model with an area under the curve (AUC) of 0.983 (95% confidence interval = 0.958–1) (Table 5).
Discussion
In our series, the morphometric dimensions of the teeth evaluated were smaller in the DS individuals than in the controls. These results confirm the findings of other authors, who also showed that the crown dimensions of permanent teeth were smaller in individuals with DS than in healthy controls28, and that the roots of most anterior teeth and premolars in the DS population were shorter than in the general population29.
Microdontia of the permanent teeth is considered a phenotypic characteristic of DS28, and for comparison with non-syndromic control groups, asymmetry of dental morphometric variables in individuals with DS should not therefore be evaluated in terms of absolute right-left differences. One of the most widely used methods proposed to correct the size dependence of variability in studies of fluctuating asymmetry (FA) is the index-trait difference divided by the trait mean (FA2 index)3. FA2 describes fluctuating asymmetry as a proportion of trait size by estimating the between-sides variance and is hardly affected by departures from normality (skew or leptokurtosis)17, although it has been criticized for the apparent lack of independence between the numerator and the denominator30.
In the present study, we did not find greater dental crown asymmetry in DS individuals than in the controls. These results contrast with those published some decades ago by other authors, who reported greater dental asymmetry in DS15,16. Although those studies are of indisputable value, they carry relevant biases, such as the absence of an age- and sex-matched control group15 and the use of parametric tests to look for differences between DS individuals and controls15,16 when it is known that some dental morphometric variables do not have a normal distribution.
A novel finding of this study has been that the root length asymmetry of tooth 13 versus 23 was significantly lower in the DS group. Twin studies have shown that the canines are the teeth with greatest genetic control of dimensional variations in the general population, and they are considered the most stable teeth in the maxillary dentition31. Despite the biases we have indicated in those previous studies, some authors have suggested that the mesiodistal crown diameter of the maxillary right versus left canines was similar in DS individuals and in the general population32.
The asymmetries detected in multiroot teeth such as the maxillary first molar must be interpreted with caution, as only the dimensions of the palatine root are evaluated. It has therefore been suggested that an additional method should be devised to achieve a more accurate crown-to-root ratio of the maxillary molars33.
The most relevant result is that crown-to-root ratio asymmetry between the maxillary right and left central incisors, right and left lateral incisors, and right and left canines was significantly lower in the DS group. This finding is surprising for both biological and anatomical reasons, as formation of the dental crowns starts in the early weeks of intrauterine life, whereas the root portion of the tooth takes several years to develop fully and, in addition, the extra-osseous part of the tooth is particularly exposed to certain environmental aggressions, such as tooth wear. As a result, this finding requires us to revise our concepts of developmental biology such as canalization and modelization in order to analyze certain traits in individuals with DS.
The binary classification capability (DS versus control groups) of the mixed additive model that includes the crown-to-root ratio of the right versus left anterior teeth, enables us to identify individuals with DS by analyzing dental morphometric variables that can be measured easily on 2-dimensional images such as periapical or panoramic x-rays34. This finding could become a useful tool for the diagnosis of DS in areas such as paleopathology35 and paleoantropathology36.
The potential limitations of this study include the teeth selected, the dental variables analyzed, the method used to quantify left-right dental asymmetry and the sample size. Applying Butler’s morphogenetic field concept, the mesial tooth of each morphological tooth group is the most developmentally stable37; this conflicts with the results in the DS series published13,15, and the need to perform morphometric measurements on all teeth in the future will have to be discussed. The use of CBCT enabled us to analyze several tooth dimensions simultaneously, some of which had not previously been evaluated in DS individuals21. Use of the FA2 index3 to quantify fluctuating dental asymmetry obviates errors derived from trait size or from the use of correlations. Although it has been stated that sample sizes of several hundred are needed to detected population differences in dental asymmetry12, some authors have suggested a minimum sample size of 30 individuals as an empirical rule for studies of fluctuating asymmetry38. As the data obtained in the present study showed a low variability after applying the FA2 index, we consider that valid statistical conclusions can be drawn with the sample size used.
In summary, taking into account the limitations of this study, fluctuating dental asymmetry would appear not only not to be greater in DS individuals than in the general population, but some dental morphometric variables may also actually be more stable in individuals with trisomy 21. This offers a new perspective on the relationship between canalization, fluctuating asymmetry, and aneuploidy.
Change history
16 January 2024
A Correction to this paper has been published: https://doi.org/10.1038/s41598-024-51672-w
References
Waddington, C.H. The Strategy of the Genes: A discussion of some aspects of theoretical biology (ed. Waddington, C.H.) (George Allen and Unwin,1957).
Waddington, C. H. Canalization of development and the inheritance of acquired characters. Nature 150, 563–565 (1942).
Palmer, A. R. & Strobeck, C. Fluctuating asymmetry: measurement, analysis, patterns. Annu Rev Ecol Sys 17, 391–421 (1986).
Opitz, J. M., Mendez, H. M. & Hall, J. G. Growth analysis in clinical genetics. Prog Clin Biol Res 200, 33–63 (1985).
Naugler, C. T. & Ludman, M. D. Fluctuating asymmetry and disorders of developmental origin. Am J Med Genet 66, 15–20 (1996).
Shapiro, B. L. Prenatal dental anomalies in mongolism: comments on the basis and implications of variability. Ann New York Acad Sci 171, 562–577 (1970).
Blum-Hoffmann, E., Rehder, H. & Langenbeck, U. Skeletal anomalies in trisomy 21 as an example of amplified developmental instability in chromosome disorders: a histological study of the feet of 21 mid-trimester fetuses with trisomy 21. Am J Med Genet 29, 155–160 (1988).
Shapiro, B. L. Amplified developmental instability in Down’s syndrome. Ann Hum Genet 38, 429–437 (1975).
Starbuck, J. M., Cole, T. M. 3rd, Reeves, R. H. & Richtsmeier, J. T. Trisomy 21 and facial developmental instability. Am J Phys Anthropol 151, 49–57 (2013).
Shapiro, B., Gorlin, R., Redman, R. & Bruhl, H. The palate and Down’s syndrome. N Engl J Med 276, 1460–1463 (1967).
Van Valen, L. A study of fluctuating asymmetry. Evolution 16, 125–142 (1962).
Smith, B. H., Garn, S. M. & Cole, P. E. Problems of sampling and inference in the study of fluctuating dental asymmetry. Am J Phys Anthropol 58, 281–289 (1982).
Garn, S. M., Cohen, M. M. & Geciauskas, M. A. Increased crown-size asymmetry in trisomy G. J Dent Res 49, 465 (1970).
Peretz, B., Shapira, J., Farbstein, H., Arieli, E. & Smith, P. Modified cuspal relationships of mandibular molar teeth in children with Down’s syndrome. J Anat 193, 529–533 (1998).
Barden, H. S. Fluctuating dental asymmetry: a measure of developmental instability in Down syndrome. Am J Phys Anthropol 52, 169–173 (1980).
Townsend, G. C. Fluctuating dental asymmetry in Down’s syndrome. Aust Dent J 28, 39–44 (1983).
Palmer, A.R. & Strobeck, C. Fluctuating asymmetry analyses revisited. 279-319 In: (ed. Polak, M.) Developmental Instability (DI): Causes and Consequences. (Oxford University Press, 2003).
Bell, E. J., Kaidonis, J. & Townsend, G. C. Tooth wear in children with Down syndrome. Aust Dent J 47, 30–35 (2002).
Banerjee, A., Kamath, V. V., Satelur, K., Rajkumar, K. & Sundaram, L. Sexual dimorphism in tooth morphometrics: An evaluation of the parameters. J Forensic Dent Sci 8, 22–27 (2016).
Palestis, B. G. & Trivers, R. A longitudinal study of changes in fluctuating asymmetry with age in Jamaican youth. Symmetry 8, 123 (2016).
Abeleira, M. T. et al. Dimensions of central incisors, canines, and first molars in subjects with Down syndrome measured on cone-beam computed tomographs. Am J Orthod Dentofacial Orthop 146, 765–775 (2014).
Abeleira, M. T. et al. Morphometry of the hard palate in Down’s syndrome through CBCT-image analysis. Orthod Craniofac Res 18, 212–20 (2015).
Pinheiro, J. & Bates, D. Mixed-Effects Models in S and S-PLUS. Series: Statistics and Computing (Springer, 2000).
Palmer, A. R. Fluctuating, asymmetry analyses: A primer (ed. Markow, T.A.) 335–364 In: Developmental Instability: Its Origins and Evolutionary Implications (Springer, 1994).
Sherrard, J. F., Rossouw, P. E., Benson, B. W., Carrillo, R. & Buschang, P. H. Accuracy and reliability of tooth and root lengths measured on cone-beam computed tomographs. Am J Orthod Dentofacial Orthop 137, S100–108 (2010).
Hastie, T. J. & Tibshirani, R. J. Generalized Additive Models. (Chapman & Hall, 1990).
Palmer, A. R. & Strobeck, C. Fluctuating asymmetry as a measure of developmental stability: Implications of non-normal distributions and power of statistical tests. Acta Zool Fenn 191, 57–72 (1992).
Townsend, G. C. Tooth size in children and young adults with trisomy 21 (Down) syndrome. Arch Oral Biol 28, 159–166 (1983).
Kelsen, A. E., Love, R. M., Kieser, J. A. & Herbison, P. Root canal anatomy of anterior and premolar teeth in Down’s syndrome. Int Endod J 32, 211–216 (1999).
Cuthill, I. C., Swaddle, J. P. & Witter, M. S. Fluctuating asymmetry. Nature 363, 217–218 (1993).
Osborne, R. H., Horowitz, S. L. & De George, F. V. Genetic variation in tooth dimensions: a twin study of the permanent anterior teeth. Am J Hum Genet 10, 350–356 (1958).
Barden, H. S. Mesiodistal crown size dimensions of permanent and deciduous teeth in Down syndrome. Hum Biol 52, 247–253 (1980).
Yun, H. J., Jeong, J. S., Pang, N. S., Kwon, I. K. & Jung, B. Y. Radiographic assessment of clinical root-crown ratios of permanent teeth in a healthy Korean population. J Adv Prosthodont 6, 171–176 (2014).
Hölttä, P., Nyström, M., Evälahti, M. & Alaluusua, S. Root-crown ratios of permanent teeth in a healthy Finnish population assessed from panoramic radiographs. Eur J Orthod 26, 491–497 (2004).
Walker, P. L., Cook, D. C., Ward, R., Braunstein, E. & Davee, M. A Down syndrome-like congenital disorder in a prehistoric California Indian. Am J Phys Anthropol 34, S179 (1991). (Abstract).
Baab, K. L. et al. A Critical Evaluation of the Down Syndrome Diagnosis for LB1, Type Specimen of Homo floresiensis. PLoS ONE 11, e0155731 (2016).
Butler, P. M. Studies of the mammalian dentition – differentiation of the postcanine dentition. Proc Zool Soc London 109B, 1–36 (1939).
Benítez, H. A. & Parra, L. E. Asimetría Fluctuante: Una herramienta morfo-funcional para medir estabilidad del desarrollo. Int J Morphol 29, 1459–1469 (2011).
Acknowledgements
The work was supported by the Medical-Surgical Dentistry Research Group (OMEQUI), University of Santiago de Compostela (USC), Spain.
Author information
Authors and Affiliations
Contributions
Marcos Matabuena Rodríguez: Contributed to analysis of data; Drafted the manuscript; Gave final approval. Pedro Diz Dios: Contributed to conception and design; Drafted the manuscript; Gave final approval. Carmen Cadarso Suárez: Contributed to analysis of data; Critically revised the manuscript; Gave final approval. Márcio Diniz Freitas: Contributed to acquisition of data; Critically revised the manuscript; Gave final approval. Mercedes Outumuro Rial: Contributed to acquisition of data; Critically revised the manuscript; Gave final approval. Maria Teresa Abeleira Pazos; Contributed to interpretation of data; Critically revised the manuscript; Gave final approval. Jacobo Limeres Posse: Contributed to conception and design; Drafted the manuscript; Gave final approval.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Matabuena Rodríguez, M., Diz Dios, P., Cadarso-Suárez, C. et al. Reassessment of fluctuating dental asymmetry in Down syndrome. Sci Rep 7, 16679 (2017). https://doi.org/10.1038/s41598-017-16798-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-16798-0
This article is cited by
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.