Abstract
Irrigation with eutrophic water containing cyanobacteria toxins poses a potential risk to soil animals. To evaluate ecotoxicological effect of microcystins (MCs) on earthworms, filter paper acute toxicity test, avoidance test and a 14-d artificial soil test were carried out. No acute toxicity was found in the filter paper test, and earthworms showed no avoidance response to MCs exposure. In the artificial soil test, Eisenia fetida were allowed to grow in presence or absence of MCs (0, 1, 10, 100, 1000 μg kg−1 of soil) for 1, 7, and 14 d. Results showed that MCs could bioaccumulated in earthworm. A stimulatory effect on catalase and glutathione oxidase activities induced by MCs was found on day 1, and both of them were significantly inhibited at 100 and 1000 μg kg−1 on days 14. The superoxide dismutase activity was relatively insensitive. Significant increase of malondialdehyde content and decrease of neutral red retention time were observed at 100 and 1000 μg kg−1 on days 7 and 14. Our results suggest that MCs induces oxidative stress on earthworms, which leads to disruption of the antioxidant system and lipid peroxidation, as well as alterations in lysosomal membrane stability.
Similar content being viewed by others
Introduction
In eutrophic fresh water lakes, a group of toxic compounds are produced by some bloom-forming cyanobacteria. The most prominent of these cyanotoxins are microcystins (MCs). Owing to their potential carcinogenicity, MCs can negatively affect both public health and fundamental ecological processes1. MCs exposure induced thyroid endocrine disruption and transgenerational effects of developmental neurotoxicity in zebrafish offspring as well as in embryo2,3,4. More than 200 different structural analogues of MCs, with a range of molecular weights from 882 to 1116 Da, have been identified from cyanobacterial blooms and cultures5,6. Microcystin-leucine-arginine (MC-LR) is found to be the most common and potent analogue, followed by Microcystin-arginine-arginine (MC-RR) and Microcystin-tyrosine-arginine (MC-YR)7. With the increasing prevalence of these cyanobacteria blooms, more attention is being paid to impacts caused by MCs.
MCs can be brought into contact with soils by irrigation source water that contains cyanobacteria; in addition, cyanobacterial blooms can be applied directly to soil as organic fertilizer after being intentionally harvested from lakes8,9,10,11. Total MC concentrations in surface waters varied from less than 1 μg L−1 to 29, 000 μg L−1 12,13,14,15,16. However, high concentrations of MC would be from very dense cyanobacterial biomass, and the concentration of MCs in most of the water samples were less than 100 μg L−1 17,18,19. Although the MCs concentration in field samples collected near Dianchi Lake were relatively low, ranged from 1.43 to 21.3 μg kg−1 20, high concentrations of MC will occur by irrigation with water containing dense cyanobacterial biomass. Moreover, once come in contact with soil, MCs can persist in soils with a half-life ranging from several days to several months21,22, depending on the efficiency of degradation, mainly photolysis and bacterial degradation. Studies on the absorption of MCs in soils are rare. It is suggested that organic carbon content and clay content in soils were important for the adsorption of MCs, and sandy soil was incapable of the removal of MCs23,24. However, Eynard et al.25 indicated that soil was unable to protect groundwater from surface water that contains cyanotoxins. Therefore, there is a possibility that soils adjacent to lakes and reservoirs may be contaminated by MCs. And the low absorption of MCs in soils could lead to their high bioavailability to soil organisms, animals and plants. However, very few studies have examined the impact of MCs on soils, especially on soil animals, which play important roles in the soil ecosystem. A recent study reported the toxic effects of MC-LR on earthworms26. In their study, MC-LR caused reproductive, biochemical, and cellular toxicity to earthworms. Li et al.27 used the nematode Caenorhabditis elegans as a model animal for assessing the toxicity induced by microcystin-LR. Reduced life span, extended generation time, decreased brood size, delayed development and suppressed locomotion behavior were found in C. elegans after exposed to MC-LR. Caenorhabditis elegans were also used for a chemotactic behavior study induced by MCs28. They found that MCs altered AWA but not AWC sensory neuron function via mechanisms other than protein phosphatases inhibition. However, the survival or reproduction of springtail Folsomia candida was not affected by water bloom biomass at concentration up to 4 g kg−1 DW soil29. As it is known to us, earthworms play vital roles in soil system, such as nutrient cycling, climate, water purification, remediation and restoration30. Moreover, earthworms can be used as bioindicators of various soil contaminations due to their sensitivity to pollutants31,32. Antioxidant enzymes in earthworms, which protect cells against adverse effects of reactive oxygen species (ROS), are used as indicators for evaluating the environmental effect of soil pollutants33,34,35. As a sensitive cellular biomarker, lysosomal membrane stability can provide valuable information on cellular damage36. Among earthworms, Eisenia fetida is sensitive to various toxicants and can be cultured easily under laboratory conditions. So it has been used as a standard animal for toxicology experiment37. In the present study, E. fetida was used for assessing the toxicological effects of MCs. In addition, biochemical responses in earthworms can be used as early warning indicators of soil contamination.
Cyanobacterial blooms have become more frequent in recent years in many large freshwater lakes and reservoirs. Surface water containing MCs used as irrigation source were reported all over the world, including but not limited to China16, India38, Australia39, United States40 and Finland12. Consequently, it is possible that large areas of farmland near contaminated lakes are exposed to irrigation source water containing cyanobacteria blooms. In the present study, we measured mortality, avoidance responses, bioaccumulation, biochemical responses, and lysosomal membrane stability in earthworms exposed to different concentrations of cyanobacteria crude extract. These findings help to give a better understanding of the potential environmental risk of irrigation with cyanobacteria bloom into agricultural soils.
Results
Filter paper assay
There was no significant effect of microcystins on earthworms with filter paper test. Survival was 100% in all treatments after the 72 h exposure. In addition, earthworm weight was not significantly affected by MCs concentration (ANOVA, p > 0.05; data not shown), nor were morphological abnormalities observed in any of the treatments.
Avoidance responses
Earthworm net avoidance response was not significantly affected by MCs treatment in soils. (ANOVA, p > 0.05; data not shown).
Bioaccumulation
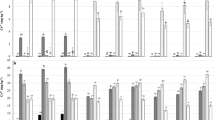
There was no evidence of MCs bioaccumulation in earthworms from control and 1 μg kg−1 treatments. For 10, 100 and 1000 μg kg−1 treatments, MCs were not detected on day 1, but increased along with increasing MCs dose on days 7 and 14 (Table 1). A sharp increase was observed from day 1 to day 14 for 100 and 1000 μg kg−1 treatments. MCs level in 10 μg kg−1 treatment increased relatively slow. At days 14, MC concentrations in earthworms were 0.18, 0.40 and 0.63 μg g−1 in 10, 100 and 1000 μg kg−1 treatments, respectively. MCs in artificial soils were also determined. After 14 d exposure, MCs content in soils decreased by 16%, 11%, 17% and 19% at 1, 10, 100 and 1000 μg kg−1 treatments, respectively (data not shown).
Biochemical responses
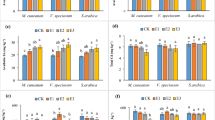
The treatment with 1 µg MCs kg−1 did not affect CAT activity regardless of exposure time. However, CAT activity responded quickly to the application of higher concentrations of MC; CAT activity at 10, 100, and 1,000 μg kg−1 increased significantly after 1 d (Fig. 1A). However, after a 7 d exposure, the only CAT activity that was significantly different from the controls was the 10 μg kg−1 treatment, similar to day 1 response. After 14 days of exposure, CAT activity at 10 μg kg−1 was still statistically greater than the controls, but significantly lower values of CAT activity were found at 100 and 1,000 μg kg−1.
The effect of MCs on GPx activity was concentration and time-dependent (Fig. 1B). Both 1 and 10 μg kg−1 treatments had no effect on GPx activity compared to controls, regardless of exposure time. However, GPx activity in the 1,000 μg kg−1 treatment was significantly higher than the controls after 1 d exposure, but then was significantly lower at days 7 and 14. GPx activity at 100 μg kg−1 was not significantly different from the control at day 1, but similar to the 1,000 μg kg−1 treatment, and was significantly lower than the controls at days 7 and 14.
SOD activity was relatively insensitive to MCs application (Fig. 1C). SOD activity at 100 μg kg−1 was enhanced after 7 d, while it was significantly inhibited at 1,000 μg kg−1 after 14 d compared to the controls. No significant differences were found in the other treatments.
The MDA content of earthworms was not significantly affected after a 1 d exposNure, while significant increases were measured in 100 μg kg−1 and 1,000 μg kg−1 treatments after 7 and 14 d of exposure (Fig. 1D). The MDA content at the two highest MC concentrations on days 7 and 14 was almost two times higher than the controls.
Significant influence of dose and duration of exposure on all studied biochemical responses was revealed by multivariate analysis. The CAT and GPx activities and MDA content were significantly affected by both dose and duration of exposure, while SOD activity wasonly affected by duration (Table 2).
Lysosomal membrane stability
MCs had a negative effect on the NRRT of earthworms at the highest concentration on day 1, but no other concentration had a significant effect compared to controls on that day (Fig. 2). A significant reduction in NRRT was observed with both 100 and 1,000 μg kg−1 treatments on days 7 and 14, with reductions of 45% and 62%, respectively.
Discussion
In this study, we found no evidence that MCs are acutely toxic to Eisenia fetida. There was no mortality or morphological changes even at an exposure of 10,000 μg L−1 MCs. Furthermore, no avoidance response was found in any of the MC-treated soils. In contrast to our findings, Wen et al.26 reported that the LC50 of MC-LR was 0.149 μg cm−1 at 72 h based on a filter paper test on Eisenia fetida. The explanation for such different might be due to the earthworms (300–400 mg) used in their study is smaller than ours (~500 mg), and MCs are more toxic to younger earthworms. Toxic effects of MCs on aquatic animals have also been reported before. Oberemm et al.41 found a dose-dependent increase of mortality rate of chub (Leuciscus cephalus) after exposure to 0.5, 5, or 50 μg MC-RR or MC-LR L−1. Liu et al.42 also reported mortality (LC50 = 593.3 μg/L) of juvenile loach (Misguruns mizolepis) after exposure to 1, 3, 10, 100, or 1,000 μg MC-LR L−1 after 7 d. Various abnormalities, as well as dose- and time-dependent survival rate was found after micro injection of 300, 750, and 900 nM MC-LR into zebrafish (Danio rerio)43. However, some animals such as mollusks showed tolerance to cyanotoxins. Vasconcelos44 reported that less than 1% mortality of mussel (Mytilus galloprovincialis) was found after fed on a toxic M. aeruginosa strain for 16 days. During this period mussels accumulated up to 10.5 µg g−1 MCs dry mussel weight. Anodonta cygnea was also found to be able to accumulate high levels of microcystins produced by Oscillatoria agardii HII without any visible damage45. There are no specific carriers that carry MCs, which are relatively lipophobic, into hepatopancreatic cells46. Moreover, MCs used in the present was cyanobaterial extract contained mainly MC-RR, which is less toxic than the most common congener MC-LR in a mice test47. Thus, toxicity of different MC congeners in earthworms should be explored in the future. Besides, in view of the toxicity of MCs to aquatic animals, short- and long-term effects of MCs on other soil organisms should also be evaluated. Our results showed that concentrations of MCs in earthworms were high when MC concentrations in treatments were above 10 μg kg−1, suggesting that MCs may bioaccumulate in earthworms. With less microbes and no light in the artificial condition, which are main factors for MCs degradation1, MCs degradation in artificial soil is much lower than that in natural soil. Degradation of MCs in the artificial soil is less than 20% at the end of the exposure experiment. So the bioaccumulation of MCs in earthworms in natural soil should be re-evaluated due to different soil type and different quantity of MCs. Moreover, we were unable to completely remove earthworm gut contents, even when gut contents of earthworms were voided with wet filter paper for 24 h after exposure. Hence, we suggest that the biological responses (such as response of the antioxidant system) observed in our study are better indicators of impacts from MCs than tissue levels.
The biochemical indicators varied with doses of MCs and duration of exposure. One of the toxicity mechanisms of MCs to animals is oxidative stress48. When cellular antioxidant defense fail to remove excessive ROS, oxidative stress results in organisms49. CAT, which eliminates hydrogen-peroxide, plays an important role in anti-oxidant systems. In our study, CAT activity in E. fetida was sensitive to MCs exposure. A significant increase was found at 10, 100, 1,000 μg kg−1 after only 1 d exposure, indicating that MCs lead to oxidative stress in this earthworm species50. However, the stimulatory effect of 100 and 1,000 μg kg−1 in our study disappeared after 7 d, with a significant decrease observed after 14 d exposure. The same time-dependent effect was found in crabs, Pinho et al.51 showed that CAT activity increased after exposed to 5.32 mg MCs kg−1 day−1 for 2 days, whereas decreased at day 7. A possible explanation is that the production of H2O2 at the early stage may induce an increase of CAT activity, whereas the natural antioxidant defenses may have become saturated as incubation time lengthened, resulting in accumulation of ROS in earthworms, thereby inhibiting CAT activity52,53. As antioxidant system vary with animal species, different result was found in freshwater clam Diplodon chilensis patagonicus, in which a continuous stimulatory effect of CAT was observed after exposed to an estimated mean weekly dose of 0.625 mg MCLR g−1 clam for six weeks54. Leão et al.55 found decreased CAT activity in an estuarine worm (Laeonereis acuta) after 48 h exposure to 2 mg mL−1 lyophilized cells of toxic M. aeruginosa. Moreover, we detected all the biochemical indicators with whole earthworm tissue with no distinction of different organs. Lower CAT level was found in gills than other organs (liver and kidney) of tilapia56. So this may raise questions on the difference of antioxidant response in different organs. Therefore, alternation of antioxidant enzyme activities in different earthworm organs after exposed to MCs should be studied in the future.
GPx is another enzyme that can remove H2O2 in organisms by using reduced glutathione as a hydrogen donor. Similar to CAT activity, a stimulatory effect was observed at 1,000 μg kg−1 after 1 d exposure, and a significant inhibition at 100 and 1,000 μg kg−1 began after 7 d exposure, which continued through day 14. Stimulatory effect of GPx activity was also observed in embryos of zebrafish exposed to 0.5 μg L−1 MC-LR57, and in hepatocytes of C. carpio exposed to 10 μg L−1 MC-LR58. However, GPx activity was increased in liver and intestine of Corydoras paleatus exposed to 2 μg L−1 MC-RR, whereas strongly inhibited in gills at all tested concentrations59. Same differential impact vary with organs was found in tilapia56. Similar to CAT, as previously discussed, antioxidant response in different earthworm organs should be re-evaluated. Interestingly, same as our results, a previous study on earthworms60 also found that GPx activity was induced by naphthenic acids after 1 d but inhibited after 14 d of exposure. The reasons might be that (i) GPx activity was inhibited by excessive ROS in cells; and (ii) large amounts of glutathione (GSH) were consumed to remove ROS.
SOD also plays a vital part in eliminating ROS. The ·superoxide anion radical (O2 −) in the cell is transformed to H2O2 by SOD and then H2O2 is transformed to harmless H2O and O2 by CAT and GPx under oxidative stress61,62. Unlike CAT and GPx activity, no significant change was found after 1 d in our study. A stimulatory effect was found only at 100 μg kg−1 after 7 d, which probably was caused by O2 − accumulation51. Inhibition was found at 1,000 μg kg−1 after 14 d. The possible reason is that hydrogen peroxide, peroxyl radicals and singlet oxygen induce inactivation of SOD or the highly reactive superoxide was eliminated63,64,65,66. Increase as well as decrease of SOD activity in organs after treated with MCs were found by previous studies56,67,68,69. Similar to CAT and GPx activities, SOD activity varies with animal species.
MDA results when unsaturated fatty acids react with free radicals in cellular membranes. It is used frequently as a sensitive indicator that reflects lipid peroxidation and indicates intracellular injury70. Various contaminants are known to induce lipid peroxidation due to excessive ROS71. In our study, MDA content in earthworms was significantly higher than that observed in controls after 7 d and 14 d of exposure to higher concentrations of MC (100 and 1,000 μg kg−1). These results suggest that MCs lead to lipid peroxidation and induce oxidative injury in earthworms.
Oxidative stress in animals and plants caused by MCs has been reported in recent years. Generation of ROS and disruption of mitochondrial electron transport chain were found in MC-treated rats48. Oxidative stress caused by excessive ROS production was found in several MC-treated aquatic and terrestrial organisms, and an increase in lipid peroxidation was found as well51. Regarding plants, SOD activity in rape (Brassica napus L.) decreased as toxin concentration increased, while activity was stimulated in rice (Oryza sativa L.)72. Stimulatory effects of MCs on SOD and GPx activities also were found by Chen et al.10 and Gehringer et al.73 in apple trees (Malus pumila) and garden cress (Lepidium sativum), respectively. Oxidative stress was found in several MC-treated animals and plants, changes in antioxidative activity varying with time and tested species.
Our results demonstrated that MCs can induce oxidative stress in earthworms, as observed in other animals and plants, which can lead to the disruption of the antioxidant system and lipid peroxidation. Nevertheless, the duration of our study was only 14 d; we recommend that earthworm responses to MCs be evaluated over longer time periods and in natural conditions. Generally, impaired lysosomal reactions precede cell and tissue pathology; therefore, lysosomal systems can respond quickly to pollutant exposure74. LMS is an early indicator of various environmental contaminants, and LMS in earthworm coelomocytes is often evaluated by the NR retention assay32,36,75. In the present study, high concentrations of MC significantly decreased LMS in earthworm coelomocytes. A same inhibition effect of NRRT was reported by Wen et al.26 in MC-LR treated earthworms. However, the mechanism causing alternations in LMS is still unknown. Evidence suggests that ROS produced in the cellular system affects various cellular organelles76,77. Consequently, generation of ROS induced by MCs may be the cause of alterations in LMS. This putative mechanism should be explored in future studies to better understand the impact of MCs on LMS on earthworm coelomocytes.
Soils irrigated with cyanobacteria-containing water may suffer varying degrees of MCs contamination, which could pose a threat to earthworms in the soil. Although no significant effect on earthworms was observed at low MC concentrations treatment, effects on earthworms after long-time and/or repeatedly contact with MCs are still unknown. In the artificial soil test, earthworms are cultivated in artificial soil according to OECD78. Although the composition of artificial soil was set to mimic natural soil, the microbial composition of artificial soil is much different from that in natural soil. Moreover microorganisms play important roles in degrading MCs in soils. The implication for natural soil systems is unclear, so field experiments are needed in the future.
Materials and Methods
Earthworms
Earthworms were purchased from a local earthworm farm in Nanjing. Adult earthworms weighing about 500 mg with well-developed clitella were selected. To void gut contents, earthworms were placed on damp filter paper in Petri dishes for 24 h before testing.
Extraction and analysis of microcystin variants
In the present study, we used a natural cyanobacterial bloom extract so as to mimic natural toxic conditions with multiple MCs. In order to prevent interference from impurities, the extract was purified with an Oasis HLB extraction cartridge (Waters). The method of extraction and purification was modified from Harada et al.79. Cyanobacterial cells obtained from Dianchi Lake (~1 g DW) were homogenized with 25 mL of 5% (v/v) aqueous acetic acid after freeze-drying. The homogenate was subjected to an ultrasonic bath for 5 min, and then centrifuged at 10,000 r min−1 at 4 °C for 15 min. After re-extracting the residue two more times as before, all the supernatant was collected and then applied to 5 g HLB extraction cartridge. The cartridge with toxin was rinsed with 50 mL of 5% (v/v) aqueous methanol. Subsequently, the cartridge was eluted with 100 mL 100% aqueous methanol. The eluant was evaporated to dryness, and then 10 mL deionized water was used to dissolve the toxin. Combined toxin-containing solutions were stored at −40 °C before use. According to Corbel et al.80, microcystin variants were analyzed by ultra-high-performance liquid chromatography tandem mass spectrometry (UHPLC/MS), using a Waters Acquity UPLC system coupled to a triple-quadruple mass spectrometer (TQD, Waters, France) via an electrospray ionization (ESI) interface. MC-LR (14.92 mg L−1), MC-YR (5.90 mg L−1) and MC-RR (61.61 mg L−1) were found in the extract after the analysis. The toxin extract was diluted as needed for subsequent use.
Filter paper acute toxicity test
The acute toxicity test was conducted according to OECD guidelines78,81. Considering there are no previous studies reporting the toxicity of MCs on earthworms, a wide range of MC concentrations was used for the acute toxicity test (MCs concentration, 0.1, 1, 10, 100, 1,000, 10,000 μg L−1). After placing single pieces of filter paper on the bottom of Petri dishes, 1 mL of each MC treatment concentration or deionised water (controls) was added to wet the entire filter paper. Then one earthworm was randomly placed in each Petri dish. Five replicates were used for each treatment. The Petri dishes were incubated in the dark at 20 ± 1 °C for 72 h and the mortality was recorded every 24 h; earthworm weight and morphological changes also were observed.
Avoidance test
According to ISO guideline82, avoidance tests were performed in plastic containers with three replicates for each treatment. Soils used in the test was collected (0–15 cm) from an area adjacent to Lake Taihu at Suzhou, with no known previous history of MCs application. Some of the physicochemical properties are as follows: 27.9% sand, 31.0% silt, 41.2% clay, pH 7.46, 9.91 g organic matter kg−1 soil, 0.78 g total nitrogen kg−1 soil, 15.4 mg available P kg−1 soil, 150 mg available K kg−1 soil. After taken back to the laboratory, the soil samples were homogenized and passed through a 2-mm sieve to sift out roots and other large debris. The soil samples were used for physico-chemical analysis after air-dried at 25 °C in 48 h. The remainders were kept fresh at 4 °C for avoidance test. Contaminated soil was produced by thoroughly mixing soil and MCs containing deionized water with final treatment concentrations of 1, 10, 100, 1,000 μg kg−1 dry soil. Control soil received an equal amount of deionized water. All treatments were adjusted to 60% of maximum water-holding capacity. Briefly, the container was filled half with contaminated soil and half with control soil at each end. After placing ten earthworms on the soil surface along the transition line between control and contaminated soil in each test container, perforated plastic films were used to cover the containers. The containers were incubated in the dark at 20 ± 1 °C for 48 h. The numbers of earthworms in the control vs. contaminated sections of each container were recorded after incubation. The net avoidance response (NR) was determined with the following formula: NR = (C − T)/N × 100%, where C, T, and N represent the numbers of earthworms in the control soil (C), the contaminated treatment soil (T), and the total number of earthworms (N), respectively.
Artificial soil test
A 14-day toxicity test was performed in artificial soil according to OECD78 guidelines. The artificial soil was composed of 10% finely ground sphagnum peat, 20% kaolin clay and 70% industrial sand with 35% water content and 7.0 ± 0.2 pH value. Before the test, earthworms were acclimated for one week in untreated artificial soil at a controlled temperature of 20 ± 1 °C with 80–85% relative humidity under 400–800 lux of continuous light. Different amount of MCs were added in the soil to make a final MC concentrations of 1, 10, 100, 1,000 μg kg−1 dry soil. The control soil received an equivalent amount of distilled water. The MCs concentrations in the artificial soil prior to the exposure were 0.65, 8.47, 90.1 and 958.3 μg kg−1 dry soil. Twelve earthworms were added to each 1 L wide-mouth bottle filled with 500 g contaminated soil. Each soil treatment included three replicates. The bottles were sealed with porous plastic film, allowing exchange of air, and incubated at 20 ± 1 °C with 80–85% relative humidity under 400–800 lux of continuous light for 14 days. Four earthworms were randomly collected on days 1, 7, and 14 following application of MCs. Prior to analysis, the gut contents of all the earthworms were voided as mentioned above. No mortality was observed during the experiment.
Analysis of MCs in earthworms
Earthworms were lyophilized after voiding gut contents. MCs in lyophilized earthworms were determined as described by Xie and Park83. The limit of detection (LOD) for MC-LR, MC-RR and MC-YR are 0.022, 0.024 and 0.030 μg kg−1, respectively. The recovery rate ranged from 83.5% to 97.5% (RSD ranged from 0.1%–2.3%). Briefly, lyophilized earthworms were homogenized with 10 mL of BuOH:MeOH:H2O (1:4:15, in volume) after ground with a mortar. The homogenate was subjected to an ultrasonic bath for 3 min, and then centrifuged at 4,000 r min−1 at room temperature for 20 min. After re-extracting the residue two more times as before, all the supernatant was collected and then applied to 500 mg Oasis HLB extraction cartridge. The cartridge with toxin was rinsed with 20 mL of 5% (v/v) aqueous methanol. Subsequently, the cartridge was eluted with 15 mL 100% aqueous methanol. The eluant was evaporated to dryness, and then 2 mL 100% aqueous methanol was used to dissolve the toxin. The dissolved toxin was then applied to a silica gel cartridge (2 g)/plus silica gel (0.69 g) tandem cartridge. The cartridge with toxin was rinsed with 20 mL of 100% aqueous methanol, and eluted with 20 mL 70% aqueous methanol. The eluant was evaporated to dryness and the residue was dissolved in 0.1 mL 100% aqueous methanol. The toxin solutions obtained were stored at −40 °C for UHPLC/MS detection.
Analysis of MCs in artificial soils
The artificial soil was lyophilized after collected at the end of the test. MCs in soils were determined as described by Li et al.20 with slight modification. The LOD for MC-LR, MC-RR and MC-YR are 0.25, 0.25 and 0.50 μg kg−1, respectively. The recovery rate ranged from 54.9% to 97.4% (RSD ranged from 4.3%−16.9%). Briefly, lyophilized soil samples were extracted 3 times with 30 mL of 0.1 M EDTA-0.1 M Na4P2O7 with 10 min ultrasonic bath after ground with a mortar. The homogenate was then centrifuged at 4,000 g for 10 min. After turning the pH value of the supernatant to pH 3 with TFA, the solution was centrifuged again with the same condition. Then the aqueous extractions were applied to an Oasis HLB extraction cartridge. The cartridge with toxin was rinsed with 15 mL of 20% (v/v) aqueous methanol, and eluted with 10 mL 90% aqueous methanol. The eluant was evaporated to dryness and the residue was dissolved in 0.1 mL 100% aqueous methanol. The toxin solutions obtained were stored at −40 °C for UHPLC/MS detection.
Biochemical assays
Enzymes were extracted according to Mishra and Dash84. Earthworms were ground under ice-cold conditions in 0.85% NaCl solution (1:9, w/v) with a prechilled mortar. The homogenate was centrifuged at 3,000 rpm at 4 °C for 10 min. The supernatants were used for various analyses. Several test kits (Nanjing Jiancheng, China) including Catalase (CAT) assay kit (Ammonium molybdate method), Glutathione Peroxidase (GPx) assay kit (Colorimetric method), Total Superoxide Dismutase (T-SOD) assay kit (Hydroxylamine method), Total protein quantitative assay kit and Malondialdehyde (MDA) assay kit (TBA method) were used to measure CAT activity, GPx activity, total SOD activity, protein content and MDA content, respectively.
Neutral red retention assay
The non-invasive extrusion method was used to obtain earthworm coelomocytes85. The extrusion medium (10 mg ml−1 guaiacol glyceryl ether, 0.85% saline, 5% ethanol, 2.5 mg ml−1 EDTA,) was used to rinse earthworms. Coelomocytes secreted in the medium were washed three times with 100 mM PBS (pH 7.3) to remove mucous. Neutral red (NR) retention assay was used to evaluate lysosomal membrane stability of coelomocytes as described by Lowe and Pipe86.
Statistical analysis
SPSS software (SPSS 22.0, Inc., 2013) was used for statistical analyses. Data were expressed as mean ± SD. One-way ANOVA followed by the Duncan test were used to test for differences between different exposure levels and the control. Means of biochemical indicators were compared by two-way ANOVA with dose and duration treatments as independent variables. The criteria for significance were set at p < 0.01 and p < 0.05.
Data availability
All data generated or analysed during this study are included in this published article (and its Supplementary Information files).
Conclusion
The results obtained from various tests and multiple biomarkers lead us to conclude that: (1) MCs do not induce acute toxicity in earthworms; (2) earthworms showed no avoidance response to MCs exposure; (3) MCs were bioaccumulated in earthworms after exposed to high concentrations of MCs; and (4) MCs induced oxidative stress in earthworms, which lead to disruption of the antioxidant system and lipid peroxidation, as well as alterations in LMS. More studies are encouraged to figure out the thorough impact of MCs on soil animals.
References
Rastogi, R. P., Sinha, R. P. & Incharoensakdi, A. The cyanotoxin-microcystins: current overview. Rev. Environ. Sci. Biotechnol. 13, 215–249 (2014).
Wu, Q. et al. Microcystin-LR exposure induces developmental neurotoxicity in zebrafish embryo. Environ. Pollut. 213, 793–800 (2016).
Wu, Q. et al. Parental transfer of microcystin-LR induced transgenerational effects of developmental neurotoxicity in zebrafish offspring. Environ. Pollut. 231, 471–478 (2017).
Cheng, H. et al. Parental exposure to microcystin-LR induced thyroid endocrine disruption in zebrafish offspring, a transgenerational toxicity. Environ. Pollut. 230, 981–988 (2017).
Spoof, L. & Catherine, A. Appendix 3: Tables of microcystins and nodularins in Handbook of cyanobacterial monitoring and cyanotoxin analysis (eds Meriluoto, J. Spoof, L. Codd, G. A.) 526–537 (John Wiley & Sons, Ltd, Chichester, 2017).
Zastepa, A., Pick, F. R., Blais, J. M. & Saleem, A. Analysis of intracellular and extracellular microcystin variants in sediments and pore waters by accelerated solvent extraction and high performance liquid chromatography-tandem mass spectrometry. Anal. Chim. Acta 872, 26–34 (2015).
Chen, L., Chen, J., Zhang, X. Z. & Xie, P. A review of reproductive toxicity of microcystins. J. Hazard. Mater. 301, 381–399 (2016).
Liu, B. B., Gong, Y., Xiao, B. D., Liu, J. T. & Liu, Y. D. A laboratory study on risk assessment of microcystin-RR in cropland. J. Environ Manage. 86, 566–574 (2008).
Saqrane, S. et al. Physiological changes in Triticum durum, Zea mays, Pisum sativum and Lens esculenta cultivars, caused by irrigation with water contaminated with microcystins: a laboratory experimental approach. Toxicon 53, 786–796 (2009).
Chen, J. et al. Bioaccumulation of microcystin and its oxidative stress in the apple (Malus pumila). Ecotoxicology 19, 796–803 (2010).
Chen, J., Han, F. X., Wang, F., Zhang, H. & Shi, Z. Accumulation and phytotoxicity of microcystin-LR in rice (Oryza sativa). Ecotoxicol. Environ. Saf. 76, 193–199 (2012).
Spoof, L., Vesterkvist, P., Lindholm, T. & Meriluoto, J. Screening for cyanobacterial hepatotoxins, microcystins and nodularin in environmental water samples by reversed-phase liquid chromatography–electrospray ionisation mass spectrometry. J. Chromatogr. A 1020, 105–119 (2003).
Billam, M. et al. Seasonal variations in the concentration of microcystin-LR in two lakes in western Texas, USA. Environ. Toxicol. Chem. 25, 349–355 (2006).
Nasri, H., El Herry, S. & Bouaïcha, N. First reported case of turtle deaths during a toxic Microcystis spp. bloom in Lake Oubeira. Algeria. Ecotox. Environ. Safe. 71, 535–544 (2008).
Giannuzzi, L., Sedan, D., Echenique, R. & Andrinolo, D. An acute case of intoxication with cyanobacteria and cyanotoxins in recreational water in Salto Grande Dam, Argentina. Mar. Drugs 9, 2164–2175 (2011).
Liu, Y. et al. Cyanobacteria-/cyanotoxin-contaminations and eutrophication status before Wuxi drinking water crisis in Lake Taihu, China. J. Environ. Sci. China 23, 575–581 (2011).
Lindholm, T. & Meriluoto, J. A. O. Recurrent depth maxima of the hepatotoxic cyanobacterium Oscillatoria agardhii. Can. J. Fish. Aquat. Sci. 48, 1629–1634 (1991).
Jones, G. J. & Orr, P. T. Release and degradation of microcystin following algicide treatment of a Microcystis aeruginosa bloom in a recreational lake, as determined by HPLC and protein phosphatase inhibition assay. Water Res. 28, 871–876 (1994).
Lahti, K., Rapala, J., Färdig, M., Niemelä, M. & Sivonen, K. Persistence of cyanobacterial hepatotoxin, microcystin-LR in particulate material and dissolved in lake water. Water Res. 31, 1005–1012 (1997).
Li, Y. W. et al. Simultaneous extraction and determination of three microcystins from soil using solid phase extraction and liquid chromatography-tandem mass spectrometry. Chin. J. Anal. Chem. 41, 88–92 (2013).
Jones, G. J., Falconer, I. R. & Wilkins, R. M. Persistence of cyclic peptide toxins in dried Microcystis aeruginosa crusts from lake Mokoan, Australia. Environ. Toxic. Water 10, 19–24 (1995).
Chen, W., Li, L., Gan, N. Q. & Song, L. R. Optimization of an effective extraction procedure for the analysis of microcystins in soils and lake sediments. Environ. Pollut. 143, 241–246 (2006).
Morris, R. J. et al. The adsorption of microcystin-LR by natural clay particles. Toxicon 38, 303–308 (2000).
Miller, M. J. & Fallowfield, H. J. Degradation of cyanobacterial hepatotoxins in batch experiments. Water Sci. Technol. 43, 229–232 (2001).
Eynard, F., Mez, K. & Walther, J. L. Risk of cyanobacterial toxins in Riga waters (Latvia). Water Res. 34, 2979–2988 (2000).
Wen, H. F. et al. Toxicological effects of microcystin-LR on earthworm (Eisenia fetida) in soil. Biol. Fertil. Soils. 2, 1–12 (2017).
Li, Y. H., Wang., Y., Yin, L. H., Pu, Y. P. & Wang, D. Y. Using the nematode Caenorhabditis elegans as a model animal for assessing the toxicity induced by microcystin-LR. J. Environ. Sci-China 21, 395–401 (2009).
Moore, C. E., Lein, P. J. & Puschner, B. Microcystins alter chemotactic behavior in Caenorhabditis elegans by selectively targeting the AWA sensory neuron. Toxins 6, 1813–1836 (2014).
Lána, J., Hofman, J. & Bláha, L. Can cyanobacterial biomass applied to soil affect survival and reproduction of springtail folsomia candida? Ecotox. Environ. Safe. 74, 840–843 (2011).
Blouin, M. et al. A review of earthworm impact on soil function and ecosystem services. Eur. J. Soil Sci. 64, 161–182 (2013).
Xiao, N., Jing, B., Ge, F. & Liu, X. The fate of herbicide acetochlor and its toxicity to Eisenia fetida under laboratory conditions. Chemosphere 62, 1366–1373 (2006).
Saint-Denis, M., Narbonne, J. F., Arnaud, C. & Ribera, D. Biochemical responses of the earthworm Eisenia fetida andrei exposed to contaminated artificial soil: effects of lead acetate. Soil Biol. Biochem. 33, 395–404 (2011).
Saint-Denis, M., Labrot, F., Narbonne, J. F. & Ribera, D. Glutathione, glutathione related enzymes, and catalase in the earthworm Eisenia fetida andrei. Arch. Environ. Contam.Toxicol. 35, 602–614 (1998).
Saint-Denis, M., Narbonne, J. F., Arnaud, C., Thybaud, E. & Ribera, D. Biochemical responses of the earthworm Eisenia fetida andrei exposed to contaminated artificial soil: effects of benzo(a)pyrene. Soil Biol. Biochem. 31, 1837–1846 (1999).
Łaszczyca, P. et al. Profiles of enzymatic activity in earthworms from zinc lead and cadmium polluted areas near Olkusz (Poland). Environ. Int. 30, 901–910 (2004).
Weeks, J. M. & Svendsen, C. Neutral-red retention by lysosomes from earthworm (Lumbricus rubellus) coelomocytes: a simple biomarker of exposure to soil copper. Environ. Toxicol. Chem. 15, 1801–1805 (1996).
OECD, Earthworm, Acute Toxicity Tests. Guideline for Testing Chemicals. No. 207. (OECD, Paris, 1984)
Prakash, S., Lawton, L. A. & Edwards, C. Stability of toxigenic Microcystis blooms. Harmful Algae 8, 377–384 (2009).
Everson, S., Fabbro, L., Kinnear, S. & Wright, P. Extreme differences in akinete, heterocyte and cylindrospermopsin concentrations with depth in a successive bloom involving Aphanizomenon ovalisporum (Forti) and Cylindrospermopsis raciborskii (Woloszynska) Seenaya and Subba Raju. Harmful Algae 10, 265–276 (2011).
Miller, M. A. et al. Evidence for a novel marine harmful algal bloom: cyanotoxin (microcystin) transfer from Land to Sea Otters. Plos One 5, e12576 (2010).
Oberemm, A., Becker, J., Codd, G. & Steinberg, C. Effects of cyanobacterial toxins and aqueous crude extracts on the development of fish and amphibians. Environ Toxicol. 14, 77–88 (1999).
Liu, Y., Song, L., Li, X. & Liu, T. The toxic effects of MC-LR on embryo-larval and juvenile development of loach, Misguruns mizolepis Gunthe. Toxicon 40, 395–399 (2002).
Wang, P. J., Chien, M. S., Wu, F. J., Chou, H. N. & Lee, S. J. Inhibition of embryonic development by microcystin-LR in zebrafish. Danio rerio. Toxicon 45, 303–308 (2005).
Vasconcelos, V. M. Uptake and depuration of the peptide toxin microcystin-LR in the mussel Mytilus galloprovincialis. Aquat. Toxicol. 32, 227–237 (1995).
Eriksson, J. E., Meriluoto, J. A. O. & Lindholm, T. Accumulation of a peptide toxin from the cyanobacterium Oscillatoria agardhii in the fresh water mussel Anodonta cygnea. Hydrobiologia 183, 211–216 (1989).
Vasconcelos, V. M. Cyanobacterial toxins in Portugal: effects on aquatic animals and risk for human health. Braz. J. Med. Biol. Res. 32, 249–254 (1999).
Gupta, N., Pant, S. C., Vijayaraghavan, R. & Rao, P. V. L. Comparative toxicity evaluation of cyanobacterial cyclic peptide toxin microcystin variants (LR, RR, YR) in mice. Toxicology 188, 285–296 (2003).
Ding, W. X., Shen, H. M. & Ong, C. N. Calpain activation after mitochondrial permeability transition in microcystin induced cell death in rat hepatocytes. Biochem. Biophys. Res. Commun. 291, 321–331 (2002).
Limon-Pacheco, J. & Gonsebatt, M. E. The role of antioxidants and antioxidant-related enzymes in protective responses to environmentally induced oxidative stress. Mutat. Res. 674, 137–147 (2009).
Di Giulio, R. T., Washburn, P. C., Wenning, R. J., Winston, G. W. & Jewell, C. S. Biochemical responses in aquatic animals: a review of determinants of oxidative stress. Environ. Toxicol. Chem. 8, 1103–1123 (1989).
Pinho, G. L. L. et al. Antioxidant responses and oxidative stress after microcystin exposure in the hepatopancreas of an estuarine crab species. Ecotoxicol. Environ. Saf. 61, 353–360 (2005).
Song, Y. et al. DNA damage and effects on antioxidative enzymes in earthworm (Eisenia foetida) induced by atrazine. Soil Biol. Biochem. 41, 905–909 (2009).
Wu, S. J., Wu, E. M., Qiu, L. Q., Zhong, W. H. & Chen, J. M. Effects of phenanthrene on the mortality, growth, and anti-oxidant system of earthworms (Eisenia fetida) under laboratory conditions. Chemosphere 83, 429–434 (2011).
Sabatini, S. E. et al. Microcystin accumulation and antioxidant responses in the freshwater clam Diplodon chilensis patagonicus upon subchronic exposure to toxic microcystis aeruginosa. Ecotoxicol. Environ. Saf. 74, 1188–1194 (2011).
Leão, J. C., Geracitano, L. A., Monserrat, J. M., Amado, L. L. & Yunes, J. S. Microcystin-induced oxidative stress in Laeonereis acuta (Polychaeta, Nereididae). Mar Environ Res. 66, 92–4 (2008).
Jos, A. et al. Toxic cyanobacterial cells containing microcystins induce oxidative stress in exposed tilapia fish (Oreochhromis sp.) under laboratory conditions. Aquat. Toxicol. 72, 261–271 (2005).
Wiegand, C. et al. Uptake and effects of microcystin-LR on detoxication enzymes of early life stages of the zebra fish (Danio rerio). Environ. Toxicol. 14, 89–95 (1999).
Li, X., Liu, Y., Song, L. & Liu, J. Responses of antioxidant systems in the hepatocytes of common carp (Cyprinus carpio L.) to the toxicity of MC-LR. Toxicon 42, 85–89 (2003).
Cazenave, J., Bistoni, M. d. l. A., Pesce, S. F. & Wunderlin, D. A. Differential detoxification and antioxidant response in diverse organs of Corydoras paleatus experimentally exposed to microcystin-RR. Aquat. Toxicol. 76, 1–12 (2006)
Wang, J. et al. Oxidative damage of naphthenic acids on the Eisenia fetida earthworm. Environ. Toxicol. 31, 1337–1343 (2016).
Velki, M. & Hackenberger, B. K. Inhibition and recovery of molecular biomarkers of earthworm Eisenia andrei after exposure to organophosphate dimethoate. Soil Biol. Biochem. 57, 100–108 (2013).
Lin, D., Xie, X., Zhou, Q. X. & Liu, Y. Biochemical and genotoxic effect of triclosan on earthworms (Eisenia fetida) using contact and soil tests. Environ. Toxicol. 27, 385–392 (2012).
Hodgson, E. K. & Fridovich, I. The interaction of bovine erythrocyte superoxide dismutase with hydrogen peroxide: inactivation of the enzyme. Biochemistry 14, 5294–5299 (1975).
Escobar, J. A., Rubio, M. A. & Lissi, E. A. SOD and catalase inactivation by singlet oxygen and peroxyl radicals. Free Radical Biol. Med. 20, 285–290 (1996).
Company, R. et al. Effect of cadmium, copper and mercury on antioxidant enzyme activities and lipid peroxidation in the gills of the hydrothermal vent mussel Bathymodiolus azoricus. Mar. Environ. Res. 58, 377–381 (2004).
Sandrini, J. Z. et al. Antioxidant responses in the nereidid Laeonereis acuta (Annelida, Polychaeta) after cadmium exposure. Ecotoxicol. Environ. Saf. 70, 115–120 (2008).
Li, X. Y., Chung, I. K., Kim, J. I. & Lee, J. A. Oral exposure to Microcystis increases activity augmented antioxidant enzymes in the liver of loach (Misgurnus mizolepis) and has no effect on lipid peroxidation. Comp. Biochem. Physiol. C 141, 292–6 (2005).
Prieto, A. I., Jos, A., Pichardo, S., Moreno, I. & Cameán, A. M. Differential oxidative stress responses to microcystins LR and RR in intraperitoneally exposed tilapia fish (Oreochromis sp.). Aquat. Toxicol. 77, 314–21 (2006).
Prieto, A. I., Pichardo, S., Jos, Á., Moreno, I. & Cameán, A. M. Time-dependent oxidative stress responses after acute exposure to toxic cyanobacterial cells containing microcystins in tilapiafish (Oreochromis niloticus) under laboratory conditions. Aquat. Toxicol. 84, 337–45 (2007).
Grundy, J. E. & Storey, K. B. Antioxidant defenses and lipid peroxidation damage in estivating toads. Scaphiopus couchii. J. Comp. Physiol. 168, 132–142 (1998).
Krauss, M., Wilcke, W. & Zech, W. Availability of polycyclic aromatic hydrocarbons (PAHs) and polychlorinated biphenyls (PCBs) to earthworms in urban soils. Environ. Sci. Technol. 34, 4335–4340 (2000).
Chen, J., Song, L., Dai, J., Gan, N. & Liu, Z. Effects of microcystins on the growth and the activity of superoxide dismutase and peroxidase of rape (Brassica napus L.) and rice (Oryza sativa L.). Toxicon 43, 393–400 (2004).
Gehringer, M. M., Kewada, V., Coates, N. & Downing, T. G. The use of Lepidium sativum in a plant bioassay system for the detection of microcystin-LR. Toxicon 41, 871–876 (2003).
Moore, M. N., Icarus, A. J. & McVeigh, A. Environmental prognostics: an integrated model supporting lysosomal stress responses as predictive biomarkers of animal health status. Mar. Environ. Res. 61, 278–304 (2006).
Svendsen, C., Spurgeon, D. J., Hankard, P. K. & Weeks, J. M. A review of lysosomal membrane stability measured by neutral red retention: is it a workable earthworm biomarker? Ecotoxicol. Environ. Saf. 54, 20–29 (2004).
Vallyathan, V., Shi, X. & Castranova, V. Reactive oxygen species: their relation to pneumoconiosis and carcinogenesis. Environ. Health Perspect. 106, 1151–1155 (1998).
Squibb, K. S. & Fowler, B. A. Relationship between metal toxicity to sub cellular systems and the carcinogenic response. Environ. Health Perspect. 40, 181–188 (1981).
OECD, Guidelines for Testing of Chemicals No. 222: Earthworm Reproduction Tests (Eisenia foetida/Eisenia andrei). (OECD, Paris, 2004)
Harada, K. I. et al. Improved method for purification of toxic peptides produced by cyanobacteria. Toxicon 26, 433–439 (1988).
Corbel, S. et al. Evaluation of phytotoxicity and ecotoxicity potentials of a cyanobacterial extract containing microcystins under realistic environmental concentrations and in a soil-plant system. Chemosphere 128, 332–340 (2015).
OECD, Earthworm reproduction test (Eisenia fetida/andrei). In: Organization for Economic Cooperation and Development (Ed.), OECD Guideline for Testing Chemicals. Proposal for a New Guideline. (OECD, Paris, 2000)
ISO, Soil Quality-avoidance Test for Testing the Quality of Soils and Effects of Chemicals. Part 1. Test with Earthworms (Eisenia fetida and Eisenia andrei) Guideline. No. 17512e1. (ISO, Geneve, 2007)
Xie, L. Q. & Park, H.-D. Determination of microcystins in fish tissues using HPLC with a rapid and efficient solid phase extraction. Aquaculture 271, 530–536 (2007).
Mishra, P. C. & Dash, M. C. Digestive enzymes of some earthworms. Experientia 36, 1156–1157 (1980).
Eyambe, G. S., Goven, A. J., Fitzpatrick, L. C., Venables, B. J. & Cooper, E. L. A noninvasive technique for sequential collection of earthworm (Lumbricus terrestris) leukocytes during subchronic immunotoxic studie. Lab. Anim. 25, 61–67 (1991).
Lowe, D. M. & Pipe, R. K. Contaminant induced lysosomal membrane damage in marine mussel digestive cells: an in vitro study. Aquat. Toxicol. 30, 357–365 (1994).
Acknowledgements
This research was supported by Creative Research Groups of China (Grant No. 41621002), the Key Program of the Chinese Academy of Sciences (Grant No. ZDRW-ZS-2017-3-4).
Author information
Authors and Affiliations
Contributions
Q.C. wrote the main manuscript text. Q.C., A.D.S., L.Y. and L.X. designed and conducted the experiments and collected and analyzed the data. A.D.S. and L.X. checked and modified the manuscript text.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Cao, Q., Steinman, A.D., Yao, L. et al. Toxicological and biochemical responses of the earthworm Eisenia fetida to cyanobacteria toxins. Sci Rep 7, 15954 (2017). https://doi.org/10.1038/s41598-017-16267-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-16267-8
This article is cited by
-
Effects of diclofenac, sulfamethoxazole, and wastewater from constructed wetlands on Eisenia fetida: impacts on mortality, fertility, and oxidative stress
Ecotoxicology (2023)
-
Toxicity analysis and biomarker response of Quinalphos Organophosphate Insecticide (QOI) on eco-friendly exotic Eudrilus eugeniae earthworm
Environmental Monitoring and Assessment (2023)
-
Antioxidant responses and microcystins accumulation in Corbicula fluminea following the control of algal blooms using chitosan-modified clays
Journal of Soils and Sediments (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.