Abstract
Reference region selection is important for proper amyloid PET analysis, especially in subcortical vascular dementia (SVaD) patients. We investigated reference region differences between SVaD and Alzheimer’s disease (AD) using Centiloid scores. In 57 [C-11] Pittsburgh compound B (PiB) positive (+) AD and 23 PiB (+) SVaD patients, we assessed standardized PiB uptake and Centiloid scores in disease-specific cortical regions, with several reference regions: cerebellar gray (CG), whole cerebellum (WC), WC with brainstem (WC + B), pons, and white matter (WM). We calculated disease group differences from young controls (YC) and YC variance according to reference region. SVaD patients showed large effect sizes (Cohen’s d > 0.8) using all reference regions. WM and pons showed larger YC variances than other regions. Findings were similar for AD patients. CG, WC, and WC + B, but not WM or pons, are reliable reference regions for amyloid imaging analysis in SVaD.
Similar content being viewed by others
Introduction
The use of amyloid PET scans for quantitative measurement of amyloid-beta deposition has grown in recent years. As a result, the development and standardization of methods for amyloid-PET data analysis, particularly methods applicable across centers, has increasingly interested clinicians and investigators1,2,3. The Centiloid project, an effort to standardize quantitative amyloid-PET plaque estimation across centers, is one such development. The ratio of target to reference region is an effective method for calculating amyloid load, with sufficient discriminatory power between Alzheimer’s disease (AD) and matched controls2,4. This method is easy, useful, has obvious merit for clinical use as it requires no arterial blood sampling2, and has been adopted widely for semi-quantification of amyloid load. In probable AD, target to reference ratio is usually large and variance of the ratio matters little clinically2. However, there are sometimes subjects with borderline amyloid PET tracer uptake. Aβ deposition occurs on a continuum; at present there is no clear a priori way to separate individuals who have Aβ in the brain from those who do not. Therefore, small differences in quantification result according to the reference region used can present an issue especially in subjects with borderline uptake. Furthermore, when target-to-reference ratios are converted to the Centiloid scale, the values are increased, as the dynamic range of the Centiloid scale is greater than that of the target-to-reference ratio; consequently, choosing an inappropriate reference region may exaggerate errors in the Centiloid procedure especially in patients with borderline amyloid burden.
Cerebrovascular disease (CVD) and amyloid burden are the most frequent pathologies in cognitively impaired subjects5, and these two distinct pathologies present concurrently at a high rate. Mixed pathology is present in approximately half of all clinically diagnosed AD cases6,7,8,9, even in clinical trials with participants extensively screened for pure AD10. Conditions such as subcortical vascular dementia (SVaD), which exhibit both CVD and amyloid pathology, also require amyloid quantification; however, reference region selection in SVaD presents additional difficulties because pathology may occur within and impact measurement of certain reference regions.
According to Thal amyloid phase, pons and cerebellum are among the latest regions presenting amyloid pathology in AD11. The cerebellum has been used as a reference region in most previous studies using [C-11] Pittsburgh compound B (PiB) PET scans2,4,12. However, there is concern that cerebellum may not be suitable as a reference region for analyses of amyloid burden in conditions other than late-onset AD, as cerebellar amyloid deposits may be present in cerebral amyloid angiopathy (CAA)13, prion diseases14, and genetic AD15,16. Therefore, alternative reference regions such as pons have been evaluated for reliability4. Edison and colleagues reported that the pons is a reliable reference region for analysis in [C-11] PiB studies where cerebellum is not an appropriate reference region4. A recent study also suggested that WM as a reference region improved discrimination between clinically-defined groups17. However, the appropriateness of different reference regions for assessing amyloid uptake in patients with SVaD has not yet been investigated.
In this study, we investigated the use of different reference regions in analyzing amyloid uptake in SVaD patients. Given that SVaD patients affect more severe white matter (WM), brainstem and cerebellum than AD patients18,19,20, we hypothesized that there might be differences between SVaD and AD in the appropriateness of different reference regions. For testing the hypothesis, we used five reference regions cerebellar gray matter (CG), whole cerebellum (WC), WC with brainstem (WC + B), pons, and WM which have been frequently used in previous studies.
Results
Participant demographics
Table 1 shows demographic information of participants in this study. There were no differences in gender proportion between PiB (−) old control (OC), PiB (+) AD, and PiB (+) SVaD groups. However, ANOVA (F = 14.2, p < 0.001) and post-hoc tests indicated the PiB (+) AD group was younger than the PiB (−) OC (t = 3.4 and p = 0.001) and PiB (+) SVaD (t = 4.3 and p < 0.001) groups.
Global cortical target (CTX) ROIs in PiB (+) AD and PiB (+) SVaD
Figure 1 shows disease-specific CTX ROIs representing large differences in group-averaged PiB images between OC and patients (see the section of “Disease-specific CTX ROI”). SUVr of every voxel in the ROIs is significantly higher in patients (see Supplementary Fig. S1). Both CTXAD and CTXSVaD were mostly located in frontal, temporal and parietal regions. When evaluating proportions by lobe, CTXSVaD showed relatively larger proportions in parietal and lower proportions in temporal regions than CTXAD (Table 2).
Disease-specific cortical target regions. AD: Alzheimer’s disease; SVaD: subcortical vascular dementia. Colored areas indicate voxels by percent threshold (upper 3% intensity) of group-averaged difference between old control and patient groups, which was defined as disease-specific cortical target regions. The regions are mapped on MNI 152 template with the coordinates of the z-axis (numbers under each slice) to the MNI space.
Effect size of Centiloid (CL) values between disease groups and young control (YC) according to reference ROIs
The differences of CL between disease groups and YC are represented as effect sizes (Table 3). The effect sizes were large enough (Cohen’s d > 0.8) to show consistent group differences in all reference ROIs. The highest effect size of differences between PiB (+) AD and YC was generated using WM ROI (5.543) followed by pons (4.363), WC + B (4.200), WC (4.043) and CG (3.698). The highest effect size of differences between PiB (+) SVaD and YC was generated using WM ROI (4.721) followed by WC + B (3.957), WC (3.928), CG (3.851) and pons (3.615).
Variance of CL values according to reference ROIs in YC group
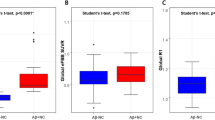
The results of assessing variance of CL values are shown in Table 3. The Bartlett’s statistics for equal variance indicated that there was within-group variability for all groups except SVaD patients; the results of post hoc tests are shown in Figs 2 and 3. The largest variances of CL values in YC were found using WM as reference region, followed in order by pons, WC + B, WC and CG (Table 3). Compared to other ROIs, WM and pons ROIs showed greater variance of CL values.
Post hoc variance tests of SUVr. Box plots display the SUVr distribution obtained by each reference. SUVr: standardized uptake value ratio; AD: Alzheimer’s disease; SVaD: subcortical vascular dementia; YC: young control; CG: cerebellar gray; WC: whole cerebellum; WC + B: WC with brainstem; WM: white matter. On each plot, the red line indicates the median (Q2), the bottom and top edges of the box represent lower quartile (Q1) and upper quartile (Q3). The outliers (red dots) are the values which falls outside of the lower/upper fences between 1.5 times the interquartile range (IQR, Q3–Q1) from Q1 and Q3, respectively. The black horizontal lines display minimum and maximum values in the fences.
Post hoc variance tests of Centiloid score. Box plots display the variances and differences of Centiloid scores within group. Lowercase letters indicate significant differences from CG (A), WC (B), WC + B (C), and pons (D). AD: Alzheimer’s disease; SVaD: subcortical vascular dementia; YC: young control; CG: cerebellar gray; WC: whole cerebellum; WC + B: WC with brainstem; WM: white matter. On each plot, the red line indicates the median (Q2), the bottom and top edges of the box represent lower quartile (Q1) and upper quartile (Q3). The outliers (red dots) are the values which falls outside of the lower/upper fences between 1.5 times the interquartile range (IQR, Q3–Q1) from Q1 and Q3, respectively. The black horizontal lines display minimum and maximum values in the fences.
Discussion
In this study, we translated our PiB PET data into the Centiloid scale as described previously1, and tested multiple reference regions for evaluating amyloid-beta deposits in cortical target regions, with a focus on patients with PiB (+) SVaD. We found that there were large magnitudes of effect sizes for differences between SVaD and YC regardless of reference ROI. However, using WM or pons ROI as a reference region showed larger variances in YC than using other ROI regions. In PiB (+) AD patients, the results were similar. Taken together, our findings show that CG, WC, and WC + B could be used as reference regions for amyloid imaging analysis in patients with PiB (+) SVaD, which are the same for AD patients.
We found that CG, WC or WC + B were reliable as reference regions for amyloid PET analyses in SVaD patients. These regions are commonly used as reference for amyloid PET analyses in AD patients because they are among the last regions where amyloid pathology is known to occur2,4,12, and are classified as Thal phase 511. Although some studies have raised the possibility of requiring different reference regions for specific diseases4,14,15,16, our findings demonstrate that appropriate reference regions for SVaD patients are the same as those for AD.
In this study, WM showed higher variability of Centiloid-scaled values in YC group than other reference regions. [C-11] PiB binding to WM is mainly non-saturable and non-specific21, but PiB retention has been found to be reduced in WMH regions compared to normal appearing WM regions22. Some have raised concerns that using a WM-containing reference region for analysis of amyloid imaging data could be problematic in a population with high WMH22. Consistent with this, our findings suggest that the quantification of amyloid deposits based on the ratio of target to WM may be an inappropriate approach in SVaD patients.
We also found that pons showed higher variability than other reference regions of Centiloid-scaled values in YC. This may appear inconsistent with a recent amyloid imaging study demonstrating the value of the pons as a reference region4. The discrepancy may be partly explained by different analysis methods, as well as by different study subjects. Unlike the previous study, patients with genetic AD and prion disease were excluded in our study. However, our findings are consistent with a previous Centiloid study showing pons with higher variability and lower effect size than other reference regions1. In addition, the inherent variability of normalization processes could affect the results of our study. For example, poor performance of normalization process in the pons can occur frequently, due to its small size and the fact that the SPM normalization algorithm seems to handle brainstem structures less well than cortical structures1, and could influence the [C-11] PiB processing and results.
One of the important findings of our study is that CG, WC, and WC + B, but not pons nor WM, are appropriate reference regions in both AD and SVaD. However, several limitations must be noted. First, our conclusions that WM and pons are not reliable reference regions, while CG, WC, and WC + B are suitable for analysis in the scaled data set, is not based on neuropathological studies. The precise performance of these reference regions will require confirmation by further neuropathological studies. Second, other pathologies such as prion disease, tau pathology, or hippocampal sclerosis are not considered because we did not perform a pathologic study. Third, due to the small number of SVaD patients used in this study, it will be necessary to replicate our results with larger samples in further study. Fourth, selecting a disease-specific CTX ROI as well as an appropriate reference region is important for calculating amyloid uptake. We therefore chose the disease-specific CTX ROI in the SVaD and AD groups, separately. Finally, we did not perform correction of PiB retention for brain atrophy, which might have effects on quantification.
Nevertheless, this study demonstrates that CG, WC, and WC + B are reliable reference regions for amyloid PET analysis in patients with SVaD, suggesting that amyloid burden in SVaD patients might be analyzed using the same reference regions as AD.
Methods
Participants
We selected 57 patients with PiB (+) AD and 23 patients with PiB (+) SVaD according to clinical diagnosis and PiB-PET status (see below for PiB-PET methods). PiB(+) AD patients were those who (1) met probable AD dementia criteria proposed by the National Institute of Neurological and Communicative Disorders and Stroke and the AD and Related Disorders Association23; (2) had minimal WM hyperintensities (WMH; periventricular WMH < 5 mm and deep WMH < 5 mm); (3) and had significant amyloid burden (PiB standardized uptake value ratio [SUVr] ≥ 1.5) as measured by PiB-PET. Patients with PiB (+) SVaD were those who (1) met the diagnostic criteria for vascular dementia as determined by the Diagnostic and Statistical Manual of Mental Disorders–Fourth Edition (DSM-IV) and also fulfilled the imaging criteria for SVaD proposed by Erkinjuntti et al.24; (2) showed a subcortical vascular feature defined as both a focal neurological symptom/sign and significant ischemia on MRI; (3) had significant ischemia on MRI, defined as a cap or band ≥ 10 mm and a deep WM lesion ≥ 25 mm, as modified from Fazekas ischemia criteria25; and (4) had significant amyloid burden (PiB SUVR ≥ 1.5)26.
Patients were evaluated by clinical interview and neurological and neuropsychological examinations as previously described27. All patients underwent laboratory tests including complete blood count, blood chemistry, vitamin B12/folate, syphilis serology, and thyroid function tests. Brain MRI confirmed the absence of structural lesions including territorial cerebral infarction, brain tumors, hippocampal sclerosis, and vascular malformation.
Ethics statement
This study protocol was approved by the Institutional Review Board of Samsung Medical Center. We obtained written consent from each participant and all methods were carried out in accordance with the approved guidelines.
MR and [C-11] PiB-PET imaging techniques
We acquired standardized three-dimensional T1 turbo field echo images from all participants at Samsung Medical Center, using the same 3.0 T MRI scanner (Philips Achieva; Philips Healthcare, Andover, MA), using the following parameters: sagittal slice thickness, 1.0 mm, over contiguous slices with 50% overlap; no gap; repetition time (TR) of 9.9 msec; echo time (TE) of 4.6 msec; flip angle of 8°, and matrix size of 240 × 240 pixels, reconstructed to 480 × 480 over a field of view (FOV) of 240 mm.
All patients underwent [C-11] PiB-PET imaging at Samsung Medical Center or Asan Medical Center (Seoul, Korea) with identical settings using a Discovery STe PET/CT scanner (GE Medical Systems, Milwaukee, WI, USA). [C-11] PiB-PET scanning was performed in 3-dimensional scanning mode examining 35 slices of 4.25-mm thickness spanning the entire brain. [C-11] PiB was injected into an antecubital vein as a bolus with a mean dose of 420 MBq (range 259–550 MBq). CT scans were performed for attenuation correction 60 minutes after injection. A 30-minute emission static PET scan was then initiated. The specific radioactivity of [C-11] PiB at time of administration was more than 1,500 Ci/mmol for patients and the radiochemical yield was more than 35%. The radiochemical purity of the tracer was more than 95% in all PET studies.
Image processing
We employed image processing described in the Centiloid paper1, and replicated the Centiloid procedure to validate methodological consistency (see Supplementary information).
Disease-specific CTX ROI
In Klunk et al.1, the CTX ROI was defined using a data-driven mask thresholded at 1.05 SUVr (using the WC reference ROI) difference between group-averaged PiB images of 19 AD subjects and 25 age-matched older controls. The threshold was designed to avoid WM regions and to minimize the number of separate clusters in the CTX ROI, and small holes and isolated voxels were manually edited. Although the typical amyloid binding regions reported in previous studies were included in the published CTX ROI, there were two reasons to generate a new CTX ROI. First, the published CTX ROI was designed specifically for AD patients, and given different progression patterns between SVaD and AD26, it was reasonable to make a new CTX ROI for SVaD patients. The second reason necessitating a new CTX ROI was the use of a numerical SUVr threshold. While amyloid deposition was well represented using a CTX ROI 1.05 SUVr unit difference in the dataset of Klunk et al.1, this value has not been validated in other datasets. CTX ROI values may be highly variable across cohorts, especially in AD patients with lower severity. Therefore, new CTX ROIs for SVaD and AD patients (CTXAD and CTXSVaD ROIs) were generated (using WC as reference) using data from the 57 PiB (+) AD and 23 PiB (+) SVaD patients, as well as control data from the Alzheimer’s Disease Neuroimaging Initiative (ADNI). When creating disease-specific CTX ROIs, we compared disease group and 14 PiB (−) OC subjects to find regions of high amyloid accumulation in elderly. As described above, individual images were transformed to MNI-152 template space and SUVr values derived. The average OC image was (separately) subtracted from averaged AD and SVaD images and spatially smoothed with a 5 mm full width at half maximum kernel. To extract CTXAD and CTXSVaD ROIs, we applied a percent threshold (upper 3% intensity) to smoothed images to retain a similar number of voxels as the published CTX ROI1. These disease-specific CTX ROIs are shown in Fig. 1.
Reference ROIs
The four data-driven ROIs (CG, WC, WC + B and pons) in Klunk et al.1 have been used as reference ROIs for quantifying of amyloid retention in numerous previous studies28,29,30,31,32,33, because no significant binding differences between AD and OC are observed. WM has also been evaluated as a reference ROI in recent [C-11] PiB-PET studies34, as those studies reveal comparable WM amyloid burden between diagnostic groups and low inter-subject variability. Therefore, we included WM as a candidate reference region. To generate the WM ROI, the WM probability map on template of SPM8 was thresholded at 0.7. This threshold provided a WM region that avoided contributions from CSF and amyloid retention in gray matter.
Statistical analysis
Individual SUVr values were calculated for normalized PiB-PET using five different reference ROIs (including WM), and CTXAD and CTXSVaD. SUVr values were obtained by ratio with each of the reference ROIs. CL values for individual subjects in AD group were computed by comparing to YC group which are considered not to have any brain amyloid pathology, and it defined as follows:
Where \(SUV{r}_{IND\ast }\) represents the individual SUVr values of all YC-0 and AD-100 subjects, and \(SUV{r}_{YC-0\ast }\) and \(SUV{r}_{AD-100\ast }\) represent each group’s mean values. For SVaD group, the term of \(SUV{r}_{AD-100\ast }\) was replaced as \(SUV{r}_{SVaD-100\ast }\).
To describe the dissimilar amyloid binding patterns between CTXAD and CTXSVaD ROIs, we measured regional volumetric proportion of CTX by dividing into frontal, parietal, temporal, occipital lobe and other regions.
Selection of the standard reference was based on the effect size of the group differences between patients and YC and on the variance of reference ROIs. Effect size for each group was evaluated with Cohen’s d with pooled standard deviation,
where:
μ p and μ n mean the average of SUVr in each patient and YC group.
\({\sigma }_{p}^{2}\) and \({\sigma }_{n}^{2}\) are the variance in each patient and YC group.
N p and N n are the number of subjects in each patient and YC group.
For variance tests, Bartlett’s statistics were employed for validating equal variances of reference ROIs in each clinical group35. Post hoc variance tests were also performed to investigate regional specificities for reference ROIs.
For quantitative analysis of lobar SUVr values among all groups, mean values of SUVr (using WC as reference) in each lobe were calculated.
References
Klunk, W. E. et al. The Centiloid Project: standardizing quantitative amyloid plaque estimation by PET. Alzheimer’s & dementia: the journal of the Alzheimer’s Association 11(1–15), e11–14, https://doi.org/10.1016/j.jalz.2014.07.003 (2015).
Ikoma, Y., Edison, P., Ramlackhansingh, A., Brooks, D. J. & Turkheimer, F. E. Reference region automatic extraction in dynamic [(11)C]PIB. Journal of cerebral blood flow and metabolism: official journal of the International Society of Cerebral Blood Flow and Metabolism 33, 1725–1731, https://doi.org/10.1038/jcbfm.2013.133 (2013).
Bullich, S. et al. Optimal reference region to measure longitudinal amyloid-beta change with 18F-florbetaben PET. Journal of nuclear medicine: official publication, Society of Nuclear Medicine. https://doi.org/10.2967/jnumed.116.187351 (2017).
Edison, P. et al. Can target-to-pons ratio be used as a reliable method for the analysis of [11 C] PIB brain scans? NeuroImage 60, 1716–1723 (2012).
Park, J. H. et al. Effects of cerebrovascular disease and amyloid beta burden on cognition in subjects with subcortical vascular cognitive impairment. Neurobiology of aging 35, 254–260, https://doi.org/10.1016/j.neurobiolaging.2013.06.026 (2014).
Jellinger, K. A. & Attems, J. Neuropathological evaluation of mixed dementia. Journal of the neurological sciences 257, 80–87, https://doi.org/10.1016/j.jns.2007.01.045 (2007).
Kalaria, R. N. The role of cerebral ischemia in Alzheimer’s disease. Neurobiol Aging 21, 321–330, doi:S0197-4580(00)00125-1 [pii] (2000).
Rockwood, K. et al. The diagnosis of “mixed” dementia in the Consortium for the Investigation of Vascular Impairment of Cognition (CIVIC). Ann N Y Acad Sci 903, 522–528 (2000).
Schneider, J. A., Arvanitakis, Z., Bang, W. & Bennett, D. A. Mixed brain pathologies account for most dementia cases in community-dwelling older persons. Neurology 69, 2197–2204 (2007).
Wang, B. W. et al. Multiple pathologies are common in Alzheimer patients in clinical trials. Can J Neurol Sci 39, 592–599, doi:H177721116452648 [pii] (2012).
Thal, D. R., Rub, U., Orantes, M. & Braak, H. Phases of A beta-deposition in the human brain and its relevance for the development of AD. Neurology 58, 1791–1800 (2002).
Perani, D. et al. A survey of FDG- and amyloid-PET imaging in dementia and GRADE analysis. Biomed Res Int 2014, 785039, https://doi.org/10.1155/2014/785039 (2014).
Johnson, K. A. et al. Imaging of amyloid burden and distribution in cerebral amyloid angiopathy. Ann Neurol 62, 229–234, https://doi.org/10.1002/ana.21164 (2007).
Mead, S. et al. Inherited prion disease with six octapeptide repeat insertional mutation–molecular analysis of phenotypic heterogeneity. Brain: a journal of neurology 129, 2297–2317, https://doi.org/10.1093/brain/awl226 (2006).
Klunk, W. E. et al. Amyloid deposition begins in the striatum of presenilin-1 mutation carriers from two unrelated pedigrees. J Neurosci 27, 6174–6184, https://doi.org/10.1523/JNEUROSCI.0730-07.2007 (2007).
Knight, W. D. et al. Carbon-11-Pittsburgh compound B positron emission tomography imaging of amyloid deposition in presenilin 1 mutation carriers. Brain: a journal of neurology 134, 293–300, https://doi.org/10.1093/brain/awq310 (2011).
Brendel, M. et al. Improved longitudinal [(18)F]-AV45 amyloid PET by white matter reference and VOI-based partial volume effect correction. Neuroimage 108, 450–459, https://doi.org/10.1016/j.neuroimage.2014.11.055 (2015).
Yoon, C. W. et al. Cerebellar atrophy in patients with subcortical-type vascular cognitive impairment. Cerebellum 12, 35–42, https://doi.org/10.1007/s12311-012-0388-0 (2013).
Sung, Y. H. et al. Midbrain atrophy in subcortical ischemic vascular dementia. Journal of neurology 256, 1997–2002, https://doi.org/10.1007/s00415-009-5226-z (2009).
Kim, Y. J. et al. White matter microstructural changes in pure Alzheimer’s disease and subcortical vascular dementia. European journal of neurology 22, 709–716, https://doi.org/10.1111/ene.12645 (2015).
Fodero-Tavoletti, M. T. et al. Characterization of PiB binding to white matter in Alzheimer disease and other dementias. Journal of nuclear medicine: official publication, Society of Nuclear Medicine 50, 198–204, https://doi.org/10.2967/jnumed.108.057984 (2009).
Goodheart, A. E. et al. Reduced binding of Pittsburgh Compound-B in areas of white matter hyperintensities. NeuroImage. Clinical 9, 479–483, https://doi.org/10.1016/j.nicl.2015.09.009 (2015).
McKhann, G. et al. Clinical diagnosis of Alzheimer’s disease: report of the NINCDS-ADRDA Work Group under the auspices of Department of Health and Human Services Task Force on Alzheimer’s Disease. Neurology 34, 939–944 (1984).
Erkinjuntti, T. et al. Research criteria for subcortical vascular dementia in clinical trials. Journal of neural transmission. Supplementum 59, 23–30 (2000).
Fazekas, F. et al. Pathologic correlates of incidental MRI white matter signal hyperintensities. Neurology 43, 1683–1689 (1993).
Lee, J. H. et al. Identification of pure subcortical vascular dementia using C-11-Pittsburgh compound B. Neurology 77, 18–25 (2011).
Seo, S. W. et al. Cortical thickness in single- versus multiple-domain amnestic mild cognitive impairment. NeuroImage 36, 289–297, https://doi.org/10.1016/j.neuroimage.2007.02.042 (2007).
Lowe, V. J. et al. Comparison of 18F-FDG and PiB PET in cognitive impairment. Journal of nuclear medicine: official publication, Society of Nuclear Medicine 50, 878–886, https://doi.org/10.2967/jnumed.108.058529 (2009).
Doherty, B. M. et al. Amyloid burden, cortical thickness, and cognitive function in the Wisconsin Registry for Alzheimer’sPrevention. Alzheimer’s & dementia 1, 160–169, https://doi.org/10.1016/j.dadm.2015.01.003 (2015).
Klunk, W. E. et al. Imaging brain amyloid in Alzheimer’s disease with Pittsburgh Compound-B. Annals of neurology 55, 306–319, https://doi.org/10.1002/ana.20009 (2004).
Mikhno, A. et al. Voxel-based analysis of 11C-PIB scans for diagnosing Alzheimer’s disease. Journal of nuclear medicine: official publication, Society of Nuclear Medicine 49, 1262–1269, https://doi.org/10.2967/jnumed.107.049932 (2008).
Jack, C. R. Jr. et al. 11C PiB and structural MRI provide complementary information in imaging of Alzheimer’s disease and amnestic mild cognitive impairment. Brain: a journal of neurology 131, 665–680, https://doi.org/10.1093/brain/awm336 (2008).
Kemppainen, N. M. et al. Voxel-based analysis of PET amyloid ligand [11C]PIB uptake in Alzheimer disease. Neurology 67, 1575–1580, https://doi.org/10.1212/01.wnl.0000240117.55680.0a (2006).
Chen, K. et al. Improved power for characterizing longitudinal amyloid-beta PET changes and evaluating amyloid-modifying treatments with a cerebral white matter reference region. Journal of nuclear medicine: official publication, Society of Nuclear Medicine 56, 560–566, https://doi.org/10.2967/jnumed.114.149732 (2015).
Rock, J. & Bartlett, M. K. Biopsy studies of human endometrium: criteria of dating and information about amenorrhea, menorrhagia, and time of ovulation. Journal of the American Medical Association 108, 2022–2028 (1937).
Acknowledgements
This research was supported by the Brain Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Science, ICT & Future Planning (2016M3C7A1913844), the Korea government (MSIP) (NRF-2016R1A2B3016609 and NRF-2017R1A2B2005081) and Research of Korea Centers for Disease Control and Prevention (2016-ER6203-00), and by National Institutes of Health (F32-AG050389).
Author information
Authors and Affiliations
Contributions
Designed the experiments (H.J.Y., S.H.M., J.M.L. and S.W.S.), performed the experiments (H.J.Y.), analyzed the data (H.J.Y. and S.W.S.), contributed reagents/materials (S.H.M., H.J.K., Y.S.C., K.H.L., D.L.N., J.M.L. and S.W.S.) and wrote the paper (H.J.Y., S.H.M., S.N.L. and S.W.S.).
Corresponding authors
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Yun, H.J., Moon, S.H., Kim, H.J. et al. Centiloid method evaluation for amyloid PET of subcortical vascular dementia. Sci Rep 7, 16322 (2017). https://doi.org/10.1038/s41598-017-16236-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-16236-1
This article is cited by
-
Multimodality imaging of neurodegenerative disorders with a focus on multiparametric magnetic resonance and molecular imaging
Insights into Imaging (2023)
-
Quantification of amyloid PET for future clinical use: a state-of-the-art review
European Journal of Nuclear Medicine and Molecular Imaging (2022)
-
Amyloid-β PET Classification on Cognitive Aging Stages Using the Centiloid Scale
Molecular Imaging and Biology (2022)
-
[11C]PIB amyloid quantification: effect of reference region selection
EJNMMI Research (2020)
-
Appropriate reference region selection of 18F-florbetaben and 18F-flutemetamol beta-amyloid PET expressed in Centiloid
Scientific Reports (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.