Abstract
Solvents play important roles in our lives, they are also of interest in molecular materials, especially for molecular magnets. The solvatomagnetic effect is generally used for trigger and/or regulation of magnetic properties in molecule-based systems, however, molecular nanomagnets showing solvatomagnetic effects are very difficult to obtain. Here we report four 3d-4f heterometallic cluster complexes containing ROH lattice solvent molecules, [Cu3Tb2(H3L)2(OAc)2(hfac)4]∙2ROH {H6L = 1,3-Bis[tris(hydroxymethyl)methylamino]propane, hfac− = hexafluoroacetylacetonate; R = CH3, 1; R = C2H5, 2; R = C3H7, 3; R = H, 4}. Single-molecule magnet (SMM) properties of these four complexes were observed to be dependent on the ROH lattice solvent molecule. There is an interesting magneto-structural correlation: the larger the R group, the higher the energy barrier. For the first time, the solvatomagnetic effect is used for the continuous fine adjustment of the energy barrier of 0D molecular nanomagnets. Additionally, [Cu3Dy2(H3L)2(OAc)2(hfac)4]∙2MeOH (5), an analogue of [Cu3Tb2(H3L)2(OAc)2(hfac)4]∙2MeOH (1), is also reported for comparison.
Similar content being viewed by others
Introduction
Solvents, especially water, are critical to the origins of life, and they have penetrated into all aspects of human life. Besides as reaction mediums and extracting agents, chemical solvents are also of interest in molecular materials. For example, in the field of molecular magnets they can be utilized as the terminal ligand to complete the coordination configuration1,2,3,4; and they can also serve as guest or lattice molecules to adjust magnetic properties5,6,7,8,9. The solvatomagnetic effect is very interesting because solvent molecules can be used for trigger and/or regulation of magnetic properties while the molecular magnetic structure is always maintained. Therefore, molecular magnets showing solvatomagnetic effects can be used as molecule devices, molecular switches and/or molecular sensors. Naturally, solvatomagnetic effects are often found in porous metal-organic frameworks (MOFs) in which solvent molecules are guest molecules5,6,7,8,9, while low-dimensional systems with solvatomagnetic effects are more difficult to obtain due to the lack of pores. Recently, we found a chain-like azido-bridged manganese(III) coordination polymer showing both solvatomagnetic effect and spin-glass behaviour10. In studies of single molecule magnets (SMMs)11, we also hope to explore SMM systems with solvatomagnetic effects. However, it is a great challenging task because most SMMs reported are concentrated on zero-dimensional (0 D) cluster or mononuclear systems.
It is well known that SMMs are of great potential for technological applications in high-density information storage, quantum computing and spintronics12,13,14,15,16,17; and the energy barrier leading to magnetic bistability and slow magnetic relaxation is a pivotal parameter. Therefore, except enhancing the relaxation energy barrier and increasing the blocking temperature18,19,20,21, tuning the relaxation energy barrier is another important target in the molecular nanomagnet field22,23,24,25,26. Surprisingly, systematic studies of SMMs with the same magnetic structure are still rare, however, some factors such as the electron-withdrawing effect27, the electrostatic potential of the key coordination atom28 have been observed to be able to modulate SMMs’ energy barriers recently. Regarding structures and magnetic properties may be affected by a small change of circumstance, solvent molecules may also be used to adjust SMMs’ properties. To the best of our knowledge, a direct correlation between energy barriers and different lattice solvent molecules of 0D molecular nanomagnets has never been documented, though a 3D Dy(III) MOF-type SMM was found to show an obvious solvatomagnetic effect in 201529, and guest-dependent single-ion magnet behaviours were observed in a 2D cobalt(II) coordination polymer in 201630. Herein we describe the lattice-solvent effect of ROH molecules (R = CH3, 1; R = C2H5, 2; R = C3H7, 3; R = H, 4) on the energy barrier of 0D SMMs with the same magnetic structure [Cu3Tb2(H3L)2(OAc)2(hfac)4] {H6L = 1,3-Bis[tris(hydroxymethyl)methylamino]propane, Fig. 1; hfac− = hexafluoroacetylacetonate}. Fine adjustment of the energy barrier (from 25.7 K to 33.1 K, H dc = 0 Oe) in this [Cu3Tb2(H3L)2(OAc)2(hfac)4] SMM system was achieved by changing the ROH lattice solvent molecule. A similar [Cu3Dy2(H3L)2(OAc)2(hfac)4] SMM system was also studied, but only the crystal structure of [Cu3Dy2(H3L)2(OAc)2(hfac)4]∙2MeOH (5) was successfully solved; complex 5 also exhibits slow magnetic relaxation under zero dc field, with the energy barrier of 30.0 K, a relatively high value for reported relaxation barriers of the Cu-Dy heterometallic SMMs.
Results and Discussion
Preparation
Bis-tris propane (H6L), an universal ligand due to flexible polydentate coordination sites, has been used to bind not only 3d transition metal ions31,32 but also 4 f lanthanide metal ions33. Furthermore, it can also be utilized to construct 3d-4f heterometallic complexes34. Recently, Murrie et al. reported a series of 3d-4f complexes formulated as {Ln2Cu3(H3L)2Xn} (X = OAc−, Ln = Gd, Tb or X = NO3 −, Ln = Gd, Tb, Dy, Ho, Er)35; they found that changing the auxiliary ligand OAc− through NO3 − may lead to a remarkable improvement of the energy barrier of {Tb2Cu3(H3L)2Xn} (X = OAc− and NO3 −) complexes, which suggests that the anion co-ligand has a great impact on the energy barrier of {Tb2Cu3(H3L)2Xn} SMMs. In the recent process of pursuing new SMMs, we observed that using Ln(OAc)(hfac)2(H2O)2 as the lanthanide (III) salt source may lead mixed co-ligands OAc− and hfac− into 3d-4f heterometallic clusters effectively36. Therefore, we adopted this synthesis strategy to obtain the [Cu3Tb2(H3L)2(OAc)2(hfac)4] SMM with different ROH lattice solvent molecules (methanol, ethanol and isopropyl alcohol), in which not only the OAc− anion but also the hfac− anion are co-ligands. Notably, our synthetic procedures were completed at room temperature rather than at 60 °C used by Murrie group35. Products using methanol, ethanol and isopropyl alcohol as reaction solvents were [Cu3Tb2(H3L)2(OAc)2(hfac)4]∙2MeOH (1), [Cu3Tb2(H3L)2(OAc)2(hfac)4]∙2EtOH (2) and [Cu3Tb2(H3L)2(OAc)2(hfac)4]∙2iso-C3H7OH (3), respectively; while [Cu3Tb2(H3L)2(OAc)2(hfac)4]∙2H2O (4) was quantitatively transformed from complex 1 by taking place of methanol molecules with water molecules. In order to yield [Cu3Tb2(H3L)2(OAc)2(hfac)4] SMMs with larger ROH lattice solvent molecules, other ROH solvents such as isobutyl alcohol, n-butyl alcohol and isoamyl alcohol were also used instead of methanol for 1, but no any crystalline products could be obtained. Furthermore, the [Cu3Dy2(H3L)2(OAc)2(hfac)4] SMM system was also explored, but only the crystal structure of [Cu3Dy2(H3L)2(OAc)2(hfac)4]∙2MeOH (5) was successfully solved, the crystal structure of [Cu3Dy2(H3L)2(OAc)2(hfac)4] SMMs with other lattice solvent molecules (H2O, ethanol and isopropyl alcohol) could not be obtained due to the severe twinning phenomenon.
Structural description
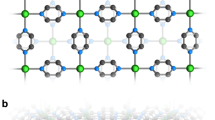
All [Cu3Tb2(H3L)2(OAc)2(hfac)4]∙2ROH SMMs have the main structure [Cu3Tb2(H3L)2(OAc)2(hfac)4] (Fig. 2). Therefore, the structure of [Cu3Tb2(H3L)2(OAc)2(hfac)4]∙2MeOH (1) is chose to be described in detail. In the main structure [Cu3Tb2(H3L)2(OAc)2(hfac)4], a {Cu3(H3L)2} linear unit is formed through bridging two terminal {Cu(H3L)}− fragments using a central Cu2+ ion, then two Tb3+ ions link to this {Cu3(H3L)2} linear unit in the opposite direction, in which each Tb3+ ion connects with the central Cu2+ ion and one external Cu2+ ion through sharing one μ 3-O atom and one μ-O atom from one H3L3− ligand, and one μ 3-O atom from the other H3L3− ligand (Fig. 2a), similar to those in {Ln2Cu3(H3L)2Xn}35. The eight-coordinate sphere of each Tb3+ ion is finally completed by two hfac− anions and one OAc− anion. Shape software37 was adopted to calculate the Tb(III) coordination polyhedron, giving a triangular dodecahedron as the most likely configuration for complex 1, and the deviation value from the ideal D 2d symmetry is 1.015 (Table S1, SI). It is worth noting that the Tb(III) coordination polyhedron can also be viewed as a biaugmented trigonal prism, but with the deviation value of 1.756 from the ideal D 2d symmetry. Moreover, the calculation result for the Tb(III) coordination polyhedra of complexes 2–4 using Shape software37 are listed in Tables S2–S4 (SI), respectively.
The external Cu atom, in a distorted square-pyramidal configuration, is coordinated with two N atoms and two μ-O atoms from one H3L3− ligand, forming the base of the pyramid; whereas the third μ-O atom from the same H3L3− ligand occupying the apical site. The central Cu2+ ion is coordinated by two μ 3-O atoms and four μ-O atoms from two H3L3− ligands, generating a distorted octahedral geometry, in which two μ-O atoms bridging the central Cu atom and the external Cu atom are in the Jahn-Teller axis’ direction, with the long Cu-O bond distance of 2.665 Å for complex 1.
There are hydrogen bonds between the methanol O atom and the carboxylate O atom with the Omethanol…Ocarboxylate distance of 2.803 Å and between the methanol O atom and the N atom from the H3L3− ligand with the Omethanol…N distance of 2.937 Å for complex 1. Similar hydrogen bonds were observed between the ethanol O atom and the carboxylate O atom with the Oethanol…Ocarboxylate distance of 2.788 Å and between the ethanol O atom and the N atom from the H3L3− ligand with the Oethanol…N distance of 2.941 Å for 2; between the isopropyl alcohol O atom and the carboxylate O atom with the Oisopropyl alcohol…Ocarboxylate distance of 2.817 Å and between the isopropyl alcohol O atom and the N atom from the H3L3− ligand with the Oisopropyl alcohol…N distance of 2.922 Å for 3; and between the water O atom and the carboxylate O atom with the Owater…Ocarboxylate distance of 2.866 Å and between the water O atom and the N atom from the H3L3− ligand with the Owater…N distance of 2.944 Å for 4. These weak intermolecular interactions play important roles in not only stabilization of crystal structures but also adjustment of magnetic properties for complexes 1–4.
Complex 5 has the same structure as 1, but Dy instead of Tb is used (Fig. S1, SI). The Dy-O bond distance (average 2.357 Å) in 5 is slightly smaller than the Tb-O bond length (average 2.368 Å) in 1 owing to the lanthanide contraction effect. The Dy(III) coordination polyhedron can also be described as a triangular dodecahedron with the deviation value of 0.975 from the ideal D 2d symmetry (Table S5, SI). This value is a little smaller than that of 1 (1.015), indicating that the Dy(III) coordination polyhedron in 5 is closer to a triangular dodecahedron than the Tb(III) coordination polyhedron in 1. The deviation value from the ideal D 2d symmetry for a biaugmented trigonal prism is 1.735 for 5, also a little smaller than that of 1 (1.756). Similar to 1, there are also hydrogen bonds between the methanol O atom and the carboxylate O atom with the Omethanol…Ocarboxylate distance of 2.796 Å and between the methanol O atom and the N atom from the H3L3− ligand with the Omethanol…N distance of 2.937 Å for complex 5.
Magnetic properties
The direct current (dc) variable-temperature magnetic susceptibility of complexes 1–4 was measured at 1000 Oe applied field (Fig. 3). The room temperature χT values of the complexes 1 (24.91 cm3 K mol−1), 2 (24.85 cm3 K mol−1), 3 (24.84 cm3 K mol−1) and 4 (24.90 cm3 K mol−1) are slightly larger than the theoretical value of 24.77 cm3 K mol−1 for three noninteracting Cu2+ ions (g = 2.0) and two uncoupled Tb3+ ions (7 F 6, J = 6, L = 3, S = 3, g = 3/2). As shown in Fig. 3, upon cooling, the χT product almost keeps a constant value or just slightly lowers; however, below about 50 K, a rapid rise appears until reaches the maximum values of 53.92 cm3 K mol−1 at 6.0 K for 1, 45.14 cm3 K mol−1 at 4.0 K for 2 and 49.80 cm3 K mol−1 at 4.0 K for 3; the χT values then decline to 49.34 cm3 K mol−1 at 2.0 K for 1, 43.48 cm3 K mol−1 at 2.0 K for 2 and 47.48 cm3 K mol−1 at 2.0 K for 3. Exceptionally, complex 4 does not reach the maximum value until 2.0 K (44.60 cm3 K mol−1). These magnetic behaviours are very similar to those of {Tb2Cu3(H3L)2Xn} (X = OAc− and NO3 −)35, suggesting that all four complexes are also ferromagnetic. The small difference in dc magnetic susceptibilities of 1–4 means that there is a solvatomagnetic effect in this [Cu3Tb2(H3L)2(OAc)2(hfac)4] SMM system.
The solvatomagnetic effect could also be detected by alternating current (ac) magnetic susceptibility investigations. Both the in-phase (χ′, Fig. S2, SI) and the out-of-phase (χ′′, Fig. 4) of variable-temperature ac magnetic susceptibility for 1–4 are frequency-dependent in zero dc field, indicating slow magnetic relaxation typical for SMMs. Such thermally induced relaxation was fitted with the Arrhenius law, τ = τ 0exp(U eff/kT), extracting U eff/k values of 30.0(0.4) K for 1, 32.4(0.2) K for 2, 33.1(0.7) K for 3 and 25.7(0.2) K for 4 as well as τ 0 values of 3.7(0.2) × 10−8 s for 1, 6.2(0.1) × 10−9 s for 2, 2.6(0.3) × 10−8 s for 3 and 2.3(0.1) × 10−8 s for 4 (Fig. 5a). All four τ 0 values are within the normal range for SMMs/SIMs (10−5–10−11 s)13. A comparison of the effective barrier value for complexes 1–4 with the R group of the ROH lattice solvent molecules (R = H, 4; R = CH3, 1; R = C2H5, 2 and R = C3H7, 3) reveals an important magneto-structural correlation for this [Cu3Tb2(H3L)2(OAc)2(hfac)4] SMM system: The larger the R group in ROH, the higher the energy barrier of the [Cu3Tb2(H3L)2(OAc)2(hfac)4]∙2ROH SMM (Fig. 5b). It is noteworthy that either the U eff/k value of 2 or the U eff/k value of 3 is one of the largest values so far for the Cu-Tb heterometallic SMMs in zero dc field, just smaller than that of (NMe4)2[Tb2Cu3(H3L)2(NO3)7(CH3OH)2](NO3) (36 K)35; the U eff/k value of 1 is also remarkable, which is comparable with that of [Cu2(valpn)2Tb2(N3)6]·2CH3OH [H2valpn = 1,3-propanediylbis(2-iminomethylene-6-methoxyphenol)] (30.1 K, H dc = 0 Oe)38. In many cases39,40,41,42,43,44, a dc field is necessary for 3d-4f heterometallic complexes to display magnetic relaxation because of the obvious quantum-tunnelling effects.
Simplified theoretical investigations by Murrie group suggested that the magnetic bistability in the [Cu3Tb2(H3L)2Xn] system is not because of single-ion behaviours, and both the Cu···Cu and Cu···Tb ferromagnetic interactions maybe quench the tunnel splitting, which are similar to acting as an internal applied field, inducing to zero-field SMM behaviours35. Nevertheless, the difference of the Tb3+ coordination configurations has influence on the SMM characteristics35. Owing to great difficulty for theoretical calculation and comparison of the Cu···Cu and Cu···Tb ferromagnetic couplings35, we tried to make a magneto-structural correlation for complexes 1–4 using the deviation value from the ideal D 2d symmetry of the biaugmented trigonal prism for the Tb3+ ion and the intermolecular distance as two main structural parameters. As shown in Table 1, the coordination configuration of the Tb3+ ions is closer to the biaugmented trigonal prism from 1 to 3, the corresponding energy barrier value becomes larger from 1 to 3, indicating the biaugmented trigonal prism configuration in the [Cu3Tb2(H3L)2(OAc)2(hfac)4] SMM system is the dominant configuration; but 4 is a bit unusual, its deviation value (1.735) is comparable with that of 1 (1.756), which suggests that other structural factors such as intermolecular distances need to be considered; as shown in Table 1, the longer the intermolecular distance (defined by the shortest Cucentral…Cucentral separation), the higher the energy barrier; which is in line with the magneto-structural correlation using the R group itself, because larger ROH lattice solvent molecules may enhance intermolecular distances correspondingly.
The SMM properties of 1–4 were also evaluated by the parameter Φ = (ΔT f/T f)/Δ(logf)45, where f represents the frequency and T f the peak temperature of χ″ curve; the Φ values of 1, 2, 3 and 4 are 0.18, 0.17, 0.17 and 0.21, respectively, which support the superparamagnet behaviour of these SMMs (Φ > 0.1), but exclude any spin glass properties (Φ ≈ 0.01)45. Further determinations of ac magnetic susceptibility revealed that the variable-frequency χ″ signals of 1–4 are evidently temperature-dependent (Fig. 6), confirming slow magnetic relaxation of SMMs. The χ″ vs χ′ plots show classical half-circular curves for all four complexes, indicating a single magnetic relaxation process (Fig. S3, SI). These Cole-Cole plots could be fitted with a generalized Debye model46,47. The α values are smaller than 0.07 for 2–4, suggesting a single relaxation mechanism; while the α values for 1 are from 0.10 to 0.22, indicating a relatively narrow distribution of the relaxation time. In addition, no any hysteresis was observed in the M vs H plot at 1.9 K for 1–4 (Fig. S4, SI).
The χT value at room temperature for complex 5 is 29.44 cm3 K mol−1 (Fig. 7a), which is in good agreement with the expected value of 29.47 cm3 K mol−1 for three uncoupled Cu2+ ions (g = 2.0) and two isolated Dy3+ ions (6 H 15/2, J = 15/2, S = 5/2, L = 5, g = 4/3). When temperature is decreased, the χT product decreases very slowly until 110 K (29.20 cm3 K mol−1), then increases very gently until about 50 K. Below this temperature, the χT value rises rapidly, reaching the maximum of 63.92 cm3 K mol−1 at 5 K and then dropping down to 62.38 cm3 K mol−1 at 2 K, these magnetic behaviors are similar to those for 1 and (NMe4)2{Dy2Cu3(NO3)7(CH3OH)2](NO3)35, and the ferromagnetic coupling obviously exists between the Cu2+ ion and the Dy3+ ion as well as among the Cu2+ ions, similar to that observed in [Gd2Cu3(H3L)2(CH3COO)6]·THF·3H2O by Murrie group35.
The magnetization dynamics of compound 5 are similar to those of complexes 1–4. Under zero dc field, the appearance of frequency-dependent χ′ (Fig. S5, SI) and χ″ signals (Fig. 7b) indicates SMM behaviors of 5. The SMM parameters extracted from the Arrhenius law for 5 are U eff/k = 30.0(0.2) K and τ 0 = 9.7(0.1) × 10−9 s (Fig. 7c). The energy barrier value of 5 is comparable with that of 1, but obviously larger than that of (NMe4)2{Dy2Cu3(H3L)2(NO3)7(CH3OH)2](NO3) [23.9(0.1) K], whose χ″ signals even do not appear peaks in zero dc field35. Notably, this U eff/k value is the third high value for the Cu-Dy heterometallic SMMs, after 47 K of [{Dy(hfac)3}2{Cu(dpk)2}] (dpk− = di-2-pyridyl ketoximate)48 and 41.6 K of [Cu4Dy4(vanox)6(Hvanox)2(NO3)4(μ-HOMe)2]·6MeOH (H2vanox = 3-methoxy-2-hydroxybenzaldoxime)49. Furthermore, this U eff/k value is remarkable larger than those of the Cu-Dy heterometallic SMMs with higher nucleus (<20 K)50,51. Additionally, the parameter Φ value of 0.16 for 5 supports the SMM nature too.
The variable-frequency ac magnetic susceptibility study of 5 revealed that the χ″ signals of 5 are temperature-dependent (Fig. 7d), confirming the SMM behavior of 5. The Cole-Cole plots were fitted to a generalized Debye model (Fig. S6, SI)46,47, giving the α values of 0.01–0.08 for 5, suggesting the magnetic relaxation happens via a single relaxation process. Additionally, the M vs H plot of 5 shows no any hysteresis at 1.9 K (Fig. S7, SI).
Conclusions
In summary, a mixed OAc−/hfac− co-ligands’ synthesis strategy was adopted to prepare 3d-4f heterometallic SMMs based on the 1,3-Bis[tris(hydroxymethyl)methylamino]propane ligand (H6L). The ROH lattice solvent molecules (R = H, CH3, C2H5 and C3H7) in the [Cu3Tb2(H3L)2(OAc)2(hfac)4] SMM system have great influences on the energy barrier; the larger the R group, the higher the energy barrier. We predict that the larger ROH molecule may enlarge the intermolecular distance and can help to change the coordination configuration of the Ln(III) ions through the hydrogen bonding interaction between the ROH lattice solvent molecule and the [Cu3Tb2(H3L)2(OAc)2(hfac)4] main-structural molecule. Our work demonstrates that solvatomagnetic effects can be used to continuously fine-tune energy barriers in SMMs. The discovery is bound to have significances in enhancing and turning energy barriers of molecular nanomagnets via chemical methods such as using lattice-solvent effects.
Methods
Physical measurements
The elemental analyses were measured on a Vario ELIII elemental analyser. The magnetic susceptibility measurements were carried out on a Quantum Design MPMS-XL5 SQUID magnetometer, and diamagnetic corrections were calculated from Pascal’s constants of all components.
Synthesis of [Cu3Tb2(H3L)2(OAc)2(hfac)4]∙2MeOH (1)
To a mixture of H6L (0.25 mmol) and Cu(ClO4)2·6H2O (0.375 mmol) in 20 mL of MeOH, was added Tb(OAc)(hfac)2(H2O)2 (0.15 mmol), a blue solution was formed after being stirred for 10 min; Et3N (0.75 mmol) was then added dropwise, the resultant solution was stirred for 3 h at room temperature and turned violet. Violet plate-like X-ray quality crystals were obtained through slow evaporation of the filtrate at room temperature over 1 week. Yield (25%). Anal. Calcd (%) for C48H64Cu3F24N4O26Tb2 (1) C 27.75; H 3.11; N 2.70. Found: C 27.80; H 3.14; N 2.67.
Synthesis of [Cu3Tb2(H3L)2(OAc)2(hfac)4]∙2EtOH (2)
The same synthetic procedure for complex 1 was followed, but using ethanol instead of methanol. Violet plate-like X-ray quality crystals were obtained through slow evaporation of the filtrate at room temperature over 10 days. Yield (27%). Anal. Calcd (%) for C50H68Cu3F24N4O26Tb2 (2): C 28.52; H 3.26; N 2.66. Found: C 28.55; H 3.29; N 2.63.
Synthesis of [Cu3Tb2(H3L)2(OAc)2(hfac)4]∙2iso-C3H7OH (3)
The same synthetic procedure for complex 1 was followed, but using isopropyl alcohol instead of methanol. Violet plate-like X-ray quality crystals were obtained through slow evaporation of the filtrate at room temperature over 15 days. Yield (22%). Anal. Calcd (%) for C52H72Cu3F24N4O26Tb2 (3): C 29.27; H 3.40; N 2.63. Found: C 29.23; H 3.43; N 2.60.
Synthesis of [Cu3Tb2(H3L)2(OAc)2(hfac)4]∙2H2O (4)
Complex 1 was kept at 60 °C for 6 h, and then exposed on air for 24 h. Violet plate-like X-ray quality crystals of 4 were obtained quantitatively. Anal. Calcd (%) for C46H60Cu3F24N4O26Tb2 (4): C 26.96; H 2.95; N 2.73. Found: C 27.02; H 2.99; N 2.69.
Synthesis of [Cu3Dy2(H3L)2(OAc)2(hfac)4]∙2MeOH (5)
The same synthetic procedure for complex 1 was followed, but using Dy(OAc)(hfac)2(H2O)2 instead of Tb(OAc)(hfac)2(H2O)2. Violet plate-like X-ray quality crystals were obtained through slow evaporation of the filtrate at room temperature over 1 week. Yield (28%). Anal. Calcd (%) for C48H64Cu3Dy2F24N4O26 (5): C 27.66; H 3.09; N 2.69. Found: C 27.69; H 3.11; N 2.67.
X-ray crystallography
A single crystal with dimensions 0.261 × 0.093 × 0.025 mm3 of 1, 0.178 × 0.063 × 0.024 mm3 of 2, 0.183 × 0.125 × 0.031 mm3 of 3, 0.108 × 0.067 × 0.025 mm3 of 4, and 0.134 × 0.125 × 0.027 mm3 of 5 was picked out to mount on a Bruker SMART APEX-CCD diffractometer with Mo-K α radiation (λ = 0.71073 Å) for data collection at 173(2) K. Empirical absorption corrections from φ and ω scan were applied. Cell parameters were calculated by the global refinement of the positions of all collected reflections for five complexes. The structures were solved by direct methods and refined by a full matrix least-squares technique based on F2 using with the SHELX-2014 program package. All hydrogen atoms were set in calculated positions and refined as riding atoms, and all non-hydrogen atoms were refined anisotropically. CCDC 1574978–1574982 contain the supplementary crystallographic data, which can be obtained free of charge from the Cambridge Crystallographic Data Centre via www.ccdc.cam.ac.uk/data_request/cif.
Crystal data for 1: P−1, a = 10.086(2) Å, b = 12.463(3) Å, c = 15.594(3) Å, α = 104.67(3)°, β = 94.07(3)°, γ = 108.97(3)°, V = 1767.9(7) Å3, M r = 2077.49, D c = 1.951 g cm−3, Z = 1, R 1 = 0.0366 (I > 2σ(I)), wR 2 = 0.0878 (I > 2σ(I)), S = 1.080.
Crystal data for 2: P−1, a = 10.243(2) Å, b = 12.469(3) Å, c = 15.602(3) Å, α = 101.71(3)°, β = 96.50(3)°, γ = 110.00(3)°, V = 1797.2(6) Å3, M r = 2105.54, D c = 1.945 g cm−3, Z = 1, R 1 = 0.0498 (I > 2σ(I)), wR 2 = 0.1085 (I > 2σ(I)), S = 1.122.
Crystal data for 3: P−1, a = 10.309(2) Å, b = 12.473(3) Å, c = 15.677(3) Å, α = 101.80(3)°, β = 96.96(3)°, γ = 110.19(3)°, V = 1811.4(7) Å3, M r = 2133.58, D c = 1.956 g cm−3, Z = 1, R 1 = 0.0345 (I > 2σ(I)), wR 2 = 0.0797 (I > 2σ(I)), S = 1.084.
Crystal data for 4: P−1, a = 10.042(2) Å, b = 12.480(3) Å, c = 15.819(3) Å, α = 107.08(3)°, β = 99.23(3)°, γ = 109.83(3)°, V = 1706.2(7) Å3, M r = 2049.44, D c = 1.995 g cm−3, Z = 1, R 1 = 0.0488 (I > 2σ(I)), wR 2 = 0.0945 (I > 2σ(I)), S = 1.153.
Crystal data for 5: P−1, a = 10.085(2) Å, b = 12.427(3) Å, c = 15.581(3) Å, α = 104.59(3)°, β = 94.21(3)°, γ = 108.86(3)°, V = 1762.5(7) Å3, M r = 2084.65, D c = 1.964 g cm−3, Z = 1, R 1 = 0.0321 (I > 2σ(I)), wR 2 = 0.0762 (I > 2σ(I)), S = 1.074.
References
Milios, C. J. et al. A Record Anisotropy Barrier for a Single-Molecule Magnet. J. Am. Chem. Soc. 129, 2754–2755 (2007).
Xu, H.-B. et al. Stringing Oxo-Centered Trinuclear [MnIII 3O] Units into Single-Chain Magnets with Formate or Azide Linkers. Angew. Chem. Int. Ed. 2007 46, 7388–7392 (2007).
Bai, Y.-L. et al. Coexistence of Magnetization Relaxation and Dielectric Relaxation in a Single-Chain Magnet. J. Am. Chem. Soc. 128, 16428–16429 (2006).
Liu, C.-M. et al. 1D Coordination Polymers Constructed from anti-anti Carboxylato-Bridged MnIII 3O(Brppz)3 Units: From Long-Range Magnetic Ordering to Single-Chain Magnet Behaviors. Inorg. Chem. 48, 4980–4987 (2009).
Halder, G. J. et al. Guest-dependent spin crossover in a nanoporous molecular framework material. Science 298, 1762–1765 (2002).
Maspoch, D. et al. A nanoporous molecular magnet with reversible solvent-induced mechanical and magnetic properties. Nat. Mater. 2, 190–195 (2003).
Rujiwatra, A. et al. Layered Cobalt Hydroxysulfates with Both Rigid and Flexible Organic Pillars: Synthesis, Structure, Porosity, and Cooperative Magnetism. J. Am. Chem. Soc. 123, 10584–10594 (2001).
Sato, Y. et al. Solvatomagnetism-Induced Faraday Effect in a Cobalt Hexacyanochromate-Based Magnet. J. Am. Chem. Soc. 125, 14590–14595 (2003).
Zhang, X.-M. et al. Dehydration-Induced Conversion from a Single-Chain Magnet into a Metamagnet in a Homometallic Nanoporous Metal-Organic Framework. Angew. Chem. Int. Ed. 46, 3456–3459 (2007).
Liu, C.-M. et al. Solvatomagnetic effect and spin-glass behavior in a 1D coordination polymer constructed from EE-azido bridged MnIII 3O units. Chem. Commun. 44, 368–370 (2008).
Liu, C.-M. et al. A Chinese Pane-Like 2D Metal-Organic Framework Showing Magnetic Relaxation and Luminescence Dual-Functions. Sci. Rep. 7, 11156, https://doi.org/10.1038/s41598-017-11006-5 (2017).
Sessoli, R. et al. Magnetic bistability in a metal-ion cluster. Nature 365, 141–143 (1993).
Woodruff, D. N., Winpenny, R. E. P. & Layfield, R. A. Lanthanide single-molecule magnets. Chem. Rev. 113, 5110–5148 (2013).
Bogani, L. & Wernsdorfer, W. Molecular spintronics using single-molecule magnets. Nat. Mater. 7, 179–186 (2008).
Komeda, T. et al. Observation and electric current control of a local spin in a single-molecule magnet. Nat. Commun. 2, 217 (2011).
Fetoh, A. et al. Photo-activation of single molecule magnet behavior in a manganese based complex. Sci. Rep. 6, 23785, https://doi.org/10.1038/srep23785 (2016).
Gao, F. et al. Tetrathiafulvalene-supported triple-decker phthalocyaninato dysprosium(III) complex: synthesis, properties and surface assembly. Sci. Rep. 4, 5928, https://doi.org/10.1038/srep05928 (2014).
Liu, J. et al. A Stable Pentagonal Bipyramidal Dy(III) Single-Ion Magnet with a Record Magnetization Reversal Barrier over 1000 K. J. Am. Chem. Soc. 138, 5441–5450 (2016).
Yao, X.-N. et al. Two-Coordinate Co(II) Imido Complexes as Outstanding Single Molecule Magnets. J. Am. Chem. Soc. 139, 373–380 (2017).
Ding, Y.-S. et al. On Approaching the Limit of Molecular Magnetic Anisotropy: A Near-Perfect Pentagonal Bipyramidal Dysprosium(III) Single-Molecule Magnet. Angew. Chem. Int. Ed. 55, 16071–16074 (2016).
Goodwin, C. A. P. et al. Molecular magnetic hysteresis at 60 kelvin in dysprosocenium. Nature 548, 439–442 (2017).
Qian, K. et al. A Single-Molecule Magnet Based on Heptacyanomolybdate with the Highest Energy Barrier for a Cyanide Compound. J. Am. Chem. Soc. 135, 13302–13305 (2013).
Langley, S. K. et al. Modulation of slow magnetic relaxation by tuning magnetic exchange in {Cr2Dy2} single molecule magnets. Chem. Sci. 5, 3246–3256 (2014).
Li, X.-L. et al. Modulation of Homochiral DyIII Complexes: Single-Molecule Magnets with Ferroelectric Properties. Chem.-Eur. J. 18, 14632–14637 (2012).
Zhang, L. et al. Anions Influence the Relaxation Dynamics of Mono-μ 3-OH-Capped Triangular Dysprosium Aggregates. Inorg. Chem. 54, 5571–5578 (2015).
Liu, C.-M. et al. Simultaneous assembly of mononuclear and dinuclear dysprosium(III) complexes behaving as single-molecule magnets in a one-pot hydrothermal synthesis. Sci. China Chem. 60, 358–365 (2017).
Habib, F. et al. Significant Enhancement of Energy Barriers in Dinuclear Dysprosium Single-Molecule Magnets Through Electron-Withdrawing Effects. J. Am. Chem. Soc. 135, 13242–13245 (2013).
Wang, Y.-L. et al. Anions Influence the Relaxation Dynamics of Mono-μ3-OH-Capped Triangular Dysprosium Aggregates. Inorg. Chem. 55, 5571–5578 (2016).
Zhang, X. et al. Influence of Guest Exchange on the Magnetization Dynamics of Dilanthanide Single-Molecule-Magnet Nodes within a Metal-Organic Framework. Angew. Chem. Int. Ed. 54, 9861–9865 (2015).
Vallejo, J. et al. Guest-dependent single-ion magnet behaviour in a cobalt(II) metal-organic framework. Chem. Sci. 7, 2286–2293 (2016).
Ferguson, A. et al. Bis-Tris Propane as a New Polydentate Linker in the Synthesis of Iron(III) and Manganese(II/III) Complexes. Inorg. Chem. 47, 9742–9744 (2008).
Milway, V. A. et al. Directed Synthesis of {Mn18Cu6} Heterometallic Complexes. Angew. Chem. Int. Ed. 52, 1949–1952 (2013).
Liu, C.-M. et al. A single-molecule magnet featuring a parallelogram [Dy4(OCH2–)4] core and two magnetic relaxation processes. Dalton Trans. 42, 14813–14818 (2013).
Han, S.-D. et al. Synthesis and Magnetic Properties of a Series of Octanuclear [Fe6Ln2] Nanoclusters. Cryst. Growth Des. 15, 2253–2259 (2015).
Ojea, M. J. H. et al. Enhancement of TbIII-CuII Single-Molecule Magnet Performance through Structural Modification. Chem.-Eur. J. 22, 12839–12848 (2016).
Liu, C.-M. et al. Hexanuclear [Ni2Ln4] clusters exhibiting enhanced magnetocaloric effect and slow magnetic relaxation. RSC Adv. 4, 53870–53876 (2014).
Casanova, D. et al. The rich stereochemistry of eight-vertex polyhedra: a continuous shape measures study. Chem.-Eur. J. 11, 1479–1494 (2005).
Huang, X.-C. et al. End-On Azido-Bridged 3d-4f Complexes Showing Single-Molecule Magnet Property. Inorg. Chem. 52, 7314–7316 (2013).
Kajiwara, T. et al. Structural Design of Easy-Axis Magnetic Anisotropy and Determination of Anisotropic Parameters of LnIII-CuII Single-Molecule Magnets. Chem. Eur. J. 17, 196–205 (2011).
Wen, H.-R. et al. Temperature-controlled polymorphism of chiral CuII-LnIII dinuclear complexes exhibiting slow magnetic relaxation. Dalton Trans. 44, 11191–11201 (2015).
Costes, J.-P. et al. Heterodinuclear Cu-Tb Single-Molecule Magnet. Inorg. Chem. 45, 5–7 (2006).
Zhu, Q. et al. A series of goblet-like heterometallic pentanuclear [LnIIICuII 4] clusters featuring ferromagnetic coupling and single-molecule magnet behavior. Chem. Commun. 48, 10736–10738 (2012).
Ishida, T. et al. Exchange coupling in TbCu and DyCu single-molecule magnets and related lanthanide and vanadium analogs. Dalton Trans. 41, 13609–13619 (2012).
Wen, H.-R. et al. A family of nickel-lanthanide heterometallic dinuclear complexes derived from a chiral Schiff-base ligand exhibiting single-molecule magnet behaviors. Inorg. Chim. Acta 435, 274–282 (2015).
Mydosh, J. A. Spin Glasses, An Experimental Introduction; Taylor and Francis Ltd.: London, pp. 1–256 (1993).
Cole, K. S. & Cole, R. H. Dispersion and absorption in dielectrics I. Alternating current characteristics. J. Chem. Phys. 9, 341–351 (1941).
Aubin, S. M. et al. Reduced anionic Mn12 molecules with half-integer ground states as single-molecule magnets. Inorg. Chem. 38, 5329–5340 (1999).
Mori, F. et al. Oximate-bridged trinuclear Dy-Cu-Dy complex behaving as a single-molecule magnet and its mechanistic investigation. J. Am. Chem. Soc. 128, 1440–1441 (2006).
Kuhne, I. A. et al. An octanuclear {CuII 4DyIII 4} coordination cluster showing single molecule magnet behaviour from field accessible states. Chem. Commun. 50, 1882–1885 (2014).
Liu, J.-L. et al. Two 3d-4f nanomagnets formed via a two-step in situ reaction of picolinaldehyde. Chem. Commun. 49, 6549–6551 (2013).
Xue, S. et al. Molecular Magnetic Investigation of a Family of Octanuclear [Cu6Ln2] Nanoclusters. Inorg. Chem. 53, 8165–8171 (2014).
Acknowledgements
This work was supported by the National Key Basic Re-search Program of China (2013CB933403), the National Natural Science Foundation of China (21471154 and 91022014), and the Strategic Priority Research Program of the Chinese Academy of Sciences (XDB12010103).
Author information
Authors and Affiliations
Contributions
C.-M. Liu designed the concept. C.-M. Liu wrote the paper. C.-M. Liu, D.-Q. Zhang and D.-B. Zhu carried out the experimental work, and analyzed the results and revised the paper.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Liu, CM., Zhang, DQ. & Zhu, DB. Fine Tuning the Energy Barrier of Molecular Nanomagnets via Lattice Solvent Molecules. Sci Rep 7, 15483 (2017). https://doi.org/10.1038/s41598-017-15852-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-15852-1
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.