Abstract
Gene regulation systems are mimicked by simple quantitative detection of non-nucleic acid molecular targets such as protein and metabolite. Here, we describe a one-tube, one-step real-time quantitative detection methodology for isothermal signal amplification of those targets. Using this system, real-time quantitative detection of thrombin and streptomycin, which were used as examples for protein and metabolite targets, was successfully demonstrated with detection limits of at most 50 pM and 75 nM, respectively. Notably, the dynamic range of target concentrations could be obtained for over four orders of magnitude. Thus, our method is expected to serve as a point-of-care or on-site test for medical diagnosis and food and environmental hygiene.
Similar content being viewed by others
Introduction
Gene expressions are generally subject to thermodynamic and kinetic controls based on association and dissociation of protein transcription factors to specific sites on DNA1,2. Once the initiation complex forms, RNA polymerase executes the designated transcription. Transcriptions are precisely regulated and never start without meeting particular conditions3. This mechanism for gene expression has been used in biological techniques for analysing molecular interactions, such as one-, two-, and three-hybrid systems functioning in living cells4,5,6.
Simplified testing for specific molecular targets has been gaining traction7,8,9,10,11 in view of recent back-to-back biomarker discoveries. Here, we anticipated that further applications using alternative materials might enable the construction of a simple and sensitive biomarker detection method for in vitro use. Ideally, we envisioned that, as with the real-time immuno-polymerase chain reaction (RT-IPCR) method12,13, the presence of the target molecule can be converted into the production of amplified polynucleotide strands, which can quantitatively be detected as a fluorescent signal; however, the detection reaction isothermally proceeds in a one-tube, one-step manner, i.e., without the requirement of temperature fluctuations or washing steps.
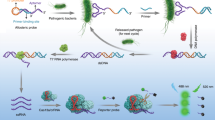
To this end, we employed split aptamers14,15,16,17,18 for target recognition and modified a φ29 DNA polymerase-catalyzed rolling circle amplification (RCA) system19 termed “signal amplification by ternary initiation complexes (SATIC)”20 as a platform of amplicon production. In principle, the formation of a four-membered initiation complex involving the target can trigger RCA, thereby generating polynucleotides containing tandemly tethered multiple G-quadruplexes that are specifically and fluorescently stained with a thioflavin T (ThT) derivative. The amplification can be chronologically monitored in a one-tube, one-step manner (Figs 1 and 2).
In the proposed system, the target (thrombin or streptomycin), capture strand (CS-thr or CS-str), and first primer (P1-thr or P1-str) associate on the circular template (cT1) to form a four-membered initiation complex to start RCA (the names and sequences of all oligonucleotides used in this study are listed in Table S1). The capture strands, CS-thr and CS-str, comprise an 11-mer and 17-mer split aptamer sequence (of thrombin and streptomycin, respectively) at the 3′-end, and a 20-mer common sequence at the 5′-end, which hybridizes to a part of cT1 (Table S1). The 3′-end of the strands is capped with monophosphates to prevent undesired extension during RCA. The primers, P1-thr and P1-str, contain a 10-mer and 21-mer split aptamer sequence at the 5′-end, respectively, and a 7-mer common sequence at the 3′-end. The 7-mer common sequence was optimized in length and sequence on the basis of nearest-neighbour thermodynamic parameters21,22. Specifically, without formation of the initiation complex, the primer (P1-thr or P1-str) cannot stably hybridize to the corresponding part on cT1, preventing extension with φ29 DNA polymerase. After the first RCA starts, the generated P1-thr- or P1-str-elongated strand attracts multiple circular templates (cT2) and, thereby, the second primer P2 is able to associate with cT2, which starts the second RCA to generate P2-elongated strands (Fig. 2). The amplicons (i.e., the P2-elongated strands) contain tandemly tethered multiple three-tiered G4s23, transcribed from cT2 that incorporate a 27-mer complementary sequence of c-Myc specifically stained with the ThT derivative (ThT-HE) (Figure S1)24,25. Overall, in principle, if the target is present, the test tubes are expected to emit fluorescence after adding the relevant reagents after maintenance of an isothermal temperature (37 °C).
Results and Discussion
Optimization of alkali metal ion concentration
Generally, the standard buffer for RCA does not contain Na+ or K+ because an increase in the salt concentration attenuates the strand displacement activity of φ29 DNA polymerase26; however, adjustment of alkali metal ion concentration was necessary. Indeed, RCA in our light-up system only worked at low concentrations up to 7.5 mM and 10 mM of Na+ and K+, respectively (Figure S2). Further, nether of the split aptamers exhibited their target-binding activities in the absence of those metal ions. As a result, RCA running and aptamer activities could simultaneously be sustained under the optimal condition of 10 mM of K+ (Figures S3 and S4).
Verification of the four-membered initiation complex formation
To verify the four-membered initiation complex formation on cT1 (Fig. 1), fluorescence resonance energy transfer (FRET) experiments were conducted using 6-carboxyfluorescein (5′-FAM)-labelled capture strands (F-CS-thr or F-CS-str) and 3′-BHQ (Black Hole Quencher 1)-labelled primers (P1-thr-B or P1-str-B) with or without the targets (thrombin or streptomycin). As shown in Fig. 3, efficient FRET quenching was observed only when all four members (cT1/F-CS-thr/P1-thr-B/thrombin or cT1/F-CS-str/P1-str-B/streptomycin) were present. These results indicate that complex formation can trigger the start of RCA and enable specific target detection using the SATIC methodology (Fig. 2).
FRET quenching from the formation of a four-membered initiation complex (target, circular template, primer, and capture strands). The FRET experiments were conducted to verify the four-membered complex formation involving (A) thrombin and (B) streptomycin. F-CS-thr or F-CS-str (spectra 1); F-CS-thr/P1-thr-B or F-CS-str/P1-str-B (spectra 2); F-CS-thr/thrombin or F-CS-str/streptomycin (spectra 3); cT1/F-CS-thr or cT1/F-CS-str (spectra 4); cT1/F-CS-thr/P1-thr-B or cT1/F-CS-str/P1-str-B (spectra 5); cT1/F-CS-thr/thrombin or cT1/F-CS-str/streptomycin (spectra 6); cT1/F-CS-thr/P1-thr-B/thrombin or cT1/F-CS-str/P1-str-B/streptomycin (spectra 7) as illustrated in (C). Only spectra 7 showed efficient FRET quenching from complex formation.
Visual and quantitative detection of a protein target
To demonstrate the feasibility of the envisioned light-up system, we first examined detection of human thrombin as a protein target. Measurement of thrombin generation, e.g., by a thrombin generation assay, has recently gained renewed attention in the clinical areas of thrombosis and haemostasis due to the increasing demand for the development of simple haemostatic ability diagnoses for individual patients against anti-coagulant drugs27. As shown in Fig. 4A, thrombin was specifically detected with greenish blue fluorescence (λex = 410 nm) and no emission was observed in test tubes containing non-targets such as lysozyme, lectin, streptavidin, and in the negative-control tube without any proteins. Without the capture strand (CS-thr), no emission was observed in the presence of thrombin, indicating that the two split aptamer strands are consistently associated by thrombin on the circular template to form the initiation complex for signal amplification by RCA. Furthermore, thrombin was clearly visualized in the presence of three non-target proteins (Fig. 4B). Next, we attempted quantitative detection of thrombin using a CFX96 real-time PCR detection system (Bio-Rad Laboratories, Inc., CA, USA). The average relative rate of reaction for each target concentration (0–5000 nM) was obtained from the increase in fluorescence intensity per unit time (Figure S5A). As shown in Fig. 4C, the logarithm of the average relative rate of reaction was linearly proportional to the logarithm of the thrombin concentration in the range of 0.050–1000 nM. Currently, the detection limit of the system is between 10 and 50 pM (i.e., at most 50 pM) for thrombin. Notably, no increase in fluorescence intensity was observed in the absence of the target, even 100 min after the start of monitoring (Figure S5). This result indicates that RCA initiation in the light-up system can be strictly regulated by the presence of the analyte.
The specific light-up system for protein target detection: photographs of the aliquots containing ThT-HE (5 μM) captured under visible light irradiation at 410 nm. Each reaction mixture was incubated at 37 °C for 2 h. (A) The aliquots in rows a and b are reaction mixtures without and with the capture strand CS-thr, respectively. Similarly, the aliquots in columns 1, 2, 3, 4, and 5 contain an analyte (1.0 nM) as described. (B) The aliquots in rows a and b are reaction mixtures without and with the target thrombin (1.0 nM), respectively. Similarly, the aliquots in columns 1, 2, 3, and 4 contain analyte(s) (1.0 nM each) as described, where + and − indicate presence and absence, respectively. Real-time quantitative analysis by the light-up system (C): linear calibration curve for target quantitation in the range from 0.050 to 1000 nM (R2 = 0.9998). Error bars reflect three separate measurements.
The 14-mer and 13-mer sequences of the thrombin-binding split aptamer imitate a thrombin-binding aptamer (TBA) consisting of a 29-mer deoxyribonucleotide (DNA) that forms a hairpin loop with a two-tiered G428,29,30,31,32. While the 29-mer TBA is well known as a high-affinity aptamer with a Kd value of 0.5 nM, the binding affinity of the split aptamer, which was divided in the middle of the G4 moiety, may be slightly degraded but was substantially retained14. Thus, as expected, our experimental results prove that the split aptamer can act as a key element for the switching machinery.
In many cases, crude biological samples contain nucleases, which may affect the outcome of target analyses using the present methodology. In φ29 buffers containing 10%, 30%, and 60% v/v human serum, 26-mer single-stranded oligodeoxyribonucleotides (T26) were greatly degraded within 2 h at 37 °C (Figure S6). From these conditions, 10% v/v human serum exhibited the strongest nuclease activity. Therefore, we attempted the specific light-up of thrombin under such conditions and confirmed that SATIC was effective, although the fluorescence intensity was somewhat decreased (Figure S7), while using a modified-type primer, P1-thr-PS, instead of a natural-type primer, P1-thr (Table S1). P1-thr-PS remained substantially intact in 10% v/v human serum for 2 h at 37 °C while P1-thr was almost entirely digested (Figure S8). These results indicate that further chemical modifications of the nucleotide components used for SATIC will yield better detection systems29,33,34,35,36.
Visual and quantitative detectio.n of a small molecular target
To demonstrate the versatility of our light-up system, we examined the specific detection of streptomycin, for which the maximum residual levels of certain animal origin products have been set and strictly monitored for food hygiene, such as small molecular targets37. The streptomycin-binding split aptamer, whose 46-mer mother aptamer is known to exhibit an apparent Kd value of 1 μM38, consists of 18-mer and 22-mer ribonucleotide (RNA) fragments. The light-up system for streptomycin detection was simply designed by replacing the sequences of the thrombin-binding split aptamer with those of the streptomycin-binding split aptamer. Namely, CS-str and P1-str as DNA/RNA chimeric oligomers were synthesized and used for the experiments instead of CS-thr and P1-thr. As with the thrombin light-up system, the target streptomycin was successfully distinguished from other coexisting metabolites such as ampicillin and kanamycin (Fig. 5A,B). Furthermore, streptomycin was quantitatively detected in the concentration range of 0.075–1000 μM when the detection limit of the system was between 0.050 and 0.075 μM (i.e., at most 0.075 μM) (Fig. 5C), which is higher than the range of thrombin attributable to the difference between the target-binding affinities of the two split aptamers.
The specific light-up system for small molecular target detection: photographs of the aliquots containing ThT-HE (5 μM) captured under visible light irradiation at 410 nm. Each reaction mixture was incubated at 37 °C for 2 h. (A) The aliquots in rows a and b are reaction mixtures without and with the capture strand CS-str, respectively. Similarly, the aliquots in columns 1, 2, 3, and 4 contain an analyte (0.10 μM) as described. (B) The aliquots in rows a and b are reaction mixtures without and with the target streptomycin (0.10 μM), respectively. Similarly, the aliquots in columns 1, 2, and 3 contain analyte(s) (0.10 μM each) as described, where + and − indicate presence and absence, respectively. Real-time quantitative analysis by the light-up system (C) linear calibration curve for target quantitation in the range from 0.075 to 1000 μM (R2 = 0.9997) for streptomycin. Error bars reflect three separate measurements.
Conclusions
We have constructed a one-tube, one-step light-up system for non-nucleic acid targets using isothermal nucleic acid amplification techniques. Furthermore, we demonstrated quantitative performance over a wide dynamic range and sequence compatibility with nucleotide type (i.e., DNA and RNA) in the aptamer parts, enabling diverse biomarker detection. Although several methods for such bioanalyses using RCA39,40,41,42,43,44,45 or loop-mediated isothermal amplification (LAMP)46,47 have been devised, the present method substantiated quantitative measurements without any operations such as annealing, washing, or transferring of samples48,49,50,51. Furthermore, introduction of chemical modifications into the nucleotide components enhanced nuclease resistance, thus offering the possibility for practical application, such as the analyses of crude biological samples. While split aptamers were employed in this study for initiating RCA, the mechanisms of riboswitches52,53,54,55,56, which are known to control gene expressions by their conformational change caused by metabolite binding to their aptamer moiety35,57,58,59,60,61, will also be applicable to this system. Thus, our concept of assembly to start amplifications will readily expand the kinds of targets that can be used for measurement and will further facilitate simple diagnoses and tests with improved accuracy.
References
Slutsky, M. & Mirny, L. A. Kinetics of protein-DNA interaction: facilitated target location in sequence-dependent potential. Biophys. J. 87, 4021–4035 (2004).
Scholes, C., DePace, A. H. & Sanchez, A. Combinatorial Gene Regulation through Kinetic Control of the Transcription Cycle. Cell Syst. 4, 97–108 (2017).
Maniatis, T. & Reed, R. An extensive network of coupling among gene expression machines. Nature 416, 499–506 (2002).
Gaudinier, A. et al. Enhanced Y1H assays for Arabidopsis. Nat. Methods 8, 1053–1055 (2011).
Lindsay, A. J. et al. Identification and characterization of multiple novel Rab-myosin Va interactions. Mol. Biol. Cell 24, 3420–3434 (2013).
Cottier, S. et al. The yeast three-hybrid system as an experimental platform to identify proteins interacting with small signaling molecules in plant cells: potential and limitations. Front. Plant. Sci. 2, https://doi.org/10.3389/fpls.2011.00101 (2011).
Wang, M. et al. Conjugating a groove-binding motif to an Ir(iii) complex for the enhancement of G-quadruplex probe behavior. Chem. Sci. 7, 2516–2523 (2016).
Ma, D. L., Lin, S., Wang, W., Yang, C. & Leung, C. H. Luminescent chemosensors by using cyclometalated iridium(iii) complexes and their applications. Chem. Sci. 8, 878–889 (2017).
Ma, D. L. et al. Metal complexes for the detection of disease-related protein biomarkers. Coord. Chem. Rev. 324, 90–105 (2016).
Wang, M., Wang, W., Kang, T. S., Leung, C. H. & Ma, D. L. Development of an Iridium(III) Complex as a G-Quadruplex Probe and Its Application for the G-Quadruplex-Based Luminescent Detection of Picomolar Insulin. Anal. Chem. 88, 981–987 (2016).
Liu, Z. et al. A Reversible Fluorescent Probe for Real-Time Quantitative Monitoring of Cellular Glutathione. Angew. Chem. Int. Ed. Engl. 56, 5812–5816 (2017).
Niemeyer, C. M., Adler, M. & Wacker, R. Detecting antigens by quantitative immuno-PCR. Nat. Protoc. 2, 1918–1930 (2007).
Sims, P. W., Vasser, M., Wong, W. L., Williams, P. M. & Meng, Y. G. Immunopolymerase chain reaction using real-time polymerase chain reaction for detection. Anal. Biochem. 281, 230–232 (2000).
Chen, J. et al. An ultrasensitive signal-on electrochemical aptasensor via target-induced conjunction of split aptamer fragments. Biosens. Bioelectron. 25, 996–1000 (2010).
Liu, X. et al. Target-induced conjunction of split aptamer fragments and assembly with a water-soluble conjugated polymer for improved protein detection. ACS Appl. Mater. Interfaces 6, 3406–3412 (2014).
Nick, T. A. et al. Stability of a Split Streptomycin Binding Aptamer. J. Phys. Chem. B 120, 6479–6489 (2016).
Lin, S. et al. Interaction of an Iridium(III) Complex with G-Quadruplex DNA and Its Application in Luminescent Switch-On Detection of Siglec-5. Anal. Chem. 88, 10290–10295 (2016).
Wang, M. et al. Label-free luminescent detection of LMP1 gene deletion using an intermolecular G-quadruplex-based switch-on probe. Biosens. Bioelectron 70, 338–344 (2015).
Johne, R., Muller, H., Rector, A., van Ranst, M. & Stevens, H. Rolling-circle amplification of viral DNA genomes using phi29 polymerase. Trends Microbiol. 17, 205–211 (2009).
Fujita, H., Kataoka, Y., Tobita, S., Kuwahara, M. & Sugimoto, N. Novel One-Tube-One-Step Real-Time Methodology for Rapid Transcriptomic Biomarker Detection: Signal Amplification by Ternary Initiation Complexes. Anal. Chem. 88, 7137–7144 (2016).
Sugimoto, N., Nakano, S., Yoneyama, M. & Honda, K. Improved thermodynamic parameters and helix initiation factor to predict stability of DNA duplexes. Nucleic Acids Res. 24, 4501–4505 (1996).
Muhuri, S., Mimura, K., Miyoshi, D. & Sugimoto, N. Stabilization of three-way junctions of DNA under molecular crowding conditions. J. Am. Chem. Soc. 131, 9268–9280 (2009).
Yu, H., Gu, X., Nakano, S., Miyoshi, D. & Sugimoto, N. Beads-on-a-string structure of long telomeric DNAs under molecular crowding conditions. J. Am. Chem. Soc. 134, 20060–20069 (2012).
Kataoka, Y. et al. Minimal thioflavin T modifications improve visual discrimination of guanine-quadruplex topologies and alter compound-induced topological structures. Anal. Chem. 86, 12078–12084 (2014).
Mohanty, J. et al. Thioflavin T as an efficient inducer and selective fluorescent sensor for the human telomeric G-quadruplex DNA. J. Am. Chem. Soc. 135, 367–376 (2013).
Blanco, L. et al. Highly efficient DNA synthesis by the phage phi 29 DNA polymerase. Symmetrical mode of DNA replication. J. Biol. Chem. 264, 8935–8940 (1989).
Shen, G., Zhang, H., Yang, C., Yang, Q. & Tang, Y. Thrombin Ultrasensitive Detection Based on Chiral Supramolecular Assembly Signal-Amplified Strategy Induced by Thrombin-Binding Aptamer. Anal. Chem. 89, 548–551 (2017).
Cao, Z. & Tan, W. Molecular aptamers for real-time protein-protein interaction study. Chemistry 11, 4502–4508 (2005).
Kasahara, Y. et al. Capillary electrophoresis-systematic evolution of ligands by exponential enrichment selection of base- and sugar-modified DNA aptamers: target binding dominated by 2′-O,4′-C-methylene-bridged/locked nucleic acid primer. Anal. Chem. 85, 4961–4967 (2013).
Nimjee, S. M. et al. Synergistic effect of aptamers that inhibit exosites 1 and 2 on thrombin. RNA 15, 2105–2111 (2009).
Tasset, D. M., Kubik, M. F. & Steiner, W. Oligonucleotide inhibitors of human thrombin that bind distinct epitopes. J. Mol. Biol. 272, 688–698 (1997).
Bock, L. C., Griffin, L. C., Latham, J. A., Vermaas, E. H. & Toole, J. J. Selection of single-stranded DNA molecules that bind and inhibit human thrombin. Nature 355, 564–566 (1992).
Fujita, H., Nakajima, K., Kasahara, Y., Ozaki, H. & Kuwahara, M. Polymerase-mediated high-density incorporation of amphiphilic functionalities into DNA: enhancement of nuclease resistance and stability in human serum. Bioorg. Med. Chem. Lett. 25, 333–336 (2015).
Kasahara, Y., Irisawa, Y., Ozaki, H., Obika, S. & Kuwahara, M. 2′,4′-BNA/LNA aptamers: CE-SELEX using a DNA-based library of full-length 2′-O,4′-C-methylene-bridged/linked bicyclic ribonucleotides. Bioorg. Med. Chem. Lett. 23, 1288–1292 (2013).
Imaizumi, Y. et al. Efficacy of base-modification on target binding of small molecule DNA aptamers. J. Am. Chem. Soc. 135, 9412–9419 (2013).
Hagiwara, K. et al. In vitro selection of DNA-based aptamers that exhibit RNA-like conformations using a chimeric oligonucleotide library that contains two different xeno-nucleic acids. Mol. Biosyst. 11, 71–76 (2015).
Liu, B. et al. Multifunctional gold-silica nanostructures for ultrasensitive electrochemical immunoassay of streptomycin residues. ACS Appl. Mater. Interfaces 3, 4668–4676 (2011).
Wallace, S. T. & Schroeder, R. In vitro selection and characterization of streptomycin-binding RNAs: recognition discrimination between antibiotics. RNA 4, 112–123 (1998).
Ali, M. M. et al. Rolling circle amplification: a versatile tool for chemical biology, materials science and medicine. Chem. Soc. Rev. 43, 3324–3341 (2014).
Shen, J. et al. Label-free electrochemical aptasensor for adenosine detection based on cascade signal amplification strategy. Biosens. Bioelectron 90, 356–362 (2017).
Li, W., Jiang, W. & Wang, L. Self-locked aptamer probe mediated cascade amplification strategy for highly sensitive and selective detection of protein and small molecule. Anal. Chim. Acta. 940, 1–7 (2016).
Guo, Y. et al. Label-free and highly sensitive electrochemical detection of E. coli based on rolling circle amplifications coupled peroxidase-mimicking DNAzyme amplification. Biosens. Bioelectron 75, 315–319 (2016).
Bi, S., Li, L. & Zhang, S. Triggered polycatenated DNA scaffolds for DNA sensors and aptasensors by a combination of rolling circle amplification and DNAzyme amplification. Anal. Chem. 82, 9447–9454 (2010).
Liu, M., Zhang, W., Zhang, Q., Brennan, J. D. & Li, Y. Biosensing by Tandem Reactions of Structure Switching, Nucleolytic Digestion, and DNA Amplification of a DNA Assembly. Angew. Chem. Int. Ed. Engl. 54, 9637–9641 (2015).
Liu, M. et al. A graphene-based biosensing platform based on the release of DNA probes and rolling circle amplification. ACS Nano 8, 5564–5573 (2014).
Yuan, Y. et al. Ultrasensitive electrochemiluminescent aptasensor for ochratoxin A detection with the loop-mediated isothermal amplification. Anal. Chim. Acta. 811, 70–75 (2014).
Xie, S., Chai, Y., Yuan, Y., Bai, L. & Yuan, R. Development of an electrochemical method for Ochratoxin A detection based on aptamer and loop-mediated isothermal amplification. Biosens. Bioelectron 55, 324–329 (2014).
Zhu, Z. et al. Strand-Exchange Nucleic Acid Circuitry with Enhanced Thermo-and Structure- Buffering Abilities Turns Gene Diagnostics Ultra-Reliable and Environmental Compatible. Sci. Rep. 6, 36605, https://doi.org/10.1038/srep36605 (2016).
Jiang, Y. S., Li, B., Milligan, J. N., Bhadra, S. & Ellington, A. D. Real-time detection of isothermal amplification reactions with thermostable catalytic hairpin assembly. J. Am. Chem. Soc. 135, 7430–7433 (2013).
Lam, B. J. & Joyce, G. F. An isothermal system that couples ligand-dependent catalysis to ligand-independent exponential amplification. J. Am. Chem. Soc. 133, 3191–3197 (2011).
Sczepanski, J. T. & Joyce, G. F. A cross-chiral RNA polymerase ribozyme. Nature 515, 440–442 (2014).
Mehdizadeh Aghdam, E., Hejazi, M. S. & Barzegar, A. Riboswitches: From living biosensors to novel targets of antibiotics. Gene 592, 244–259 (2016).
Nutiu, R. & Li, Y. Aptamers with fluorescence-signaling properties. Methods 37, 16–25 (2005).
Schneider, C. & Suess, B. Identification of RNA aptamers with riboswitching properties. Methods 97, 44–50 (2016).
Winkler, W., Nahvi, A. & Breaker, R. R. Thiamine derivatives bind messenger RNAs directly to regulate bacterial gene expression. Nature 419, 952–956 (2002).
Cheah, M. T., Wachter, A., Sudarsan, N. & Breaker, R. R. Control of alternative RNA splicing and gene expression by eukaryotic riboswitches. Nature 447, 497–500 (2007).
Minagawa, H. et al. Selection, Characterization and Application of Artificial DNA Aptamer Containing Appended Bases with Sub-nanomolar Affinity for a Salivary Biomarker. Sci. Rep. 7, 42716, https://doi.org/10.1038/srep42716 (2017).
Velu, R., Frost, N. & DeRosa, M. C. Linkage inversion assembled nano-aptasensors (LIANAs) for turn-on fluorescence detection. Chem. Commun. 51, 14346–14349 (2015).
McKeague, M. et al. Comprehensive analytical comparison of strategies used for small molecule aptamer evaluation. Anal. Chem. 87, 8608–8612 (2015).
Matsunaga, K. I., Kimoto, M. & Hirao, I. High-Affinity DNA Aptamer Generation Targeting von Willebrand Factor A1-Domain by Genetic Alphabet Expansion for Systematic Evolution of Ligands by Exponential Enrichment Using Two Types of Libraries Composed of Five Different Bases. J. Am. Chem. Soc. 139, 324–334 (2017).
de-los-Santos-Alvarez, N., Lobo-Castañón, M. J., Miranda-Ordieres, A. J. & Tuñón-Blanco, P. Modified-RNA aptamer-based sensor for competitive impedimetric assay of neomycin B. J. Am. Chem. Soc. 129, 3808–3809 (2007).
Acknowledgements
This study was partly supported by the Basic Science and Platform Technology Program for Innovative Biological Medicine from the Japan Agency for Medical Research and Development (AMED), by a Grant for Adaptable and Seamless Technology Transfer Program through Target-Driven R & D, No. AS2525029M, from Japan Science and Technology Agency (JST), by the MEXT (Ministry of Education, Culture, Sports, Science and Technology, Japan)-Supported Program for the Strategic Research Foundation at Private Universities (2014–2019), and by the Hirao Taro Foundation of KONAN GAKUEN for Advanced Scientific Research. H.F. and Y.K. are grateful for JSPS Research Fellowships for Young Scientists.
Author information
Authors and Affiliations
Contributions
H.F. performed most of the analytical experiments and processed the data. Y.K. and R.N. contributed to chemical syntheses, data collection and assembly. Y.N., M.Y. and N.S. contributed to the analysis and interpretation of the data. M.K. contributed to the conception and design of the study and led the research process. The manuscript was written through contributions of all authors. All authors have given approval to the final version of the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Fujita, H., Kataoka, Y., Nagano, R. et al. Specific Light-Up System for Protein and Metabolite Targets Triggered by Initiation Complex Formation. Sci Rep 7, 15191 (2017). https://doi.org/10.1038/s41598-017-15697-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-15697-8
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.