Abstract
Boreal forests play critical roles in global carbon, water and energy cycles. Recent studies suggest drought is causing a decline in boreal spruce growth, leading to predictions of widespread mortality and a shift in dominant vegetation type in interior Alaska. We took advantage of a large set of tree cores collected from random locations across a vast area of interior Alaska to examine long-term trends in carbon isotope discrimination and growth of black and white spruce. Our results confirm that growth of both species is sensitive to moisture availability, yet show limited evidence of declining growth in recent decades. These findings contrast with many earlier tree-ring studies, but agree with dynamic global vegetation model projections. We hypothesize that rising atmospheric [CO2] and/or changes in biomass allocation may have compensated for increasing evaporative demand, leaving recent radial growth near the long-term mean. Our results highlight the need for more detailed studies of tree physiological and growth responses to changing climate and atmospheric [CO2] in the boreal forest.
Similar content being viewed by others
Introduction
The boreal forest is a critical component of global carbon, water and energy cycles1. However, recent studies point to increasing wildfire2, decreasing landscape greenness3,4, greater tree mortality5 and declining spruce growth6,7,8,9,10,11. Over the last two decades, numerous tree-ring studies have been completed in interior Alaska, with most concluding that declining growth of black (Picea mariana) and white spruce (Picea glauca) is a result of drought stress, induced by rising air temperature and increasing evaporative demand6,7,8,9,10,11,12,13,14,15. The evidence for drought stress is derived from negative correlations between growing season air temperature and growth, positive correlations between precipitation and growth and decreasing carbon isotope discrimination in tree-rings. The common conclusion that growth of black and white spruce is declining in interior Alaska as a result of temperature-induced drought stress has led some investigators to predict widespread spruce mortality and suggest that coniferous forests of interior Alaska are in the midst of a transition to temperate forests and/or grasslands8,11.
There is a wide range of variation in the degree to which plants regulate water loss through stomatal closure under warm and dry conditions16. At one end of the spectrum are anisohydric species, which exhibit relatively little stomatal regulation and allow leaf water potential to decline with soil water potential under drought. Anisohydric species tend to occupy drier habitats and have xylem conduits that are more resistant to cavitation under low water potential. At the other end of the spectrum are isohydric species, which have xylem conduits that are more vulnerable to cavitation, display strong stomatal regulation of water loss and typically maintain leaf water potential within a limited range under drought. Drought-induced mortality of plants at the anisohydric end of the spectrum is more likely to occur as a result of hydraulic failure. Meanwhile, isohydric species are thought to be more prone to mortality related to reduced carbohydrate availability, as a result of prolonged stomatal closure17. Carbohydrates are essential for phloem transport, turgor maintenance, re-filling of embolized xylem and production of defensive compounds. Depletion of carbohydrate reserves by maintenance respiration during extended periods of stomatal closure could lead to mortality as a result of any of the above physiological mechanisms alone or in combination with attack by insects or pathogens18.
Spruce are relatively isohydric in the sense that complete stomatal closure and loss of hydraulic conductance both occur at high xylem water potential. In white spruce, near complete stomatal closure was observed at a xylem water potential of ~−2.0 MPa19, while 50% loss of hydraulic conductance was detected at a xylem water potential of ~−4.0 MPa20. While anisohydric species may exhibit mortality in response to extreme climate events, drought-induced mortality of isohydric species is expected to occur in response to warm and dry conditions over a longer time period, during which stomatal closure constrains the supply of photosynthate and the demands of maintenance respiration eventually exhaust carbohydrate reserves17. Because drought-induced mortality of isohydric species often occurs slowly and involves depletion of carbohydrate reserves that fuel both maintenance respiration and growth, tree-ring studies commonly show a prolonged growth decline prior to drought-induced mortality21,22,23,24. Thus, identification of a climate-driven growth decline in the tree-ring record could be a useful tool to identify isohydric species that are at risk of drought-induced mortality.
We took advantage of a large set of tree cores collected from randomly located plots across a vast area of interior Alaska (Fig. 1) and tested many of the conclusions that have been drawn from previous tree-ring studies in the region. In an earlier study, using the same tree-ring data, we examined the effect of detrending method on apparent long-term growth trends in black and white spruce25. We found that choice of detrending method had important implications for apparent growth trends and the strength of climate-growth correlations. All of the methods tested revealed a pronounced peak in growth of both species centered near 1940, providing important historical context for studies that have focused on growth trends during the second half of the 20th century. Of the detrending methods tested, signal-free multiple curve RCS26 produced the strongest and/or greatest number of significant climate-growth correlations. As observed in many previous studies, growth was negatively correlated with growing season temperature and positively correlated with August precipitation. In the present study, we carried out a more thorough evaluation of the relationships between climate and growth, used stable isotopes in tree-ring alpha-cellulose to examine potential changes in gas exchange physiology over time and analyzed a wide range of habitat variables with the aim of identifying important controls on spatial variation in recent growth of black and white spruce. Specifically, we investigated the following set of linked questions and hypotheses:
-
1.
How have correlations between growth of black and white spruce and growing season temperature and precipitation changed over the past century? We hypothesized that temperature would have a positive influence on growth in the early part of the tree-ring records and a negative influence on growth in recent decades. We anticipated that precipitation would be weakly correlated with growth early and positively correlated with growth later in our tree-ring chronologies.
-
2.
What are the forms (e.g., linear, sigmoidal, etc.) of the relationships between growth of black and white spruce and growing season temperature and precipitation? We hypothesized that the relationship between temperature and growth would be strongly non-linear, with a positive effect at low temperatures, a negative effect at high temperatures and a clear optimum at intermediate air temperatures. We anticipated an overall positive correlation between precipitation and growth.
-
3.
How has the balance between photosynthesis and stomatal conductance of black and white spruce changed over the past century? In keeping with previous results, we anticipated a large decrease in carbon isotope discrimination (Δ13C) in tree-ring alpha-cellulose from the turn of the 20th to the turn of the 21st century in black and white spruce.
-
4.
Which habitat characteristics are the most important determinants of recent growth of black and white spruce? We hypothesized that habitat characteristics that relate to moisture availability (aspect, slope, elevation, topographic position, stand density, etc.) would be the most important determinants of recent growth in both species.
-
5.
Do black and white spruce growing in “good” and “poor” habitats show different sensitivities to climate and different long-term growth trends? We hypothesized that “poor” habitats for both species would be those in which moisture availability is expected to be most limited. Further, we anticipated that growth in “poor” habitats would be more negatively correlated with air temperature and more positively correlated with precipitation than growth in “good” habitats. Finally, we expected that tree-ring chronologies constructed separately for trees growing in “poor” habitats would show the most marked recent growth declines.
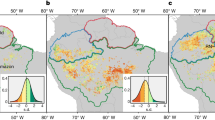
Approximate locations of the FIA and AIRIS plots where increment cores were collected in interior Alaska. Locations of Tanana, Fairbanks, Eagle, Tok and Dawson are shown for reference. The plots tend to cluster into four distinct groups, which we refer to as northwest (NW), northcentral (NC), southcentral (SC) and southeast (SE). The map was created using ArcGIS 10.4.1 (Esri, Redlands, CA). The base layer is the intellectual property of Esri and is used herein under license. Copyright © 2016 Esri and its licensors. All rights reserved.
Results
Potential Changes in Climate-Growth Relationships over Time
The growing season air temperature record (May- August) for Fairbanks shows a strong warming trend that began around 1965 (Fig. 2). August precipitation, which was identified as the precipitation variable best correlated with growth of both species25, does not show evidence of a significant trend over time in the Fairbanks station record. However, 18 of the 50 years between 1915 and 1964 had August precipitation greater than 60 mm, while only 10 of the 50 years between 1965 and 2014 had August precipitation totals exceeding that amount.
Long-term growing season air temperature and August precipitation records for Fairbanks, AK. The bottom two panels show four-curve RCS tree-ring chronologies for white and black spruce. The grey shading indicates 95% confidence intervals, the dark blue bars show variation in sample size over time, while the green shading indicates the five time periods selected for isotopic analysis. Both chronologies have a mean of 1.0 over their respective lengths.
There was limited statistical evidence that climate-growth correlations have changed over the past century for either species (Fig. 3). Warmer growing seasons have been associated with reduced growth, while more abundant August precipitation has been associated with greater growth throughout the Fairbanks climate record. One possible exception is the relationship between May air temperature and growth. Both species showed greater negative sensitivity to warm May air temperature in recent decades than in the early part of the Fairbanks climate record. However, this change in the May temperature effect was not greater than might be expected by random chance.
Moving window correlations between the black and white spruce ring width indices and growing season air temperature and precipitation. Blue shading indicates a positive correlation, red shading indicates a negative correlation and an asterisk indicates a statistically significant correlation. The P-values refer to a test of whether low frequency variation in the strength and sign of correlations over time is greater than would be expected by random chance (Zang & Biondi, 2015).
Form of Relationships between Climate and Tree Growth
Our climate-growth BRT models explained 71% of the variation in our black spruce chronology and 75% of the variation in our white spruce chronology (Fig. 4). Both models tended to slightly underestimate periods of greater growth and overestimate periods of lesser growth. Relationships between climate and tree growth were non-linear and remarkably similar for both species. Relationships between growing season temperature and growth were negative and sigmoidal in form, with growth declines observed when monthly mean temperatures passed through apparent thresholds (Fig. 4). Growth declines were generally observed over a 2–3 °C range, with growth stabilizing at lower levels under warmer air temperatures. Growth of both species showed a saturating response to increasing August precipitation. Growth was limited when August precipitation in Fairbanks was less than 40 mm, increased with rising precipitation between 40 and 80 mm and was stable at a greater level when August precipitation exceeded 80 mm. There was little evidence of strong interactions among the climate variables, suggesting that effects of warm growing seasons and low precipitation on growth were additive and not synergistic.
Partial dependence plots from BRT analyses designed to examine the form of relationships between the ring width indices and growing season air temperature and precipitation. The relative influence of each variable for each species is shown in parentheses, while the distribution of the climate data is shown by vertical bars. The final two panels show the relationship between the BRT climate-growth model predictions and the observed data for each species. While there were many years with warm growing season air temperature that help to constrain the lower tails of the sigmoidal relationships between temperature and growth, there were relatively few years with cool air temperature. The upper tails of the sigmoids are, therefore, rather poorly constrained.
Changes in Gas Exchange Physiology over Time
Random Forest regression analyses designed to isolate the effects of time period and ring age on Δ13C revealed that ring age was a more important determinant of Δ13C than time period for both black (ring age importance: 211.2, time period importance: 45.3) and white spruce (ring age importance: 171.6, time period importance: 34.3). Δ13C decreased strongly until ring age reached 50–70 years (~0.5‰ in black and ~1.5‰ in white spruce) and subsequently increased very slowly with rising ring age (~0.25‰/century in both species) (Supplemental Figure S1).
In white spruce, the lowest Δ13C was observed during and immediately following the mid-20th century growth peak, leading to an inverse relationship between growth and Δ13C (Fig. 5). For black spruce, the lowest Δ13C was observed during the 1993–2002 interval. Over the five time periods examined, Ci/Ca varied by less than 0.03 in both species. In the context of rising Ca, this relative constancy in Ci/Ca was associated with a ~50 μmol/mol increase in Ci and a ~30% increase in iWUE of both species.
Measured and modeled Δ13C plotted in relation to smoothed four-curve RCS chronologies of black and white spruce growth. Bars on the measured data are standard error. The modeled data are partial dependence plots from a Random Forest analysis designed to separate the effects of time period and ring age and show Δ13C for a ring age of 100 years. The right-hand panels show atmospheric [CO2] (Ca) (McCarroll & Loader, 2004), along with intercellular [CO2] (Ci), Ci/Ca and intrinsic water-use-efficiency (iWUE), which were calculated based on the modeled Δ13C estimates for each species.
Determinants of Spatial Variation in Recent Tree Growth
Our BRT models designed to explain spatial variation in recent growth (2003–2012) explained considerably less of the variation (23% for black and 39% for white spruce) than our models aimed at explaining temporal variation in the mean chronologies. In contrast with our temporal climate-growth analyses, which showed very small differences between species, our spatial analyses showed large differences in the habitat variables that most strongly influenced growth. For black spruce growth, the most influential variables were slope, aspect, moss cover, topographic position and stand age (Table 1), with limited evidence of strong interactions among predictors. Black spruce growth was greatest where the slope exceeded 15%, the aspect was not east-facing or flat ground, where moss cover was greater than 60%, on upper and mid-slopes and where the stand was younger (Supplemental Figure S2). For white spruce growth, the most influential variables were moss cover, duff depth, stand density and aspect. Recent white spruce growth was greater where moss cover was less than 50%, duff depth was shallow, stand density was higher and on flat ground and south-facing aspects. There was a strong interaction between duff depth and aspect. Increasing duff depth led to the greatest decline in growth on flat ground and south-facing aspects, where growth was greatest. Region, elevation and Δ13C had limited influence on spatial variation in recent growth of both species.
Construction of separate chronologies for black and white spruce growing in good and poor habitats revealed strong differences following the mid-20th century growth peak (Fig. 6). When growing in poor habitat, both species showed clear evidence of the growth peak, followed by a decline to relatively stable growth from ~1970 to 2013. Black spruce growing in poor habitat may have shown a very slight decreasing trend following the growth peak, through 2013. When growing in good habitat, the mid-20th century growth peak was less apparent for both species. Following an increase from ~1900 to ~1940, white spruce in good habitat generally maintained consistent growth through 2013. Meanwhile, black spruce growing in good habitat showed a slight increase during the early years of the 20th century with very little trend thereafter.
Chronologies of black and white spruce growing in good (light green) and poor habitats (dark green) during the 20th century. Habitat quality was determined by examining partial dependence plots from a BRT analysis relating plot locational and structural variables to recent growth of both species (Supporting Figure S4). Sample sizes for white spruce are 86 trees from good habitats and 44 trees from poor habitats, while those for black spruce are 41 trees from good habitats and 53 trees from poor habitats. The trees and habitats selected represent the extremes of the population in terms of recent growth.
Comparison of climate-growth correlations among black and white spruce growing in good and poor habitats revealed strong differences in temperature sensitivity (Table 2). Both species showed much stronger negative sensitivity to growing season air temperature when growing in poor habitats. Interestingly, white spruce growing in good habitat showed greater positive sensitivity to August precipitation than those growing in poor habitats. The opposite was true for black spruce, as trees growing in poor habitats generally showed greater positive sensitivity to growing season precipitation.
Discussion
Numerous studies have concluded that growth of black and white spruce in boreal Alaska is declining as a result of temperature-induced drought stress. The results of our study point to more nuanced responses of spruce growth to changes in climate. Our data confirm that growth of black and white spruce in the Tanana Valley is sensitive to moisture availability6,8,9,10,11,12,27,28, as evidenced by negative correlations with growing season air temperature, positive correlations with August precipitation and slight decreases in Δ13C over the past century. Fewer studies have examined the temporal stability of climate-growth relationships. Those that have29 also found that warm growing seasons and low August precipitation have been associated with limited growth of black and white spruce throughout the ~100 year Fairbanks climate record. The form of relationships between growth and growing season air temperature (sigmoidal) and August precipitation (saturating) also agree very well with results of an earlier study with a similar spatial extent13.
Our results differ from past work in several important respects. Many previous studies have produced tree-ring chronologies that show a steep growth decline during the latter half of the 20th century6,8,9,11,14. These studies either did not detrend their ring width data6,8, detrended the majority of their tree-ring series by fitting a horizontal line9,11 or focused their analysis on just the latter portion of the 20th century10,14. When exclusively old trees of similar age are sampled6,8, choosing not to detrend the data may allow the common age-related ring width decline to dominate the resulting tree-ring chronology. A similar effect can occur when trees of a wider age range are sampled, but tree-ring series are detrended by fitting a horizontal line, which eliminates variation in mean ring width across trees and allows the age-related ring width declines of both old and young trees to dominate the chronology25. Finally, while restricting the length of the chronology can be a means to avoid artifacts of the detrending process, doing so limits the opportunity to place recent growth in a historical context. Our black and white spruce chronologies both show a distinct growth peak near the middle of the 20th century, followed by a decline to relatively stable growth in recent decades. Decline from a peak may have very different ecological consequences than decline from a trough.
Relationships between growing season air temperature and growth were sigmoidal, rather than linear, suggesting that growth may stabilize at a lower level at the warm end of the observed temperature range. While the sigmoidal relationships between growing season air temperature and growth of both species are well constrained at higher temperatures, they are poorly constrained at cooler temperatures, as there were relatively few years in Fairbanks with mean July air temperature <14 °C, for instance, during the 20th century. We hypothesize that more limited growth of both species during the 19th century was associated with generally cooler air temperature than observed during the 20th century. Further, we anticipate that incorporating 19th century climate data into our analyses (if they were available) would reveal temperature optima for growth at the lower end of the air temperature range observed during the 20th century. While the flattening of the air temperature-growth relationships is well constrained at the warm end, it would not be wise to extrapolate beyond the air temperature range observed over the past century. Our results apply only to the range of climate conditions observed over the past century.
Several previous studies have examined Δ13C in rings of black and white spruce. One study restricted analysis of Δ13C in whole wood of black spruce to a ~25 year period (1979–2003) and found no evidence of a trend over time, although Δ13C was negatively correlated with growing season air temperature and positively correlated with growing season precipitation14. Another study showed overall variation of ~3.0‰ in white spruce holocellulose during the 20th century and a decline of more than ~2.0‰ from ~1930 to 19966. A third study showed a ~2.0‰ decline in Δ13C of black spruce holocellulose from ~1915 to 19968. These dramatic declines in Δ13C over time dwarf the overall changes of <0.5‰ observed in our study. The difference between our results and these earlier studies may reflect contrasting sampling designs, differences in sample size and/or geographic extent and handling of potential age-related effects, particularly for rings formed during the first 50–70 years after a tree reaches breast height.
The very small overall changes that we observed in Δ13C during the past century are consistent with the set-point theory, which predicts that plants will respond to rising atmospheric [CO2] by reducing stomatal conductance to maintain constant Ci/Ca 30. An earlier study in the northern boreal forest of Eurasia found very little change in Ci/Ca of Larix and Pinus between the periods 1861–1890 and 1961–199031. In their study, Picea showed a tendency toward lower Ci/Ca in the more recent period at some sites. Our Picea data from interior Alaska and recent data from the Brooks Range32 are consistent with results for Larix and Pinus and suggest that maintenance of constant Ci/Ca in the context of rising atmospheric [CO2] and associated changes in climate may be a common response of boreal trees at a global scale. Maintenance of near constant Ci/Ca in the context of rising atmospheric [CO2] over the past century is associated with a ~50 μmol/mol increase in intercellular [CO2] and a ~30% increase in water-use-efficiency. Greater iWUE has important implications for the cycling of water and energy in boreal forests, as it is likely associated with reduced transpiration, more limited latent heat flux and a warmer and drier boundary layer.
In our study, growth of black and white spruce showed remarkably similar responses to climate, but very different effects of habitat on spatial variation in recent growth. Black spruce were most productive when growing on a slope and when moss cover was greater than 60%. Meanwhile, white spruce were most productive where moss cover was less than 50% and when duff depth was shallow. Moss cover and duff depth are likely integrator variables that reflect underlying site conditions and may not affect tree growth directly. For example, moss cover and duff depth are likely correlated with soil temperature, soil moisture and/or depth to permafrost and it may be these other variables that more directly affect tree growth. We hypothesize that greater black spruce growth may be associated with cool, moist soils without near-surface permafrost, while greater white spruce growth may be associated with relatively warm soils.
Separate chronologies created for black and white spruce growing in good and poor habitats revealed areas of the landscape where white spruce have sustained the relatively vigorous growth observed during the mid-20th century growth peak. Meanwhile, black spruce growing on relatively flat ground with <60% moss cover in recent years showed greater negative sensitivity to temperature and slightly more positive sensitivity to growing season precipitation than other combinations of species and habitats. When Δ13C was examined separately for black spruce growing in good and poor habitats, two important differences emerged (Supplemental Figure S3). First, Δ13C was lower in black spruce growing in poor habitats, despite their lower mean elevation (Δ13C tends to decrease with increasing elevation)33. Second, there was evidence of a ~1.0‰ decline in Δ13C of black spruce growing in good habitats, but there was no evidence of a change in Δ13C of black spruce growing in poor habitats over the past century. These results are similar to observations in New Mexico, where lower elevation ponderosa pine that died in response to prolonged drought showed little variation in Δ13C during the years preceding mortality, while the higher elevation trees that survived showed strong decreases in Δ13C34. The authors concluded that the lower elevation trees were moisture limited prior to the onset of drought conditions and had a limited range over which to adjust foliar gas exchange, while higher elevation trees were able to reduce Gs in response to drought. It is somewhat counterintuitive that black spruce growing in flat or gently sloping areas might be the most sensitive to drought. When these habitats occur in upland areas, they are typically characterized by near surface permafrost that impedes drainage and leads to relatively high soil water content. However, several studies of white spruce further north in the Brooks Range have shown that Gs is reduced under low soil temperature19,35, presumably as a result of limited membrane permeability and greater water viscosity. Thus, it is possible the combination of high evaporative demand and cold permafrost-affected soils leads to conditions that are more chronically stressful than the coincidence of high evaporative demand and low soil water availability during the late summer in better drained hill slope habitats. In contrast, flat or gently sloping habitats that occur in lowland areas often exhibit patchy permafrost and sandy, well-drained soils, particularly in areas closer to the Alaska Range36. In these habitats, black spruce growth may be responding to both soil and atmospheric drought during warm and dry summers.
Some investigators have suggested that drought stress is leading to a decline in growth of black and white spruce, that widespread spruce mortality is imminent and that the boreal forest of interior Alaska is in the early stages of a shift in dominance from coniferous forests to temperate forests and/or grasslands8,11. Our data support a more nuanced interpretation that is generally more consistent with recent work in nearby Denali National Park and Preserve, where researchers have concluded that habitat diversity will likely provide resistance to a widespread transition in dominant vegetation type37,38 and with dynamic global vegetation models, which generally project greater boreal tree growth in a warmer climate39. Our results confirm that growth of black and white spruce in interior Alaska is sensitive to moisture availability, but also show that warm growing seasons with low August precipitation have been associated with limited spruce growth throughout the Fairbanks climate record. Overall, Δ13C in tree-ring alpha-cellulose showed limited variation over the past century, consistent with observations from across northern Eurasia and with the prediction that plants will respond to rising atmospheric CO2 by reducing stomatal conductance to maintain constant Ci/Ca. Collectively, our results beg the question: if spruce growth in interior Alaska is sensitive to moisture availability and the climate is becoming warmer and presumably drier, why has spruce growth not declined to historically low levels? We hypothesize that rising atmospheric [CO2] has been associated with a decrease in Gs that has left tree water use, photosynthetic CO2 uptake and growth of black and white spruce relatively unchanged, despite increasing evaporative demand. We stress that this is merely a hypothesis, but one that would benefit from testing with more detailed studies of tree physiological responses to changes in climate and atmospheric [CO2] in the boreal forest.
There are several limitations to our study. First, our results do not address the possibility that increasing wildfire frequency and severity may promote greater dominance by deciduous tree species at the expense of spruce in boreal Alaska40. Second, while isohydric species like spruce are expected to exhibit a prolonged growth decline prior to drought-induced mortality, episodic mortality events remain a possibility. Synergistic effects of unfavorable climate conditions and insect outbreaks like the spruce bark beetle outbreak of the late 1990s in southcentral Alaska41,42 could lead to episodic mortality events that would be difficult to predict based upon growth trends. Third, like all tree-ring studies, our results apply to only one aspect of growth (radial growth in the main stem), yet trees are well known to shift allocation patterns in response to changes in climate43. In that context, our results might not conflict with observations of decreasing landscape greenness in interior Alaska3. It is possible, if not likely, that black and white spruce of interior Alaska have shifted allocation from foliage to fine roots in response to increasing evaporative demand, while maintaining radial growth in the main stem near the long-term mean. Warming-induced deepening of the active layer may have facilitated greater belowground investment and the combination of deeper thaw and larger root systems may be another reason why spruce growth has not declined to a greater degree. Finally, like most tree-ring studies, our inferences are limited to individual trees that were alive when sampled and do not address changes over time in stand productivity. It is possible that mortality within the black and white spruce populations improved resource availability to surviving trees, thereby preventing growth declines. On the basis of our results and the limits to our inferences, we argue that more detailed studies of tree physiological responses to climate and atmospheric [CO2] that account for growth in all major organs and address changes in stand-level productivity are desperately needed in interior Alaska and throughout the boreal forest.
Methods
Increment Core Collection and Processing
Increment cores (1 core/tree) were collected at breast height (1.37 m) during the 2013 and 2014 growing seasons from United States Forest Service Forest Inventory and Analysis (FIA) and Alaska Integrated Resource Inventory System (AIRIS) plots in the Tanana Valley of interior Alaska (Fig. 1). FIA plots were restricted to forested land within the Tanana Valley State Forest (~730,000 ha) and Tetlin National Wildlife Refuge (~280,000 ha), while the AIRIS plots were more widely distributed. Approximately 5 cores/species were collected from each FIA plot (n = 100 plots), while approximately 10 cores/species were collected from each AIRIS plot (n = 9 plots), when the species was present on the plot. On the FIA plots, cores were collected from trees that appeared healthy and were representative of the most abundant size class. On the AIRIS plots, tree size cohorts were visually defined and cores were collected from representative trees within each cohort, regardless of apparent health. Our dataset contains a bias favoring visibly healthy trees that is lessened by incorporation of cores collected on the AIRIS plots44. Greater detail regarding the plots and the trees selected for sampling can be found in our earlier publication25.
Increment cores were air dried, mounted, sanded to 600 grit and measured to the nearest 0.001 mm using a sliding bench micrometer and digital encoder (Velmex Inc. Bloomfield, NY). Ring width data were analyzed in COFECHA to identify potential dating errors45. The pith was present in 27% of the white spruce and 40% of the black spruce cores that were included in the dataset. For cores that missed the pith, but passed close enough for the innermost ring to form a complete arc, the missing radius was estimated using the geometric method46. The number of missing rings was estimated by dividing the mean width of the first ten rings into the missing radius. Increment cores that did not include the pith or pass close enough for the innermost ring to form a complete arc were eliminated from the dataset. Additionally, 14 of the FIA plots sampled are known to have burned within the past 50 years. To limit the effect of disturbance on our tree-ring chronologies, we eliminated all post-fire ring width data from plots with a known fire history. A total of 339 white spruce and 213 black spruce cores (trees) were used to construct our tree-ring chronologies.
Detrending
Ring width data were detrended using four-curve regional curve standardization (RCS) in CRUST47. We evaluated a wide range of common detrending methods and concluded that our four-curve RCS chronologies were the most unbiased and best correlated with the climate data of the methods tested25. Multiple curve RCS involves grouping the individual tree-ring series by mean ring width (>40 series/group), fitting an empirical curve describing the age-related ring width decline for each group and then calculating indices of observed versus expected ring width as a function of age. We calculated ring width indices as ratios of observed to expected growth and assembled chronologies using Tukey’s biweight robust mean. Data were processed to produce signal-free chronologies48, in which the raw ring widths are repeatedly divided by the detrended chronology (<10 iterations) with the goal of correcting for inadvertent removal of some of the climate signal during detrending. We truncated our chronologies when the sample size dropped below 50 trees.
Contrasting sampling sampling design between the FIA and AIRIS plots could lead to differences in growth trends. It would be reasonable to hypothesize that trees sampled on AIRIS plots might show greater evidence of declining growth in recent decades, as both apparently healthy and unhealthy trees were sampled. To examine this possibility, we produced separate multiple curve RCS chronologies for each species and plot type when data for at least 25 trees contributed to the mean (Supplemental Figure S4). For both species, inter-annual and inter-decadal variation tended to be greater in trees sampled on the AIRIS plots, although this could be attributable to smaller sample sizes. There was no evidence of a greater growth decline in recent decades among trees sampled on the AIRIS plots. On the contrary, white spruce sampled on the AIRIS plots showed a strong positive growth trend since the mid-1970s. Given the nature of the differences between the FIA and AIRIS tree-ring chronologies, we elected to conduct our analyses without regard to plot type.
Potential Changes In Climate-Growth Relationships Over Time
Air temperature and precipitation data for Fairbanks, AK (1915–2013) were obtained from the Alaska Climate Research Center at the University of Alaska Fairbanks. We considered the possibility of using gridded and downscaled climate data in an effort to incorporate differences in climate means and trends across the Tanana Valley, but we were concerned about temporal heterogeneities associated with differences in station record lengths in those data sources49. Comparison of the Fairbanks growing season air temperature record with the records from five other climate stations in the interior boreal forest of Alaska and western Canada revealed good agreement in terms of inter-annual variability and long-term trends (Supplemental Figure S5).
In an earlier study using the same datasets25, we examined the strength and sign of correlations between our four-curve RCS chronologies and monthly mean air temperature (May-August) and both monthly and seasonal precipitation totals (May-August and October-April) for the full length of the Fairbanks climate record. These analyses were performed using the treeclim package50 in R 3.1.251 and included climate data for the growth year and the previous year. Here, we extended our earlier analyses to examine potential changes in the strength and sign of climate-growth correlations over time for each species using moving window analyses in treeclim. The same climate variables used in the earlier static analyses were retained for the moving window analyses. Precipitation data were log-transformed prior to analysis, because they were positively skewed. We used a 25-year moving window beginning in 1916 with a 1-year step between windows. After completing the moving window analyses, we tested whether low-frequency variations in the strength and sign of correlations between climate and growth are significantly stronger than would be expected by chance, with significance assessed using exact bootstrap resampling.
Form of Relationships between Climate and Tree Growth
The static and moving window correlation analyses are limited in the sense that they assume linear and non-interactive relationships between climate and tree growth. To examine the potential for non-linear relationships and interactions among climate variables, we conducted boosted regression tree (BRT) analyses52 separately for each species using the gbm53 and dismo54 packages in R 3.1.2. The same climate variables used in the moving window analyses were included in the BRT analyses. We used a tree complexity of 2, a learning rate of 0.001, a bag fraction of 0.5 and we set the maximum number of regression trees at 3000013. The final models were constructed using 3850 trees for white spruce and 3575 trees for black spruce. We examined the potential for interactions among climate variables and constructed partial dependence plots, which depict the modeled relationship between each climate variable and the ring width indices for each species, while holding all other variables at their mean values.
Changes in Gas Exchange Physiology over Time
Carbon isotope discrimination (Δ13C) in tree-ring alpha-cellulose was examined to provide insight into potential changes in gas exchange physiology of black and white spruce over time. Following examination of the tree-ring chronologies, five time periods of interest were identified: 1895–1904, which was a period of relatively low and stable growth, 1930–1949, when growth of both species rose to a distinct peak, 1950–1959, when growth of both species declined from the peak, 1993–2002, which was a period of recent relatively stable growth and 2003–2012, which includes one of the warmest, driest and most severe wildfire seasons in recorded history (2004). A total of 85 black and 85 white spruce trees were selected for isotopic analysis with the aim of maximizing the inter-series correlation of the selected trees, obtaining an even distribution of both species across the study area and sampling similar numbers of old and young trees. The mean inter-series correlation of the selected trees was 0.420 for black and 0.503 for white spruce. Trees were selected for isotopic analysis on all but 26 of the 109 plots. The desire to sample young trees as well as old trees reduced the sample size for the 1895–1904 period to 53 black and 51 white spruce.
The time periods of interest were separated from each increment core and homogenized by slicing into fine fragments with a razor blade. The homogenized samples were then reduced to alpha-cellulose using the water-modified Brendel method55,56. The alpha-cellulose was dried overnight at 40 °C and 0.3 mg of each sample was weighed into a tin capsule for analysis using an elemental analyzer (Costech 4010, Costech Analytical, Valencia, CA), coupled with a continuous-flow isotope ratio mass spectrometer (Thermo-Finnigan Delta Plus XP, Thermo Electron Corp., Waltham, MA) in the Environment and Natural Resource Institute’s Stable Isotope Laboratory at the University of Alaska Anchorage. Carbon isotope discrimination (Δ13C) was calculated as
where δ13Ca is the isotopic value of atmospheric CO2, which has decreased progressively as a result of fossil fuel combustion since the Industrial Revolution. Annual estimates of δ13Ca were obtained from the literature57. Data for 2003–2012 were estimated by linear extrapolation of the trend between 1993 and 2002.
Tree-ring Δ13C may be influenced by the age (size) of a tree. When a tree is young, it is likely to show greater Δ13C because it may assimilate a larger proportion of soil-respired CO2, because shade may lead to lower photosynthesis and/or because shorter trees exhibit lower resistance to xylem water flow, potentially allowing for greater stomatal conductance58. To address potential age effects on Δ13C, we conducted Random Forest regression analyses separately for each species with ring age and time period as independent variables and Δ13C as the dependent variable using the randomForest package59 in R 3.1.2. We then examined modeled Δ13C over time for each species with ring age held constant at 100 years, which was very close to the mean age of the trees selected for isotopic analysis.
To gain further insights into changes in gas exchange physiology over time, we calculated the ratio of intercellular to atmospheric [CO2] (Ci/Ca) from modeled Δ13C60:
where a is fractionation associated with diffusion of CO2 through the stomata (4.4‰), and b is fractionation during carboxylation (27‰). We then used annual estimates of Ca 57 to solve for Ci. Again, estimates of Ca for 2003 to 2012 were obtained by linear extrapolation of the trend between 1993 and 2002. The relationship between Ci/Ca and Δ13C was developed for whole leaf tissue, while our data are for tree-ring alpha-cellulose, which is enriched relative to whole wood and whole leaf tissue. To improve estimates of Ci and Ci/Ca, we applied an offset of −1.33‰ to δ13C of tree-ring alpha-cellulose32,61.
Finally, to further examine changes in the balance between photosynthesis (A) and stomatal conductance (Gs) over time, we calculated intrinsic water-use efficiency (iWUE)31:
Carbon isotope discrimination in tree-rings is widely used to assess potential changes in moisture limitation to tree growth over time. Use of Δ13C in this context is complicated by two key factors. First, changes in Δ13C over time can be influenced by shifts in either A or Gs, with changes in the former potentially masking or overriding changes in the latter. Second, there is a growing awareness that the Δ13C is actually related to the chloroplast CO2 concentration (Cc), rather than Ci, meaning that Δ13C is influenced both by Gs and by mesophyll conductance (Gm)62. While Gm generally decreases with moisture limitation63 and most studies show a positive correlation between Gs and Gm 64, there may be instances when they are not well correlated and this may add uncertainty to interpretation of trends in Δ13C over time.
Determinants of Spatial Variation in Recent Tree Growth
The FIA program collects a wide range of plot locational and structural variables. To investigate the most important drivers of spatial variation in recent growth of black and white spruce (2003–2012), we conducted BRT analyses similar to those used to examine relationships between climate and tree growth. Ring width indices were averaged for each tree over the 10-year period and related to the following variables: aspect (north, south, east, west), basal area (m2/ha), Δ13C (‰), duff depth (cm, the layer of decomposing organic matter below litter and above mineral soil), elevation (m), litter depth (cm), moss cover (%), region (northwest, northcentral, southcentral, southeast), slope (%), stand age (years), stand density (trees/ha), topographic position (upper slope, mid-slope, lower slope, alluvial flat, dry flat, wet flat) and stand type (black spruce, white spruce, paper birch (Betula neoalaskana), balsam poplar (Populus balsamifera), trembling aspen (Populus tremuloides)). The final models were constructed using 3925 regression trees for white spruce and 1090 trees for black spruce. Again, we examined the potential for interactions among plot variables and constructed partial dependence plots, which depict the modeled relationship between each variable and the ring width indices for each species, while holding all other plots variables at their mean values.
After identifying the two most influential plot locational or structural variables for each species, we examined partial dependence plots for obvious breaks or inflection points that would allow us to construct separate chronologies for black and white spruce growing in “good” and “poor” habitats. These separate chronologies represent the positive and negative extremes of our datasets with regard to recent growth. To examine the possibility that trees growing in good and poor habitats may respond to climate differently, we conducted static correlation analyses in treeclim using the same settings described above for the time period from 1916 to 2013. It is important to note that, while some plot variables are fixed (e.g., aspect, slope, elevation, etc.) others have likely varied through time (e.g., moss cover, duff depth, stand density, etc.). Our definitions of good and poor habitats relied on both fixed and temporally variable plot attributes. In the case of the latter, the underlying assumption is that recent moss cover, for instance, is a function of the edaphic characteristics of the plot, which have likely been more constant over time than moss cover itself.
Data Availability
Data presented in this article have been submitted to the International Tree-Ring Data Bank for archival.
References
Pan, Y., Birdsey, R. A. & Fang, J. et al. A large and persistent carbon sink in the World’s forests. Science 333, 988–993 (2011).
Turetsky, M. R. et al. Recent acceleration of biomass burning and carbon losses in Alaskan forest and peatlands. Nature Geoscience 4, 27–31 (2011).
Verbyla, D. The greening and browning of Alaska based on 1982–2003 satellite data. Global Ecology and Biogeography 17, 547–555 (2008).
Ju, J. & Masek, J. G. The vegetation greenness trend in Canada and US Alaska from 1984-2012 Landsat data. Remote Sensing of the Environment 176, 1–16 (2016).
Peng, C., Ma, Z. & Lei, X. et al. A drought-induced pervasive increase in tree mortality across Canada’s boreal forests. Nature Climate Change 1, 467–471 (2011).
Barber, V. A., Juday, G. P. & Finney, B. P. Reduced growth of Alaskan white spruce in the twentieth century from temperature-induced drought stress. Nature 405, 668–673 (2000).
McGuire, A. D. et al. Vulnerability of white spruce tree growth in interior Alaska in response to climate variability: dendrochronological, demographic, and experimental perspectives. Canadian Journal of Forest Research 40, 1197–1209 (2010).
Beck, P. S. A., Juday, G. P. & Alix, C. et al. Changes in forest productivity across Alaska consistent with biome shift. Ecology Letters 14, 373–379 (2011).
Juday, G. P. & Alix, C. Consistent negative temperature sensitivity and positive influence of precipitation on growth of floodplain Picea glauca in interior Alaska. Canadian Journal of Forest Research 42, 561–573 (2012).
Walker, X. & Johnstone, J. F. Widespread negative correlations between black spruce growth and temperature across topographic moisture gradients in the boreal forest. Environmental Research Letters 9, 064016 (2014).
Juday, G. P., Alix, C. & Grant, T. A. III Spatial coherence and change of opposite white spruce temperature sensitivities on floodplains in Alaska confirms early-stage boreal biome shift. Forest Ecology and Management 350, 46–61 (2015).
Lloyd, A. H. & Fastie, C. L. Spatial and temporal variability in the growth and climate response of treeline trees in Alaska. Climatic Change 52, 481–509 (2002).
Lloyd, A. H., Duffy, P. A. & Mann, D. H. Nonlinear responses of white spruce growth to climate variability in interior Alaska. Canadian Journal of Forest Research 43, 331–343 (2013).
Walker, X., Johnstone, J. F. & Mack, M. C. Stable carbon isotope analysis reveals widespread drought stress in boreal black spruce forests. Global Change Biology 21, 3102–3113 (2015).
Wolken, J. M. et al. Climate-growth relationships along a black spruce toposequence in interior Alaska. Arctic, Antarctic and Alpine Research 48, 637–652 (2016).
Tardieu, F. & Simonneau, T. Variability among species of stomatal control under fluctuating soil water status and evaporative demand: modelling isohydric and ansiohydric behaviors. Journal of Experimental Botany 49, 419–432 (1998).
McDowell, N., Pockman, W. T. & Allen, C. D. et al. Mechanisms of plant survival and mortality during drought: why do some plants survive while others succumb to drought? New Phytologist 178, 719–739 (2008).
McDowell, N. G. et al. The interdependence of mechanisms underlying climate-driven vegetation mortality. Trends in Ecology and Evolution 26, 523–532 (2011).
Sullivan, P. F. & Sveinbjörnsson, B. Environmental controls on needle gas exchange and growth of white spruce on a riverside terrace near the Arctic treeline. Arctic, Antarctic and Alpine Research 43, 279–288 (2011).
Sperry, J. S., Nichols, K. L., Sullivan, J. E. M. & Eastlack, S. E. Xylem embolism in ring-porous, diffuse-porous and coniferous trees of northern Utah and interior Alaska. Ecology 75, 1736–1752 (1994).
Pedersen, B. S. The role of stress in the mortality of Midwestern oaks as indicated by growth prior to death. Ecology 79, 79–93 (1998).
Suarez, M. L., Ghermandi, L. & Kitzberger, T. Factors predisposing episodic drought-induced tree mortality in Nothofagus – site, climatic sensitivity and growth trends. Journal of Ecology 92, 954–966 (2004).
Jump, A. S., Hunt, J. M. & Peñuelas, J. Rapid climate change-related growth decline at the southern range edge of Fagus sylvatica. Global Change Biology 12, 2163–2174 (2006).
Liu, H., Williams, A. P. & Allen, C. D. et al. Rapid warming accelerates tree growth decline in semi-arid forests of Inner Asia. Global Change Biology 19, 2500–2510 (2013).
Sullivan, P. F., Pattison, R. R., Brownlee, A. H., Cahoon, S. M. P. & Hollingsworth, T. N. Effect of tree-ring detrending method on apparent growth trends of black and white spruce in interior Alaska. Environmental Research Letters 11, 114007 (2016).
Briffa, K. R. & Melvin, T. M. A closer look at regional curve standardization of tree-ring records: justification of the need, a warning of some pitfalls, and suggested improvements in its application. In: Dendroclimatology: Progress and Prospects. (eds Hughes, M. K., Diaz, H. F. & Swetnam, T. W.), pp. 113–145, Springer, Berlin (2011).
Yarie, J., Van Cleve, K. & Schlentner, R. Interaction between moisture, nutrients and growth of white spruce in interior Alaska. Forest Ecology and Management 30, 73–89 (1990).
Yarie, J. & Van Cleve, K. Long-term monitoring of climatic and nutritional effects on tree growth in interior Alaska. Canadian Journal of Forest Research 40, 1325–1335 (2010).
Barber, V. A., Juday, G. P., Finney, B. P. & Wilmking, M. Reconstruction of summer temperatures in interior Alaska from tree-ring proxies: evidence for changing synoptic climate regimes. Climatic Change 63, 91–120 (2004).
Ehleringer, J. R. & Cerling, T. E. Atmospheric CO2 and the ratio of intercellular to ambient CO2 concentrations in plants. Tree Physiology 15, 105–111 (1995).
Saurer, M., Siegwolf, R. T. W. & Schweingruber, F. H. Carbon isotope discrimination indicates improving water-use efficiency of trees in northern Eurasia over the last 100 years. Global Change Biology 10, 2109–2120 (2004).
Brownlee, A. H., Sullivan, P. F., Csank, A. Z., Sveinbjörnsson, B. & Ellison, S. B. Z. Drought-induced stomatal closure probably cannot explain divergent white spruce growth in the Brooks Range, Alaska, USA. Ecology 97, 145–159 (2016).
Körner, C., Farquhar, G. D. & Wong, S. C. Carbon isotope discrimination by plants follows latitudinal and altitudinal trends. Oecologia 88, 30–40 (1991).
McDowell, N., Allen, C. D. & Marshall, L. Growth, carbon-isotope discrimination, and drought-associated mortality across a Pinus ponderosa elevational transect. Global Change Biology 16, 399–415 (2010).
Goldstein, G. H., Brubaker, L. B. & Hinckley, T. M. Water relations of white spruce (Picea glauca (Moench) Voss) at tree line in north central Alaska. Canadian Journal of Forest Research 15, 1080–1087 (1985).
Nowacki, G. J., Spencer, P., Fleming, M., Brock, T. & Jorgenson, T. Unified ecoregions of Alaska: 2001. U.S. Geological Survey Open-File Report 02–297 (2003).
Roland, C. A., Schmidt, J. H. & Nicklen, E. F. Landscape-scale patterns of tree occupancy and abundance in subarctic Alaska. Ecological Monographs 83, 19–48 (2013).
Nicklen, E. F., Roland, C. A., Ruess, R. W., Schmidt, J. H. & Lloyd, A. H. Local site conditions drive climate-growth responses of Picea mariana and Picea glauca in interior Alaska. Ecosphere 7, e01507 (2016).
Tei, S. et al. Tree-ring analysis and modeling approaches yield contrary response of circumboreal forest productivity to climate change. Global Change Biology, 10.111/gcb.13780 (2017).
Johnstone, J. F., Hollingsworth, T. N., Chapin, F. S. III & Mack, M. C. Changes in fire regime break the legacy lock on successional trajectories in Alaskan boreal forest. Global Change Biology 16, 1281–1295 (2010).
Berg, E. E., Henry, J. D., Fastie, C. L., De Volder, A. D. & Matsuoka, S. M. Spruce beetle outbreaks on the Kenai Peninsula, Alaska, and Kluane National Park and Reserve, Yukon Territory: Relationship to summer temperatures and regional differences in disturbance regimes. Forest Ecology and Management 227, 219–232 (2006).
Csank, A. Z., Miller, A. E., Sheriff, R. L., Berg, E. E. & Welker, J. M. Tree-ring isotopes reveal drought sensitivity in trees killed by spruce beetle outbreaks in south-central Alaska. Ecological Applications 26, 2001–2020 (2016).
Lapenis, A., Shvidenko, A., Shepaschenko, D., Nilsson, S. & Aiyyer, A. Acclimation of Russian forests to recent changes in climate. Global Change Biology 11, 2090–2102 (2005).
Sullivan, P. F. & Csank, A. Z. Contrasting sampling designs among archived datasets: implications for synthesis efforts. Tree Physiology 36, 1057–1059 (2016).
Holmes, R. L. Computer-assisted quality control in tree-ring dating and measurement. Tree-Ring Bulletin 43, 69–78 (1983).
Duncan, R. An evaluation of errors in tree age estimates based on increment cores in Kahikatea (Dacrycarpus dacrydiodes). New Zealand Natural Sciences 16, 31–37 (1989).
Melvin, T. M. & Briffa, K. R. CRUST: Software for the implementation of Regional Chronology Standardisation: Part 1. Signal-free RCS. Dendrochronologia 32, 7–20 (2014).
Melvin, T. M. & Briffa, K. R. A. “signal-free” approach to dendroclimatic standardisation. Dendrochronologia 26, 71–86 (2008).
McAfee, S., Guentchev, G. & Eischeid, J. Reconciling precipitation trends in Alaska: 2. Gridded data analyses. Journal of Geophysical Research Atmospheres 119, 13820–13837 (2014).
Zang, C. & Biondi, F. treeclim: an R package for the numerical calibration of proxy-climate relationships. Ecography 38, 1–6 (2015).
R Core Team R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria (2014).
Elith, J., Leathwick, J. R. & Hastie, T. A working guide to boosted regression trees. Journal of Animal Ecology 77, 802–813 (2008).
Ridgeway, G. gbm: Generalized Boosted Regression Models. R package version 2.1 (2013).
Hijmans, R. J., Phillips, S., Leathwick, J. & Elith, J. dismo: Species distribution modeling. R package version 0, 9–3 (2013).
Brendel, O., Iannetta, P. P. M. & Stewart, D. A rapid and simple method to isolate pure alpha-cellulose. Phytochemical Analysis 11, 7–10 (2000).
Gaudinski, J. B., Dawson, T. E. & Quideau, S. et al. Comparative analysis of cellulose preparation techniques for use with 13C, 14C, and 18O isotopic measurements. Analytical Chemistry 77, 7212–7224 (2005).
McCarroll, D. & Loader, N. J. Stable isotopes in tree rings. Quaternary Science Reviews 23, 771–801 (2004).
Francey, R. J. & Farquhar, G. D. An explanation of 13C/12C variation in tree rings. Nature 297, 28–31 (1982).
Liaw, A. & Wiener, M. Classification and regression by randomForest. R News 2, 18–22 (2002).
Farquhar, G. D., O’Leary, M. H. & Berry, J. A. On the relationship between carbon isotope discrimination and the inter-cellular carbon-dioxide concentration in leaves. Australian Journal of Plant Physiology 9, 121–137 (1982).
Borella, S., Leuenberger, M., Saurer, M. & Siegwolf, R. Reducing uncertainties in δ13C analysis of tree rings: pooling milling and cellulose extraction. Journal of Geophysical Research 103, 19,519–19,526 (1998).
Seibt, U., Rajabi, A., Griffiths, H. & Berry, J. A. Carbon isotopes and water use efficiency: sense and sensitivity. Oecologia 155, 441–454 (2008).
Flexas, J., Ribas-Carbó, M., Diaz-Espejo, A., Galmés, J. & Medrano, H. Mesophyll conductance to CO2: current knowledge and future prospects. Plant, Cell and Environment 31, 602–621 (2008).
Flexas, J., Niinemets, Ü. & Gallé, A. et al. Diffusional conductances to CO2 as a target for increasing photosynthesis and photosynthetic water-use efficiency. Photosynthesis Research 117, 45–59 (2013).
Acknowledgements
This study was funded by two “Focus Area” awards to RRP, TNH and H. Andersen from the Pacific Northwest Research Station (PNWRS), by a NASA Carbon Monitoring System (CMS) grant and by the PNWRS’s Resource Monitoring Assessment Program. HA, B. Cook, D. Morton, R. Nelson and A. Finley assisted in acquiring CMS funding. Involvement of PFS, AHB and SMPC was funded through a joint venture agreement #14-JV-11261919-030 between the PNWRS and the University of Alaska Anchorage. J. Hollingsworth and S. Frost collected increment cores on the AIRIS plots. JH made Figure. 1.
Author information
Authors and Affiliations
Contributions
R.R.P. and T.N.H. oversaw collection of the increment cores; A.H.B. participated in sampling and measured ring widths; S.M.P.C. measured pith offsets; P.F.S. analyzed the data and wrote the manuscript; all authors contributed to revisions.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Sullivan, P.F., Pattison, R.R., Brownlee, A.H. et al. Limited evidence of declining growth among moisture-limited black and white spruce in interior Alaska. Sci Rep 7, 15344 (2017). https://doi.org/10.1038/s41598-017-15644-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-15644-7
This article is cited by
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.