Abstract
Video analysis of animal behaviour is widely used in fields such as ecology, ecotoxicology, and evolutionary research. However, when tracking multiple animals, occlusion and crossing are problematic, especially when the identity of each individual needs to be preserved. We present a new algorithm, ToxId, which preserves the identity of multiple animals by linking trajectory segments using their intensity histogram and Hu-moments. We verify the performance and accuracy of our algorithm using video sequences with different animals and experimental conditions. The results show that our algorithm achieves state-of-the-art accuracy using an efficient approach without the need of learning processes, complex feature maps or knowledge of the animal shape. ToxId is also computationally efficient, has low memory requirements, and operates without accessing future or past frames.
Similar content being viewed by others
Introduction
Animal behaviour is important in many research fields such as ecology, medicine, neurology, ecotoxicology or evolutionary research1. In these fields, automatic tracking often relies on video tracking software to detect animal positions in controlled arenas2,3. While several methods provide a reliable tool for tracking one single individual4,5, preserving the identity of multiple individuals after an occlusion remains a challenging problem1, see Fig. 1 for an example. The complexity of this problem is illustrated in Pérez-Escudero et al.6, in a scenario where they solved correctly 99% of all crossings, but when considering error propagation only 11% of the animals were correctly identified after 2 minutes of tracking. Many state-of-the-art techniques, however, usually report a much lower accuracy. For example, in Itskovits et al.4 a multiple animal tracker system is proposed, which is able to solve only 77% of crossings between 2 animals.
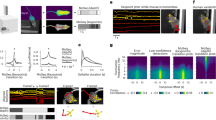
Occlusions. When animals cross or overlap, general tracking algorithms cannot preserve the identity of each individual; this scenario is called an occlusion. Occlusion examples are provided from the following datasets used in this study: (a) Mice326, (b) Zebrafish1118, (c) Zebrafish56, (d) Ant56. For the occlusion shown in (c), trajectory fragments before and after the occlusion are shown in (e). To solve the occlusion, the correct fragments should be merged. An incorrect identification is shown in (f) and the correct identification is shown in (g).
To address the occlusion problem, some techniques tag the organisms with a visual marker to preserve their identity7. These solutions, however, may be invasive and are not applicable to the vast majority of animals. Other methods rely on improving the detection by using several cameras with different perspectives8,9. This technique adds complexity to the experimental setup and dramatically increases the amount of data generated. To improve detection and tracking, some techniques use a specific model of the animal body based on the head shape10,11, the body geometry12,13,14 or the symmetry axis15. Additionally, some authors discuss the use of features such as face properties16 or bilateral symmetry17. Nevertheless these methods can only be applied for animals geometrically compatible with the used model.
Other approaches to reduce occlusion problems rely on pattern recognition, matching specific texture maps6 or using convolutional neural networks18 to identify the animals. These techniques are computationally and memory expensive and require access to past and future frames (offline tracking). Thus, offline tracking is generally slow and cannot be applied in real-time, streaming applications, or other situations where only the current frame can be accessed. In these situations the use of an online tracking is required19.
To overcome the mentioned limitations - animal specific algorithms, slow algorithms that operate only offline, and low accuracy - we developed a new online algorithm called ToxId. ToxId can handle a large variety of animals and it does not use future or past frames, thus it can be used as a post processing stage in real-time applications. We show that ToxId achieves the same accuracy as the best state-of-the-art algorithm, also when considering error propagation. ToxId identifies animals in 97% of the frames and achieves this using the intensity histogram and the Hu-moments of detected animals. ToxId requires no training or complex configuration steps, it does not use features or characteristic fingerprint-like maps, and it requires significantly less memory than other algorithms6,18. ToxId is implemented in the free tracking software ToxTrac 5, which allows the user to extract locomotor activity such as; average speed, acceleration and distance traveled per time unit. In addition, it can also measure the time an organism spends near aquaria or terrarium walls. The software, the user manual and the documentation are available at https://toxtrac.sourceforge.io.
Results and Discussion
ToxId can be included into the workflow of any online tracking algorithm and can detect and handle occlusions of multiple animals in an efficient way. When an occlusion takes place, ToxId cuts the tracked trajectories of the animals involved in the occlusion. The resulting trajectory fragments then needs to be fused in order to reconstruct the complete movement of each animal. To do this in a fast and reliable manner, ToxId extracts a set of visual features and positions of each animal in every frame and, at the end of the process, uses these features to fuse the fragments that have the highest probability to originate from the same individual. The features and positions of the animals are the only information needed to solve the occlusions, thus making the algorithm very computational efficient. We implemented ToxId in the free tracking software ToxTrac to run the analysis and we recommend not to use more than 10–20 animals in a single experiment to have reliable tracking.
To validate ToxId, we analyse multiple animals of different species under different experimental conditions and with different video resolutions and framerates. The main characteristics of these datasets, which are in total 48 minutes long and consist of approximately 80,000 frames, are summarized in Table 1. We first generate trajectory fragments from each video, where each fragment f J is formed by a set of samples \(\{{d}_{J0},\ldots ,{d}_{Jn}\}\), representing consecutive animal detections. Thereafter, we compare how ToxId merges trajectory fragments with a manually labelled ground truth. We define the validation metrics by counting the number of samples of correctly and incorrectly assigned fragments. The validation procedure is in short done as follows:
-
We chose the first detected fragment of the animals as their reference identity.
-
The fragments subsequently assigned by ToxId are marked as correct if they correspond to the same animal as the reference identity, and marked as incorrect otherwise. The remaining fragments are marked as not assigned.
To quantify the algorithm performance we then use the following classification; the Identity Error Rate (IER) defined in equation (1), the Correct Fragment Rate (CFR) defined in equation (2), and the Correct Sample Rate (CSR) defined in equation (3).
CF, IF, NF represent the correct, incorrect and unassigned fragments; and \(|{\rm{CF}}|,|{\rm{IF}}|,|{\rm{NF}}|\) represent the corresponding number of samples. We discard short fragments without a sufficient number of samples (less than 25), and for the IER, we discard fragments that are shorter than one second.
A direct quantitative comparison with most published algorithms is not possible since different algorithms create the trajectory fragments differently. For example, tracking small features of the animal head will result in less occlusions than tracking the whole body, regardless of the occlusion solving strategy. Additionally, different algorithms cannot usually be applied to the same animals (restrictions in the animal shape) or to the same video (restrictions in resolution due to memory requirements). We therefore compare the performance of different techniques using the reported CSR when possible and the CFR or IER in other cases (Fig. 2).
Note that we use more strict CFR and ER definitions for ToxId than for other techniques. The reason for this is that other techniques do not consider error propagation. Thus, for ToxId a fragment is marked as correct if it is assigned to the original animal, while the compared techniques count correct and incorrect identifications independently for each crossing. In other words, we count identifications as incorrect if the same animal is identified before and after the crossing but the identity before the crossing was incorrect due to a previous error, other techniques will however count this as correct.
ToxId obtains a CSR of 97%, a CFR of 94% and an IER of 0.22 (Table 2). This is in line with the best published algorithm, Idtracker 6, which results are discussed in the next paragraph. ToxId achieves very good results when processing fish and aquatic invertebrates (e.g. waterlouse), but does not perform equally well with ants and cockroaches. Thus, if we consider these datasets separately (Ant5, Cockroach3 and WingedAnt7) ToxId obtains a CSR of 94%, a RCF of 85% and an IER of 0.40. For the other datasets ToxId obtains a CSR of 99%, a CFR of 96% and a IER of 0.12. We explain this difference by the complex orientations and deformations of ant and cockroach bodies combined with a high similarity between individuals. We also find that ToxId achieves a very good accuracy for a wide range of video resolutions and animal sizes, regardless of the illumination conditions, as long as conditions are uniform and constant. Video sequences with backlight illumination and opaque organisms of the same size are slightly more problematic to track since this illumination type hides texture differences of the organisms. To reproduce these results follow the instructions in the supplementary materials.
The Idtracker 6 occlusion handling algorithm is based on extracting feature maps that code the texture and act as characteristic fingerprints of each animal. This algorithm has been tested with several datasets of different animals, reporting a CSR of 99%. This approach, however, requires the access to past and future frames and cannot be applied for scenarios where online tracking is required19. Furthermore, Idtracker requires a huge number of samples for each individual to work (3,000 samples are recommended). Also, this method has been criticized for being limited to small animal groups and for requiring long video sequences to acquire enough reference frames18.
Rectangles 14 uses a chain of rectangles model to represent fish-like bodies. They obtained a mean CFR of 90% using two video datasets of 2,000 frames with 10 and 20 fish. These sequences are too short to measure the algorithm behaviour over time and do not consider error propagation.
HeadEnd 11 uses the head position and orientation of fish-like animals to predict their future positions. This method is tested using two short video datasets of 2,000 frames with 20 and 40 fishes. An average IER of 0.31 is achieved. They compared these results with: Prediction-Matching 10, with a IER of 0.55; Nearest-Neighbor 20, with a IER of 2.27; and Idtracker 6, with a IER of 0.23. We conclude that the dataset used in this comparison was too small and no error propagation was considered. Therefore, these results have a limited validity and are difficult to compare to more thorough tests.
CNN 18 uses fish head detection algorithm and a Convolutional Neural Network to match the fish heads with the animals. Five video datasets (from 2,000 to 15,000 frames) with 5 to 25 fishes were used to test this algorithm and they reached an IER of 1.58. This algorithm is rather slow and cannot be applied for real-time processing, though it is reportedly able to process more individuals than Idtracker 6. However, it has the disadvantages of being one of the less accurate algorithms, only applicable to fish, and very inefficient, since it relies on the use of neural networks, which require a training stage and are generally slow. Additionally, they did not consider error propagation in their reported results.
Finally, the MAT 4 technique is an extension of the Kalman filter21 to predict the future position of multiple animals. They validated their algorithm by analysing two worms crossings, reporting a CFR of 77%. However, they did not consider error propagation in the reported results.
When considering proposed online algorithms that can handle occlusions4,10,11,14, we find that they usually can only be applied to a specific kind of animal10,11,14 or have significant accuracy problems when solving occlusions4,20. In contrast, offline techniques6,18 can in theory achieve a significantly higher accuracy than online techniques by accessing future frames of the sequence. ToxId however, achieves a CSR (97%) similar to the best technique, Idtracker 6 (99%), without accessing future or past frames. Furthermore, Idtracker also requires the use of complex texture maps with a heavy memory and computational cost. For example, to extract the texture maps of a sample of n pixels, Idtracker computational complexity22 is O(n 2)(has order of n 2 complexity), and the memory required is O(n). ToxId computational complexity is only O(n), and its required memory is O(1). This means that the Idtracker computational cost grows with a quadratic rate according to the pixel-number of the animal and its memory requirements grow linearly. ToxId computational cost grows only linearly with the pixel-number of the animal and its memory requirements do not grow.
Conclusion
Quantitative analysis of animal behaviour is important in many fields1. These studies often generate a vast amount of data and many individuals need to be tracked simultaneously. Therefore, automatic techniques that can detect and track multiple organisms with accuracy, and at the same time are able preserve the identity of each individual, need to be developed. However, keeping the identity of the animals in a reliable way has proven problematic.
Current strategies to solve the identity problem use either an expensive strategy of texture analysis which requires access to past and future frames6, rely on features only valid for a particular kind of animal11,14, or combine both strategies18. Our new algorithm (ToxId) overcomes these limitations. ToxId requires no information of the shape of the animal, does not access to past or future frames and has low memory and computational costs.
We validated ToxId using 8 datasets with multiple animals of different species and in different experimental conditions by implementing ToxId in the latest version of the free tracking software, ToxTrac. The results show that, in most cases, ToxId achieves the same accuracy as the best state-of-the-art algorithm but with a significantly faster computational speed. We believe that ToxId represent a significant contribution in the study of multiple interacting organisms, as it overcomes some fundamental problems of current techniques.
Methods
Trajectory fragment creation
Animals are detected as dark moving objects in a constant bright background using a threshold intensity value defined by the user, who also introduces the number of animals in the experiment. The obtained objects or blobs are filtered by size to remove false-positives. During the detection, a number of image features representing each object are saved. Numerically, for a body B formed by a set of pixels {p}, we define the detection d B as follows:
where CM B is the center of mass of B 23; S B is the number of pixels of B and represents its size; and \({H}_{B,kn}\) is the histogram of B, calculated by normalizing from 0 to 255 the rectangular area enclosing B, we denote this as ROI; Hu B represent the Hu’s Seven Moments Invariants24 of B; and t B represents the time of detection. Hu’s moment invariants are used to characterize patterns in images and they consists of six absolute orthogonal invariants and one skew orthogonal invariant that are calculated using weighted averages of the image intensity. The seven values characterize the image intensity distribution regardless of its location, scale and rotation. Therefore, it is very efficient for identifying rigid and moving objects regardless their orientation in the image.
To assign new detections d i to existing trajectory fragments formed by sets of previous detections \({f}_{J}=\{{d}_{J0},\mathrm{...},{d}_{Jn}\}\), we use the Kalman21 algorithm. Thus, to every pair fragment–detection, we estimate the change in the size \(({\rm{\Delta }}{S}_{i,Jn})\) and the Euclidean distance from the detection to the predicted fragment position the \(({{\rm{cost}}}_{i,J})\) as follows:
Detections are assigned to fragments minimizing globally the costs with an Hungarian optimization algorithm25. To prevent assigning detection to a fragment incorrectly, the fragment is marked as inactive if the corresponding cost or change in size is higher than a certain limit, or if the fragment has not been assigned to any detection for a certain number of frames. We also mark fragments as inactive when two fragments are too close to the same detection.
Fragment similarity
The goal of the algorithm is to connect the trajectory fragments that belong to the same individual; for simplicity, we note two fragments of the same individual with the equal sign. The first step is to construct a square matrix IdMatrix of size NxN, where N is the number of fragments and where the element \(I{d}_{R,C}\) in the row R and column C, is a value representing the likelihood of \({f}_{R}={f}_{C}\). We initialize IdMatrix as an identity matrix and then set the values that are incompatible with each other (for example the fragments that coexist at a certain moment time) to 0. For the remaining values we will estimate a value \(P({f}_{R},{f}_{C})\), this is expressed as follows:
where \(P({f}_{R},{f}_{C})\) is the correlation of the similarity values Sim of f R and f J with all the fragments in the matrix. The idea behind this is that if two fragments belong to the same individual, not only their Sim value will be high, but also their Sim values will be similar regarding the remaining fragments. This can be expressed as follows:
where R is the Pearson correlation coefficient, and the similarity function between two fragments is defined as follows:
The first factor A represents a similarity amplitude equal to the correlation maximum of the histograms of the two fragments. It is expressed as,
The second factor of the similarity function, \(\sum {w}_{L}{r}_{L}\), represents a normalized distribution of the histogram correlation. This distribution uses L n levels and is weighted by a Gaussian function. This is expressed as,
where μ and σ are the mean and the standard deviation of the Gaussian curve, with default values of 1 and 0.05 respectively, and Rn × Cn is the total number of correlations. Thus, r L is the rate of histogram correlations defined by L, and w L is a weight assigned to this rate. In practice, we do not calculate the correlation of all samples, but only of those with a difference in shape and size below a certain value. We estimate the difference in shape of two samples as follows:
Fragment assignment
To assign the different tracks to each other, the first step is to divide the fragments into long and short fragments according to a limit selected by the user (50 samples by default) and then choosing the groups of long fragments where all individuals are observed. Since this is an assignment problem with an optimal solution we use the classic Hungarian optimization algorithm25. To assign the remaining tracks, we iteratively select the best correlation value in the idMatrix, first with the long and then with the short tracks, till we reach a minimum correlation value selected by the user. With every assignment, we update the idMatrix and propagate the knowledge we obtain in each iteration, thus reducing the uncertainty for the remaining fragments. The fragment assignment algorithm workflow is shown in Fig. 3.
ToxId workflow. The matrix idMatrix contains the similarity between every two trajectory fragments. The fragments are divided between short and long according to the number of samples they contain. The assignment algorithm first uses a Hungarian optimization to assign groups of long fragments where all individuals are present. The remaining fragments are assigned one by one from higher to lower similarity.
Data availability
The ToxId algorithm has been implemented in the free tracking software ToxTrac 5, available at https://toxtrac.sourceforge.io v.2.70. The datasets analysed during the current study are available for public download at https://toxtrac.sourceforge.io and to reproduce the results in this work follow the steps provided in the Reproducing the results section in the supplementary materials. If you have problems accessing the files please contact the corresponding author.
Statement
All experiments and methods were performed in accordance with relevant guidelines and regulations. All procedures and experimental protocols were conducted as stated and permitted by the Ethical Committee on Animal Experiments in Umeå (license no A41-12) and comply with current Swedish law.
References
Delcourt, J., Denoël, M., Ylieff, M. & Poncin, P. Video multitracking of fish behaviour: a synthesis and future perspectives. Fish Fish. 14, 186–204 (2013).
Rodriguez, A. et al. Optical fish trajectory measurement in fishways through computer vision and artificial neural networks. J. Comput. Civ. Eng. 25, 291–301 (2011).
Rodriguez, A., Bermúdez, M., Rabuñal, J. & Puertas, J. Fish tracking in vertical slot fishways using computer vision techniques. J. Hydroinformatics (2014).
Itskovits, E., Levine, A., Cohen, E. & Zaslaver, A. A multi-animal tracker for studying complex behaviors. BMC Biol. 15, 29 (2017).
Rodriguez, A. et al. ToxTrac: a fast and robust software for tracking organisms. Methods Ecol. Evol. 2017, 1–5 (2017).
Pérez-Escudero, A., Vicente-Page, J., Hinz, R. C., Arganda, S. & de Polavieja, G. G. idTracker: tracking individuals in a group by automatic identification of unmarked animals. Nat. Methods 11, 743–748 (2014).
Crall, J. D., Gravish, N., Mountcastle, A. M. & Combes, S. A. BEEtag: a low-cost, image-based tracking system for the study of animal behavior and locomotion. PLoS One 10, e0136487 (2015).
Saberioon, M. M. & Cisar, P. Automated multiple fish tracking in three-dimension using a structured light sensor. Comput. Electron. Agric. 121, 215–221 (2016).
Maaswinkel, H., Zhu, L. & Weng, W. Using an automated 3D-tracking system to record individual and shoals of adult zebrafish. JoVE (Journal Vis. Exp.) e50681–e50681 (2013).
Qian, Z. M., Cheng, X. E. & Chen, Y. Q. Automatically Detect and track multiple fish swimming in shallow water with frequent occlusion. PLoS One 9, (2014).
Qian, Z.-M., Wang, S. H., Cheng, X. E. & Chen, Y. Q. An effective and robust method for tracking multiple fish in video image based on fish head detection. BMC Bioinformatics 17, 251 (2016).
Maitra, P., Schneider, S. & Shin, M. C. Robust bee tracking with adaptive appearance template and geometry-constrained resampling. in Workshop on Applications of Computer Vision (WACV) 1–6 (2009).
Clausen, S. et al. Automatic segmentation of overlapping fish using shape priors. Scandinavian conference on image analysis 11–20 (2007).
Wang, S. H., Cheng, X. E., Qian, Z.-M., Liu, Y. & Chen, Y. Q. Automated planar tracking the waving bodies of multiple zebrafish swimming in shallow water. PLoS One 11, e0154714 (2016).
Fontaine, E. et al. Automated visual tracking for studying the ontogeny of zebrafish swimming. J. Exp. Biol. 211, 1305–1316 (2008).
Burghardt, T. & Ćalić, J. Analysing animal behaviour in wildlife videos using face detection and tracking. IEE Proceedings-Vision, Image Signal Process. 153, 305–312 (2006).
Rodriguez, A. et al. Refining particle positions using circular symmetry. PLoS One 12, e0175015 (2017).
Zhiping, X. U. & Cheng, X. E. Zebrafish tracking using convolutional neural networks. Sci. Rep. 7, 42815 (2017).
Stamou, G., Krinidis, M., Loutas, E., Nikolaidis, N. & Pitas, I. 2D and 3D motion tracking in digital video. in Handbook of image and video processing 491–517 (Academic press, 2005).
Bar-Shalom, Y., Fortmann, T. E. & Cable, P. G. Tracking and data association. J. Acoust. Soc. Am. 87, 918–919 (1990).
Welch, G. & Bishop, G. An introduction to the Kalman filter. (2006).
Bovet, D. P., Crescenzi, P. & Bovet, D. Introduction to the theory of complexity. (Prentice Hall London, 1994).
Cauchy, A.-L. Considérations nouvelles sur les intégrales définies qui s’ étendent à tous les points d’une courbe fermée, et sur celles qui sont prises entre des limites imaginaires. CR Acad. Sci. Paris 23, 689 (1846).
Hu, M.-K. Visual pattern recognition by moment invariants. IRE Trans. Inf. theory 8, 179–187 (1962).
Kuhn, H. W. The Hungarian method for the assignment problem. Nav. Res. Logist. Q. 2, 83–97 (1955).
Giancardo, L. et al. Automatic visual tracking and social behaviour analysis with multiple mice. PLoS One 8, e74557 (2013).
Acknowledgements
The Swedish Research Council (2013-5379) and Kempestiftelserna supported this work to MA.
Author information
Authors and Affiliations
Contributions
A.R. wrote the main manuscript text and implemented ToxId. A.R., H.Z., J.K., T.B. and M.A. collaborated in planning experiments and analysing the data. A.R., H.Z., J.K., T.B. and M.A. edited the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Rodriguez, A., Zhang, H., Klaminder, J. et al. ToxId: an efficient algorithm to solve occlusions when tracking multiple animals. Sci Rep 7, 14774 (2017). https://doi.org/10.1038/s41598-017-15104-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-15104-2
This article is cited by
-
The connexin hemichannel inhibitor D4 produces rapid antidepressant-like effects in mice
Journal of Neuroinflammation (2023)
-
Fabricating a dielectrophoretic microfluidic device using 3D-printed moulds and silver conductive paint
Scientific Reports (2023)
-
Influence of the direct and indirect effects of habitat fragmentation, via microclimate change, on animal locomotion
Landscape Ecology (2023)
-
Beyond the three-chamber test: toward a multimodal and objective assessment of social behavior in rodents
Molecular Autism (2022)
-
Walking activity of a predatory ladybird, Cheilomenes propinqua: impacts of photoperiod, temperature, and starvation
BioControl (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.