Abstract
Long interspersed nuclear elements-1 (L1s) are a large family of retrotransposons. Retrotransposons are repetitive sequences that are capable of autonomous mobility via a copy-and-paste mechanism. In most copy events, only the L1 sequence is inserted, however, they can also mobilize the flanking non-repetitive region by a process known as 3′ transduction. L1 insertions can contribute to genome plasticity and cause potentially tumorigenic genomic instability. However, detecting the activity of a particular source L1 and identifying new insertions stemming from it is a challenging task with current methodological approaches. We developed a long-distance inverse PCR (LDI-PCR) based approach to monitor the mobility of active L1 elements based on their 3′ transduction activity. LDI-PCR requires no prior knowledge of the insertion target region. By applying LDI-PCR in conjunction with Nanopore sequencing (Oxford Nanopore Technologies) on one L1 reported to be particularly active in human cancer genomes, we detected 14 out of 15 3′ transductions previously identified by whole genome sequencing in two different colorectal tumour samples. In addition we discovered 25 novel highly subclonal insertions. Furthermore, the long sequencing reads produced by LDI-PCR/Nanopore sequencing enabled the identification of both the 5′ and 3′ junctions and revealed detailed insertion sequence information.
Similar content being viewed by others
Introduction
Long interspersed nuclear elements (LINE)-1, also known as L1 elements, are active mobile repeat elements in the human genome. Germline L1 polymorphic insertions are suggestive of L1’s contribution to genomic diversity1. Nevertheless, somatic L1 insertions can drive tumorigenesis and high L1 expression is emerging as a common trait of several cancers2. For an L1 to be potentially active, it needs to be a full-length element with a 5′ promoter and two intact open reading frames, ORF1 and ORF2, terminating with a 3′ polyadenylation signal.
Out of more than 500,000 copies of L1 sequences in the human reference genome only 90 to 100 are potentially active3,4. Upon activation, the L1 is transcribed into an RNA intermediate, which is then translated into two proteins, ORF1p and ORF2p. The L1 mRNA, ORF1p and ORF2p form a ribonucleoprotein complex that nicks the DNA at the target location via the endonuclease function of ORF2p. The polyadenylated 3′ end of the L1 mRNA anneals to the T-rich region of the target site, which then acts as a primer for reverse transcription5. Thus, this process, referred to as target-primed reverse transcription (TPRT), integrates L1 sequence into the target region with a signature polyA sequence marking initiation of reverse transcription. TPRT sometimes is accompanied by a process called twin-priming that leads to inversion of the L1 inserts. This results when one overhang produced on the target region by ORF2p-mediated staggered double-strand cleavage serves as a T-rich or polyT primer for TPRT, and the other anneals to the internal L1 mRNA sequence serving as an internal primer6.
Not all potentially active L1 elements contribute equally to retrotransposition events however, and only few “hot” L1 elements are responsible for most retrotranspositions3. These hot L1 elements are of considerable interest as they show frequent somatic insertions in various types of cancers (Fig. 1a)7,8,9,10,11. Particularly, an L1 located in the first intron of TTC28 (chromosomal position 22q12.1) (L1Base ID: 13512; dbRIP ID: 200014413), also shown to be an active element in an in vitro retrotransposition assay3, is highly active in colorectal cancer9,11. It was possible to identify this particular L1 (hereafter referred to as TTC28 L1) as the source of the insertions by using short paired-end read sequencing based on the mobilization of its non-repetitive (unique) 3′ flanking sequence, via a mechanism known as 3′ transduction14. 3′ transduction occurs when the canonical 3′ polyadenylation signal of the source L1 is weak, causing the transcription machinery to skip it and to continue transcribing the non-repetitive region downstream in the 3′ flanking region until it reaches a stronger polyadenylation/termination signal. Consequently, some of this unique sequence is included in the RNA intermediate and subsequently incorporated into the new chromosomal location, thereby serving as a unique sequence tag that reveals the L1’s origin.
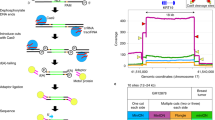
LDI-PCR based method to detect the activity of a hot L1. (a) Schematic showing mobility of hot L1 from the TTC28 locus upon activation. (b) LDI-PCR to detect 3′ transduction arising from the L1 at TTC28: Schematic representation of a hypothetical TTC28 specific L1 retrotransposition including transduction of 3′ flanking region or the “unique tag” (=region between the canonical polyadenylation signal and an alternative polyadenylation signal downstream), into an unknown target locus. NsiI produces restriction fragments of two different sizes that are self-ligated to form a circular template. Upon LDI-PCR, an inverse primer pair directed at the unique tag produces a native product and an insertion-specific target product. In addition to NsiI, two further restriction enzymes (PstI and SacI) and primer pairs (not depicted here) were used; see Materials and Methods for details.
Although whole genome sequencing (WGS) can identify 3′ transductions, detecting these L1-mediated insertions is still a great challenge, principally due to the repetitive nature of L1 sequences and our limited capacity to sequence long fragments of DNA. Several next-generation sequencing strategies targeting young L1s or 3′ transduced regions have been developed (ATLAS15, L1-seq.16, RC-seq.17, transduction-specific ATLAS18, TIP-seq.19) and used to identify somatic L1 insertions, but they are quite extensive if the aim is to simply assess the activity of a few L1 loci. Furthermore, all current methods targeting L1 insertions are limited in their capacity to simultaneously resolve full insertion sequence. Thus we have developed a direct molecular approach to detect 3′ transductions from specific L1s and hence monitor their activity; this method requires no prior knowledge of the insertion target regions. We apply long-distance inverse (LDI)-PCR20 to a particular source L1 (TTC28 specific L1 in this study) by targeting inverse primers to its frequently transduced 3′ flanking sequence (here referred to as the “unique tag”) (Fig. 1b). Note, however, that L1 insertions exhibiting 3′ transduction represent a quarter of the total L1 insertions emanating from any particular source11.
We utilized previously published WGS data21 to select two colorectal tumour samples with high number of TTC28 L1 3′ transductions (hereafter referred simply as insertions) for our proof-of-concept analysis and comparison. By selective amplification of the transduced region using LDI-PCR (Fig. 1b) followed by Nanopore sequencing, we were able to detect 14 out of 15 previously detected insertions, and additionally identified several highly subclonal insertions not detected by WGS. Long reads produced by Nanopore sequencing allowed detailed sequence analysis of the LDI-PCR products, including full inserted sequence and identification of hallmarks of retrotransposition, such as target-site duplications and deletions, polyA sequence and genomic aberrations such as inversions and deletions22.
Results
Detection of somatically acquired TTC28 insertions using LDI-PCR/Nanopore sequencing
We selected the L1 located in the first intron of TTC28 for LDI-PCR analysis, as it had been previously reported to be highly active in colorectal cancer9. We performed LDI-PCR (Fig. 1b) on DNA obtained from two tumour samples, selected from a previously reported WGS data set21 and on DNA from the corresponding normal samples, using three restriction enzymes (PstI, NsiI (Fig. 1b) and SacI) and three different primer pairs (Supplementary Table S1). The LDI-PCR product corresponding to the source TTC28 L1 (the “native PCR product”) was observed in almost all the samples digested with PstI and SacI (Fig. 2) but only sporadically observed in NsiI digested samples (e.g. as seen in LDI-PCR using primer pair 2, Fig. 2). In addition to the native PCR product, tumour samples exhibited additional PCR products which indicated mobilization of the TTC28-specific unique tag via L1 3′ transduction to different target locations. In order to identify the target location of each insertion, we sequenced the LDI-PCR products from the tumour samples using a single-molecule sequencing technique, Nanopore sequencing.
Agarose gel image of the LDI-PCR products. LDI-PCR using three different inverse primer pairs and restriction enzymes on two colorectal tumours (c985T and c368T) and their matching normal (c985N and c368N) DNA samples. The “native” PCR product of expected size and several additional tumour-specific products representing different putative 3′ transduction targets were detected. (Sizes corresponding to native LDI-PCR products: PstI~6.3 kb, NsiI~10.2 kb, SacI~5.6 kb.) Digested/self-ligated blood genomic DNA sample of an unrelated individual was run in the lane labelled “b” and PCR without any template in “−”.
Nanopore sequencing generated 644,669 reads and no bias in read frequency towards particular read lengths was apparent (Supplementary Fig. S1). We developed and applied the LDI-PCR software (LDI-PCR.py) to identify TTC28 specific 3′ transductions in both tumour samples (c985T and c368T). After filtering the LDI-PCR.py calls, we were able to identify 14 out of 15 previously detected insertions. Additionally, we detected 25 novel insertions not identified by WGS, despite visual inspection of the paired-end read data (Supplementary Fig. S2) (Table 1). Notably, these 25 novel candidate insertions were supported by fewer reads than the 14 WGS-detected insertions (p = 2.43 × 10−6 by Wilcoxon rank-sum test) (Table 1 and Fig. 3). The median number of supporting reads was 98 for novel candidate insertions and 11,428 for WGS-detected insertions, suggesting that the novel insertions are subclonal events and therefore difficult to detect by 40x WGS.
Read counts for insertions called by LDI-PCR.py. On the left, boxplots of read counts for insertions that were either detected (n = 14) or undetected (n = 25) by WGS. On the right, boxplots of read counts for insertions undetected by WGS that were either validated (n = 7) or unvalidated (n = 18). For better visualization, data are presented on a base-10 log scale.
In order to validate the novel candidate insertions, we first performed conventional PCR and Nanopore sequencing and were able to validate two insertions (Y:15633117 and 14:79638932). Subsequently we performed allele-specific PCR and Sanger sequencing on another ten insertions, of which we successfully sequenced the insertion-to-target junctions for six of them (Table 1, Supplementary Fig. S2). Again, non-validated insertions were supported by fewer reads as compared to validated insertions (p = 7.53 × 10–4 by Wilcoxon rank-sum test) (Fig. 3). Furthermore, the insertion located at Y:15633117 was confirmed with both methods.
Analysis of consensus sequence generated by LDI-PCR/Nanopore sequencing to elucidate insertion characteristics
Consensus sequences, generated from the LDI-PCR.py calls after filtering, provided us with complete inserted sequences for 35 out of 39 L1 insertions which were analysed to decipher the insertion mechanism and retrotransposition hallmarks (Table 1). 8/35 consensus sequences contained short alignment gaps, ranging from 3–10 bp, due to mismatches affecting the alignment. Most of these alignment gaps (7) arose from insertions supported by less reads indicating, as expected, that higher number of reads improves consensus accuracy. 29 out of 35 insertions involved a target-site duplication while 6 insertions involved a target-site deletion. We also detected 2 target-site duplications and 1 target-site deletion in insertions with incomplete sequence (Table 1). The size of L1 insertions ranged from 142 bp to 1124 bp with an average insertion size of 493 bp. All detected L1 insertions were heavily truncated at their 5′ end, the majority (~77%) to the extent that they were composed of the 3′ transduced region only, without any L1 sequence (also known as “orphan transductions”). We were able to locate the terminal sequence in all 35 insertions with complete inserted sequence. Variation in the 3′ most genomic coordinate (Table 1) of the L1 3′ transduction suggested the use of more than one polyadenylation signal. By following the criteria explained in the methods section to determine which polyadenylation signal was preferred, we observed that more than 90% of the TTC28 L1 3′ transductions used either the 6th or the 9th polyadenylation signal instead of the canonical polyadenylation signal (Fig. 4, Supplementary Table S2). Preference for the 9th polyadenylation signal is in agreement with polyadq prediction23 (Supplementary Table S2), a web-based polyadenylation signal prediction tool. However, the 6th polyadenylation signal was defined as a false signal by polyadq, even though it had the highest score among the ATTAAA polyadenylation signals (Supplementary Table S2).
Polyadenylation signal of choice for TTC28 L1 mediated 3′ transduction. 8 polyadenylation signals following the L1 canonical polyadenylation signal (no. 2–9) were present in the unique tag assessed for TTC28 L1 3′ transduction. Most of the 3′ transductions identified in this study terminated utilizing the 6th and 9th polyadenylation signal instead of the L1’s canonical polyadenylation signal (no. 1). The height of the orange bars indicates the frequency of L1 3′ transductions terminating at each polyadenylation signal; number of termination events is given above the bars.
Strand inversion of the inserted sequence due to twin-priming was observed in 16 out of 35 insertions for which complete inserted sequence was available. One more twin-priming event was observed in three insertions with incomplete sequence (Table 1). Furthermore, the point of inversion was identifiable in all 16 cases of twin-priming. We were able to resolve the 5′ junction for 29 out of 35 L1 insertions, as remaining 6 contained short alignment gaps as mentioned earlier. In addition we were also able to resolve 5′ junction of 4 insertions that had incomplete inserted sequence. Out of these total 33 insertions 28 of them showed microhomology of 1–13 bp at the 5′ junction. Microhomology of 1–5 bp was also observed in 10 out of 16 twin-priming inversion point.
We then used information on the inserted sequences to better understand the integration process of the L1-derived sequence. To extrapolate the stepwise mechanism of L1 insertion, we selected two insertions displaying two different well-characterized modes of insertion: (a) TPRT, exemplified by the L1 insertion at GRID2 locus on chromosome 4 and (b) TPRT with twin-priming, exemplified by the L1 insertion at CNTNAP2 locus in chromosome 7 (Supplementary Fig. S3, Fig. 5). L1-transduced sequence was inserted on the “+” strand of GRID2 target locus and on the “−” strand of CNTNAP2 locus (Supplementary Fig. S3). Target site duplication (TSD) observed at both loci indicated L1 endonuclease mediated staggered double-stranded cleavage in the target region (Fig. 5ai,bi). This staggered double-strand cleavage at both target loci generated a T-rich overhang. We infer that these T-rich overhangs produced on the “−” and “+” strand of GRID2 and CNTNAP2 target loci, respectively, annealed to the polyA tail at the end of the L1 mRNA (Fig. 5aii,bii) and were used as a polyT primer for reverse transcription Fig. 5ai,bi; stepwise mechanism illustrated in Fig. 5aiii,biii). In addition to polyT priming, reverse transcription at the CNTNAP2 target locus most likely also used as an internal primer the other overhang generated, causing an inversion of the inserted sequence (Fig. 5biii). Upon close examination we found that the region of inversion (22:29,065,715–29,065,721) did in fact show nucleotide complementarity with the 5′ overhang generated by ORF2p on the reverse strand (7:146,783,223) (Fig. 5bii,biii). This twin-priming (first by a polyT primer and then by an internal primer) at the CNTNAP2 locus led to reverse-transcription at two different locations causing strand inversion (Fig. 5biii and biv). We also observed a deletion of 3 base pairs (22:29,065,722–29,065,724) at the inversion site and microhomology of 3 base pairs between the reverse transcribed sequence produced by the polyT primer and the internal primer at the point of inversion (Fig. 5b). Microhomology was also observed at the 5′ junction of L1 insertion at the GRID2 target locus. Thus, LDI-PCR/Nanopore sequencing provided complete information for most of the somatic L1 insertions, enabling us to analyse the insertion process in great detail.
Extrapolated mechanism of L1 insertion at (a) GRID2 locus and (b) CNTNAP2 locus In both (a) and (b) (i) shows locations of staggered double-stranded cleavage in the target region with PolyT primer in the 3′ overhang and an internal primer (when used) in the 5′ overhang (b). (ii) Extrapolated L1 mRNA with the TTC28 3′ unique tag. Regions complementary to the polyT primer and internal primer at target site is highlighted by the same colour scheme. (iii) Schematic representation of the TPRT (a) and twin-priming (b) mechanism of TTC28 specific 3′ transduction in the GRID2 (a) and CNTNAP2 (b) locus. Figures are not drawn to scale.
Comparison of the local assembly of WGS data and the consensus sequences generated by LDI-PCR/Nanopore sequencing
In order to interpret the advantages and disadvantages of long read sequencing, we compared the local assembly of paired-end read data to the LDI-PCR/Nanopore consensus sequences of those insertions in tumour sample c985T that were detected by both methods. After local assembly, we were able to reconstruct four out of seven insertions into one contiguous fragment (contig) (Supplementary Table S3) with defined parameters (Supplementary Table S4). However, the remaining three insertions were represented in two contigs and therefore the sequence in between was lost. The length of missed sequences, ranging from 88 bp to 614 bp, was estimated based on the genomic coordinates of both insertion junctions. However, in cases where the twin priming point of inversion was located within the missed sequence, estimation of the insertion size was not possible, as shown for chr7:152661937 (Supplementary Table S3). All insertions with missing sequence information were longer than 400 bp and were the longest insertions based on the consensus sequence analysis of Nanopore data. The deviation of the estimated length of the insertions ranged from 0–7 bp and 10/14 target coordinates were consistently predicted by both analysis (Supplementary Table S3). The remaining four target coordinates were located right after a tract of Ns where the consensus polyA/T junction was predicted, henceforth, target sequence after the polyA/T was probably missed by the local assembly. In conclusion, local assembly of paired-end read data was of limited value in reconstructing full insertions but also interpreting polyA/T tails in our study, thus hampering the elucidation of insertion features.
Discussion
Due to the repetitive nature of L1 elements and their abundance in the human genome, it is possible to determine the lineage or source of the L1 insertions either (a) when the insertion contains signature single nucleotide polymorphisms associated with the source L110 or (b) when it involves 3′ transduction of the unique flanking sequence9,11. Thus, in order to determine the activity of a particular source L1, one needs to scan the genome of interest for either of the aforementioned signs. Assessing the activity of a particular source L1, in particular when active in a tumour type, can be of great interest, as these L1 insertions can cause tumorigenic events or serve as a clinical biomarker24. However, the specific detection of L1 3′ transductions and/or sequencing the whole length of L1 insertions remains a great challenge. In this study we used a LDI-PCR based assay to study the activity of a source L1 at the TTC28 locus, previously shown to be highly active in colorectal cancer, and tested it on two colorectal cancer samples with already published whole genome sequencing data21.
By applying LDI-PCR in conjunction with Nanopore sequencing to as low as 300 ng of tumour DNA per sample, we were able to detect 14 out of 15 previously identified TTC28 L1 mediated 3′ transductions, and also discovered 25 3′ transductions not detected by WGS (Table 1). The read count difference between the two groups (WGS-detected versus not detected) indicated that these insertions could be subclonal events and thus not detectable with 40x sequencing. Furthermore, high coverage provided us with enough data to reconstruct accurate consensus sequences, which permitted analyses of full inserted sequence in 90% of the insertions. About 45% of the 3′ transductions analysed showed strand inversion due to twin-priming. We speculate that this high incidence of twin-priming in TTC28 L1 3′ transduction is due to (a) the nature of nucleotide sequence downstream of the L1 3′ end, possibly due to many small stretches of Ts present which could complement with a stretch of As generated by the ORF2p endonuclease action on the target region which in turn can be used as the second or “internal” primer leading to twin-priming, or (b) detection of more L1 insertions with these inversion properties than by conventional methods due to the sequencing of the whole inserted sequence. We were also able to sequence the entire source TTC28 L1 (Supplementary Fig. S4). In addition, we were able to identify that TTC28 3′ transductions terminated preferentially on two polyadenylation signals (Fig. 4), one predicted to be a true signal with the highest score (9th), and another defined as a false signal (6th) (Supplementary Table S2) by polyadq.
One insertion predicted by WGS was not identified in this study, however. LDI-PCR and Nanopore sequencing could have been hindered due to the formation of a secondary structure due to homology between the insertion polyT and a nearby (567 bp upstream) polyA present in the reference genome.
LDI-PCR/Nanopore success principally relies on (i) a careful selection of the restriction enzymes so as to produce PCR-amplifiable target templates and (ii) the design of multiple primer pairs covering several downstream polyadenylation signals predicted by tools such as polyadq. In our initial pilot LDI-PCR/Nanopore sequencing experiment using one restriction enzyme (SacI) and two primer pairs on one sample (c985T), we were able to detect only 4 insertions out of 8 detected by WGS analysis. Detection sensitivity was substantially improved by updated sequencing chemistry, the use of two additional restriction enzymes and by designing more primers that covered additional polyadenylation signals in a well-dispersed manner.
LDI-PCR follows a similar targeting strategy as transduction-specific ATLAS (TS-ATLAS)18. However, LDI-PCR allows the amplification of both 5′ and 3′ junctions and sequencing the entire insertion simultaneously, which cannot be accomplished by any other L1 targeted approach or by WGS. The only region that remains unsequenced in a single read is the nucleotide bases in between the primer pairs, however this limitation can be minimized by reducing the distance between the primers and using more than one set of primers. Furthermore, the inverse PCR primers at the unique sequence enable the detection of L1 orphan transductions, which are not detected by other targeted sequencing techniques such as ATLAS, L1-seq, RC-seq or TIP-seq.15,16,17,19. Additionally, LDI-PCR/Nanopore sequencing is customizable for any full-length L1 allowing the implementation of this assay on a handful of “hot” L1 elements that contributes to a large fraction of 3′ transductions in a cancer genome11.
To conclude, we demonstrated that LDI-PCR/ Nanopore sequencing is suitable for sequencing the entire L1 insertion and for detecting highly subclonal events. Consequently, applying LDI-PCR in conjunction with Nanopore sequencing in larger sample sets and different tumour types enables a more detailed characterization of L1 insertions providing new insights into L1 biology and cancer genetics.
Material and Methods
Samples
The colorectal adenocarcinoma (CRC) samples utilized in this study were obtained from a population based series of 1042 CRCs previously described25,26. The tumours were fresh frozen and the corresponding normal tissues were obtained from blood (c985T) and from colon tissue (c368T). The study was reviewed and approved by the Ethics committee of the Hospital district of Helsinki and Uusimaa, Finland. A signed informed consent or authorization from the National Supervisory Authority for Welfare and Health was obtained for all the samples.
LDI-PCR and Nanopore sequencing
Digestion and ligation of DNA
To detect insertions arising from TTC28 L1, genomic DNA was separately digested by three restriction enzymes: SacI, PstI and NsiI. SacI and PstI make a 5′ cut at ORF1 of the L1 and a 3′ cut downstream of the unique tag producing a native restriction fragment of 5.7 kb and 6.3 kb respectively, whereas NsiI makes a 5′ cut 3.1 kb upstream of the intact L1 sequence and a 3′ cut downstream of the unique tag generating a native restriction fragment of ~10.2 kb (Fig. 1b, Supplementary Table S5). L1 retrotransposition usually involves a 5′ truncation, and the average L1 insert size including the 3′ transduced region is 1000 bp11. Therefore it is unlikely that the somatically acquired L1 insertion will contain the same SacI or PstI cut sites as the source region, hence increasing the likelihood that the target site restriction fragment is of different size compared to the native one. The infrequent cases of full-length somatic L1 insertion can be captured by the digestion library produced by NsiI. At least one out of three enzymes always generated a predicted restriction fragment of less than 8.2 kb in all the WGS predicted targets (Supplementary Table S5). Digested DNA was then self-ligated using T4 DNA ligase (Thermo) to form circular templates for LDI-PCR.
Primer Design and Optimization
Inverse primers for LDI-PCR were designed on the unique tag, that is, the genomic region between the canonical polyadenylation signal of the L1 and the next strongest polyadenylation signal on its 3′ flanking region (Fig. 1b, Supplementary Table S2). Strength of polyadenylation signals at this region was estimated using polyadq scores23 (http://rulai.cshl.edu/tools/polyadq/polyadq_form.html). Primers were designed using Primer3 (http://primer3.ut.ee) and their specificity was checked using NCBI Primer-BLAST (available at http://www.ncbi.nlm.nih.gov/tools/primer-blast/). Since there were several polyadenylation signals at the 3′ flanking region of the L1 (Fig. 1b), we designed several primer pairs between the canonical polyadenylation signal (marked 1) and following downstream polyadenylation signals. Distance between the primers was kept as short as possible (≤51bp).
LDI-PCR
1.25 ng circular templates generated by restriction enzyme digestion and T4 ligation were used in LDI-PCR as eight replicates (Supplementary Fig. S5) as previously described20 using three primer pairs (Supplementary Table S1). These PCR products were then analysed on a 1% agarose gel and purified using NucleoSpin Gel and PCR Clean-up kit (Macherey-Nagel). Replicate reactions showing reproducible patterns of PCR amplification were pooled, and sequenced using MinION (Oxford Nanopore Technologies). Replicates that did not show reproducibile patterns were discarded and the reaction was repeated again.
Oxford Nanopore MinION™ sequencing
LDI-PCR products from 18 different reactions (with three different primer pairs, three different restriction enzymes and from two tumour samples) were pooled into nine different barcodes in equal molarity. Tape Station 2200 (Agilent Technologies) was used to estimate the relative molarity based on the fragment distribution in each reaction. Libraries were constructed according to the manufacturer’s instructions using SQK-LSK108 and EXP-NBD103 sequencing and barcoding kits (Oxford Nanopore Technologies). Equal molarity was preserved throughout the protocol. The MinION Flow cell (FLO-MIN106) was run for 6 hours using MinION Mk1B. The raw signal from MinION was basecalled with ONT Albacore Sequencing Pipeline Software (version 1.0.2). The reads passing base calling were aligned to GRCh37 genome reference augmented with viral and 1000 genomes decoy sequences. The alignment was performed with bwa mem v0.7.12 using option -x ont2d27.
LDI- PCR software
We separated the reads produced by different samples, PCR primers and restriction enzymes by using the sequencing barcodes and comparing the read mapping with restriction enzyme cut sites in the reference genome. To systematically detect the insertions, we developed the LDI-PCR.py software which identifies reads that display hallmark features of LDI-PCR products (Fig. 1b). Hallmarks of a LDI-PCR product in this experiment are: that it contains at least one alignment to TTC28 L1 (22:29060420–29067335) and at least one alignment to a single target locus in the genome. The read can have multiple supplementary alignments to the target locus but only if located in close proximity of each other (maximum distance between alignments 100 kb). All considered alignments had to have mapping quality of at least 20. The insertion breakpoint was defined by the location where the alignment switched from TTC28 L1 to the target locus and vice versa for each hallmark read. All insertion breakpoints were clustered together allowing a maximum gap of 3 kb from all reads defining the different insertion locations. The most frequent genomic coordinate was called as the insertion breakpoint defining each LDI-PCR insertion call. The software is available at https://github.com/kpalin/LDI-PCR-call and https://github.com/kpalin/ampcorrect.
Furthermore, LDI-PCR insertion calls had to be supported by at least 5 reads and, to filter away random ligation products generated by LDI-PCR, the insertion breakpoint had to be located at least 35 bp from the closest corresponding restriction enzyme cut site. Moreover, due to barcoding ligation crosstalk produced by EXP-NBD103 barcoding kit (Oxford Nanopore Technologies), several insertion calls were present in both samples. In order to circumvent this issue, in cases where the insertion call was present in two samples, only the calls coming from the sample that contained at least 95% of the reads were included in further analyses. Insertions in mitochondria and unplaced sequence were filtered away. All insertions that fulfilled the abovementioned criteria in at least one reaction were defined as candidate insertions and selected further for consensus sequence analysis of the insertion characteristics. The reads from the candidate insertions were processed with ampCorrect, a Nanopore read correction method similar to nanocorrect28, to obtain accurate consensus sequences for the amplicons. Briefly, ampCorrect uses sumaclust29 (http://metabarcoding.org/sumatra) to cluster the reads, requiring 60% sequence similarity and poaV2 -do global30 to align multiple reads. The consensus sequence is treated as corrected sequence of the analysed amplicon. The processed sequences were aligned to the human reference genome using bwa mem with default parameters. The analysis of the insertion characteristics was performed on a consensus sequence which was constructed from 20 random candidate insertion call reads using UCSC BLAT (https://genome.ucsc.edu/cgi-bin/hgBlat) (Consensus sequence of all insertions analysed are provided in FASTA format in Supplementary dataset 1).
Determining the polyadenylation signal used for each L1 3′ transduction
Since the transcription termination and polyadenylation occurs 10–30 bp downstream of the selected polyadenylation signal31, we analysed how many 3′ coordinates of the L1 insertions were located within a 10–30 bp window downstream of each of the polyadenylation signal (stop signal window) located downstream of the L1 sequence (Supplementary Table S2). 9 out of 35 L1 insertion terminal sequence did not fall within any of the 8 defined windows, and were assigned to the closest available window (maximum distance was 9 bp) (Table 1 and Supplementary Table S2).
Statistical analysis
To test for differences in read counts, we used Wilcoxon rank-sum test with continuity correction in R version 3.3.2. Read counts refer to numbers of reads supporting each candidate insertion called by LDI-PCR.py. In cases where the same candidate insertion was detected in different reactions (three different restriction enzymes and three different primer pairs) the reaction with higher read count was used.
Whole genome sequencing analysis
We utilized the WGS dataset described in Katainen et al.21 to select for LDI-PCR and Nanopore sequencing those colorectal cancer samples with a high number of somatically acquired insertions originating from TTC28. Structural variant breakpoints located at the 3′ end of the TTC28 L1 were extracted to calculate the number of transductions. The 3′ end of the L1 was defined as GRCh37 coordinates 22:29065455–29066124. To examine whether the novel candidate insertions were detected by WGS or not, we performed a thorough visual inspection of the paired-end read data using BasePlayer32.
In order to compare LDI-PCR Nanopore sequencing results to WGS data we performed local assembly of the WGS data. We selected those chimeric reads and discordant read pairs that aligned within 1kb upstream and downstream the predicted insertion breakpoint, with the exception of one insertion (chr15:97602708) where, due to a long target site deletion, a 3kb window was used. The local assembly of the reads was performed using Velvet 1.2.1033. All hash lengths within default parameters (11,13,15,17,19,21,23,25,27,29,31) were tested and the hash length that produced the longest and most contiguous contig was selected for each insertion33 (Supplementary Table S4). We aligned the assembled contigs with UCSC Blat (https://genome.ucsc.edu/cgi-bin/hgBlat) to the GRCh37 genome reference.
Validation of highly subclonal L1 insertions by conventional PCR
Two approaches were used to validate novel candidate insertions detected by LDI-PCR but not by WGS: (i) First, primer pairs were designed on the target genomic region across the insertion breakpoint and sequenced by Nanopore. The library was prepared as described in the section “Oxford Nanopore MinION™ sequencing”. This approach included all candidate subclonal insertions, however only 3/25 novel candidate insertions were validated (ii) Second, primers were designed based on the consensus sequence, with one primer at the target site and the other primer at the inserted sequence, followed by Sanger sequencing of the resulting PCR product; this was performed for 10 selected novel candidate insertions. Primer pairs were designed with primer3Plus (http://www.bioinformatics.nl/cgi-bin/primer3plus/primer3plus.cgi). Primer sequences are in Supplementary Table S6. Sanger sequencing was performed by the Biomedicum Sequencing Unit, Helsinki, on ABI Prism 3130xl Genetic Analyzer (Applied Biosystems) using BigDye Terminator v3.1 cycle sequencing kit (Applied Biosystems). Sequences were manually analysed using FinchTV v.1.4.0 (http://www.freewarefiles.com/FinchTV_program_17782.html).
Data availability
The datasets generated during and analysed during the current study are available from the corresponding author on request.
References
Beck, C. R. et al. LINE-1 retrotransposition activity in human genomes. Cell 141, 1159–1170, https://doi.org/10.1016/j.cell.2010.05.021 (2010).
Burns, K. H. Transposable elements in cancer. Nat Rev Cancer 17, 415–424, https://doi.org/10.1038/nrc.2017.35 (2017).
Brouha, B. et al. Hot L1s account for the bulk of retrotransposition in the human population. Proc Natl Acad Sci USA 100, 5280–5285, https://doi.org/10.1073/pnas.0831042100 (2003).
Lander, E. S. et al. Initial sequencing and analysis of the human genome. Nature 409, 860–921, https://doi.org/10.1038/35057062 (2001).
Luan, D. D., Korman, M. H., Jakubczak, J. L. & Eickbush, T. H. Reverse transcription of R2Bm RNA is primed by a nick at the chromosomal target site: a mechanism for non-LTR retrotransposition. Cell 72, 595–605 (1993).
Ostertag, E. M. & Kazazian, H. H. Jr. Twin priming: a proposed mechanism for the creation of inversions in L1 retrotransposition. Genome Res 11, 2059–2065, https://doi.org/10.1101/gr.205701 (2001).
Lee, E. et al. Landscape of somatic retrotransposition in human cancers. Science 337, 967–971, https://doi.org/10.1126/science.1222077 (2012).
Miki, Y. et al. Disruption of the APC gene by a retrotransposal insertion of L1 sequence in a colon cancer. Cancer Res 52, 643–645 (1992).
Pitkanen, E. et al. Frequent L1 retrotranspositions originating from TTC28 in colorectal cancer. Oncotarget 5, 853–859, https://doi.org/10.18632/oncotarget.1781 (2014).
Scott, E. C. et al. A hot L1 retrotransposon evades somatic repression and initiates human colorectal cancer. Genome Res 26, 745–755, https://doi.org/10.1101/gr.201814.115 (2016).
Tubio, J. M. et al. Mobile DNA in cancer. Extensive transduction of nonrepetitive DNA mediated by L1 retrotransposition in cancer genomes. Science 345, 1251343, https://doi.org/10.1126/science.1251343 (2014).
Penzkofer, T. et al. L1Base 2: more retrotransposition-active LINE-1s, more mammalian genomes. Nucleic Acids Res 45, D68–D73, https://doi.org/10.1093/nar/gkw925 (2017).
Wang, J. et al. dbRIP: a highly integrated database of retrotransposon insertion polymorphisms in humans. Hum Mutat 27, 323–329, https://doi.org/10.1002/humu.20307 (2006).
Moran, J. V., DeBerardinis, R. J. & Kazazian, H. H. Jr. Exon shuffling by L1 retrotransposition. Science 283, 1530–1534 (1999).
Badge, R. M., Alisch, R. S. & Moran, J. V. ATLAS: a system to selectively identify human-specific L1 insertions. Am J Hum Genet 72, 823–838, https://doi.org/10.1086/373939 (2003).
Ewing, A. D. & Kazazian, H. H. Jr. High-throughput sequencing reveals extensive variation in human-specific L1 content in individual human genomes. Genome Res 20, 1262–1270, https://doi.org/10.1101/gr.106419.110 (2010).
Baillie, J. K. et al. Somatic retrotransposition alters the genetic landscape of the human brain. Nature 479, 534–537, https://doi.org/10.1038/nature10531 (2011).
Macfarlane, C. M. et al. Transduction-specific ATLAS reveals a cohort of highly active L1 retrotransposons in human populations. Hum Mutat 34, 974–985, https://doi.org/10.1002/humu.22327 (2013).
Rodic, N. et al. Retrotransposon insertions in the clonal evolution of pancreatic ductal adenocarcinoma. Nat Med 21, 1060–1064, https://doi.org/10.1038/nm.3919 (2015).
Pradhan, B. et al. Detection and screening of chromosomal rearrangements in uterine leiomyomas by long-distance inverse PCR. Genes Chromosomes Cancer 55, 215–226, https://doi.org/10.1002/gcc.22317 (2016).
Katainen, R. et al. CTCF/cohesin-binding sites are frequently mutated in cancer. Nat Genet 47, 818–821, https://doi.org/10.1038/ng.3335 (2015).
Gilbert, N., Lutz-Prigge, S. & Moran, J. V. Genomic deletions created upon LINE-1 retrotransposition. Cell 110, 315–325 (2002).
Tabaska, J. E. & Zhang, M. Q. Detection of polyadenylation signals in human DNA sequences. Gene 231, 77–86 (1999).
Ardeljan, D., Taylor, M. S., Ting, D. T. & Burns, K. H. The Human Long Interspersed Element-1 Retrotransposon: An Emerging Biomarker of Neoplasia. Clin Chem 63, 816–822, https://doi.org/10.1373/clinchem.2016.257444 (2017).
Aaltonen, L. A. et al. Incidence of hereditary nonpolyposis colorectal cancer and the feasibility of molecular screening for the disease. N Engl J Med 338, 1481–1487, https://doi.org/10.1056/NEJM199805213382101 (1998).
Salovaara, R. et al. Population-based molecular detection of hereditary nonpolyposis colorectal cancer. J Clin Oncol 18, 2193–2200, https://doi.org/10.1200/JCO.2000.18.11.2193 (2000).
Heng, L. Aligning sequence reads, clone sequences and assembly contigs with BWA-MEM. arXiv (2013).
Loman, N. J., Quick, J. & Simpson, J. T. A complete bacterial genome assembled de novo using only nanopore sequencing data. Nat Methods 12, 733–735, https://doi.org/10.1038/nmeth.3444 (2015).
Mercier, C. B. F., Bonin, A. & Coissac, E. SUMATRA and SUMACLUST: fast and exact comparison and clustering of sequences. Programs Abstr SeqBio 27 (2013).
Grasso, C. & Lee, C. Combining partial order alignment and progressive multiple sequence alignment increases alignment speed and scalability to very large alignment problems. Bioinformatics 20, 1546–1556, https://doi.org/10.1093/bioinformatics/bth126 (2004).
Colgan, D. F. & Manley, J. L. Mechanism and regulation of mRNA polyadenylation. Genes Dev 11, 2755–2766 (1997).
Katainen, R. et al. BasePlayer: Versatile Analysis Software For Large-Scale Genomic Variant Discovery. bioRxiv, https://doi.org/10.1101/126482 (2017).
Zerbino, D. R. & Birney, E. Velvet: algorithms for de novo short read assembly using de Bruijn graphs. Genome Res 18, 821–829, https://doi.org/10.1101/gr.074492.107 (2008).
Acknowledgements
We are grateful to the anonymous reviewers whose comments on the first version of this manuscript inspired several highly informative experiments. We would like to thank Alison Ollikainen, Iina Vuoristo and Heikki Metsola for excellent technical assistance. In addition, we would like to thank Alison Ollikainen for language support. This work was supported by grants from the Academy of Finland [Finnish Center of Excellence Program 2012–2017 250345, personal grant to O.K. 274474, grants 256996, 263870, 292789 and 306026 to L.K.]; the Finnish Cancer Society [personal grants to K.P. and to L.K.]; the European Research Council [ERC; 268648]; the Sigrid Juselius Foundation; SYSCOL [a European Union Framework Programme 7 Collaborative project, 258236]; the Nordic Information for Action eScience Center (NIASC), the Nordic Center of Excellence financed by NordForsk [project 62721, personal grant to K.P.]; Ida Montinin Säätio foundation [personal grant to TC]; Biocentrum Helsinki and Jane and Aatos Erkko Foundation. L.K. is the recipient of a Marie Curie Career Integration Grant (PCIG11-GA-2012–321983) of the European Union. B.P. is the recipient of University of Helsinki Science Foundation funded PhD studentship and Biomedicum Helsinki Foundation Grant. The authors wish to acknowledge CSC-IT Center for Science, Finland, for computational resources.
Author information
Authors and Affiliations
Contributions
B.P. and T.C. conceived the experiments, analysed the results and wrote the manuscript. R.K. contributed to the analysis of the results. P.S. performed the local assembly of WGS data and T.T. the statistical analysis. O.K., E.P. supervised and contributed to writing the manuscript. L.K. conceived and supervised the experiments, contributed to the analysis of the results and helped to write the manuscript. K.P. supervised and contributed to the experimental procedure, contributed to the analysis of the results and writing the manuscript. All authors reviewed the manuscript.
Corresponding authors
Ethics declarations
Competing Interests
LAA has received a lecture fee from Roche Oy. Other authors have no competing financial interests to declare.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Pradhan, B., Cajuso, T., Katainen, R. et al. Detection of subclonal L1 transductions in colorectal cancer by long-distance inverse-PCR and Nanopore sequencing. Sci Rep 7, 14521 (2017). https://doi.org/10.1038/s41598-017-15076-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-15076-3
Keywords
This article is cited by
-
PCIP-seq: simultaneous sequencing of integrated viral genomes and their insertion sites with long reads
Genome Biology (2021)
-
Nanopore sequencing technology, bioinformatics and applications
Nature Biotechnology (2021)
-
A potential new mechanism for pregnancy loss: considering the role of LINE-1 retrotransposons in early spontaneous miscarriage
Reproductive Biology and Endocrinology (2020)
-
Transposon insertion profiling by sequencing (TIPseq) for mapping LINE-1 insertions in the human genome
Mobile DNA (2019)
-
Diseases of the nERVous system: retrotransposon activity in neurodegenerative disease
Mobile DNA (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.