Abstract
Two-dimensional molybdenum disulfide (2D MoS2) presents extraordinary optical, electrical, and chemical properties which are highly tunable by engineering the orientation of constituent 2D layers. 2D MoS2 films with vertically-aligned layers exhibit numerous 2D edge sites which are predicted to offer superior chemical reactivity owing to their enriched dangling bonds. This enhanced chemical reactivity coupled with their tunable band gap energy can render the vertical 2D MoS2 unique opportunities for environmental applications that go beyond the conventional applications of horizontal 2D MoS2 in electronics/opto-electronics. Herein, we report that MoS2 films with vertically-aligned 2D layers exhibit excellent visible light responsive photocatalytic activities for efficiently degrading organic compounds in contaminated water such as harmful algal blooms. We demonstrate the visible light-driven rapid degradation of microcystin-LR, one of the most toxic compounds produced by the algal blooms, and reveal that the degradation efficiency can be significantly improved by incorporating noble metals. This study suggests a high promise of these emerging 2D materials for water treatment, significantly broadening their versatility for a wide range of energy and environmental applications.
Similar content being viewed by others
Introduction
Viable solutions for efficiently degrading emerging organic contaminants in drinking water supplies have urgently been demanded with their increasing threats to environment and human health. For instance, harmful algal blooms (HABs), a rapid increase and/or accumulation in the population of algae which can severely damage aquatic ecosystems, have recently gained substantial public attention1,2,3. The primary concern over HABs is that they release harmful cyanotoxins4,5, which can be fatal if ingested and/or inhaled6,7. However, traditional water purification methods are designed to primarily remove suspended solids and/or individual elements of carbon, nitrogen, and phosphorus in contaminated water, which are not well suited to directly degrade algal toxins. Photocatalysis, an alternative approach based on a solar energy-driven oxidation process, has drawn substantial interest for its intrinsic simplicity and efficient operation8,9,10,11. In this approach, photoactive catalytic materials in contact with contaminated water generate electron-hole (e−h+) pairs upon absorbing the solar energy. The photo-generated charge carriers dissociate dissolved oxygen (DO) in water, generating reactive oxygen species (ROS) such as hydroxyl groups and superoxide anions, which in turn disinfect pathogens12. Photocatalytic materials (usually, oxide semiconductors) accelerate the rate of the associated chemical reactions (oxidation/reduction) in the microorganisms. However, conventional photocatalytic methods have relied on the use of ultraviolet (UV) light for e−h+ generation, which is limited to harness a very small portion of the available solar energy13. This is because photocatalytic semiconductors possess large band gap (Eg) energies which match the UV spectrum corresponding to only ~4–5% of the entire solar spectrum, thus, inevitably resulting in prolonged exposure and slow reaction rates. For instance, titanium dioxide (TiO2), one of the most sought-after photocatalytic semiconductors presents Eg > 3.0 eV8,9,10,11 and only harvests UV light while neglecting the broad range of the visible light which corresponds to >40% of the entire solar spectrum14.
Molybdenum disulphide (MoS2), a recently rediscovered semiconductor classified as two-dimensional (2D) transition metal dichalcogenides (TMDs), presents a rich set of optical and structural properties uniquely suitable for photocatalytic reactions. In terms of optical properties; (1) It presents a band gap energy (~1.2–1.8 eV) matching the spectral range of the visible light. Moreover, the band gap energy is highly tunable by varying the number of 2D atomic layers15. (2) It exhibits exceptionally large sunlight absorption; for example, over an order of magnitude higher than conventional semiconductors such as silicon (Si) or gallium arsenide (GaAs) for similar thicknesses16. In terms of structural advantages; (1) It can be grown vertically standing on growth substrates (e.g. silicon dioxide (SiO2)) exposing the edges of individual 2D layers17,18,19. In this vertical orientation, atomically unsaturated 2D edge sites full of molybdenum (Mo) and sulfur (S) dangling bonds are maximally exposed on the surface. Consequently, the surface is highly reactive, offering large chemical/physical adsorption capacity for capturing molecules20,21,22. (2) It presents suitable energy band structure with respect to the redox potentials for hydrogen- or oxygen evolution reactions (HER or OER) as its conduction (valence) band edge lies above (below) the electrostatic potential of H2 (O2) evolution, respectively22,23,24,25,26. A proof-of-concept demonstration of its application to microbial inactivation in contaminated water has recently been reported12, which studies the disinfection of Escherichia (E.) coli for drinking water purification via ex-situ photocatalytic measurements.
In this work, we demonstrate rapid and efficient photocatalytic degradation of algal toxins using MoS2 films with vertically-aligned 2D layers under visible light illumination. Particularly, we study the degradation of one of the most toxic organic compounds generated from harmful algal blooms, microcystin-LR (MC-LR), which is recognized as an emerging threat to a wide range of water sources, including seawater, river, and lakes. We investigate its photocatalytic reaction kinetics by in situ characterization of ROS generation using microsensors. Moreover, we identify that the coating of thin noble metal layers on top of pristine MoS2 films significantly improves the photocatalytic efficiency, enabling the rapid and complete removal of MC-LR. The underlying mechanisms for the observed photocatalytic reactions as well as their governing parameters are also discussed. The study suggests the promise and versatility of MoS2 films with vertically-aligned 2D layers for a broad range of water purification and environmental applications.
Result and Discussion
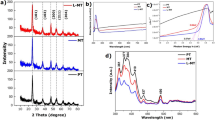
Figure 1(a) is the schematic illustration demonstrating the concept of photocatalytic degradation of emerging water contaminants using semiconducting 2D MoS2 photocatalysts. MoS2 films with vertically aligned 2D layers grown on SiO2/Si substrates are immersed in a water bath containing algal toxins (MC-LR in this case) absorbing photons from the visible light, which readily generates e−h+. These charge carriers migrate to the semiconducting surface where they react with hydroxyl ions and oxygen compounds to generate highly reactive species (i.e. O2 •−,.OH, and H2O2) that can degrade the algal toxins. Figure 1(b) is an image of a MoS2 film with vertically aligned 2D layers grown on a SiO2/Si wafer of >2 cm2 in size. The growth of the MoS2 film with vertically aligned 2D layers was achieved by the sulfurization of Mo-deposited wafers following the previously reported method17. Details for the material growth are in Materials and Methods. Figure 1(c) is a representative high-resolution transmission electron microscopy (HRTEM) image of a MoS2 film with vertically-aligned 2D layers. It is apparent that the MoS2 film predominantly exposes the edge sites of vertically-aligned 2D layers perpendicular to the substrate surface. The film presents continuously connected vertically-aligned 2D MoS2 layers, uniformly covering the entire surface (Supplementary Information, S1). The annular dark field (ADF) scanning TEM (STEM) image in Fig. 1(d) allows for a near atomic-scale investigation of a single MoS2 grain with vertically-aligned 2D layers. The image clearly identifies the individual atomic planes of molybdenum (Mo) and sulfur (S) organized in an S-Mo-S sequence with heavier Mo atoms appearing brighter than S atoms. The projected atomic structure of MoS2 is superimposed on the image, indicating an interlayer spacing of ~0.62 nm. The presented structural model matches the image of the projected MoS2 atomic structure, indicating that the MoS2 film predominantly expose the semiconducting 2H hexagonal phases27,28. For the purpose of photocatalytic performance tests, some MoS2 films with vertically-aligned 2D layers were coated with thin layers of noble metals. Figure 1(e) shows a HRTEM image of a platinum (Pt; ~3 nm thick)-deposited MoS2 film with vertically-aligned 2D layers. The image reveals that individual Pt nanoparticles are uniformly and discontinuously distributed, while the vertical 2D layer edges are well maintained even with the Pt coating (Fig. 1(f)).
Concept for photocatalytic degradation of MC-LR and TEM characterizations of MoS2 films with vertically-aligned layers. (a) Schematic illustration for photocatalytic degradation using MoS2 films. (b) Image of an as-grown MoS2 film. (c) HRTEM image to show vertically-aligned 2D MoS2 layers. (d) ADF-STEM image and atomic structure comparison of vertically-aligned 2D layers. (e) HRTEM image to show the uniform distribution of Pt nanoparticles on the vertically-aligned 2D layer edges. (f) Close-up image to show that vertical 2D layer edges are well maintained even after Pt incorporation.
Detailed structural and chemical characterizations were further performed. In Fig. 2(a), Raman spectra obtained from the MoS2 films grown with Mo seeds of various thicknesses are presented. For all film thicknesses, the Raman spectra show strong signatures of both the in-plane (E1 2g) and out-of-plane (A1g) phonon modes of MoS2. The intensity ratio of A1g mode to E1 2g mode (A1g/E2g) increases with increasing Mo thickness, which indicates the pronounced exposure of 2D edge sites and is consistent with previous studies17. Figure 2(b) shows the change in the A1g/E2g intensity ratio as a function of Mo thickness (red), while there is no significant thickness-dependent change in the frequency difference of A1g-E1 2g (blue). The thickness of the MoS2 film with vertically-aligned 2D layers was identified by cross-section TEM characterizations (Supplementary Information, S2). The thickness of initial Mo seeds increases by ~3 times after the sulfurization, which was consistently observed for Mo thickness from 5 nm to 20 nm. Figure 2(c) is the photoluminescence (PL) spectra obtained from the same MoS2 films with vertically-aligned 2D layers in Fig. 2(a) and (b). We observe that MoS2 films with a thickness of <10 nm exhibit two emission peaks centered around 1.81 eV and 1.96 eV, corresponding to the A and B excitons of MoS2 27. The observation of such strong PL peaks is interesting as they are typically observed in mono-to-few layer horizontal 2D MoS2 flakes of much smaller thickness (<a few nm)27. Similar observations of PL peaks in vertically-aligned 2D MoS2 layers have previously been reported29, while the exact mechanism for the PL emission remains unclear at present. X-ray photoelectron spectroscopy (XPS) characterizations were performed to investigate the chemical composition and the atomic bonding characteristics of the MoS2 film with vertically-aligned 2D layers. Figure 2(d) and (e) show the XPS spectra for the Mo3d and S2p core levels of the studied sample. The absence of a noticeable peak at 235.2 eV corresponding to Mo-O bonds indicates a negligible formation of molybdenum oxides. No noticeable peaks corresponding to the S-O formation in the S2p states are observed, which indicates the negligible oxidation of sulfur in the sample. The chemical compositions of the MoS2 film was determined by analyzing the Mo3d and S2p peak areas with relative sensitivity factors of 9.5 and 1.67, respectively. The atomic ratio of Mo and S is identified to be ~1:1.85, indicating a small density of S vacancies, which is commonly observed chemically synthesized MoS2 and is known to introduce n-type intrinsic doping30.
Structural and chemical characterizations by Raman, PL, and XPS. (a) Raman spectra obtained from the MoS2 films grown with Mo seed layers of various thicknesses. (b) A1g/E1 2g intensity ratio and frequency difference of A1g−E1 2g as a function of Mo thickness. (c) PL spectra obtained from the corresponding MoS2 films. XPS spectra of a MoS2 film for (d) Mo3d and (e) S2p core levels.
Electronic band structures of the MoS2 films with vertically-aligned 2D layers were identified to assess their feasibility for visible light-driven photocatalytic reactions. Figure 3(a) shows the UV-vis spectra obtained from a MoS2 film (thickness: ~20 nm) transferred on a transparent sapphire substrate. Two prominent exciton absorption peaks corresponding to A and B excitons at ~1.8 eV and ~2.0 eV are observed, which originates from the strong spin-orbit splitting of the valence band. The absorbance tail observed in the regime I at a wavelength < 1.8 eV indicates the indirect band transition (Fig. 3(a) inset). The band gap (Eg) of the sample was estimated by using the Tauc’s equation, (α(ν)*hν)1/n = A(hν − E g ), where α(ν), A, n are the absorption coefficient, proportionality constant, and gap-type depending exponent, respectively31,32. In this case, n = 2 is assigned such that multi-layered MoS2 presents indirect band gap originating from the maximum of valence band at Γ and the minimum of conduction band halfway between Γ and K33. The inset of Fig. 3(a) presents the variations of (αhν) 1/2 vs. hν for the MoS2 film with vertically-aligned 2D layers. The red straight dashed line indicates the indirectly allowed transition region in the sample, from which Eg of ~1.59 eV is extracted. This value of Eg belongs to the visible light regime of the solar spectrum, indicating that the MoS2 film with vertically-aligned 2D layers can absorb sun light up to ~780 nm. This enhanced absorption corresponds to ~50% increase in energy compared to other oxide semiconductor photocatalysts which are sensitive to UV light only (i.e. 4–5% of the whole solar spectrum). The positions of the valence band (VB) and the work function of the MoS2 film were determined by ultraviolet photoemission spectroscopy (UPS) and scanning kelvin probe microscopy (SKPM). The characterizations were performed on the MoS2 films transferred onto conductive (e.g. gold (Au)-deposited) Si/SiO2 substrates. Details are in Materials and Methods. The VB position is identified to be ~−6.07 eV, which is ~1.5 eV lower than the Fermi level of Au as shown in Fig. 3(c). The work function of the MoS2 film is extracted to be ~−4.57 eV based on the surface potential difference of ~0.3 V in Fig. 3(c). Figure 3(d) presents the band structure of the MoS2 film with respect to ROS reaction potentials. It is noted that the Fermi level of the MoS2 film is close to the conduction band (CB) edge implying its n-type carrier transport, as also predicted from XPS analysis which indicates intrinsic n-doping owing to S vacancies. The results indicate that MoS2 films with vertically-aligned 2D layers possess electronic structures suitable for ROS generation and photocatalytic reactions12. The carrier transport properties of the MoS2 film with vertically-aligned 2D layers were also evaluated by measuring its sheet resistance (Rs). Two metal contacts were deposited on the either sides of the as-grown MoS2 film channel defined by e-beam lithography (Fig. 3(e) inset). The current-voltage characteristic in Fig. 3(e) shows Ohmic transport with Rs of ~2.63 × \({10}^{9}\) Ω/□. The sheet resistance of the MoS2 film is observed to be three orders of magnitude larger than that of monolayer horizontal MoS2 flakes where the carrier transport occurs on the basal planes of 2D layers. This large in-plane Rs of the vertically-aligned 2D MoS2 layers indicates the hopping-dominated carrier transports accompanied by significant carrier scattering across the van der Waals gaps in between vertical 2D layers34.
Band structure determination by optical and electrical characterizations. (a) UV-vis spectrum from a MoS2 film with thickness of 20 nm. The inset shows the extraction of band gap (Eg). (b) UPS measurement for the determination of VB position in the MoS2 film. (c) Surface potentials of MoS2 and reference Au measured by scanning kelvin probe microscopy. (d) Band structure of the MoS2 film with respect to the redox potentials for hydrogen- or oxygen evolution reactions. (e) Current-voltage characteristics of a MoS2 film on a SiO2/Si substrate. Inset shows the optical image of the corresponding device.
Photocatalytic performances of various MoS2 films were evaluated for the visible light-driven production of ROS and degradation of MC-LR. The tested samples include pristine MoS2 and MoS2 films coated with thin (5 nm) noble metals (platinum (Pt), copper (Cu), gold/palladium (Au/Pd)). The production of ROS was monitored using H2O2 detection microsensors and 2,3-bis (2-methoxy-4-nitro-5-sulfophenyl)-2H-tetrazolium-5-carboxanilide inner salt (XTT) essay. In photocatalysis, chemical reactions occur at the photo-reactive surfaces in aqueous solutions, thus in situ measurements of the mass transfer of reactants and products are essential to understand the overall photocatalytic reactions35. The concentration gradient microprofiles of O2 and H2O2, an indication of ROS production, were directly measured using a commercial DO microsensor and a home-built Pt-based H2O2 microsensor (Fig. 4(a)). For in situ microprofiling, MoS2 samples were placed inside a microprofile chamber36 with a 2 mLmin−1 continuous flow of deionized (DI) water and a silver/silver chloride (Ag/AgCl) was used as a reference electrode (Fig. 4(b)). The ROS production from various MoS2 films immersed in MC-LR baths was quantified by the optical absorption of XTT at 470 nm wavelength. An increase in the optical density (OD) indicates an increase of ROS concentration (i.e. O2 •−) under illumination37. Figure 4(c) shows the ROS production (denoted as OD470) from MoS2 films of various types as a function of illumination time. It is clear that all the tested films present noticeable ROS production, exhibiting a linear relationship in OD470 vs. time. Amongst them, the Pt-coated MoS2 film exhibits the highest ROS production rate (determined from the slopes of the plots). DO concentration microprofiles of the corresponding samples are shown in Fig. 4(d), which indicates O2 concentrations in solutions as a function of the distance from the photo-reactive surface. The Pt-coated MoS2 exhibits the largest consumption of oxygen with a surface concentration of 5.8 mg O2 L−1, which corresponds to 2.2 mg O2 L−1 consumption in comparison to the bulk concentration (8.02 ± 0.2 mg O2 L−1). The result suggests that Pt-coated MoS2 films are highly photocatalytic under visible light illumination, and are consistent with the XTT characterizations (Fig. 4(c)). The detailed kinetics of ROS production in Pt-coated MoS2 films were further revealed by quantifying the conversion ratio of O2 to H2O2 using the H2O2 and DO microsensors (Fig. 4(e)). Details for the microsensor characterizations are in Materials and Methods. The microprofiles reveal that 60% of the O2 consumed at the film surface is being stoichiometrically converted to H2O2, following 2H2O2 ↔ O2 + 2H2O. The overall results obtained from XTT analysis and in situ microsensor characterizations confirm that Pt-coated MoS2 films efficiently produce ROS under visible light.
In situ monitoring of ROS production. (a) H2O2 and dissolved oxygen (DO) microsensors for in situ characterizations of H2O2 production and O2 consumption (b) Image of H2O2 and DO microsensors immersed in a water bath (c) ROS production from MoS2 films of various types measured by absorbance of XTT-formazan at 470 nm (optical density [OD] 470). (d) DO concentration microprofiles of various MoS2 films (e) H2O2 and DO concentration microprofiles at the surface of Pt-coated MoS2 film. 0 µm represents the top surface of the film. All the microprofiles were obtained after ~30 min exposure to water.
The direct photocatalytic removal of MC-LR with various MoS2-based films using a visible light illuminating fluorescent lamp (Spectrum data is in Supplementary Information, S3) was further investigated in Fig. 5. Figure 5(a) presents the relative concentration (normalized to initial concentration) of removed MC-LR as a function of time. Pt-coated MoS2 films present the fastest removal of MC-LR under identical conditions (pH = 5.8 and initial MC-LR concentration = 250 µg L−1), achieving a complete removal within 2 hours after the onset of illumination. It is worth mentioning that pristine MoS2 films also remove MC-LR slightly faster than Cu-only film, indicating the intrinsic photocatalytic activity of vertical MoS2 with Eg tailored to visible light. Cu-coated MoS2 films exhibit higher removal rate than both pristine MoS2 and Cu-only, indicating that metal coatings noticeably improve the overall degradation efficiency. Moreover, it is interesting to note that the removal of MC-LR occurs even before illumination (in the dark), which is attributed to the adsorption of MC-LR to the samples. Figure 5(b) compares the contribution of adsorption and photocatalytic activity that account for the total degradation of MC-LR. The plots reveal that pristine MoS2 films do not exhibit a noticeable removal of MC-LR by adsorption-only, further indicating their intrinsic photocatalytic activity. Thus, the significantly improved removal efficiency achieved in Pt-coated MoS2 films is likely the result of the synergetic effects of adsorption (mainly contributed by Pt) and photocatalytic reaction (contributed by both Pt and MoS2).
Photocatalytic degradation of MC-LR at pH 5.8. (a) Removal of MC-LR as a function of illumination time (initial MC-LR concentration of 250 μg L−1). (b) Comparison of adsorption with various samples after two hour illumination. (c) Comparison of MC-LR removal for Pt + MoS2 and Au/Pd + MoS2 at concentrations of 250 μg L−1 and 500 μg L−1. (d) Determination of rate constants for the photocatalytic degradation of MC-LR with Pt + MoS2 and Au/Pd + MoS2 based on the pseudo first-order kinetics. (e) XPS characterizations to show the appearance of N1s peaks in MC-LR tested samples (sample 1 and 2) in comparison to pristine samples (sample 3 and 4). Sample 1,3 and 2,4 were prepared from Mo films of 10 nm and 15 nm thickness, respectively.
Additional experiments were performed using Pt- and Au/Pd-coated MoS2 samples using MC-LR with initial concentration of 500 µg L−1 and were compared to the results obtained with 250 µg L−1 (Fig. 5(a)). Figure 5(c) shows that both samples efficiently degrade MC-LR under illumination while the degradation rate decreases with increasing MC-LR concentrations. The detailed kinetics for the degradation of MC-LR were analyzed. Figure 5(d) presents the degradation kinetics for both Pt- and Au/Pd-coated MoS2 in the logarithmic plots of MC-LR concentration as a function of illumination time. The plots indicate that MC-LR degradation follows pseudo first-order kinetics, yielding rate constants of 0.2451 and 0.4884 h−1 for Au/Pd and 0.4084 and 0.6769 h−1 for Pt, respectively, obtained from 500 µg L−1 and 250 µg L−1 of MC-LR. These results indicate that the incorporation of noble metals into photocatalytic MoS2 films significantly improve the efficiency of e−h+ separation resulting in enhanced photocatalytic activity. Similar results have previously been reported with other photocatalytic semiconductors which possess much larger Eg, thus, are sensitive to UV light-only38,39,40,41. In order to clarify the adsorption-driven removal of MC-LR in the dark, we performed additional XPS characterizations (Fig. 5(e)). We focused on comparing the characteristics of N1s signals for MoS2 samples exposed to MC-LR in the dark and pristine MoS2. Figure 5(e) shows the XPS spectra from the MC-LR tested (sample 1 and 2) and pristine (sample 3 and 4) samples for the binding energies between 390 eV–420 eV. The results reveal that the MC-LR tested samples present pronounced N1s peaks which are absent in pristine MoS2 films, indicating the significant adsorption of MC-LR in the dark. We further investigated the exclusive contribution of MC-LR adsorption in the dark on its overall degradation (Fig. 5(b)) by fitting a type II pseudo second order adsorption model to the MC-LR degradation kinetics obtained under dark/illumination conditions (Supplementary Information, S4). The analysis successfully decouples the adsorption and photocatalytic effects, and further verifies that the observed MC-LR degradation under illumination is a combine result of both the effects.
The effect of noble metals on enhancing photocatalytic activity can be understood considering the following factors; (a) Noble metals of high work functions in contact with n-type MoS2 form low-barrier Schottky junctions which facilitate the separation of e−h+, promoting their migration and participation in ROS generation42,43,44,45. We identify that our n-type MoS2 with vertically-aligned layers (Fig. 3(d)) in contact with Pt form low-barrier Schottky junctions with the Fermi level pinning of MoS2 close to its conduction band (Schematic in Supplementary Information, S5). Significantly enhanced photocatalytic activities have also been reported in oxide semiconductor photocatalysts of large Eg after incorporating metals39, consistent with our studies. (b) Nanoscale noble metals introduce localized surface plasmon resonances (SPR) which significantly increases optical absorption in the visible light spectrum46, leading to further enhancement in photocatalytic activity. (c) 2D MoS2 with vertically-aligned layers present ~five orders of magnitude greater chemical/physical absorbance compared to horizontal 2D MoS2 layers owing to their highly reactive 2D edge sites47. It is anticipated that an optimal mass loading of noble metals exists for optimized photocatalytic reactions39,40,48, which is to be determined by a balance between enhanced optical absorption and increased electrical conductivity (e−h+ separation efficiency).
Conclusion
In summary, we report the visible-light driven photocatalytic degradation of MC-LR using 2D MoS2 films with vertically-aligned layers. We reveal that coating thin noble metal layers on top of pristine MoS2 films significantly improves the degradation efficiency, resulting in a rapid ROS production and consequent MC-LR removal. The efficient degradation of MC-LR in metal-coated MoS2 is attributed to the combined result of the intrinsic photocatalytic activity of MoS2 with band gap energy tailored to visible light and enhanced adsorption enabled by noble metals. This study suggests the high potential of 2D MoS2 films with vertically-aligned layers for photocatalysts, thus have great implications for a wide range of environmental applications for sustainable emerging contaminants degradation and water purification.
Materials and Methods
Synthesis of MoS2 films with vertically aligned layers
MoS2 films with vertically aligned layers were grown via the sulfurization of Mo-deposited SiO2/Si substrates in a chemical vapor deposition (CVD) furnace. High-quality Mo films were deposited on Si/SiO2 wafers using an e-beam evaporation system (Thermionics VE-100) with the deposition rate of 0.15 Å/s. The Mo/Si/SiO2 substrates were placed at the center of a CVD furnace (Lindberg/Blue M Mini-Mite) while an alumina boat containing S powder is located at the upstream side. Following the evacuation down to the base pressure of ~1 mTorr and Ar purging, the CVD furnace was heated to the reaction temperature 650 °C in 15 min and was held for 10 minute under the continuous supply of Ar gas (100 SCCM). After the reaction, the furnace was naturally cooled down to room temperature. The deposition of various metals on MoS2 films with vertically-aligned layers was achieved via e-beam evaporation with the deposition rate of 0.15 Å/s.
Structural, optical, and electrical characterizations. Transmission electron microscopy (TEM) characterization
The crystalline structure and the chemical composition of 2D MoS2 films with vertically-aligned layers were characterized using a JEOL ARM200F FEG-TEM/STEM with a Cs-corrector. TEM samples were prepared by transferring the 2D MoS2 films from SiO2/Si substrates to holey carbon TEM grids by using diluted hydrogen fluoride which etches away the SiO2. All TEM/ADF-STEM operations were performed at an accelerating voltage of 200 kV.
Raman and Photoluminescence (PL) characterizations
Raman and PL characterizations were performed with a Raman spectroscopy (Renishaw) with a diode-pumped solid state laser of 532 nm wavelength and a spot size of 1 µm. Raman shifts and PL peaks were obtained under illumination for 10 seconds with power of 156 µW and 2 mW, respectively.
X-ray photoelectron spectroscopy (XPS) and ultraviolet photoemission spectroscopy (UPS) characterizations
XPS measurements were performed using PHI-5700 spectrometer with monochromatic Al Kα x-ray (1486.6 eV) below 4 × 10−9 Torr. The Shirley-type background was removed from the measured XPS spectra. For UPS and absorbance measurements, as-prepared MoS2 films on SiO2/Si growth substrates were first spin-coated with polymethyl methacrylate (PMMA). The samples were subsequently transferred onto transparent sapphire substrates followed by the etching of SiO2 and removal of PMMA.
UV-Vis absorption and electron transport characterizations
The UV-Vis absorption spectra were obtained using a UV-Vis spectrophotometer (Cary5000, Agilent). The absorption coefficient and the optical energy gap have been determined by characterizing the transmission T(λ) and reflection R(λ) spectra of the samples in the spectral wavelength range of 170~3300 nm. For electrical characterizations, the metal contacts of aluminum/chrome/gold (Al/Cr/Au) (20/5/20 nm) electrodes are fabricated on top of as-prepared MoS2 films on SiO2/Si substrates using e-beam lithography. Electrical transport properties were measured by a Keithley 4200 semiconductor parameter analyzer.
Photocatalytic degradation of MC-LR using microsensors
Dissolved oxygen (DO) and hydrogen peroxide (H2O2) concentration microprofiles were measured using a commercial DO microsensor (10 µm tip size, UNISENSE A/S, Denmark) and a platinum (Pt)-based H2O2 microsensor fabricated with a 50 µm tip diameter. A 3% hydrogen peroxide solution (H324-500, Fisher) was used to calibrate the H2O2 microsensor. The DO microsensor was calibrated in oxygen saturated (21% DO, 8.6 mg O2 L−1 at 23 °C) and deionized (DI) water under nitrogen bubbling (0% DO). The microsensors were calibrated before and after each measurement. For microprofiling, MoS2 samples were placed in a microprofile chamber with a 2 mL min−1 continuous flow of DI water. Positioning and movement of the microsensor tip towards the sample was accomplished using a three-dimension (3D) micromanipulator (UNISENSE A/S, Denmark) and observed using a stereomicroscope with a CCD camera (World Precision Instruments, Sarasota, FL, USA)16. A silver/silver chloride (Ag/AgCl) reference electrode (MI-401, Microelectrodes Inc., Bedford, NH, USA) was positioned using a helping hand (VTHH, Veleman Inc., Forth Worth, TX, USA) and a lab jack (Swiss Boy Model 110, Fisher Scientific) was used to position the MoS2 samples in view of the microscope. Microsensor electrical signal (mV or pA) was measured using a multimeter (UNISENSE A/S, Denmark) and the experiments were performed in a Faraday cage (81-334-04, Technical Manufacturing Co. Peabody, MA) to minimize electrical interference. Microprofile measurements were conducted at 50–100 µm intervals with 5 seconds of wait time between each measurement. Three replicate profiles were obtained for each parameter and the microprofiles shown in Fig. 4 are the averaged values of these replicates. Microprofiles were taken from 2,000 µm above the MoS2 film surface to the solution surface.
XTT reduction assay for monitoring ROS production
2-methoxy-4-nitro-5-sulfophenyl-2H-tetrazolium-5-carboxanilide inner salt (XTT) assay (X4626, Sigma Aldrich, St. Louis, MO) was used to investigate ROS production. XTT reduced by superoxide radical anions (O2 •−) generates water-soluble XTT-formazan with a maximum absorption at 470 nm, and the formazan produced by the reaction can be used to determine the relative amount of produced ROS49. Metal-coated MoS2 films of identical size (3 cm2) were tested. 40 mL of XTT (0.4 mM) dissolved in phosphate buffered saline was used to submerge the samples while being exposed to 16,000 Lux (4.47 × 10−3 W cm−2) continuous cool-white fluorescent light illumination. A shaker table was used at 90 RPM to keep the samples well mixed. Absorbance at 470 nm was taken using a spectrophotometer (DR 900, HACH Co., Loveland, CO) for two days to determine the rate of ROS generation.
Degradation of MC-LR
The photocatalytic removal of MC-LR in pristine MoS2 and metal-coated MoS2 films was investigated under an illumination with a fluorescent lamp. As reactors, PYREX™ reusable borosilicate petri-dishes (diameter: 60 mm and depth: 15 mm, Fisher Scientific) were used to contain MC-LR solutions (total volume of 10 mL in each reactor). A stack solution of MC-LR (475815, Calbiochem) of 500 mg L−1 was first prepared using SQ water (18 mega ohm of a resistivity) and then diluted to 250 μg L−1 or 500 μg L−1 with pH = 5.8. Subsequently, MoS2 samples were loaded in the reactors covered with aluminum foils and were kept for 3 hours to reach the adsorption equilibrium before illumination (light intensity of 4.75 × 10−4 W cm−2 measured with Newport broadband radiant power meter (Newport Corporation)). After illumination, the concentration of the MC-LR taken out of the reactors was determined by Agilent series 1100 high-performance liquid chromate graph (HPLC) with a C18 reversed-phase column (Supelco C18 Discover HS column, 150 mm × 2 mm, 5 μm particle size, Supelco, USA). Liquid with a unit volume of 20 μL was constantly injected under the flow rate of 0.2 mL min−2, following the previously reported method50,51,52.
References
Heisler, J. et al. Eutrophication and harmful algal blooms: A scientific consensus. Harmful Algae 8, 3–13 (2008).
Paerl, H. W., Hall, N. S. & Calandrino, E. S. Controlling harmful cyanobacterial blooms in a world experiencing anthropogenic and climatic-induced change. Sci. Total Environ. 409, 1739–1745 (2011).
Paerl, H. W. et al. Mitigating cyanobacterial harmful algal blooms in aquatic ecosystems impacted by climate change and anthropogenic nutrients. Harmful Algae 54, 213–222 (2016).
Kaushik, R. & Balasubramanian, R. Methods and Approaches used for Detection of Cyanotoxins in Environmental Samples: A Review. Crit. Rev. Env. Sci. Technol 43, 1349–1383 (2013).
Moreira, C., Ramos, V., Azevedo, J. & Vasconcelos, V. Methods to detect cyanobacteria and their toxins in the environment. Appl. Microbiol. Biotechnol. 98, 8073–8082 (2014).
Carmichael, W. W. et al. Human fatalities from cyanobacteria: Chemical and biological evidence for cyanotoxins. Environ. Health Perspect. 109, 663 (2001).
Codd, G. A. Cyanobacterial toxins, the perception of water quality, and the prioritisation of eutrophication control. Ecol. Eng. 16, 51–60 (2000).
Antoniou, M. G., Nicolaou, P. A., Shoemaker, J. A., de la Cruz, A. A. & Dionysiou, D. D. Impact of the morphological properties of thin TiO2 photocatalytic films on the detoxification of water contaminated with the cyanotoxin, microcystin-LR. Appl. Catal. B 91, 165–173 (2009).
Robertson, P. K. J., Bahnemann, D. W., Lawton, L. A. & Bellu, E. A study of the kinetic solvent isotope effect on the destruction of microcystin-LR and geosmin using TiO2 photocatalysis. Appl. Catal. B 108–109, 1–5 (2011).
Liu, I., Lawton, L. A., Bahnemann, D. W. & Robertson, P. K. J. The photocatalytic destruction of the cyanotoxin, nodularin using TiO2. Appl. Catal. B 60, 245–252 (2005).
Feitz, A. J., Waite, T. D., Jones, G. J., Boyden, B. H. & Orr, P. T. Photocatalytic Degradation of the Blue Green Algal Toxin Microcystin-LR in a Natural Organic-Aqueous Matrix. Environ. Sci. Technol. 33, 243–249 (1999).
Liu, C. et al. Rapid water disinfection using vertically aligned MoS2 nanofilms and visible light. Nat. Nanotechnol. 11, 1098–1104 (2016).
McGuigan, K. G. et al. Solar water disinfection (SODIS): A review from bench-top to roof-top. J. Hazar. Mater. 235–236, 29–46 (2012).
Chen, X. & Mao, S. S. Titanium Dioxide Nanomaterials: Synthesis, Properties, Modifications, and Applications. Chem. Rev. 107, 2891–2959 (2007).
Wang, Q. H., Kalantar-Zadeh, K., Kis, A., Coleman, J. N. & Strano, M. S. Electronics and optoelectronics of two-dimensional transition metal dichalcogenides. Nat. Nanotechnol. 7, 699–712 (2012).
Bernardi, M., Palummo, M. & Grossman, J. C. Extraordinary Sunlight Absorption and One Nanometer Thick Photovoltaics Using Two-Dimensional Monolayer Materials. Nano Lett. 13, 3664–3670 (2013).
Kong, D. et al. Synthesis of MoS2 and MoSe2 Films with Vertically Aligned Layers. Nano Lett. 13, 1341–1347 (2013).
Jung, Y., Shen, J., Sun, Y. & Cha, J. J. Chemically Synthesized Heterostructures of Two-Dimensional Molybdenum/Tungsten-Based Dichalcogenides with Vertically Aligned Layers. ACS Nano 8, 9550–9557 (2014).
Jung, Y. et al. Metal Seed Layer Thickness-Induced Transition from Vertical to Horizontal Growth of MoS2 and WS2. Nano Lett. 14, 6842–6849 (2014).
Heydari-Bafrooei, E. & Shamszadeh, N. S. Synergetic effect of CoNPs and graphene as cocatalysts for enhanced electrocatalytic hydrogen evolution activity of MoS2. RSC Adv. 6, 95979–95986 (2016).
Chen, J. et al. One-pot Synthesis of CdS Nanocrystals Hybridized with Single-Layer Transition-Metal Dichalcogenide Nanosheets for Efficient Photocatalytic Hydrogen Evolution. Angew. Chem. Int. Ed. 54, 1210–1214 (2014).
Caslake, L. F. et al. Disinfection of Contaminated Water by Using Solar Irradiation. Appl. Environ. Microbiol. 70, 1145–1151 (2004).
Singh, A. K., Mathew, K., Zhuang, H. L. & Hennig, R. G. Computational Screening of 2D Materials for Photocatalysis. J. Phys. Chem. Lett. 6, 1087–1098 (2015).
Rasmussen, F. A. & Thygesen, K. S. Computational 2D Materials Database: Electronic Structure of Transition-Metal Dichalcogenides and Oxides. J. Phys. Chem. C 119, 13169–13183 (2015).
Tong, H. et al. Nano-photocatalytic Materials: Possibilities and Challenges. Adv. Mater. 24, 229–251 (2011).
Sakthivel, S. et al. Enhancement of photocatalytic activity by metal deposition: characterisation and photonic efficiency of Pt, Au and Pd deposited on TiO2 catalyst. Water Res. 38, 3001–3008 (2004).
Deokar, G. et al. Large area growth of vertically aligned luminescent MoS2 nanosheets. Nanoscale 9, 277–287 (2017).
Li, H., Wu, H., Yuan, S. & Qian, H. Synthesis and characterization of vertically standing MoS2 nanosheets. Sci. Rep. 6, 21171 (2016).
Wang, H. et al. Electrochemical tuning of vertically aligned MoS2 nanofilms and its application in improving hydrogen evolution reaction. Proc. Nat. Aca. Sci. 110, 19701–19706 (2013).
McDonnell, S., Addou, R., Buie, C., Wallace, R. M. & Hinkle, C. L. Defect-dominated Doping and Contact Resistance in MoS2. ACS Nano 8, 2880–2888 (2014).
Choi, W. et al. High-Detectivity Multilayer MoS2 Phototransistors with Spectral Response from Ultraviolet to Infrared. Adv. Mater. 24, 5832–5836 (2012).
Saha, N. et al. Highly active spherical amorphous MoS2: facile synthesis and application in photocatalytic degradation of rose bengal dye and hydrogenation of nitroarenes. RSC Adv. 5, 88848–88856 (2015).
Mak, K. F., Lee, C., Hone, J., Shan, J. & Heinz, T. F. Atomically Thin MoS2: A New Direct-Gap Semiconductor. Phys. Rev. Lett. 105, 136805 (2010).
Li, S.-L. et al. Thickness Scaling Effect on Interfacial Barrier and Electrical Contact to Two-Dimensional MoS2 Layers. ACS Nano 8, 12836–12842 (2014).
Lazar, M. A., Varghese, S. & Nair, S. S. Photocatalytic Water Treatment by Titanium Dioxide: Recent Updates. Catalysts 2, 572–601 (2012).
Lee, W. H. et al. Needle-type environmental microsensors: design, construction and uses of microelectrodes and multi-analyte MEMS sensor arrays. Meas. Sci. Technol. 22, 042001 (2011).
Okyay, T. O. et al. Antibacterial properties and mechanisms of toxicity of sonochemically grown ZnO nanorods. RSC Adv. 5, 2568–2575 (2015).
Wang, X. & Lim, T.-T. Highly efficient and stable Ag–AgBr/TiO2 composites for destruction of Escherichia coli under visible light irradiation. Water Res. 47, 4148–4158 (2013).
Han, C. et al. UV-visible light-activated Ag-decorated, monodisperse TiO2 aggregates for treatment of the pharmaceutical oxytetracycline. Environ. Sci. Pollut. Res. 21, 11781–11793 (2013).
Pulido Melián, E. et al. Effect of deposition of silver on structural characteristics and photoactivity of TiO2-based photocatalysts. Appl. Catal. B 127, 112–120 (2012).
Lee, M.-K., Kim, T. G., Kim, W. & Sung, Y. M. Surface Plasmon Resonance (SPR) Electron and Energy Transfer in Noble Metal-Zinc Oxide Composite Nanocrystals. J. Phys. Chem. C 112, 10079–10082 (2008).
Yoon, H. S. et al. Layer dependence and gas molecule absorption property in MoS2 Schottky diode with asymmetric metal contacts. Sci. Rep. 5, 10440 (2015).
Tanabe, I. & Ozaki, Y. Consistent changes in electronic states and photocatalytic activities of metal (Au, Pd, Pt)-modified TiO2 studied by far-ultraviolet spectroscopy. Chem. Commun. 50, 2117–2119 (2014).
Ismail, A. A. Mesoporous PdO–TiO2 nanocomposites with enhanced photocatalytic activity. Appl. Catal. B 117–118, 67–72 (2012).
Subramanian, V., Wolf, E. E. & Kamat, P. V. Catalysis with TiO2/Gold Nanocomposites, Effect of Metal Particle Size on the Fermi Level Equilibration. J. Am. Chem. Soc. 126, 4943–4950 (2004).
Zhou, G. et al. Vertically aligned MoS2/MoOx heterojunction nanosheets for enhanced visible-light photocatalytic activity and photostability. Cryst. Eng. Comm. 16, 9025–9032 (2014).
Cho, S.-Y. et al. Highly Enhanced Gas Adsorption Properties in Vertically Aligned MoS2 Layers. ACS Nano 9, 9314–9321 (2015).
De Souza, M. L. & Corio, P. Effect of silver nanoparticles on TiO2-mediated photodegradation of Alizarin Red S. Appl. Catal. B 136–137, 325–333 (2013).
Yang, X. et al. Antibacterial activity of two-dimensional MoS2 sheets. Nanoscale 6, 10126–10133 (2014).
Antoniou, M. G., Shoemaker, J. A., de la Cruz, A. A. & Dionysiou, D. LC/MS/MS structure elucidation of reaction intermediates formed during the TiO2 photocatalysis of microcystin-LR. Toxicon. 51, 1103–1118 (2008).
Pelaez, M., de la Cruz, A. A., Stathatos, E., Falaras, P. & Dionysiou, D. Visible light-activated N-F-codoped TiO2 nanoparticles for the photocatalytic degradation of microcystin-LR in water. Catal. Today 144, 19–25 (2009).
Han, C. et al. Innovative visible light-activated sulfur doped TiO2 films for water treatment. Appl. Catal. B 107, 77–87 (2011).
Acknowledgements
H.-S.C. was supported by National Research Foundation of Korea (NRF) grant funded by the Korea Government (MSIP) (No. 2015R1C1A1A01052727). G.H.L. acknowledges supports by the Basic Science Research Programs through the National Research Foundation of Korea (NRF) funded by the Ministry of Science, ICT & Future Planning (NRF-2016M3A7B4910940 and NRF-2017R1A2B2006568). Y.J. acknowledges the start-up fund from the University of Central Florida.
Author information
Authors and Affiliations
Contributions
W.H.L., and Y.J. conceived the idea and led the project. M.A.I., and N.C. synthesized MoS2 films under the guidance of Y.J. J.C. carried out the microsensor characterization under the guidance of W.H.L. C.H. characterized the degradation of MC-LR. H.-S.C. performed the TEM characterization of MoS2 films. E.J., J.H.K. performed the optical and electrical characterizations of MoS2 films under the guidance of G.H.L. All authors participated in writing the paper.
Corresponding authors
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Islam, M.A., Church, J., Han, C. et al. Noble metal-coated MoS2 nanofilms with vertically-aligned 2D layers for visible light-driven photocatalytic degradation of emerging water contaminants. Sci Rep 7, 14944 (2017). https://doi.org/10.1038/s41598-017-14816-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-14816-9
This article is cited by
-
Tuning of the Optical Properties of Monolayer Blue Phosphorene
Plasmonics (2021)
-
Scalable lateral heterojunction by chemical doping of 2D TMD thin films
Scientific Reports (2020)
-
Centimeter-scale Green Integration of Layer-by-Layer 2D TMD vdW Heterostructures on Arbitrary Substrates by Water-Assisted Layer Transfer
Scientific Reports (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.