Abstract
In 2015/16, a marine heatwave associated with a record El Niño led to the third global mass bleaching event documented to date. This event impacted coral reefs around the world, including in Western Australia (WA), although WA reefs had largely escaped bleaching during previous strong El Niño years. Coral health surveys were conducted during the austral summer of 2016 in four bioregions along the WA coast (~17 degrees of latitude), ranging from tropical to temperate locations. Here we report the first El Niño-related regional-scale mass bleaching event in WA. The heatwave primarily affected the macrotidal Kimberley region in northwest WA (~16°S), where 4.5–9.3 degree heating weeks (DHW) resulted in 56.6–80.6% bleaching, demonstrating that even heat-tolerant corals from naturally extreme, thermally variable reef environments are threatened by heatwaves. Some heat stress (2.4 DHW) and bleaching (<30%) also occurred at Rottnest Island (32°01’S), whereas coral communities at Ningaloo Reef (23°9’S) and Bremer Bay (34°25’S) were not impacted. The only other major mass bleaching in WA occurred during a strong La Niña event in 2010/11 and primarily affected reefs along the central-to-southern coast. This suggests that WA reefs are now at risk of severe bleaching during both El Niño and La Niña years.
Similar content being viewed by others
Introduction
Coral reefs are in serious decline worldwide due to a combination of increasing local and global anthropogenic pressures1,2. Rising atmospheric CO2-concentrations are causing ocean warming, which leads to more intense and frequent mass coral bleaching events. To date, three global mass bleaching events (1998, 2010, and 2015/16) have been documented since the 1980s and were associated with El Niño-Southern Oscillation (ENSO) driven warming events3,4,5, highlighting the sensitivity of corals to climate-driven marine heatwaves6. Bleaching most commonly occurs during periods of thermal stress when corals lose their algal dinoflagellate symbionts (Symbiodinium spp.), resulting in a pale or white appearance of the coral colony7,8,9. Given that the majority of scleractinian corals meet most of their metabolic demand from carbon derived from symbiont photosynthesis10, bleaching results in severe resource limitation and thus significantly weakens them. While bleached corals can sometimes recover, the physiological damage caused during bleaching often results in extensive coral mortality11,12,13. Therefore, warming-related mass bleaching events are among the greatest threats to coral reefs today5,14. Since these events can lead to mass mortality from regional to global scales, they impact both the diversity and functioning of coral reef ecosystems15, and also threaten the socio-economic services on which millions of people worldwide depend16.
In 2015/16, unusually high ocean temperatures associated with one of the strongest El Niño events on record triggered an unprecedented global coral reef crisis, initiating what would become the third documented global mass bleaching event17. This event was estimated to have impacted 38% of the world’s coral reefs and became the longest and most severe mass bleaching event on record5,17. It has devastated coral reefs across all three major ocean basins3,5,11,18,19,20, and caused annually recurring mass bleaching in several locations for the first time3,21. Coral reefs in the South China Sea, for example, experienced unprecedented mass bleaching with 40% coral mortality in 201511, while in the Maldives live coral cover declined by 75% due to severe bleaching in 201619. Similarly, the Great Barrier Reef experienced the worst bleaching event in its history in 20165, followed by another severe bleaching event just one year later21.
In late 2015, the U.S. National Oceanic and Atmospheric Administration (NOAA)’s Coral Reef Watch predicted significant coral bleaching and/or mortality (alert levels 1 and 2) for most coral reefs in Western Australia (WA) during the austral summer 201617. NOAA’s bleaching forecasts showed that the greatest heat stress would occur along the northern WA coast, particularly in the remote Kimberley region. The macrotidal Kimberley region is one of the most extreme natural coral reef environments in the world, with tidal ranges up to 12 m, turbid waters associated with strong tidal currents and terrestrial sediment discharge, and sea surface temperatures (SST) exceeding 30 °C for five months per year22,23,24,25. Despite these extreme environmental conditions, highly diverse coral reefs exist throughout the Kimberley25. Interestingly, the highly fluctuating temperatures of intertidal reef habitats (up to 7 °C daily) have been shown to enhance coral thermal tolerance26, consistent with other work on thermally variable reef environments27,28. However, Kimberley corals were nevertheless not immune to severe heat stress simulated in a tank experiment26, raising the question how they would respond to periods of elevated temperatures associated with marine heat waves.
To date, regional-scale mass bleaching (i.e. extending 100 s of kilometres) has been documented in WA only once during a strong La Niña event in 2010/1129,30,31,32,33, but not during strong El Niño years (e.g. 1997/98, 2010) that caused mass bleaching in many other locations around the world. Although some offshore oceanic coral atolls in northern WA (e.g. Scott Reef) did bleach severely in 199834, the coastal WA region has largely been considered to be at low risk from bleaching during strong El Niño events. During the La Niña-driven heatwave in 2010/11, sea surface temperatures exceeded normal summer temperatures by an average of 3 °C along the WA coast between 22°S (Ningaloo Reef) and 34°S (Cape Leeuwin)33 (Fig. 1a). This resulted, for example, in 12–100% bleaching at the Houtman Abrolhos Islands29,35 and 79–92% at Ningaloo Reef12. However, coral reefs in northern WA, including the Kimberley, escaped this marine heatwave and associated bleaching. Early forecasts in late-2015 by NOAA’s Coral Reef Watch of an impending heatwave during the austral summer 2016 raised significant concern for northern WA, given that regional-scale mass bleaching has never occurred in WA during a strong El Niño. As a consequence we conducted extensive coral health surveys at five sites in four different bioregions along the WA coast between December 2015 and May 2016 (Fig. 2), both prior to and following the peak warming. Since El Niño events will likely increase in frequency and intensity due to climate change36, understanding how these extreme climatic events impacts coral reefs in WA is critical to predict their persistence under continued ocean warming and climate change.
Large-scale patterns of sea surface temperature (SST) anomalies within the Eastern Indian Ocean along Western Australia, monthly-averaged for the periods when SST anomalies were near maximum: (a) Feb 2011 during the La Niña; and (b) Apr 2016 during the El Niño. SST anomalies are based on NOAA 1/4° daily optimally interpolated SST version 2 (OISST V2) relative a 1971–2000 climatological mean for the respective month using data available from https://www.ncdc.noaa.gov/oisst/data-access 63. Map figures are plotted using MATLAB R2015b (http://www.mathworks.com/).
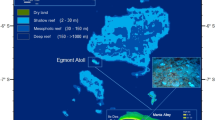
Map of Western Australia showing the locations of all survey sites and corresponding sea surface temperature (SST) plots for (a) Montgomery Reef, (b) Cygnet Bay Intertidal, (c) Cygnet Bay Subtidal, (d) Ningaloo Reef, (e) Rottnest Island, and (f) Bremer Bay. SSTs are in situ temperature data for all sites except Montgomery Reef, for which satellite-derived data were used (NOAA Coral Reef Watch 5-km virtual station North Western Australia). SSTs are shown for the 12 weeks prior to the first survey time point through to the second survey time point at each site. Dashed vertical lines indicate survey time points. MMM = maximum monthly mean, bleaching threshold = local MMM + 1 °C, DHW = Degree Heating Weeks (see Methods for more details). Line art imagery created in Adobe Illustrator (version CS6) based on a map image created in R software (version 3.4.0, R Core Team, 2017, https://www.R-project.org) by S. Comeau.
Methods
Underwater coral health surveys were conducted at five sites in four different bioregions along the WA coast, spanning ~2900 km and ~17 degrees of latitude, and ranging from a tropical climate in the north to a temperate climate in the south. From north to south, these sites/bioregions were Montgomery Reef and Cygnet Bay (Kimberley region), Coral Bay (Ningaloo Reef), Rottnest Island (subtropical Southwest), and Bremer Bay (temperate Southwest) (Fig. 2). Coral health was assessed at all sites except Montgomery Reef prior to and during the predicted heat stress period between December 2015 and May 2016 (Table 1).
Survey sites
Kimberley region (Montgomery Reef, Cygnet Bay)
Surveys in the macrotidal Kimberley region were conducted at remote Montgomery Reef (15°59′33.29″S, 124°4′9.34″E; Fig. 2), which is Australia’s largest inshore reef (380 km2). The tidal range in this area is up to 12 m during spring tide, resulting in vast areas of the reef being regularly exposed to air and forming a large plateau at low tide. While the plateau itself is dominated by coralline algae, seaweed and solitary corals, diverse hard coral communities are found along the slopes of the plateau, consisting mostly of sub-massive corals37. Due to the remote location of Montgomery Reef, surveys in the shallow subtidal were conducted only in April 2016 when severe bleaching was detected at Cygnet Bay ~120 km to the southwest (Fig. 2). These surveys were conducted concurrently with extensive aerial surveys of the region5. In situ temperature data are not available for Montgomery Reef, but seasonal monthly temperatures generally range from ~25.5 °C to ~31 °C in the region, with a long-term maximum monthly mean (MMM) sea surface temperature (SST) of 30.827 °C38.
Surveys were also conducted in the subtidal and intertidal environments at Shell Island, Cygnet Bay (16°28′46.8″S, 123°2′36.6″E; Fig. 2) in January and April 2016. These environments are described in detail elsewhere24,26. The spring tidal range in Cygnet Bay is ~8 m. At low tide, the intertidal represents a shallow tide pool with a slack water period of several hours, resulting in extreme temperature fluctuations (up to 7 °C daily, maxima of up to 37 °C) and regular aerial exposure of shallow corals. In contrast, the subtidal zone experiences more moderate temperatures where aerial exposure of coral occurs only a few days per year during extreme low tides24. Branching Acropora colonies dominate coral cover in both environments. In situ seasonal temperatures in both environments are similar and range from ~22.0 °C to 31.5 °C (daily averages)24. Bleaching thresholds for both intertidal and subtidal corals were previously experimentally established to be ~32 °C, ~1 °C above the local MMM26. We therefore used the long-term MMM of 30.827 °C from NOAA’s 5-km virtual station North Western Australia38 for our analyses.
Ningaloo Reef (Coral Bay)
Surveys were conducted at Coral Bay (23°9′21.47″S, 113°45′7.79″E; Fig. 2) located in the southern section of Ningaloo Reef, which is Australia’s largest fringing reef encompassing over 5000 square kilometers of ocean. Surveys were conducted on the reef flat during February and May 2016, at a depth of ~2 m. Coral cover at this site primarily consists of branching Acropora corals12. Seasonal monthly temperatures range from ~22–23 °C to ~27–28 °C39. We used the NOAA long-term MMM of 27.399 °C for our analyses40.
Subtropical Southwest (Rottnest Island)
Rottnest Island is located ~20 km off the coast of Perth in southwestern Australia (32°01′00.4″S, 115°31′06.0″E; Fig. 2), and is characterized by a combination of subtropical and temperate marine habitats due to the influence of the Leeuwin Current, which transports warm oligotrophic water poleward along the Western Australian coastline. These waters support over 25 species of scleractinian corals at this high-latitude location41. Surveys were conducted in February and April 2016 at a depth of ~1–2 m in Salmon Bay where coral communities are dominated by Acropora and Pocillopora 42. This is the southernmost documented occurrence of the genus Acropora in WA. In situ seasonal temperatures in 2013–2014 ranged from ~18 °C to 23 °C42. We used the NOAA long-term MMM of 22.323 °C for our analyses43.
Temperate Southwest (Bremer Bay)
Bremer Bay is located on the southern coast of WA, ~400 km southeast of Perth (Fig. 2), and features extensive high-latitude coral communities dominated by Turbinaria reniformis colonies growing at the geographical limits of their distribution in WA. Surveys were conducted in December 2015 and March 2016 at a depth of ~8 m at both Back Beach (34°25′16.57″S, 119°23′35.43″E) and Little Boat Harbour (34°28′8.50′S, 119°21′34.09″E). In situ seasonal temperatures range from ~16 °C to ~21 °C, with a MMM of 21.3 °C (historical data records obtained from the Bremer Bay “888 Abalone” farm located ~20 m from Back Beach for August 2009 to December 2014 were combined with in situ temperature data from December 2014 onwards).
Coral health surveys and sea surface temperature monitoring
The following methodology was used for surveys at all sites except Bremer Bay (see Table 1 for details). Four to six 15 m transects were conducted at randomized locations via intertidal walking or snorkeling. Care was taken to ensure that all transects were conducted at a similar depth. High-resolution photos of a 50×50 cm quadrat were taken every 0.5–1 m along the transect line. At Bremer Bay, a different methodology was used due to the sporadic coral cover at this high-latitude site. Divers conducted roving diver surveys44,45, photographing and scoring any coral colonies that they encountered within a 30 min time period.
HOBO U22 v2 temperature loggers (±0.2 °C; Onset Computer Corp) were deployed at each site except the remote Montgomery Reef, and continuously recorded in situ water temperature every 15 minutes during the study period (i.e. from 22 October 2015 to 06 April 2016 at Cygnet Bay, from 24 November 2015 to 17 May 2016 at Coral Bay, from 14 November 2015 to 01 April 2016 at Rottnest Island, and 16 September 2015 to 01 March 2016 at Bremer Bay). These time periods were chosen to assess heat stress during the 12 weeks prior to the first survey time point and between the first and second survey time point. For Montgomery Reef, satellite-derived SSTs from NOAA’s 5-km virtual station North Western Australia were used for a similar time period as at nearby Cygnet Bay (9 November 2015 to 23 April 2016). To assess cumulative heat stress, degree heating days (DHD)46 were calculated as the sum of all positive temperature anomalies (i.e. daily average SST exceeding the local MMM) over the previous 12 weeks; this was found to provide more realistic estimates of heat stress than NOAA’s methodology of accumulating for only positive temperature anomalies ≥1 °C (see Discussion). Large DHD values were converted to Degree Heating Weeks (DHW) by dividing by 7. The bleaching threshold was set at 1 °C above the local MMM, which is generally thought to be the threshold for bleaching in most coral species47,48.
Photoquadrat analysis
Photoquadrats were analyzed using the software photoQuad49 by one person to keep observer bias equal across all photos. The outline of the quadrat within the photo was manually defined. Substrate type was defined using stratified random point counts (100 points), such that the spawn canvas was divided into sub-cells and points spawned within each cell in a random manner. This method ensures that at least one point is present in each sub-cell. The following types of substrate were distinguished: hard coral, soft coral, seaweed/seagrass/encrusting coralline algae/turf, sand/rubble, rock, and unknown. The ‘unknown’ category was applied to quadrat areas that could not be unequivocally assigned to a substrate category; this was primarily the case for Kimberley surveys where high water turbidity and rapidly changing water levels created challenging optical conditions. Hard corals were identified to genus level when possible and further assigned a morphology (i.e. branching, plate-like/plating, encrusting and mounding/sub-massive/massive). Each coral colony was scored using the following four health categories as a categorical bleaching score13: unbleached (UB), moderately bleached (M: <50% of the colony bleached or colony pale), severely bleached (S: >50% bleached), and dead (D) (see Supplementary Figure S1).
Statistical analysis
Since survey methodology differed between Bremer Bay and the other sites, different statistical tests were used to analyse the data. For all sites except Bremer Bay, count data from the photoquadrat analyses were converted to percent abundance data and square root transformed prior to multivariate statistical analysis. The variability of the four health categories (UB, M, S and D) for each coral genus was statistically tested for differences between sites and time periods using Permutational Multivariate Analyses of Variance (PERMANOVAs), with transects as replicates, the Bray-Curtis similarity index and 9999 iterations. Several PERMANOVAs were conducted to accommodate the fact that not all sites were surveyed at all time points (i.e. Montgomery Reef), and that one site (Cygnet Bay) had two different environments (intertidal and subtidal) where coral communities are known to differ in their heat tolerance26:
-
1.
For sites that were surveyed before and during the peak heat stress, a two-way PERMANOVA was conducted to test the effects of time (2 levels) and site (4 levels: Cygnet Bay intertidal, Cygnet Bay subtidal, Ningaloo Reef, Rottnest Island) on coral health across all genera.
-
2.
To assess whether there were any differences between sites during the peak heat stress, a one-way PERMANOVA was conducted to test the effect of site (5 levels: Montgomery Reef, Cygnet Bay intertidal, Cygnet Bay subtidal, Ningaloo Reef, Rottnest Island) on coral health across all genera at the second time point only.
-
3.
To assess whether intertidal and subtidal environments at Cygnet Bay differed in their response to heat stress, two-way PERMANOVAs were conducted to test the effects of time (2 levels) and environment (2 levels: intertidal and subtidal) on both coral health across all genera and the four health categories summarized for all hard corals.
Post-hoc pairwise comparisons were calculated using a sequential Bonferroni correction to adjust p-values. Principal Component Analyses (PCAs) were used to visualize these data.
For Bremer Bay, data from the roving diver surveys at Back Beach and Little Boat Harbour were pooled after a χ² test showed no statistically significant difference in coral health between the two locations for both December 2016 and March 2016 (Table 2). A second χ² test was then used to test whether coral health differed between these two time points. All analyses were conducted using the software PAST version 3.1250. P-values ≤ 0.05 were considered significant. The raw data for this manuscript will be submitted to the Pangaea Database after publication.
Results
Sea surface temperatures and heat stress
Substantial heat stress occurred in the Kimberley region (i.e. Montgomery Reef, Cygnet Bay) and, to a lesser degree, in the subtropical Southwest (i.e. Rottnest Island) during the study period (Figs 1 and 2). Montgomery Reef was at highest risk of bleaching, with daily-averaged SSTs exceeding the local MMM for 113 days from 9 November 2015 to 23 April 2016, resulting in 64.9 DHD (9.3 DHW) by late April 2016 when surveys were conducted (Fig. 2a). Similarly, daily-averaged SSTs in the intertidal and subtidal environments at nearby Cygnet Bay exceeded the local MMM for 99 and 95 days, respectively, between 22 October 2015 and 6 April 2016, and by early April 2016 accumulated to 4.5 and 4.3 DHW, respectively (Fig. 2b,c). However, heat stress from mid-November 2015 onward had already reached ~3 DHW by the time the surveys were conducted in mid-January 2016. At Rottnest Island, SSTs exceeded the local MMM for 39 days between 14 November 2015 and 1 April 2016, resulting in 16.4 DHD (2.4 DHW) by the second survey (Fig. 2e). However, some heat stress had already occurred throughout January 2016, resulting in 1.4 DHW at the first survey. In contrast, SSTs at Ningaloo Reef and Bremer Bay did not exceed the local MMM on any day during the respective study periods (Fig. 2d,f).
2015/16 coral bleaching along the WA coastline
Initially in January 2016, no major bleaching was observed at any of the sites in the Kimberley; however, severe bleaching was observed at all Kimberley sites in April 2016. Some bleaching was observed at Rottnest Island in May 2016, whereas Ningaloo Reef and Bremer Bay did not experience major bleaching at either time point (Figs 3, 4). These results are consistent with patterns of heat stress across WA (Fig. 2). However, the site at Ningaloo Reef experienced high levels of coral mortality throughout the study period, despite no evidence of heat stress (Figs 1, 2d).
Principal components analysis (PCA) of coral health across all hard coral genera for (a) all sites that were surveyed at the two time points, (b) all sites (except Bremer Bay) at the second time point only, and (c) Cygnet Bay only at the two time points to compare coral health in the intertidal and subtidal environments. Bremer Bay was not included in these analyses due to the different survey methodology (see Methods). Vectors represent specific coral genera and their associated health status that had the greatest influence on overall coral health. Symbols represent individual transects. UB = unbleached, M = moderately bleached, S = severely bleached, D = dead.
Box plots showing percent cover of the 4 health categories summarized for all hard coral genera at (a) Montgomery Reef, (b) Cygnet Bay Intertidal, (c) Cygnet Bay Subtidal, (d) Ningaloo Reef, (e) Rottnest Island, and (f) Bremer Bay at the first and the second survey time point. The four health categories are unbleached (UB) in green, moderately bleached (M) in yellow, severely bleached (S) in orange, and dead (D) in red. The first survey time points are in December (Dec) 2015, January (Jan) or February (Feb) 2016 and the second survey time points are in March (Mar), April (Apr) or May 2016. Box plots show median and 25th and 75th percentiles, with whiskers (error bars) indicating the 10th and 90th percentile.
Sites that were surveyed before and during the predicted peak heat stress
At Cygnet Bay, Ningaloo Reef and Rottnest Island, a significant interactive effect of site and time was observed for coral health across all coral genera (Table 2). This was consistent with the PCA plot (Fig. 3a), where transects conducted prior to and during the predicted peak heat stress were clearly separated along principal component 2, driven by differences in the abundance of unbleached versus bleached Acropora corals. Transects conducted at Ningaloo Reef at the second survey time clustered along the vector for dead Acropora. Rottnest Island was clearly distinct from the other sites, particularly prior to the peak heat stress, due to unbleached Pocillopora contributing substantially to live coral cover.
These statistical findings were confirmed by trends in coral health categories summarized for all hard coral genera (Fig. 4, Table 3). Prior to the predicted peak heat stress, most corals were unbleached in the intertidal (88.6 ± 4.1% (mean ± standard error)) and subtidal (94.3 ± 2.6%) at Cygnet Bay, although some bleached and dead corals were observed (Fig. 4b,c). Similarly, most corals were unbleached at Rottnest Island (95.8 ± 1.3%, Fig. 4e). Coral cover at Ningaloo Reef was also dominated by unbleached corals (50.6 ± 17.1%, Fig. 4d), but with a high percentage of moderately bleached and dead corals compared to the other sites (21.6 ± 6.9 and 23.7 ± 10.9%, respectively). Three months later during the peak of heat stress, severely bleached corals dominated coral cover at Cygnet Bay, although there were more severely bleached corals in the subtidal (75.6 ± 4.8%) than intertidal (52.6 ± 4.0%). In contrast, the majority of corals at Rottnest Island were still unbleached (70.0 ± 14.8%) but moderately bleached corals increased from 3.7 ± 1.3% to 28.5 ± 14.3%. At Ningaloo Reef, unbleached corals decreased in abundance (31.5 ± 4.7%) and dead corals now dominated coral cover (68.3 ± 4.6%).
Comparing all sites (except Bremer Bay) during predicted peak heat stress
During the predicted peak heat stress in April 2016, all sites (excluding Bremer Bay) differed significantly from one another (Table 2). This pattern was also evident in the PCA plot (Fig. 3b), with Cygnet Bay transects having a large cover of severely bleached Acropora, particularly in the subtidal. Montgomery Reef represented a separate cluster due to coral cover generally being dominated by Montipora rather than Acropora and Pocillopora (Table 3). A large percentage (38.2 ± 2.5%) of the total coral cover at Montgomery Reef was severely bleached in April 2016 (Fig. 4a). In contrast, the typical coral cover at sites with no major bleaching was driven by unbleached corals (Fig. 3b and Fig. 4d,e). At Rottnest Island, unbleached Acropora and Pocillopora dominated coral cover, whereas unbleached Acropora dominated cover at Ningaloo Reef, despite there also being a significant percentage of dead corals (Fig. 4d,e).
Comparing intertidal and subtidal environments at Cygnet Bay
Coral health across all genera differed significantly between January and April 2016 at Cygnet Bay, but not between intertidal and subtidal environments (Table 2, Fig. 3c). However, when the same analysis was conducted with coral health pooled for all coral genera, a significant interactive effect of time and site was observed (Table 2). This is consistent with the substantially greater percentage of severely bleached corals in the subtidal (75.6 ± 4.8%) than intertidal (52.6 ± 4.0%) in April 2016 (Fig. 4b,c).
Changes in coral health at Bremer Bay
At the temperate site in Bremer Bay, a total of 123 T. reniformis colonies ranging from 2–150 cm in diameter were surveyed in December 2015 (n = 45) and March 2016 (n = 78). At both survey times, more than 90% of the surveyed colonies were unbleached (Table 3), with the remaining corals being moderately bleached (n = 4 in December, n = 6 in March; Fig. 4). Therefore, coral health was not significantly different between the two time points (Table 2).
Further details about the health status of each coral genus at the different survey time points and sites are provided in Supplementary Table S1 online.
Discussion
We show here that marine heatwaves associated with extreme climatic events such as the record-strength 2015/16 El Niño have the potential to cause unprecedented regional-scale mass bleaching, even in coral reef regions that harbour naturally heat-resistant corals that have escaped mass bleaching in previous El Niño years. This occurred in the macrotidal Kimberley region in northwestern Australia during the austral summer of 2016, where highly diverse and naturally stress-resistant coral reef communities have been observed to thrive under conditions that corals from more typical reef environments would usually not survive (e.g. long aerial exposure, daily temperature fluctuations of up to 7 °C and temperature maxima of up to 38 °C during low tide24,26,51). A recent study showed that these highly fluctuating temperatures enhance the thermal tolerance of Kimberley corals26. Nevertheless, Kimberley coral reefs experienced unprecedented mass bleaching in April 2016 in response to severe heat stress (~4–9 DHW, Fig. 2), with more than 56% and 71–80% of live coral cover being bleached at our two Kimberley study sites (Montgomery Reef and Cygnet Bay, respectively) (Fig. 4). In addition, broader-scale aerial surveys of 25 reefs in the southern Kimberley, conducted over the same time period, further confirmed the regional scale of this mass bleaching event and showed that most reefs in this region had 30–60% bleaching5. This demonstrates that even naturally heat-resistant corals from extreme temperature environments such as the Kimberley region are not immune to marine heatwaves and extreme climatic events. These findings are consistent with experimental work on Kimberley corals showing that heat stress equivalent to ~3 DHW resulted in severe bleaching and mortality, although heat stress in that study was applied over a much shorter time period (<2 weeks)26. Coral communities in other naturally extreme temperature environments, such as the Persian/Arabian Gulf where corals have the world’s highest known bleaching thresholds (~35–36 °C), are also not immune to severe heat stress and have suffered from multiple episodes of bleaching associated with significant mortality over the last three decades52. This suggests that even naturally heat-resistant corals are significantly threatened by periods of sustained ocean warming, as it is currently unclear whether they can increase their heat tolerance over the time scales required to cope with future climate change.
The marine heatwave causing the 2016 mass bleaching in the Kimberley was characterized by long-lasting exposure to small positive temperature anomalies that rarely exceeded the local MMM by more than 1 °C (Fig. 2a–c). SSTs already rose above the local MMM in November 2015, resulting in increasing heat stress and DHW from that point onwards. As a consequence, some coral genera (i.e. Seriatopora and Stylophora) were already severely bleached in January 2016 but did not substantially influence overall coral community health due to their overall low abundance. Thus, Kimberley reefs experienced cumulative heat stress for ~5 consecutive months, demonstrating that even small positive temperature anomalies can cause severe bleaching and mortality when persisting over a long duration. The severity of the bleaching event was further confirmed by the significant bleaching of massive corals, which are typically more resistant to heat stress53.
Surprisingly, Montgomery Reef appeared to experience greater heat stress, yet a lower percentage of bleaching was observed compared to Cygnet Bay (Fig. 4a–c). This could be due to several factors, such as satellite-derived SSTs overestimating in situ heat stress, and/or coral cover being dominated by plate-like Montipora rather than Acropora corals (Table 3). Importantly, the choice of the MMM value can have a substantial influence on calculated heat stress and DHW values even when in situ temperature data are available. NOAA’s MMM of 30.827 °C (version 2) for the virtual station North Western Australia shows close agreement with experimentally established bleaching thresholds (MMM + 1 °C) of ~32 °C for Kimberley corals26, verifying the applicability of this MMM value for determining the bleaching threshold. However, NOAA’s climatology was recently updated (i.e. in 2017), emphasizing the challenge of accurately defining baseline temperatures that corals are adapted to. The latest version (i.e. version 3) now lists a much lower MMM (29.903 °C) for this station than version 2. This lower MMM value is most likely far too conservative because Kimberley corals do not show any signs of visible bleaching or declines in their photochemical efficiency (Fv/Fm) when exposed to daily average temperatures of ~31 °C for almost two weeks26. While we cannot exclude the possibility that Kimberley corals may have acclimatized to rising SSTs over the last few decades, these discrepancies highlight the importance of in situ temperature records and physiological data, especially in complex macrotidal reef environments that create particular challenges for satellite-derived SST monitoring.
The large spatial scale of the 2016 mass bleaching event in northern WA was, to the best of our knowledge, unprecedented. Kimberley offshore oceanic atolls (e.g. Scott Reef) have bleached previously54,55, and bleached again in 20165; however, the vast inshore Kimberley region has escaped any bleaching prior to 2016, although it is possible that such events may have gone unnoticed or undocumented due to the remoteness of this region. The 2015/16 El Niño coincided with an extremely unusual and dry wet season in the Kimberley56, and also with the most extreme tides of the year. This likely resulted in increased temperature, light and UV stress as well as longer aerial exposure of shallow corals. Furthermore, the absence of major storms and cyclones would have prevented mitigation of both heat and light stress57. The combination of these factors most likely contributed or exacerbated heat stress58, thus resulting in unprecedented bleaching.
On finer spatial scales, the bleaching susceptibility of Kimberley coral communities differed significantly depending on small-scale differences in their thermal environment. During peak heat stress, coral health of subtidal coral communities at Cygnet Bay had declined significantly more than in intertidal communities, as indicated by a much higher percentage of severely bleached corals (Table 2, Fig. 4). This was the case despite similar exposure to heat stress (4.3 and 4.5 DHW in the subtidal and intertidal, respectively) and community composition (dominated by Acropora corals). These observations confirm experimental work showing that subtidal Acropora and Dipsastraea corals at Cygnet Bay have a lower thermal tolerance than their intertidal counterparts26. Although both intertidal and subtidal environments have similar average temperatures, they differ substantially with regard to daily temperature fluctuations and the frequency of aerial exposure during low tide26. Given that symbiont types did not differ between intertidal and subtidal corals26, our findings provide further evidence that extreme temperature fluctuations (up to 7 °C daily in the intertidal) represent a mechanism that enhances coral thermal tolerance26,27,28.
The 2016 mass bleaching event in the Kimberley region is the first such event to occur in WA during a strong El Niño year32 as regional-scale mass bleaching in WA extending over 100 s of kilometres has to date only occurred once during a strong La Niña year in 2010/1129,30,31,32,33. The 2010/11 heatwave primarily affected the central-to-southern WA coastal region (Fig. 1a), devastating coral communities from Ningaloo Reef to Rottnest Island. In the Ningaloo region, for example, the Exmouth Gulf had the highest amount of bleaching (~95%), whereas Coral Bay suffered ~25% bleaching12. In the Perth region, ~17% bleaching was observed12, which also extended to deeper reef communities at Rottnest Island (24–28 m)59. During the heatwave in 2016, satellite SST anomalies were negative (cooler) along the central-to-southern WA, which would suggest that no heat stress should occur (Fig. 1b). However, in situ temperature records showed that Rottnest Island experienced substantial heat stress (2.4 DHW, Fig. 2e). These reef-scale temperature anomalies are likely due to the positive local anomalies in air-sea heat fluxes that occurred over large scales across Western Australia at the time, which would have caused coastal warming in shallow reef waters off the coast of Perth60. This may explain why we observed 29.1% bleaching in Rottnest Island, although most corals were only moderately bleached (i.e. <50% bleached or colony pale) (Fig. 4). In contrast, no heat stress occurred in Coral Bay as the in situ SSTs stayed well below the local MMM for most of the summer period (Fig. 2d). We therefore consider it unlikely that the large proportion of dead corals observed at Coral Bay (Fig. 4), particularly in May 2016, was in some way related to unusual temperatures. Rather, we suspect that coral spawn slicks may have caused significant coral mortality at Coral Bay between our survey time points. Unusual weather conditions can trap coral spawn slicks in shallow bays, which then severely deplete oxygen concentrations in the water, resulting in mass mortality of coral and other reef organisms61,62. Such events have been documented at Coral Bay and other reefs in WA previously61,62. The high-latitude site Bremer Bay also did not experience any heat stress (Fig. 2f) and the small proportion of moderately bleached corals (Fig. 4f), thus, likely represents naturally lighter pigmentation due to seasonally higher light levels. Furthermore, it is unlikely that any heat stress occurred after early March given that SSTs generally peak in February at this site and remained below the MMM of 21.3 °C during all of March and April (C. Ross, unpublished data). To our knowledge, this site has not been surveyed during previous El Niño or La Niña years and it is therefore unclear whether it was affected by the marine heatwave in 2010/11.
This study shows that the geographic footprint of the 2010/11 and 2016 mass bleaching events in WA differed substantially (Fig. 1). While the 2010/11 event affected a larger geographic area, impacting coral reefs across 12° of latitude along the WA coast30, the 2016 event was regionally restricted to the northwestern parts of the state (although it should be noted that heat stress and bleaching occurred throughout northern Australia, including the Northern Territory and the Great Barrier Reef30,32). The 2010/11 event also coincided with a very active cyclone season30, which may have mitigated bleaching impacts to some extent57, whereas the opposite was the case for the 2016 event56, likely exacerbating heat stress. Both the 2010/11 and the 2016 event were prolonged marine heatwaves that lasted for several months, but heat stress peaked earlier (between January and early March30) in 2010/11 compared to 2016 (April-May). Bleaching patterns across these two events suggest that northern WA is particularly at risk of bleaching during strong El Niño years, whereas central-to-southern WA is vulnerable during strong La Niña years32. Rottnest Island could be particularly susceptible to frequent bleaching since it is the only site that bleached during both events, which threatens the potential of this high-latitude site to serve as climate change refuge42. Moreover, our findings highlight that regional-scale mass bleaching may now occur throughout WA during both El Niño and La Niña events32. As El Niño Southern Oscillation events will likely become more frequent and intense with continued climate change36, these findings have significant implications for the future resilience of coral reefs in WA.
References
Pandolfi, J. M., Connolly, S. R., Marshall, D. J. & Cohen, A. L. Projecting Coral Reef Futures Under Global Warming and Ocean Acidification. Science 333, 418–422 (2011).
Wilkinson, C. Status of Coral Reefs of the World: 2008. Glob. Coral Reef Monit. Netw. Reef Rainfor. Res. Cent. Townsvill Aust 296 (2008).
Eakin, C. M. et al. Global Coral Bleaching 2014–2017, Status and an Appeal of Observations. Reef Encount. News J. Int. Soc. Reef Stud. Reef Curr. Glob. Coral Bleach 7 (2016).
Hoegh-Guldberg, O. Climate change, coral bleaching and the future of the world’s coral reefs. Mar. Freshw. Res 50, 839–866 (1999).
Hughes, T. P. et al. Global warming and recurrent mass bleaching of corals. Nature 543, 373–377 (2017).
Hobday, A. J. et al. A hierarchical approach to defining marine heatwaves. Prog. Oceanogr 141, 227–238 (2016).
Hoegh-Guldberg, O. & Smith, G. J. The effect of sudden changes in temperature, light and salinity on the population density and export of zooxanthellae from the reef corals Stylophora pistillata Esper and Seriatopora hystrix Dana. J. Exp. Mar. Biol. Ecol 129, 279–303 (1989).
Jokiel, P. L. & Coles, S. L. Response of Hawaiian and other Indo-Pacific reef corals to elevated temperature. Coral Reefs 8, 155–162 (1990).
Porter, J. W., Fitt, W. K., Spero, H. J., Rogers, C. S. & White, M. W. Bleaching in reef corals: Physiological and stable isotopic responses. Proc. Natl. Acad. Sci 86, 9342–9346 (1989).
Muscatine, L., McCloskey, L. R. & Marian, R. E. Estimating the daily contribution of carbon from zooxanthellae to coral animal respiration. Limnol. Oceanogr 26, 601–611 (1981).
DeCarlo, T. M. et al. Mass coral mortality under local amplification of 2 °C ocean warming. Sci. Rep 7, 44586 (2017).
Depczynski, M. et al. Bleaching, coral mortality and subsequent survivorship on a West Australian fringing reef. Coral Reefs 32, 233–238 (2013).
McClanahan, T. R., Baird, A. H., Marshall, P. A. & Toscano, M. A. Comparing bleaching and mortality responses of hard corals between southern Kenya and the Great Barrier Reef, Australia. Mar. Pollut. Bull 48, 327–335 (2004).
Hoegh-Guldberg, O. et al. Coral Reefs Under RapidClimate Change and Ocean Acidification. Science 318, 1737–1742 (2007).
Baker, A. C., Glynn, P. W. & Riegl, B. Climate change and coral reef bleaching: An ecological assessment of long-term impacts, recovery trends and future outlook. Estuar. Coast. Shelf Sci 80, 435–471 (2008).
Moberg, F. & Folke, C. Ecological goods and services of coral reef ecosystems. Ecol. Econ 29, 215–233 (1999).
NOAA. NOAA declares third ever global coral bleaching event. Available at: http://www.noaanews.noaa.gov/stories2015/100815-noaa-declares-third-ever-global-coral-bleaching-event.html. (Accessed: 19th March 2017) (2015).
McClanahan, T. Changes in coral sensitivity to thermal anomalies. Mar. Ecol. Prog. Ser 570, 71–85 (2017).
Perry, C. T. & Morgan, K. M. Bleaching drives collapse in reef carbonate budgets and reef growth potential on southern Maldives reefs. Sci. Rep 7, (2017).
Xie, J. Y. et al. The 2014 summer coral bleaching event in subtropical Hong Kong. Mar. Pollut. Bull. https://doi.org/10.1016/j.marpolbul.2017.03.061 (2017).
ARC Centre of Excellence for Coral Reef Studies. Scientists mobilise as bleaching resumes on Great Barrier Reef. Available at: https://www.coralcoe.org.au/media-releases/scientists-mobilise-as-bleaching-resumes-on-great-barrier-reef (2017).
Purcell, S. Intertidal reefs under extreme tidal flux in Buccaneer Archipelago, Western Australia. Coral Reefs 21, 191–192 (2002).
Rosser, N. L. & Veron, J. E. N. Australian corals thriving out of water in an extreme environment. Coral Reefs 30, 21–21 (2011).
Dandan, S. S., Falter, J. L., Lowe, R. J. & McCulloch, M. T. Resilience of coral calcification to extreme temperature variations in the Kimberley region, northwest Australia. Coral Reefs 34, 1151–1163 (2015).
Richards, Z. T., Garcia, R. A., Wallace, C. C., Rosser, N. L. & Muir, P. R. A Diverse Assemblage of Reef Corals Thriving in a Dynamic Intertidal Reef Setting (Bonaparte Archipelago, Kimberley, Australia). PLOS ONE 10, e0117791 (2015).
Schoepf, V., Stat, M., Falter, J. L. & McCulloch, M. T. Limits to the thermal tolerance of corals adapted to a highly fluctuating, naturally extreme temperature environment. Sci. Rep 5, 17639 (2015).
Castillo, K. D., Ries, J. B., Weiss, J. M. & Lima, F. P. Decline of forereef corals in response to recent warming linked to history of thermal exposure. Nat. Clim. Change 2, 756–760 (2012).
Oliver, T. A. & Palumbi, S. R. Do fluctuating temperature environments elevate coral thermal tolerance? Coral Reefs 30, 429–440 (2011).
Abdo, D. A., Bellchambers, L. M. & Evans, S. N. Turning up the Heat: Increasing Temperature and Coral Bleaching at the High Latitude Coral Reefs of the Houtman Abrolhos Islands. PLoS ONE 7, e43878 (2012).
Moore, J. A. Y. et al. Unprecedented Mass Bleaching and Loss of Coral across 12° of Latitude in Western Australia in 2010–11. PLOS ONE 7, e51807 (2012).
Caputi, N. et al. The marine heat wave off Western Australia during the summer of 2010/11: 2 years on. (2014).
Zhang, N., Feng, M., Hendon, H. H., Hobday, A. J. & Zinke, J. Opposite polarities of ENSO drive distinct patterns of coral bleaching potentials in the southeast Indian Ocean. Sci. Rep 7, 2443 (2017).
Pearce, A. F. & Feng, M. The rise and fall of the ‘marine heat wave’ off Western Australia during the summer of 2010/2011. J. Mar. Syst 111–112, 139–156 (2013).
Goreau, T., McClanahan, T., Hayes, R. & Strong, A. Conservation of Coral Reefs after the 1998 Global Bleaching Event. Conserv. Biol 14, 5–15 (2000).
Smale, D. A. & Wernberg, T. Ecological observations associated with an anomalous warming event at the Houtman Abrolhos Islands, Western Australia. Coral Reefs 31, 441–441 (2012).
Cai, W. et al. Increasing frequency of extreme El Nino events due to greenhouse warming. Nat. Clim. Change 4, 111–116 (2014).
Heyward, A. & Moore, C. Benthic habitat characterisation of Montgomery reef: Assessing the distribution and relative abundance of dominant benthic communities. (2009).
NOAA. NOAA Coral Reef Watch 5-km virtual station North Western Australia, version 2. Available at: https://coralreefwatch.noaa.gov/vs/gauges/north_western_aus.php. (Accessed: 5th September 2017).
Falter, J. L., Lowe, R. J., Atkinson, M. J. & Cuet, P. Seasonal coupling and de-coupling of net calcification rates from coral reef metabolism and carbonate chemistry at Ningaloo Reef, Western Australia. J. Geophys. Res. Oceans 117, C05003 (2012).
NOAA. NOAA Coral Reef Watch 5-km virtual station Ningaloo, version 2. Available at: https://coralreefwatch.noaa.gov/vs/gauges/ningaloo.php. (Accessed: 5th September 2017).
Veron, J. & Marsh, L. Hermatypic corals of Western Australia. Records and annotated species list. Supplement. Records of the Western Australian Museum 29, 1–136 (1988).
Ross, C. L., Falter, J. L., Schoepf, V. & McCulloch, M. T. Perennial growth of hermatypic corals at Rottnest Island, Western Australia (32°S). PeerJ 3, e781 (2015).
NOAA. NOAA Coral Reef Watch 5-km virtual sation Marmion and Rottnest Island. Available at: https://coralreefwatch.noaa.gov/vs/gauges/marmion_rottnest.php. (Accessed: 5th September 2017).
Pattengil-Semmens, C. V. Learning to see underwater. Underw. Nat 25, 37–40 (2001).
Pattengil-Semmens, C. V. The reef fish assemblage of Bonaire Marine Park: and analysis of REEF Fish Survey Project data. Proc Gulf Caribb. Fish. Inst 52, (1998).
Maynard, J. A. et al. ReefTemp: An interactive monitoring system for coral bleaching using high-resolution SST and improved stress predictors. Geophys. Res. Lett 35, L05603 (2008).
Atwood, D. K., Hendee, J. C. & Mendez, A. An Assessment of Global Warming Stress on Caribbean Coral Reef Ecosystems. Bull. Mar. Sci 51, 118–130 (1992).
Glynn, P. W. & D’Croz, L. Experimental evidence for high temperature stress as the cause of El Niño-coincident coral mortality. Coral Reefs 8, 181–191 (1990).
Trygonis, V. & Sini, M. photoQuad: A dedicated seabed image processing software, and a comparative error analysis of four photoquadrat methods. J. Exp. Mar. Biol. Ecol 424-425, 99–108 (2012).
Hammer, Ø., Harper, D. A. T. & Ryan, P. D. Paleontological Statistics Software Package for Education and Data Analysis. Available at: http.palaeo-electronica. org/2001_1/past/issue1_01.htm. Palaeontologia Electronica, 2001, vol. 41, no 1. (2001).
Lowe, R. J., Pivan, X., Falter, J., Symonds, G. & Gruber, R. Rising sea levels will reduce extreme temperature variations in tide-dominated reef habitats. Sci. Adv 2, e1600825 (2016).
Coles, S. L. & Riegl, B. M. Thermal tolerances of reef corals in the Gulf: A review of the potential for increasing coral survival and adaptation to climate change through assisted translocation. Mar. Pollut. Bull 72, 323–332 (2013).
Loya, Y. et al. Coral bleaching: the winners and the losers. Ecol. Lett 4, 122–131 (2001).
AIMS. About the Australian Institute of Marine Science. AIMS northwest Australian coral bleaching update (2016). Available at: http://www.aims.gov.au/docs/media/latest-news/-/asset_publisher/EnA5gMcJvXjd/content/aims-northwest-australia-coral-bleaching-update. (Accessed: 19th March 2017).
Gilmour, J. P., Smith, L. D., Heyward, A. J., Baird, A. H. & Pratchett, M. S. Recovery of an Isolated Coral Reef System Following Severe Disturbance. Science 340, 69–71 (2013).
Australia Climate in April 2016. Available at: http://www.bom.gov.au/climate/current/month/aus/archive/201604.summary.shtml. (Accessed: 6th May 2017).
Carrigan, A. D. & Puotinen, M. Tropical cyclone cooling combats region-wide coral bleaching. Glob. Change Biol 20, 1604–1613 (2014).
Takahashi, S. & Murata, N. How do environmental stresses accelerate photoinhibition? Trends Plant Sci 13, 178–182 (2008).
Thomson, D. P., Bearham, D., Graham, F. & Eagle, J. V. High latitude, deeper water coral bleaching at Rottnest Island, Western Australia. Coral Reefs 30, 1107 (2011).
Zaker, N. H., Imberger, J. & Pattiaratchi, C. Dynamics of the coastal boundary layer off Perth, Western Australia. J. Coast. Res 1112–1130 (2007).
Hobbs, J.-P. A. & Macrae, H. Unusual weather and trapped coral spawn lead to fish kill at a remote coral atoll. Coral Reefs 31, 961–961 (2012).
Simpson, C. J., Cary, J. L. & Masini, R. J. Destruction of corals and other reef animals by coral spawn slicks on Ningaloo Reef, Western Australia. Coral Reefs 12, 185–191 (1993).
Banzon, V., Smith, T., Chin, T., Liu, C. & Hankins, W. A long-term record of blended satellite and in situ sea-surface temperature for climate monitoring, modeling and environmental studies. Earth Syst Sci Data 8, 165–176 (2016).
Acknowledgements
We thank J. Brown, A.-M. Nisumaa-Comeau, C. Lebens, A. Kuret, and the staff at Cygnet Bay Pearl Farm for assistance in the field. We also thank Frazer McGregor and the Murdoch University Coral Bay Marine Research station for logistical and field support. We thank the Bardi Jawi and the Dambimangari people who enabled this research through their advice and consent to access their traditional lands. This work was funded by the Western Australian Marine Science Institution, as part of the Kimberley Marine Research Programme, and the ARC Centre of Excellence for Coral Reef Studies (CE140100020). MTM was supported by an Australian Research Council Laureate Fellowship. VS was supported by a PADI Foundation Research Grant. SC was supported by an ARC Discovery Early Career Researcher Award (DE160100668).
Author information
Authors and Affiliations
Contributions
All authors conceived and designed components of the research. M.L.N., C.L.R., C.E.C., S.C., and V.S. conducted field work. M.L.N analyzed the data with guidance from C.E.C., S.C., and V.S. RL conducted the data analysis for Fig. 1. All authors contributed critically to the manuscript drafts and gave final approval for publication.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Le Nohaïc, M., Ross, C.L., Cornwall, C.E. et al. Marine heatwave causes unprecedented regional mass bleaching of thermally resistant corals in northwestern Australia. Sci Rep 7, 14999 (2017). https://doi.org/10.1038/s41598-017-14794-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-14794-y
This article is cited by
-
Diversity, composition and potential roles of sedimentary microbial communities in different coastal substrates around subtropical Okinawa Island, Japan
Environmental Microbiome (2024)
-
Evidence of corallivorous gastropod Drupella cornus breeding on the higher latitude reefs of Rottnest Island (32°S), Western Australia
Marine Biology (2024)
-
Optimal Proxy Indices for Annual Marine Heatwave Characteristics Using Monthly Sea Surface Temperature
Ocean Science Journal (2024)
-
Vertical structures of marine heatwaves
Nature Communications (2023)
-
Added value of a regional coupled model: the case study for marine heatwaves in the Caribbean
Climate Dynamics (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.