Abstract
Climate change vulnerability assessment (CCVA) has become a mainstay conservation decision support tool. CCVAs are recommended to incorporate three elements of vulnerability – exposure, sensitivity and adaptive capacity – yet, lack of data frequently leads to the latter being excluded. Further, weighted or unweighted scoring schemes, based on expert opinion, may be applied. Comparisons of these approaches are rare. In a CCVA for 17 Australian lizard species, we show that membership within three vulnerability categories (low, medium and high) generally remained similar regardless of the framework or scoring scheme. There was one exception however, where, under the warm/dry scenario for 2070, including adaptive capacity lead to five fewer species being classified as highly vulnerable. Two species, Eulamprus leuraensis and E. kosciuskoi, were consistently ranked the most vulnerable, primarily due to projected losses in climatically suitable habitat, narrow thermal tolerance and specialist habitat requirements. Our findings provide relevant information for prioritizing target species for conservation and choosing appropriate conservation actions. We conclude that for the species included in this study, the framework and scoring scheme used had little impact on the identification of the most vulnerable species. We caution, however, that this outcome may not apply to other taxa or regions.
Similar content being viewed by others
Introduction
Climate change poses a substantial threat to global biodiversity. Prioritizing conservation actions to ameliorate the impacts of climate change requires that we assess the vulnerability of species to climate change and identify which traits or characteristics drive their vulnerability1,2,3. To this end, climate change vulnerability assessment (CCVA) has become a mainstay conservation decision support tool4,5,6,7,8. However, most do not assess all the elements of vulnerability and hence paint an incomplete picture of a species’ vulnerability to climate change8,9.
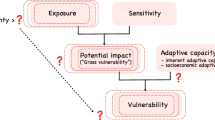
The vulnerability of a population, species or community is considered a function of three elements: exposure, sensitivity and adaptive capacity3,4,10. Exposure is the magnitude of climate change likely to be experienced by a species across its range, and depends on the rate and magnitude of climate change10. It is typically quantified using species distribution models (SDMs) fitted with climate variables, and projected onto downscaled output from regional or global circulation models (RCMs, GCMs)10,11. Most studies have measured exposure using projected change in a species’ climatically suitable habitat, overlap between the current and future suitable habitat and projected loss of suitable habitat within protected areas12,13,14,15. Other studies have also included changes in food availability, extreme weather and sea level16,17.
Sensitivity is the degree to which the performance, survival and persistence of species is affected by climate change10. Most CCVAs have assessed sensitivity using life-history traits, including physiological, behavioural or ecological traits, such as tolerance to temperature or hydrological regimes, habitat specificity and dietary specialization, occupied area, population size, reproductive rate, temperature-dependent sex determination, clutch size, growth rate, generation length, and life span3,7,10,11,13,15,18. In general, the number and combination of life-history traits employed in CCVAs varies with the study species, their habitats and data availability5.
Adaptive capacity is the potential for species or populations to tolerate or adapt to climate change10. Like sensitivity, adaptive capacity is governed by intrinsic traits, but can be influenced by extrinsic traits, such as habitat loss and fragmentation19. Although the distinction between sensitivity and adaptive capacity is somewhat ambiguous, dispersal and colonization ability, microevolution and phenotypic plasticity are generally regarded as the components of adaptive capacity10,19. Dispersal allows organisms to move to regions with suitable habitat20,21. It also promotes gene flow that increases genetic diversity, fitness and evolutionary potential of geographically isolated populations22. However, it is unclear whether the natural dispersal rates of species, particularly philopatric species, will be sufficient to track the movement of climate zones23,24. Also, anthropogenic and natural barrier to dispersal (e.g., roads, large water bodies, mountain ranges), threatening processes (e.g., predation, disease, hunting) in the landscape and aspects of climate that affect dispersal capacity may prevent organisms from tracking their climate niche25,26,27.
Evolutionary adaptive capacity is the ability for species or populations to adapt in situ through micro-evolution28. Evolution is a change in allele frequency, and thus requires adequate heritable genetic variation in populations29,30. Although the rate of evolutionary adaptation varies among species and populations, and in space and time28, it occurs faster in species with high genetic diversity, large population size, high fecundity and short generation time31. For many species, it remains unclear if they can adapt at a sufficient speed to counter the projected rate of climate change. Recent studies, however, have suggested that microevolution may be more rapid than previously thought28,32.
In addition to evolutionary adaptation, species may exhibit phenotypic plasticity, whereby individuals change their phenology, physiology or morphology without undergoing changes in their genetic makeup33. Individuals have greater fitness when their phenotypes suit the environment34,35, yet as climate changes, the phenotype and phenology of populations may no longer confer high fitness35,36. Although phenotypic plasticity alone may not be sufficient for the long-term persistence of species under rapid climate change37 and in fact, may buffer selection and slow evolution (i.e., the Baldwin effect)29,38, it can increase the rate of evolution or buy time for evolutionary adaption35.
The components of adaptive capacity can be assessed by empirical, observational and modelling studies10. Yet, for most species, there is very little available information on dispersal rates, evolutionary capacity and phenotypic plasticity, or the thresholds at which they are considered adequate to counter the impacts of climate change19. Therefore, like sensitivity, adaptive capacity is relative and a better understanding of the contributions of its components to a species’ resilience and resistance to climate change is crucial for advancing its quantification3.
A robust CCVA should account for all three elements, thereby facilitating identification of the most vulnerable species and the characteristics that determine their vulnerability8,10. However, recent reviews indicate that CCVAs are commonly based on sensitivity and exposure9,39. It has been suggested that assessments that fail to account for any of the three elements may be incomplete and produce biased outcomes, thereby rendering them less reliable for guiding conservation decisions4,8,15.
Another difference in the approach to CCVA is whether all traits incorporated into the analysis contribute equally to a species’ vulnerability score. Both unweighted and weighted scoring schemes (where the latter attempts to capture the perceived relative contribution of the various traits to vulnerability) have been used e.g.13,14,15,40, but the outcome of these scoring systems have been rarely compared.
Here, we adopted and extended existing CCVA frameworks to include the capacity of species to adapt to climate change and explored the different approaches to undertaking CCVAs. Our main objective was to assess the relative vulnerability of 17 lizard species distributed partly or wholly along the Great Dividing Range of southeastern Australia, and the factors that make them susceptible to climate change. In doing so, we evaluated the extent to which the omission of adaptive capacity in CCVA, and the weighting of scoring schemes, influences the assessment outcome. Specifically, we asked the following questions: (i) Which species are most vulnerable to climate change and what factors are responsible for their vulnerability? (ii) Does the omission of adaptive capacity influence the outcome of CCVAs? (iii) What effect does unweighted and weighted systems have on the outcome of CCVAs? We hypothesize that the number and composition of species ranked as highly vulnerable could change when adaptive capacity is included in the CCVA. We also expect the weighted system to influence the composition and order of species listed under different vulnerability categories.
Results
Exposure
Loss of suitable habitat varied across climate trajectories and time horizons, with up to 15 lizards (88%) projected to lose portions of their climatically suitable habitat by 2070 (Supporting Information Table S1). Seven species (41%), including Eulamprus leuraensis, Eulamprus kosciuskoi, Eulamprus heatwolei, Eulamprus tympanum, Egernia frerei, Egernia kingii and Egernia cunninghami were projected to lose at least 50% of their current suitable habitat by 2070. Lissolepis coventryi was the only species projected to gain (26%-136%) climatically suitable habitat under all climate scenarios and time horizons considered (Supporting Information Table S1). The proportion of species assigned to the three exposure categories (low, moderate and high) also varied with climate trajectory and time horizon. In general, most species were assigned to the moderate and low exposure categories (Fig. 1), with no significant difference between the weighted and unweighted scoring schemes (Fisher’s exact test: df = 32; p > 0.05). Eulamprus leuraensis was the only species assigned to the high exposure category under all the climate trajectories and time horizons considered.
Weighted and unweighted exposure scores for 17 Australian lizards under projected climate change by 2050. Exposure was assessed using four contrasting future climate scenarios that are equally plausible: hot/wet; warm/dry; hot/similar precipitation; and warm/ wet scenarios. Score ≥0.67 is high, from 0.33 to 0.66 is moderate and <0.33 is low. See Supporting Information Figures S1 and S2 for exposure scores at 2030 and 2070.
Sensitivity
Two species were categorised as having high sensitivity to climate change: E. leuraensis and L. coventryi, both of which are habitat specialists, have small population sizes and narrow geographic range sizes. The remaining species were assigned to the moderate sensitivity category, with scores between 55–66 for the unweighted score and 40–64 for the weighted score. Sensitivity scores for the two scoring schemes were strongly correlated (Spearman’s correlation, r s = 0.91, df = 32, p < 0.01), and although the unweighted scores were higher than the weighted scores (Fig. 2) this had no effect on the proportion and identity of the species assigned to the various sensitivity categories.
Adaptive capacity
None of the 17 species were classified as having low adaptive capacity (Ac), and those with high Ac were characterised by high genetic diversity and dispersal capacity. Depending upon the scoring scheme and time period, up to 76% of the species were assigned to the moderate Ac category. Six species, Cyclodomorphus gerrardii, Varanus varius, Egernia striolata, E. cunninghami, E. kingii and E. frerei were classified as having high Ac under the unweighted scoring scheme. Four of these (excluding E. frerei and E. striolata) were also assigned to the high Ac category by the weighted scoring scheme.
Overall vulnerability
Accounting for exposure, sensitivity and adaptive capacity
Although the vulnerability scores for individual species varied across the four climate scenarios and three time horizons (Supporting Information Table S6), membership within the three vulnerability categories remained relatively similar. Generally, there were no significant differences between the two scoring schemes or between the CCVA with (ES) and without (ESA) adaptive capacity (Fisher’s exact test: df = 32, p > 0.05) (Fig. 3). There was an exception, however. In the unweighted scoring scheme, under the warm/dry scenario for 2070, omitting adaptive capacity led to five additional species being classified as highly vulnerable, compared to the two (E. leuraensis and E. kosciuskoi) included in the ESA framework. Indeed, E. leuraensis and E. kosciuskoi were classified as highly vulnerable in both frameworks and scoring schemes, across most of the climate scenarios and time horizons.
According to the unweighted scheme, E. frerei, E. cunninghami and E. kingii are borderline potential adapters because they are highly exposed under at least one climate scenario and time slice, and have high adaptive capacity, but moderate sensitivity to climate change. Under both unweighted and weighted schemes, Lissolepis coventryi is on the borderline of high latent risk because it has low exposure, high sensitivity and moderate adaptive capacity across all the climate scenarios and at least two time slices.
Discussion
In this era of rapid climate change, making informed decisions about where and how to allocate resources for conservation is crucial8,41. Here, we applied a CCVA framework that integrates species’ exposure, sensitivity and capacity for adapting to climate change, to 17 lizard species distributed across the Great Dividing Range of Australia. As hypothesized, we found that incorporating adaptive capacity into a CCVA influenced the composition of species assigned to the various vulnerability categories, although there were exceptions. Our results also highlighted how the degree of exposure to climate change is temporally dynamic, and ought to be assessed over multiple time horizons to facilitate informed, species-specific management decisions. Our findings provide relevant information to guide conservation strategies for Australian lizards under climate change.
When all three elements of vulnerability were integrated, the difference between the number of species within the various vulnerability categories under the weighted and unweighted schemes were not significant (Fisher’s exact test: df = 32; p > 0.05). The composition of species within the three vulnerability categories also did not change. The majority (≥59%) of lizard species were classified as moderately vulnerable under both schemes, with only one to two assigned to the high vulnerability category (weighted and unweighted schemes, respectively). The two species, E. leuraensis and E. kosciuskoi, were assigned to the high vulnerability category. The key contributors to their vulnerability were substantial losses in the spatial extent of climatically suitable habitat, narrow thermal tolerance, small geographic range, and low dispersal capacity. However, it has been suggested that species assigned to a high vulnerability category may persist if they are able to adapt to the novel future climate3,10. Although both E. leuraensis and E. kosciuskoi have moderate adaptive capacity, this measure is relative and does not highlight a species’ true capacity to adapt to changes in climate19. As such, active monitoring and management of these species might be necessary to prevent climate-driven extinction. Actions should be aimed at reducing their sensitivity to climate change; external stressors, such as habitat loss and degradation, predation by feral cats, and competition with invasive species should be managed. Where there is evidence that their populations are declining over time, assisted migration may be necessary to safeguard the species from extinction.
The species that are not currently at risk from climate change could also be categorized as being potential adapters, potential persisters, or as having high latent risk, depending on their exposure, sensitivity and adaptive capacity rankings. Each of these classes has particular implications for conservation management3. Potential adapters are the species that are highly exposed and sensitive to climate change, but have high capacity to adapt and tolerate the impacts of the change3,10. Potential persisters are species that are highly exposed and have low adaptive capacity, but are not sensitive to climate change3,10. Species in this class may be able to tolerate changes in climate conditions. Species with high latent risk are those with high sensitivity and low adaptive capacity, but are currently not highly exposed to climate change. These species are not of immediate management concern, but could become highly vulnerable if exposed beyond the modelled time frame or if the rate of change in exposure outpaces that in the GCMs that we used3,10.
According to the unweighted scheme, E. frerei, E. cunninghami and E. kingii are highly exposed under at least one climate scenario and time slice, and have high adaptive capacity, but moderate sensitivity to climate change. These species are therefore borderline potential adapters and must be monitored to ensure they retain stable populations over time. Under both schemes, Lissolepis coventryi has low exposure, high sensitivity and moderate adaptive capacity across all the climate scenarios and at least two time slices. This species lies at the border of high latent risk and not of immediate conservation concern. Species that are highly exposed, but have moderate sensitivity and adaptive capacity, such as E. leuraensis, E. tympanum are also not of immediate concern, but represent new priorities for conservation. Changes to external stressors, such as habitat degradation and high predation, competition and disease, which increase their sensitivity and decrease their adaptive capacity may result in these species becoming more vulnerable to climate change.
The species assigned to the high exposure category are projected to experience greater losses in climatically suitable habitat than those assigned to the moderate and low categories. This supports the findings of other studies on Australian reptiles13,14 and suggests that loss in climatically suitable habitat will be a key driver of the vulnerability of lizards in Australia. For species with sufficient dispersal capabilities, creating and maintaining connectivity between current and future habitat may be necessary to facilitate their movement across the landscape42,43. Connecting populations that are in close proximity, but are separated by physical barriers, could also enhance gene flow, genetic diversity and the capacity of species to adapt to climate change.
A frequent challenge to CCVAs has been the lack of data on adaptive capacity8,19. Yet, we demonstrate that ignoring adaptive capacity can influence the results; more species were classified as highly vulnerable, especially under the weighted scheme. This may impede reliable identification of conservation priorities, leading to suboptimal use of limited conservation resources. This emphasizes the need to comprehensively assess all three elements of vulnerability where such data are available, as advocated by recent studies3,8.
Our study identified only two species as highly vulnerable to climate change. However, these results should be interpreted with caution, because we only considered 17 species in our analysis, and it is possible that we excluded several species that are highly vulnerable to climate change. For example, the montane specialist skink Lampropholis robertsi and its congener L. coggeri are endemic to rainforests of northeast Australia, have relatively narrow distributions, occur within a narrow climatic zone, and have relatively low dispersal capacity. These characteristics suggest they might be highly vulnerable to climate change, but they were not included in our analysis because their occurrence records were too few to allow for reliable models.
Like exposure and sensitivity, adaptive capacity scores are relative and are sensitive to the kind and number of variables used. Also, there is no consensus on how to assess the relative importance the components of adaptive capacity. However, using the same set of fundamental adaptive capacity variable, genetic diversity, dispersal and migratory capacity and phenotypic plasticity19 in the same order of importance across the 17 lizard species increases the level of confidence of the vulnerability rankings of these species. This can guide prioritization of the species for conservation, although future studies will be crucial to evaluate the actual contributions of each variable to adaptive capacity across species in space and time.
Majority of our study lizards have high neutral genetic diversity (Supporting Information Table S5) and should this indeed be a good proxy for adaptive genetic variation, then it mean that they could adapt to climate change (Supporting Information Table S5). However, it is still debated if neutral genetic diversity is a good proxy for variation under natural selection44. Recent technological advances in next-generation sequencing show promise in identifying genetic markers for traits that confer thermal tolerance45. These markers could be useful to forecast adaptive responses of species to climate change in future studies46.
We ranked the dispersal capacity of lizards based on their body size, however, in general, lizards have limited dispersal capacity compared to large mammals and most birds. As yet, it is unclear if the localized mobility of lizards is sufficient to keep up with the pace of climate change12,24,47. Genetic characterization of the Cunningham’s skink48 and other lizard species49,50,51,52 showed population differentiation considerably above the FST level of 0.35, which is approximately the point at which the spread of advantageous alleles across a species’ range is prevented53. This suggests that the natural dispersal rates of these species are insufficient for them to track their climate niche even where habitats are contiguous and with no barriers to dispersal.
Moreover, the use of species distribution models to project changes in climatically suitable habitat has its own limitations that have been well documented elsewhere54,55,56. Species distribution models are sensitive to the choice of modelling algorithm57,58,59 and climate trajectory60. Therefore, different algorithms, and future changes in climate that do not conform to the climate trajectories used in the present study, could provide dramatically different exposure estimates and hence species vulnerability rankings. Other external stressors that were not accounted for, such as predation, competition and invasion, and potential disease outbreaks could substantially influence vulnerability to climate change61.
It has been shown that combining correlative and mechanistic SDMs improves the predictive accuracy of models62. Although we used only correlative SDM, future studies will benefit from the inclusion of mechanistic models in the projection of species distributions under climate change. Also, given that vulnerability categorization is relative, it is crucial to validate the vulnerability index, from even a small geographical area. Although this was beyond the scope of this study, it can be done via laboratory experiment or long-term field studies.
We have shown that the choice of CCVA frameworks and scoring schemes can influence the proportion and composition of species assigned to the different vulnerability categories. Sensitivity, exposure and adaptive capacity traits are unlikely to contribute equally to scores of species vulnerability to climate change13,14, and intuitively, weighted scores may provide a more realistic assessment. However, given the lack of empirical evidence to support vulnerability trait scores, it remains difficult to select an appropriate weighting scheme3,5. This is further complicated by the large number of variables that may be relevant for different taxonomic groups. Furthermore, even within a relatively small clade such as that investigated here, different variables may vary dramatically in their importance, across the species. Our study indicates that there is greater congruence between weighted and unweighted species’ vulnerability rankings when all three elements of vulnerability (exposure, sensitivity, and adaptive capacity) are integrated. Further studies are needed on other taxa, such as mammals and birds, and from various geographical areas to test the generality of our findings.
Materials and Methods
Study area and species
We assessed the vulnerability of lizards distributed along the Great Dividing Range (GDR) in southeastern Australia to climate change. The GDR traverses almost 3,500 km of the Australian continent, running from the west of Alps in southeast Victoria to north of Atherton in northeast Queensland49. The GDR is a key conservation area because it harbours globally endemic and endangered species63. As a result, it has been earmarked for the creation of a connectivity corridor to enhance species’ mobility and capacity to adapt to climate change64.
To demonstrate the generality and transferability of our framework, we selected lizards with a broad spectrum of natural history traits (e.g., body size, reproductive mode, thermal tolerance, habitat association) and for which data on life-history traits, ecology, genetics and occurrence records are readily available. We restricted our analysis to species whose entire distributional range fell within the boundary of the climate data used in this study (approx. 135.7–153.7 °E, 23.6–39.5 °S). Overall, 17 species, belonging to the families Scincidae (88%), Agamidae (6%) and Varanidae (6%), were analyzed.
Assessing exposure
We used Maxent (version 3.3.3k)65,66 to model the current and future distribution of suitable habitat for the 17 lizard species. Maxent is one of the most frequently used correlative SDMs because of its high predictive performance, computational efficiency and ease of use (Elith et al. 2006; Phillips and Dudik, 2008; Phillips et al. 2009). We obtained species occurrence records from the Atlas of Living Australia (ALA; www.ala.org.au). We removed duplicate (points that occurred twice or more) and questionable records (i.e., records that had low positional accuracy and those fell outside the species’ known range or known records outside the range) and those that were collected before 1950. Overall, we included more than 31,000 unique occurrence points (i.e., one point per the spatial resolution considered) for the 17 species.
Current and future climate data, at a spatial resolution of 1 × 1 km, were derived from projections developed for the NSW and ACT Regional Climate Modelling (NARCliM) project67. Projections correspond to future climates simulated by four global climate models (GCMs): MIROC3.2-medres68, ECHAM5/MPI-OM69, CGCM3.1-T4770 and CSIRO-Mk3.071, dynamically downscaled from 50 km resolution to 1 km and 250 m for south-eastern Australia using the Weather and Research Forecasting (WRF) Regional Climate Model67. Three alternate parameterizations of the WRF model (hereafter R1, R2, and R3), were used for downscaling, resulting in 12 future climate scenarios. The NARCliM project assumed the A2 emissions scenario72, which approximates the relative forcing and mean temperature trajectories of the RCP8.5 scenario73. In general, CGCM3.1 is a relatively hot/wet scenario, MIROC3.2 is a warm/wet scenario, CSIRO-Mk3.0 is a warm/dry scenario, while ECHAM5 projects major increases in temperature but little change in rainfall (hereafter: hot/similar precipitation scenario)74.
We use a suite of five predictor climatic variables that have been shown to predict the distribution of reptiles very well13,14. These included annual mean temperature, temperature seasonality, maximum temperature of the warmest month, minimum temperature of the coldest month and annual precipitation. Because microhabitats have been shown to play a significant role in thermal buffering and determining the presence of species at fine spatial resolutions75,76, and given that the most of the study species dwell in crevices of granite rock outcrops, we included an index of rock cover (Weathering Intensity Index)77 as a static predictor variable. We consider this to be an important addition because the presence of rock outcrops with suitable crevices will largely determine range filling (i.e., colonization of suitable habitats within the species range) under climate change.
We fitted the model using different combinations of Maxent features (i.e., linear, quadratic, product, threshold and hinge features), and varying levels of regularization, to control how tightly the model fitted the given occurrence points. The most realistic model settings as assessed by the smoothness of the response curves for our modelled species were linear, product, quadratic features, and a regularization multiplier of 1.5. To reduce over-prediction, background points were sampled from areas within 100 kilometres of occurrence localities. The fitted model was projected onto the current (20-year time period centred on 2000) and three future time slices (2030, 2050 and 2070). Future projections were constrained using a buffer of 300 km around the current distribution across all species to prevent the model predicting climatically suitable habitats in areas too far beyond the species’ range.
Model performance was evaluated using the area under the receiver operating characteristic curve (AUC) and the True Skill Statistic (TSS) based on 10-fold cross-validation. AUC scores range from 0 to 1, with values of 1 indicating perfect binary classifier accuracy and 0.5 suggesting that model performance is no better than random. TSS scores range from −1 to 1, where 1 indicates perfect agreement between test data and model predictions, and scores of 0 or less indicate performance no better than random78. Variables’ predictive ability and importance to the model were assessed by referring to their percentage contribution, and to the impacts of jack-knifing on model performance. A final model was fitted using all occurrence data, and habitat suitability maps were generated by projecting these models to current and future predictor data. The projected continuous habitat suitability maps were transformed into climatically suitable and unsuitable habitat using the equal training sensitivity and specificity logistic threshold79,80.
Analyses of model output
For each GCM, we computed the change in climatically suitable habitat between the current and future projections as the percentage change in the number of suitable grid cells. We also calculated the percentage of overlap between current and future suitable habitat and the percentage of suitable habitat lost or gained within protected areas. For the latter, we obtained a GIS layer of the Australian protected area network from the Collaborative Australian Protected Area Database (CAPAD 2014; available at www.environment.gov.au/parks/science/capad). Following Cabrelli and Hughes13, we assigned higher exposure scores to species projected to lose a higher proportion of their range and suitable habitat within protected areas, and to those with no overlap between current and future suitable habitat (Supporting Information Tables S1, S2 & S3).
Assessing sensitivity
We undertook an intensive survey of the published literature to compile values for 11 traits that capture the sensitivity of each species to climate change. As described below, sensitivity was based on: narrow thermal tolerance3,81; geographically localised or restricted to a single climatic zone15; specialised habitat10,15 or dietary requirements3, slow reproductive rate40; temperature-dependent sex determination or small clutches13; low rate of offspring survival; and long generation length7 or life span40. Although some of these traits are correlated (e.g., generation length and life span), we included them in the analysis because these traits often interact to determine a species sensitivity to climate change. Unless otherwise stated, values for the above traits were obtained from Chapple82 and Greer83. We assessed each trait as follows (see also Supporting Information Table 4):
Physiological thermal tolerance
Changes in body temperature (Tb) influences the physiological sensitivity and fitness of ectotherms. Very high Tb reduces an organisms’ fitness and can be lethal at the organism’s critical maximum temperature (CTmax). An organism’s integrated fitness over some time is a function of its performance curve and the Tb it experiences. Therefore, the physiological impact of climate change will depend on an organism’s field Tb relative to its maximum performance temperature84,85,86. However, because some species may already occur in regions where environmental temperature is relatively close to their CTmax 84,85,86, we used the ratio of CTmax to the median temperature across the species’ range as a proxy for physiological thermal tolerance. The median temperature each species may be exposed to in the future was calculated by overlaying occurrence records with data describing future scenarios of mean annual temperature in southeastern Australia67. Species with a relatively lower ratio of CTmax to median temperature were given higher sensitivity scores than those with a higher ratio.
Range size
We estimated each species’ range size using the number of 100 × 100 km grid cells currently occupied. Species with relatively large range sizes were given lower sensitivity scores than those with smaller ranges.
Climatic zone
To obtain the number of climatic zones occupied by our study species, we overlaid their occurrence records with Koppen’s climate classification of Australia83. Species that occurred in multiple climatic zones were given lower sensitivity scores than those that occurred in only one zone.
Habitat requirements
Habitat generalists are more likely to adapt to changing conditions with climate change87. Indeed, species with more specialized habitats have been shown to respond negatively to climate change3,88. Consequently, habitat specialists were given higher sensitivity scores than generalists (those that occurred in multiple habitats).
Dietary requirements
Cabrelli and Hughes13 classified Australian skinks as specialists, borderline specialists and generalists based on the breadth of their prey types. We adopted this classification and gave diet specialists higher sensitivity scores than generalists.
Reproductive rate
Organisms that have fast reproductive rates respond less to climate change88. Thus, species that reproduce once or more within a year were given lower sensitivity scores than those that reproduce less frequently.
Reproductive mode
The nest temperature of many reptiles determines the sex of offspring (temperature-dependent sex determination, TSD). The correlation between temperature and offspring sex ratio implies that even modest increases in mean temperature may dramatically skew the sex ratio. High increases in temperature (>4 °C) could potentially eliminate production of male offspring thereby reducing population viability89. Although females may compensate for climatic variation via behavioral changes, such as nesting earlier in the season, digging deeper, or nesting in shade90, this may not compensate completely for climate change91. For this reason, species that lay eggs were given higher sensitivity scores than those that give birth to live-young.
Number of offspring
Organisms that have higher clutch sizes are more likely to produce more genetic variant individuals to trigger adaptation87, hence species that produce more offspring (≥5) per reproductive event were given a lower sensitivity score than those that have fewer offspring per event.
Offspring survival
Organisms with more offspring reaching sexual maturity are more likely to reproduce and have enough genetic variability to trigger adaptation87. For this reason, species with less than 50% of their offspring dying before reaching sexual maturity were given higher sensitivity scores than those with more than 50% of their offspring reaching sexual maturity.
Generation length
Species with longer generation times have slower life histories and lower reproductive output87. Consequently, species reaching sexual maturity within a long time (≥5 years) were given higher sensitivity scores than those with shorter juvenile stages.
Life span
Species that live longer are less susceptible to climate change because adaptation and range shifts occur over a long time88. Therefore, species that live for 10 years or more were given lower sensitivity scores than those with shorter lifespans.
Assessing adaptive capacity
Although the theoretical basis of adaptive capacity is well understood, its quantification remains difficult and little information and guidance exists to inform its objective assessments16,19. We used four measures of adaptive capacity: genetic diversity; body size; habitat fragmentation; and microhabitat buffering (Supporting Information Table S5).
Genetic diversity
Given that evolutionary adaptation and plastic responses to climate change depend on the extent of genetic variation within species and among populations32, we used measures of genetic diversity as a proxy for adaptive potential. There are two type of genetic diversity: adaptive genetic diversity, which influences the fitness of individuals and populations and neutral genetic diversity, which confers no advantage, but may provide proxy for adaptive variation and means for monitoring gene flow and other demographic processes53. Recent genomics analyses suggest that most adaptive genetic responses are based on small effect in many genes, rather than a few genes of large effect92. These considerations suggest that evolutionary potential (a key component of adaptive capacity) is better predicted by overall genetic diversity than a focus on individual genes93. Here we used neutral genetic variation (as measured by expected heterozygosity) as a proxy for adaptive potential. Expected heterozygosity (He) for the lizards (measured by microsatellite markers) were obtained from the published literature (for references see Supporting Information Table S5). In principle, species with relatively high genetic diversity are expected to have greater adaptive potential, hence we assigned higher adaptive capacity scores to species with mean He > 0.8.
Dispersal capacity
Empirical data on the dispersal of most lizards is rare. However, given their strong correlation, we used body size as a surrogate for dispersal capacity94. In general, the larger the organism, the more mobile it is, hence larger lizards (measured by snout-vent length, SVL) were assigned relatively higher dispersal capacity, and thus adaptive capacity, than smaller ones.
Habitat loss and fragmentation
The availability of suitable habitat and degree of connectivity of the landscape can impede or enhance movement of organisms, thereby influencing their response to climate change15,95,96. To assess the availability of suitable habitat ant degree of habitat fragmentation for individual species, we overlaid species’ current range maps with a GIS layer of the land use/land cover of Australia (Australian Land Use and Management [ALUM] Classification, version 7, May 2010, available at www.abs.gov.au). For each species, we calculated the percentage of occupied grid cells that fell within pasture, crop and modified lands. We assigned a higher adaptive capacity score to species with more than 40% of their occupied grid cells within pasture, crop and modified lands, and a lower score to those with less than 10% of the occupied grid cells within these land cover types.
Microhabitat buffering
The use of microhabitat features that moderate temperature and extreme weather conditions can influence the capacity of species to cope with climate change75,76. Hence, species that used rock crevices and tree hollows as retreat sites were given higher adaptive capacity scores than those that used leaf litter or bare ground.
Vulnerability framework and scoring
We scored the variables of the three elements of vulnerability on an ordinal scale using two scoring schemes: unweighted and weighted (Table 1). In the unweighted scoring scheme, we considered the variables within each element as equally important and were awarded a maximum score of three points and a minimum score of one point following Gardali, et al.16. In the weighted scheme, the variables within each element were ranked according to their perceived relative contribution to vulnerability as deemed by a panel of experts13,14. Higher maximum scores were awarded to the variables that contributed more to climate vulnerability, with categories within variables awarded a minimum score of one and a median score of half the maximum score following Cabrelli, et al.14. For example, of the 11 sensitivity traits used in this study, thermal tolerance was regarded the most important trait and so it was awarded the highest maximum score of 11 for species with narrow thermal tolerance, a score of 5.5 for species with moderate tolerance, and a score of one for species with wide tolerance. Life span was regarded the least important of the traits and was therefore awarded the lowest maximum score of three for short-lived species, a score of two for species with moderate life-spans, and a score of one for long-lived species (Table 1).
Although these scoring schemes are highly arbitrary, they provide an easy-to-use approach to convert continuous variables into ordinal (high or low) categorization. This approach has been used in similar assessments3,16,87,97.
Climate vulnerability score and ranking threshold
To generate a climate change vulnerability score, we first divided the sum of the scores for each of the three elements of vulnerability by their respective potential maximum score to generate exposure score (Es), sensitivity score (Ss) and adaptive capacity score (As). We then computed vulnerability score with adaptive capacity (ESA) as (Es + Ss) − As and without adaptive capacity (ES) as Es + Ss. Computing ESA as (Es × Ss)/As did not change the outcome of the vulnerability rankings. We applied two of the commonly used vulnerability ranking thresholds. Firstly, following Cabrelli, et al.14, species with ESA/ES ≥67 were ranked as high vulnerability, those with ESA/ES from 33 to 66 were ranked as moderate vulnerability and those with ESA/ES < 33 were classified as low vulnerability. Secondly, following Dawson, et al.10 and Foden, et al.3, we assigned species to four vulnerability categories: high, potential adapters, potential persisters and high latent risk. High vulnerability species are those that have high sensitivity, high exposure and low adaptive capacity to climate change. Potential adapters are the species that are highly exposed and sensitive to climate change, but have high capacity to adapt and tolerate the impacts of the change (Dawson et al., 2011; Foden et al., 2013). Potential persisters are species that are highly exposed and have low adaptive capacity, but are not sensitive to climate change, while species with high latent risk are those with low exposure, but high sensitivity and low adaptive capacity to climate change (Dawson et al., 2011; Foden et al., 2013).
In the first classification, vulnerability categorization based on the raw continuous values produced similar results as categorization based on ranked values for exposure, sensitivity and adaptive capacity. The second classification however required that we first rank the species as either high, moderate or low exposure, sensitivity and adaptive capacity before determining their vulnerability status. We evaluated the relationship between the two assessment methods (i.e., with and without considering adaptive capacity) and scoring schemes (i.e., unweighted and weighted) using the Spearman’s rank correlation coefficient and analysed the difference between them using the Fisher’s exact test.
References
Carvalho, S. B., Brito, J. C., Crespo, E. J. & Possingham, H. P. Fromclimate change predictions to actions – conserving vulnerable animal groups in hotspots at a regional scale. Global Change Biology 16, 3257–3270, https://doi.org/10.1111/j.1365-2486.2010.02212.x (2010).
Williams, S. E., Shoo, L. P., Isaac, J. L., Hoffmann, A. A. & Langham, G. Towards an integrated framework for assessing the vulnerability of species to climate change. PLoS biology 6, e325 (2008).
Foden, W. B. et al. Identifying the world’s most climate change vulnerable species: a systematic trait-based assessment of all birds, amphibians and corals. PLoS One 8, e65427 (2013).
Small-Lorenz, S. L., Culp, L. A., Ryder, T. B., Will, T. C. & Marra, P. P. A blind spot in climate change vulnerability assessments. Nature Climate Change 3, 91–93 (2013).
Lankford, A. J., Svancara, L. K., Lawler, J. J. & Vierling, K. Comparison of climate change vulnerability assessments for wildlife. Wildlife Society Bulletin 38, 386–394 (2014).
Rowland, E. L., Davison, J. E. & Graumlich, L. J. Approaches to evaluating climate change impacts on species: a guide to initiating the adaptation planning process. Environmental management 47, 322–337 (2011).
Pearson, R. G. et al. Life history and spatial traits predict extinction risk due to climate change. Nature Climate Change 4, 217–221 (2014).
Butt, N. et al. Challenges in assessing the vulnerability of species to climate change to inform conservation actions. Biological Conservation 199, 10–15 (2016).
Pacifici, M. et al. Assessing species vulnerability to climate change. Nature Climate Change 5, 215–224 (2015).
Dawson, T. P., Jackson, S. T., House, J. I., Prentice, I. C. & Mace, G. M. Beyond predictions: biodiversity conservation in a changing climate. Science 332, 53–58 (2011).
Thomas, C. D. et al. A framework for assessing threats and benefits to species responding to climate change. Methods in Ecology and Evolution 2, 125–142 (2011).
Araújo, M. B. et al. Ecology Letters 14, 484–492 (2011).
Cabrelli, A. L. & Hughes, L. Assessing the vulnerability of Australian skinks to climate change. Climatic Change 130, 223–233 (2015).
Cabrelli, A. L., Stow, A. J. & Hughes, L. A framework for assessing the vulnerability of species to climate change: a case study of the Australian elapid snakes. Biodiversity & Conservation 23, 3019–3034 (2014).
Heikkinen, R. K. et al. Assessing the vulnerability of European butterflies to climate change using multiple criteria. Biodiversity and conservation 19, 695–723 (2010).
Gardali, T., Seavy, N. E., DiGaudio, R. T. & Comrack, L. A. A climate change vulnerability assessment of California’s at-risk birds. PLoS One 7, e29507 (2012).
Bush, A. A. et al. Continental-scale assessment of risk to the Australian Odonata from climate change. PloS one 9, e88958 (2014).
Tuberville, T. D., Andrews, K. M., Sperry, J. H. & Grosse, A. M. Use of the NatureServe Climate Change Vulnerability Index as an Assessment Tool for Reptiles and Amphibians: Lessons Learned. Environmental management 56, 822–834 (2015).
Nicotra, A. B., Beever, E. A., Robertson, A. L., Hofmann, G. E. & O’Leary, J. Assessing the components of adaptive capacity to improve conservation and management efforts under global change. Conservation Biology 29, 1268–1278 (2015).
Taylor, K., Brummer, T., Taper, M. L., Wing, A. & Rew, L. J. Human‐mediated long‐distance dispersal: an empirical evaluation of seed dispersal by vehicles. Diversity and Distributions 18, 942–951 (2012).
Trakhtenbrot, A., Nathan, R., Perry, G. & Richardson, D. M. The importance of long‐distance dispersal in biodiversity conservation. Diversity and Distributions 11, 173–181 (2005).
Sexton, J. P., Strauss, S. Y. & Rice, K. J. Gene flow increases fitness at the warm edge of a species’ range. Proceedings of the National Academy of Sciences 108, 11704–11709 (2011).
Niculae, M.-I., Nita, M. R., Vanau, G. O. & Patroescu, M. Evaluating the Functional Connectivity of Natura 2000 Forest Patch for Mammals in Romania. Procedia Environmental Sciences 32, 28–37 (2016).
Shaw, R. G. & Etterson, J. R. Rapid climate change and the rate of adaptation: insight from experimental quantitative genetics. New Phytologist 195, 752–765 (2012).
Bateman, B. L., Murphy, H. T., Reside, A. E., Mokany, K. & VanDerWal, J. Appropriateness of full‐, partial‐and no‐dispersal scenarios in climate change impact modelling. Diversity and Distributions 19, 1224–1234 (2013).
Castillo, J. A., Epps, C. W., Davis, A. R. & Cushman, S. A. Landscape effects on gene flow for a climate‐sensitive montane species, the American pika. Molecular ecology 23, 843–856 (2014).
Schwalm, D. et al. Habitat availability and gene flow influence diverging local population trajectories under scenarios of climate change: a place‐based approach. Global change biology (2016).
Hoffmann, A. A. & Sgro, C. M. Climate change and evolutionary adaptation. Nature 470, 479–485 (2011).
Ghalambor, C. K., McKay, J. K., Carroll, S. P. & Reznick, D. N. Adaptive versus non-adaptive phenotypic plasticity and the potential for contemporary adaptation in new environments. Functional Ecology 21, 394–407, https://doi.org/10.1111/j.1365-2435.2007.01283.x (2007).
Bell, G. & Gonzalez, A. Adaptation and evolutionary rescue in metapopulations experiencing environmental deterioration. Science 332, 1327–1330 (2011).
Gomulkiewicz, R. & Houle, D. Demographic and genetic constraints on evolution. The American Naturalist 174, E218–E229 (2009).
Sgro, C. M., Lowe, A. J. & Hoffmann, A. A. Building evolutionary resilience for conserving biodiversity under climate change. Evolutionary Applications 4, 326–337 (2011).
Pigliucci, M. Evolution of phenotypic plasticity: where are we going now? Trends in Ecology & Evolution 20, 481–486 (2005).
Kingsolver, J. G., Pfennig, D. W. & Servedio, M. R. Migration, local adaptation and the evolution of plasticity. Trends in Ecology & Evolution 17, 540–541 (2002).
Reed, T. E., Schindler, D. E. & Waples, R. S. Interacting effects of phenotypic plasticity and evolution on population persistence in a changing climate. Conservation Biology 25, 56–63 (2011).
Fuller, A. et al. Physiological mechanisms in coping with climate change. Physiological and Biochemical Zoology 83, 713–720 (2010).
Urban, M. C., Richardson, J. L. & Freidenfelds, N. A. Plasticity and genetic adaptation mediate amphibian and reptile responses to climate change. Evolutionary Applications 7, 88–103 (2014).
Robinson, B. W. & Dukas, R. The influence of phenotypic modifications on evolution: the Baldwin effect and modern perspectives. Oikos, 582–589 (1999).
Willis, S. et al. Integrating climate change vulnerability assessments from species distribution models and trait-based approaches. Biological Conservation 190, 167–178 (2015).
Moyle, P. B., Kiernan, J. D., Crain, P. K. & Quinones, R. M. Climate change vulnerability of native and alien freshwater fishes of California: a systematic assessment approach. PLoS One 8, e63883 (2013).
Ferraro, P. J. & Pattanayak, S. K. Money for nothing? A call for empirical evaluation of biodiversity conservation investments. PLoS Biol 4, e105 (2006).
Crooks, K. R. & Sanjayan, M. Connectivity conservation. Vol. 14 (Cambridge University Press, 2006).
Doerr, V. A., Barrett, T. & Doerr, E. D. Connectivity, dispersal behaviour and conservation underclimate change: a response to Hodgson et al. Journal of Applied Ecology 48, 143–147 (2011).
Holderegger, R., Kamm, U. & Gugerli, F. Adaptive vs. neutral genetic diversity: implications for landscape genetics. Landscape Ecology 21, 797–807 (2006).
Manel, S. & Holderegger, R. Ten years of landscape genetics. Trends in Ecology & Evolution 28, 614–621 (2013).
Sunday, J. M. et al. Evolution in an acidifying ocean. Trends in Ecology & Evolution 29, 117–125 (2014).
Huntley, B. & Barnard, P. Potential impacts of climatic change on southern African birds of fynbos and grassland biodiversity hotspots. Diversity and Distributions 18, 769–781 (2012).
Ofori, B. Y., Beaumont, L. J. & Stow, A. J. Cunningham’s skinks show low genetic connectivity and signatures of divergent selection across its distribution. Ecology and Evolution 7, 48–57, https://doi.org/10.1002/ece3.2627 (2017).
Pepper, M., Barquero, M. D., Whiting, M. J. & Keogh, J. S. A multi-locus molecular phylogeny for Australia’s iconic Jacky Dragon (Agamidae: Amphibolurus muricatus): Phylogeographic structure along the Great Dividing Range of south-eastern Australia. Molecular Phylogenetics and Evolution 71, 149–156 (2014).
Smissen, P. J., Melville, J., Sumner, J. & Jessop, T. S. Mountain barriers and river conduits: phylogeographical structure in a large, mobile lizard (Varanidae: Varanus varius) from eastern Australia. Journal of Biogeography 40, 1729–1740 (2013).
Bell, R. C. et al. Patterns of persistence and isolation indicate resilience to climate change in montane rainforest lizards. Molecular Ecology 19, 2531–2544 (2010).
Symula, R., Keogh, J. S. & Cannatella, D. C. Ancient phylogeographic divergence in southeastern Australia among populations of the widespread common froglet, Crinia signifera. Molecular phylogenetics and evolution 47, https://doi.org/10.1016/j.ympev.2008.01.011 (2008).
Lowe, W. H. & Allendorf, F. W. What can genetics tell us about population connectivity? Molecular Ecology 19, 3038–3051 (2010).
Pearson, R. G. et al. Model‐based uncertainty in species range prediction. Journal of Biogeography 33, 1704–1711 (2006).
Sinclair, S. J., White, M. D. & Newell, G. R. How useful are species distribution models for managing biodiversity under future climates. Ecology and Society 15 (2010).
Wiens, J. A., Stralberg, D., Jongsomjit, D., Howell, C. A. & Snyder, M. A. Niches, models, and climate change: assessing the assumptions and uncertainties. Proceedings of the National Academy of Sciences 106, 19729–19736 (2009).
Pearson, R. G. Climate change and the migration capacity of species. Trends in Ecology & Evolution 21, 111–113 (2006).
Beaumont, L. J. et al. Which species distribution models are more (or less) likely to project broad-scale, climate-induced shifts in species ranges? Ecological Modelling 342, 135–146 (2016).
Elith, J. & Graham, C. H. Do they? How do they? WHY do they differ? On finding reasons for differing performances of species distribution models. Ecography 32, 66–77 (2009).
Beaumont, L. J., Hughes, L. & Pitman, A. Why is the choice of future climate scenarios for species distribution modelling important? Ecology Letters 11, 1135–1146 (2008).
Brook, B. W., Sodhi, N. S. & Bradshaw, C. J. Synergies among extinction drivers under global change. Trends in Ecology & Evolution 23, 453–460 (2008).
Kearney, M. R., Wintle, B. A. & Porter, W. P. Correlative and mechanistic models of species distribution provide congruent forecasts under climate change. Conservation Letters 3, 203–213 (2010).
Steffen, W. et al. Australia’s biodiversity and climate change. (Csiro Publishing, 2009).
Worboys, G. & Pulsford, I. Connectivity conservation in Australian landscapes. Report prepared for the Australian Government Department of Sustainability, Environment, Water, Population and Communities on behalf of the State of the Environment 2011 Committee, Canberra (2011).
Phillips, S. J., Dudík, M. & Schapire, R. E. In Proceedings of the twenty-first international conference on Machine learning. 83 (ACM).
Phillips, S. J., Anderson, R. P. & Schapire, R. E. Maximum entropy modeling of species geographic distributions. Ecological Modelling 190, 231–259 (2006).
Evans, J. et al. Design of a regional climate modelling projection ensemble experiment–NARCliM. Geoscientific Model Development 7, 621–629 (2014).
K-I Model Developers. K-1 coupled model (MIROC) description. Tokyo, Japan: Center for Climate System Research, University of Tokyo (2004).
Jungclaus, J. et al. Ocean circulation and tropical variability in the coupled model ECHAM5/MPI-OM. Journal of Climate 19, 3952–3972 (2006).
Flato, G. The third generation coupled global climate model (CGCM3). Available on line at http://www. cccma. bc. ec. gc. ca/models/cgcm3. shtml (2005).
Gordon, H. et al. The CSIRO Mk3 climate system model. (CSIRO Atmospheric Research technical paper, 2002).
Nakicenovic, N. et al. Special report on emissions scenarios: a special report of Working Group III of the Intergovernmental Panel on Climate Change. (Pacific Northwest National Laboratory, Richland, WA (US), Environmental Molecular Sciences Laboratory (US), 2000).
Stocker, T. F. et al. Climate change 2013: The physical science basis. (Cambridge University Press Cambridge, UK, and New York, 2014).
Evans, J. & Ji, F. Choosing GCMs. (NARCliM Technical Note 1, 7pp, NARCliM Consortium, Sydney, Australia, 2012).
Scheffers, B. R. et al. Thermal buffering of microhabitats is a critical factor mediating warming vulnerability of frogs in the Philippine biodiversity hotspot. Biotropica 45, 628–635 (2013).
Scheffers, B. R., Edwards, D. P., Diesmos, A., Williams, S. E. & Evans, T. A. Microhabitats reduce animal’s exposure to climate extremes. Global Change Biology 20, 495–503 (2014).
Wilford, J. A weathering intensity index for the Australian continent using airborne gamma-ray spectrometry and digital terrain analysis. Geoderma 183, 124–142 (2012).
Allouche, O., Tsoar, A. & Kadmon, R. Assessing the accuracy of species distribution models: prevalence, kappa and the true skill statistic (TSS). Journal of Applied Ecology 43, 1223–1232 (2006).
Liu, C., Berry, P. M., Dawson, T. P. & Pearson, R. G. Selecting thresholds of occurrence in the prediction of species distributions. Ecography 28, 385–393 (2005).
Jiménez-Valverde, A. & Lobo, J. M. Threshold criteria for conversion of probability of species presence to either–or presence–absence. Acta oecologica 31, 361–369 (2007).
Deutsch, C. A. et al. Impacts of climate warming on terrestrial ectotherms across latitude. Proceedings of the National Academy of Sciences 105, 6668–6672 (2008).
Chapple, D. G. Ecology, life-history, and behavior in the Australian scincid genus Egernia, with comments on the evolution of complex sociality in lizards. Herpetological Monographs 17, 145–180 (2003).
Greer, A. Critical thermal maximum temperatures in Australian scincid lizards: their ecological and evolutionary significance. Australian Journal of Zoology 28, 91–102 (1980).
Clusella-Trullas, S. & Chown, S. L. Lizard thermal trait variation at multiple scales: a review. Journal of Comparative Physiology B 184, 5–21 (2014).
Huey, R. B. et al. Why tropical forest lizards are vulnerable to climate warming. Proceedings of the Royal Society of London B: Biological Sciences, rspb. 2008, 1957 (2009).
Diamond, S. E. et al. Who likes it hot? A global analysis of the climatic, ecological, and evolutionary determinants of warming tolerance in ants. Global Change Biology 18, 448–456 (2012).
Böhm, M. et al. Hot and bothered: using trait-based approaches to assess climate change vulnerability in reptiles. Biological Conservation 204, 32–41 (2016).
Pacifici, M. et al. Species’ traits influenced their response to recent climate change. Nature Climate Change 7, 205–208 (2017).
Mitchell, N. & Janzen, F. Temperature-dependent sex determination and contemporary climate change. Sexual Development 4, 129–140 (2010).
Doody, J. S. et al. Nest site choice compensates for climate effects on sex ratios in a lizard with environmental sex determination. Evolutionary Ecology 20, 307–330 (2006).
Telemeco, R. S., Elphick, M. J. & Shine, R. Nesting lizards (Bassiana duperreyi) compensate partly, but not completely, for climate change. Ecology 90, 17–22 (2009).
Pritchard, J. K., Pickrell, J. K. & Coop, G. The genetics of human adaptation: hard sweeps, soft sweeps, and polygenic adaptation. Current Biology 20, 208–215 (2010).
Pritchard, J. K. & Di Rienzo, A. Adaptation–not by sweeps alone. Nature Reviews Genetics 11, 665–667 (2010).
Sutherland, G. D., Harestad, A. S., Price, K. & Lertzman, K. P. Scaling of natal dispersal distances in terrestrial birds and mammals. Conservation Ecology 4, 16 (2000).
Broennimann, O. et al. Do geographic distribution, niche property and life form explain plants’ vulnerability to global change? Global Change Biology 12, 1079–1093 (2006).
Baguette, M., Blanchet, S., Legrand, D., Stevens, V. M. & Turlure, C. Individual dispersal, landscape connectivity and ecological networks. Biological Reviews 88, 310–326 (2013).
Carr, J., Hughes, A. & Foden, W. A climate change vulnerability assessment of West African species. UNEP-WCMC Technical Report, Cambridge, UK (2014).
Acknowledgements
We thank Daniel Attuquayefio, David Nipperess, Manuel Esperón-Rodríguez, Mohasin Haque, Anindita Roy, Sabira Sultana and Joseph Maina for their insightful comments on the manuscript.
Author information
Authors and Affiliations
Contributions
B.Y.O., L.J.B. and A.J.S. designed study and B.Y.O. performed research. B.Y.O. and J.B.B. analysed data. J.B.B. prepared figures. B.Y.O. wrote main manuscript text with contributions from L.J.B., A.J.S., and J.B.B. All authors reviewed manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ofori, B.Y., Stow, A.J., Baumgartner, J.B. et al. Influence of adaptive capacity on the outcome of climate change vulnerability assessment. Sci Rep 7, 12979 (2017). https://doi.org/10.1038/s41598-017-13245-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-13245-y
This article is cited by
-
Flooded lives: socio-economic implications and adaptation challenges for riverine communities in Bangladesh
International Journal of Environmental Science and Technology (2024)
-
Climate Change Vulnerability Assessment for the Rondo Dwarf Galago in Coastal Forests, Tanzania
Environmental Management (2023)
-
The costs of living in the city: influence of urbanization on the trophic niche and body condition of the African rainbow lizard Agama picticauda
European Journal of Wildlife Research (2023)
-
Global warming leads to larger bats with a faster life history pace in the long-lived Bechstein’s bat (Myotis bechsteinii)
Communications Biology (2022)
-
Sustainability initiative for a Malaysian university campus: living laboratories and the reduction of greenhouse gas emissions
Environment, Development and Sustainability (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.