Abstract
Two distinct forms of the erythropoietin receptor (EPOR) mediate the cellular responses to erythropoietin (EPO) in different tissues. EPOR homodimers signal to promote the maturation of erythroid progenitor cells. In other cell types, including immune cells, EPOR and the ß-common receptor (CD131) form heteromers (the innate repair receptor; IRR), and exert tissue protective effects. We used dextran sulphate sodium (DSS) to induce colitis in C57BL/6 N mice. Once colitis was established, mice were treated with solvent, EPO or the selective IRR agonist cibinetide. We found that both cibinetide and EPO ameliorated the clinical course of experimental colitis in mice, resulting in improved weight gain and survival. Correspondingly, DSS-exposed mice treated with cibinetide or EPO displayed preserved tissue integrity due to reduced infiltration of myeloid cells and diminished production of pro-inflammatory disease mediators including cytokines, chemokines and nitric oxide synthase-2. Experiments using LPS-activated primary macrophages revealed that the anti-inflammatory effects of cibinetide were dependent on CD131 and JAK2 functionality and were mediated via inhibition of NF-κB subunit p65 activity. Cibinetide activation of the IRR exerts potent anti-inflammatory effects, especially within the myeloid population, reduces disease activity and mortality in mice. Cibinetide thus holds promise as novel disease-modifying therapeutic of inflammatory bowel disease.
Similar content being viewed by others
Introduction
Erythropoietin (EPO) is a type I cytokine that fulfills dual functions. In its classical endocrine role, EPO produced by the kidney acts as the key regulator of erythropoiesis via inhibition of erythroid progenitor cell apoptosis and stimulation of differentiation1,2. Additionally, locally-produced EPO mediates extra-erythropoietic activity in non-hematopoietic cells and tissues3. Accordingly, basic research has characterized EPO as a tissue-protective and anti-apoptotic cytokine in animal models of ischemia, ischemia-reperfusion injury or mechanical trauma in various tissues including the nervous system, retina, myocardium, lung, kidney, liver or transplanted pancreatic islets4,5,6,7,8,9,10,11,12. Moreover, it has been shown that EPO ameliorates disease activity in experimental arthritis, encephalomyelitis and colitis in mice, suggesting that EPO exerts anti-inflammatory and tissue protective effects in autoimmune diseases13,14,15,16.
These pleiotropic effects of EPO are transduced by two different receptor isoforms3. The erythropoietic response to EPO is initiated via a receptor formed by two identical EPO receptor (EPOR) subunits, which mediate Janus kinase-2 (JAK2)-dependent activation of signal transducer and activator of transcription-5 (STAT5)17. STAT5 subsequently translocates to the nucleus to regulate hemoglobin (Hb) synthesis, control cell cycle progression and inhibit the apoptosis of erythroid progenitors18,19. In parallel to the STAT5 pathway, EPOR homodimers activate mitogen-activated kinases (MAPKs) and nuclear factor (NF)-κB20. In contrast, the anti-apoptotic and anti-inflammatory effects of EPO are transduced via an alternative receptor composed of subunits EPOR and CD131, the beta common receptor shared by granulocyte macrophage (GM)-CSF, interleukin (IL)−3 and IL-521, also termed the innate repair receptor (IRR). Activation of the IRR is paracrine in nature, requiring high local concentrations of EPO, yet facilitates similar signal transduction events including STAT5 and MAPK activation. The effects of IRR activation on NF-kB function are diverse and may be dependent on the experimental model or cell type studied. For example, EPO protected cultured cerebrocortical neurons form excytotoxic and nitrosative stress through activation of JAK2 and NF-κB, resulting in increased expression of anti-apoptotic gene products4. In contrast, in vivo models of myocardial and hepatic ischemia/reperfusion injury have shown that EPO treatment protected from tissue damage via inhibition of NF-κB activation10,22. Similarly, sensitization to chemotherapeutics by EPO in vitro involved the inhibition of both JAK2 and NF-κB23.
Cibinetide (also termed ARA290 or pyroglutamate helix B surface peptide (pHBSP)), is a synthetic oligo-peptide rationally designed to selectively activate the IRR24. Cibinetide interacts with the external hydrophilic domain of the EPOR/CD131 complex and has been shown to exert benefical effects in animal models of myocardial infarction, dilated cardiomyopathy, experimental ischemic stroke, peripheral nerve trauma and wound healing. Furthermore, randomized placebo controlled trials have demonstrated its safety and efficacy in human subjects affected by peripheral neuropathy secondary to sarcoidosis or diabetes mellitus25,26,27. Cibinetide improves symptoms in patients with sarcoidosis-associated small nerve fiber loss and increases corneal small nerve fiber abundance.
Cibinetide lacks erythropoietic effects as it does not activate EPOR homodimers. Correspondingly, unchanged Hb levels have been reported following repeated applications of cibinetide in patients. Thus, cibinetide is not associated with unintended side effects such as thromboembolism secondary to stimulation of erythropoiesis as ascribed for full length recombinant erythropoietin.
The purpose of this study was to evaluate the therapeutic potential of cibinetide in the model of experimental DSS-colitis and to investigate possible mechanisms that mediate its immuno-modulatory effects. We report that cibinetide had multiform anti-inflammatory properties in inflamed tissues. While cibinetide did not affect erythropoiesis, it reduced the influx of myeloid cells into the lamina propria and inhibited the local production and systemic availability of TNF, IL-6, IL12/IL-23, myeloid derived chemokines and nitric oxide (NO) in mice suffering from DSS colitis. Mechanistically, these effects of cibinetide were transduced via CD131 and JAK2 signaling which resulted in impaired activation of the NF-κB subunit p65 thereby blocking transcriptional activation of NF-κB driven inflammatory mediators. Consequently, cibinetide exerted beneficial effects in DSS-colitis by improving disease activity and survival, recommending cibinetide as a promising therapy for human inflammatory bowel disease.
Results
Cibinetide ameliorates the clinical course of dextran sulphate sodium-induced colitis
Full length EPO has anti-inflammatory effects in a model of inflammatory bowel disease, namely DSS-colitis, that are mediated via NF-κB de-activation16. We thus questioned whether this effect depended on IRR activation by determining whether the specific IRR agonist cibinetide improved DSS-colitis, where pathophysiology and tissue damage are centrally driven by NF-κB regulated pathways. C57BL/6N mice were therefore administered 3% DSS via the oral route for 7 days (days 1 to 7). From day 8 to 14, mice had ad libitum access to drinking water and were injected intraperitoneally daily with either EPO, cibinetide or an equal volume of PBS (solvent).
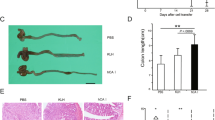
In comparison to DSS-exposed mice treated with solvent, animals receiving either EPO or cibinetide presented with a significantly improved weight gain (Fig. 1A). Expectedly, EPO-treated animals exhibited increased hemoglobin (Hb) levels (Fig. 1B), whereas cibinetide treatment did not result in such erythropoietic activity in vivo or in vitro. Specifically, EPO but not cibinetide stimulated the differentiation of murine erythropoietic progenitors (for gating strategy see Supplementary Fig. S1A) in the bone marrow in vivo (Supplementary Fig. S1B–S1G) and increased Hb protein and CD71 mRNA levels in human mononuclear cell cultures (Supplementary Fig. S1H and S1I). Cibinetide prevented the occurrence of anemia in DSS-exposed mice (Fig. 1B), possibly by reducing intestinal bleeding as evident from reduced fecal Hb content (Fig. 1C). Importantly, lethality was also significantly lower in the EPO and cibinetide treatment groups, respectively (Fig. 1D). Correspondingly, histopathologic changes as quantified by a colitis score28 and immune cell tissue invasion/mucosal thickening were significantly and similarly reduced in colitis mice treated with EPO or cibinetide (Fig. 1E and F–H).
Cibinetide and EPO treatment improve disease activity in DSS-induced colitis. (A) C57BL/6N mice received drinking water or 3% DSS from days 1 to 7. Through days 8 to 14, all animals received drinking water. Starting on day 8, mice were daily injected with PBS (Ctrl.; given as solvent), 1.5 pmol/g body weight rhu erythropoietin (EPO) or 1.5 pmol/g body weight cibinetide (CIB) as indicated by arrows. The change in body weight is expressed as percentage of body weight from day 0, and data are shown as means ± S.E.M. for 5–12 mice per group. Results were compared by ANOVA using Bonferroni correction. Statistical significant differences between DSS-mice treated with EPO or CIB in comparison to PBS treatment are indicated next to the corresponding symbol. (B) Hemoglobin (Hb) levels of the animals presented in (a) were measured on day 14 by an automated veterinary cell counter. Data were compared by Kruskal-Wallis testing. Data are shown as means ± S.E.M. n = 5–12 mice per group. (C) Fecal Hb levels were measured on day 14 and normalized for dry weight. Data were compared by Kruskal-Wallis testing. Data are shown as means ± S.E.M. n = 5–9 mice per group. (D) Survival of DSS-exposed mice treated with PBS, CIB, or EPO was followed for 14 days. Data were compared by log-rank test and are depicted as Kaplan-Meier plot. n = 3 per group for mice on drinking water, n = 14–24 per group for mice on DSS. (E) Histopathological colitis scores of mice receiving drinking water or 3% DSS and subsequently treated with PBS, CIB, or EPO (n = 5–12 per group). Each point represents an individual mouse, horizontal bars represent medians. Statistically significant differences between treatment groups as determined by ANOVA using Bonferroni correction are indicated. n = 5–12 mice per group. (F–H) Photomicrographs of HE-stained colonic sections showed mucosal thickening, epithelial hyperplasia and inflammation in PBS-treated DSS-mice (F) and nearly unaffected mucosa observed in EPO- (G) or CIB-treated DSS-mice (H). Colon histology is shown, inserts are indicated. Scale bars: 100 μm.
Cibinetide inhibits immune cell influx into the colonic lamina propria
Flow cytometric characterization of immune cells (for gating strategies see Supplementary Fig. S2A, S2B and S3A) infiltrating the lamina propria in response to DSS showed that treatment with EPO or cibinetide resulted in reduced accumulation of neutrophils, monocytes and eosinophils in the colon (Fig. 2A–C). In addition, we noted a significant reduction in CD4+ T cell numbers expressing either IFN-γ or IL-17A in the colon of mice having received EPO or cibinetide as compared to control treated littermates (Fig. 2E and F). A substantial reduction of TNF positivity was found in myeloid cells, especially of Ly-6C+ monocytes and CD11b+ CD11c−macrophages (Fig. 2G and H) but not in the CD4+ T cell population (Fig. 2D). In addition, either population of myeloid cells displayed reduced intracellular Nos2 staining in the EPO and cibinetide treatment groups (Supplementary Fig. S2C and S2D). No change was observed in intralesional numbers of CD11c+ DC (Supplementary Fig. S2E), mast cells (Supplementary Fig. S2F), IL-10+ CD4+ T, CD8+ T or B cells (Supplementary Fig. S3B, S3C and S3D, respectively).
Cibinetide (CIB) or EPO treatment reduces immune cell infiltration into lamina propria. Leukocyte populations in the colonic lamina propria were analyzed on day 14 by flow cytometry. To assess cytokine expression by flow cytometry, single cell suspensions were re-stimulated with PMA and ionomycin in the presence of Golgi blockage. Cells were permeabilized for intracellular staining. Gating strategies are detailed in Supplementary Fig. S2A, S2B and S3A. Neutrophils (A), monocytes (B), eosinophils (C), TNF+ CD4+ T cells (D), IFN-γ+ CD4+ T cells (E), IL-17A+ CD4+ T cells (F), TNF+ monocytes (G) and TNF+ CD11b+ CD11c− macrophages (H) were quantified. Numbers represent cell abundance relative to PBS-injected controls (Ctrl.) kept on drinking water. Data were compared by Kruskal-Wallis testing. n = 5–12 mice per group.
Lamina propria macrophages express the innate repair receptor and respond to cibinetide
Next, we FAC-sorted CD11b+ CD11c− lamina propria macrophages and found constitutive expression of Epor mRNA (Fig. 3A). CD131 mRNA expression was increased by DSS-induced inflammation, while treatment with EPO or cibinetide inhibited this effect (Fig. 3B). In addition, we observed reduced p65 binding activity in sorted CD11b+ CD11c− lamina propria macrophages from DSS-exposed mice treated with EPO or cibinetide as compared to cells from solvent treated littermates (Fig. 3C).
EPO and cibinetide counteract NF-κB p65 activation in lamina propria macrophages. CD11b+ CD11c− macrophages were FAC-sorted from the colonic lamina propria on d14 of DSS exposure to prepare total RNA and nuclear proteins. Epor (A) and CD131 (B) mRNA expression was assessed by qPCR. Activation of p65 (C) was measured by an ELISA-based assay. Data were compared by Kruskal-Wallis testing. n = 5–12 mice per group.
Cibinetide acts on myeloid cells and inhibits the production of soluble disease mediators
In line with these results, FAC-sorting of macrophages, monocytes, dendritic cells and CD4+ T cells from the lamina propria of DSS-exposed mice revealed that EPO and cibinetide both substantially reduced Ccl2, Ccl3, Ccl11, TNF, IL-1ß and IL-6 production by myeloid cells, but had only little effect on respective cytokine/chemokine expressions by CD4+ T cells (Fig. 4A–F). The observed clinical and histopathological improvement of colitis upon EPO or cibinetide treatment was also associated with reduced levels of nitrite, TNF, IL-1ß, IL-6, IL-12p70, IL-23, IFN-γ and IL-17A in supernatants of colonic organ cultures (Supplementary Fig. S4A–S4E). Corresponding results were found upon determination of cytokine and chemokine mRNA levels in colonic tissue samples from control and DSS mice demonstrating an inhibitory effect of EPO or cibinetide on the expression of several pro-inflammatory immune mediators (Supplementary Table 1).
EPO and cibinetide inhibit chemokine and cytokine production by myeloid cells. The indicated immune cell populations were FAC-sorted from the lamina propria of DSS-exposed mice and cells were cultured for 24 hrs to allow for ex vivo mediator secretion. Concentrations of Ccl2 (A), Ccl3 (B), Ccl11 = Eotaxin; depicted in (C), TNF (D), IL-1ß (E) and IL-6 (F) were measured by specific ELISA kits. Data are from 3 to 4 individual mice and were compared using the unpaired student’s t-test. *p < 0.05, **p < 0.01, ***p < 0.001 as compared to DSS/PBS stimulation. n.d. denotes not detectable.
CD131 is necessary for macrophages to respond to cibinetide
We next generated BMDMs from WT and CD131 −/− mice and treated cells with EPO or cibinetide followed by stimulation with the TLR4 ligand LPS. We found that both, EPO and cibinetide, significantly reduced concentrations of Ccl2, TNF, IL-1ß and IL-6 in culture supernatants of LPS-stimulated WT BMDMs, but both compounds failed to affect cytokine concentrations in the absence of CD131 (Fig. 5A–D).
Cibinetide inhibits pro-inflammatory immune responses in BMDMs in vitro via CD131. BMDMs differentiated from CD131 −/− mice or C57BL/6 N mice were pre-treated with PBS as solvent (Ctrl.), 2 pmol/mL rhu erythropoietin (EPO), 2 pmol/mL cibinetide (CIB) or 50 µg/ml dexamethasone (DEX) 30 min before the addition of LPS (200 ng/mL) or solvent. Supernatants were analyzed for concentrations of cytokines after 24 hours. Data from 4 independent experiments were compared by means of ANOVA using Bonferroni correction for multiple testing. Values are depicted as means ± S.E.M. and statistical significances between PBS and EPO, cibinetide or dexamethasone treatment, respectively, are indicated. Ccl2 (A), TNF (B), IL-1ß (C) and IL-6 (D) levels in supernatants are depicted. IL-1ß and IL-6 levels of solvent-treated control macrophages remained below the reported detection limits of the corresponding ELISA kits. *p < 0.05 as compared to LPS/solvent stimulation in the respective genotype group; n.d. denotes not detectable.
Cibinetide acts on JAK2 and PI3 kinase and inhibits p65 activation
Moreover, pre-treatment of LPS-stimulated cells with EPO or cibinetide caused a significant reduction of p65 DNA binding activity in a time-dependent manner (Fig. 6A), which translated into reduced activation of the Nos2 promoter, because NF-κB is a central activator of Nos2 transcription. Notably, the effects of EPO and cibinetide were diminished in cells transfected with Nos2 promoter constructs carrying site-specific mutations in the NF-κB consensus sequence (Fig. 6B). The inhibitory effects of EPO and cibinetide on p65 activation were not limited to the TLR4 ligand LPS, but were also observed in BMDMs treated with PMA and ionomycin (data not shown) or in cells co-stimulated with LPS and TNF, IL-1ß, IFN-γ or IL-17A, respectively (Fig. 6C).
Cibinetide induces JAK2 phosphorylation and inhibits NF-κB p65 activation in BMDMs in vitro. (A) BMDMs were treated with PBS, 2 pmol/mL EPO, 2 pmol/mL cibinetide (CIB) and 200 ng/mL LPS. NF-κB p65 DNA binding activity in nuclear extracts was evaluated using a chemi-luminescent transcription factor assay at the indicated time points. Data of 3 independent experiments are expressed as arbitrary light units and shown as means ± S.E.M. and were compared using ANOVA. Statistically significant differences between LPS-stimulated cells pre-treated with either PBS, EPO or cibinetide are indicated. (B) BMDMs were transiently transfected with a full-length murine Nos2 promoter luciferase reporter or with constructs carrying site-specific mutations in one or both NF-κB binding sites (mut-κBI-Nos2-luc and mut-κBII-Nos2-luc or mut-κBI/II-Nos2-luc, respectively). Relative luciferase activity after pre-treatment with PBS, EPO or cibinetide and subsequent stimulation with LPS is shown. Data from 6 independent experiments were compared by ANOVA. (C) BMDMs were pre-treated as above and stimulated with LPS without and with co-stimulation with rmu TNF, IL-1ß, IFN-γ and IL-17A (10 ng/ml each). Nuclear NF-κB p65 binding activity was quantified. Data of 4 independent experiments are expressed as relative (to PBS-treatment) arbitrary light units and shown as means ± S.E.M. and were compared by ANOVA. Statistically significant differences between LPS-stimulated cells pre-treated with either PBS, EPO or cibinetide are indicated. (D,E) Following the addition of solvent, 10 µM WP1066, 200 µM M573108 or 50 µM LY294002, BMDMs were treated with PBS, EPO, cibinetide and LPS. Nuclear NF-κB p65 binding activity was measured. Data of 3 independent experiments are expressed as arbitrary light units and shown as means ± S.E.M. and were compared by means of ANOVA. *p < 0.001 as compared to the addition of LPS/solvent. (F) BMDMs were isolated from Jak2 −/− mice and Jak2 -+/+ littermates. Cells were stimulated with PBS, EPO or cibinetide to verify the presence or absence of Jak2, respectively. (G) In independent experiments, BMDMs were stimulated with PBS, EPO, cibinetide (CIB) or 50 µg/ml dexamethasone (DEX). NF-κB p65 binding activity was quantified. *p < 0.05 as compared to LPS/solvent stimulation in the respective genotype group.
To determine whether the anti-inflammatory effects of EPO and cibinetide are dependent on JAK-STAT-dependent signaling or alternative pathways, we pre-incubated BMDMs with a panel of pharmacological inhibitors prior to the addition of EPO or cibinetide and stimulation with LPS, respectively. Inhibitors of STAT3 (WP1066) or STAT5 (M573108) did not interfere with EPO’s or cibinetide’s ability to de-activate p65 (Fig. 6D). Neither did we see an effect using inhibitors of the MAP kinases MEK or p38 (Data not shown). In contrast, pre-treatment with the PI3 kinase inhibitor LY294002 blunted the effects of EPO or cibinetide on p65 binding activity (Fig. 6E).
To corroborate these results, we generated BMDMs lacking JAK2 by Cre-lox technology (LysMCre-(LMC)-Jak2 −/−). While stimulated LMC-Jak2 +/+-BMDMs responded to EPO and cibinetide with JAK2-phosphorylation, no effect was found in LMC-Jak2 -/-BMDMs (Fig. 6F). Notably, pre-treatment with EPO or cibinetide failed to suppress p65 binding activity in LMC-Jak2 -/-BMDMs (Fig. 6G). In contrast, the glucocorticoid dexamethasone (DEX) did inhibit p65 binding activity independent of the presence or absence of JAK2.
Cibinetide has little effect on cytokine production by T helper cells
As a range of T helper cell cytokines was significantly suppressed in the colon of DSS-exposed mice treated with EPO or cibinetide (Supplementary Table 1), we tested whether EPO or cibinetide may also act on isolated CD4+ T cells stimulated in vitro with PMA/ionomycin or with conditioned media (CM) of BMDMs treated with PMA/ionomycin and PBS (as solvent), EPO or cibinetide. We found a fairly small and non-significant inhibitory effect of EPO and cibinetide on IL-17A mRNA expression in isolated CD4+ cells stimulated with PMA/ionomycin (Supplementary Fig. S5C). In contrast, conditioned media of BMDMs treated with PMA/ionomycin and PBS (as solvent), EPO or cibinetide had differential effects on the mRNA expression of TNF, IFN-γ, IL-17A, IL-4 and IL-6 dependent on whether or not conditioning of BMDMs included EPO or cibinetide (Supplementary Fig. S5A–S5E), further evidence that EPO and cibinetide act primarily on myeloid cells.
Discussion
The results of this study demonstrate that cibinetide exerts broad and potent anti-inflammatory effects on myeloid cells. In vitro, cibinetide significantly reduced the production of several macrophage-derived inflammatory mediators in response to LPS stimulation. In vivo, the proportion of TNF+ monocytes and macrophages in the colonic lamina propria was reduced after treatment of DSS-exposed mice with cibinetide or EPO. The observation that in vivo cibinetide treatment dampens the ex vivo secretion of Ccl2, Ccl3, Cxcl1 and Ccl11 by FAC-sorted monocytes and macrophages further suggests that the recruitment of inflammatory cells to the colonic lamina propria is dampened by activation of the IRR of myeloid cells. Since cibinetide has no effect on erythropoiesis, it appears unlikely that the compound may influence the ratio of erythropoiesis/myelopoiesis. In fact, the proportion of Ter119+ vs. CD11b+ cells in the bone marrow was not affected by cibinetide. Moreover, IL-10 and TGF-ß mRNA levels in the colon were dampened by cibinetide or EPO treatment in the same manner as were pro-inflammatory cytokines and chemokines. We therefore propose a direct anti-inflammatory effect of cibinetide downstream of the IRR. The in vitro results obtained with JAK2 deficient BMDMs and with selective kinase inhibitors support this concept and further suggest that JAK2 and PI3K transduce this anti-inflammatory signal to the nucleus. Although EPO is known to influence several signal transduction cascades, we found that in LPS- or cytokine-stimulated macrophages, EPO and cibinetide primarily targeted the NF-κB subunit p65 activation pathway following JAK2 phosphorylation. Since NF-κB is one of the crucial transcription factors in the initiation and amplification of the inflammatory cascade, cibinetide and EPO both influence a key event of macrophage activation.
Inflammatory mediators such as TNF and IL-1ß auto-regulate their own expression via a positive amplification loop involving NF-κB. Moreover, NF-κB coordinates the expression of a variety of other inflammatory genes, including adhesion molecules and additional transcription factors. NF-κB has further been shown to be centrally involved in the pathophysiology of human inflammatory bowel disease (IBD) and experimental colitis. The increased risk of Crohn’s disease observed in individuals with pathogenic nucleotide-binding oligomerization domain (NOD)−2 polymorphisms is believed to result from increased NF-κB binding activity in the colon and enhanced secretion of IL-1ß29. Furthermore, lamina propria macrophages in IBD patients have increased NF-κB binding activity along with increased production of TNF, IL-1ß and IL-630,31. Experimental treatment strategies which specifically target p65 functions ameliorate disease severity in different models of murine colitis32,33,34,35,36.
Inhibition of NF-κB activation is thus a direct and promising intervention for the treatment of distinct forms of colitis. Cibinetide may have the capacity to beneficially interfere with the autocrine and paracrine stimulation of cytokine production by macrophages and other immune cells within inflamed microenvironments. Of relevance, medications used in the treatment of IBD such as glucocorticoids, sulfasalazine, mesalazine and leflunomide act, at least in part, via inhibition of NF-κB activity37. Consequently, the addition of cibinetide to established medications may help to accomplish dose reductions, thus limiting toxic side effects associated with current therapeutic regimens. Moreover, cibinetide may be especially valuable for patients who are primarily resistant to conventional therapies or experience tachyphylaxis38,39. An additional benefit is that cibinetide lacks side effects that are ascribed to the erythropoiesis inducing activity of EPO, such as hyperviscosity of the blood and an increased risk for thromboembolic events40.
In these studies we have demonstrated profound anti-inflammatory and disease modifying activity of cibinetide in the setting of DSS-induced colitis without evidence of unintended erythropoietic effects. The results of our studies are encouraging to initiate a randomized controlled trial to study the therapeutic potential of cibinetide in human IBD.
Materials and Methods
Compounds
Cibinetide (CIB), also known as ARA 290 or pyroglutamate helix B surface peptide (pHBSP), was obtained from Araim Pharmaceuticals (Tarrytown, NY, USA). Recombinant human (rhu) EPO theta (Eporatio®) was purchased from Ratiopharm (Vienna, Austria). Both compounds were diluted in phosphate-buffer saline (PBS).
Animals
Conditional deletion of Jak2 in lysozyme M positive cells was performed by crossing Jak2f/f mice, kindly provided by K.U. Wagner (Eppley Institute for Research in Cancer and Allied Diseases, University of Nebraska, Omaha, NE), with LysMCre transgenic mice41 and by further breeding to generate Jak2f/f;LysMCre (LMC-Jak2 −/−) animals. Jak2 −/− animals and Jak2 +/+ littermates were used on a mixed C57BL/6N-Sv129 background42,43. CD131 −/− mice were generated and used as described44,45.
Establishment of DSS-Colitis
DSS-colitis was induced in male, pathogen-free, 8–12 weeks old C57BL/6N mice (Charles River Laboratories) following a reported protocol with slight modifications46. Briefly, 3% dextran sulfate sodium (DSS; MW 36,000–50,000; obtained from MP Biomedicals) was added to the drinking water with ad libitum availability to mice. Animals were treated with DSS or water for 7 consecutive days, followed by 7 days of water. Starting on day 8 after the initiation of DSS treatment, mice were daily administered 1.5 pmol/g body weight rhuEPO or cibinetide, while control mice received PBS. All animal experiments were performed according to the guidelines of the Medical University of Innsbruck based on the Austrian Animal Testing Act of 1988. All animal experiment protocols were approved by the ‘MUI Animal ethics committee’ and the Austrian Ministry for Science and Education (approval no. BMWF-66.011/0020-II/3b/2012).
Cell Isolation and Culture
Age and sex matched mice were sacrificed, bone marrow cells were recovered from tibiae and femora and erythrocytes were lysed. BMDMs were initially cultured on non-tissue culture dishes using 100 ng/ml recombinant murine (rmu) M-CSF (Prepotech). BMDMs used in assays were between 6 d and 8 d in culture and were grown overnight on tissue culture 6-well plates prior use.
CD4+ T cells were isolated from spleen by positive selection using a specific MACS system (Miltenyi Biotec) according to the manufacturer’s instructions.
Cells were incubated with 2 pmol/mL rhuEPO or cibinetide diluted in PBS or PBS alone. After another 30 min, cells were stimulated with 200 ng/mL LPS (lipopolysaccharide from Escherichia coli 055:B5), 100 ng/ml PMA (phorbol-12-myristate-13acetate), 500 ng/ml ionomycin (Sigma) and/or 10 ng/mL rmu TNF, IL-1ß, IFN-γ or IL-17A (R&D) while controls were treated with PBS. Thereafter, supernatants were harvested and cells were subjected to RNA or protein preparation. The use of either rhuEPO theta (Eporatio®; Ratiopharm) or of rmuEPO (R&D) yielded very similar results. Thus, rhuEPO was used for subsequent experiments. Where indicated, 100 µM AG490 (JAK2 inhibitor), 10 µM WP1066 (STAT3 inhibitor), 200 µM M573108 (STAT5 inhibitor), 50 µM PD98059 (MEK inhibitor), 500 µM SB203540 (p38 MAPK inhibitor; all obtained from Merck), 50 µg/ml CAPE (NF-κB inhibitor; from Marlingen), 50 µM LY294002 (PI3K inhibitor; Merck) or 50 µg/ml dexamethasone (Sigma) were used. PBS, DMSO (Sigma) or SB202474 (negative control for SB203540; Merck) served as appropriate controls.
Colonic lamina propria cells were isolated from the large intestine as described previously47,48. In brief, the colon was opened longitudinally, cut into small pieces and washed extensively in PBS to remove feces and debris. Following incubation in HBSS containing 5% FCS, 1.5 mg/ml collagenase VIII and 40 µg/ml DNase I (Sigma), suspensions of lamina propria cells were directly stained for flow cytometry or cell sorting. Individual cell populations were sorted as detailed in the Supplementary information section. Purity of isolated cell populations was at least 98% as determined by FACS analysis.
For human mononuclear cell cultures, 1–3 × 105/ml mononuclear cells (MNC) isolated by lymphoprep from peripheral blood of healthy donors were dissolved in IMDM/5% FCS, counted and mixed with methylcellulose (MethocultTM GF+ H4535 without EPO, StemCell Technologies, Germany) and either solvent or increasing concentrations of EPO (0.4 or 1.2 pmol/mL rhuEPO) or CIB (0.4, 1.2 or 12 pmol/mL cibinetide). Cells were cultured for 14 days and BFU-E were identified according to their morphology with an inverted microscope and a 100 mm culture dish marked with the scoring grid. For analysis of hemoglobin content and mRNA expression, colonies were picked according to their morphology, pooled in PBS and frozen for later analyses. All methods were carried out in accordance with the guidelines and regulations of the Medical University Innsbruck. All experimental protocols were approved by the bioethical committee ‘Ethikkommission’ of the Medical University Innsbruck. Written informed consent was obtained from all subjects.
Flow Cytometry
Cell suspensions were stained in PBS supplemented with sterile 1% FBS and 0.5% BSA using the antibodies listed in the Supplementary information section.
RNA extraction and quantitative real-time PCR
Preparation of total RNA and quantification of mRNA expression by quantitative Taqman RT-PCR following reverse transcription was performed exactly as described16. For colon samples, the Dynabeads® mRNA kit was used (Ambion).
Protein extraction and Western blot analysis
Protein extraction and Western blotting were performed exactly as described49. NF-κB p65 binding activity in nuclear extracts was assessed using a chemi-luminescent assay kit according to the manufacturer’s instructions (Pierce).
Transient Transfections
The full-length Nos2 promoter (WT-Nos2-luc), cloned into the pGL2 basic luciferase reporter gene vector, as well as Nos2-luciferase reporter constructs bearing point mutations at the NF-κB consensus sequences of either the proximal region I (mut-κBI-Nos2-luc), the distal region II (mut-κBII-Nos2-luc), or at both sites (mut-κBI/II-Nos-luc) were used50,51. Nos2 promoter activity was determined by the Dual Luciferase system (Promega) according to the manufacturer’s instructions.
Detection of cytokines, reactive species and hemoglobin
Determination of cytokines in culture supernatants was performed with specific ELISA kits (from R&D). Hemoglobin content was measured in a spectrophotometer at a wave length of 660 nm based on the heme-catalyzed oxidation of tetramethylbenzidine by H2O2 using serial dilutions of purified human hemoglobin (Sigma) as standard52. Values were normalized for total protein of bone marrow cells as determined by BCA (Pierce) or for dry weight of fecal content.
Histopathology
Histological examinations of tissues from in vivo infection and colitis experiments, respectively, were performed on formalin-fixed tissue sections stained with hematoxylin and eosin (HE) according to a standard protocol. The degree of colonic inflammation on microscopic sections was graded semi-quantitatively as described28. Histological scoring was performed by an experienced pathologist blinded to study design and sample identity.
Statistical analysis
Statistical analyses were carried out using SPSS using unpaired two-tailed student’s t-tests or by Mann-Whitney U test to assess data where only two groups existed. Analysis of variance combined with Bonferroni correction or Kruskall-Wallis test, as appropriate, was used for all other experiments. Survival was compared by log-rank test. P values less than 0.05 were considered significant.
References
De Maria, R. et al. Apoptotic role of Fas/Fas ligand system in the regulation of erythropoiesis. Blood 93, 796–803 (1999).
Liu, Y. et al. Suppression of Fas-FasL coexpression by erythropoietin mediates erythroblast expansion during the erythropoietic stress response in vivo. Blood 108, 123–133, https://doi.org/10.1182/blood-2005-11-4458 (2006).
Brines, M. & Cerami, A. Erythropoietin-mediated tissue protection: reducing collateral damage from the primary injury response. J Intern Med 264, 405–432, https://doi.org/10.1111/j.1365-2796.2008.02024.x (2008).
Digicaylioglu, M. & Lipton, S. A. Erythropoietin-mediated neuroprotection involves cross-talk between Jak2 and NF-kappaB signalling cascades. Nature 412, 641–647, https://doi.org/10.1038/35088074 (2001).
Toth, C. et al. Local erythropoietin signaling enhances regeneration in peripheral axons. Neuroscience 154, 767–783, https://doi.org/10.1016/j.neuroscience.2008.03.052 (2008).
Junk, A. K. et al. Erythropoietin administration protects retinal neurons from acute ischemia-reperfusion injury. Proc Natl Acad Sci USA 99, 10659–10664, https://doi.org/10.1073/pnas.152321399 (2002).
Sepodes, B. et al. Recombinant human erythropoietin protects the liver from hepatic ischemia-reperfusion injury in the rat. Transpl Int 19, 919–926, https://doi.org/10.1111/j.1432-2277.2006.00366.x (2006).
Parsa, C. J. et al. A novel protective effect of erythropoietin in the infarcted heart. J Clin Invest 112, 999–1007, https://doi.org/10.1172/JCI18200 (2003).
Wu, H. et al. Erythropoietin attenuates ischemia-reperfusion induced lung injury by inhibiting tumor necrosis factor-alpha and matrix metalloproteinase-9 expression. Eur J Pharmacol 602, 406–412, https://doi.org/10.1016/j.ejphar.2008.11.037 (2009).
Liu, X. et al. Mechanism of the cardioprotection of rhEPO pretreatment on suppressing the inflammatory response in ischemia-reperfusion. Life Sci 78, 2255–2264, https://doi.org/10.1016/j.lfs.2005.09.053 (2006).
Chen, J., Connor, K. M., Aderman, C. M. & Smith, L. E. Erythropoietin deficiency decreases vascular stability in mice. J Clin Invest 118, 526–533, https://doi.org/10.1172/JCI33813 (2008).
Watanabe, M. et al. A Nonhematopoietic Erythropoietin Analogue, ARA 290, Inhibits Macrophage Activation and Prevents Damage to Transplanted Islets. Transplantation 100, 554–562, https://doi.org/10.1097/TP.0000000000001026 (2016).
Agnello, D. et al. Erythropoietin exerts an anti-inflammatory effect on the CNS in a model of experimental autoimmune encephalomyelitis. Brain Res 952, 128–134, doi:S0006899302032390 (2002).
Cuzzocrea, S. et al. Erythropoietin reduces the degree of arthritis caused by type II collagen in the mouse. Arthritis Rheum 52, 940–950, https://doi.org/10.1002/art.20875 (2005).
Yuan, R. et al. Erythropoietin: a potent inducer of peripheral immuno/inflammatory modulation in autoimmune EAE. PLoS One 3, e1924, https://doi.org/10.1371/journal.pone.0001924 (2008).
Nairz, M. et al. Erythropoietin contrastingly affects bacterial infection and experimental colitis by inhibiting nuclear factor-kappaB-inducible immune pathways. Immunity 34, 61–74, https://doi.org/10.1016/j.immuni.2011.01.002 (2011).
Bittorf, T. et al. Activation of STAT5 during EPO-directed suppression of apoptosis. Cell Signal 12, 23-30, S0898-6568(99)00063-7 (2000).
Livnah, O. et al. Crystallographic evidence for preformed dimers of erythropoietin receptor before ligand activation. Science 283, 987–990 (1999).
Konstantinopoulos, P. A., Karamouzis, M. V. & Papavassiliou, A. G. Selective modulation of the erythropoietic and tissue-protective effects of erythropoietin: time to reach the full therapeutic potential of erythropoietin. Biochim Biophys Acta 1776, 1–9, https://doi.org/10.1016/j.bbcan.2007.07.002 (2007).
Broxmeyer, H. E. Erythropoietin: multiple targets, actions, and modifying influences for biological and clinical consideration. J Exp Med 210, 205–208, https://doi.org/10.1084/jem.20122760 (2013).
Brines, M. et al. Erythropoietin mediates tissue protection through an erythropoietin and common beta-subunit heteroreceptor. Proc Natl Acad Sci USA 101, 14907–14912, https://doi.org/10.1073/pnas.0406491101 (2004).
Hochhauser, E. et al. Recombinant human erythropoietin attenuates hepatic injury induced by ischemia/reperfusion in an isolated mouse liver model. Apoptosis 13, 77–86, https://doi.org/10.1007/s10495-007-0155-8 (2008).
Carvalho, G. et al. Chemosensitization by erythropoietin through inhibition of the NF-kappaB rescue pathway. Oncogene 24, 737–745, https://doi.org/10.1038/sj.onc.1208205 (2005).
Brines, M. et al. Nonerythropoietic, tissue-protective peptides derived from the tertiary structure of erythropoietin. Proc Natl Acad Sci USA 105, 10925–10930, https://doi.org/10.1073/pnas.0805594105 (2008).
Culver, D. A. et al. Cibinetide Improves Corneal Nerve Fiber Abundance in Patients With Sarcoidosis-Associated Small Nerve Fiber Loss and Neuropathic Pain. Invest Ophthalmol Vis Sci 58, BIO52–BIO60, https://doi.org/10.1167/iovs.16-21291 (2017).
Dahan, A. et al. ARA 290 improves symptoms in patients with sarcoidosis-associated small nerve fiber loss and increases corneal nerve fiber density. Mol Med 19, 334–345, https://doi.org/10.2119/molmed.2013.00122 (2013).
Heij, L. et al. Safety and efficacy of ARA 290 in sarcoidosis patients with symptoms of small fiber neuropathy: a randomized, double-blind pilot study. Mol Med 18, 1430–1436, https://doi.org/10.2119/molmed.2012.00332 (2013).
Dieleman, L. A. et al. Chronic experimental colitis induced by dextran sulphate sodium (DSS) is characterized by Th1 and Th2 cytokines. Clin Exp Immunol 114, 385–391 (1998).
Maeda, S. et al. Nod2 mutation in Crohn’s disease potentiates NF-kappaB activity and IL-1beta processing. Science 307, 734–738, https://doi.org/10.1126/science.1103685 (2005).
Neurath, M. F. et al. Cytokine gene transcription by NF-kappa B family members in patients with inflammatory bowel disease. Ann N Y Acad Sci 859, 149–159 (1998).
Schreiber, S., Nikolaus, S. & Hampe, J. Activation of nuclear factor kappa B inflammatory bowel disease. Gut 42, 477–484 (1998).
Fichtner-Feigl, S., Fuss, I. J., Preiss, J. C., Strober, W. & Kitani, A. Treatment of murine Th1- and Th2-mediated inflammatory bowel disease with NF-kappa B decoy oligonucleotides. J Clin Invest 115, 3057–3071, https://doi.org/10.1172/JCI24792 (2005).
De Vry, C. G. et al. Non-viral delivery of nuclear factor-kappaB decoy ameliorates murine inflammatory bowel disease and restores tissue homeostasis. Gut 56, 524–533, https://doi.org/10.1136/gut.2006.096487 (2007).
Neurath, M. F. et al. Local administration of antisense phosphorothioate oligonucleotides to the p65 subunit of NF-kappa B abrogates established experimental colitis in mice. Nat Med 2, 998–1004 (1996).
Lawrance, I. C. et al. A murine model of chronic inflammation-induced intestinal fibrosis down-regulated by antisense NF-kappa B. Gastroenterology 125, 1750–1761, doi:S0016508503015051 (2003).
Wu, F. & Chakravarti, S. Differential expression of inflammatory and fibrogenic genes and their regulation by NF-kappaB inhibition in a mouse model of chronic colitis. J Immunol 179, 6988–7000, doi:179/10/6988 (2007).
Auphan, N., DiDonato, J. A., Rosette, C., Helmberg, A. & Karin, M. Immunosuppression by glucocorticoids: inhibition of NF-kappa B activity through induction of I kappa B synthesis. Science 270, 286–290 (1995).
Neurath, M. F. New targets for mucosal healing and therapy in inflammatory bowel diseases. Mucosal Immunol 7, 6–19, https://doi.org/10.1038/mi.2013.73 (2014).
Peyrin-Biroulet, L. et al. Selecting Therapeutic Targets in Inflammatory Bowel Disease (STRIDE): Determining Therapeutic Goals for Treat-to-Target. Am J Gastroenterol 110, 1324–1338, https://doi.org/10.1038/ajg.2015.233 (2015).
Tonia, T. et al. Erythropoietin or darbepoetin for patients with cancer. Cochrane Database Syst Rev 12, CD003407, https://doi.org/10.1002/14651858.CD003407 (2012).
Clausen, B. E., Burkhardt, C., Reith, W., Renkawitz, R. & Forster, I. Conditional gene targeting in macrophages and granulocytes using LysMcre mice. Transgenic Res 8, 265–277 (1999).
Asshoff, M. et al. Momelotinib inhibits ACVR1/ALK2, decreases hepcidin production, and ameliorates anemia of chronic disease in rodents. Blood 129, 1823–1830, https://doi.org/10.1182/blood-2016-09-740092 (2017).
Wagner, K. U. et al. Impaired alveologenesis and maintenance of secretory mammary epithelial cells in Jak2 conditional knockout mice. Mol Cell Biol 24, 5510–5520, https://doi.org/10.1128/MCB.24.12.5510-5520.2004 (2004).
Reed, J. A. et al. Distinct changes in pulmonary surfactant homeostasis in common beta-chain- and GM-CSF-deficient mice. Am J Physiol Lung Cell Mol Physiol 278, L1164–1171 (2000).
Weber, G. F. et al. Pleural innate response activator B cells protect against pneumonia via a GM-CSF-IgM axis. J Exp Med 211, 1243–1256, https://doi.org/10.1084/jem.20131471 (2013).
Kaser, A. et al. XBP1 links ER stress to intestinal inflammation and confers genetic risk for human inflammatory bowel disease. Cell 134, 743–756, https://doi.org/10.1016/j.cell.2008.07.021 (2008).
Becker, C. et al. TGF-beta suppresses tumor progression in colon cancer by inhibition of IL-6 trans-signaling. Immunity 21, 491–501, https://doi.org/10.1016/j.immuni.2004.07.020 (2004).
Hirotani, T. et al. The nuclear IkappaB protein IkappaBNS selectively inhibits lipopolysaccharide-induced IL-6 production in macrophages of the colonic lamina propria. J Immunol 174, 3650–3657, doi:174/6/3650 (2005).
Theurl, I. et al. Dysregulated monocyte iron homeostasis and erythropoietin formation in patients with anemia of chronic disease. Blood 107, 4142–4148 (2006).
Nairz, M. et al. Nitric oxide-mediated regulation of ferroportin-1 controls macrophage iron homeostasis and immune function in Salmonella infection. J Exp Med 210, 855–873, https://doi.org/10.1084/jem.20121946 (2012).
Morris, K. R. et al. Role of the NF-kappaB signaling pathway and kappaB cis-regulatory elements on the IRF-1 and iNOS promoter regions in mycobacterial lipoarabinomannan induction of nitric oxide. Infect Immun 71, 1442–1452 (2003).
Welch, C. L. & Young, D. S. Spectrophotometry of occult blood in feces. Clin Chem 29, 2022–2025 (1983).
Acknowledgements
The authors are grateful to Sylvia Berger, Ines Brosch, Sabine Engl, Elisabeth Hoflehner, Yoshiko Iwamoto and Markus Seifert for excellent technical support. We thank Drs. J.A. Whitsett (Cincinnati Children’s Hospital Medical Center, Cincinnati, OH) and K.U.Wagner (Eppley Institute for Research in Cancer and Allied Diseases, University of Nebraska, Omaha, NE) for generously providing breeding pairs of CD131 −/− (Csf2rb –/–) and Jak2f/f mice, respectively. The authors are indebted to Drs. Kristin R. Morris and Edward D. Chan (Department of Medicine, University of Texas-Houston Health Science Center, Houston, TX) for generously providing site-specifically mutated Nos2-luciferase constructs. This work was supported by grants from the Austrian Research Fund (FWF; project TRP-188 to G.W. and the FWF sponsored doctoral programme W-1253 HOROS to G.W.), the ‘Theodor Körner Fonds’ (to M.N.), the intramural funding program of the Medical University Innsbruck for young scientists MUI-START (project 2012032003 to M.N.) and by the ‘Verein zur Förderung von Forschung und Weiterbildung in Infektiologie und Immunologie an der Medizinischen Universität Innsbruck’.
Author information
Authors and Affiliations
Contributions
M.N. received funding, conceived the study, performed experiments, analyzed and interpreted the data and drafted the manuscript. D.H., S.D., T.S., A.S., M.A. and J.E.M. performed experiments and analyzed data. P.L.M. scored histology. D.W., F.K.S., I.T., A.C. and M.B. critically revised the manuscript for important intellectual input. G.W. received funding, conceived and supervised the study.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Nairz, M., Haschka, D., Dichtl, S. et al. Cibinetide dampens innate immune cell functions thus ameliorating the course of experimental colitis. Sci Rep 7, 13012 (2017). https://doi.org/10.1038/s41598-017-13046-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-13046-3
This article is cited by
-
Erythropoietin and its derivatives: from tissue protection to immune regulation
Cell Death & Disease (2020)
-
EPO does not promote interaction between the erythropoietin and beta-common receptors
Scientific Reports (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.