Abstract
Whether opioid use in cancer surgery would promote tumor dissemination in humans is inconclusive. We investigated the effect of intraoperative fentanyl dose on colorectal cancer (CRC) prognosis following resection in this retrospective study. A total of 1679 patients with stage I-III CRC undergoing tumor resection between January 2011 and December 2014 were evaluated through August 2016. Postoperative recurrence-free survival (RFS) and overall survival (OS) were analyzed using Cox regression models. Multivariable Cox regression analysis demonstrated no dose-response association between the amount of fentanyl dose and RFS (adjusted hazard ratio: 1.03, 95% CI: 0.89–1.19) or OS (adjusted hazard ratio: 0.84, 95% CI: 0.64–1.09). Patients were further classified into the high- and low-dose groups by the median of fentanyl dose (3.0 μg·kg−1), and there was no significant difference in RFS or OS between groups, either (adjusted hazard ratio: 0.93, 95% CI: 0.74–1.17 for RFS; 0.79, 95% CI: 0.52–1.19 for OS). We concluded that intraoperative fentanyl consumption has no impact on recurrence-free or overall survival in patients after curative CRC resection.
Similar content being viewed by others
Introduction
Opioids have long been the mainstay of analgesics for cancer-related pain and perioperative acute pain. However, it has been established that opioids inhibit cellular and humoral immune function in humans1, 2. Although there are no data directly implicating opioids in cancer genesis in humans, animal models strongly suggest they may contribute to cancer recurrence in the clinical setting3. This tumor-promoting property was also demonstrated in fentanyl2, 4. Nonopioid analgesia helps to preserve the function of natural killer (NK) cell, the primary defense against cancer cells, and reduces metastatic spread of cancer in animals5.
It has been proposed that opioids exert their effects on tumor growth by activation of the mu-opioid receptor (MOR). Overexpression of the MOR in human non–small cell lung cancer (NSCLC) was suggested to promote tumor growth and progression6. Higher MOR expression and greater opioid requirement are associated with poor oncologic outcomes in patients with metastatic prostate cancer7. MORs have also been demonstrated in the nuclei of human colon cancer cells, and exposure of these cells to morphine increased secretion of urokinase plasminogen activator, a promoter of tumor metastasis8.
Since opioids are common analgesics administered during and after cancer surgery, it is of great importance to evaluate their effects on oncologic outcomes in clinical setting. We conducted this retrospective cohort study in patients following CRC surgery to analyze the associations between intraoperative fentanyl dose and cancer recurrence or overall mortality using multivariable Cox regression models. Dose-response associations between the amount of fentanyl dose and cancer outcomes were further evaluated and major prognostic predictors of CRC outcomes were also considered in the analysis to reduce potentially confounding effects9,10,11.
Methods
Setting and patient selection
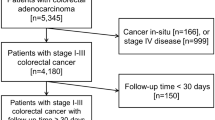
The study was approved by the Institutional Review Board of Taipei Veterans General Hospital, Taipei, Taiwan (IRB-TPEVGH No. 2015-11-010CC). Written informed consent was waived, and all the study materials were anonymized and de-identified before analysis. We utilized the electronic medical database to identify all the patients with CRC undergoing curative resection of primary cancer at the Taipei Veterans General Hospital, Taipei, Taiwan, between January 2011 and December 2014. There was no significant alteration in clinical practice at the centre in the time period of the study. We excluded patients with carcinoma in-situ or stage IV disease determined at the time of surgery, unknown cancer stage, pathology other than adenocarcinoma, re-do surgeries due to recurrence or colostomy, or missing data about demographics or fentanyl dose (Fig. 1).
Anesthetic management
All patients undergoing major abdominal surgery at our hospital were given balanced general anesthesia, typically inducted with fentanyl 1–2 μg·kg−1 and propofol 1–2 mg·kg−1, and neuromuscular antagonism with rocuronium 0.8 mg·kg−1 or cisatracurium 0.2 mg·kg−1. Anesthesia was maintained with sevoflurane 2–3 vol% or desflurane 6–8 vol% in oxygen, with a fraction of inspired oxygen of 40 to 60% at the anesthetist’s discretion.
Fentanyl was the mainstay narcotics used during cancer surgery at this centre. In addition to anesthetic induction, an intravenous bolus of fentanyl 50 μg may be administered before surgical incision if patients did not receive epidural analgesia. If uncontrolled pain was suspected based on hemodynamic changes (e.g. tachycardia and hypertension), the anesthetist may escalate the concentration of volatile anesthetics. Alternatively, single or consecutive intravenous doses of fentanyl would be administered to relieve pain at the anesthetist’s discretion. When an epidural was used, the typical regimen included a loading dose of local anesthetics (lidocaine 1% or 2%) with or without fentanyl 50 μg, followed by an continuous infusion of local anesthetics (bupivacaine 0.25% or 0.5%) with fentanyl 5 μg·ml−1 at a rate of 5–10 ml·hour−1.
Patients would have regular postoperative surveillance at our hospital if they would be eligible for curative-intent surgery. Carcinoembryonic antigen (CEA) was tested every 3 to 6 months for 2 years, then every 6 months for 3 years. Abdomen and chest computed tomography (CT) scans were performed for colon cancer every 6 to 12 months for up to 5 years for patients at high risk of recurrence. For rectal cancer, pelvis CT was added every 3 to 6 months for 2 years, then every 6 to 12 months for up to 5 years for those at high risk of recurrence. Colonoscopy surveillance was conducted at 1 year and repeated 1 to 3 years thereafter.
An electronic medical database was used to determine the baseline characteristics and risk factors for cancer recurrence and mortality, including demographics, pre-treatment CEA level12, perioperative packed red blood cell (pRBC) transfusion13, pathologic findings (tumor differentiation, mucinous or signet-ring histology9, lymphovascular invasion10, and perineural invasion11); whether preoperative or postoperative adjuvant chemotherapy or radiotherapy was used. Current status of each patient was determined by documentation of follow-up visits to the hospital’s outpatient clinic or subsequent admissions. Tumor nodes metastasis (TNM) staging was obtained from the pathologic reports and translated into stages I to III according to the American Joint Committee on Cancer criteria (AJCC-7 staging system)14. Tumor location was divided into right-sided tumor (cecal to splenic flexure) and left-sided colon (splenic flexure to rectum). Adjuvant therapy given in the form of chemotherapy (leucovori and oxaliplatin or fluorouracil, capecitabine, tegafur-uracil for stage II or III disease) or radiotherapy was at the discretion of surgeons and patients. Any adjuvant therapy was defined as given within 90 days of surgery. Cancer recurrence was determined by imaging examinations (CT, magnetic resonance imaging, sonography, bone scan, or plain radiogram) and defined according to response evaluation criteria in solid tumours (RECIST) guidelines15.
Medical records of all the patients included were reviewed and data extracted by research assistants who were not involved in the data analysis. Specialist anesthetists assisted them with data extraction when necessary. The quality of the extracted data was verified through random sampling by the authors. Data were collected up to the end of August 2016.
The primary endpoint was recurrence-free survival (RFS), which was defined as time from the date of surgery to the date of cancer recurrence. The secondary endpoint was overall survival (OS), defined as time from the date of surgery to the date of death. For those without the event of cancer recurrence or death, their survival times are regarded as the corresponding censored observations.
Data analysis and statistics
Patient characteristics, surgical data and pathologic findings were compared between groups using t tests, Mann-Whitney U tests and chi square tests as appropriate. The Kaplan–Meier method was used to calculate the recurrence-free and overall survival of patients from the date of surgery to the date of detection of cancer recurrence and death, respectively; patients without recurrence or alive were censored in the corresponding survival analyses at the last observed day or end of follow-up time (August 31, 2016). Cox proportional hazards regression models were used to evaluate the correlation between the exposure amount of fentanyl and risks of cancer recurrence or overall mortality in the univariate analysis. Multivariable models were applied to adjust other independent risk factors obtained from forward model selection processes with an entry criterion of 0.05. Furthermore, we used the median of fentanyl dose (3.0 μg·kg−1) to define the high-dose (>3.0 μg·kg−1) and low-dose (<3.0 μg·kg−1) groups. Fentanyl dose was regarded as a categorical variable in the univariate and multivariable analyses as well. A two-sided significance level of 0.05 was used to assess statistically significant difference. All the statistical analyses were conducted with IBM SPSS Statistics, Version 23.0 (Armonk, NY: IBM Corp.).
Results
A total of 1679 patients were selected for further analyses after the exclusion processes (Fig. 1). The minimum and maximum fentanyl dosages were 0.2 and 7.8 μg·kg−1, respectively, and the interquartile range of fentanyl dose was from 2.5 to 3.5 μg·kg−1. The median of fentanyl dose administered intraoperatively was 3.0 μg·kg−1 and this number was subsequently utilized to separating patients into the high- and low-dose groups. The differences in mean fentanyl dose between the two groups were 1.24 μg·kg−1 (p < 0.001). The high-dose group was more likely to be female and have epidural blocks, neoadjuvant chemotherapy or radiotherapy, perioperative pRBC transfusion, lower body height and weight and longer follow-up intervals (Table 1).
The association between fentanyl dose and recurrence-free survival
No dose-response relationship was noted between the amount of fentanyl dose and RFS in the univariate analysis. No significant difference in RFS was noted between high- and low-dose groups, either (p = 1.00 by log-rank test, Fig. 2A). Univariate analysis revealed several significant risk factors of cancer recurrence (Table 2), including chronic kidney disease, higher pretreatment CEA level, longer anesthesia time, perioperative pRBC transfusion, advanced cancer stage, specific pathologic findings (poor differentiation, signet-ring histology, lymphovascular invasion, and perineural invasion), preoperative chemotherapy and/or radiotherapy, and postoperative chemotherapy or radiotherapy. After the model selection, eight independent prognostic factors for RFS were identified, including chronic kidney disease (HR = 1.53), preoperative CEA level (on base-10 logarithmic scale, HR = 1.73), cancer stage (II vs. I, HR = 2.78; III vs. I, HR = 6.77), perioperative pRBC transfusion (HR = 1.39), pathologic lymphovascular invasion (HR = 1.37) and perineural invasion (HR = 1.68), preoperative chemotherapy and/or radiotherapy (HR = 2.46), and postoperative radiotherapy (HR = 1.95) (Table 3). After taking these significant predictors into account, the effect of fentanyl dose on RFS after resection surgery was non-significant in the multivariable model, either as a linear (adjusted HR = 1.03, 95% CI = 0.89–1.19) or a categorical variable (adjusted HR = 0.93, 95% CI = 0.74–1.17).
The association between fentanyl dose and overall survival
No dose-dependent association was identified between the amount of fentanyl dose and OS in the univariate analysis. The difference in OS after surgery was not significant between the high- and low-dose groups, either (p = 0.77, Fig. 2B). In the univariate analysis, variables associated with shorter survival were older age, ASA physical status >3, lower body weight, comorbidities (diabetes, ischemic heart disease, heart failure, old stroke, and chronic kidney disease), higher pretreatment CEA level, longer anesthesia time, perioperative pRBC transfusion, advanced cancer stage, specific pathologic findings (poor differentiation, mucinous and signet-ring histology, lymphovascular invasion, and perineural invasion) and postoperative radiotherapy (Table 2). Nine independent prognostic determinants for OS were identified after multivariable analysis (Table 4), including older age (HR = 1.03), lower body weight (HR = 1.02), chronic kidney disease (HR = 1.74), higher pretreatment CEA (on base-10 logarithmic scale, HR = 1.53), longer anesthesia time (on base-2 logarithmic scale, HR = 2.33), perioperative pRBC transfusion (HR = 2.17), cancer stage (II vs. I, HR = 1.87; III vs. I, HR = 3.01), signet-ring histology (HR = 2.41), and pathologic perineural invasion (HR = 1.83). Adjusting for covariates, no association between fantanyl dose and overall mortality after surgery was noted, either as a linear (adjusted HR = 0.84, 95% CI = 0.64–1.09) or a categorical variable (adjusted HR = 0.79, 95% CI = 0.52–1.19).
Discussion
This study showed no definite association between intraoperative fentanyl dose and oncologic outcomes in stage I-III CRC patients after surgical resection. Despite the contradiction to the findings of some prior investigations, our study provided new evidence to reject the hypothetical relationships between intraoperative fentanyl dose and CRC prognosis after surgery with two strengths. We regarded the amount of fentanyl dose as a linear predictor to evaluate the dose-response association between CRC cancer outcomes and intraoperative fentanyl dose. Furthermore, compared with previous studies, we collected comprehensive information about important clinicopathologic predictors (e.g. pre-treatment CEA level and pathologic lymphovascular invasion) to minimize confounding effects of critical prognostic factors.
Intraoperative administration of sufentanil has been reported to be associated with shorter biochemical-free recurrence survival in patients with prostate cancer16. Large opioid requirement and high expression of the MOR were associated with worse progression-free survival and overall survival in patients with metastatic prostate cancer7. Higher opioid consumption during surgery has been reported to be a risk factor for overall survival in patients with stage I but not stage II-III NSCLC17. Maher and colleagues reported an association between increased opioid doses during initial 96-hours postoperative period and higher recurrence rate of NSCLC within 5 years18. However, they found no difference in intraoperative opioid administration among those with or without recurrence of NSCLC at the 5 year follow-up. Similarly, the association between the intraoperative administration of sufentanil and higher rate of recurrence after breast cancer surgery was not significant19. Presumably narcotics played little role in postoperative cancer outcomes but factors increasing narcotic demand (e.g. more aggressive tumor and more extensive surgical resection) affected recurrence and survival after cancer surgery.
Although most clinically used opioids exert their analgesic effects through MOR, different mu opioids may have varying effects on immune function20. Beilin and co-workers showed patients receiving high-dose fentanyl (75–100 µg·kg−1) had more prolonged suppression of NK cell than those receiving low-dose fentanyl (up to 6 µg·kg−1)2. Nevertheless, our result did not suggest the tumour-promoting effect of fentanyl in the perioperative period. Additionally, the effect of morphine on tumor growth is mixed and conflicting in previous literature21. Morphine was demonstrated to suppress cell-mediated immunity in healthy volunteers, including NK cell cytotoxicity1. Morphine at clinically relevant doses increases angiogenesis and promotes breast tumor growth in mice3. However, administration of morphine in rats undergoing laparotomy attenuated a surgery-induced increase in tumor retention22. Tramadol stimulates NK cell activity, both in rodents and humans and can block the enhancement of lung metastasis induced by surgery and prevent the surgery-induced suppression of NK cell activity in a rat model23.
Several retrospective clinical studies revealed the association between the expression of MOR and tumor progression in specific cancer types, including breast, prostate, lung, and esophageal squamous cell carcinoma24, 25. Although there was no report focused on the effect of MOR on cancer outcomes in CRC, a preclinical study suggested the metastasis-promoting potential of opioids in colorectal tumor cells8. It has been proposed mu-opioids stimulate tumor proliferation and invasion through proangiogenic effects3, and angiogenesis is also found to be an important mechanism in tumor growth and invasion in CRC26,27,28,29,30. Our observation that fentanyl dose does not influence recurrence rate contrasts with some previous studies. Apart from the difference in cancer types, the discrepancy may come from the relatively small doses of opioids used in our subjects compared with previous studies. Whether there exists a threshold dose of opioids to produce tumor-promoting effects in human remains to be seen.
We would like to acknowledge the limitations of the present investigation. First, this is a retrospective cohort study and not a randomized controlled trial. Although many important prognostic factors had been considered in this study, it is difficult to evaluate potential effects from unmeasured confounders. Second, the total amount of opioids administered postoperatively was not available for analysis due to the limitations of databank. Third, we did not further assess the effect of non-opioid analgesics that might interact with opioids to alter immune responses which may affect oncologic outcomes after surgery by reason of data availability.
Conclusions
Intraoperative fentanyl consumption has no impact on cancer recurrence or overall survival in patients after curative colorectal cancer resection. Our findings provided new evidence to reject the association between intraoperative fentanyl dose and long-term outcomes after colorectal cancer surgery. Further prospective studies are needed to elucidate the relationship between intraoperative opioid use and cancer prognosis.
References
Yeager, M. P. et al. Morphine inhibits spontaneous and cytokine-enhanced natural killer cell cytotoxicity in volunteers. Anesthesiology 83, 500–508 (1995).
Beilin, B. et al. Effects of anesthesia based on large versus small doses of fentanyl on natural killer cell cytotoxicity in the perioperative period. Anesth Analg 82, 492–497 (1996).
Gupta, K. et al. Morphine stimulates angiogenesis by activating proangiogenic and survival-promoting signaling and promotes breast tumor growth. Cancer Res 62, 4491–4498 (2002).
Shavit, Y., Ben-Eliyahu, S., Zeidel, A. & Beilin, B. Effects of fentanyl on natural killer cell activity and on resistance to tumor metastasis in rats. Dose and timing study. Neuroimmunomodulation 11, 255–260 (2004).
Ben-Eliyahu, S., Shakhar, G., Rosenne, E., Levinson, Y. & Beilin, B. Hypothermia in barbiturate-anesthetized rats suppresses natural killer cell activity and compromises resistance to tumor metastasis: A role for adrenergic mechanisms. Anesthesiology 91, 732–740 (1999).
Lennon, F. E. et al. Overexpression of the μ-opioid receptor in human non-small cell lung cancer promotes Akt and mTOR activation, tumor growth, and metastasis. Anesthesiology 116, 857–867 (2012).
Zylla, D. et al. Opioid requirement, opioid receptor expression, and clinical outcomes in patients with advanced prostate cancer. Cancer 119, 4103–4110 (2013).
Nylund, G. et al. Functional expression of mu-opioid receptors in the human colon cancer cell line, HT-29, and their localization in human colon. Dig Dis Sci 53, 461–466 (2008).
Nitsche, U. et al. Mucinous and signet-ring cell colorectal cancers differ from classical adenocarcinomas in tumor biology and prognosis. Ann Surg 258, 775–782 (2013).
Hogan, J. et al. Lymphovascular invasion: a comprehensive appraisal in colon and rectal adenocarcinoma. Dis Colon Rectum 58, 547–555 (2015).
Liebig, C. et al. Perineural invasion is an independent predictor of outcome in colorectal cancer. J Clin Oncol 27, 5131–5137 (2009).
Thirunavukarasu, P. et al. C-stage in colon cancer: implications of carcinoembryonic antigen biomarker in staging, prognosis, and management. J Natl Cancer Inst 103, 689–697 (2011).
Amato, A. & Pescatori, M. Perioperative blood transfusions for the recurrence of colorectal cancer. Cochrane Database Syst Rev 1, CD005033 (2006).
AJCC Cancer Staging Manual, 7th edition (ed. Edge SB, Byrd SR, Compton CC, Fritz AG, Greene FL, Trotti A.) 143–164 (Springer-Verlag, 2010).
Eisenhauer, E. A. et al. New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur J Cancer 45, 228–247 (2009).
Forget, P. et al. Do intraoperative analgesics influence oncological outcomes after radical prostatectomy for prostate cancer? Eur J Anaesthesiol 28, 830–835 (2011).
Cata, J. P. et al. A retrospective analysis of the effect of intraoperative opioid dose on cancer recurrence after non-small cell lung cancer resection. Cancer Med 3, 900–908 (2014).
Maher, D. P. et al. Association of increased postoperative opioid administration with non-small-cell lung cancer recurrence: a retrospective analysis. Br J Anaesth 113, i88–94 (2014).
Forget, P. et al. Do intraoperative analgesics influence breast cancer recurrence after mastectomy? A retrospective analysis. Anesth Analg 110, 1630–1635 (2010).
Al-Hashimi M., Scott S. W., Thompson J. P. & Lambert D. G. Opioids and immune modulation: more questions than answers. Br J Anaesth 111, 80–88 (2013).
Snyder, G. L. & Greenberg, S. Effect of anaesthetic technique and other perioperative factors on cancer recurrence. Br J Anaesth 105, 106–115 (2010).
Page, G. G., McDonald, J. S. & Ben-Eliyahu, S. Pre-operative versus postoperative administration of morphine: impact on the neuroendocrine, behavioural, and metastatic-enhancing effects of surgery. Br J Anaesth 81, 216–223 (1998).
Gaspani, L., Bianchi, M., Limiroli, E., Panerai, A. E. & Sacerdote, P. The analgesic drug tramadol prevents the effect of surgery on natural killer cell activity and metastatic colonization in rats. J Neuroimmunol 129, 18–24 (2002).
Lennon, F. E., Moss, J. & Singleton, P. A. The μ-opioid receptor in cancer progression: is there a direct effect? Anesthesiology 116, 940–945 (2012).
Singleton, P. A., Moss, J., Karp, D. D., Atkins, J. T. & Janku, F. The mu opioid receptor: A new target for cancer therapy? Cancer 121, 2681–2688 (2015).
Rmali, K. A., Puntis, M. C. & Jiang, W. G. Tumour-associated angiogenesis in human colorectal cancer. Colorectal Dis 9, 3–14 (2007).
Chia, Y. Y., Liu, K., Wang, J. J., Kuo, M. C. & Ho, S. T. Intraoperative high dose fentanyl induces postoperative fentanyl tolerance. Can J Anaesth 46, 872–877 (1999).
Fletcher, D. & Martinez, V. Opioid-induced hyperalgesia in patients after surgery: a systematic review and a meta-analysis. Br J Anaesth 112, 991–1004 (2014).
Zhang, Y. L. et al. Effect of intraoperative high-dose remifentanil on postoperative pain: a prospective, double blind, randomized clinical trial. PLoS One 9, e91454 (2014).
Fechner, J., Ihmsen, H., Schüttler, J. & Jeleazcov, C. The impact of intra-operative sufentanil dosing on post-operative pain, hyperalgesia and morphine consumption after cardiac surgery. Eur J Pain 17, 562–570 (2013).
Acknowledgements
This work was supported by the grants from Taipei Veterans General Hospital (V104C-096), Taipei, Taiwan, R.O.C., Ministry of Science and Technology (MOST104-2314-B-075-015), Taipei, Taiwan, R.O.C. and Anesthesiology Research and Development Foundation (ARDF10401), Taipei, Taiwan, R.O.C.
Author information
Authors and Affiliations
Contributions
Y.H.T.: Data collection and manuscript drafting; H.L.W.: Data collection and verification; W.K.C. and M.Y.T.: Manuscript revision; H.H.C.: Study design and statistical analysis; K.Y.C.: Statistical review, manuscript revision, and final approval of the version to be published. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Tai, YH., Wu, HL., Chang, WK. et al. Intraoperative Fentanyl Consumption Does Not Impact Cancer Recurrence or Overall Survival after Curative Colorectal Cancer Resection. Sci Rep 7, 10816 (2017). https://doi.org/10.1038/s41598-017-11460-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-11460-1
This article is cited by
-
Influence of Perioperative Anesthesia on Cancer Recurrence: from Basic Science to Clinical Practice
Current Oncology Reports (2023)
-
Fentanyl activates ovarian cancer and alleviates chemotherapy-induced toxicity via opioid receptor-dependent activation of EGFR
BMC Anesthesiology (2022)
-
Epidural analgesia does not impact recurrence or mortality in patients after rectal cancer resection
Scientific Reports (2021)
-
Opioids and Cancer Mortality
Current Treatment Options in Oncology (2020)
-
The Effect of Anaesthetic and Analgesic Technique on Oncological Outcomes
Current Anesthesiology Reports (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.