Abstract
Staphylococcus capitis TE8 was isolated from skin surface of a healthy human foot, and exhibited a strong antibacterial activity against Gram-positive bacteria, including Staphylococcus aureus. Whole genome sequence of S. capitis TE8 was obtained by shotgun and paired-end pyrosequencing with a coverage of 109-fold. The draft genome contains 2,516,639 bp in 8 scaffolds with 209 total contigs. The genome contains 2319 protein coding sequences, 58 tRNA and 3 rRNA. Genome sequence analysis revealed 4 distinct gene loci with the ability to encode antimicrobial peptides: (i) an epidermicin gene cluster; (ii) a gallidermin gene cluster; (iii) a gene cluster encoding six phenol soluble modulin (PSM) β-type peptides (PSMβ1-β6) and (iv) an additional gene that belonged to PSMβ family and encoded a 44 residues long peptide, HTP2388. Synthetic peptides with sequence identical to seven PSMβ-like peptides i.e. PSMβ1-β6 and peptide HTP2388 showed antibacterial activity. Genome sequence also revealed genes for adhesins, intracellular adhesins, osmoadaptation, oxidative and acid stress tolerance possibly responsible for initial attachment, colonization and survival of S. capitis TE8 on human skin. Comparative genome analysis revealed presence of a gamut of genes in S. capitis strains in comparison to Staphylococcus epidermidis and Staphylococcus caprae indicating towards their possible role in better adaptation and survival on human skin.
Similar content being viewed by others
Introduction
The skin is the outermost covering of human body and provides protection from numerous invading agents and toxic substances1. Human skin is a host to a large number of microbes, which exhibit distinct topographical niches and significant diversity depending on the skin site2, 3. While a general commensal role vs. a mutually beneficial symbiotic relationship with the host cells is not clear for the microbial populations, several of these microbes are known to protect against colonisation and invasion by pathogenic or harmful organisms in different ways4,5,6. For instance, resident bacteria on the skin may secrete antimicrobial agents and thereby help in maintaining a safe environment on the skin7. Recently, the large diversity of microbial populations in the human microbiome has been found to encode biosynthetic gene clusters that offer a rich potential for bioactive compounds with an ability to modulate host-microbe interactions8, 9. At the same time, the human microbiota offers an enormous resource to look for new antimicrobial agents4.
Microbe-derived natural products have been the classical source of antibiotics and antimicrobial compounds10. The emergence of antimicrobial resistance in bacteria over the last few decades mandates an urgent need to search for new sources of therapeutic agents. The skin microbiota is envisaged to harbour a large repertoire of antimicrobial agents. Staphylococci are common colonizers of human skin; represented as 3rd largest genera out of the 205 genera identified in human skin microbiome using the 16S rRNA gene phylotyping2. S. epidermidis and Staphylococcus hominis, Staphylococcus capitis, Staphylococcus caprae, Staphylococcus auricularis, Staphylococcus warneri, Staphylococcus aureus and Staphylococcus haemolyticus are among the commonly isolated species from human skin11. They are abundant at sebaceous sites and also represented at the moist sites of human skin2. These staphylococci play an important role in skin protection by their ability to reduce the pathogen load on the skin surface and also maintain community structure on the skin surface effectively12, 13. For instance, S. epidermidis has the ability to inhibit S. aureus colonization in nasal cavities by secreting a serine protease, Esp5 while S. lugdunensis impairs S. aureus colonization by the production of a bioactive compound, Lugdunin4.
In this study, we isolated a bacterium Staphylococcus capitis TE8 from the skin surface of a healthy human foot, which exhibited a strong antibacterial activity against Gram positive bacteria, including S. aureus. The genes encoding for antimicrobial peptides were identified using a whole genome sequencing approach. Whole genome sequencing of S. capitis TE8 enabled us to identify 4 distinct gene loci with the ability to encode antimicrobial peptides: (i) an epidermicin gene cluster; (ii) a gallidermin gene cluster; (iii) a gene cluster encoding six phenol soluble modulin (PSM) β-type peptides (PSMβ1-β6) and (iv) an additional gene that belonged to PSMβ family and encoded a 44 residues long peptide HTP2388. We further demonstrate that the antibacterial activity of S. capitis TE8 was associated with all the seven PSMβ-like peptides i.e. six PSMβ peptides and peptide HTP2388. This work adds to the growing understanding that the microbial community in the skin may be a rich source of novel antimicrobial molecules that provide a secure environment both to the microbe from competing microbes and to the human host against skin infections from other pathogens.
Results and Discussion
Identification of human skin isolate TE8 with antimicrobial activity
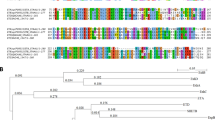
The bacterial isolate TE8 was isolated from the skin surface of the foot of a healthy individual and identified by 16S rRNA gene sequence analysis. The amplified region of the 16S rRNA gene of isolate TE8 showed 99.86% sequence identity with rrs of both S. caprae ATCC 35538 (T) and S. capitis subsp. capitis ATCC 27840 (T) (Fig. 1). In-silico DNA-DNA hybridization (DDH) by genome to genome distance calculator revealed that S. capitis AYP1020, with a score of 88.5, is the closest species followed by 26.4 for S. caprae and 22.5 for S. epidermidis, indicating that isolate TE8 may be a new strain of S. capitis. The strain was hence designated as S. capitis TE8.
S. capitis TE8 showed antibacterial activity against Gram positive organisms Micrococcus luteus, S. aureus and Bacillus subtilis (Fig. 2a) but not against Gram negative strains i.e. Escherichia coli, and Pseudomonas aeruginosa or fungus Candida glabrata (data not shown). The antibacterial activity of the 1-butanol extract was examined against the Gram positive test strains, namely, M. luteus, S. aureus, and B. subtilis (Fig. 2b). M. luteus was found to be relatively more susceptible to the extract of S. capitis TE8, showing a clear and wider zone of inhibition. The extract also exhibited antibacterial activity against B. subtilis and S. aureus. Proteinase K treatment of the extract resulted in complete loss of antimicrobial activity on M. luteus (Fig. 2c), suggesting the antimicrobial agent to be proteinaceous in nature. Various Staphylococcus species have also been shown to produce proteins or peptides that inhibit the growth of S. aureus and other Gram positive organisms, e.g. S. gallinarum produces gallidermin14, S. epidermidis 224 produces epidermicin N10115, S. warneri produces warnerin16 and S. lugdunensis produces Lugdunin4; likewise, S. capitis TE8 may encode protein/peptides with antimicrobial properties.
Antibacterial activity of Staphylococcus capitis TE8. (a) Antibacterial activity of S. capitis TE8 against Bacillus subtilis, Micrococcus luteus and Staphylococcus aureus. (b) Antibacterial activity of crude 1-butanol extracts of isolate TE8 against B. subtilis, M. luteus and S. aureus. (c) Proteinase K treatment effect on 1-butanol extract: 1- Crude extract, 2 - Proteinase K treated extract on M. luteus.
Genome analysis of Staphylococcus capitis TE8
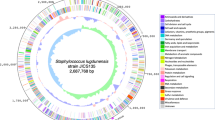
Whole genome sequencing of S. capitis TE8 yielded a total of 1,465,735 reads (105,156 shotgun reads and 1,360,579 paired-end reads). The estimated genome size of S. capitis TE8 is 2.8 Mb with a coverage of 109-fold. The draft genome contains 2,516,639 bp in 8 scaffolds with 209 total contigs. Genome annotation on the NCBI PGAP annotation pipeline identified 2319 protein coding sequences, 58 tRNA and 3 rRNA in the genome (Table 1). The predicted protein coding sequences were annotated and assigned to putative biological functions (Fig. 3). The genome possesses 8 genomic islands (Supplemental Fig. S1) and the genes present in them are included in Supplemental Table S1. Most of the islands harbor genes encoding for hypothetical proteins and one of them appears to be a prophage. Presence of genes encoding for antimicrobial peptides and nutritional stress resistance proteins reflect evolutionary advantage that the predicted GIs might confer on S. capitis TE8.
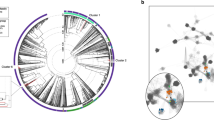
Comparative genome analysis using OrthoMCL revealed clustering of S. capitis TE8 CDSs in 2116 families, out of which 1723 (67.8%) were shared with other three bacteria, S. capitis AYP1020, S. caprae and S. epidermidis (Fig. 4, Supplemental Fig. S2). The remaining 393 orthologous groups were defined the accessory genome, including 57 unique genes present only in S. capitis TE8. Comparison between genomes of these strains using RAST (Supplemental Table S2) revealed that S. capitis TE8 shared a majority of subsystems with S. capitis AYP1020 but also have few distinct differences. S. capitis TE8 contains genes for polyhydroxybutyrate metabolism, inorganic sulphur assimilation and antibiotic & toxic compounds resistance genes, which are absent in S. capitis AYP1020. While genes for some important subsystems were absent in S. capitis TE8 genome but were present in S. capitis AYP1020, such as genes in bacterial check point control, cell division and cell control, lysine biosynthesis, carotenoids, isoprenoid biosynthesis and polyprenyldiphosphate biosynthesis.
Staphylococcus capitis TE8 encodes distinct antimicrobial peptides
The annotated genome of S. capitis TE8 was analysed using different servers viz. AntiSMASH, BAGEL3 and RAST to identify the genes which may encode antibacterial proteins/peptides in the genome. Genome analysis revealed (i) a gene cluster for an epidermicin-type protein, (ii) a gene cluster for gallidermin-type protein, (iii) a PSMβ-type peptide gene cluster of six genes and another (iv) PSMβ-related peptide, HTP2388.
Epidermicin-like antimicrobial peptide
A gene encoding a 51-residue long epidermicin-like protein was identified in the genome of S. capitis TE8 using RAST server and BAGEL3 software. Epidermicins are ribosomally synthesized antibacterial proteins/peptides of type II bacteriocin family. Most significant homology was found with epidermicin N101 (accession number AFD03077) of S. epidermidis 22415 with 96% sequence identity and only two residue variations. Predicted epidermicin of S. capitis TE8 contains glycine and serine at 3rd and 51st positions instead of alanine at corresponding positions for epidermicin N101 of S. epidermidis 224 (Fig. 5a). The genetic organization of the epidermicin cluster of S. capitis TE8 is also similar to that of epidermicin N101 in S. epidermidis 224 with a similar arrangement of neighboring genes (Fig. 5b). It is present on a genomic island of length 6307 bp (Supplemental Table S1). Epidermicins are potent antibacterial agents that display activity against Gram positive pathogens like S. epidermidis, methicillin resistant S. aureus (MRSA) isolates, and vancomycin-resistant enterococci (VRE)15. High sequence similarity and similar gene organization of putative epidermicin from S. capitis TE8 with epidermicin of S. epidermidis 224 indicates that epidermicin of S. capitis TE8 is likely to have similar potent antibacterial activity.
Genetic organization and sequence of putative epidermicin locus of Staphylococcus capitis TE8 compared to epidermicin N101. Epidermicin gene cluster arrangement of S. capitis TE8 and S. epidermidis 224. 1- YbdS like protein, 2- YbdT like protein, 3- epidermicin, 4, 5, 6- hypothetical proteins, 7- RND efflux like protein, 8- ABC transport ATP-binding protein. Accession no JQ025380 - JQ025387 and KJ702950 for epidermicin gene cluster of S. epidermidis 224 and nucleotide sequence (having epidermicin gene cluster) of S. capitis TE8, respectively.
Gallidermin biosynthesis genes cluster
A putative gallidermin-encoding gene in the genome of S. capitis TE8 was identified using antiSMASH server (Fig. 6a). Gallidermins are lanthionine-containing peptide antibiotics having inhibitory activity against propionibacteria14. Gallidermins consist of a leader peptide and an active core region with antimicrobial activity. Previously characterized gallidermin of S. gallinarum was identified as a 52 residue-long peptide with 30 residues associated with leader peptide and 22 residues with the core peptide17. The predicted gallidermin of S. capitis TE8 as identified by antiSMASH server was manually curated to a 44-residue long peptide with ACG as the possible start codon of the gene encoding the gallidermin (Fig. 6b). Blastp search of this putative gallidermin-like protein shows homology with a hypothetical protein (98% identity; WP_016898795) encoded by S. capitis, a gallidermin-type hypothetical peptide (81% identity; WP_002469996) encoded by S. capitis, gallidermin (63% identity; P21838) of S. gallinarum 17 and epidermin (59% identity; P08136) of S. epidermidis 18, suggesting similar antimicrobial functions for the S. capitis TE8 encoded peptide.
Genetic organization and sequence alignment of putative gallidermin locus of Staphylococcus capitis TE8. (a) Putative gallidermin gene cluster arrangement of S. capitis TE8 along with S. capitis AYP1020 and S. gallinarum. 1- LanC cyclase, 2- LanB dehydratase, 3- gallidermin. (b) Sequence alignment of gallidermin protein of S. capitis TE8, S. capitis AYP1020 and S. gallinarum. Arrow indicates the start site predicted by antiSMASH.
Cluster of PSMβ peptides (PSMβ1-β6) and peptide HTP2388
A gene cluster encoding six PSMβ-type peptides (PSMβ1-β6) was identified in S. capitis TE8 using RAST server. The number of PSMβ peptides encoded in the genomes of closely related staphylococci usually varies from two to six (Fig. 7). All the six predicted PSMβ peptides are closely related to each other with identity ranging from 55% to 93% (Supplemental Table S3) and contain the conserved domain of staph_haemo superfamilies.
RAST server identified another gene encoding a hypothetical peptide, HTP2388 (AIK23238) with a conserved domain of staph_haemo superfamilies. HTP2388 shows 27% identity to PSMβ1 and low similarity to other identified PSMβ peptides encoded in the S. capitis TE8 genome. A BLAST search for similar peptides in other bacterial genomes identified several hits suggesting that the peptide is conserved across several staphylococci genomes. The sequence of HTP2388 was found to have 100% sequence identity to several other peptides, namely, hypothetical protein (YP_006939115) of S. epidermidis, a hemolysin-like peptide (WP_002436888) of S. capitis, a hypothetical protein (EEE48298) of S. capitis SK14 and to another hypothetical protein (EES40760) of S. caprae.
The conserved staph_haemo domain found in PSMβ1-β6 and in HTP2388 is present in several small staphylococcal proteins, such as SLUSH proteins, haemolysin, gonococcal growth inhibitors and PSMβ peptides. Phylogenetic analysis of PSMβ and HTP2338 showed the clustering of predicted PSMβ peptides of S. capitis TE8 with the previously reported PSMβ peptides of various Staphylococcus species whereas the peptide HTP2388 clusters with other putative hemolysin-like proteins of staphylococci, confirming it to be a member of a separate family (Fig. 8). All the predicted PSMβ-type peptides of S. capitis TE8 are phylogenetically closer to PSMβ of S. aureus and gonococcal growth inhibitors. Gonococcal growth inhibitors are 44 amino acid peptides which are recently included in PSMβ families on the basis of size and are known to have inhibitory activity against Neisseria gonorrhoeae 19. PSMs have been shown to be involved in Staphylococcal pathogenesis and virulence and play a major role in survival of staphylococci on epithelial surfaces19. PSMβ peptides are devoid of signal peptides and are known to be secreted by PMT transporter in S. aureus 20. A putative PMT transporter was also identified in the genome of S. capitis TE8 consisting of four genes (pmtA, pmtB, pmtC and pmtD), which may be involved in secretion of PSMβ peptides.
Phylogenetic analysis of antibacterial peptides from Staphylococcus capitis TE8 along with other peptides of PSM family. Neighbor-joining phylogenetic tree of PSMβ peptides (PSMβ1-β6) and hypothetical peptide HTP2388 of S. capitis TE8 and PSM of other Staphylococcus spp. Evolutionary analyses were performed using MEGA7.
Synthetic PSMβ1-6 and HTP2388 peptides exhibit antimicrobial activity
The six putative PSMβ peptides and peptide HTP2388 from S. capitis TE8 were synthesized by GL Biochem (Shanghai) Ltd. and tested for possible antimicrobial activity. All the seven synthetic peptides were found to have antibacterial activity against M. luteus with the highest activity in PSMβ6 and least in PSMβ1 (Fig. 9a and b). Further analysis suggested that net charge on the peptides may play a role in the observed antimicrobial activity as also reported earlier21. PSMβ4, PSMβ5, PSMβ6 having neutral net charge showed higher antibacterial activity in comparison to the negatively charged peptides (PSMβ1, PSMβ2, PSMβ3). PSMβ1/β2 of S. aureus with net negative charge are also reported to have no significant antibacterial activity22. Sequence comparison of PSMβ1-6 of S. capitis TE8 with PSMβ1/β2 of S. aureus and gonococcal growth inhibitor peptide sequences of S. aureus indicated the importance of conserved residues to maintain the amphipathic nature of the peptides and in imparting antimicrobial activity (Fig. 9c and Supplemental Fig. S3). A lysine at 3rd and a tryptophan at 20th residue position were found to be conserved across gonococcal growth inhibitors as well as in PSMβ peptides of S. capitis TE8, which have been demonstrated to have antibacterial activity. These residues were found to be replaced by glycine in both PSMβ1/β2 of S. aureus, which do not possess antibacterial activity22. It is tempting to associate the role of lysine at 3rd and/or tryptophan at 20th position in providing antibacterial activity of PSMβ family peptides but would require further validation on a larger set of PSMβ family peptides from different organisms.
Antibacterial activity of synthesized peptides PSMβ1-β6 and HTP2388. (a) and (b) Antibacterial activity of synthesized peptides. Peptides were dissolved in DMSO: water (1:1) and agar well diffusion assay were done against Micrococcus luteus test plate. Test plates were grown overnight and zones of inhibition were measured. (c) Sequence alignment of PSMβ1-β6 of Staphylococcus capitis TE8 along with gonococcal growth inhibitors from Staphylococcus haemolyticus [GGI I (P11697.1), GGI II (P11698.1), GGI III (P11699.1)], PSMβ1 (ABD22369.1) and PSMβ2 (ABD20853.1) of Staphylococcus aureus USA300_FPR3757. Net charge on peptides was written in the parentheses.
Genomic insights into adaptation of Staphylococcus capitis TE8 on skin
S. capitis TE8 was isolated from the human skin and expected to harbour genes required for its invasion, adhesion and virulence. Detailed sequence analysis of S. capitis TE8 genome identified several putative proteins required for adherence, invasion, virulence, biofilm formation, osmotolerance and acid tolerance (Supplemental Table S2). This gamut of genes is expected to play important role in adherence and colonization of strain TE8 on skin. Strain TE8 possess 14 adhesins including 7 cell wall anchored proteins with LPXTG motif, a virulence associated cell wall anchored protein, 4 bifunctional autolysins, a SdrD-like adhesion and an elastin binding protein. S. capitis TE8 genome also contains genes for intercellular adhesions (icaADBC) and the regulatory gene icaR. The genes in ica locus are found to be important for biofilm formation in S. aureus and S. epidermidis 23. Biofilm formation in S. epidermidis is known as a beneficial mechanism in order to get resistance against various antibiotics and protection from host defence system24. Development of biofilm formation begins with primary attachment of bacteria to surface and human matrix proteins. Microbial surface components recognizing adhesive matrix molecules (MSCRAMMs) are required for initial attachment of S. aureus and S. epidermidis to human matrix proteins25, 26. A bifunctional adhesion and autolysin (AtlE) surface protein also has been shown to play important roles in adhesion to abiotic surface27. The 14 putative adhesins (including 4 bifunctional autolysins) along with intracellular adhesins may be responsible for initial attachment and colonization of S. capitis TE8 on human skin.
Apart from possessing double the number of adhesins, S. capitis strains also possess genes for aureusimine (Supplemental Table S4) in comparison to S. caprae and S. epidermidis. Genes for cAMP signalling, LysR family regulatory proteins, Grams positive competence and late competence genes, mannitol utilization, twin arginine translocation system and restriction modification systems were present in S. capitis TE8, S. capitis AYP1020 and S. caprae but were absent in S. epidermidis, while genes for lactose and galactose uptake and utilization, butanol biosynthesis and mixed acid fermentation genes were present only in S. epidermidis. Mannitol utilization is shown to be important for protection of S. aureus from human skin antimicrobial fatty acids28; also disruption of twin-arginine translocation pathway in S. aureus resulted in significantly reduced virulence in mouse kidney abscess model29. A putative LysR family transcription factor and a putative cyclic di-GMP synthetase were shown to regulate hemolysin expression in S. aureus 30. Presence of higher number of adhesins, auresusimine biosynthesis, mannitol utilization, LysR family regulatory protein may indicate towards higher adaptability and virulence of S capitis TE8 and S. capitis AYP1020 in comparison to S. epidermidis.
Skin is the first physical barrier to the environment. Skin microbiome is hence likely to be exposed to various stress factors and the members of skin microbiome expected to possess mechanisms of stress tolerance for survival on the skin surface. Strain TE8 genome has 16 genes for osmoadaptation, which include 2 putative OpuC-type osmolyte uptake systems, 2 putative betaine uptake systems, genes for choline uptake and conversion to glycine-betaine and 2 putative low affinity proline transporters. To overcome the oxidative stress, S. capitis TE8 possess 27 genes encoding for oxidative stress tolerance including genes for superoxide dismutase (SOD), catalase, ferroxidase and OsmC. The strain contains a eukaryotic-type low affinity urea transporter and all components for a functional urease. Along with this, the strain also possesses arginine and ornithine degradation pathway, which are expected to help in adaptation to acidic skin environment. The presence of a large gamut of osmotic/salinity stress, oxidative and acid stress protection mechanisms in S. capitis TE8 may be a reflection on adaptation of this strain to human skin.
Conclusion
We have isolated a skin resident bacterium with the ability to produce a repertoire of antimicrobial peptides with activity against Gram positive strains including S. aureus. Comparative genomics revealed features which might be responsible for adaptation and colonization of this strain on human skin.
Methods
Sample isolation and antimicrobial activity assay
For isolation of skin microbiota, consent from human subjects and permission of Institutional Ethical Committee of CSIR-Institute of Genomics and Integrative Biology, New Delhi 110020, India was taken before the sampling. Guidelines of the Institutional Ethical Committee were followed for the work. S. capitis TE8 was isolated from human skin of healthy volunteer using a sterile cotton swab, pre-soaked in a sterile saline solution containing 0.15 M NaCl and 0.1% Tween-20 and grown on LB agar at 37 °C. It was screened for antimicrobial activity using agar overlay method. The antagonistic property of the isolate was checked against B. subtilis, C. glabrata, E. coli, M. luteus, P. aeruginosa and S. aureus. Briefly, the strain was first grown on LB agar plates at 37 °C for 48 h and then an overlay of LB top agar (0.7%) with the above mentioned test strains, pre-grown to A600 of 0.4–0.5, was carried out on the plates. The plates were then incubated at 37 °C overnight, followed by the detection of the antimicrobial activity based on the zone of inhibition.
Bacterial identification and phylogenetic analysis
Isolate TE8 exhibiting antimicrobial activity against the test strains was further grown in Luria Bertani (LB) medium (pH 7.2) in a shaking incubator at 37 °C with 200 rpm for identification and further analysis. This isolate was identified by rrs (16S rRNA) gene sequence analysis. Briefly, rrs gene was amplified from the genomic DNA using universal primers, 27 F (AGAGTTTGATCMTGGCTCAG) and 1492 R (TACGGYTACCTTGTTACGACTT). The amplified product was sequenced and the obtained sequence was used to identify the bacterium and its closely related members using BLAST and EzTaxon31. The sequence of the amplified region of 16S rRNA was submitted to NCBI with an accession number of KJ420579.
Phylogenetic analysis of the 16S rRNA gene sequence of S. capitis TE8 with other rrs gene sequences obtained from BLAST analysis was carried out by the Maximum Likelihood method based on the Le Gascuel, 2008 model in MEGA v.7.032 and the gaps were treated using partial deletion method.
Extraction of antimicrobial agents
Extraction of agents exhibiting antimicrobial activity was done using 1-butanol extraction method22. In brief, isolate TE8 was grown in 100 ml of LB medium at 37 °C for 24 h. The cells were then harvested by centrifugation at 8228 × g for 10 min and the supernatant collected. The supernatant was mixed with 30 ml of 1-butanol (3:1 ratio) and extraction of secreted antimicrobial compounds was carried out by shaking the mixed solutions at room temperature for 2 h. After brief centrifugation 1-butanol phase was collected and dried by vacuum centrifugation. Dried sample was dissolved in 200 μl of sterile water and 50 μl was checked again for antagonistic properties against B. subtilis, M. luteus and S. aureus.
Effect of Proteinase K enzyme on antibacterial activity
To confirm whether the antimicrobial activity is due to a proteinaceous agent or other secondary metabolites, the extract was treated with Proteinase K (Proteinase K:1-butanol extract:: 1:10) at 37 °C for 1 h. The proteinase K treated extract was checked for antibacterial activity against M. luteus using agar well diffusion assay. After overnight incubation, zone of inhibition was measured to monitor the residual antibacterial activity.
Genome sequencing and analysis
To extract the genomic DNA of isolate TE8, the culture was grown in 5 ml LB broth for 14–16 h. The cells were harvested by centrifugation at 9000 g for 10 min and the genomic DNA was isolated using Microbial DNA purification kit (Mo Bio Laboratories Inc) according the manufacturer’s protocol.
The genome sequencing of isolate TE8 was carried out with a combined strategy using whole genome shotgun (Roche GS-Junior) and paired-end pyrosequencing (3 kb) (Roche FLX). De novo assembly of the raw reads was done by Newbler v2.9 assembler software. Assembled contigs were used for genome annotation by NCBI Prokaryotic Genome Annotation pipeline (PGAP) and Rapid Annotation using Subsystems Technology (RAST) server33, which use tRNAscan-SE and GLIMMER2 for prediction of tRNA and protein encoding genes, respectively. The automated annotation was followed by manual curation/inspection. The genomic islands were predicted using IslandViewer4.34 Secondary metabolites were predicted by Antibiotics & Secondary Metabolite Analysis Shell (antiSMASH)35 and bacteriocin were identified by BAGEL336.
The genome and protein sequences of S. capitis subsp. capitis AYP102037 (GCA_001028645.1), S. caprae M23864:W1 (GCA_000160215.1) and S. epidermidis ATCC 1222838 (GCA_000007645.1) were downloaded from NCBI Repository for in-silico DNA to DNA hybridization (DDH) and comparative genomics analysis. Accession numbers and general features of the selected genomes are provided in Supplemental Table S5. In-silico prediction of DDH value was calculated by using Genome-to-Genome Distance Calculator tool 2.139. Grouping of the proteins into orthologs based on their sequence similarity was performed by OrthoMCL40. OrthoMCL uses the reciprocal best BLAST hit approach and makes an adjustment for species distance (normalization) to distinguish orthologs from in-paralogs and the resulting groups are generated from the normalized BLAST scores between proteins using Markov Clustering. The set of orthologous genes for each strain determined by orthoMCL were presented in the form of a Venn diagram using Venny 2.1 (http://bioinfogp.cnb.csic.es/tools/venny/index.html).
Synthesis and activity of antimicrobial peptides
The predicted six PSMβ peptides (PSMβ1-β6) and the hypothetical peptide HTP2388 were commercially synthesised by GL Biochem (Shanghai) Ltd. at purity greater than 95%. Synthesized peptides were dissolved to a concentration of 10 mg/ml in DMSO: water (1:1) and 20 µl of this was checked for antibacterial activity against M. luteus. DMSO:water (1:1) without any peptide was used as a control.
Nucleotide sequence accession numbers
The genome sequence of S. capitis TE8 has been deposited at GenBank under the accession no. JMGB00000000. The gene clusters of epidermicin, gallidermin, six phenol soluble modulin-like peptides and peptide HTP2388 were deposited in GenBank under accession #KJ702950, KJ728983, KJ702949 and KJ867527, respectively.
Ethical statement
The Ethical Committee of the CSIR-Institute of Genomics and Integrative Biology has approved the work and the subjects gave informed consent to the work.
References
Segre, J. A. Epidermal barrier formation and recovery in skin disorders. J. Clin. Invest. 116, 1150–8 (2006).
Grice, E. A. et al. Topographical and temporal diversity of the human skin microbiome. Science 324, 1190–2 (2009).
Costello, E. K. et al. Bacterial Community Variation in Human Body Habitats Across Space and Time. Science (80-.). 326, 1694–1697 (2009).
Zipperer, A. et al. Human commensals producing a novel antibiotic impair pathogen colonization. Nature 535, 511–516 (2016).
Iwase, T. et al. Staphylococcus epidermidis Esp inhibits Staphylococcus aureus biofilm formation and nasal colonization. Nature 465, 346–349 (2010).
Grice, E. A. & Segre, J. A. The skin microbiome. Nat. Rev. Microbiol. 9, 244–253 (2011).
Cogen, A. L. et al. Selective Antimicrobial Action Is Provided by Phenol-Soluble Modulins Derived from Staphylococcus epidermidis, a Normal Resident of the Skin. J. Invest. Dermatol. 130, 192–200 (2010).
Donia, M. S. et al. A Systematic Analysis of Biosynthetic Gene Clusters in the Human Microbiome Reveals a Common Family of Antibiotics. Cell 158, 1402–1414 (2014).
Donia, M. S. & Fischbach, M. A. Small molecules from the human microbiota. Science (80-.). 349, 1254766–1254766 (2015).
Milshteyn, A., Schneider, J. S. & Brady, S. F. Mining the Metabiome: Identifying Novel Natural Products from Microbial Communities. Chem. Biol. 21, 1211–1223 (2014).
Otto, M. Staphylococcus colonization of the skin and antimicrobial peptides. Expert Rev. Dermatol. 5, 183–195 (2010).
Janek, D. et al. High Frequency and Diversity of Antimicrobial Activities Produced by Nasal Staphylococcus Strains against Bacterial Competitors. PLOS Pathog. 12, e1005812 (2016).
Nakatsuji, T. et al. Antimicrobials from human skin commensal bacteria protect against Staphylococcus aureus and are deficient in atopic dermatitis. Sci. Transl. Med. 9 (2017).
Kellner, R. et al. Gallidermin: a new lanthionine-containing polypeptide antibiotic. Eur. J. Biochem. 177, 53–9 (1988).
Sandiford, S. & Upton, M. Identification, characterization, and recombinant expression of epidermicin NI01, a novel unmodified bacteriocin produced by Staphylococcus epidermidis that displays potent activity against Staphylococci. Antimicrob. Agents Chemother. 56, 1539–47 (2012).
Prema, P., Bharathy, S., Palavesam, A., Sivasubramanian, M. & Immanuel, G. Detection, purification and efficacy of warnerin produced by Staphylococcus warneri. World J. Microbiol. Biotechnol. 22, 865–872 (2006).
Schnell, N. et al. Structural gene isolation and prepeptide sequence of gallidermin, a new lanthionine containing antibiotic. FEMS Microbiol. Lett. 49, 263–7 (1989).
Schnell, N. et al. Prepeptide sequence of epidermin, a ribosomally synthesized antibiotic with four sulphide-rings. Nature 333, 276–278 (1988).
Cheung, G. Y. C., Joo, H. S., Chatterjee, S. S. & Otto, M. Phenol-soluble modulins - critical determinants of staphylococcal virulence. FEMS Microbiol. Rev. 38, 698–719 (2014).
Chatterjee, S. S. et al. Essential Staphylococcus aureus toxin export system. Nat. Med. 19, 364–7 (2013).
Jiang, Z. et al. Effects of net charge and the number of positively charged residues on the biological activity of amphipathic α-helical cationic antimicrobial peptides. Biopolymers 90, 369–383 (2008).
Joo, H.-S., Cheung, G. Y. C. & Otto, M. Antimicrobial Activity of Community-associated Methicillin-resistant Staphylococcus aureus Is Caused by Phenol-soluble Modulin Derivatives. J. Biol. Chem. 286, 8933–8940 (2011).
Arciola, C. R., Campoccia, D., Ravaioli, S. & Montanaro, L. Polysaccharide intercellular adhesin in biofilm: structural and regulatory aspects. Front. Cell. Infect. Microbiol. 5, 7 (2015).
Costerton, J. W., Stewart, P. S. & Greenberg, E. P. Bacterial biofilms: a common cause of persistent infections. Science 284, 1318–22 (1999).
Patti, J. M., Allen, B. L., McGavin, M. J. & Hook, M. MSCRAMM-Mediated Adherence of Microorganisms to Host Tissues. Annu. Rev. Microbiol. 48, 585–617 (1994).
Askarian, M. et al. Evaluation of the Patients’ Queue Status at Emergency Department of Nemazee Hospital and How to Decrease It, 2014. Glob. J. Health Sci. 9 (2017).
Heilmann, C., Hussain, M., Peters, G. & Götz, F. Evidence for autolysin-mediated primary attachment of Staphylococcus epidermidis to a polystyrene surface. Mol. Microbiol. 24, 1013–24 (1997).
Kenny, J. G. et al. Mannitol utilisation is required for protection of Staphylococcus aureus from human skin antimicrobial fatty acids. PLoS One 8, e67698 (2013).
Biswas, L. et al. Role of the Twin-Arginine Translocation Pathway in Staphylococcus. J. Bacteriol. 191, 5921–5929 (2009).
Burnside, K. et al. Regulation of Hemolysin Expression and Virulence of Staphylococcus aureus by a Serine/Threonine Kinase and Phosphatase. PLoS One 5, e11071 (2010).
Kim, O.-S. et al. Introducing EzTaxon-e: a prokaryotic 16S rRNA gene sequence database with phylotypes that represent uncultured species. Int. J. Syst. Evol. Microbiol. 62, 716–21 (2012).
Kumar, S., Stecher, G. & Tamura, K. MEGA7: Molecular Evolutionary Genetics Analysis Version 7.0 for Bigger Datasets. Mol. Biol. Evol. msw054, doi:10.1093/molbev/msw054 (2016).
Aziz, R. K. et al. The RAST Server: Rapid Annotations using Subsystems Technology. BMC Genomics 9, 75 (2008).
Bertelli, C. et al. IslandViewer 4: expanded prediction of genomic islands for larger-scale datasets. Nucleic Acids Res. 45, W30–W35 (2017).
Medema, M. H. et al. antiSMASH: rapid identification, annotation and analysis of secondary metabolite biosynthesis gene clusters in bacterial and fungal genome sequences. Nucleic Acids Res. 39, W339–W346 (2011).
van Heel, A. J., de Jong, A., Montalban-Lopez, M., Kok, J. & Kuipers, O. P. BAGEL3: automated identification of genes encoding bacteriocins and (non-)bactericidal posttranslationally modified peptides. Nucleic Acids Res. 41, W448–W453 (2013).
Cameron, D. R. et al. Insights on virulence from the complete genome of Staphylococcus capitis. Front. Microbiol. 6, 980 (2015).
Zhang, Y.-Q. et al. Genome-based analysis of virulence genes in a non-biofilm-forming Staphylococcus epidermidis strain (ATCC 12228). Mol. Microbiol. 49, 1577–93 (2003).
Meier-Kolthoff, J. P., Auch, A. F., Klenk, H.-P. & Göker, M. Genome sequence-based species delimitation with confidence intervals and improved distance functions. BMC Bioinformatics 14, 60 (2013).
Fischer, S. et al. In Current Protocols in Bioinformatics Chapter 6, Unit 6.12.1–19 (John Wiley & Sons, Inc., 2011).
Acknowledgements
This work was funded by Council of Scientific and Industrial Research (CSIR) grant “Towards Understanding of skin Cell Homeostasis (TOUCH), BSC0302 to RS and BT”. We thank Neha Sahu, Sonam Nain and Garima Suneja for help in preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
R.S. conceived and designed the experiments. R.K. isolated and characterized strain and performed experiments. P.K.J. performed the genome sequencing. All authors analyzed the genomic data and helped in writing of manuscript. All authors reviewed the manuscript before submission.
Corresponding authors
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kumar, R., Jangir, P.K., Das, J. et al. Genome Analysis of Staphylococcus capitis TE8 Reveals Repertoire of Antimicrobial Peptides and Adaptation Strategies for Growth on Human Skin. Sci Rep 7, 10447 (2017). https://doi.org/10.1038/s41598-017-11020-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-11020-7
This article is cited by
-
Scalp microbiome of healthy women wearing hijab compared to those not wearing hijab: a cross-sectional study
Scientific Reports (2023)
-
Microbiome in Atopic Dermatitis: Is It All About Staphylococcus aureus?
Current Treatment Options in Allergy (2023)
-
The microbiome-shaping roles of bacteriocins
Nature Reviews Microbiology (2021)
-
Complete genome sequencing of three human clinical isolates of Staphylococcus caprae reveals virulence factors similar to those of S. epidermidis and S. capitis
BMC Genomics (2018)
-
High throughput in situ metagenomic measurement of bacterial replication at ultra-low sequencing coverage
Nature Communications (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.