Abstract
Nucleotide excision repair (NER) plays a vital role in platinum-induced DNA damage during chemotherapy. We hypothesize that regulatory single nucleotide polymorphisms (rSNPs) of the core NER genes modulate clinical outcome of patients with advanced non-small cell lung cancer (NSCLC) treated with platinum-based chemotherapy (PBS). We investigated associations of 25 rSNPs in eight NER genes with progression free survival (PFS) and overall survival (OS) in 710 NSCLC patients. We found that ERCC1 rs3212924 AG/GG and XPC rs2229090 GC/CC genotypes were associated with patients’ PFS (HRadj = 1.21, 95% CI = 1.03–1.43, P adj = 0.021 for ERCC1 and HRadj = 0.80, 95% CI = 0.68–0.94, P adj = 0.007 for XPC), compared with the AA and GG genotypes, respectively. The association of XPC rs2229090 was more apparent in adenocarcinoma than in squamous cell carcinoma patients. Additionally, ERCC4 rs1799798 GA/AA genotypes were associated with poorer OS (HRadj = 1.32, 95% CI = 1.04–1.69, P adj = 0.026), compared with the GG genotype. The expression quantitative trait loci analysis revealed that ERCC1 rs3212924 and XPC rs2229090 might regulate transcription of their genes, which is consistent with their associations with survival. Larger studies are needed to validate our findings with further functional studies to elucidate the mechanisms underlying these observed associations.
Similar content being viewed by others
Introduction
Lung cancer is the leading cause of cancer-related deaths worldwide, accounting for an estimation of 1.59 million deaths in 20121, while China attributed to 529,153 deaths in 20112. Non-small cell lung cancer (NSCLC) patients, representing approximately 85% of the total lung cancer incident cases, are often diagnosed at an advanced stage of IIIB or IV3, 4. Because surgical treatment for these advanced-stage patients is not possible, palliative chemotherapy remains the mainstream therapeutic option. The overall five-year survival for lung cancer has remained less than 15%, and the prognosis for the advanced-stage disease is even poorer, with a median overall survival (OS) of approximately one year5, 6.
Platinum-based regimes are the standard first-line chemotherapy for NSCLC patients, although recent targeted therapies have presented benefits for a small portion of the patients who have activating EGFR mutations or EML-ALK translocations7, 8. However, the efficacy of platinum-based chemotherapy (PBC) varies remarkably among the patients, with an overall response rate of 26~60%9. It is speculated that this may be related to individual variability in repairing DNA damage induced by PBC10, 11. Increasing body of evidence highlights the importance of genetic factors, such as single nucleotide polymorphisms (SNPs), and gene expression in individual response to the treatment, which have an impact on subsequent survival12, particularly for genetic variations in nucleotide excision repair (NER) genes13,14,15,16.
The DNA repair pathways are the safeguard of genomic stability by restoring damaged DNA induced by mutagens (i.e. UV, tobacco or chemicals), of which NER is the major mechanism removing bulky DNA lesions caused by chemicals. NER has been frequently associated with survival in NSCLC patients treated with PBC13. NER functions by repairing platinum-DNA (Plt-DNA) adducts, involving the coordination of 20–30 proteins that replace the bulky adduct DNA segment with a newly synthesized DNA segment using the intact complementary strand as the template17. The hypotheses of NER genes affecting lung cancer prognosis are two-folds, a double edged sword: on one side, suboptimal DNA repair may promote carcinogenesis by weakening mutation-fixation of DNA damage induced by both exogenous and endogenous carcinogens and subsequent development of tumours18 and the other side, efficient DNA repair in the tumour may lead to fast removal of plt-DNA adducts, reducing the efficacy of PBC13, 19.
NER comprises of three main events: recognition of base damage, the bimodal incision of DNA, and excision of DNA fragments17, 20, 21. The specific recognition of substrate sites consists of several key proteins: the initial step involves the XPC-HHRAD23 complex, which recognizes the base damage caused by exogenous carcinogens22. The XPE/DDB1 protein has been studied for its auxiliary role for the recognition of cyclobutane pyrimidine lesions, due to its affinity for UV-damaged DNA23. The XPC/HHRAD23 complex further binds to several other proteins (i.e. XPA, RPA, TFIIH and XPG/ERCC5), in which transcription factor IIH (TFIIH) is a subcomplex of the RNA polymerase II transcription initiation machinery, and XPB/ERCC3 and XPD/ERCC2 are two central DNA helicases that unwind the DNA duplex in the close vicinity of the base damage; XPG and ERCC1-XPF heterodimeric protein are two endonucleases that cut the damaged DNA strand 3′ and 5′ to the site of the base damage, respectively20, 22. These core proteins work in concert to maintain NER function, and hence their respective roles in the NER pathway have been more extensively studied.
In the present study, we undertook a hypothesis-based approach to evaluate the impact of regulatory SNPs (rSNPs) in the core NER genes (ERCC1, ERCC2, ERCC3, ERCC4, ERCC5, RAD23B, XPA, XPC and XPE) on survival of NSCLC patients treated with PBC by analysing a pool of 25 rSNPs in 710 patients with advanced disease stages. All these 25 rSNPs were predicted by bioinformatics tools to be potentially functional in regulating their gene expression (Table 1 and Supplemental S1).
Results
Characteristics of the study population
The present study consisted of 710 patients diagnosed with NSCLC24, who had DNA samples, complete data on demographic, clinical characteristics, progression free survival (PFS) and overall survival (OS). Of all the patients, 508 were males and 202 were females, with a median age at diagnosis of 58 (a range of 23–83) years, and 334 (47%) were never, 41 (5.8%) former, and 335 (47.2%) current smokers. All subjects had an advanced TNM stage (III or IV) cancer, with 478 (67.3%) being adenocarcinoma. For different chemotherapy combinations, 237 (33.4%) received platinum-docetaxel/paclitaxel, whereas 300 (42.3%) received platinum-pemetrexed treatment. Furthermore, 219 (30.8%) and 257 (36.2%) of the patients received palliative radiotherapy and tyrosine-kinase inhibitor (TKI) treatment, respectively. The associations of these demographic characteristics and the known risk factors with NSCLC survival were also described in a previous publication24. The characteristics of demographic and clinical variables are described in Supplemental Table S2.
NER rSNPs and NSCLC survival
The details of the eight (after excluding DDB1/XPE that does not have any rSNPs) core NER genes. We selected 25 rSNPs that are located in a regulatory region in either of the eight genes, and those rSNPs under investigation are shown in Table 1. We then performed the genotyping with DNA samples extracted from the whole blood cells. Call rates of the majority of the SNPs were >95%, except for three rSNPs (rs2607735, rs1007616 and rs7507745), which were then excluded from further analysis. In the univariate analysis without and multivariate analysis with adjustment for clinical variables, three rSNPs (ERCC1 rs3212924, XPC rs2229090 and ERCC4 rs1799798) consistently showed a significant association with either PFS or OS in NSCLC patients (Tables 2). Further subgroups analysis was performed for adenocarcinoma and squamous cell carcinoma patients, as well as by the dominant chemotherapy treatment, for these two histological types.
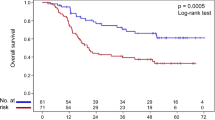
Specifically, the ERCC1 rs3212924 G allele was found to be significantly associated with a poor PFS [AG/GG vs. AA: median survival time (MST) 6.5 vs. 7.6 months, P log-rank = 0.030; adjusted hazards ratio (HRadj) = 1.21, 95% CI = 1.03–1.43, P adj = 0.021, under a dominant model] (Table 2). This variant was not significantly associated with PFS in patients with adenocarcinoma, nor in patients with squamous cell carcinoma alone in our dataset (Supplemental Table S3). The XPC rs2229090 C allele was associated with a longer PFS for all patients (GC/CC vs. GG: MST 7.4 vs. 6.5 months, P log-rank = 0.063; HRadj = 0.80, 95% CI = 0.68–0.94, P adj = 0.007, under a dominant model) (Table 2). In the stratified analysis by histological type and treatment, this variant was significantly associated with a longer PFS only in adenocarcinoma patients alone and squamous cell carcinoma patients who had received docetaxel-cisplatin (GC/CC vs. GG: MST 7.4 vs.6.1 and 10.3 vs. 6.5, P log-rank = 0.109 and 0.030, HRadj = 0.79 and 0.44, 95% CI = 0.65–0.96 and 0.22–0.90, P adj = 0.021 and 0.025, respectively) (Supplemental Tables S3 and S4). Patients carrying ERCC4 rs1799798 GA/AA genotypes showed a significantly increased risk of death, compared with those with the GG genotype (MST 24 vs. 29.3 months, P log-rank = 0.032; HRadj = 1.32, 95% CI = 1.04–1.69, P adj = 0.026, also under a dominant model) (Table 2; Fig. 1D). This variant has a borderline association with OS in adenocarcinoma patients, but not in squamous cell lung cancer patients, which is likely due to sample size reduction in the subgroup analysis (Supplemental Table S3). When we combined all risk genotypes into the number of risk genotypes (NRGs, i.e., the number of ERCC1 rs3212924 GG/AG and XPC rs2229090 GG genotypes) for assessing their joint effect on PFS, the frequencies of patients with a score of 0, 1 or 2 for NRGs were 205, 347 and 156, respectively (Table 2). A dose-dependent trend was observed for patients carrying at least one of these genotypes and patients carrying two of these genotype had the highest risk for disease progression, compared with those carrying zero risk genotypes (HRadj = 1.50, 95% CI = 1.19–1.90, P log-rank = 0.017, P adj = 0.0006) (Table 2; Fig. 1A). After dichotomizing patients into a low-risk (0 risk genotype) (LRi) or a high-risk (1–2 risk genotypes) (HRi) group, patients in the HRi group exhibited a significant shorter survival time before progression (HRadj = 1.32, 95% CI = 1.10–1.58, P adj = 0.003), compared to those in the LRi group (Table 2).
Kaplan-Meier survival curves for non-small cell lung cancer (NSCLC) patients by risk genotypes. (A) Progression-free survival (PFS) curves for a score of 0, 1, 2 for the number of risk genotypes (NRGs, i.e. rs3212924 AG/GG, rs2229090 GG); (B) Overall survival (OS) curve for a score of 0, 1, 2 for the NRGs (i.e. rs3212924 AG/GG, rs2229090 GG); (C) PFS curves for ERCC4 rs1799798 risk genotypes (GA/AA vs. GG); (D) OS curves for ERCC4 rs1799798 risk genotypes (GA/AA vs. GG).
Stratified analysis between the risk genotypes and NSCLC survival
Stratified analysis was also performed to assess differential effects of demographic or clinical variables (such as tumour histological type and treatment) on survival risk associated with genotype groups (LRi or HRi) or risk genotypes. Overall, the risk genotype group carriers (ERCC1 rs3212924 AG/GG and XPC rs2229090 GG) tended to have a significantly increased risk of disease progression in subgroups of younger (≤58 years old), males, current smokers, TNM stage III, no radiotherapy, ECOG status 2, poorly differentiated, platinum-docetaxel/paclitaxel recipients. Most homogeneity tests did not provide any evidence to support for differences in HRs between the strata, except for the performance status (P = 0.006), which may be caused by unbalanced distribution of risk genotype groups between different subgroups. For ERCC4 rs1799798 GA/AA carriers, an increased risk of death was observed in older patients (>58 years), non-smokers or former smokers, well-moderately differentiated tumours, and recipients of carboplatin-based or TKI chemotherapies, compared with the GG carriers (Supplemental Table S5).
Correlations between ERCC1 and XPC risk genotypes and mRNA expression levels
To examine genotypic effect of the survival-associated rSNPs on gene expression, the eQTL analysis of the three NER rSNPs was further performed by using two publically available datasets. One included the GTEx samples of normal lung tissues, in which the ERCC1 rs3212924 G allele was associated with a significantly higher ERCC1 mRNA expression level (P = 0.038, effect size = 0.13) (Fig. 2A). The XPC rs2229090 protective C allele was associated with a lower expression level of XPC (z-score = −6.83, P = 8.39E-12) and a nearby gene TMEM43 in peripheral blood cells (z-score = −6.29, P = 3.17E-10, Fig. 2D). Therefore, it is biologically plausible that the associations between those variants and NSCLC survival may be explained by the difference in gene expression levels regulated by those variants. That is, an increased expression of ERCC1 was associated with a poor survival, whereas a decreased expression of XPC was associated with a better survival, and these support the notion that DNA repair is a double-edged sword.
The eQTL analysis of ERCC1 rs3212924 and XPC rs2229090 by using two publically available databases. (A) mRNA expression levels of ERCC1 by rs3212924 genotypes in lung tissues from 278 individuals (P = 0.038, effect size = 0.13); (B) mRNA expression levels of VN1R20P by rs2229090 in tibial artery tissues from 285 individuals (P = 4.5E-12, effect size = −0.49); (C) Genomic position of XPC, VN1R20P and TMEM43; (D) XPC rs2229090 and gene expression in peripheral blood samples from 5,311 individuals and 2,775 replications (blood eQTL browser). Abbreviations: SNP, single nucleotide polymorphism; Chr, chromosome; FDR, false discovery rate.
Discussion
According to the American Society of Clinical Oncology and National Comprehensive Cancer Network (NCCN) guidelines, lung cancer patients with a performance status of 0 or 1 should be treated with a combination of a platinum drug (cisplatin or carboplatin) and a non-platinum drug (e.g. paclitaxel) in the first-line therapy25. Cytotoxicity of platinum compounds results from formation of Plt-DNA adducts, leading to bulky distortion of DNA, destabilization of the double helix, inhibition of DNA replication, transcription and ultimately death of tumour cells26. Better clinical outcome was observed in patients with higher levels of Plt-DNA adducts in the tumours13. DNA repair capacity, particularly of the NER pathway, has been associated with the PBC efficacy. This is because NER primarily repairs bulky DNA adducts caused by mutagens and guanine-cisplatinium adducts formed during PBC17. Likewise, in vitro studies have also shown that NER is the major DNA repair pathway responsible for the repair of cisplatin-DNA damage10.
Previous association studies on SNPs of NER genes and the survival of NSCLC have mainly focused on missense variants or coding regions of individual genes, with very few studies focusing on all the core genes in the pathway, linkage disequilibrium (LD) blocks or non-coding variants27. We adopted a hypothesis-based approach with a main focus on regulatory variants predicted to be biologically functional in NER. In the present study, we found that two rSNPs (ERCC1 rs3212924 and XPC rs2229090) were associated with PFS and one rSNP (ERCC4 rs1799798) associated with OS of NSCLC patients, and these associations were not previously reported for lung cancer. The rs3212924 variant resides at the upstream or an intron of different ERCC1 transcripts, with a predicted function of altering transcription factor binding, which may further affect gene expression. Additional evidence from the eQTL analysis also indicated a significantly higher mRNA expression level in lung tissues containing the risk ERCC1 G allele. Difference in gene expression by the rs3212924 G allele has been observed not only in lung tissue, but also in artery, skin, and ovary tissues, suggesting a genetically determined regulatory role of this variant in its gene expression. In the rs3212924 LD block, none of the other SNPs in high LD (r2 > 0.8) have been previously reported to be associated with cancer survival (Fig. 3A). Taken together, the associations between this variant with high tumour tissue levels of ERCC1 mRNA may have led to cisplatin resistance28, 29, which may have independently affected disease progression in NSCLC patients.
LD block of ERCC1 and XPC risk variants. (A) LD block of ERCC1 rs3212924; (B) LD block of XPC rs2229090. The blue circle indicates the SNP under study. R2 indicates the linkage disequilibrium value between the two pointed SNPs. Abbreviations: NER, nucleotide excision repair; LD, linkage disequilibrium.
The rs2229090 variant is located at the 3′UTR of XPC, and the G to C allele substitution is predicted to affect miRNA binding. In fact, the eQTL analysis indicated a genotypic effect of rs2229090 on expression of a pseudogene (Vomeronasal 1 Receptor 20 Pseudogene, VN1R20P) downstream of XPC in tibial artery tissues (Fig. 2B) and XPC expression in peripheral blood cells (Fig. 2D). In the corresponding LD block (Fig. 3B ), only variant rs2228000 (Ala499Val) (r2 = 0.875) was reported to be associated with survival of patients with various cancers including lung cancer30,31,32,33. Prior evidence indicated that subjects carrying rs2228000 CT/TT genotypes exhibited a better DNA repair capacity, and a poorer survival or risk of recurrence in oropharynx squamous cell carcinoma34 and acute myeloid leukaemia35. However, no detailed molecular mechanism of how rs2228000 T allele functions in these associations has ever been reported. It is likely that the phenotypic change of XPC function associated with rs2228000 may have been responsible for the observed association with rs2229090 that is within the same LD block (Fig. 3B). Because XPC plays a key role in recognizing DNA damage and initiation of the NER process, these collective findings suggest that XPC variants at the rs2229090 block may have an impact on PFS in NSCLC patients treated with PBC by affecting PBC outcome through changing XPC expression and thus the DNA repair capacity. These findings call for further functional studies to reveal the biological mechanisms underlying those associations.
In the subgroup analysis by histological type and chemotherapy treatment, XPC rs2229090 GC/CC exhibited a significant association with a longer PFS, while ERCC4 rs1799798 GA genotype was significantly associated with a shorter OS in 477 adenocarcinoma patients, but not in 138 squamous cell carcinoma patients, suggesting a potential histological difference in genetic regulation of lung cancer survival outcome in response to the treatments. Although XPC rs2229090 GC/CC genotypes were significantly associated with PFS in squamous cell carcinoma patients who received docetaxel-cisplatin, the sample size of this treatment group was relatively small (n = 56); hence, this result needs to be interpreted with caution (Supplemental Table S4). It is also possible that the sample size in most of the subgroups was not large enough to reveal the real associations, suggesting that future larger validation studies are required to substantiate our findings.
The ROC curve prediction model for PFS incorporating ERCC1 and XPC risk genotypes exhibited a statistically significant improvement in discriminatory power, compared with that of the clinical factors only (I/C AUC 0.59 vs. 0.58, P = 0.019) (Supplemental Fig. S1A and B; Table S7). There was a trend towards a higher AUC of ROC and C index in the genotype-inclusive prediction models for the five-year overall survival (Supplemental Fig. S1C and D; Table S7).
There are inherent limitations in the present study. First, the recruitment of patients treated in the same hospital may lead to selection bias in generalization of the results to the general population; therefore, additional results of patients from other hospitals of other populations are necessary to confirm our findings. Second, with the aim of studying potentially functional SNPs in the regulatory regions of eight NER core genes, we did not incorporate the other known effect of non-synonymous SNPs on survival outcome of NSCLC patients, although they are not in the LD block with the ones under investigation in the present study (except for rs2228080). Third, multiple testing correction was not conducted in the present study, because this was an exploratory study with a limited study power. Prospective studies in larger populations are warranted to substantiate the findings in the present study.
Conclusions
The present study provided evidence that rSNPs in the core NER genes may modulate PBC-related survival outcome in Chinese NSCLC patients with an advanced stage disease. Potential gene regulation by rSNPs of two NER genes associated with outcomes of patients with NSCLC call for further functional studies to unravel the molecular mechanisms underlying the observed associations, which will also allow for further development of predictive biomarkers to facilitate personalized chemotherapy regime.
Material and Methods
Study populations
The present study was conducted on patients diagnosed with histologically advanced NSCLC from Fudan University Shanghai Cancer Centre (FUSCC) between February 1, 2009 and November 30, 2013. The recruitment criteria included the following: (1) unrelated Han Chinese with inoperable TNM stages III to IV tumours of NSCLC without prior history of cancer other than in situ carcinoma; (2) received PBC as the first-line treatment; (3) having Eastern Cooperative Oncology Group performance (ECOG) status 0 to 2 with laboratory testing for blood tests and uronoscopy in normal range; (4) no active infection and serious medical or psychological conditions that might prevent patients from adhering to treatment; and (5) patients with recent myocardial infarction, cardiac arrhythmia, active congestive heart failure or cerebral apoplexy, crankiness or depression were excluded from this study. The clinical data including age at treatment, sex, smoking history, ECOG performance, TNM stage, histological type and grade, chemotherapy regimens, radiotherapy, tyrosine-kinase (TKI) treatment were collected from patients’ medical records.
Survival data
Survival data were collected from patients’ next of kin through a telephone follow-up and inpatient and outpatient clinical medical records. OS time was calculated from the starting date of the treatment until the date of the last follow-up or death. PFS time was measured from the starting date of the treatment until the last follow-up, progression of disease or death. Patients without progression were censored at the date of last contact. The median follow-up time was 32.1 months. The Institutional Review Board of FUSCC approved this study, with all methods performed in accordance with the guidelines and regulations of FUSCC. All participants provided an informed consent for using their blood samples in future research.
Chemotherapy Regimens
All patients enrolled in the study were given the first-line PBC, which consists of ten combinations: cisplatin (75 mg/m2) or carboplatin (area under the curve [AUC] 6 mg/ml·min), administered with paclitaxel (175 mg/m2) on day 1 every 3 weeks, docetaxel (75 mg/m2) on day 1 every 3 weeks, gemcitabine (1250 mg/m2) on day 1 and 8 every 3 weeks, pemetrexed (500 mg/m2) on day 1 every 3 weeks or vinorelbine (25 mg/m2) on days 1 and 8 every 3 weeks, and cisplatin (100 mg/m2) or carboplatin (AUC 6 mg/ml·min) administered on day 1 every 4 weeks, in combination with etoposide (100 mg/m2) on days 1 to 3 every 4 weeks. All chemotherapeutic drugs were administered intravenously.
SNP selection
To specifically explore the association between rSNPs in core NER genes and survival of NSCLC in response to PBC, all rSNPs were queried from the NER gene regions under the study by using SNP/GeneView in dbSNP database (http://www.ncbi.nlm.nih.gov/snp/) using the GRCh38 reference build of the human genome. A total of 25 rSNPs in eight (out of nine) core NER genes were chosen, with detailed characteristics of all investigated genes and rSNPs shown in (Table 1 and Supplemental Table S1 ). The selection criteria were based on the following: minor allele frequency (MAF) ≥ 5% in Han Chinese, in the regulatory region (5′ near gene, 5′UTR, intron, 3′ near gene, or 3′UTR), in low LD with each other (r2 < 0.8), have predicted functions (transcription factor binding site, splicing, miRNA binding site or significant eQTL) by SNPinfo (http://snpinfo.niehs.nih.gov/snpfunc.htm) and GTEx portal (http://www.gtexportal.org/home/). A full list of the NER genes analysed in this study, their region coordinates, their start sites and stop sites, and the characteristics of genotyped variants are summarized in Table 1.
SNPseq genotypin
Genomic DNA was extracted from the whole blood of all study subjects by using DNA Blood Mini Kit (Qiagen, Valencia, CA). The purity [optical density (OD)260/280 at 1.7~2.0] and concentration ( >20 ng/μl) that met the sequencing requirements. Genotyping of all rSNPs was conducted by FastTarget, a next generation sequencing-based method using Illumina Miseq. 2000 Platform (2 × 250 bp, Illumina, CA, USA). Prior to sequencing, 5% of the samples were randomly selected and subjected to 1% agarose gel electrophoresis quality control. Genomic regions containing the investigating rSNPs were amplified using the FastTargetTM technology (Genesky Biotechnologies Inc, Shanghai, China). A total of 25 amplicons were amplified, with the primers information attached in Supplemental Table S6. After multiple PCR reactions, DNA fragments were ligated with the adaptor by using Q5 DNA polymerase Kit (New England Biolabs, MA, USA), and further purified by Agencourt AMPure XP (Beckman Coulter, CA, USA). Next-generation sequencing of the amplification products was carried out by MiSeq 2000 Sequencer (Illumina, Inc., San Diego, CA, USA), following the manufacturer’s standard protocols. Sequencing depth of more than 30x was achieved for over 90% of the samples. Output sequence data were trimmed and then compared with fragment reference sequences (hg19) using the Blat program28. Burrows- Wheeler Aligner (BWA, V 0.7.5a) was used to map the reads36, followed by Sequence Alignment/Map (SAM)-to-BAM conversion, sorting, and removal of duplicates using SAM tools (v0.1.19)37. Combined rSNP calling was performed on the resulting BAM files using Genome Analysis Toolkit (GATK, https://software.broadinstitute.org/gatk/best-practices/) and VarScan programs38. Finally SNP annotation was done by the Annovar program39.
Statistical analysis
The association between each genetic variant and PFS/OS was estimated by Cox proportional hazards regression model, calculated as HRs with their corresponding 95% CIs. The covariates used for adjusted HR for PFS included age-at-treatment, sex, smoking status, TNM stage, histological type, histologic grade, ECOG performance status, chemotherapy regimens, grade 3/4 chemotherapy toxicity and palliative radiotherapy, whereas TKI treatment was included for adjusted HR for OS in addition to the covariates mentioned above. Kaplan-Meier test was used to assess each genetic variant on the cumulative probability of PFS and OS40. Log-rank test was used to examine the difference in survival between groups. The observed associations were stratified by selected demographic and clinical variables. The heterogeneity between subgroups was assessed by the χ 2-based Q test. For survival prediction model construction, independent predictors including selected clinical variables and genetic variants were included. ROC analysis was used to compare sensitivity and specificity of the OS and PFS prediction by the included parameters. Predictive values of selected variables were evaluated by I/D AUC of the ROC curves for censored data and C index for comparison of survival models. The I/D ROC and I/D AUC were calculated and plotted by RisksetROC package of R software (version 3.2.3; The R Foundation for Statistical Computing)41. All statistical analyses were performed by SAS software (version 9.4; SAS Institute, Cary, NC). Unless stated otherwise, all P values were two-sided with a significance level of P < 0.05.
The eQTL analysis
Two large-scale eQTL datasets were used to assess the correlation between survival-related genetic variants and NER gene expression levels: one is the GTEx project using 278 lung tissue samples, and the other is the blood eQTL browser (http://www.genenetwork.nl/bloodeqtlbrowser/) encompassing 5,311 individuals and 2,775 replicates42.
References
Stewart BW, W. C. World Cancer Report 2014 (WHO Press, 2014).
Chen, W., Zheng, R., Zeng, H., Zhang, S. & He, J. Annual report on status of cancer in China, 2011. Chin J Cancer Res 27, 2–12, doi:10.3978/j.issn.1000-9604.2015.01.06 (2015).
Li, C. et al. Polymorphisms of nucleotide excision repair genes predict melanoma survival. J Invest Dermatol 133, 1813–1821, doi:10.1038/jid.2012.498 (2013).
Herbst, R. S., Heymach, J. V. & Lippman, S. M. Lung cancer. N Engl J Med 359, 1367–1380, doi:10.1056/NEJMra0802714 (2008).
Sandler, A. et al. Paclitaxel-carboplatin alone or with bevacizumab for non-small-cell lung cancer. N Engl J Med 355, 2542–2550, doi:10.1056/NEJMoa061884 (2006).
Scagliotti, G. V. et al. Phase III study comparing cisplatin plus gemcitabine with cisplatin plus pemetrexed in chemotherapy-naive patients with advanced-stage non-small-cell lung cancer. J Clin Oncol 26, 3543–3551, doi:10.1200/JCO.2007.15.0375 (2008).
Rosell, R. et al. Erlotinib versus standard chemotherapy as first-line treatment for European patients with advanced EGFR mutation-positive non-small-cell lung cancer (EURTAC): a multicentre, open-label, randomised phase 3 trial. Lancet Oncol 13, 239–246, doi:10.1016/S1470-2045(11)70393-X (2012).
Camidge, D. R. et al. Activity and safety of crizotinib in patients with ALK-positive non-small-cell lung cancer: updated results from a phase 1 study. Lancet Oncol 13, 1011–1019, doi:10.1016/S1470-2045(12)70344-3 (2012).
Bahl, A. & Falk, S. Meta-analysis of single agents in the chemotherapy of NSCLC: what do we want to know? Br J Cancer 84, 1143–1145, doi:10.1054/bjoc.2000.1740 (2001).
Reed, E. Platinum-DNA adduct, nucleotide excision repair and platinum based anti-cancer chemotherapy. Cancer Treat Rev 24, 331–344 (1998).
Zamble, D. B. & Lippard, S. J. Cisplatin and DNA repair in cancer chemotherapy. Trends Biochem Sci 20, 435–439 (1995).
Rosell, R., Cecere, F., Santarpia, M., Reguart, N. & Taron, M. Predicting the outcome of chemotherapy for lung cancer. Curr Opin Pharmacol 6, 323–331, doi:10.1016/j.coph.2006.01.011 (2006).
Bosken, C. H., Wei, Q., Amos, C. I. & Spitz, M. R. An analysis of DNA repair as a determinant of survival in patients with non-small-cell lung cancer. J Natl Cancer Inst 94, 1091–1099 (2002).
Campayo, M. et al. Single nucleotide polymorphisms in tobacco metabolism and DNA repair genes and prognosis in resected non-small-cell lung cancer. J Surg Res 167, e5–12, doi:10.1016/j.jss.2011.01.007 (2011).
Cheng, H. et al. Predictive effect of XPA and XPD polymorphisms on survival of advanced NSCLC patients treated with platinum-based chemotherapy: a three-dimensional (3-D), polyacrylamide gel-based DNA microarray method. Technol Cancer Res Treat 12, 473–482, doi:10.7785/tcrt.2012.500337 (2013).
Gurubhagavatula, S. et al. XPD and XRCC1 genetic polymorphisms are prognostic factors in advanced non-small-cell lung cancer patients treated with platinum chemotherapy. J Clin Oncol 22, 2594–2601, doi:10.1200/JCO.2004.08.067 (2004).
de Boer, J. & Hoeijmakers, J. H. Nucleotide excision repair and human syndromes. Carcinogenesis 21, 453–460 (2000).
Rosell, R. et al. Reduced survival in patients with stage-I non-small-cell lung cancer associated with DNA-replication errors. Int J Cancer 74, 330–334 (1997).
Shellard, S. A., Fichtinger-Schepman, A. M., Lazo, J. S. & Hill, B. T. Evidence of differential cisplatin-DNA adduct formation, removal and tolerance of DNA damage in three human lung carcinoma cell lines. Anticancer Drugs 4, 491–500 (1993).
Lindahl, T. & Wood, R. D. Quality control by DNA repair. Science 286, 1897–1905 (1999).
Hoeijmakers, J. H. Genome maintenance mechanisms for preventing cancer. Nature 411, 366–374, doi:10.1038/35077232 (2001).
Friedberg, E. C., Walker, G. C. & Siede, W. DNA Repair and Mutagenesis (ASM Press, Washington, 1995).
Keeney, S., Chang, G. J. & Linn, S. Characterization of a human DNA damage binding protein implicated in xeroderma pigmentosum E. J Biol Chem 268, 21293–21300 (1993).
Jia, M. et al. The P38alpha rs3804451 Variant Predicts Chemotherapy Response and Survival of Patients with Non-Small Cell Lung Cancer Treated with Platinum-Based Chemotherapy. Transl Oncol 9, 531–539, doi:10.1016/j.tranon.2016.09.006 (2016).
Azzoli, C. G. et al. American Society of Clinical Oncology Clinical Practice Guideline update on chemotherapy for stage IV non-small-cell lung cancer. J Clin Oncol 27, 6251–6266, doi:10.1200/JCO.2009.23.5622 (2009).
Qiu, M. et al. Predictive value of XPD polymorphisms on platinum-based chemotherapy in non-small cell lung cancer: a systematic review and meta-analysis. PLoS One 8, e72251, doi:10.1371/journal.pone.0072251 (2013).
Li, F. et al. Association between polymorphisms of ERCC1 and XPD and clinical response to platinum-based chemotherapy in advanced non-small cell lung cancer. Am J Clin Oncol 33, 489–494, doi:10.1097/COC.0b013e3181b9cedc (2010).
Dabholkar, M., Vionnet, J., Bostick-Bruton, F., Yu, J. J. & Reed, E. Messenger RNA levels of XPAC and ERCC1 in ovarian cancer tissue correlate with response to platinum-based chemotherapy. J Clin Invest 94, 703–708, doi:10.1172/JCI117388 (1994).
Furuta, T. et al. Transcription-coupled nucleotide excision repair as a determinant of cisplatin sensitivity of human cells. Cancer Res 62, 4899–4902 (2002).
He, B. S. et al. Nucleotide excision repair pathway gene polymorphisms are linked to breast cancer risk in a Chinese population. Oncotarget 7, 84872–84882, doi:10.18632/oncotarget.12744 (2016).
Hua, R. X. et al. Association of XPC Gene Polymorphisms with Colorectal Cancer Risk in a Southern Chinese Population: A Case-Control Study and Meta-Analysis. Genes (Basel) 7, doi:10.3390/genes7100073 (2016).
Ravegnini, G. et al. Polymorphisms in DNA repair genes in gastrointestinal stromal tumours: susceptibility and correlation with tumour characteristics and clinical outcome. Tumour Biol 37, 13413–13423, doi:10.1007/s13277-016-5276-7 (2016).
Farnebo, L. et al. DNA repair genes XPC, XPD, XRCC1, and XRCC3 are associated with risk and survival of squamous cell carcinoma of the head and neck. DNA Repair (Amst) 31, 64–72, doi:10.1016/j.dnarep.2015.05.003 (2015).
Song, X. et al. Variants in nucleotide excision repair core genes and susceptibility to recurrence of squamous cell carcinoma of the oropharynx. Int J Cancer 133, 695–704, doi:10.1002/ijc.28051 (2013).
Strom, S. S., Estey, E., Outschoorn, U. M. & Garcia-Manero, G. Acute myeloid leukemia outcome: role of nucleotide excision repair polymorphisms in intermediate risk patients. Leuk Lymphoma 51, 598–605, doi:10.3109/10428190903582804 (2010).
Li, H. & Durbin, R. Fast and accurate long-read alignment with Burrows-Wheeler transform. Bioinformatics 26, 589–595, doi:10.1093/bioinformatics/btp698 (2010).
Li, H. et al. The Sequence Alignment/Map format and SAMtools. Bioinformatics 25, 2078–2079, doi:10.1093/bioinformatics/btp352 (2009).
Koboldt, D. C. et al. VarScan 2: somatic mutation and copy number alteration discovery in cancer by exome sequencing. Genome Res 22, 568–576, doi:10.1101/gr.129684.111 (2012).
Wang, K., Li, M. & Hakonarson, H. ANNOVAR: functional annotation of genetic variants from high-throughput sequencing data. Nucleic Acids Res 38, e164, doi:10.1093/nar/gkq603 (2010).
Klein, J. a. M., M. Survival Analysis:Techniques for Censored and Truncated Data (Springer, 1997).
Heagerty, P. J. & Zheng, Y. Survival model predictive accuracy and ROC curves. Biometrics 61, 92–105, doi:10.1111/j.0006-341X.2005.030814.x (2005).
Westra, H. J. et al. Systematic identification of trans eQTLs as putative drivers of known disease associations. Nat Genet 45, 1238–1243, doi:10.1038/ng.2756 (2013).
Acknowledgements
The authors acknowledge Dr. Haiquan Chen at the Department of Thoracic Surgery for contributing lung cancer tissues, the staff members at the tissue bank of the Fudan University Shanghai Cancer Centre for their tremendous support in providing stored blood samples. This research was supported by China’s Thousand Talents Program at Fudan University, Shanghai, China.
Author information
Authors and Affiliations
Contributions
R.Z. and M.J. contributed equally to this work by coordinating and implementing the study, analysing data and writing the manuscript. H.X. and M.Z. helped collected and organized the clinical data information and contributed to the sample collection and DNA extraction. Y.X. and M.W. helped with sample preparation and genotyping. M.S. helped provide lung cancer patient blood samples. J.C. and Q.W. conceived the design, participated in coordination of the study, provided financial support, and participated in writing. All authors have read and approved of the final manuscript for submission.
Corresponding authors
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zhang, R., Jia, M., Xue, H. et al. Genetic variants in ERCC1 and XPC predict survival outcome of non-small cell lung cancer patients treated with platinum-based therapy. Sci Rep 7, 10702 (2017). https://doi.org/10.1038/s41598-017-10800-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-10800-5
This article is cited by
-
Hsa_circ_0069244 acts as the sponge of miR-346 to inhibit non-small cell lung cancer progression by regulating XPC expression
Human Cell (2021)
-
Epigenetic based synthetic lethal strategies in human cancers
Biomarker Research (2020)
-
DNA repair in cancer initiation, progression, and therapy—a double-edged sword
Journal of Applied Genetics (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.