Abstract
Ultrafine particles, more heterojunction interfaces and amorphous materials can effectively enhance the photocatalytic activity of photocatalysts. In this work, a facile in-situ precipitation method was developed to prepare ultrafine amorphous iron oxyhydroxide/ultrathin g-C3N4 nanosheets heterojunction composites. The amorphous iron oxyhydroxide possessed an ultrafine particle size and a wide range of visible light absorption. In this process, the ultrafine particles not only shortened the diffusion distance of photogenerated carriers, but also facilitated the formation of more heterojunctions with ultrathin g-C3N4 nanosheets. The photocatalytic activities were evaluated using rhodamine B, methylene blue, and methyl orange as pollution models under visible light irradiation. Notably, the optimal photocatalytic activity of a-FeOOH/CNNS-800 composite is ~17.8 times higher than that of CNNS towards the degradation of rhodamine B under visible light. The outstanding photocatalytic activities were ascribed to the narrower band gap, the enhanced visible light absorbance, abundant heterojunction interfaces, and the effective separation of the photogenerated charges driven by the matched band edge in the heterostructures. We trusted that the facile and easy-to-extend synthesis method can be further expanded to synthesize other ultrafine semiconductors coupled with g-C3N4 for enhancing the photocatalytic activities.
Similar content being viewed by others
Introduction
In recent years, the environmental pollution is becoming more and more serious with the development of economy, which has become a serious threat to human survival and development1,2,3,4. The semiconductor photocatalytic technique has been regarded as an important way to solve this issue by decomposing toxic and hazardous organic pollutants into non-toxic products5,6,7. However, the traditional semiconductor photocatalytic technique has some drawbacks, such as the only absorb UV light and the low photocatalytic efficiencies3, 8,9,10. Therefore, the development of novel photocatalysts with visible-light response and high photocatalytic efficiencies has become urgent8, 11, 12.
Graphite carbon nitride (g-C3N4), as metal-free semiconductor, has received great attention in photocatalytic degradation of organic pollutants, due to its unique three-dimensional layered structure, relatively narrow band gap (~2.7 eV) and low cost13,14,15,16. However, a low quantum efficiency and narrow visible-light response range restricted its application5, 17. To conquer these defects, numerous strategies have been explored, including combination with carbon materials, metal and/or non-metallic materials doping, formation of heterojunctions, and so on9, 12, 18, 19. For example, a series of g-C3N4-based photocatalysts was constructed by Zhang group and G. Mamba group, which enhanced the visible-light photocatalytic performance through the formation of heterojunctions4, 20. Among these, the coupling of other semiconductors with suitable bandgap on g-C3N4 to form heterojunctions is an effective method to enhance visible-light absorption ranges and improve photogenerated charge separation and transfer18. However, it is still a challenge to explore suitable semiconductors to satisfy these requirements.
The amorphous iron oxyhydroxide (a-FeOOH), as one kind of iron oxides/hydroxides, has attracted attention for hydrogen production from water splitting and degradation of toxic organic pollutants under UV or visible-light irradiation21,22,23. The amorphous iron oxyhydroxide is easy to get in the natural environment with the features of non-toxic, corrosion resistant and low cost19, 23,24,25,26. In addition, the narrow band gap endows wide light response ranges to ensure harvest visible light, which is the premise to achieve high photocatalytic efficiency20, 25, 27, 28. Accordingly, it is expected that the amorphous iron oxyhydroxide coupled g-C3N4 would show high photocatalytic performance due to the following reasons: (i) it could obtain a wide range of visible light absorption; (ii) it has a suitable band structure, which could form heterojunctions, decreased the recombination of photogenerated charge and improved the photocatalytic efficiency17, 29.
As is known to all, interfacial charge transfer between the discrete energy levels of molecules and the continuous energy levels of solids has been an effective method to raise the photocatalytic activity of semiconductor photocatalysts6, 30. Furthermore, abundant heterojunction interface will separate the photogenerated electron-hole pairs more effectively18, 31. Therefore, it is feasible to enhance the photocatalytic activities of g-C3N4-based photocatalysts by tuning the size and distribution of the coupled nanoparticles benefited from the two merits: (i) the smaller particles obviously shorten the diffusion distance of photogenerated carriers, improving the photocatalytic performance; (ii) ultrafine particles coupling with g-C3N4 can generate more effective heterojunctions and further facilitate the synergistic reaction between semiconductors and g-C3N4 32,33,34,35. In other words, ultrafine nanoparticles and more efficient heterojunctions can also enhance the catalytic properties of photocatalysts32,33,34,35.
In this work, we prepared the novel ultrafine amorphous iron oxyhydroxide/g-C3N4 nanosheets heterojunction nanocomposites through a simple in-situ precipitation method by uniformly dispersing ultrafine amorphous iron oxyhydroxide to the surface of g-C3N4 nanosheets. During the reaction, the designed amorphous iron oxyhydroxide/g-C3N4 nanosheets heterostructures are featured with an ultrafine amorphous iron oxyhydroxide anchored on the ultrathin g-C3N4 nanosheets, providing a large surface area and abundant heterojunction interfaces. The photocatalytic activities of amorphous iron oxyhydroxide/g-C3N4 nanosheets were evaluated by degrading rhodamine B (RhB), methylene blue (MB) and methyl orange (MO) under visible light irradiation. In addition, the degradation mechanism was also proposed.
Experimental sections
Preparation of the amorphous iron oxyhydroxide (a-FeOOH)
a-FeOOH was synthesized by a simple in-situ precipitation method. Firstly, 1 mmol FeCl3·6H2O was dissolved in 150 mL anhydrous ethanol under stirring. Then, 3 mmol ammonium bicarbonate (NH4HCO3) was added with stirring, and the solution kept stirring for 8 hours. Finally, the precipitates were collected and washed with anhydrous ethanol, followed by vacuum freeze drying.
Synthesis of g-C3N4 nanosheets (CNNS)
Urea was placed in the covered crucible and heated under static air at 550 °C for 4 h with a ramp rate of 2.5 °C/min. The as-obtained powders were placed in an open crucible and further heated at 500 °C for 2 h with a ramp rate of 5 °C/min. After cooling down to room temperature, the desired ultrathin CNNS were obtained by washing with deionized water and further dried in a vacuum oven.
Synthesis of amorphous iron oxyhydroxide/g-C3N4 nanosheets composites (a-FeOOH/CNNS)
The a-FeOOH/CNNS composites were synthesized by a facile in-situ precipitation method, similar as the synthesis of a-FeOOH. Specifically, 1 mmol FeCl3·6H2O was dissolved in 150 mL of anhydrous ethanol. Then, a certain amount of CNNS were added into the solution and sonicated for 2 h to disperse CNNS. After that, 3 mmol ammonium bicarbonate (NH4HCO3) was added into the suspension and the reaction was continued another 8 hours. The precipitates were collected and washed with anhydrous ethanol, followed by vacuum freeze drying. a-FeOOH/CNNS composites prepared with different amounts of CNNS in 300, 400, 500, 600, 700, 800 and 900 mg were denominated as a-FeOOH/CNNS-300, a-FeOOH/CNNS-400, a-FeOOH/CNNS-500, a-FeOOH/CNNS-600, a-FeOOH/CNNS-700, a-FeOOH/CNNS-800 and a-FeOOH/CNNS-900, respectively.
Characterization
Powder X-ray diffraction (XRD) data were obtained via a D/MAX2500 V diffractometer equipped with Cu Kα radiation (λ = 1.5418 Å). The infrared absorption spectra of the materials were measured by a Fourier transform spectrophotometer (FT-IR, Avatar 370, Thermo Nicolet) using the standard KBr disk method. X-ray photoelectron spectroscopy (XPS) measurements were conducted on ESCALAB250 with Mg Kα as the source and the C 1s peak at 284.6 eV as an internal standard. The morphologies of the materials were observed using a FEI QUANTA FEG250 field emission scanning electron microscope (SEM) and a Tecnai G2 F20 S-TWIN transmission electron microscope (TEM) at an accelerating voltage of 200 kV as well as the composites. UV-vis diffuse reflection spectroscopy (DRS) was performed on a Shimadzu UV-2500 spectrophotometer using BaSO4 as the reference.
Evaluation of photocatalytic activity
The photocatalytic activity of as-prepared catalysts was evaluated by degrading RhB, MB and MO under a 500 W Xe lamp equipped with a cutoff filter (λ ≥ 420 nm) as a light source. In short, 50 mg photocatalysts were added into 50 mL of RhB (or MB, MO) aqueous solution (10 mg/L). The mixed solution was magnetically stirred for 60 min to achieve adsorption–desorption equilibrium before turning on Xe lamp. During the photocatalytic test, 3 mL suspension was sampled, followed by centrifugation to separate the photocatalyst at a certain irradiation time interval, and the photocatalytic efficiency was tested by a UV-vis spectro-photometer (UV-2500, Shimadzu).
Results and Discussion
A simple in-situ precipitation synthesis method was designed to prepare a-FeOOH and a-FeOOH/CNNS hybrid hierarchical three-dimensional architectures, repesctively, and the preparation process was schematically shown in Fig. 1. The process may be described as follows22:
For the preparation of a-FeOOH, NH4HCO3 was added to the FeCl3 solution with the formation of a-FeOOH. The chemical reaction was carried out very homogeneously forming ultrafine a-FeOOH nanoparticles. CNNS could be easily obtained by thermal oxidation etching of bulk g-C3N4 in air. The dispersed CNNS were negatively charged, with a zeta potential of about −27.6 mV (Figure S1). By adding CNNS into FeCl3 solution, Fe3+ cations would be bound tightly to the surface of CNNS via electrostatic interactions. Then, with the addition of NH4HCO3, the fixed Fe3+ cations would further react with NH4HCO3 to generate a-FeOOH, resulting in a-FeOOH/CNNS. In this case, CNNS not only provided a large surface area, but also acted as the support to form heterostructures. This synthetic route to a-FeOOH/CNNS photocatalysts was economic, facile and could be produced in a large scale, which provided new opportunities for the preparation of amorphous metal oxide nanoparticles.
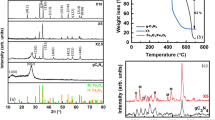
Figure 2 showed the XRD patterns of pure CNNS and a-FeOOH/CNNS composites with different amounts of CNNS. For pure CNNS, the diffraction peaks at 13.1° and 27.8° corresponded to (100) and (002) diffraction planes of graphitic carbon nitride, respectively3, 7, 18, 36. All XRD patterns of composites did not show the diffraction peaks of a-FeOOH, probably indicating that the amorphous feature of a-FeOOH or the few content of a-FeOOH in the composites22.
Figure 3 exhibited the FT-IR spectra of pure CNNS and a-FeOOH/CNNS composites, which was used to reflect the difference in chemical structure of materials. For pure CNNS and its composites, the broadened band in the range of 3000–3700 cm−1 represented the N-H stretching vibration of CNNS1, 3, 6. The several strong peaks within 1400–1800 cm−1 belonged to C = N and C-N heterocycles skeletal vibration of the aromatic ring7,8,9. The peaks at 1245 and 1325 cm−1 indicated the stretching vibrations of C-NH-C bridges6,7,8,9. And the sharp peak at 814 cm−1 was in line with the C-N stretching vibration of the feature of the triazine cycles, which confirmed the successful introduction of CNNS in the composites9, 11, 12. A small peak at 2356 cm−1 was attributed to the appearance of C≡N and N = C = N, which might be from the small fragment of diazo groups adhered to the surface, but in fact these may not have an impact on the formation of this organic network structure7, 8. For a-FeOOH/CNNS composites, no obvious differences were observed in comparing with CNNS, probably manifesting the introduction of a-FeOOH did not change the chemical structure of CNNS or the content of a-FeOOH was very small1, 6. So XPS was used to demonstrate the existence of a-FeOOH.
The elements of the synthesized a-FeOOH/CNNS-800 by the XPS spectra were shown in Fig. 4. The XPS spectra demonstrated the presence of C, N, Fe and O in a-FeOOH/CNNS-800 hybrid (Figure S2). In the C 1s spectra of a-FeOOH/CNNS-800 (Fig. 4A), the C 1s peak with a binding energy of 284.75 eV was considered to be the standard reference carbon from the background5, 6, 37. In addition to the standard reference carbon, the high resolution C 1s spectrum was also fitted into three carbonaceous species with different binding energy38. The main peak with a binding energy of 288.41 eV represented sp3-C = N bonds, which made up the main structure of CNNS18, 37, 38. There were two small peaks with the binding energy of 285.49 and 289.09 eV. The peak with a binding energy of 285.49 eV could be explained as surface hydroxyl (C-OH) bond, while the peak with a binding energy of 289.09 eV was identified as carbonyl (O-C = O) bond8, 9. These two peaks were often used as the evidence of successful surface modification6.
For the N 1s spectrum (Fig. 4B), there displayed three nitrogen states. The strong peak at 398.89 eV could be attributed to sp2-hybridizednitrogen (C = N-C) coordination in CNNS structures5, 6, 39. The peak locating at 399.99 eV indicated tertiary nitrogen (N-(C)3) groups (quaternary N), while the peak with a binding energy of 401.29 eV was assigned to amino functions (C-N-H)6, 7. These states corresponded to three nitrogen units that constituted the basic units of CNNS17, 18. The weak peak with a binding energy of 404.60 eV was attributed to the charging effects or positive charge localization in the heterocycles38. It meant that the nanostructure of CNNS was not changed after it has been compounded with a-FeOOH20. The Fe 2p spectrum (Fig. 4C) depicted four peaks with binding energy of 710.90, 725.01, 718.04 and 733.02 eV. The formation of a-FeOOH was proven by two major peaks located at 710.93 eV for Fe 2p3/2 and 725.06 eV for Fe 2p1/2, which corresponded to Fe3+.40, 41 Shake-up satellite peaks of Fe 2p3/2 and Fe 2p1/2 can also be observed around 718.02 and 733.07 eV42,43,44. In the O 1s spectrum (Fig. 4D), the peaks located at 530.05 and 531.80 eV were assigned to Fe-O-Fe bond and Fe-O-H bond, respectively, while the weak peak at 533.40 eV was assigned to H-O-H bond38, 40.The discovery of Fe-O-Fe and Fe-O-H bonds also demonstrated the presence of a-FeOOH22.
Figure 5A indicated the light absorption properties of CNNS, a-FeOOH and a-FeOOH/CNNS by UV-vis DRS. The light absorption edge of CNNS was ~454 nm, corresponded to a band gap of ~2.73 eV, which was slightly blue shift relative to the band gap of the bulk g-C3N4, this indicated that CNNS has thinner nanosheets18. The light absorption edge of a-FeOOH was more than 800 nm, so a-FeOOH can absorbed nearly all visible lights. The a-FeOOH/CNNS composites presented the hybrid absorption features of both a-FeOOH and CNNS. Compared with CNNS, all a-FeOOH/CNNS composites exhibited broader visible light absorptions due to intimate interfacial contact between a-FeOOH and CNNS, resulted in the change of optical properties of the composites6. The strength of the absorption bands enhanced with the quantity increase of a-FeOOH corresponding to the color change of the composites (Fig. 5B), which turned from light yellow to reddish brown with the increase of a-FeOOH. It is deduced that the light absorption abilities of CNNS could be significantly enhanced through heterojunctions by way of loading a-FeOOH as “nanometer island” on the surface of CNNS, resulting in a decrease in the interface contact barrier and an enhancement of the electron coupling of the semiconductor, which were beneficial to generate more photogenerated electrons/holes with improved photocatalytic performance18.
The energy levels and band gap of semiconductor photocatalysts play a crucial role in determining photocatalytic performance. The band gap energy (Eg) of a semiconductor can be calculated by the following Eq.:
where α, h, ν, Eg and A represent the light absorption coefficient, Planck constant, light frequency, band gap energy, and constant, respectively. Among them, n is determined by the type of optical transition of a semiconductor (n = 1 for direct transition and n = 4 for indirect transition). The band gaps are estimated to be 2.56 eV and 0.91 eV for CNNS and a-FeOOH respectively (Figure S3), according to a plot of (αhν)1/2 versus energy (hν).
The morphologies of a-FeOOH, a-FeOOH/CNNS-800 and CNNS were characterized using electron microscopes. The related SEM and TEM images are shown in Figs 6 and 7, respectively. It can be seen that CNNS, a-FeOOH/CNNS-400, a-FeOOH/CNNS-600 and a-FeOOH/CNNS-800 exhibit three-dimensional layered structures from the SEM images (Fig. 6), which was favorable for the increase of the reaction surface and surface active sites. As shown in Fig. 7A and D, the particle size of pure a-FeOOH is very small (1–8 nm). The a-FeOOH/CNNS-800 (Fig. 7B and E) and CNNS (Fig. 7C and F) were observed as thin nanosheets with wrinkles, in which overlapped layers were described as black stripes. The SEM and TEM images of CNNS and a-FeOOH/CNNS composites suggested that the morphology of a-FeOOH/CNNS composites did not change compared with pure CNNS. However, a-FeOOH cannot be differentiated from CNNS in the SEM and TEM images due to the ultrafine granularity, so Mapping and EDX was used to further detect the presence of a-FeOOH. The uniformly dispersing ultrafine a-FeOOH on the entire surface of CNNS could be further verified by the elemental mapping and EDS images (Fig. 8), where Fe, C, N and O elements were homogeneously distributed over the whole profile, demonstrating the successful coupling of a-FeOOH with CNNS to form heterostructures.
To evaluate the photocatalytic activities of a-FeOOH/CNNS composites under visible light irradiation, RhB was chosen as model pollutant for photocatalytic degradation (Fig. 9A). The adsorption of RhB on a-FeOOH/CNNS in the darkness reached equilibrium within 60 min (Figure S4) and negligible self-degradation was also observed for RhB under visible light irradiation. As expected, all a-FeOOH/CNNS composites exhibited higher photocatalytic activities than either a-FeOOH or CNNS by visible light irradiation with a sequence of a-FeOOH/CNNS-800 > a-FeOOH/CNNS-700 > a-FeOOH/CNNS-900 > a-FeOOH/CNNS-600 > a-FeOOH/CNNS-500 > a-FeOOH/CNNS-400 > a-FeOOH/CNNS-300 > CNNS, indicating the positive effect of CNNS contents to promote the photocatalytic activities of a-FeOOH/CNNS composites. Full degradation of RhB was observed within 240 min by visible light irradiation in the presence of a-FeOOH/CNNS-800 composite, illustrating the significantly improved photocatalytic activity of the ultrafine a-FeOOH NPs/CNNS composites. However, further increment of CNNS (a-FeOOH/CNNS-900) resulted in decreased photocatalytic activity, which may be attributed to the recombination of photogenerated electrons and holes to restrain the photocatalytic efficiency. Figure 10A shows the variation of the absorption spectra of RhB under visible light irradiation by using a-FeOOH/CNNS-800. The characteristic peak intensities of RhB gradually decreased by prolonging the irradiation time, and the adsorption peaks disappeared within ~240 min irradiation. The maximum absorption wavelength edisplayed a blue shift, which indicated that the de-ethylation process46. These corresponding optical photographs of RhB degradation using different photocatalysts under different irradiation times were collected and displayed in Fig. 10B.
(A) The photocatalytic activities of the as-prepared photocatalysts for the degradation of RhB under visible light irradiation; (B) The rate constants of the as-prepared photocatalysts for the degradation of RhB; (C) The photocatalytic activities of the as-prepared photocatalysts for the degradation of RhB, MB, MO under visible light irradiation; (D) The rate constants of the as-prepared photocatalysts for the degradation of RhB, MB, MO.
In order to further assess the mineralization of RhB in water, total organic carbon (TOC) was also monitored during the reaction process, and a-FeOOH/CNNS-800 was selected as the representative photocatalyst, and the result is shown in Figure S5. TOC of RhB is degraded by 55% after exposure to visible light irradiation for 240 min, indicating that the produced organic intermediates continue to decompose into inorganic species. It is verified that RhB is indeed photocatalytic degraded by a-FeOOH/CNNS-800 composite, rather than just decolorized by the light irradiation.
To investigate the photodegradation kinetics of RhB, the photodegradation data were further analyzed by the pseudo-first-order model. This analysis was deduced from the pseudo-first-order model in the following Eq.12, 20
K (min−1) is the rate constant, C 0 (mg/L) is the initial concentration of RhB, and C (mg/L) is the concentration of RhB at time t (min). The highest degradation rates for RhB (Fig. 9B) were calculated to be 0.0107 min−1 for a-FeOOH/CNNS-800, which were ~17.8, ~4.6, ~3.1, ~2.5, ~1.8, ~1.3 and ~1.5 times higher than those of CNNS (0.0006 min−1), a-FeOOH/CNNS-300 (0.0023 min−1), a-FeOOH/CNNS-400 (0.0034 min−1), a-FeOOH/CNNS-500 (0.0042 min−1), a-FeOOH/CNNS-600 (0.0057 min−1), a-FeOOH/CNNS-700 (0.008 min−1) and a-FeOOH/CNNS-900 (0.0071 min−1), respectively.
In order to test the photocatalytic activity of a-FeOOH/CNNS-800 for different pollutants (MO and MB), were chosen as model pollutants for photocatalytic degradation under visible light irradiation (Fig. 9C). As expected, a-FeOOH/CNNS-800 can also degrade MO (0.0015 min−1) and MB (0.0022 min−1) under visible light irradiation (Fig. 9D), indicating the positive effect of a-FeOOH to expand the universality of a-FeOOH/CNNS composites.
The excellent photocatalytic performance a-FeOOH/CNNS composites could be attributed to the synergistic interactions from the following aspects: (i) the ultrafine a-FeOOH NPs can shorten diffusion distance of photogenerated charge to the surface; (ii) for the synthesis of a-FeOOH/CNNS composites, CNNS is simply introduced into the reaction system and the a-FeOOH is tightly anchored by strong electrostatic interaction, which can ensure the stability of the nanocomposites; (iii) the three-dimensional layered structures can provide more reactive sites for the adsorption and degradation of organic pollutants18, 45, 47.
Recycling experiments were also performed on a-FeOOH/CNNS-800 to evaluate the stability of this photocatalyst (Fig. 11A). Within the same irradiation time (~240 min) more than ~95% RhB were degraded even after seven successive cycles, illustrating its high stability and great promise in practical applications.48
The organic pollutants can be effectively degraded by reactive species, including h+, ∙OH and ∙O2 −, which may vary for different photocatalysts due to their different band structures and phase compositions12. Therefore, to explore the mechanism of the high photocatalytic activity and to assess the contribution of the reactive species, trapping experiments of reactive species were conducted using ethylenediaminetetraacetate (EDTA-2Na) and isopropyle alcohol (IPA) as h+ and ∙OH scavenges, respectively49, 50. By adding two scavenges into the degradation solutions, the reactive species in the degradation process can be revealed, as illustrated in Fig. 11B for the degradation of RhB by a-FeOOH/CNNS-800. The addition of IPA and EDTA-2Na has a certain effect on the photocatalytic efficiency, indicating that the •OH and h+ species play a positive role in the photocatalytic degradation of RhB.
Based on experimental results and previous researches, Fig. 12 depicted a diagrammatic sketch for the energy band positions of CNNS and a-FeOOH6, 20, 51. The conduction bands of CNNS and a-FeOOH are −1.38 eV and 0.58 eV6, 18, 51, so the valence bands CNNS and a-FeOOH are 1.18 eV and 1.49 eV, respectively. Under visible light irradiation, CNNS and a-FeOOH could absorb visible light, leading to the excitation of e- to the conduction band (CB) and whilst keeping h+ in the valence bands (VB). For a-FeOOH/CNNS heterojunctions, the photogenerated e- on the CNNS CB could easily migrate to the CB of a-FeOOH while the photogenerated h+ in the VB of a-FeOOH could migrate to CNNS. That is to say, the appropriately aligned band edges of CNNS and a-FeOOH indicated that the migration of effective photogenerated charges could occur via the heterojunctions with strong interfacial coupling effect in the composite. The migration of photogenerated charges limited the transmission of photogenerated e- and h+ on different sides, which reduced the recombination rate of photogenerated electron-hole pairs and improved the abundance and stability of photogenerated charge in the composite. At the same time, the isolated photogenerated charge promoted the production of reactive oxidative species, i.e. h+ and •OH, which were responsible for degrading RhB. And the conclusion can be further confirmed by Fig. 11B. So the photocatalytic efficiency of the composite was improved.
Generally, higher specific surface areas and larger pore volumes of photocatalysts were favorable for photocatalytic reaction, which can provide abundant reaction sites3, 50. The nitrogen adsorption-desorption isotherms and the pore size distributions curves of pure CNNS and a-FeOOH/CNNS-800 are showed in Figure S6. The BET surface areas of pure CNNS and a-FeOOH/CNNS-800 are 90.59 and 90.68 m2/g respectively, which means that they have higher BET surface areas. At the same time, the pure CNNS and a-FeOOH/CNNS-800 also have some mesopores. Compared with pure CNNS, a-FeOOH/CNNS-800 has more mesopores, which is consistent with the degradation performance of the photocatalysts. Therefore, the pure CNNS and a-FeOOH/CNNS-800 can provide abundant reaction sites for photocatalytic reactions due to their higher specific surface areas and larger pore volumes.
In the field of photocatalysis, photo absorption induced electron–hole separation character is extremely important3. To further comprehend the separation and recombination of electron–hole pairs in pure CNNS and a-FeOOH/CNNS composites, the photocurrent test is carried out under visible light, as illustrated in Figure S7. As we all know, the higher the photocurrent, the higher electrons–holes separation efficiency, thus resulting in higher photocatalytic activity1,2,3. In this study, a-FeOOH/CNNS-800 composite shows the highest photocurrent intensity, which is more than 2 times than that of pure CNNS. This indicates that a-FeOOH/CNNS-800 composite has the lowest electrons and holes recombination rate. Thus we think a-FeOOH/CNNS composites may have very good photocatalyst performance.
In order to study the effect of a-FeOOH modification, photoluminescence (PL) spectral analysis was carried out to bright to light the migration, transfer and recombination processes of photo-generated electron–hole pairs in pure CNNS and a-FeOOH/CNNS composites2, 11. As shown in Figure S8, the main fluorescence emission peak is centered at 500 nm for CNNS. With the addition of a-FeOOH nanoparticles, the emission intensity of the PL spectra of a-FeOOH/CNNS composites is significantly lower than that of CNNS and the order of the a-FeOOH/CNNS composites is consistent with the photocatalytic performance. Therefore, the a-FeOOH/CNNS composites inhibit more effectively the recombination of photo-generated charge carriers to improve photocatalytic activity. This give further an evidence to support the above photocurrent response curves results.
Conclusions
In summary, a series of a-FeOOH/CNNS composites was successfully synthesized via a facile in-situ precipitation method. The hierarchically ultrathin g-C3N4 nanosheets not only provided a large surface area, but also performed as the support to form heterostructures. The as-synthesized a-FeOOH/CNNS-800 photocatalyst showed superior visible light photocatalytic activities than others, which could be ascribed to the synergetic effect between a-FeOOH and CNNS, including the maximum heterojunction interface with intimate contact, enhanced photogenerated charge separation efficiency, and fully exposed reactive sites as well as excellent visible light response in the composite. This work could give insights into the importance of rational design of heterojunction systems, and provide a potential method for the construction of efficient heterojunction photocatalysts with controllable sizes and space distributions.
References
Zhu, Z. et al. Construction of high-dispersed Ag/Fe3O4/g-C3N4 photocatalyst by selective photo-deposition and improved photocatalytic activity. Appl. Catal. B: Environ. 182, 115–122 (2016).
Ma, S. L., Zhan, S. H., Jia, Y. N., Shi, Q. & Zhou, Q. X. Enhanced disinfection application of Ag-modified g-C3N4 composite under visible light. Appl. Catal. B: Environ. 186, 77–87 (2016).
Ong, W.-J., Tan, L.-L., Ng, Y. H., Yong, S.-T. & Chai, S.-P. Graphitic carbon nitride (g-C3N4)-based photocatalysts for artificial photosynthesis and environmental remediation: are we a step closer to achieving sustainability? Chem. Rev. 116, 7159–7329 (2016).
Mamba, G. & Mishra, A. Graphitic carbon nitride (g-C3N4) nanocomposites: A new and exciting generation of visible light driven photocatalysts for environmental pollution remediation. Appl. Catal. B: Environ. 198, 347–377 (2016).
Zhao, W. et al. A novel ternary plasmonic photocatalyst: ultrathin g-C3N4 nanosheet hybrided by Ag/AgVO3 nanoribbons with enhanced visible-light photocatalytic performance. Appl. Catal. B: Environ. 165, 335–343 (2015).
Liu, Q., Guo, Y., Chen, Z., Zhang, Z. & Fang, X. Constructing a novel ternary Fe (III)/graphene/g-C3N4 composite photocatalyst with enhanced visible-light driven photocatalytic activity via interfacial charge transfer effect. Appl. Catal. B: Environ. 183, 231–241 (2016).
Katsumata, H., Sakai, T., Suzuki, T. & Kaneco, S. Highly efficient photocatalytic activity of g-C3N4/Ag3PO4 hybrid photocatalysts through Z-Scheme photocatalytic mechanism under visible light. Ind. Eng. Chem. Res. 53, 8018–8025 (2014).
Shan, W., Hu, Y., Bai, Z., Zheng, M. & Wei, C. In situ preparation of g-C3N4/bismuth-based oxide nanocomposites with enhanced photocatalytic activity. Appl. Catal. B: Environ. 188, 1–12 (2016).
Yao, Y. J. et al. Magnetic core-shell CuFe2O4@C3N4 hybrids for visible light photocatalysis of Orange II. J. Hazard. Mater. 297, 224–233 (2015).
Shi, H., Chen, G., Zhang, C. & Zou, Z. Polymeric g-C3N4 coupled with NaNbO3 nanowires toward enhanced photocatalytic reduction of CO2 into renewable fuel. ACS Catal. 4, 3637–3643 (2014).
Dai, K. et al. Sonication assisted preparation of graphene oxide/graphitic-C3N4 nanosheet hybrid with reinforced photocurrent for photocatalyst applications. Dalton Trans. 43, 6295–6299 (2014).
Christoforidis, K. C. et al. Synthesis and photocatalytic application of visible-light active β-Fe2O3/g-C3N4 hybrid nanocomposites. Appl. Catal. B: Environ. 187, 171–180 (2016).
Dong, F. et al. An advanced semimetal-organic Bi spheres-g-C3N4 nanohybrid with SPR-enhanced visible-light photocatalytic performance for NO purification. Environ. Sci. Technol. 49, 12432–12440 (2015).
Xiong, T., Cen, W. L., Zhang, Y. X. & Dong, F. Bridging the g-C3N4 Interlayers for Enhanced Photocatalysis. ACS Catal. 6, 2462–2472 (2016).
Ni, Z. L., Dong, F., Huang, H. W. & Zhang, Y. X. New insights into how Pd nanoparticles influence the photocatalytic oxidation and reduction ability of g-C3N4 nanosheets. Catal. Sci. Technol. 6, 6448–6458 (2016).
Wen, J. et al. Fabricating the Robust g-C3N4 Nanosheets/Carbons/NiS Multiple Heterojunctions for Enhanced Photocatalytic H2 Generation: An Insight into the Trifunctional Roles of Nanocarbons. ACS Sustain. Chem. Eng. 5, 2224–2236 (2017).
Yan, S., Lv, S., Li, Z. & Zou, Z. Organic–inorganic composite photocatalyst of g-C3N4 and TaON with improved visible light photocatalytic activities. Dalton Trans. 39, 1488–1491 (2010).
Zhang, S. et al. Hybrid 0D-2D nanoheterostructures: in-situ growth of amorphous silver silicates dots on g-C3N4 nanosheets for full spectrum photocatalysis. ACS Appl. Mater. Interfaces 8, 35138–35149 (2016).
Li, Y. F. et al. Ultrathin g-C3N4 nanosheets coupled with AgIO3 as highly efficient heterostructured photocatalysts for enhanced visible-light photocatalytic activity. Chem. Eur. J. 21, 17739–17747 (2015).
Zheng, Y., Zhang, Z. & Li, C. Beta-FeOOH-supported graphitic carbon nitride as an efficient visible light photocatalyst. J. Mol. Catal. A: Chem. 423, 463–471 (2016).
Lima, L. V. et al. Synergism between n-type WO3 and p-type δ-FeOOH semiconductors: high interfacial contacts and enhanced photocatalysis. Appl. Catal. B: Environ. 165, 579–588 (2015).
Liu, J., Zheng, M., Shi, X., Zeng, H. & Xia, H. Amorphous FeOOH quantum dots assembled mesoporous film anchored on graphene nanosheets with superior electrochemical performance for supercapacitors. Adv. Funct. Mater. 26, 919–930 (2016).
Chemelewski, W. D., Lee, H.-C., Lin, J.-F., Bard, A. J. & Mullins, C. B. Amorphous FeOOH oxygen evolution reaction catalyst for photoelectrochemical water splitting. J. Am. Chem. Soc. 136, 2843–2850 (2014).
Sun, Y. et al. Synthesis of amorphous FeOOH/reduced graphene oxide composite by infrared irradiation and its superior lithium storage performance. ACS Appl. Mater. Interfaces 5, 10145–10150 (2013).
Padhi, D. K. & Parida, K. Facile fabrication of α-FeOOH nanorod/RGO composite: a robust photocatalyst for reduction of Cr (VI) under visible light irradiation. J. Mater. Chem. A 2, 10300–10312 (2014).
Li, Y. & Zhang, F.-S. Catalytic oxidation of Methyl Orange by an amorphous FeOOH catalyst developed from a high iron-containing fly ash. Chem. Eng. J. 158, 148–153 (2010).
Sherman, D. M. Electronic structures of iron (III) and manganese (IV)(hydr) oxide minerals: Thermodynamics of photochemical reductive dissolution in aquatic environments. Geochim. Cosmochim. Ac. 69, 3249–3255 (2005).
Guo, H. B. & Barnard, A. S. Modeling the iron oxides and oxyhydroxides for the prediction of environmentally sensitive phase transformations. Phys. Rev. B 83(9), 094112 (2011).
Cheng, R. L. et al. One-step construction of FeOx modified g-C3N4 for largely enhanced visible-light photocatalytic hydrogen evolution. Carbon 101, 62–70 (2016).
Zhang, Z. Y., Huang, J. D., Zhang, M. Y., Yuan, L. & Dong, B. Ultrathin hexagonal SnS2 nanosheets coupled with g-C3N4 nanosheets as 2D/2D heterojunction photocatalysts toward high photocatalytic activity. Appl. Catal. B: Environ. 163, 298–305 (2015).
Pei, Z. et al. Texturing in situ: N, S-enriched hierarchically porous carbon as a highly active reversible oxygen electrocatalyst. Energ. Environ. Sci. (2017).
Yang, W. L. et al. Microwave-assisted synthesis of porous Ag2S-Ag hybrid nanotubes with high visible-light photocatalytic activity. Angew. Chem. Int. Edit. 51, 11501–11504 (2012).
Xia, J. X. et al. Construction of ultrathin C3N4/Bi4O5I2 layered nanojunctions via ionic liquid with enhanced photocatalytic performance and mechanism insight. Appl. Catal. B: Environ. 191, 235–245 (2016).
Hu, Y. et al. Carbon-coated CdS petalous nanostructures with enhanced photostability and photocatalytic activity. Angew. Chem. Int. Edit. 525, 636–5639 (2013).
Gao, X. H. et al. Formation of mesoporous heterostructured BiVO4/Bi2S3 hollow discoids with enhanced photoactivity. Angew. Chem. Int. Edit. 53, 5917–5921 (2014).
Liu, C. et al. Chlorine intercalation in graphitic carbon nitride for efficient photocatalysis. Appl. Catal. B: Environ. 203, 465–474 (2017).
Pei, Z. et al. Toward enhanced activity of a graphitic carbon nitride-based electrocatalyst in oxygen reduction and hydrogen evolution reactions via atomic sulfur doping. J. Mater. Chem. A 4, 12205–12211 (2016).
Ye, L., Liu, J., Jiang, Z., Peng, T. & Zan, L. Facets coupling of BiOBr-g-C3N4 composite photocatalyst for enhanced visible-light-driven photocatalytic activity. Appl. Catal. B: Environ. 142, 1–7 (2013).
Wan, W., Yu, S., Dong, F., Zhang, Q. & Zhou, Y. Efficient C3N4/graphene oxide macroscopic aerogel visible-light photocatalyst. J. Mater. Chem. A 4, 7823–7829 (2016).
Zhu, T., Ong, W. L., Zhu, L. & Ho, G. W. TiO2 fibers supported β-FeOOH nanostructures as efficient visible light photocatalyst and room temperature sensor. Sci. Rep-uk. 5, 10601 (2014).
Feng, J. X. et al. FeOOH/Co/FeOOH hybrid nanotube arrays as high-performance electrocatalysts for the oxygen evolution reaction. Angew. Chem. Int. Edit. 55, 3694–3698 (2016).
Xu, J. et al. Large scale preparation of Cu-doped a-FeOOH nanoflowers and their photo-Fenton-like catalytic degradation of diclofenac sodium. Chem. Eng. J. 291, 174–183 (2016).
Huang, J., Ding, Y., Luo, X. & Feng, Y. Solvation effect promoted formation of p–n junction between WO3 and FeOOH: A high performance photoanode for water oxidation. J. Catal. 333, 200–206 (2016).
Zhang, S. et al. Visible‐light photocatalytic degradation of methylene blue using SnO2/α‐Fe2O3 hierarchical nanoheterostructures. ChemPlusChem 78, 192–199 (2013).
McIntyre, N. & Zetaruk, D. X-ray photoelectron spectroscopic studies of iron oxides. Anal. Chem. 49, 1521–1529 (1977).
Chen, Y. et al. Exploring the different photocatalytic performance for dye degradations over hexagonal ZnIn2S4 microspheres and cubic ZnIn2S4 nanoparticles. ACS Appl. Mater. Interfaces 4, 2273–2279 (2012).
Shao, P. H., Tian, J. Y., Shi, W. X., Gao, S. S. & Cui, F. Y. Eco-friendly one-pot synthesis of ultradispersed TiO2 nanocrystals/graphene nanocomposites with high photocatalytic activity for dye degradation. J. Mater. Chem. A 3, 19913–19919 (2015).
Dong, F., Xiong, T., Sun, Y. J., Zhang, Y. X. & Zhou, Y. Controlling interfacial contact and exposed facets for enhancing photocatalysis via 2D-2D heterostructures. Chem. Commun. 51, 8249–8252 (2015).
Liu, L. et al. A stable Ag3PO4@g-C3N4 hybrid core@shell composite with enhanced visible light photocatalytic degradation. Appl. Catal. B: Environ. 183, 133–141 (2016).
Zhang, Z. Y., Jiang, D. L., Li, D., He, M. Q. & Chen, M. Construction of SnNb2O6 nanosheet/g-C3N4 nanosheet two-dimensional heterostructures with improved photocatalytic activity: Synergistic effect and mechanism insight. Appl. Catal. B:Environ. 183, 113–123 (2016).
Gao, B., Liu, L., Liu, J. & Yang, F. Photocatalytic degradation of 2, 4, 6-tribromophenol on Fe2O3 or FeOOH doped ZnIn2S4 heterostructure: Insight into degradation mechanism. Appl. Catal. B: Environ. 147, 929–939 (2014).
Acknowledgements
Financial supports from National Natural Science Foundation of China (Grant Nos 51672109 and 21505050), Natural Science Foundation of Shandong Province for Excellent Young Scholars (Grant No. ZR2016JL015), and the Natural Science Foundation of Shandong Provience (Grant No. ZR2016FM30).
Author information
Authors and Affiliations
Contributions
Y.H.C. and Z.S.W. designed the experiments. C.R.Y. performed the experiments. D.X.L. performed the SEM observations. L.Z.P. performed TEM observations. Z.S.W. and X.X.J. discussed and commented on the experiments and results, and wrote the paper.
Corresponding authors
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Yang, H., Zhang, S., Cao, R. et al. Constructing the novel ultrafine amorphous iron oxyhydroxide/g-C3N4 nanosheets heterojunctions for highly improved photocatalytic performance. Sci Rep 7, 8686 (2017). https://doi.org/10.1038/s41598-017-09283-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-09283-1
This article is cited by
-
Photoelectrocatalytic hydrogen evolution and synchronous degradation of organic pollutants by pg-C3N4/β-FeOOH S-scheme heterojunction
Science China Technological Sciences (2024)
-
Small molecules derived carbon dots: synthesis and applications in sensing, catalysis, imaging, and biomedicine
Journal of Nanobiotechnology (2019)
-
Influence of Cu + g-C3N4 incorporation on the photocatalytic dye decomposition of ZnO film coated on stainless steel wire meshes
Journal of Materials Science: Materials in Electronics (2019)
-
Fabrication of Hierarchical ZnO@NiO Core–Shell Heterostructures for Improved Photocatalytic Performance
Nanoscale Research Letters (2018)
-
Fabrication of Ag-modified porous ZnMgO nanorods with enhanced photocatalytic performance
Journal of Materials Science: Materials in Electronics (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.