Abstract
Rodent models of maternal obesity have been associated with kidney damage and dysfunction in offspring. However, the underlying mechanisms are yet to be elucidated. In this study, female rats were fed a high-fat diet (HFD) for 6 weeks prior to mating, throughout gestation and lactation; both male and female offspring were examined at weaning. Our results demonstrate that renal lipid deposition was increased in male offspring only, which is associated with reduced protein expression of Sirtuin (SIRT) 1, an essential regulator of lipid metabolism and stress response. Other components in its signalling network including phosphorylated 5′-AMP-activated protein kinase (pAMPKα), Forkhead box FOXO3a and Peroxisome proliferator-activated receptor (PPAR)γ coactivator 1-alpha (PGC-1α) were also downregulated. By contrast, in female offspring, renal fat/lipid distribution was unchanged in coupling with normal SIRT1 regulation. Specific autophagy and antioxidant markers were suppressed in both sexes. On the other hand, fibronectin and Collagen type IV protein expression was significantly higher in the offspring born HFD-fed dams, particularly in the males. Collectively, these findings suggest that maternal HFD consumption can induce sex-specific changes in offspring kidney lipid metabolism and stress responses at early ages, which may underpin the risk of kidney diseases later in life.
Similar content being viewed by others
Introduction
Chronic kidney disease (CKD) is a global health crisis, characterised by a gradual loss of kidney function and ultimately requiring renal replacement therapy. Up to 90% loss of kidney function can occur before any symptoms are perceived1. There is increasing recognition that a predisposition to CKD is evident in children and young adults, although overt renal failure may not manifest till older age is reached2. Such phenomenon has been attributed in part to “fetal programming”, a term for fetal metabolic and epigenetic alterations induced by an abnormal intrauterine environment3,4,5. Maternal obesity during pregnancy is one of the factors that can induce fetal programming, leading to increased body weight and adiposity in the offspring6, 7, and pathophysiological changes in the kidney in adulthood8. Oxidative stress and inflammation have been identified as two main factors contributing to such outcomes9. Nevertheless, the changes are often detected at later stages when other permanent damages may have already occurred9. Therefore, it is important to identify earlier targets for a preventative therapy.
Lipid metabolism and autophagy are among the earliest possible mechanisms that can be affected by maternal obesity considering their direct relevance to energy intake and dietary composition. Indeed, perirenal fat and intrarenal lipid accumulation can be used as predicting factors for renal fibrosis and CKD10, 11. On the other hand, autophagy is negatively regulated by nutrition availability, which functions to dispose of and recycle damaged and dysfunctional cellular components that otherwise accumulate and interfere with cellular homeostasis and activities. Prolonged deficiency in autophagy for example in obesity and aging increase renal susceptibility to glomerular and tubulointerstitial pathology12, 13. Apart from the two pathways, antioxidant shortage is a direct cause of oxidative imbalance that can result in oxidative damage and cellular dysfunction in the offspring.
Sirtuins (SIRTs) are a group of d NAD+-dependent deacetylases and mono-ADP-ribosyl transferases. In mammals, seven members of the family (SIRT1–7) have been identified, among which SIRT1 is the most extensively studied, having pivotal roles in multiple processes especially stress responses and glucose/fat metabolism14. Its expression and activities are known to be promoted during nutrient deprivation15 and suppressed when nutrient is in excess, such as in obese individuals16. With regard to the kidney, activation of the SIRT1 signalling pathway has been shown to reduce renal lipotoxicity17, improve renal autophagy18 and antioxidant defence19, thereby attenuate kidney diseases in obese and diabetic animals.
In models of maternal obesity, it has been shown that SIRT1 expression is reduced in the fetus20 and specifically in the offspring liver21, suggesting a potential role of SIRT1 as a mediator of the programming effects by maternal obesity22. Nevertheless, the regulation of renal SIRT1 signalling in the offspring has yet to be demonstrated. In this study, we examine the hypothesis that maternal obesity induces renal lipid metabolic disorders and autophagy deficiency in the offspring and SIRT1 signalling plays a central role in this mechanism. As the risk of CKD differs between sexes23 and so does the fetal programming of kidney disorders24,25,26,27, we also examined whether these mechanisms are regulated in a sex-specific manner.
Results
Maternal HFD consumption increased body weight, fat deposition, plasma glycerides and kidney weight in both male and female offspring
At weaning, HFD-fed dams had significantly higher body weight, retroperitoneal fat mass and liver weight compared to chow-fed dams (P < 0.05, Table 1). Both male and female offspring of the HFD-fed dams showed significantly greater body weight than those born to chow-fed dams (P < 0.05, Table 2). Moreover, female offspring were significantly smaller than males (P < 0.05, sex effect). Mesenteric, retroperitoneal and perigonadal fat mass in both MHF male and female offspring was significantly higher in the offspring from HFD-fed dams (P < 0.05). The difference remained significant when adjusted for body weight (P < 0.01) except for male offspring’s mesenteric fat. Similar to the body weight, the overall percentage of retroperitoneal fat in female offspring was significantly smaller than in the males (P < 0.05, sex effect). The net liver weight was significantly increased by MHF only in female offspring. The sex effect for liver weight was significant (P < 0.05, sex effect), as well as the interaction between sex and maternal diet (P < 0.05, interaction). Kidney weight was also significantly increased in the offspring (P < 0.05 in male and P < 0.01 in female respectively). Overall, the females have lighter kidneys than the males. Consistent with the increased body weight and adiposity, plasma triglyceride levels were also increased in both sexes in the offspring (P < 0.05 and P < 0.01 respectively), whereas the levels of non-esterified fatty acid were unchanged.
Maternal HFD consumption increased perirenal fat deposition and renal triglyceride concentration in male but not female offspring
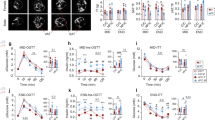
Male offspring from HFD-fed dams demonstrated increased perirenal fat deposition (Fig. 1), which was unchanged in female offspring. Similarly, kidney levels of triglycerides in male offspring were significantly higher when born to dams fed a HFD (P < 0.01), whereas in female offspring, no difference was observed between the groups. The sex-specific effect is reflected by a significant interaction (P < 0.05) between the two factors: sex and maternal diet.
Maternal high-fat diet increases perirenal fat deposition and triglyceride concentration in male but not female offspring. MC (offspring of dams fed chow); MHF (offspring of dams fed high-fat diet). Results are expressed as mean ± SD. Data were analysed by two-way ANOVA followed by Bonferroni post hoc test. (a) (P < 0.05, Maternal diet effect), (b) (P < 0.5, Sex effect), (c) (P < 0.05, interaction effect), **P < 0.01 (vs MC offspring). n = 5.
The effects of maternal HFD consumption on lipid metabolism regulators in offspring kidney
As kidney triglyceride levels were increased in the male offspring only, we examined the mRNA expression of potential regulator(s) involved in lipid metabolism. As shown in Table 3, only mRNA expression of SIRT1 and PGC-1α was significantly downregulated in the male offspring due to maternal HFD consumption (P < 0.01 and P < 0.05 respectively). In female offspring, no differences at the transcriptional level were detected. The sex-specific regulation of SIRT1 mRNA is reflected by a significant interaction (P < 0.05) between the two factors of sex and maternal diet.
Together with SIRT1 and PGC-1α, PPARα and PPARγ are also essential regulators of lipid metabolism. In this study, we showed that the mRNA expression of PPARα in the offspring kidney were generally lower in females (P < 0.05, sex effect, Table 3). By a sharp contrast, the levels of PPARγ were significantly higher in the females (P < 0.05, sex effect). The effect of maternal HFD consumption on the transcription of the two genes was minor except a slight tendency of downregulation of PPARα in the MHF males (Table 3).
Similar to PPARγ, the overall mRNA expression of FABP3 was also significantly higher in the females (P < 0.05, sex effect) and unchanged by maternal HFD consumption. No difference in the renal expression of other regulators of the lipogenesis pathway including SREBP1c and FAS were detected.
Maternal HFD consumption regulates renal SIRT1 signalling network in the offspring in a sex-specific manner
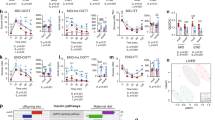
To confirm our observations, the protein expression of SIRT1 and other markers of its signalling pathway were analysed. Our results demonstrated that SIRT1 protein level was indeed reduced in the kidney of MHF male offspring (P < 0.05, Fig. 2), whereas in the females, no change was detected. The level of total AMPK, a positive regulator of the SIRT1 signalling network, was substantially reduced by approximately 50% (P = 0.052) in the MHF males. More importantly, the level of its active phosphorylated form pAMPK was also significantly suppressed correspondingly (P < 0.05). In the female offspring, the renal levels of AMPK and pAMPK were both unaffected. The sex-maternal diet interactions were near-significant (P = 0.07) and significant (P < 0.05) for SIRT1 and pAMPK respectively.
Protein levels of markers of SIRT1 signalling network in male and female offspring kidney. MC (offspring of dams fed chow); MHF (offspring of dams fed high-fat diet). SIRT1 (Sirtuin), PGC-1α (Peroxisome proliferator-activated receptor gamma coactivator 1-alpha), AMPKα (5′ AMP-activated protein kinase alpha), pAMPKα (Phosphorylated AMPKα) and FOXO3a (Forkhead box O3a). Results are expressed as mean ± SD. Data were analysed by two-way ANOVA followed by Bonferroni post hoc test. (a) (P < 0.05, Maternal diet effect), (b) (P < 0.5, Sex effect), (c) (P < 0.05, interaction effect), *P < 0.05, **P < 0.01 (vs MC offspring). n = 6.
Similarly, the expression of FOXO3a, a downstream regulator of SIRT1, was also regulated in such interactive fashion (P < 0.05) with a near-significant reduction in MHF male offspring (P = 0.053). Interestingly, despite the unchanged mRNA expression in MHF females, the protein levels of PGC-1α were significantly reduced by maternal HFD consumption in both sexes (P < 0.001 and P < 0.05 respectively, Fig. 2).
SIRT1 is known as a suppressor of PPARγ transcription28. However, in this study, the expression of PPARγ seems to be independent of SIRT1 signalling. The protein expression of both PPARγ isoforms 1 and 2 were substantially reduced (P = 0.059 and P < 0.01 respectively, Fig. 3) in MHF female offspring kidney. However, in the male offspring, no change was detected. The sex-specific effect is reflected by a significant interaction between the two factors of sex and maternal diet for the regulation of PPARγ2 (P < 0.05).
Protein levels of renal Peroxisome proliferator-activated receptor gamma isoform 1 and 2 in male and female offspring. MC (offspring of dams fed chow); MHF (offspring of dams fed high-fat diet). Results are expressed as mean ± SD. Data were analysed by two-way ANOVA followed by Bonferroni post hoc test. (a) (P < 0.05, Maternal diet effect), (c) (P < 0.05, interaction effect), **P < 0.01 (vs MC offspring). n = 6.
Maternal HFD consumption downregulates renal autophagy markers
Autophagy can be regulated by SIRT1 in multiple ways: either through the action of FOXO3a to upregulate the transcription of autophagy-related genes29, or by direct deacetylation of Atg proteins thereby increasing their activity30. Our results indicate that maternal HFD consumption has significant impacts on the renal expression of essential autophagy markers including Beclin-1, Atg12-Atg5 complex, LC3-I, LC3-II and p62 (P < 0.05, maternal diet effect, Fig. 4). Notably, while Beclin-1, LC3-I and II were suppressed to the same levels in both sexes, Atg12-Atg5 and p62 were regulated in a sex-specific fashion with significant reductions of the former in the female (P < 0.001) and the latter in the male offspring (P < 0.05) respectively. There was also a trend of reduction in the expression of Atg7 only in the females (Fig. 4).
Regulation of renal autophagy markers in male and female offspring. MC (offspring of dams fed chow); MHF (offspring of dams fed high-fat diet). Atg (Autophagy-related protein); LC3-I (Microtubule-associated proteins 1A/1B light chain 3), LC3-II (lipidated LC3); p62 (Sequestome 1). Results are expressed as mean ± SD. Data were analysed by two-way ANOVA followed by Bonferroni post hoc test. (a) (P < 0.05, Maternal diet effect), (b) (P < 0.5, Sex effect), (c) (P < 0.05, interaction effect), *P < 0.05, ***P < 0.001 (vs MC offspring). n = 6.
Maternal HFD consumption reduces renal levels of antioxidant marker GPx-1
SIRT1 signalling has been associated with cellular antioxidant defence. MnSOD and GPx-1 were examined as representative markers of antioxidant defence, each of which is involved in one of the two-step reaction to convert reactive oxygen species into water and oxygen (O− 2 → H2O2 → H2O + O2). As shown in Fig. 5, while the renal levels of MnSOD were not affected by maternal HFD consumption, GPx-1 proteins were significantly and consistently reduced in the offspring in both sexes (P < 0.01).
Regulation of renal antioxidant markers in male and female offspring. MC (offspring of dams fed chow); MHF (offspring of dams fed high-fat diet). MnSOD (Manganese superoxide dismutase); GPx1 (Glutathione Peroxidase 1). Results are expressed as mean ± SD. Data were analysed by two-way ANOVA followed by Bonferroni post hoc test. a(P < 0.05, Maternal diet effect), **P < 0.01 (vs MC offspring). n = 6.
The effects of maternal HFD consumption on renal markers of inflammation and extracellular matrix
Obesity is often associated with a low-grade inflammation31. Particularly in the kidney, it is associated with the elevation of TGFβ1 as well as structural modifications including glomerulosclerosis and interstitial fibrosis32, 33. In this study, TGFβ1 mRNA expression in the male offspring kidney was not affected by maternal HFD consumption. Higher levels of TGFβ1 were detected in the females (P < 0.05, sex effect, Table 4). By contrast, the renal expression of TGFβ receptor 1 was reduced in the kidney of MHF offspring (P < 0.05, maternal diet effect) and not different between the two sexes. Similarly, Col3A mRNA expression was gender-dependent (P < 0.05, sex effect), whereas Col1A was reduced by maternal HFD consumption (P < 0.05, maternal diet effect). Fibronectin mRNA expression showed a trend of increase by maternal HFD consumption (Table 4). In addition, immunohistochemistry staining also indicated an overall elevation of fibronectin protein in the offspring born to HFD-fed dams (Fig. 6B, P < 0.05, maternal diet effect). The renal protein level of Col4 was also significantly increased in MHF offspring (Fig. 6C, P < 0.05, maternal diet effect), especially in the males (P < 0.05).
Renal histological staining in male and female offspring. (A) Picro Sirius Red staining of collagen type I and III. (B) Fibronectin immunohistochemistry. (C) Collagen type IV immunohistochemistry. MC (offspring of dams fed chow); MHF (offspring of dams fed high-fat diet). FN (Fibronectin); Col4 (Collagen type IV). Results are expressed as mean ± SD. Data were analysed by two-way ANOVA followed by Bonferroni post hoc test. a(P < 0.05, Maternal diet effect), *P < 0.05 (vs MC offspring). n = 5.
Discussion
Maternal high-fat diet consumption and obesity have been associated with kidney damage and dysfunction in the offspring in adulthood8, 9, which are likely the result of accumulative cellular/molecular events occurred during early developmental periods. In this study, we attempted to clarify the underlying mechanisms. We demonstrate that maternal overnutrition is associated with increased lipid deposition and the suppression of multiple stress responses including autophagy, antioxidant defence and inflammation in the offspring kidney with a significant relevance to the SIRT1 signalling network. In addition, the study also suggests that many of these early changes are sex-dependent.
Consistent with previous studies6,7,8, 34, maternal HFD consumption preconception, during gestation and laction significantly increased ofsspring body weight, fat deposition, plasma triglyceride level and kidney weight at weaning, suggesting increased caloric intake and cell proliferation. Male offspring showed higher body weight and fat mass compared to females, which is also in line with previous studies in rodents26 and human35, suggesting a higher tendency of males for obesity development. In addition, the fact that maternal HFD consumption significantly increased the liver weight of female but not male offsrping may imply a higher capacity of female liver for growth and perhaps also lipid storage and processing, which in turn may reduce lipid spillover to other tissues such as kidney.
Indeed, the levels of perirenal fat as well as intrarenal lipid content were higher only in MHF males but not females, suggesting the effects of maternal HFD consumption on offspring kidney are also sex-specific. Sex-dependent differences in fetal programming of nephropathy are well-established in maternal undernutritional conditions24, 26, 27 and to a lesser extend maternal overnutrition, for instance, a high-protein diet25. Increased intrarenal lipid content has been associated with glomerulopathy and tubulointerstitial sclerosis in obese patients10, while perirenal lipid deposition has also been suggested as an independent predictor of CKD11. Such early changes in lipid metabolism are likely to induce lipotoxicity and predispose to kidney dysfunction later in life, especially when a second insult to the kidney occurs, such as the development of diabetes or obesity in the offspring5, 8.
SIRT1 and its downstream factor PGC-1α are two stimuli of fatty acid oxidation (52, 53). As a consequence of maternal HFD consumption, renal mRNA and protein levels of SIRT1 and PGC-1α levels were reduced specifically in the male offspring, which may explain the male-specific elevation of renal fat. Together with SIRT1 and PGC-1α, the protein levels of phosphorylated AMPK was also downregulated. AMPK can indirectly activate SIRT1 by increasing the NAD+/NADH ratio through the upregulation of Nicotinamide phosphoribosyltransferase36. Conversely, SIRT1 is also required for AMPK activity37. As renal AMPK has been shown to be reduced in obesity and diabetes17, 38, its reduced expression and activation due to maternal obesity is consistent with the downregulation of SIRT1 signalling and increased lipid deposition in the male offspring kidney. Both SIRT1 and AMPK are activators of FOXO3a, while FOXO3a can also promote SIRT1 transcription by interfering with the suppressor p53 on the SIRT1 promoter39. As such, a positive feedback loop is formed within the SIRT1 signalling network.
In MHF female offspring, there was no change in renal expression of SIRT1, pAMPK or FOXO3a. Alternatively, the protein level of PPARγ2 was significantly reduced. As PGC-1α is a co-activator of PPARγ, its reduced level in MHF females supports downregulated PPARγ signalling. Although PPARγ is well-known as an indispensable controller of adipocyte differentiation and insulin sensitivity40, its role in the kidney is not clearly understood. It has been shown that PPARγ+/− mice showed resistance to obesity-associated kidney lipid accumulation and injuries41. The reduction of renal PPARγ2 reduction in MHF female offspring may reflect an adaptive response to the increased fatty acid influx due to maternal HFD consumption. The regulation of PPAR signalling by maternal overnutrition has been previously reported in a transcriptome study42.
In this study, the protein levels of multiple autophagy markers were significantly reduced in the offspring kidney, suggesting suppression of autophagic processes. Hence, accumulation of misfolded proteins and dysfunctional organelles such as malfunctioning mitochondria is likely to occur43. Although autophagy was suppressed by maternal HFD consumption in both sexes, there were sex-dependent differences in the regulation of individual markers. The fact that the reduction of Atg12-Atg5 complex only occurred in the MHF females suggests that autophagosome elongation was likely to be impaired. On the other hand, p62 expression was only affected in the MHF males, indicating autophagosome degradation was interfered specifically in MHF male offspring. Prolonged deficiency in autophagy can increase renal susceptibility to glomerular and tubulointerstitial pathology12, 13.
Antioxidant defence systems have a close relationship with autophagy as both are essentially involved in the adaptive responses to oxidative stress44, 45. In this study, together with autophagy, renal antioxidant defence was also suppressed, reflected in the marked reduction in GPx-1 levels. The fact that the expression of MnSOD was unchanged indicates that the impairment of antioxidant defence was only partial and may not necessarily lead to oxidative stress at such young neonatal age. However, there is a certainly higher risk for oxidative stress and inflammation to occur in the offspring kidney if the defects is prolonged. Indeed, our group has recently demonstrated that maternal obesity can exacerbate kidney oxidative stress, inflammation and fibrosis induced by type 1 diabetes in the offspring5.
It has been shown that the SIRT1/AMPK/FOXO3a axis positively regulates autophagy43, 46, 47 and antioxidant defence48, 49, thereby preventing oxidative damage, particularly in kidney tubular cells50, 51. As such, the reduction of these markers in male MHF offspring kidney is likely to underline the deficiency of kidney autophagy and antioxidant defence markers. In female offspring, as the renal levels of SIRT1, pAMPK and FOXO3a were normal, the alterations in autophagy and antioxidant defence are likely to be mediated through a different pathway, for example the PPARγ/PGC-1α pathway48, 52. Further investigations in female offspring are required to confirm this hypothesis.
In the study, the renal mRNA expression of TGFβ1, Col3A and FN in the MHF offspring was unchanged, while TGFβR1 and Col1A mRNA expression was moderately decreased. On the other hand, fibronectin and Col4 protein level was significantly upregulated in MHF offspring, suggesting that maternal HFD consumption can induce renal fibrogenesis in the offspring at weaning. Such effect has been shown to extend to adulthood9. Interestingly, only in the male offspring was the Col4 protein expression significantly upregulated. This result further supports higher levels of kidney disorders and damage in male offspring than the female offspring caused by maternal HFD consumption, which is likely linked to increased renal lipid distribution and reduced SIRT1 signalling in the male kidney. It has been demonstrated that SIRT1 activation can suppress renal fibrogenic signalling in vitro and in vivo 53. Whether such therapy is applicable in the offspring at early age to attenuate or reverse the programming effects of maternal HFD consumption on kidney structure requires further investigations.
In summary, the current study demonstrates that maternal HFD consumption can increase renal fat deposition in male offspring and impair renal autophagy and antioxidant defences. Importantly, there are sex-specific differences in the magnitude of effects in the regulatory pathways. In male offspring there is downregulation of the SIRT1/AMPK/FOXO3a/PGC-1α regulatory network; whereas in females, downregulation of PPARγ/PGC-1α is evident. The more marked abnormalities in the male offspring may underpin the well documented increased risk of CKD in males than females in adulthood23. Such significant sex differences emphasize the need to independently study both sexes and to employ sex-specific approaches to overcome the adverse effects of maternal obesity on the development of CKD in the offspring. Further investigations with a longitudinal observation targeting SIRT1 signalling pathway are required to confirm sex specificity and the implication of SIRT1 regulation in the development of CKD in the context of maternal obesity.
Methods
Animals
The study was approved by the Animal Care and Ethics Committee of the University of Technology Sydney (ACEC# 2009–350). All methods were performed in accordance with the relevant guidelines and regulations in the Australian Code of Practice for the Care and Use of Animals for Scientific Purposes. Female Sprague-Dawley rats (8 weeks) were fed HFD (HFD, 20 kJ/g, 43.5% calorie as fat, Specialty Feed, WA, Australia) or standard rodent chow (11 kJ/g, 14% calorie as fat, Gordon’s Speciality Stockfeeds, NSW, Australia) for 6 weeks before mating, throughout gestation and lactation54. On postnatal day (P) 1 the litter size was adjusted to 10 pups/litter (sex ratio controlled at approximately 1:1). Both male and female offspring were investigated. At weaning (P20), all pups were sacrificed after overnight fasting. Blood was collected via cardiac puncture after anaesthesia (Pentothal, 0.1mg/g, i.p., Abbott Australasia Pty Ltd, NSW, Australia). Fat tissues and liver were weighed. The whole kidney was dissected, snap frozen and stored at −80 °C for later analysis. For each type of analysis, 5–6 renal tissues/group were randomly selected.
Kidney histology
Kidneys were fixed in 10% formalin for 36-h and embedded in paraffin. Paraffin sections were prepared at 4 μm thickness and mount on microscope slides. The sections were stained with hematoxylin and eosin (H&E) for general renal structure and perirenal fat visualisation. Immunohistochemistry (IHC) staining was performed as previously described55. Briefly, the tissue sections underwent dewaxing (with xylene), rehydration (with ethanol), antigen-retrieval (99 °C for 20 min in 0.01M, pH 6.0 citric buffer), washing (with water), endogenous peroxidase deactivation with 3% H2O2 (Sigma-Aldrich, Dublin, Ireland), blocking (with Protein Block Serum-Free, Dako, Glostrup, Denmark), incubation with primary antibodies against fibronectin (1:1000, Abcam, Cambridge, UK) and collagen IV (1:1000, Abcam, Cambridge, UK) followed by biotinylated secondary anti-rabbit IgG antibodies and finally horseradish peroxidase (HRP)-conjugated streptavidin (Dako, Glostrup, Denmark). The tissue specimens were examined by brightfield microscopy (Olympus, Japan). Six consecutive non-overlapping fields from each section of renal cortex were photographed under high magnification. Image J (National Institutes of Health, USA) was used for estimation of the specific staining area. IHC score was determined by log transformation of the staining area.
Renal protein and lipid extraction
The kidney was homogenized in Triton X-100 lysis buffer (pH 7.4, 150 mM NaOH, 50 mM Tris-HCl, 1% Triton X-100, Roche protease inhibitor) using TissueRuptor (Qiagen, Hilden, Germany). A part of the homogenate was used for lipid extraction according to a published protocol56. The lipid extract of each sample was then added to a 96-well plate, followed by incubation with Roche triglyceride reagent GPO-PAP (Roche Life Science, NSW, Australia) at 37 °C for 20 min. The absorbance was read at 490 nm. Glycerine was used in constructing the standard curve.
The remaining tissue homogenate was centrifuged at 10,000 g for 15 min. The supernatant containing total protein was collected and quantitated for protein concentration using Pierce BCA Protein Assay Kit (Thermo Scientific, VIC, Australia) according to the manufacturer’s instructions. The protein concentration of all samples was standardised as 5 μg/μl. The samples were stored at −80 °C for further analyses.
Quantitative real time PCR (qRT-PCR)
Total RNA was isolated from the kidney tissue using RNeasy Plus Mini Kit (Qiagen Pty Ltd, CA, USA) according to the manufacturer’s instructions. The purified total RNA was used as a template to generate first-strand cDNA using the First Strand cDNA Synthesis Kit (Roche Life Science, NSW, Australia). The amplicons of target genes were amplified with SYBR Green probes. Primer sequences were: SIRT1 (Forward: GCA GGT TGC GGG AAT CCA A, Reverse: GGC AAG ATG CTG TTG CAA A); PPARγ (Forward: 5′-ATC TAC ACG ATG CTG GC-3′, Reverse: 5′-GGA TGT CCT CGA TGG G-3′); PPARα (F: 5′-AGA CAC CCT CTC TCC AGC TTC-3′, R: 5′-GAA TCT TGC AGC TTC GAT CAC-3′); PGC-1α (Forward: AAA CTT GCT AGC GGT CCT CA, Reverse: TGG CTG GTG CCA GTA AGA G-3′); Fatty acid synthase (FAS, Forward: 5′-TGC TCC CAG CTG CAG GC-3′, Reverse: 5′-GCC CGG TAG CTC TGG GTG TA-3′); Sterol regulatory element-binding protein (SREBP)−1c (Forward: 5′-GGA GCC ATG GAT TGC ACA TT-3′, Reverse: 5′-GCT TCC AGA GAG GAG GCC AG-3′), Transforming growth factor beta (TGFβ1, Forward: 5′-TCA GAC ATT CGG GAA GCA GT-3′, Reverse: 5′-ACG CCA GGA ATT GTT GCT AT-3′), TGFβ receptor 1 (TGFβR1, Forward: 5′-CAG CTC CTC ATC GTG TTG G-3′, Reverse: CAG AGG TGG CAG AAA CAC TG-3′), Collagen (Col)1A (Forward: 5′-CAT GTT CAG CTT TGT GGA CCT-3′, Reverse: 5′-GCA GCT GAC TTC AGG GAT GT-3′), Col3A (Forward: 5′-TCC CCT GGA ATC TGT GAA TC-3′, Reverse: 5′- TGA GTC GAA TTG GGG AGA AT-3′), Fibronectin (FN, Forward: 5′-CGG AGA GAG TGC CCC TAC TA-3′, Reverse: 5′-CGA TAT TGG TGA ATC GCA GA-3′) (). Gene expression was standardized to β-actin mRNA (Forward: 5′-ATC GTG CGT GAC ATT AAG; Reverse: 5′-ATT GCC AAT GGT GAT GAC). A sample of the MC-male group was assigned as a calibrator against which all other samples were expressed as fold difference.
Western-Blot
The proteins were electrophoresed and electroblotted onto the Hybond nitrocellulose membrane (Amersham Pharmacia Biotech, Amersham, UK), which was then incubated with a primary antibody at 4 °C overnight. Antibodies against 5′ AMP-activated protein kinase alpha AMPKα (1:1000), phosphorylated AMPKα (pAMPKα, 1:1000), Autophagy related gene (Atg)6/Beclin-1 (1:1000), Atg12-Atg5 complex (1:2000), Atg7 (1:2000), Atg8/LC3 (1:2000) were obtained from Cell Signalling (MA, USA). FOXO3a (1:2000 LSBio, WA, USA). SIRT1 and Peroxisome proliferator-activated receptor gamma (PPARγ) antibodies were obtained from Santacruz (1:2000, CA, USA). PPARγ coactivator-1 alpha (PGC-1α, 1:2000) and p62 (1:2000) were obtained from Novus Biologicals (VIC, Australia). The anti-oxidative markers manganese superoxide dismutase (MnSOD, 1:2000, Merck Millipore, MA, USA) and Glutathione Peroxidase 1 (GPx-1, 1:200, R&D System, MN, USA) were examined. β-actin (1:2000, Santacruz, CA, USA) was used as the housekeeping protein. All primary antibodies were derived from rabbit except GPx-1 and β-actin antibodies, which were goat-derived. Subsequently the membrane was incubated with a horseradish peroxidase-conjugated secondary antibody (donkey anti-goat for β-actin, otherwise goat anti-rabbit). The immunoblots were developed by adding the Luminata Western HRP Substrates (Millipore, MA, USA) to the membrane and exposed for an appropriate duration using ImageQuant LAS 4000 (Fujifilm, Tokyo, Japan). ImageJ (National Institutes of Health, USA) was used for densitometric analyses.
Statistical analysis
The results are expressed as mean ± SD and analysed by two-way ANOVA with Bonferroni post hoc test. P values < 0.05 were considered significant.
References
ABS. Australian Health Survey: Biomedical Results for Chronic Diseases, 2011–12. (Australian Bureau of Statistics, Canberra, 2013).
Harambat, J., Van Stralen, K. J., Kim, J. J. & Tizard, E. J. Epidemiology of chronic kidney disease in children. Pediatric nephrology 27, 363–373 (2012).
Stangenberg, S. et al. Oxidative stress, mitochondrial perturbations and fetal programming of renal disease induced by maternal smoking. The international journal of biochemistry & cell biology 64, 81–90 (2015).
Nguyen, L. T. et al. L-Carnitine reverses maternal cigarette smoke exposure-induced renal oxidative stress and mitochondrial dysfunction in mouse offspring. American Journal of Physiology-Renal Physiology 308, F689–F696 (2015).
Glastras, S. et al. Maternal Obesity Promotes Diabetic Nephropathy in Rodent Offspring. Scientific reports 6, 27769–27769 (2015).
Chen, H. & Morris, M. J. Differential responses of orexigenic neuropeptides to fasting in offspring of obese mothers. Obesity 17, 1356–1362 (2009).
Chan, Y. L. et al. Short term exendin-4 treatment reduces markers of metabolic disorders in female offspring of obese rat dams. International Journal of Developmental Neuroscience 46, 67–75 (2015).
Jackson, C. M. et al. Exposure to maternal overnutrition and a high-fat diet during early postnatal development increases susceptibility to renal and metabolic injury later in life. American Journal of Physiology - Renal Physiology 302, F774 (2012).
Glastras, S. J. et al. Effect of GLP-1 Receptor Activation on Offspring Kidney Health in a Rat Model of Maternal Obesity. Scientific Reports 6, 23525, doi:10.1038/srep23525 (2016).
de Vries, A. P. J. et al. Fatty kidney: emerging role of ectopic lipid in obesity-related renal disease. The Lancet Diabetes & Endocrinology 2, 417–426, doi:10.1016/S2213-8587(14)70065-8 (2014).
Lamacchia, O. et al. Para- and perirenal fat thickness is an independent predictor of chronic kidney disease, increased renal resistance index and hyperuricaemia in type-2 diabetic patients. Nephrology Dialysis Transplantation 26, 892–898, doi:10.1093/ndt/gfq522 (2011).
Hartleben, B. et al. Autophagy influences glomerular disease susceptibility and maintains podocyte homeostasis in aging mice. The Journal of Clinical Investigation 120, 1084–1096, doi:10.1172/JCI39492.
Yamahara, K. et al. Obesity-Mediated Autophagy Insufficiency Exacerbates Proteinuria-induced Tubulointerstitial Lesions. Journal of the American Society of Nephrology. doi:10.1681/asn.2012111080 (2013).
Nogueiras, R. et al. Sirtuin 1 and Sirtuin 3: Physiological Modulators of Metabolism. Physiological Reviews 92, 1479 (2012).
Cohen, H. Y. et al. Calorie Restriction Promotes Mammalian Cell Survival by Inducing the SIRT1 Deacetylase. Science 305, 390 (2004).
Jukarainen, S. et al. Obesity Is Associated With Low NAD+/SIRT Pathway Expression in Adipose Tissue of BMI-Discordant Monozygotic Twins. The Journal of Clinical Endocrinology & Metabolism 101, 275–283 (2015).
Kim, M. Y. et al. Resveratrol prevents renal lipotoxicity and inhibits mesangial cell glucotoxicity in a manner dependent on the AMPK–SIRT1–PGC1α axis in db/db mice. Diabetologia 56, 204–217, doi:10.1007/s00125-012-2747-2 (2013).
Kitada, M. et al. Dietary restriction ameliorates diabetic nephropathy through anti-inflammatory effects and regulation of the autophagy via restoration of Sirt1 in diabetic Wistar fatty (fa/fa) rats: a model of type 2 diabetes. Experimental diabetes research 2011 (2011).
Kitada, M., Kume, S., Imaizumi, N. & Koya, D. Resveratrol Improves Oxidative Stress and Protects Against Diabetic Nephropathy Through Normalization of Mn-SOD Dysfunction in AMPK/SIRT1-Independent Pathway. Diabetes 60, 634 (2011).
Suter, M. A. et al. A maternal high-fat diet modulates fetal SIRT1 histone and protein deacetylase activity in nonhuman primates. The FASEB Journal 26, 5106–5114 (2012).
Borengasser, S. J. et al. High fat diet and in utero exposure to maternal obesity disrupts circadian rhythm and leads to metabolic programming of liver in rat offspring. PloS one 9 (2014).
Nguyen, L. T., Chen, H., Pollock, C. A. & Saad, S. Sirtuins—mediators of maternal obesity-induced complications in offspring? The FASEB Journal 30, 1383–1390, doi:10.1096/fj.15-280743 (2016).
Cobo, G. et al. Sex and gender differences in chronic kidney disease: progression to end-stage renal disease and haemodialysis. Clinical Science 130, 1147 (2016).
McMullen, S. & Langley-Evans, S. C. Sex-Specific Effects of Prenatal Low-Protein and Carbenoxolone Exposure on Renal Angiotensin Receptor Expression in Rats. Hypertension 46, 1374–1380, doi:10.1161/01.hyp.0000188702.96256.46 (2005).
Thone-Reineke, C. et al. High-protein nutrition during pregnancy and lactation programs blood pressure, food efficiency, and body weight of the offspring in a sex-dependent manner. American Journal of Physiology-Regulatory, Integrative and Comparative Physiology 291, R1025–R1030 (2006).
Howie, G. J., Sloboda, D. M. & Vickers, M. H. Maternal undernutrition during critical windows of development results in differential and sex-specific effects on postnatal adiposity and related metabolic profiles in adult rat offspring. The British journal of nutrition 108, 298–307, doi:10.1017/s000711451100554x (2012).
Gallo, L. A. et al. Transgenerational programming of fetal nephron deficits and sex-specific adult hypertension in rats. Reproduction, Fertility and Development 26, 1032–1043, doi:10.1071/RD13133 (2014).
Han, L. et al. SIRT1 is regulated by a PPARγ–SIRT1 negative feedback loop associated with senescence. Nucleic Acids Research 38, 7458–7471, doi:10.1093/nar/gkq609 (2010).
Zhao, J. et al. FoxO3 Coordinately Activates Protein Degradation by the Autophagic/Lysosomal and Proteasomal Pathways in Atrophying Muscle Cells. Cell Metabolism 6, 472–483, doi:10.1016/j.cmet.2007.11.004 (2007).
Lee, I. H. et al. A role for the NAD-dependent deacetylase Sirt1 in the regulation of autophagy. Proceedings of the National Academy of Sciences 105, 3374–3379 (2008).
Bastard, J.-P. et al. Recent advances in the relationship between obesity, inflammation, and insulin resistance. Eur Cytokine Netw 17, 4–12 (2006).
Henegar, J. R., Bigler, S. A., Henegar, L. K., Tyagi, S. C. & Hall, J. E. Functional and structural changes in the kidney in the early stages of obesity. Journal of the American Society of Nephrology 12, 1211–1217 (2001).
Deji, N. et al. Structural and functional changes in the kidneys of high-fat diet-induced obese mice. American Journal of Physiology - Renal Physiology 296, F118–F126, doi:10.1152/ajprenal.00110.2008 (2009).
Glastras, S. J. et al. FXR expression is associated with dysregulated glucose and lipid levels in the offspring kidney induced by maternal obesity. Nutrition & metabolism 12, 1 (2015).
Power, M. L. & Schulkin, J. Sex differences in fat storage, fat metabolism, and the health risks from obesity: possible evolutionary origins. British Journal of Nutrition 99, 931–940, doi:10.1017/S0007114507853347 (2008).
Ruderman, N. B. et al. AMPK and SIRT1: a long-standing partnership? American Journal of Physiology - Endocrinology and Metabolism 298, E751–E760, doi:10.1152/ajpendo.00745.2009 (2010).
Price, N. L. et al. SIRT1 is required for AMPK activation and the beneficial effects of resveratrol on mitochondrial function. Cell metabolism 15, 675–690, doi:10.1016/j.cmet.2012.04.003 (2012).
Declèves, A.-E. et al. Regulation of lipid accumulation by AMK-activated kinase in high fat diet–induced kidney injury. Kidney international 85, 611–623 (2014).
Nemoto, S., Fergusson, M. M. & Finkel, T. Nutrient Availability Regulates SIRT1 Through a Forkhead-Dependent Pathway. Science 306, 2105–2108, doi:10.1126/science.1101731 (2004).
Ahmadian, M. et al. PPAR [gamma] signaling and metabolism: the good, the bad and the future. Nature medicine 99, 557–566 (2013).
Kume, S. et al. Role of Altered Renal Lipid Metabolism in the Development of Renal Injury Induced by a High-Fat Diet. Journal of the American Society of Nephrology 18, 2715–2723 (2007).
Tain, Y.-L., Hsu, C.-N., Chan, Y. J. & Huang, L.-T. Renal Transcriptome Analysis of Programmed Hypertension Induced by Maternal Nutritional Insults. International Journal of Molecular Sciences 16, doi:10.3390/ijms160817826 (2015).
Kume, S. et al. Calorie restriction enhances cell adaptation to hypoxia through Sirt1-dependent mitochondrial autophagy in mouse aged kidney. The Journal of Clinical Investigation 120, 1043–1055, doi:10.1172/JCI41376.
Lee, J., Giordano, S. & Zhang, J. Autophagy, mitochondria and oxidative stress: cross-talk and redox signalling. Biochemical Journal 441, 523–540 (2012).
Giordano, S., Darley-Usmar, V. & Zhang, J. Autophagy as an essential cellular antioxidant pathway in neurodegenerative disease. Redox Biology 2, 82–90, doi:10.1016/j.redox.2013.12.013 (2014).
Sanchez, A. M. et al. AMPK promotes skeletal muscle autophagy through activation of forkhead FoxO3a and interaction with Ulk1. Journal of cellular biochemistry 113, 695–710 (2012).
Ng, F. & Tang, B. L. Sirtuins’ modulation of autophagy. Journal of cellular physiology 228, 2262–2270 (2013).
Olmos, Y. et al. SirT1 Regulation of Antioxidant Genes Is Dependent on the Formation of a FoxO3a/PGC-1α Complex. Antioxidants & Redox Signaling 19, 1507–1521, doi:10.1089/ars.2012.4713 (2013).
Li, X.-N. et al. Activation of the AMPK-FOXO3 Pathway Reduces Fatty Acid–Induced Increase in Intracellular Reactive Oxygen Species by Upregulating Thioredoxin. Diabetes 58, 2246 (2009).
Hasegawa, K. et al. Sirt1 protects against oxidative stress-induced renal tubular cell apoptosis by the bidirectional regulation of catalase expression. Biochemical and biophysical research communications 372, 51–56 (2008).
Funk, J. A., Odejinmi, S. & Schnellmann, R. G. SRT1720 Induces Mitochondrial Biogenesis and Rescues Mitochondrial Function after Oxidant Injury in Renal Proximal Tubule Cells. Journal of Pharmacology and Experimental Therapeutics 333, 593 (2010).
Salazar, G., Huang, J. & Zhao, Y. PGC-1alpha a Novel Regulator of Autophagy. The FASEB Journal 30, 1062.1064–1062.1064 (2016).
Huang, X. Z. et al. Sirt1 Activation Ameliorates Renal Fibrosis by Inhibiting the TGF-βSirt1 Activation. Journal of cellular biochemistry 115, 996–1005 (2014).
Chen, H. et al. Exendin-4 is effective against metabolic disorders induced by intrauterine and postnatal overnutrition in rodents. Diabetologia 57, 614–622 (2014).
Zhang, J. et al. A Cationic-Independent Mannose 6-Phosphate Receptor Inhibitor (PXS64) Ameliorates Kidney Fibrosis by Inhibiting Activation of Transforming Growth Factor-β1. PLOS One 10, e0116888, doi:10.1371/journal.pone.0116888 (2015).
Oka, S. et al. PPARα-Sirt1 complex mediates cardiac hypertrophy and failure through suppression of the ERR transcriptional pathway. Cell metabolism 14, 598–611 (2011).
Acknowledgements
Long Nguyen was supported by Sydney Medical School’s ECR PhD Scholarship and Amgen research scholarship. This study was supported by a Gift Grant Awarded to Dr Hui Chen from Perpetual Ramaciotti Foundation, Australia.
Author information
Authors and Affiliations
Contributions
L.T.N. and H.C. designed the experiments. H.C. conducted animal work. L.T.N. conducted bench-top experiments and most data analyses. L.T.N. wrote the manuscript. C.P. provided research facilities and partially funded the project. H.C., S.S. and C.P. supervised the work, helped with data analysis and reviewed the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Nguyen, L.T., Chen, H., Pollock, C. et al. SIRT1 reduction is associated with sex-specific dysregulation of renal lipid metabolism and stress responses in offspring by maternal high-fat diet. Sci Rep 7, 8982 (2017). https://doi.org/10.1038/s41598-017-08694-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-08694-4
This article is cited by
-
Caloric and time restriction diets improve acute kidney injury in experimental menopausal rats: role of silent information regulator 2 homolog 1 and transforming growth factor beta 1
Molecular Biology Reports (2024)
-
The programming of kidney injury in offspring affected by maternal overweight and obesity: role of lipid accumulation, inflammation, oxidative stress, and fibrosis in the kidneys of offspring
Journal of Physiology and Biochemistry (2023)
-
Sirtuins and the circadian clock interplay in cardioprotection: focus on sirtuin 1
Cellular and Molecular Life Sciences (2021)
-
Renoprotective effects of vitamin D3 supplementation in a rat model of metabolic syndrome
European Journal of Nutrition (2021)
-
Exposure to maternal cafeteria diets during the suckling period has greater effects on fat deposition and Sterol Regulatory Element Binding Protein-1c (SREBP-1c) gene expression in rodent offspring compared to exposure before birth
Nutrition & Metabolism (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.