Abstract
The early evolution of archosauromorphs (bird- and crocodile-line archosaurs and stem-archosaurs) represents an important case of adaptive radiation that occurred in the aftermath of the Permo-Triassic mass extinction. Here we enrich the early archosauromorph record with the description of a moderately large (3–4 m in total length), herbivorous new allokotosaurian, Shringasaurus indicus, from the early Middle Triassic of India. The most striking feature of Shringasaurus indicus is the presence of a pair of large supraorbital horns that resemble those of some ceratopsid dinosaurs. The presence of horns in the new species is dimorphic and, as occurs in horned extant bovid mammals, these structures were probably sexually selected and used as weapons in intraspecific combats. The relatively large size and unusual anatomy of Shringasaurus indicus broadens the morphological diversity of Early–Middle Triassic tetrapods and complements the understanding of the evolutionary mechanisms involved in the early archosauromorph diversification.
Similar content being viewed by others
Introduction
The evolutionary radiation of archosauromorphs (archosaurs – crocodylians and dinosaurs – and several extinct stem-clades) in the aftermath of the catastrophic Permo-Triassic mass extinction (~252 Ma) contributed to reshape Mesozoic terrestrial ecosystems and lead to the dominance of dinosaurs1, 2. As part of the outstanding diversification of dinosaurs, multiple theropods and ornithischians (e.g. ceratosaurians, oviraptorosaurians, hadrosaurids, ceratopsids) developed elaborate cranial structures, including bony weapons and ornaments3,4,5,6,7. Cranial weapons have been considered exclusive of dinosaurs during the Mesozoic and generally interpreted as sexually selected traits and evidence of social behaviour6, 7. Here we describe a new herbivorous allokotosaurian stem-archosaur, Shringasaurus indicus gen. et sp. nov., from the early Middle Triassic of central India (Fig. 1) that possesses a pair of anterodorsally projecting and sub-conical supraorbital horns, closely resembling those of some ceratopsid dinosaurs. The presence of horns in Shringasaurus indicus is dimorphic and as in horned extant mammals, these structures were probably used as weapons in intrasexual combats driven by sexual selection8. Shringasaurus indicus expands the ecomorphotypes recorded during the early diversification of archosauromorphs and shows that morphologies driven by sexual selection were also involved in this macroevolutionary process.
Geographic and geological occurrence of Shringasaurus indicus gen. et sp. nov. (a) Map of the major Gondwana basins of peninsular India (after45), in which the red rectangle indicates the Satpura Basin. (b) Complete geological map of the Satpura Gondwana Basin (after45). (c) Close up of the geology of the area (marked in red rectangle in (b) from where Shringasaurus was collected (after9). Note that the Shringasaurus bone-bed is in the Denwa Formation and the red rectangle marked in the map indicates the Shringasaurus locality.
Results
Geological and palaeontological background
The holotype and referred specimens of Shringasaurus indicus have been collected from the Denwa Formation of the Satpura Gondwana Basin9 (Fig. 1). The Denwa Formation overlies the Lower Triassic Pachmarhi Formation and is overlain unconformably by the Cretaceous Bagra Formation. Maulik et al.10 divided the Denwa Formation into lower and upper parts on the basis of their lithology. The lower Denwa comprises multistoreyed, 3–15 metres thick, sheet-like medium to fine grained sandstone bodies interleaved by red mudstones. By contrast, the upper Denwa is a mudstone-dominated unit characterised by the presence of layers of ribbon-shaped channel-fill bodies and sandy to heterolithic sheet sandstones encased within mudstones10. The sandy or muddy heterolithic sheets and the red mudstones represent rapidly emplaced splay deposits and slowly accumulated floodplain deposits, respectively11. The upper unit of the Denwa Formation has been interpreted as deposited by an anabranching fluvial system11.
The fossil bones of Shringasaurus indicus were recovered from a red mudstone in the upper part of the Denwa Formation. At least seven individuals of different ontogenetic stages were excavated from an area of 25 square metres. Most of them were disarticulated, with exception of a partially articulated skeleton.
The vertebrate fossil assemblage of the Denwa Formation includes the dipnoan Ceratodus sp., the capitosaurid Paracyclotosaurus crookshanki, the mastodonsaurid Cherninia denwai, an undescribed brachyopid, a lonchorhynchine trematosaurid, an undescribed rhynchosaurid rhynchosaur, and small to large-sized dicynodonts9. In the nineteenth century, the Denwa Formation was considered as Late Triassic in age based on the presence of a partial skull bone originally assigned to the temnospondyl genus “Mastodontosaurus”12. Later, Chatterjee & Roy-Chowdhury13 suggested a late Early Triassic to early Middle Triassic age; an assignment also supported by Mukherjee & Sengupta14 on the basis of the recovery of additional temnospondyl remains that they assigned to Parotosuchus. Nandi & Raha15 suggested that the carboniferous shale of the Denwa Formation could be given a Late Triassic age based on its microfloral assemblage. Veevers & Tewari16 assigned a Middle Triassic (Anisian to early Ladinian) age to the Denwa Formation on the basis of its vertebrate assemblage, but without giving further details. Bandyopadhyay & Sengupta17 proposed an early Anisian age to the upper part of the Denwa Formation based on its brachyopid temnospondyl assemblage and also suggested a late Spathian to earliest Anisian age for the lower part of the unit. Abdala et al.18 also recognized that the upper Denwa Formation can be assigned to the early Anisian based on the presence of a brachyopid temnospondyl related to Hadrokkosaurus bradyi from the lower Anisian Holbrook Member of the Moenkopi Formation, USA. However, those authors also pointed out that the Denwa Formation can be directly correlated with the late Anisian subzone C of the Cynognathus Assemblage Zone of South Africa and the approximately coeval Ashfield Shale of the Wianamatta Group of the Sydney Basin (Australia) based on the presence of the temnospondyl genus Paracyclotosaurus. Damiani (in Abdala et al.18) argued that the presence of Paracyclotosaurus and the mastodonsaurid Cherninia denwai in the upper Denwa Formation indicate that a younger age (i.e. latest Anisian) is also possible for this unit. Unfortunately, the archosauromorph content of the formation currently does not shed light on the age of the unit. As a result, here we consider the upper Denwa Formation as broadly Anisian in age.
Systematic Palaeontology
Diapsida Osborn, 1903
Archosauromorpha Huene, 1946 sensu Dilkes19
Allokotosauria Nesbitt et al., 2015
Azendohsauridae Nesbitt et al., 2015
Shringasaurus indicus gen. et sp. nov.
Etymology
‘Śṛṅga’ (Shringa), horn (ancient Sanskrit), and ‘sauros’ (σαῦρος), lizard (ancient Greek), referring to the horned skull; ‘indicus’, Indian (Latin English), refers to the country where such species was discovered.
Holotype
ISIR (Indian Statistical Institute, Reptile, India) 780: partial skull roof (prefrontal, frontal, postfrontal, and parietal) with a pair of large supraorbital horns (Fig. 2e,i).
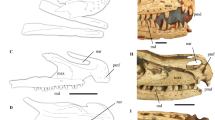
Cranial anatomy of Shringasaurus indicus gen. et sp. nov. and comparison with the skull of a ceratopsid dinosaur that possesses convergent supraorbital horns. (a) Reconstruction of the skull of Shringasaurus indicus in left lateral view. (b) Drawing of the skull of Arrhinoceratops brachyops in left lateral view (based on ROM 79648). (c) Reconstruction of the skull of Shringasaurus indicus in dorsal view. (d–g) Partial skull tables of Shringasaurus indicus in dorsal views (ISIR 781, 780, 786, 789, 790 from left to right), one side has been digitally mirrored in (d–f). (h–k) Partial skull tables of Shringasaurus indicus in left lateral views (ISIR 781, 780, 786, 790 from left to right). Specimens (d–f) and (h–j) possesses horns and specimen/s (g) and (k) lacks horns. Scales = 4 cm for (a) and ( c–k), and 20 cm for (b). en, external naris; ho, horn; or, orbit; stf, supratemporal fenestra.
Skeletal anatomy of Shringasaurus indicus gen. et sp. nov. (a) Left premaxilla (ISIR 793) in lateral view. (b) Left maxilla (ISIR 795) in lateral view. (c) Left quadrate (ISIR 797) in lateral view. (d) Axis (ISIR 803) in left lateral view. (e) Posterior cervical vertebra (ISIR 820) in left lateral view. (f,g) Anterior dorsal vertebra (ISIR 825) in left lateral view in (f), and anterior view in (g). (h) Two anterior caudal vertebrae (ISIR 875) in right lateral view (mirrored). (i) Posterior caudal vertebra (ISIR 892) in left lateral view. (j) Right femur (ISIR 1016) in ventral view. (k) Left ilium (ISIR 991) in lateral view. (l), (m) Right astragalus and fused lateral centrale (ISIR 1059) in proximal view in (l), and dorsal view in (m). (n) Interclavicle (ISIR 950) in ventral view. (o) Left clavicle (ISIR 948) in medial view. (p) Left humerus (ISIR 951) in ventral view. (q) Left scapula (ISIR 929) and coracoid (ISIR 941) in lateral view. (r) Tooth crown (ISIR 801A) in labial view. Scales = 1 cm for (a–c,i,m,l), 2 cm for (d–h,j,k,n–q), and 1 mm for (r), and skeleton = 25 cm. a. articulates with; ac, acetabulum; ain, axial intercentrum; ap, anterior process; ca, calcaneum; ce, lateral centrale; de, denticles; dpc, deltopectoral crest; fi, fibula; gf, glenoid fossa; gr, groove; hqh, hooked quadrate head; icl, interclavicle; itr, internal trochanter; la, lacrimal; lp, lateral process; mp, mammillary process; nag, non-articular gap; pcdl, posterior centrodiapophyseal lamina; pgp, postglenoid process; pnp, postnasal process; pof, popliteal fossa; pop, postacetabular process; pp, parapophysis; ppr, posterior process; ppdl, paradiapophyseal lamina; prdl, prezygodiapophyseal lamina; prp, preacetabular process; prz, prezygapophysis; qj, quadratojugal; sac, supraacetabular crest; sc, scapula; sgl, subglenoid lip; spdl, spinodiapophyseal lamina; sprdl, spinoprezygapophyseal lamina; ti, tibia.
Paratypes
ISIR 781–1072. Cranial and postcranial bones of at least seven individuals of different ontogenetic stages collected from a single, monospecific 5 metres × 5 metres bone-bed (Figs 2 and 3, S1–S3; Supplementary Tables S1, S2).
Locality and horizon
Near Tekapar village, Hoshangabad district, Madhya Pradesh, India (Fig. 1); Denwa Formation, Anisian, early Middle Triassic18, Satpura Gondwana Basin.
Diagnosis
Relatively large (3–4 m total body length; Fig. S1) allokotosaurian archosauromorph that differs from other stem-archosaurs in the following combination of character-states: confluent external nares; pair of anterodorsally oriented supraorbital horns; similar sized and leaf-shaped marginal and palatal teeth with large denticles; middle-posterior cervical, dorsal, and at least the first two caudal vertebrae with mammillary processes on the neural spines; middle-posterior cervical, dorsal, and sacral vertebrae with hyposphene-hypantrum accessory articulations; cervical vertebrae 2–5 with epipophyses (unknown in Cv6); dorsal vertebrae with spinoprezygapophyseal and spinopostzygapophyseal laminae; dorsal vertebrae 1–12 with spinodiapophyseal laminae; anterior dorsal vertebrae with neural spines two times taller than its respective centrum (see Supplementary Information for differential diagnosis).
Description
Shringasaurus indicus has a proportionally small skull with a short, rounded snout and confluent external nares (Figs 2 and 3). The premaxilla lacks a prenarial process and the postnarial process is plate-like and possesses an anteroventrally oriented lateral groove at its base, as occurs in Azendohsaurus madagaskarensis 20. The premaxilla has four tooth positions. The nasal has a long anterior process that arcs ventrally. The marginal tooth crowns are low, with a slightly bulbous base and large denticles on both margins, resembling those of Pamelaria dolichotrachela (ISIR 316/1). The prefrontal and postfrontal are thick and almost exclude the frontal from the border of the orbit. The prefrontal, nasal, frontal, and postfrontal of each side of the skull are fused to each other in large individuals (i.e. bones remain unfused to their counterpart on the sagittal line). The skull roof possesses an anteriorly curved, conical bony horn, almost equal in height to the rest of the skull in large individuals (Fig. 2). Specimens without co-ossified skull roof bones show that the base of the horn occupies the frontal and most of the transverse width of the pachyostotic skull roof. The surface of the horn is ornamented by tangential rugosities and grooves, which are features that have been identified as osteological correlates of cornified sheaths21. The parietal has a very narrow supratemporal fossa that is separated from its counterpart by a flat, broad surface, lacking a sagittal ridge. The quadrate has a hook-shaped dorsal end, as occurs in other allokotosaurians20. Vomerine crowns are more lanceolate than those of the marginal dentition. The parabasisphenoid has an oblique, anteroventrally slanting, main axis (Fig. S2).
The anterior-middle cervical centra of Shringasaurus indicus are approximately 1.5 times longer than tall, indicating a relatively long neck (Figs 3, S1), but proportionally shorter than in Azendohsaurus madagaskarensis 20 and Pamelaria dolichotrachela 22. Besides, the cervical neural spines are proportionally taller than in the latter two species. The first to twelfth dorsal vertebra possess well developed paradiapophyseal, posterior centrodiapophyseal, prezygodiapophyseal, spinodiapophyseal, and spinoprezygapophyseal laminae that bound deep fossae, similar to those of basal sauropods23. Epipophyses are present in the anterior cervical vertebrae and are absent in the seventh to ninth cervical vertebra. Mammillary processes (a pair of transverse expansions on the distal portion of the neural spine that is not confluent with the apex of the spine, see ref. 2) are low, laterally projecting, and displaced anteriorly to the anteroposterior mid-depth of the distal end of the neural spine in, at least, the fifth to the ninth cervical, all recovered dorsal vertebrae, and the first two caudal vertebrae (Fig. S3a). The first sacral vertebra is slightly longer than the second and both possess similar-sized ribs. An intercentrum is preserved between two anterior caudal vertebrae.
The clavicle is constricted close to its ventral end and the interclavicle is T-shaped with a short anterior process and a long, paddle-shaped posterior process, similar to Azendohsaurus madagaskarensis 20. The scapular blade has a distinctly concave anterior margin, as in Azendohsaurus madagaskarensis 20, but unlike the sub-rectangular blade of Pamelaria dolichotrachela 22. The scapular blade is moderately expanded anteroposteriorly at its distal end. The coracoid forms part of a posterolaterally oriented glenoid fossa and has a short post-glenoid process. The humerus is strongly constricted at mid-length and the deltopectoral crest occupies half of the length of the bone. The ulna has a low olecranon process.
The ilium possesses a well-developed, semi-circular preacetabular process, and a longer and dorsoventrally shallower postacetabular process. The acetabulum is fully closed and anterodorsally bounded by a low and thick supraacetabular crest. The pubis has a transversely broad apron that contacts its counterpart and, proximally, an extensive plate-like contact with the ischium. The femur is sigmoid with a prominent internal trochanter that does not converge with the femoral head, as in Azendohsaurus madagaskarensis 20 and Trilophosaurus buettneri 24. The distal end of the femur is transversely broader than the proximal end and the fibular condyle is slightly more distally extended than the tibial condyle. The fibular shaft is approximately two times narrower than the tibial shaft. The astragalus has tibial and fibular facets separated by a broad non-articular surface and a laterally facing concavity to receive the calcaneum, resembling other early archosauromorphs1, 2. The lateral centrale is fused to the astragalus and has a broad articular facet for the reception of the tibia.
Phylogeny
A comprehensive phylogenetic analysis focused on Permo-Triassic stem-archosaurs found Shringasaurus indicus as a non-archosauriform crocopod2, within the clade Allokotosauria (Fig. 4a). Among allokotosaurians, the new species was recovered as an azendohsaurid, together with Pamelaria dolichotrachela and both species of Azendohsaurus. Several cranial, pectoral and pelvic girdle, and limb synapomorphies support the placement of Shringasaurus indicus as an allokotosaurian, azendohsaurid, and the sister-taxon of the genus Azendohsaurus (Supplementary Information).
Phylogenetic relationships of Shringasaurus indicus gen. et sp. nov. and evolution of body size among early archosauromorphs. (a) Time calibrated strict consensus tree found in the data set analysed here (Supplementary Information). Diapsids more basal than Protorosaurus are not shown and all clades except Allokotosauria have been collapsed for clarity. Numbers at the nodes are Bremer support values higher than 1. (b) Evolution of femoral length (as proxy of body size) optimized as a continuous character using maximum parsimony among non-archosauriform archosauromorphs (Supplementary Information). The horizontal axis represents phylogenetic distance. Green circles represent non-allokotosaurian species, red circles represent allokotosaurians, light blue circles represent non-allokotosaurian ancestral femoral lengths, and violet circles represent allokotosaurian ancestral femoral lengths. The dotted line represents a branch not included in the phylogenetic analysis of this study and the horizontal bar with dotted vertical lines on the right side of the graphic represents the median and standard deviation of Permian to Middle Triassic non-allokotosaurian, non-archosauriform archosauromorph femoral length. a, Crocopoda; b, Allokotosauria; c, Trilophosauridae; d, Azendohsauridae; e, Azendohsaurus.
The topology of the strict consensus tree generated from the MPTs is identical to that recovered by Ezcurra2 and Nesbitt et al.25, with exception of the relationships within Allokotosauria (Fig. S4). Contrasting with previous analyses (Ezcurra2; Nesbitt et al.20, 25), Pamelaria dolichotrachela was found as the most basal member of Azendohsauridae, and not as the sister-taxon to all other allokotosaurians. The relationships among the other allokotosaurians are congruent with the result of Nesbitt et al.20, including the taxonomic content of the clade Trilophosauridae, in which Teraterpeton hrynewichorum is the sister-taxon of Spinosuchus caseanus and Trilophosaurus buettneri. Within Azendohsauridae, both species of Azendohsaurus are sister-taxa to one another.
Suboptimal alternative relationships for Shringasaurus indicus within Allokotosauria and outside this clade were explored using heuristic tree searches under monophyly or non-monophyly constraints. Two additional steps are necessary to force a sister-taxon relationship between the two Indian allokotosaurians Shringasaurus indicus and Pamelaria dolichotrachela, in which case the clade is sister to all other azendohsaurids. Four extra steps are required to place Shringasaurus indicus as the sister-taxon to all other azendohsaurids, in which case Pamelaria dolichotrachela is found as the sister-taxon to all other allokotosaurians. Alternatively, five extra steps forces Shringasaurus indicus as the most basal azendohsaurid if Pamelaria dolichotrachela is the sister-taxon of Azendohsaurus spp. Fourteen additional steps forces Shringasaurus indicus to be the most basal allokotosaurian and under this constraint Pamelaria dolichotrachela is the sister-taxon of Azendohsauridae + Trilophosauridae. Ten extra steps are necessary to find Shringasaurus indicus as a trilophosaurid, being the sister-taxon to all other members of the clade. Finally, 26 additional steps are required to place Shringasaurus indicus as a non-crocopod archosauromorph (as the sister-taxon of Crocopoda), 25 steps to find it as a rhynchosaur (as the sister-taxon to all other rhynchosaurs), and 29 steps to find it as a crocopod more derived than rhynchosaurs and allokotosaurians (as the sister-taxon to Boreopricea funerea plus other archosauromorphs).
The Bremer support of Allokotosauria is relatively low (=2), but it is two times higher for Azendohsauridae (=4). It is interesting to note that all the absolute and GC bootstrap frequencies within Allokotosauria are higher than 50% (Fig. S4), thus indicating a rather robust topology for the group. When Prolacertoides jimusarensis and Azendohsaurus laaroussi are pruned a posteriori, the Bremer values are of 4 for both Allokotosauria and Azendohsauridae (Fig. S5). As a result, the position of Shringasaurus indicus as an azendohsaurid allokotosaurian is very well supported in this dataset.
Discussion
The most striking feature of Shringasaurus indicus is its pair of large supraorbital horns (Figs 2 and 3). These horns should have resulted in a more physiologically costly phenotype than a species with a similar body plan but without such elaborate cranial structures (e.g. Pamelaria dolichotrachela, Azendohsaurus madagaskarensis) because of the required investment in growth, transport, and maintenance6, 26. Horned individuals of Shringasaurus indicus of different ontogenetic stages show that the size and robustness of the horns were exacerbated towards the adulthood and possess a distinct variability in their orientation and anterior curvature in large individuals (Fig. 2d–f,h–j). In extant amniotes, the exacerbation of horns and other elaborate cranial structures during ontogeny allows a lower physiological cost to young individuals and their variability is involved in honest quality-signalling27,28,29,30,31. The above mentioned traits (i.e. costliness, positive allometry) characterize sexually selected –a subset of natural selection in which the resource at stake is mates8– features and, as a result, have been considered as key criteria to recognize secondary sexual characters in the fossil record6, 7, 32,33,34,35,36. Besides, the presence of substantial variation in the morphology of the horns of Shringasaurus indicus (size and shape) and their potential costliness weakens a species recognition hypothesis as a possible explanation. Species recognition signals have a very low or zero physiological cost (e.g. differences in colour of skin, feathers or fur, vocalisations, chemical signals)32, 37 and are likely to exhibit minimal variation within a species because high levels of variation would increase the probability of recognition error34. Hence, following the conclusion of previous authors for the elaborate cranial structures of fossil archosaurs (e.g. crests of pterosaurs, hadrosaurids, and oviraptorosarians, horns of ceratopsians)6, 32, 33, a non-adaptive, neutral selection, or species recognition hypothesis are not supported as evolutionary drivers for the origin of the horns of the new species (Supplementary Information).
Strong, robust, unbranched, and sub-conical supraorbital horns very similar to those of Shringasaurus indicus are found among several amniotes and are mainly used as signals of individual quality and directly as weapons in intraspecific agonistic behaviours (e.g. bovid mammals, chamaeleonid lepidosaurs), usually in male-male combats to get access to receptive females8, 27,28,29. The independent evolution of similar horn shapes and robustness among different groups can be explained as the result of sexual selection acting on the biomechanical performance of weapons29. These lines of evidences, including the similarity with the horns of bovids and chamaeleonids, had been used to infer the origin and function of the horns of extinct dinocephalian synapsids and ceratopsian dinosaurs (Fig. 2b) as sexually selected weapons and maybe also used for status signalling3, 6, 38, and the same can be interpreted for Shringasaurus indicus.
A pair of frontals found in the bone-bed of Shringasaurus indicus completely lacks horns, but otherwise is identical to those of horned individuals (Fig. 2g,k). These frontals are approximately of the same size as one specimen with well developed, but still gracile horns (Fig. 2f,j), thus indicating a probable sexual dimorphism. A dimorphic presence/absence of horns is not documented among extinct and extant archosaurs5, 6, but occurs in several horned mammals, in which horns are not effective against predators and function only in intraspecific fighting29. Thus, the dimorphism is favoured by sexual selection, where females usually lack weapons8, 27, 28. In the Shringasaurus indicus bone-bed there are at least six horned individuals and only one or two lack horns. Females may be interpreted as those lacking weapons if we consider extant analogues8, 27, but this apparent sex ratio can be a result of taphonomic biases because horned skull roofs with fused circumorbital bones are more massive, probably favouring their differential transport and preservation.
Shringasaurus indicus attained a relatively large size (3–4 m of total length) that distinctly exceeds the size range of other Early-Middle Triassic archosauromorphs (Fig. 4b). Though there are other probable causes for increases in body size, this could be potentially related with sexual selection because intrasexual competition tends to favour the evolution of larger body sizes27, 28. Besides, the new species shows convergences with sauropodomorph dinosaurs, including the shape of marginal teeth, which seems to be related with an herbivorous habit, as previously suggested for Azendohsaurus spp.39. Thus, it is interpreted that Shringasaurus indicus occupied an ecological role as a large primary consumer in its ecosystem, a role previously thought to be restricted to synapsids in Palaeozoic and Early–Middle Triassic terrestrial communities (e.g. edaphosaurids, dinocephalians, anomodonts)40, but subsequently successfully exploited by Late Triassic archosauromorphs, such as rhynchosaurs, aetosaurs, and sauropodomorphs41, 42. The large size and unusual anatomy of Shringasaurus indicus broadens the morphological diversity of Early–Middle Triassic tetrapods and complements the understanding of the evolutionary mechanisms involved in their diversification after the Permo-Triassic mass extinction.
Methods
Phylogenetic analysis
The relationships of Shringasaurus indicus were analyzed in the most comprehensive phylogenetic dataset available for Permo-Triassic archosauromorphs2 as modified by Nesbitt et al.25. The matrix was analyzed under equally weighted parsimony using TNT 1.543, 44. A heuristic search with 100 replicates of Wagner trees (with a random addition sequence) followed by TBR branch-swapping (holding 10 trees per replicate) was performed. The best trees obtained from the replicates were subjected to a final round of TBR branch swapping. Zero length branches in any of the recovered MPTs were collapsed. Decay indices (=Bremer support values) were calculated and a bootstrap resampling analysis, using 1,000 pseudoreplicates, was performed reporting both absolute and GC (i.e. difference between the frequencies of recovery in pseudoreplicates of the original group and the most frequently recovered contradictory group) frequencies.
We added Shringasaurus indicus and three other allokotosaurian species (Azendohsaurus laaroussi, “Spinosuchus combined”, Teraterpeton hrynewichorum) to the original dataset. Two additional terminals were included after splitting the scorings of “Spinosuchus combined” into Trilophosaurus jacobsi and Spinosuchus caseanus in order to test the synonym hypothesis of Nesbitt et al.20 (Supplementary Information). Two characters were modified, some scorings were changed, and 14 characters were added (601–614; Supplementary Information). The modified data matrix (including “Spinosuchus combined”) includes 88 terminals and 620 characters. The following characters were ordered: 1, 2, 7, 10, 17, 19–21, 28, 29, 36, 40, 42, 50, 54, 66, 71, 75, 76, 122, 127, 146, 153, 156, 157, 171, 176, 177, 187, 202, 221, 227, 263, 266, 279, 283, 324, 327, 331, 337, 345, 351, 352, 354, 361, 365, 370, 377, 379, 398, 410, 424, 430, 435, 446, 448, 454, 458, 460, 463, 472, 478, 482, 483, 489, 490, 504, 510, 516, 529, 537, 546, 552, 556, 557, 567, 569, 571, 574, 581, 582, 588.
Femoral length optimization
Femoral length, as proxy of body size, was optimized as a continuous character45 using maximum parsimony in TNT 1.543. Measurements are based on personal observations and published references (Supplementary Information). The median and standard deviation of the femoral length of late Permian to Middle Triassic archosauromorphs (excluding allokotosaurians) were calculated using the software environment R44.
Nomenclatural acts
This published work and the nomenclatural acts it contains have been registered in ZooBank, the proposed online registration system for the International Code of Zoological Nomenclature. The ZooBank LSIDs (Life Science Identifiers) can be resolved and the associated information viewed through any standard web browser by appending the Life Science Identifier to the prefix ‘http://zoobank.org/’. The LSIDs for this publication are urn:lsid:zoobank.org:act:DD9F3C0A-1107-4033-8A6E-8B94A4BD9718 and urn:lsid:zoobank.org:act:2CB56E16-EC8B-4691-99BD-A8F3DF4B26E8.
References
Nesbitt, S. J. The early evolution of archosaurs: relationships and the origin of major clades. Bulletin of the American Museum of Natural History 352, 1–292 (2011).
Ezcurra, M. D. The phylogenetic relationships of basal archosauromorphs, with an emphasis on the systematics of proterosuchian archosauriforms. PeerJ 4, e1778 (2016).
Farlow, J. O. & Dodson, P. The behavioral significance of frill and horn morphology in ceratopsian dinosaurs. Evolution 29, 353–361 (1975).
Molnar, R. E. Analogies in the evolution of combat and display structures in ornithopods and ungulates. Evolutionary Theory 3, 165–190 (1977).
Padian, K. & Horner, J. R. The evolution of ‘bizarre structures’ in dinosaurs: biomechanics, sexual selection, social selection or species recognition? Journal of Zoology 283, 3–17 (2011).
Hone, D. W. E., Naish, D. & Cuthill, I. C. Does mutual sexual selection explain the evolution of head crests in pterosaurs and dinosaurs? Lethaia 45, 139–156 (2012).
Knell, R. J., Naish, D., Tomkins, J. L. & Hone, D. W. E. Sexual selection in prehistoric animals: detection and implications. Trends in Ecology and Evolution 28, 38–47 (2013).
Tobias, J. A., Montgomerie, R. & Lyon, B. E. The evolution of female ornaments and weaponry: social selection, sexual selection and ecological competition. Philosophical Transactions of the Royal Society B 367, 2274–2293 (2012).
Sengupta, S., Sengupta, D. P. & Bandyopadhyay, S. Stratigraphy of the Upper Gondwana Formations around Sohagpur, Western Part of the Satpura Gondwana Basin, Central India. Journal Geological Society of India 87, 503–519 (2016).
Maulik, P. K., Chakraborty, C., Ghosh, P. & Rudra, D. Meso- and Macro-Scale architecture of a Triassic Fluvial Succession: Denwa Formation, Satpura Gondwana Basin, Madhya Pradesh. Journal Geological Society of India 56, 489–504 (2000).
Ghosh, P., Sarkar, S. & Maulik, P. Sedimentology of a muddy alluvial deposit: Triassic Denwa Formation, India. Sedimentary Geology 191, 3–36 (2006).
Lydekker, R. Catalogue of the fossil Reptilia and Amphibia in the British Museum (Natural History), 145–146 (London, Longmans, 1890).
Chatterjee, S. & Roy-Chowdhury, T. Triassic Gondwana vertebrates of India. Indian Journal of Earth Sciences 1, 96–112 (1974).
Mukherjee, R. N. & Sengupta, D. P. New capitosaurid amphibians from the Triassic Denwa Formation of the Satpura Gondwana Basin, central India. Alcheringa 22, 317–327 (1998).
Nandi, A. & Raha, P. K. Palynoflora from Motur Formation, Satpura Basin, Madhya Pradesh. Indian Minerals 52, 129–132 (1998).
Veevers, J. J. & Tewari, R. C. Gondwana master basin of Peninsular India between Tethys and the interior of the Gondwanaland province of Pangea. Geological Society of America Memoirs 187, 1–72 (1995).
Bandyopadhyay, S. & Sengupta, D. P. Middle Triassic vertebrates of India. Journal of African Earth Sciences 29, 233–241 (1999).
Abdala, F., Hancox, P. J. & Neveling, J. Cynodonts from the uppermost Burgersdorp Formation, South Africa, and their bearing on the biostratigraphy and correlation of the Triassic Cynognathus Assemblage Zone. Journal of Vertebrate Paleontology 25, 192–199 (2005).
Dilkes, D. W. The Early Triassic rhynchosaur Mesosuchus browni and the interrelationships of basal archosauromorph reptiles. Philosophical Transactions of the Royal Society B 353, 501–541 (1998).
Nesbitt, S. J. et al. Postcranial anatomy and relationships of Azendohsaurus madagaskarensis. Bulletin of the American Museum of Natural History 398, 1–126 (2015).
Hieronymus, T. L., Witmer, L. M., Tanke, D. H. & Currie, P. J. The facial integument of centrosaurine ceratopsids: morphological and histological correlates of novel skin structures. The Anatomical Record 292, 1370–1396 (2009).
Sen, K. Pamelaria dolichotrachela, a new prolacertid reptile from the Middle Triassic of India. Journal of Asian Earth Sciences 21, 663–681 (2003).
Wilson, J. A. Vertebral laminae in sauropods and other saurischian dinosaurs. Journal of Vertebrate Paleontology 19, 639–653 (1999).
Spielmann, J. A., Lucas, S. G., Rinehart, L. F. & Heckert, A. B. The Late Triassic archosauromorph. Trilophosaurus. New Mexico Museum of Natural History & Science Bulletin 43, 1–177 (2008).
Nesbitt, S. J. et al. The earliest bird-line archosaurs and the assembly of the dinosaur body plan. Nature 544, 484–487 (2017).
Taylor, R. A. The vascularity and possible thermoregulatory function of the horns in goats. Physiological Zoology 39, 127–139 (1960).
Darwin, C. The descent of man and selection in relation to sex. London: John Murray; p. 828 (1871).
McCullough, E. L., Miller, C. W. & Emlen, D. J. Why sexually selected weapons are not ornaments. Trends in Ecology and Evolution 31, 742–751 (2016).
Geist, V. The evolution of horn-like organs. Behaviour 27, 173–214 (1966).
Tomkins, J. L., Lebas, N. R., Witton, M. P., Martill, D. M. & Humphries, S. Positive allometry and the prehistory of sexual selection. American Naturalist 176, 141–148 (2010).
Berglund, A., Bisazza, A. & Pilastro, A. Armaments and ornaments: an evolutionary explanation of traits of dual utility. Biological Journal of the Linnean Society 58, 385–399 (1996).
Knell, R. J. & Sampson, S. Bizarre structures in dinosaurs: species recognition or sexual selection? A response to Padian and Horner. Journal of Zoology 283, 18–22 (2011).
Hone, D. W. E. & Naish, D. The ‘species recognition hypothesis’ does not explain the presence and evolution of exaggerated structures in non-avialan dinosaurs. Journal of Zoology 290, 172–180 (2013).
Borkovic, B. & Russell, A. Sexual selection according to Darwin: a response to Padian and Horner’s interpretation. Comptes Rendus Palevol 13, 701–707 (2014).
Knell, R. J., Naish, D., Tomkins, J. L. & Hone, D. W. E. Is sexual selection defined by dimorphism alone? A reply to Padian and Horner. Trends in Ecology and Evolution 28, 250–251 (2013).
Hone, D. W. E. & Faulkes, C. G. A proposed framework for establishing and evaluating hypotheses about the behaviour of extinct organisms. Journal of Zoology 292, 260–267 (2014).
Maynard Smith, J. & Harper, D. Animal signals. (Oxford University Press, Oxford, 2003).
Barghusen, H. R. A review of fighting adaptations in dinocephalians (Reptilia, Therapsida). Paleobiology 1, 295–311 (1975).
Flynn, J. J., Nesbitt, S. J., Parrish, J. M., Ranivoharimanana, L. & Wyss, A. R. A new species of Azendohsaurus (Diapsida: Archosauromorpha) from the Triassic Isalo Group of southwestern Madagascar: cranium and mandible. Palaeontology 53, 669–688 (2010).
Bakker, R. T. Tetrapod mass extinctions—a model of the regulation of speciation rates and immigration by cycles of topographic diversity. Developments in Palaeontology and Stratigraphy 5, 439–468 (1977).
Benton, M. J. Dinosaur success in the Triassic: a noncompetitive ecological model. The Quarterly Review of Biology 58, 29–55 (1983).
Bonaparte, J. F. Faunal replacement in the Triassic of South America. Journal of Vertebrate Paleontology 2, 362–371 (1982).
Goloboff, P., Farris, J. & Nixon, K. TNT: a free program for phylogenetic analysis. Cladistics 24, 774–786 (2008).
Goloboff, P. A. & Catalano, S. A. TNT version 1.5, including a full implementation of phylogenetic morphometrics. Cladistics 32, 221–238 (2016).
Goloboff, P. A., Mattoni, C. I. & Quinteros, A. S. Continuous characters analysed as such. Cladistics 22, 589–601 (2006).
R: A language and environment for statistical computing. (R Foundation for Statistical Computing, Vienna, Austria, 2013).
Ghosh, S., Sarkar, S. & Ghosh, P. Petrography and major element geochemistry of the Permo-Triassic sandstones, central India: Implication for provenance in an intracratonic pull-apart basin. Journal of Asian Earth Sciences 43, 207–240 (2012).
Mallon, J. C., Holmes, R., Anderson, J. S., Farke, A. A. & Evans, D. C. New information on the rare horned dinosaur Arrhinoceratops brachyops (Ornithischia: Ceratopsidae) from the Upper Cretaceous of Alberta, Canada. Canadian Journal of Earth Sciences 51, 618–634 (2014).
Acknowledgements
SS and SB thank the financial support and infrastructure facilities provided by the Indian Statistical Institute. SB appreciates the help of D. P. Sengupta and S. S. Das during the excavation and preparation of the specimens. We thank the Reprography Unit of the ISI for photographs of the specimens. The authors are thankful to Dr. Upal Sen of the Sanskrit College and University, Kolkata, for his advice on etymology of the specimen. The comments and suggestions of Max Langer and two anonymous reviewers helped to improve the manuscript. Funding was provided by a National Geographic Society Young Explorers grant (#9467-14 to MDE), and a Marie Curie Career Integration Grant (630123 to Richard J. Butler). We thank W. Simpson (FMNH), R. Allain (MNHN), M. Brett-Surman and H.-D. Sues (both USNM) for access to allokotosaurian comparative material. Discussions with G. Cassini on horn evolution are greatly appreciated.
Author information
Authors and Affiliations
Contributions
S.B. designed the research project and conducted fieldwork; S.S. and M.D.E. conducted the phylogenetic analysis and made the figures; all the authors described the material, discussed the results, and wrote the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Sengupta, S., Ezcurra, M.D. & Bandyopadhyay, S. A new horned and long-necked herbivorous stem-archosaur from the Middle Triassic of India. Sci Rep 7, 8366 (2017). https://doi.org/10.1038/s41598-017-08658-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-08658-8
This article is cited by
-
A new short-faced archosauriform from the Upper Triassic Placerias/Downs’ quarry complex, Arizona, USA, expands the morphological diversity of the Triassic archosauriform radiation
The Science of Nature (2021)
-
Cultural Geology, Cultural Biology, Cultural Taxonomy, and the Intangible Geoheritage as New Strategies for Geoconservation
Geoheritage (2021)
-
New embryological and palaeontological evidence sheds light on the evolution of the archosauromorph ankle
Scientific Reports (2020)
-
A new specimen of Prolacerta broomi from the lower Fremouw Formation (Early Triassic) of Antarctica, its biogeographical implications and a taxonomic revision
Scientific Reports (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.