Abstract
The abundance of fluorescent dissolved organic matter (FDOM) in the surface ocean plays a critical role in the growth of marine microorganisms and corals by affecting the optical properties (i.e., the penetration of UV radiation) of seawater. In general, it is known that rivers are the main source of FDOM to surface ocean waters. Here, however, we show that the concentrations of FDOM in coastal seawater off a volcanic island, Jeju, Korea, are dependent primarily on submarine groundwater discharge (SGD). Based on a significant correlation between 222Rn and salinity in seawater, fresh groundwater was found to be the main source of groundwater as well as fresh water in the bay. The addition of dissolved organic carbon (DOC) and protein-like FDOM to the bay via SGD was generally negligible or negative. However, SGD enhanced the inventory of humic-like FDOM (FDOMH) in seawater by 2–3 times over all seasons, with conservative behavior of FDOMH in bay seawater. These results suggest that SGD-driven fluxes of FDOM regulate its inventory in seawater and consequently play a significant role in determining the optical properties of coastal waters off islands and associated coastal ecosystems (i.e., corals).
Similar content being viewed by others
Introduction
Fluorescent dissolved organic matter (FDOM) is a major fraction of the sunlight absorbing dissolved organic matter (DOM) in water1. In particular, it absorbs harmful UV and visible radiation with a short wavelength2. Thus, the abundance of FDOM in the surface water influences optical conditions in aquatic environments3. It has been reported that FDOM contributes significantly to coastal ecosystems in terms of optically dependent components, such as the growth of coral reefs, juvenile fish, and photosynthesis4,5,6,7,8. FDOM is divided mainly into two different components, i.e., humic-like FDOM (FDOMH) and protein-like FDOM (FDOMP), depending on its origin and optical properties9. Typically, the fluorescence spectra of FDOMH show broad and long excitation and emission wavelengths, and its peak location is similar to that of humic and fulvic acids9. The fluorescence spectra of FDOMP show relatively narrow and short wavelengths, and its peak location is similar to that of amino acids (e.g., tryptophan and tyrosine amino acids)10.
In nearshore coastal oceans, both FDOM components are produced by autochthonous and allochthonous processes in waters and sediments9. Active photosynthesis and subsequent biological processes result in the production of FDOMP in the euphotic zone. On the other hand, microbial degradation of organic debris produces FDOMH in the water column and sediments11, 12. In addition, significant amounts of terrestrial FDOM are transported to oceans via rivers. River waters contain dissolved organic substances derived from the efflux from soils and produced in situ in the water. Thus, the distribution of FDOM shows significant relationships with salinity or other terrestrial tracers (e.g., lignin) in coastal oceans13,14,15.
More recently, in addition to riverine fluxes, submarine groundwater discharge (SGD) has been recognized as a potential source of FDOM in coastal oceans16,17,18,19,20. SGD is the submarine groundwater outflow across the interface between the ocean and the land, regardless of its salinities and chemical properties21, 22. Chen, et al.16 successfully used FDOM to identify the groundwater-driven DOM in the Florida Coastal Everglades. Nelson, et al.18 used the spectral characteristics of FDOM as a tracer for identifying two different SGD sources in the coral reefs of Hawaii. In other studies, it was reported that, in subterranean estuaries (STE), the concentrations of FDOM in groundwater were considerably higher than those in seawater17, 19. In STE, FDOM showed either non-conservative behavior in the Florida coastal region19 and tidal flats23 or conservative behavior in waters off Jeju, a volcanic island in Korea17. However, a quantitative evaluation of SGD-driven FDOM in coastal waters has not been documented yet.
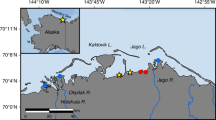
Jeju Island is an ideal place for examining the influence of SGD-driven chemical constituents in the coastal ocean because most of the precipitation permeates through the porous ground and moves to the coastal ocean through SGD rather than surface runoff from the island, on the pathway of a branch of the oligotrophic Kuroshio Current17, 24,25,26,27 (Fig. 1). In this study, we attempted to determine (1) SGD-driven fluxes and behaviors of FDOM in a bay where SGD is pronounced, (2) seasonal changes in the contribution of SGD to the FDOM budget in the bay, and (3) the contribution of SGD to the budgets of protein-like and humic-like components of FDOM in the bay.
A map showing the study region and the sampling locations for DOC and FDOM in Jochun Bay in Jeju Island, Korea. Contour lines indicate the bottom depth based on in situ CTD casting. The number in the asterisk indicates the sampling station of the coastal spring well for the fresh groundwater. The maps and the sampling stations were drawn using Adobe Illustrator CC software version 2015.0.1. (http://www.adobe.com/).
Results and Discussion
The salinities of groundwater samples ranged from 0.17–6.87, with almost constant values (average: 1.24 ± 1.89) except for two samples in August 2015 (13.63 and 20.69) (Table S1). Based on a sharp density gradient in seawater, mainly due to the flow of fresh spring water, the surface layer (salinity: 31.06 ± 2.61) in the bay is separated from the deep layer (salinity: 33.51 ± 1.06) at approximately 1 m depth. Although the salinities of deep water in Jochun Bay were relatively constant during the four sampling campaigns, surface water salinities showed a large seasonal variation: low in summer (26.21–32.20, average: 29.96 ± 1.78, in July and August 2015) and high in winter (26.90–34.50, average: 32.05 ± 2.44, in February and March 2016). The lower salinity of seawater in summer seems to be associated with larger fluxes of fresh SGD due to heavy rainfall in the summer monsoon season. The contour plots of salinity in surface water showed lower salinity values in the innermost region of Jochun Bay (Fig. 2). These trends indicate that the main springs are located mainly in shallow depths because there are no rivers in the bay.
Distributions of salinity, DOC, FDOMP, and FDOMH in the surface water in Jochun Bay. The contour plots were created using Ocean Data View software version 4.7.10. (https://odv.awi.de) and the background maps were drawn using Adobe Illustrator CC software version 2015.0.1. (http://www.adobe.com/).
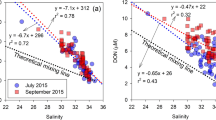
The average dissolved organic carbon (DOC) concentrations in the groundwater were 26 ± 11 μM during four sampling campaigns, except two unusually higher values (126 and 173 μM at wells 5 and 8, respectively) observed in August 2015 (Table S1). These DOC concentrations in groundwater samples were lower than those in seawater and rainwater samples (Fig. 3), typical of aged groundwater resulting from DOC degradation in the aquifer17, 18. In August 2015, relatively higher concentrations of DOC and a negative correlation between DOC concentrations and salinities (y-intercept: 165 μM) were observed (Table S2; Fig. 4). The y-intercept value was similar to the concentrations observed in the contaminated spring waters (i.e., wells 5 and 8). However, the y-intercept value was much higher than those in other wells (26 ± 11 μM). Thus, the high DOC concentrations in seawater in August 2015 seem to be linked to the introduction of unusually high, contaminated, DOC from hydraulically upgradient groundwater systems following heavy precipitation which occurred for three days (~90 mm/day) about one month before the sampling campaign. However, fluvial DOC inputs are unlikely since all sampling campaigns were performed at least one week after the precipitation event, which is longer than the water residence time of the bay.
The parallel factor analysis (PARAFAC) model identified three components from the 195 excitation-emission matrix spectroscopy (EEMs) data. The peaks of components 1 (C1) and 2 (C2) showed short excitation and emission spectra at Ex/Em = 300/360 nm and 320/336 nm, respectively. Based on a previous study, the location of the peak for C1 is similar to that of the protein-like “T” peak reported by Coble9. C2 is not exactly matched with the previous studies. However, C2 seems to be associated with FDOMP because its concentration is highly correlated with that of C1 (r2 = 0.99). Component 3 (C3) showed broad excitation and emission spectra at Ex/Em = 325–365/400–450 nm. Its spectral location is similar to that of the terrestrial FDOMH described as “C” peak in the previous study9. In this study, we use C1 and C3 to represent FDOMP and FDOMH, respectively.
In general, all three components of FDOM in groundwater samples (average: 38.4 ± 89.9 QSU for C1, 25.2 ± 56.4 QSU for C2, and 2.1 ± 1.4 QSU for C3) were approximately 2–45 times higher than those in seawater samples (average: 0.9 ± 0.8 QSU for C1, 1.2 ± 1.4 QSU for C2, and 0.8 ± 0.3 QSU for C3) (Fig. 3). Although C3 showed a significant correlation (r2 = 0.62) against salinity in surface and deep seawater, C1 showed large scattering against salinity. This difference suggests that the sources and behaviors of FDOMP and FDOMH are different in this bay.
The relationships between salinity and C1 (FDOMP) in the surface water were relatively significant in July and August 2015, but not in February and March 2016 (Table S2; Fig. 4). The distributions of C1 in surface seawater and groundwater (Table S1; Fig. 4) suggest that the relative enrichment of FDOMP in most of the groundwater influences the high FDOMP stations in summer seawater. The distributions of FDOMP in the winter season showed larger scattering. In general, the deep water showed large variations of FDOMP for a constant salinity, with unusually higher concentrations in some stations (Fig. 4). Thus, the concentrations of FDOMP in this bay seem to be affected by various sources such as groundwater inputs, biological production, and vertical water mixing.
The concentrations of C3 (FDOMH) in groundwater samples were considerably higher than those in seawater (Fig. 3). The FDOMH in surface seawater was negatively correlated with salinity (Table S2; Fig. 4), indicating that FDOMH originated mainly from the fresh groundwater. The linear relationship between FDOMH and salinity suggests its conservative behavior during mixing between the groundwater and seawater. The relatively higher concentrations of FDOMH in summer seawater seem to be associated with higher concentrations in most of the summer groundwater samples (Table S1; Fig. 4). The FDOMH concentrations and salinities in deep-water samples fall within the trend of the data for the surface layer samples (as open-ocean water background concentrations), although slightly higher FDOMH concentrations were observed in near-shore deep-water samples owing to surface-water mixing. The distribution patterns also clearly show that fresh groundwater is a dominant source of FDOMH in this region (Fig. 4).
In order to determine the contribution of the groundwater-driven DOM in Jochun Bay, the flux of DOM from the coastal groundwater was compared with that from the open ocean during the water residence time of the bay. Based on the tidal prism model, the residence time of Jochun Bay was 1.0, 0.8, 0.6, and 1.3 d in July 2015, August 2015, February 2016, and March 2016, respectively28. The volume of the bay is 7.4 × 105 m3, and its mean depth is 2.3 m. Based on the 222Rn mass balance model, the fluxes of submarine fresh groundwater discharge (SFGD) in Jochun Bay were estimated to be 6.9, 5.5, 5.0, and 4.1 (×104) m3 d−1 in July 2015, August 2015, February 2016, and March 2016, respectively28. The volume of SGD accounted for approximately 4–9% of the total water volume in Jochun Bay during the water residence time. In Jeju Island, precipitation is concentrated in the summer monsoon season (June–September) and is strongly associated with the seepage rate of groundwater24. The relatively higher discharge rates of SFGD in July and August 2015 seem to be supported by the lower salinity of the surface water (average: 29.91 and 30.56), as compared to that in February and March 2016 (average: 31.72 and 32.29). The SFGD originating from the main coastal springs seems to affect the surface layer (within a depth of 1 m) based on the gradient of salinity.
Combining all data from four sampling campaigns, one endmember of FDOMH in the fresh groundwater was derived from the y-intercept of the relationship between FDOMH and salinity in the coastal seawater (Fig. 4). However, the y-intercept value was not in good agreement with the measured concentrations of FDOMH in the spring waters. This may be due to the various hidden pathways of the aquifer and various contributions from each spring well. This result suggests that the averages of DOC and FDOM in groundwater can be decoupled from the actual endmember of groundwater owing to the complex nature of groundwater seepages. Therefore, the y-intercept value of the correlation between FDOM and salinity in the coastal seawater is a more reliable and representative endmember. Moreover, these results suggest that conservative organic substances (i.e., DOC and FDOM) can provide further information for locating SGD sources and pathways, in addition to natural SGD tracers18.
Therefore, in this study, we calculated the inventories of DOC in summer and FDOMH in all seasons, which showed a significant correlation against salinities. Here, the inventory is obtained by multiplying the fluxes of DOC and FDOMH (μmol per day and μg QS per day) by the water residence time in the bay (day). During the water residence time in the bay, the inventories of FDOMH originating from the fresh groundwater in summer were higher relative to those in winter (Fig. 5). The endmembers of FDOMH in the fresh groundwater were 5.4 and 6.4 QSU in summer and 2.6 and 3.8 QSU in winter (Table S2). Using the endmember of FDOMH and the rate of SFGD, the inventories of FDOMH resulting from SGD were estimated to be 3.3 × 108 and 1.4 × 108 μg QS in summer and winter, respectively (Fig. 5). The background inventories of FDOMH originating from the open ocean were calculated using the endmember value of FDOMH in the open ocean water and the volume of the surface water layer. The endmembers of FDOMH in the offshore were 0.49 and 0.68 QSU in summer and 0.40 and 0.41 QSU in winter. The background inventories of FDOMH originating from the seawater were estimated to be 1.9 × 108 and 1.3 × 108 μg QS in summer and winter, respectively (Fig. 5). The measured inventories of FDOMH in Jochun Bay were calculated by multiplying the average concentration of FDOMH by the volume of the surface layer. The sum of the inventories of FDOMH originating from fresh groundwater and the open ocean seawater during the water residence time was similar to the measured inventory in Jochun Bay (Fig. 5), indicating that the estimations are reasonable.
In the bay waters, the contributions of FDOMH originating from the groundwater accounted for 170% and 110% of the offshore water in summer and winter, respectively (Fig. 5). In contrast, the contributions of DOC originating from SGD in the coastal seawater were negligible or lower than those from seawater in July 2015, February 2016, and March 2016 (Figs 3 and 4). The inventory of DOC originating from SFGD in the exceptional season (summer) was estimated to be approximately 7.3 × 109 μmol (Fig. 5), which accounted for approximately 28% of the background offshore water. These results suggest that SGD is a significant source of FDOMH but an insignificant source of DOC in coastal waters off volcanic islands. This seems to be associated with the production of FDOMH in coastal aquifers due to microbial activity, resulting in the production of fulvic and humic acids, during DOM degradation. Therefore, SGD-borne FDOMH is determined to be an important hidden source of FDOMH in coastal waters, which is critical for coastal ecosystems including corals, seaweeds, and biological production. Corals need sunlight for building reefs by symbiotic zooxanthellae, but ironically they are highly vulnerable to harmful UV radiation. Zepp, et al.6 reported that FDOM accounts for 70–95% of the total UV attenuation in surface waters. Since FDOM efficiently protects the labile cells of microorganisms and corals from harmful UV radiation, FDOM is considered to be a key factor in sustaining the coral ecosystem3, 4, 6, 29. Additionally, high DOC concentrations are another major threat to coral reefs30, 31. The relatively low DOC concentrations in groundwater from the volcanic islands, Jeju17 and Hawaii18, can provide optimal conditions for the growth of corals. Thus, we surmise that SGD provides favorable conditions for the growth of coral reefs regarding the properties of organic matters in the ocean.
Conclusions
In Jochun Bay, off the volcanic island, Jeju, SFGD accounted for 4–9% of the total water volume. The high enrichments of DOC, FDOMP, and FDOMH in the bay seawater were linked to those in fresh groundwater inputs. In particular, based on the high concentrations of FDOMH in the groundwater and its negative correlations with salinity in the seawater, SFGD accounted for approximately 170% and 110% of the background inventory originating from the open ocean water in summer and winter, respectively. However, the contribution of SFGD to DOC and FDOMP inventory in seawater was negligible. This study highlights that SGD is a key factor controlling FDOM distributions in seawater, potentially affecting the optical and chemical conditions in coastal ecosystems. Because corals are susceptible to contamination by DOC and damage from strong UV radiation, the groundwater in Jeju Island potentially induces a favorable condition for the growth of coral reefs. More extensive studies are necessary to quantify the SGD-driven fluxes of FDOM in coastal seawater, particularly in islands where the optical properties significantly influence ecosystems.
Materials and Methods
Study region
Jeju Island is a volcanic island located in the southern sea of Korea, on the pathway of a branch of the oligotrophic Kuroshio Current (Fig. 1). Because the island bed is mainly composed of porous basalt rocks, most of the rainwater permeates through the porous ground and is transferred to the coastal ocean through groundwater24. Approximately 1000 artesian springs and wells are located along the coast. The groundwater from coastal springs and wells supports approximately 90% of the fresh water resources in Jeju Island. The study area, i.e., Jochun Bay, is located in the northern part of the island (Fig. 1). The bay is semi-enclosed by rocks and a harbor and has several coastal springs. The water residence time of the bay is approximately one day28. Owing to the East Asian monsoon system, heavy precipitation occurs in this region from June to September (~1900 mm per year).
Sampling
Four sampling campaigns for seawater and coastal spring water were conducted in Jochun Bay, Jeju, Korea, in 2015 and 2016 (July and August 2015; February and March 2016) (Fig. 1). Seawater samples in the surface and deep-water layers were collected into acid-washed 1-L Nalgene Polypropylene bottles using a submersible pump. Within a few hours after the seawater sampling was completed, the fresh groundwater samples were collected from artesian spring waters into the acid-washed bottles by hand, using gloves (Fig. 1). Salinity was measured in situ using a conductivity-temperature-depth sensor (OCEAN SEVEN 304 CTD, IDRONAUT Co., Milano, Italy).
FDOM analysis
The seawater and groundwater samples were filtered using pre-combusted Whatman GF/F filters with a pore size of 0.7 μm to remove large-sized materials. Filtered samples were transferred into pre-combusted vials and kept in a refrigerator at a temperature less than 4 °C until spectral analysis was performed. The filtrate was re-filtered using Nuclepore polycarbonate filters with a pore size of 0.2 μm to eliminate fine particles and microorganisms. Optical measurements of FDOM fluorescence were performed using a fluorescence spectrometer (FluoroMate FS-2, SCINCO, Korea) within one week after each sampling campaign. The scanning wavelengths of the excitation and emission spectra ranged from 250 to 600 nm (2 nm intervals) and 250 to 500 nm (5 nm intervals), respectively. The inner filter effect was not corrected for because the influence of this artifact was negligible (only 1–3% of the fluorescence intensity) for groundwater and seawater using this spectrofluorometer23. Rayleigh and Raman scatters were eliminated and interpolated based on Delaunay triangulation, which is developed by Zepp, et al.32, to prevent any problems with the blank subtraction method32, 33.
The PARAFAC model was applied to a combined set of 195 EEMs data. An appropriate number of components were evaluated statistically through the split-half test using the DOMFluor toolbox for MATLAB34. The concentrations of FDOM were normalized every day using the values of a quinine sulfate (QS) dihydrate diluted in 0.1 N sulfuric acid at a specific wavelength (Ex/Em = 350/450 nm). The concentrations are expressed in quinine sulfate equivalent units (QSU) (i.e., in μg QS/L). The variation of the measured QS fluorescence was less than 3% of the average value throughout the all measurements.
DOC analysis
Samples for DOC were vacuum filtered simultaneously with FDOM samples using pre-combusted Whatman GF/F filters. To prevent microbial activity, the filtered samples were acidified using 6 M HCl, followed by hermetic sealing in pre-combusted (500 °C for at least 4 h) 20 mL glass ampoules (Wheaton Scientific, Millville, NJ). The concentration of DOC was measured using a TOC analyzer (TOC-VCPH, Shimadzu, Japan). The accuracy of DOC concentration was verified every day using deep sea reference (DSR: 41–44 μM for DOC, University of Miami) samples.
Data Availability
The datasets analysed during the current study are available from the corresponding author upon reasonable request.
References
Kirk, J. T. O. Light and photosynthesis in aquatic ecosystems. (Cambridge University Press, 1994).
Stedmon, C. A. & Nelson, N. B. In Biogeochemistry of Marine Dissolved Organic Matter (Second Edition) (eds Dennis A. Hansell & Craig A. Carlson) Ch. 10, 481–508 (Academic Press, 2015).
Mizubayashi, K. et al. Monsoon variability of ultraviolet radiation (UVR) attenuation and bio-optical factors in the Asian tropical coral-reef waters. Estuarine, Coastal and Shelf Science 126, 34–43, doi:10.1016/j.ecss.2013.04.002 (2013).
Ayoub, L. M., Hallock, P., Coble, P. G. & Bell, S. S. MAA-like absorbing substances in Florida Keys phytoplankton vary with distance from shore and CDOM: Implications for coral reefs. Journal of Experimental Marine Biology and Ecology 420–421, 91–98, doi:10.1016/j.jembe.2012.03.026 (2012).
Anderson, S. et al. Indicators of UV Exposure in Corals and Their Relevance to Global Climate Change and Coral Bleaching. Human and Ecological Risk Assessment: An International Journal 7, 1271–1282, doi:10.1080/20018091094998 (2001).
Zepp, R. G. et al. Spatial and temporal variability of solar ultraviolet exposure of coral assemblages in the Florida Keys: Importance of colored dissolved organic matter. Limnology and Oceanography 53, 1909–1922 (2008).
Gleason, D. F. & Wellington, G. M. Ultraviolet radiation and coral bleaching. Nature 365, 836–838 (1993).
Arrigo, K. R. & Brown, C. W. Impact of chromophoric dissolved organic matter on UV inhibition of primary productivity in the sea. Marine Ecology Progress Series 140, 207–216 (1996).
Coble, P. G. Marine Optical Biogeochemistry: The Chemistry of Ocean Color. Chem. Rev. 107, 402–418, doi:10.1021/cr050350+ (2007).
Yamashita, Y. & Tanoue, E. Chemical characterization of protein-like fluorophores in DOM in relation to aromatic amino acids. Mar. Chem. 82, 255–271, doi:10.1016/S0304-4203(03)00073-2 (2003).
Burdige, D. J., Kline, S. W. & Chen, W. Fluorescent dissolved organic matter in marine sediment pore waters. Mar. Chem. 89, 289–311, doi:10.1016/j.marchem.2004.02.015 (2004).
Kim, J. & Kim, G. Significant anaerobic production of fluorescent dissolved organic matter in the deep East Sea (Sea of Japan). Geophysical Research Letters, doi:10.1002/2016GL069335 (2016).
Stedmon, C. A. & Markager, S. Behaviour of the optical properties of coloured dissolved organic matter under conservative mixing. Estuarine, Coastal and Shelf Science 57, 973–979, doi:10.1016/S0272-7714(03)00003-9 (2003).
Fichot, C. G. et al. Predicting Dissolved Lignin Phenol Concentrations in the Coastal Ocean from Chromophoric Dissolved Organic Matter (CDOM) Absorption Coefficients. Frontiers in Marine Science 3, doi:10.3389/fmars.2016.00007 (2016).
Kim, J. & Kim, G. Importance of colored dissolved organic matter (CDOM) inputs from the deep sea to the euphotic zone: Results from the East (Japan) Sea. Mar. Chem. 169, 33–40, doi:10.1016/j.marchem.2014.12.010 (2015).
Chen, M., Price, R. M., Yamashita, Y. & Jaffé, R. Comparative study of dissolved organic matter from groundwater and surface water in the Florida coastal Everglades using multi-dimensional spectrofluorometry combined with multivariate statistics. Appl. Geochem. 25, 872–880, doi:10.1016/j.apgeochem.2010.03.005 (2010).
Kim, T.-H., Kwon, E., Kim, I., Lee, S.-A. & Kim, G. Dissolved organic matter in the subterranean estuary of a volcanic island, Jeju: Importance of dissolved organic nitrogen fluxes to the ocean. Journal of Sea Research 78, 18–24, doi:10.1016/j.seares.2012.12.009 (2013).
Nelson, C. E. et al. Fluorescent dissolved organic matter as a multivariate biogeochemical tracer of submarine groundwater discharge in coral reef ecosystems. Mar. Chem. 177(Part 2), 232–243, doi:10.1016/j.marchem.2015.06.026 (2015).
Suryaputra, I. G. N. A., Santos, I. R., Huettel, M., Burnett, W. C. & Dittmar, T. Non-conservative behavior of fluorescent dissolved organic matter (FDOM) within a subterranean estuary. Continental Shelf Research 110, 183–190, doi:10.1016/j.csr.2015.10.011 (2015).
Tedetti, M., Cuet, P., Guigue, C. & Goutx, M. Characterization of dissolved organic matter in a coral reef ecosystem subjected to anthropogenic pressures (La Réunion Island, Indian Ocean) using multi-dimensional fluorescence spectroscopy. Sci. Total Environ. 409, 2198–2210, doi:10.1016/j.scitotenv.2011.01.058 (2011).
Burnett, W. C., Bokuniewicz, H., Huettel, M., Moore, W. S. & Taniguchi, M. Groundwater and pore water inputs to the coastal zone. Biogeochemistry 66, 3–33, doi:10.1023/b:biog.0000006066.21240.53 (2003).
Kim, G. & Hwang, D.-W. Tidal pumping of groundwater into the coastal ocean revealed from submarine 222Rn and CH4 monitoring. Geophysical Research Letters 29, 23-21–23-24, doi:10.1029/2002GL015093 (2002).
Kim, T.-H., Waska, H., Kwon, E., Suryaputra, I. G. N. & Kim, G. Production, degradation, and flux of dissolved organic matter in the subterranean estuary of a large tidal flat. Mar. Chem. 142–144, 1–10, doi:10.1016/j.marchem.2012.08.002 (2012).
Kim, G., Lee, K.-K., Park, K.-S., Hwang, D.-W. & Yang, H.-S. Large submarine groundwater discharge (SGD) from a volcanic island. Geophysical Research Letters 30, 2098, doi:10.1029/2003GL018378 (2003).
Jeong, J., Kim, G. & Han, S. Influence of trace element fluxes from submarine groundwater discharge (SGD) on their inventories in coastal waters off volcanic island, Jeju, Korea. Appl. Geochem. 27, 37–43 (2012).
Kim, I. & Kim, G. Large fluxes of rare earth elements through submarine groundwater discharge (SGD) from a volcanic island, Jeju, Korea. Mar. Chem. 127, 12–19 (2011).
Hwang, D., Lee, Y.-W. & Kim, G. Large submarine groundwater discharge and benthic eutrophication in Bangdu Bay on volcanic Jeju Island, Korea. Limnology and Oceanography 50, 1393–1403 (2005).
Kwon, H. K., Kang, H., Oh, Y. H., Park, S. R. & Kim, G. Green tide development associated with submarine groundwater discharge in a coastal harbor, Jeju, Korea. Scientific Reports (Accepted).
Shank, G. C., Zepp, R. G., Vähätalo, A., Lee, R. & Bartels, E. Photobleaching kinetics of chromophoric dissolved organic matter derived from mangrove leaf litter and floating Sargassum colonies. Mar. Chem. 119, 162–171, doi:10.1016/j.marchem.2010.01.003 (2010).
Lapointe, B. E. Nutrient thresholds for bottom-up control of macroalgal blooms on coral reefs in Jamaica and southeast Florida. Limnology and Oceanography 42, 1119–1131, doi:10.4319/lo.1997.42.5_part_2.1119 (1997).
Fabricius, K. E. Effects of terrestrial runoff on the ecology of corals and coral reefs: review and synthesis. Marine Pollution Bulletin 50, 125–146, doi:10.1016/j.marpolbul.2004.11.028 (2005).
Zepp, R. G., Sheldon, W. M. & Moran, M. A. Dissolved organic fluorophores in southeastern US coastal waters: Correction method for eliminating Rayleigh and Raman scattering peaks in excitation-emission matrices. Mar. Chem. 89, 15–36, doi:10.1016/j.marchem.2004.02.006 (2004).
Nelson, N. B. & Coble, P. G. In Practical Guidelines for the Analysis of Seawater 401 (CRC Press Baco Raton, NJ, 2009).
Stedmon, C. A. & Bro, R. Characterizing dissolved organic matter fluorescence with parallel factor analysis: A tutorial. Limnology and Oceanography: Methods 6, 572–579 (2008).
Acknowledgements
We are grateful to Yonghwa Oh and Hyekyung Kang for the assistance with sampling campaigns. This research was supported by the National Research Foundation (NRF) of Korean (NRF-2015R1A2A1A10054309) and by the Ministry of Oceans and Fisheries, Korea, through the project titled “Long-term change of structure and function in marine ecosystems of Korea”.
Author information
Authors and Affiliations
Contributions
J.K. performed sampling, chemical analysis, PARAFAC modeling, and calculation under the supervision of G.K. The manuscript was written in a joint effort between the two authors.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kim, J., Kim, G. Inputs of humic fluorescent dissolved organic matter via submarine groundwater discharge to coastal waters off a volcanic island (Jeju, Korea). Sci Rep 7, 7921 (2017). https://doi.org/10.1038/s41598-017-08518-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-08518-5
This article is cited by
-
Sponges and their prokaryotic communities sampled from a remote karst ecosystem
Marine Biodiversity (2024)
-
Molecular Signatures of Dissolved Organic Matter in the Central Huaihe River and Its Tributaries
Water, Air, & Soil Pollution (2023)
-
Impact of aquaculture on distribution of dissolved organic matter in coastal Jeju Island, Korea, based on absorption and fluorescence spectroscopy
Environmental Science and Pollution Research (2022)
-
Estimating submarine groundwater discharge in Jeju volcanic island (Korea) during a typhoon (Kong-rey) using humic-fluorescent dissolved organic matter-Si mass balance
Scientific Reports (2021)
-
Factors controlling the distributions of dissolved organic matter in the East China Sea during summer
Scientific Reports (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.