Abstract
Herbivory and disturbance are major drivers of biological invasions, but it is unclear how they interact to determine exotic vs. native seedling recruitment and what consequences arise for biodiversity and ecosystem functioning. Previous studies neglected the roles of different, potentially interacting, guilds of generalist herbivores such as rodents and gastropods. We therefore set up a full-factorial rodent exclusion x gastropod exclusion x disturbance x seed-addition experiment in a grassland community in Central Germany and measured early seedling recruitment, as well as species richness, species composition and aboveground biomass. Gastropod herbivory reduced the positive effect of disturbance on seedling recruitment, particularly for exotic species. Rodent herbivory had weak positive effects on seedling recruitment at undisturbed sites, irrespective of species origin. This effect was likely driven by their strong negative effect on productivity. Interactive effects between both herbivore guilds became only evident for species richness and composition. How many species established themselves depended on disturbance, but was independent of species origin. The fewer exotic species that established themselves increased productivity to a stronger extent compared to native species. Our study highlights that joint effects of disturbance, herbivory and species origin shape early recruitment, while they only weakly affect biodiversity and ecosystem functioning.
Similar content being viewed by others
Introduction
Biological invasions are a major component of global change and known to alter biodiversity and ecosystem functions of plant communities1. Despite increasing evidence that biological invasions are driven by the combination of different processes, most studies so far have focused mainly on one or a few processes and rarely, if ever, consider their interactions2.
To initially colonize a given site and to become established, seedling recruitment is of high importance for exotic as well as for native species3,4. Herbivory and disturbance shape seedling recruitment in plant communities5,6, and thus, their roles in driving invasion success or failure have been extensively studied and discussed7,8,9. The enemy release hypothesis10 suggests that exotic species gain advantage over native species because they are released from their co-evolved specialist herbivores, and are less attacked by resident generalist herbivores compared to native species, e.g. because they produce novel defenses11. In contrast, the biotic resistance hypothesis12 states that resident generalist herbivores may constrain exotic plant invasions by suppressing those exotic species that have not evolved effective defense mechanisms against them. However, so far studies on the effects of generalist herbivores on early seedling recruitment and establishment success have provided inconsistent results, as they found stronger13,14, equal2,15,16 or weaker17,18 effects on exotic compared to native species. This apparent inconsistency may at least be partly explained by the different feeding preferences of the key herbivores in the particular study system.
Two important generalist herbivore guilds of Central European grasslands, that may show such specific feeding preferences, are gastropods and rodents. Feeding preferences of generalists depend on various plant traits, such as carbon/nitrogen ratio, specific leaf area, leaf toughness and chemical defenses19, which, however, do not only differ between exotic and native plant species but also between functional groups and across life-cycle stages of plants20,21. A consequence of such feeding preferences may be non-additive effects of herbivore guilds, e.g. on early establishment22 or community structure23. Gastropods often prefer seedlings over adult plants and legumes or non-legume herbs over grasses16,24. Feeding preferences of rodents differ between granivorous and herbivorous rodents: while granivorous rodents are known to preferentially consume seeds of large-seeded species, herbivorous rodents such as voles consume biomass of grasses that is easily available25, or of legumes that is highly nutritious26.
There are comparably few studies on the importance of generalist herbivores for biological invasions, and those mostly concentrated on granivorous rodents in North America13,14. In contrast to North American grasslands, where granivores were found to play a key role in structuring plant communities27, many studies in Central European grasslands pointed out the importance of gastropods and herbivorous rodents16,23,28. Granivorous rodents can mediate biotic resistance against exotic plant invasions by particularly suppressing early recruitment or population growth of large-seeded weak invaders13. In contrast, in a previous study16, herbivorous rodents were found to equally affect seedling recruitment of exotic and native plant species. The very few studies on the effect of gastropod herbivory showed that they either equally suppressed native and exotic species15,16 or preferred native over exotic plant species17,29. Thus, there is evidence that the outcome of biological invasions differs, depending on the groups of the dominant herbivores in the system.
Another factor suggested to drive invasion success of exotic plant species is disturbance which removes resident competitors9,30. Disturbance of the vegetation cover and exposure of soil (in the following disturbance), for example through human activities such as ploughing, increases the availability of microsites and thereby provides invasion opportunities for exotic species31,32. Compared to native species exotic species are suggested to have a superior ability to exploit resources released by disturbance30. It was, however, also found that native species similarly benefitted from disturbance as exotic species5,6. Therefore, controlled experiments are needed where exotic and native species are introduced to sites with both intact and disturbed vegetation. The few studies using this approach, though, provided inconsistent results, as exotic species were found to have lower20, equal2 or higher seedling recruitment compared to native species18.
Disturbance and herbivory may not act independently, but interactively determine establishment success of plant species: if competitors are removed by disturbance and germination is no longer microsite-limited, the effects of herbivory on seedling recruitment should be greater compared to undisturbed sites33. Despite the knowledge of potentially interactive effects of disturbance and herbivory, it is still unclear how both processes jointly affect the establishment success of exotic compared to native species. Indeed, a study by Maron, et al.18 showed that the joint effects of disturbance and rodent granivory on seedling recruitment varies with species origin, but no such three-way interaction was found by Mueller, et al.2 for invertebrate herbivory. Again, these results may depend on the key herbivores in the study system (i.e. herbivorous rodents, granivorous rodents, gastropods), and thus, it is important to disentangle effects of different herbivore guilds and disturbance on exotic vs. native plant species.
Studies on the role of disturbance and herbivory for biological invasions either focused on early seedling recruitment or on community structure and ecosystem functioning32,34,35. The seedling stage is a vulnerable time in the life-cycle of plants36 and changes in seedling recruitment as an effect of disturbance or herbivory can influence plant abundance, community composition, species richness and productivity (reviewed in refs6,33). However, it is not known whether complex interactions that are evident during early seedling recruitment result in changes in species richness, community composition and productivity. Maron, et al.27 showed that the positive impact of exotic vs. native species on the productivity of the plant community was enhanced when local filters such as competition from residents and generalist herbivory were removed. Moreover, their finding that exotic species increased productivity to larger amounts compared to native species in spite of similar species richness27, suggests that different mechanisms underlie the diversity-productivity relationship in exotic and native dominated communities37,38,39. While it is generally assumed that exotic species directly contribute to productivity through intrinsically higher growth rates which are related to faster resource uptake and use40, the diversity-productivity relationship may also be shaped by selective herbivory on exotic legumes and herbs41.
To address these multiple interactions we devised a full-factorial experiment in a grassland community in Central Germany, where we excluded rodents and gastropods, and added seeds of 20 exotic and native species to plots that were either subjected to a soil disturbance treatment or remained undisturbed. Specifically we seek to answer the following research questions:
-
1)
How do disturbance and the different herbivore guilds, separately and interactively, affect the establishment of exotic vs. native plant species? We expect exotic species, compared to native species, to benefit more from disturbance and to be less affected by gastropods and equally affected by herbivorous rodents. Moreover, we expect that disturbance and herbivory interact with each other and in combination change the establishment of exotic and native species.
-
2)
What are the consequences for species diversity, composition and productivity of plant communities that are experimentally invaded by exotic and native species? We expect that changes in seedling recruitment of exotic vs. native species as an effect of disturbance, gastropod and rodent herbivory result in changes in diversity, composition and productivity over time. Moreover, we expect that, compared to native species, introductions of exotic species lead to a stronger increase in productivity, while species richness generally increases, irrespective of species origin.
Results
How do disturbance and the different herbivore guilds, separately and interactively, affect the establishment of exotic vs. native plant species?
Out of the 40 species sown 33 species (14 exotic, 19 native) germinated and more than 4,000 seedlings recruited from sown seeds. Disturbance substantially enhanced seedling recruitment, both in terms of the number of species with seedling recruitment (disturbed: 10.4 ± 0.7, undisturbed: 6.8 ± 0.7; mean ± SE) and the proportion of seedling recruitment (disturbed: 6.4 ± 1.2%, undisturbed: 2.4 ± 1.2%). However, for the proportion of seedling recruitment this effect differed between functional groups and species origins (Table 1). Specifically, disturbance tended to decrease seedling recruitment for exotic legumes (exotic legumes disturbed: 5.2 ± 2.9%, exotic legumes undisturbed: 6.9 ± 2.9%) while native legumes were not affected by disturbance (native legumes disturbed: 0.4 ± 2.2%, native legumes undisturbed: 0.5 ± 2.2%). Moreover, a strong positive effect of disturbance on the proportion of seedling recruitment was much more pronounced for exotic grasses (exotic grasses disturbed: 18.7 ± 2.9%, exotic grasses undisturbed: 2.5 ± 2.9%) and herbs (exotic herbs disturbed: 5.2 ± 1.4%, exotic herbs undisturbed: 2.5 ± 1.4%) compared to native grasses (native grasses disturbed: 8.9 ± 2.9%, native grasses undisturbed: 1.8 ± 2.9%) and herbs (native herbs disturbed: 2.4 ± 1.3%, native herbs undisturbed: 0.9 ± 1.3%; Table 1; marginally significant interaction disturbance x functional group x species origin).
Gastropod herbivory reduced the proportion of seedling recruitment (gastropod exclusion: 3.4 ± 1.2%, gastropod control: 5.5 ± 1.2%) but not the number of species with seedling recruitment (Table 1). Seedling recruitment of legumes was significantly reduced by gastropod herbivory in disturbed (legumes disturbed gastropod exclusion: 5.5 ± 2.0%, legumes disturbed gastropod control: 0.5 ± 2.0%) compared to undisturbed plots (legumes undisturbed gastropod exclusion: 4.8 ± 2.0%, legumes undisturbed gastropod control: 2.6 ± 2.0%; Table 1, significant interaction gastropod x disturbance x functional group). Moreover, gastropod herbivory reduced seedling recruitment of exotic species (exotics gastropod exclusion: 8.5 ± 1.6%, exotics gastropod control: 4.5 ± 1.6%), to a stronger extent as compared to native seedling densities (natives gastropod exclusion: 2.4 ± 1.4%, natives gastropod control: 2.6 ± 1.4%; Table 1, significant interaction gastropod x species origin). The interaction between gastropod herbivory and species origin, however, also varied between disturbed and undisturbed plots (Fig. 1a), as well as between the three functional groups (Fig. 2a). Gastropod herbivory reduced the positive effect of disturbance on seedling densities only for exotic but not for native species (Table 1, significant interaction gastropods x disturbance x species origin; Fig. 1a). The effects of disturbance and gastropod herbivory on the number of exotic and native species with seedling recruitment showed the same direction, but were not significant (Table 1, Fig. 1b). Nevertheless, compared to native legumes, the number of exotic legumes was lower in plots with gastropod herbivory while neither recruitment of exotic nor native grasses or non-legume herbs showed significant responses to gastropod herbivory (Table 1, significant three-way interaction gastropod x species origin x functional group; Fig. 2a).
Effect of gastropod herbivory (+Gas = gastropod control, −Gas = gastropod exclusion), disturbance (disturbed, undisturbed) and species origin (exotic, native) on (a) the proportion of seedling recruitment (number of recruited seedlings divided by the number of added seeds) and (b) the number of species with seedling recruitment. Note that data shown are the untransformed least square means ± SE of a linear mixed model.
(a) Effect of gastropod herbivory (+Gas = gastropod control, −Gas = gastropod exclusion), disturbance (disturbed, undisturbed) and species origin (exotic, native) on the on the proportion of seedling recruitment (number of recruited seedlings divided by the number of added seeds), (b) effect of rodent herbivory (+Rod = rodent control, −Rod = rodent exclusion) disturbance (disturbed, undisturbed) and species origin (exotic, native) on the proportion of seedling recruitment (number of recruited seedlings divided by the number of added seeds). Note that data shown are the untransformed least square means ± SE of a linear mixed model.
Overall, rodent herbivory had only weak effects on seedling recruitment. In contrast to expectations, the presence and not the absence of rodent herbivory increased the number of species with seedling recruitment by about two species at undisturbed (rodent exclusion undisturbed: 5.8 ± 0.8, rodent control undisturbed: 7.9 ± 0.8) compared to disturbed plots (rodent exclusion disturbed: 10.5 ± 0.8, rodent control disturbed: 10.4 ± 0.8, Table 1). Moreover, irrespective of species origin, the presence of rodents marginally increased the proportion of seedling recruitment of non-legume herbs, while there were was no such effect on grasses and legumes (Table 1, significant interaction of rodent herbivory x functional group; Fig. 2b).
What are the consequences for species diversity, composition and productivity of plant communities experimentally invaded by exotic and native species?
Generally, with seed addition total species richness was increased by about five species and with disturbance by about one species per 0.25 m2 plot (Table 2). Regardless of species origin disturbance in combination with seed addition increased total species richness by about three species (Fig. 3), and herb species richness about five species (Table 2). Across both study years (2012 and 2013) disturbance substantially changed the composition of the plant community (Fig. S1 and Table S1). The indicator species analysis showed that, consistently across both years, the exotic herb Dianthus giganteus benefitted the most from disturbance (Table S2). Furthermore, the grasses Bromus inermis and Bromus hordeaceus were indicators of disturbance in the first year, while the herbs Dipsacus sylvestris and Sanguisorba minor were associated with disturbance in the second year. However, disturbance and seed addition of exotic and native species also interacted with each other and community composition was shifted towards sown exotic and native species particularly at disturbed plots (Fig. S1 and Table S1). Disturbance substantially reduced aboveground productivity by 30% (Table 2).
Effect of seed addition (control = no seed addition, exotic = exotic seed addition, native = native seed addition) and disturbance (undisturbed, disturbed) on total species richness (including resident and sown species). Data shown are least square means of repeated-measure linear mixed models ± SE averaged across two years. Note that species richness is given on a square root scale.
Gastropod herbivory significantly decreased total species richness by about one species and tended to reduce the positive effect of disturbance on total, legume and herb species richness (marginally significant interaction gastropod herbivory x disturbance, Table 2). Gastropod herbivory affected community composition in the second study year only, characterized by a decrease in the abundance of the resident legume species Trifolium pratense. Gastropod herbivory led to a moderate decrease in productivity by about 12%. In contrast to seedling recruitment the effect of gastropod herbivory on species richness and productivity was independent of species origin. Rodent herbivory alone had no effects on species richness, but community composition was affected by rodent herbivory across both study years (Table S1). This finding was also supported by the indicator species analysis (Table S2). Trifolium dubium was associated with rodent exclusion in the first study year, while the resident species Achillea millefolium and the sown native species Lactuca serriola were associated with the presence of rodents in the first and second study year, respectively.
Rodent and gastropod herbivory interacted to determine the species richness of our grassland community: rodent herbivory enhanced total species richness by about two species only in the absence of gastropod herbivory (Fig. 4). Moreover, rodent herbivory decreased the number of grasses in the presence of gastropod herbivory, although this effect was small (rodent control gastropod exclusion: 3.6 ± 1.1, rodent control gastropod control: 2.9 ± 1.1). These interactions between both herbivore guilds were also reflected in slight changes of the community composition within the first study year (Table S1), but there was no significant indicator species for this interaction.
Effect of gastropod herbivory (+Gas = gastropod control, −Gas = gastropod exclusion) and rodent herbivory (+Rod = rodent control, −Rod = rodent exclosure) on total species richness (including resident and sown species). Data shown are least square means of repeated-measure linear mixed models ± SE averaged across two years. Note that species richness is given on a square root scale.
For the species richness of legumes the direction of the rodent x gastropod herbivory interaction was additionally dependent on disturbance (Table 2): at disturbed plots, both gastropods and rodents reduced the number of legume species and their combined effects were mainly additive, while at undisturbed plots particularly the presence of rodents in the absence of gastropods increased the number of legume species compared to the absence of either rodents alone or both herbivore guilds.
Aboveground productivity was generally increased by seed addition, with exotic seed addition plots displaying a marginally significant higher increase in productivity than native seed addition plots (Fig. 5). Seed addition of native species increased total species richness more strongly than exotic seed addition (natives: 14.6 ± 0.7, exotics: 13.1 ± 0.7). When separating the total species richness into species richness of the three functional groups, particularly the number of native compared to exotic herbs (natives: 8.2 ± 0.5, exotics: 6.9 ± 0.4) increased following seed addition but not grasses or legumes. Moreover, plots experimentally invaded by exotic and native species changed in community composition (Fig. S1 and Table S1).
Effect of species origin (control = no seed addition, exotic = exotic seed addition, native = native seed addition) and disturbance (undisturbed, disturbed) on aboveground biomass. Data shown are least square means of repeated-measure linear mixed models ± SE averaged across two years. Note that aboveground biomass is given on a logarithmic scale.
Discussion
Our experiment provided important insights into processes determining seedling recruitment of native and exotic species as well as into the consequences for species richness, species composition and productivity. Against expectations, gastropod herbivory reduced the positive effect of disturbance during seedling recruitment for exotic but not for native species. Independent of species origin, rodent herbivory had weak but positive effects on seedling recruitment, particularly on that of herbs and on undisturbed plots. Contrary to the effects observed for seedling recruitment, interactive effects between gastropod herbivory, disturbance and species origin were not evident for species richness, composition and productivity. However, we found interactive effects of gastropod and rodent herbivory on community composition and species richness, which were not previously found on seedling recruitment. As expected, addition of exotic species led to a stronger increase in aboveground productivity compared to native seed addition, while the opposite was true for species richness, suggesting that the relationship between productivity and species richness was different for native and exotic species.
How do disturbance and the different herbivore guilds, separately and interactively, affect the establishment of exotic vs. native plant species?
As expected, the positive effect of disturbance on seedling recruitment was stronger for exotic compared to native species, which is in line with a study by Maron, et al.18 (but see refs2,20). However, this effect also depended on the affiliation to functional groups, as only exotic herbs and grasses responded positive to soil disturbance, while exotic legumes responded negative to soil disturbance. We can only speculate here, but a higher resource use efficiency and colonization ability of exotic species30,40 together with the different resource use strategies of the different functional groups (i.e. nitrogen-fixing legumes vs. non-fixing herbs and grasses) might explain the pattern observed in this study. However, we also have to note that the effect of disturbance on exotic vs. native species affiliated to different functional groups was not independent of gastropod herbivory.
Against our expectations and results from previous studies, gastropods suppressed exotic over native seedling recruitment. Thus, our study is the first one to demonstrate that selective feeding of gastropods on exotic species may represent a strong form of biotic resistance, at least during the establishment phase of exotic plants9,12. Cates and Orians42 found that exotic, early successional species were preferred by slugs in palatability tests, but whether such preferences result in different seedling densities in the field has only rarely been investigated2,15,16. Biotic resistance by gastropods against exotic plant species was mainly driven by the strong herbivore pressure on exotic legumes, which, in the case of the species included in our experiment, were all intentionally introduced as forage or cover crops43. We may therefore assume a human pre-selection of these species for high productivity and faster growth44, being associated with lower levels of resistance but probably higher levels of tolerance45. Lower herbivore resistance would be in line with the preference of gastropods for these exotics. Another important finding of our study was that gastropods reduced seedling recruitment of exotic legumes particularly at disturbed plots, which also explains the negative response of exotic legumes to disturbance (see above). Thus, gastropods can even diminish invasion opportunities provided by disturbances of the vegetation cover30,31. A possible mechanism underlying this result may be that disturbance increased the apparency of palatable exotic species for gastropods46.
In contrast to the strong negative effects of gastropod herbivory on seedling recruitment of exotic species, rodent herbivory had weak but positive effects on seedling densities of herbs, irrespective of their origin. We have to mention here, that due to the split-plot design of our study we had fewer replicates for the factor rodent herbivory (5 replicates) as it represents the main-plot level, compared to the factor gastropod herbivory (10 replicates) as it represents the subplot level, which leads to a lower power to detect significant effects of rodents compared to gastropods. Moreover, the presence of rodents increased the opportunities for sown species, in particular for legumes, to recruit at undisturbed plots. Most likely, both effects were indirectly mediated by their strong negative effects on aboveground biomass and thereby reducing competition for light. The consistently positive effect of rodent herbivory on the number of recruiting species, irrespective of their exotic or native origin, opposes several studies that provided evidence for biotic resistance by rodents in North American grassland13,14. Major players in these grasslands are granivorous rodents which have been frequently shown to selectively consume large-seeded exotic species, reducing establishment success and abundance of these species. However, in our Central European grassland we primarily find herbivorous rodents which do not selectively feed on large-seeded exotic species. From our study, we might suggest that herbivorous rodents rather facilitate the establishment of exotic (and native) species particularly at undisturbed plots likely by reducing completion for light. These findings may have important implications for our understanding of biological invasions and suggest that the fate of exotic species recruitment will depend on which herbivore guild dominates in a grassland system (i.e. facilitative effects of herbivorous rodents vs. suppressive effects of gastropods and granivorous rodents).
What are the consequences for species diversity, composition and productivity of plant communities experimentally invaded by exotic and native species?
Against expectations, changes in the density and number of recruiting species, as a result of the interactive effect between gastropod herbivory, disturbance and species origin, did not result in changes in species richness, composition and productivity. Probably this is related to the ability of plants to tolerate herbivore damage47. However, we found that rodent and gastropod herbivory showed opposing and interactive effects on species richness and community composition, an effect which was not seen during seedling recruitment. The positive effect of rodents on species richness, most likely indirectly mediated via strong negative effects on aboveground biomass, became only visible in the absence of gastropods that were feeding on sub-dominant species (particularly legumes), which benefitted from the release of aboveground competition in the presence of rodent herbivory. Allan and Crawley23 found non-additive effects for gastropods and insects: insects were feeding on dominant grasses, thereby increased species richness, but only if gastropods were absent as they suppressed sub-dominant herb or legume species. The opposing effects of gastropods and rodents on diversity may be related to their difference in body size. Compared to smaller sized herbivores, larger herbivores are frequently less selective in food choice, but have stronger effects on the performance or productivity of plants48,49. In our experiment, gastropods particularly reduced the numbers and abundance of legume species, known to be highly nutritious. This gastropod x rodent interaction was, however, not evident during early recruitment, where the effects of both herbivore guilds were independent of each other.
Finally, in agreement with our third hypothesis and the study by Maron, et al.27, addition of exotic species increased productivity to a greater extent than addition of native species, although this difference was only marginally significant. However, in contrast to our third hypothesis and findings by Maron, et al.27, the effect of exotic vs. native species on productivity was neither amplified by disturbance nor reduced by generalist herbivory. Independent of any alteration in resource availability caused by disturbance, exotic species may thus possess higher aboveground growth rates50, which is in line with observational studies finding increased productivity at invaded sites compared to uninvaded ones1. Moreover, the positive effect of seed addition on total species richness demonstrates that our grassland community was not saturated with species due to dispersal and seed limitation which frequently occurs in grassland systems51,52. As the effect of seed addition on species richness and composition was enhanced by disturbance, we suggest that dispersal limitation in combination with microsite limitation shape species richness and composition in our rather productive grassland community6. However, the magnitude of this disturbance x seed addition effect on species richness did not differ between species origins, thus resident native species mediate biotic resistance9 but similarly for native and exotic species27 but see53. A surprising result was that fewer exotic than native species were able to establish themselves in the absence of experimental disturbance, with the consequence that adding seeds of native species resulted in a stronger increase in species richness particularly at undisturbed plots. This result may indicate a stronger niche complementarity between resident species and native colonizers, possibly resulting from a common co-evolution54. This also supports the finding by Breitschwerdt, et al.52, that species which commonly co-occur with the resident vegetation have the better chances to colonize. Finally, our result that exotic species colonized in fewer numbers but produced more biomass is consistent with other experimental results suggesting that mechanisms driving the diversity-productivity relationship differ between native and exotic-dominated communities37,38,39,41.
Conclusions
Our multi-factorial experiment revealed major insights in how disturbance, herbivory by gastropods and rodents, and plant species origin separately, and interactively, affected seedling recruitment, biodiversity and ecosystem functioning, and thus advances our general understanding of ecological processes driving community assembly and dynamics. Quality and quantity of interactions between herbivores, disturbance and species origin differed, depending on whether seedling recruitment, community species richness, composition and productivity were considered as response. For example, during early establishment selective gastropod herbivory conferred biotic resistance particularly against exotic plant species, even balancing the positive influence of disturbance. No such herbivore x disturbance x species origin interaction could be observed for other properties of the plant community. The primary factor influencing how many exotic and native species colonized was disturbance, suggesting that biotic resistance through native competitors is a major determinant of colonization success of introduced plant species. However, the fewer exotic species which were able to colonize still had a stronger impact on the productivity of the community. Our study demonstrates the need to account for the interactions between multiple processes as drivers of early recruitment of invasive species, as well as to investigate underlying mechanisms of biodiversity-ecosystem functioning in exotic dominated “novel” vs. native communities.
Material and Methods
In 2011, we set up a multi-factorial split-split plot experiment at a grassland site at the field experimental station of the Helmholtz Centre for Environmental Research – UFZ at Bad Lauchstädt, Central Germany (51°23′29.47″N, 11°52′27.76″E). The moderately productive temperate grassland community was characterized by perennial grasses, such as Arrhenatherum elatius, Calamagrostis epigejos and Dactylis glomerata, herbs such as Taraxacum officinale and Plantago major, and the legumes Trifolium repens and Trifolium pratense. Abundant rodents were voles (mostly Microtus arvalis); in addition hares (Lepus europaeus) occurred occasionally. By far the most common gastropods were slugs from the native genus Deroceras and the invasive species Arion vulgaris16. However, recent molecular analyses question the invasive status of Arion vulgaris in Europe55. The grassland was mown regularly twice a year, in summer and autumn.
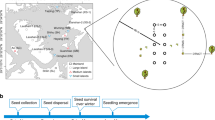
The split-split plot experiment was aranged in five blocks and included the experimental factors rodent herbivory, gastropod herbivory, disturbance and seed addition. Each of the five blocks contained a rodent exclosure and a rodent control plot. Within each of these plots one gastropod exclosure and gastropod control was set-up, summing up to each 10 gastropod exclosure and control subplots. Within each subplot we established six 0.5 × 0.5 m experimental sub-subplots. Each of the sub-subplots was randomly assigned to one of the six combinations of disturbance (yes/no) x seed-addition (no seeds, native or exotic seed mixture) (Fig. 6), summing up to a total of 120 sub-subplots.
The graph exemplifies the experimental design showing one out of five blocks. Each block includes one rodent exclusion plot (indicated by a bold solid line) and rodent control plot (indicated by a bold dashed line) plot. Within each plot, one gastropod exclusion subplot (indicated by a solid line) and gastropod control subplot (indicated by a dashed line) were established. Within each subplot, sub-subplots (depicted with smaller squares) were established that were either subjected to disturbance (shaded squares) or were left undisturbed (open squares). These sub-subplots were furthermore randomly assigned to receive no seeds (C), a seed mixture of 20 exotic species (E) or 20 native species (N).
Rodent exclosures (7 × 9.5 m) were constructed using a fence made of 6 mm mesh wire, buried 0.4 m belowground and extending 0.6 m aboveground, and topped with metal flashing to prevent rodents from climbing into the exclosure. We used bait boxes equipped with rodenticide to eliminate any rodents that might have entered the rodent exclosures. Gastropod exclosures (5 × 3.5 m) were constructed of a metal sheet, buried 0.1 m belowground and extending 0.2 m aboveground. The upper edge of the metal was bent outward and lubricated with lemon-based slug repellent (IRKA® “Schneckenabwehrpaste”, Germany), to prevent gastropods from climbing into the fence. Inside the exclosure we displayed slug baits based on metaldehyde (Spiess-Urania® “Schneckenkorn”) to kill gastropods which entered exclosures. Despite there were no infrastructure controls, we think that the side effects of our exclosures were rather negligible. First, there was a 1.75 m buffer strip between the bigger rodent exclosures and the experimental plots and a 0.75 m buffer strip between the smaller gastropod exclosures and the experimental plots, to keep shade effects as minimal as possible (see Fig. 6). Second, there was no obvious difference between rodent activity inside and outside gastropod exclosures. Moreover, we noticed herbivory by other herbivores, such as deer and aphids, inside and outside our exclosures.
In late October 2011, sub-subplots either received no seeds (control), a native or an exotic seed mixture each consisting out of 20 species. Out of the sown species pool the native species Daucus carota, Dactylis glomerata, Bromus hordeaceus, Vicia tetrasperma and the exotic species Solidago canadensis were already present in the grassland community albeit at low abundance, while all other exotic and native species were absent from that site. Native and exotic species pools were kept as similar as possible with respect to distribution of seed size, life span and functional group identity (Table S1). Seeds of all species were collected in wild-growing populations in Central Germany in summer and autumn 2011 just prior to the start of the experiment. Seeds were cleaned and weighed to determine mean propagule weight per species. To account for the fact that small-seeded species typically produce more seeds than large-seeded species we added 50, 100 or 175 seeds per species depending on their propagule weight (Table S1). Prior to adding seeds, half of the sub-sub-subplots were disturbed by removing the aboveground vegetation and turning the top 0.15 to 0.20 m of soil to reduce competition from resident vegetation.
Measurements
The identity and number of seedlings of all species was determined on all 0.25 m2 sub-subplots in April 2012, including control sub-subplots to account for spontaneous recruitment from the seed bank and colonization from surrounding vegetation. Moreover, we quantified how our treatments subsequently influenced total species richness and composition of the studied grassland community in summer 2012 and 2013. At every census date we recorded the abundance of all species occurring on a certain sub-subplot including control sub-subplots. Central European semi-natural, extensively used grasslands need to be managed to be maintained and conserved, because otherwise succession will take place and bushes will encroach. Imitating the common management practice of such grasslands, we harvested aboveground biomass in July and September, both in 2012 and 2013. Bagged samples were dried at 60 °C for 48 hours and weighed.
Statistical analysis
How do disturbance and the different herbivore guilds, separately and interactively, affect the establishment of exotic vs. native plant species?
To examine how rodent herbivory, gastropod herbivory and disturbance and the affiliation to functional groups affected seedling recruitment of sown species, we calculated the number of exotic and native species producing seedlings on the 0.25 m2 sub-subplot minus their values at the respective control sub-subplot. As residuals were already normally distributed, we used the untransformed data of the number of species with seedling recruitment as response variable and applied generalized linear mixed model with Gaussian error distribution. Rodent exclusion, gastropod exclusion, disturbance, species origin were considered fixed factors. Following the split-split-plot design of our experiment, block, block x plot (mainplot error) and block x plot x subplot (subplot error) were included as random effects in the model. The effects of disturbance, seed addition and their interactions were tested against the residual error term.
For those sown exotic and native species with successful recruitment (see above), we calculated the difference between seedling numbers in seed addition sub-subplots and those in control sub-subplots to account for spontaneous colonization from the seed bank or from the surrounding vegetation. We then used the corrected numbers of recruited individuals in relation to the number of added propagules per species as response variable (i.e. proportion of seedling recruitment). Because a binomial model did not converge, data were logit-transformed and linear mixed models were applied56. Rodent exclusion, gastropod exclusion, disturbance, species origin and functional group were considered fixed factors, while species nested within functional group and species origin were considered random factors. Moreover, following the split-split-plot design of our experiment, block, block x plot (mainplot error) and block x plot x subplot (subplot error) were included as random effects in the model. The effects of disturbance, seed addition and their interactions were tested against the residual error term. For the two response variables (number of species with seedling recruitment and the proportion of seedling recruitment) only sub-subplots with exotic and native species addition were included in the analysis. Moreover, in case of significant interactions we tested simple main effects (i.e. the effect of one factor at each level of the other factors57,58; using the SLICE option in SAS.
What are the consequences for species diversity, composition and productivity of plant communities experimentally invaded by exotic and native species?
In contrast to the analysis on seedling recruitment, all sub-subplots including the controls were incorporated in the analysis of total species richness and productivity. Total species richness per sub-subplot was calculated from the number of sown plus resident species. To distinguish how the different functional groups (i.e. grasses, herbs, legumes) responded to our experimental treatments we also calculated species richness including resident species for the functional groups separately. Productivity was assessed as the sum of both aboveground biomass harvests (July and September). For the statistical analysis total species richness and species richness of the three functional groups were square root-transformed while productivity was log-transformed. We used separate repeated-measures linear mixed models to test the effect of our experimental treatments on species richness (i.e. total, grass, herb and legume species richness) as well as aboveground productivity. Here, block, block x plot and block x plot x subplot were considered random effects in the between-subject part of the model, while the two replicates in time (2012, 2013) were considered random in the within subject part of the model. Sub-subplots were tested against residuals of the between subject model. We used orthogonal contrasts to test the a priori expectations and decompose significant interactions. Following our third hypothesis, for aboveground biomass and total species richness we tested whether seed addition sub-subplots differed from seed addition control sub-subplots, whether exotic seed addition differed from native seed addition and whether this effect was enhanced by disturbance.
To determine the effects of all experimental treatments on the species composition in 2012 and 2013 we applied different multivariate analysis techniques based on the abundances of recorded species, separately by census year. We calculated permutational multivariate analysis of variance (PERMANOVA). Rodent herbivory, gastropod herbivory, disturbance and species origin were considered fixed effects while block was considered random (“strata”). Differences in vegetation composition in response to treatments were visualized by using nonmetric multidimensional scaling (NMDS) based on Bray-Curtis dissimilarities and k = 4 dimensions. Furthermore, we calculated indicator species analysis (INDVAL) to determine which species were associated with our experimental treatments. Both PERMANOVA and INDVAL analysis were based on 999 permutations. All multivariate analysis were calculated with R 3.0.1 using the vegan59 and indicspecies package60, while all other analyses were done with SAS 9.2 using the GLIMMIX procedure.
Change history
08 March 2018
A correction to this article has been published and is linked from the HTML and PDF versions of this paper. The error has not been fixed in the paper.
References
Vilà, M. et al. Ecological impacts of invasive alien plants: a meta-analysis of their effects on species, communities and ecosystems. Ecol. Lett. 14, 702–708 (2011).
Mueller, G., Horstmeyer, L., Roenneburg, T., van Kleunen, M. & Dawson, W. Alien and native plant establishment in grassland communities is more strongly affected by disturbance than above-and below-ground enemies. J. Ecol. 104, 1211–1218 (2016).
Sakai, A. K. et al. The population biology of invasive specie. Annu. Rev. Ecol. Sys., 305–332 (2001).
Grubb, P. J. The maintenance of species-richness in plant communities: the importance of the regeneration niche. Biol. Rev. 52, 107–145 (1977).
Myers, J. A. & Harms, K. E. Seed arrival, ecological filters, and plant species richness: a meta‐analysis. Ecol. Lett. 12, 1250–1260 (2009).
Turnbull, L. A., Crawley, M. J. & Rees, M. Are plant populations seed‐limited? a review of seed sowing experiments. Oikos 88, 225–238 (2000).
Mitchell, C. E. et al. Biotic interactions and plant invasions. Ecol. Lett. 9, 726–740 (2006).
Catford, J. A. et al. The intermediate disturbance hypothesis and plant invasions: Implications for species richness and management. Plant Ecol. Evol. Syst. 14, 231–241 (2012).
Levine, J. M., Adler, P. B. & Yelenik, S. G. A meta‐analysis of biotic resistance to exotic plant invasions. Ecol. Lett. 7, 975–989 (2004).
Keane, R. M. & Crawley, M. J. Exotic plant invasions and the enemy release hypothesis. Trends Ecol. Evol. 17, 164–170 (2002).
Schaffner, U. et al. Plant invasions, generalist herbivores, and novel defense weapons. Ecology 92, 829–835 (2011).
Maron, J. L. & Vilà, M. When Do Herbivores Affect Plant Invasion? Evidence for the Natural Enemies and Biotic Resistance Hypotheses. Oikos 95, 361–373 (2001).
Pearson, D. E., Callaway, R. M. & Maron, J. L. Biotic resistance via granivory: establishment by invasive, naturalized, and native asters reflects generalist preference. Ecology 92, 1748–1757 (2011).
Connolly, B. M., Pearson, D. E. & Mack, R. N. Granivory of invasive, naturalized, and native plants in communities differentially susceptible to invasion. Ecology 95, 1759–1769 (2014).
Strauss, S. Y. et al. Cryptic seedling herbivory by nocturnal introduced generalists impacts survival, performance of native and exotic plants. Ecology 90, 419–429 (2009).
Korell, L. et al. Stronger effect of gastropods than rodents on seedling establishment, irrespective of exotic or native plant species origin. Oikos 125, 1467–1477 (2016).
Hahn, P. G. & Dornbush, M. E. Exotic consumers interact with exotic plants to mediate native plant survival in a Midwestern forest herb layer. Biol. Invasions 14, 449–460 (2012).
Maron, J. L., Pearson, D. E., Potter, T. & Ortega, Y. K. Seed size and provenance mediate the joint effects of disturbance and seed predation on community assembly. J. Ecol. 100, 1492–1500 (2012).
Crawley, M. J. Herbivory. The dynamics of animal-plant interactions (Blackwell Scientific Publications, 1983).
Kempel, A., Chrobock, T., Fischer, M., Rohr, R. P. & van Kleunen, M. Determinants of plant establishment success in a multispecies introduction experiment with native and alien species. Proc. Natl. Acad. Sci. USA 110, 12727–12732 (2013).
Loranger, J. et al. Predicting invertebrate herbivory from plant traits: evidence from 51 grassland species in experimental monocultures. Ecology 93, 2674–2682 (2012).
Latorre, L., Larrinaga, A. R. & Santamaría, L. Combined impact of multiple exotic herbivores on different life stages of an endangered plant endemism, Medicago citrina. J. Ecol. 101, 107–117 (2013).
Allan, E. & Crawley, M. J. Contrasting effects of insect and molluscan herbivores on plant diversity in a long-term field experiment. Ecol. Lett. 14, 1246–1253 (2011).
Scheidel, U. & Bruelheide, H. Selective slug grazing on montane meadow plants. J. Ecol. 87, 828–838 (1999).
Stenseth, N., Hansson, L. & Myllymäki, A. Food selection of the field vole Microtus agrestis. Oikos, 511–524 (1977).
Lantová, P. & Lanta, V. Food selection in Microtus arvalis: the role of plant functional traits. Ecol Res 24, 831–838 (2009).
Maron, J. L. et al. Staged invasions across disparate grasslands: effects of seed provenance, consumers and disturbance on productivity and species richness. Ecol. Lett. 17, 12250 (2014).
Hulme, P. E. Seedling Herbivory in Grassland: Relative Impact of Vertebrate and Invertebrate Herbivores. J. Ecol. 82, 873–880, doi:10.2307/2261451 (1994).
Hahn, P. G., Draney, M. L. & Dornbush, M. E. Exotic slugs pose a previously unrecognized threat to the herbaceous layer in a midwestern woodland. Restor. Ecol. 19, 786–794 (2011).
Davis, M. A., Grime, J. P. & Thompson, K. Fluctuating resources in plant communities: a general theory of invasibility. J. Ecol. 88, 528–534 (2000).
Shea, K. & Chesson, P. Community ecology theory as a framework for biological invasions. Trends Ecol. Evol 17, 170–176 (2002).
Jauni, M., Gripenberg, S. & Ramula, S. Non-native plant species benefit from disturbance: a meta-analysis. Oikos 124, 122–129, doi:10.1111/oik.01416 (2014).
Maron, J. L. & Crone, E. Herbivory: effects on plant abundance, distribution and population growth. Proc. Roy. Soc. B 273, 2575–2584 (2006).
Lake, J. C. & Leishman, M. R. Invasion success of exotic plants in natural ecosystems: the role of disturbance, plant attributes and freedom from herbivores. Biol. Cons. 117, 215–226 (2004).
St Clair, S. B., O’Connor, R., Gill, R. & McMillan, B. Biotic resistance and disturbance: rodent consumers regulate post‐fire plant invasions and increase plant community diversity. Ecology 97, 1700–1711 (2016).
Fenner, M. & Thompson, K. The ecology of seeds. (Cambridge University Press, 2005).
Wilsey, B. J., Teaschner, T. B., Daneshgar, P. P., Isbell, F. I. & Polley, H. W. Biodiversity maintenance mechanisms differ between native and novel exotic‐dominated communities. Ecol. Lett. 12, 432–442 (2009).
Cook-Patton, S. C. & Agrawal, A. A. Exotic plants contribute positively to biodiversity functions but reduce native seed production and arthropod richness. Ecology 95, 1642–1650 (2014).
Kuebbing, S. E., Classen, A. T., Sanders, N. J. & Simberloff, D. Above- and below-ground effects of plant diversity depend on species origin: an experimental test with multiple invaders. New Phytol. 208, 727–735 (2015).
Van Kleunen, M., Weber, E. & Fischer, M. A meta‐analysis of trait differences between invasive and non‐invasive plant species. Ecol. Lett. 13, 235–245 (2010).
Korell, L., Schmidt, R., Bruelheide, H., Hensen, I. & Auge, H. Mechanisms driving diversity–productivity relationships differ between exotic and native communities and are affected by gastropod herbivory. Oecologia 180, 1025–1036 (2016).
Cates, R. G. & Orians, G. H. Sucessional status and the palatability of plants to generalized herbivores. Ecology, 410–418 (1975).
Hanelt, P. Mansfeld’s Encyclopedia of Agricultural and Horticultural Crops. 1st edn. edn, (Springer, 2001).
Van Kleunen, M., Dawson, W. & Maurel, N. Characteristics of successful alien plants. Mol. Ecol. 24, 1954–1968 (2015).
Chun, Y. J., Van Kleunen, M. & Dawson, W. The role of enemy release, tolerance and resistance in plant invasions: linking damage to performance. Ecol. Lett. 13, 937–946 (2010).
Hahn, P. G. & Orrock, J. L. Land‐use legacies and present fire regimes interact to mediate herbivory by altering the neighboring plant community. Oikos 124, 497–506 (2015).
Ashton, I. W. & Lerdau, M. T. Tolerance to herbivory, and not resistance, may explain differential success of invasive, naturalized, and native North American temperate vines. Divers. Distrib. 14, 169–178 (2008).
Olff, H. & Ritchie, M. E. Effects of herbivores on grassland plant diversity. Trends Ecol. Evol. 13, 261–265 (1998).
Ritchie, M. & Olff, H. Herbivore diversity and plant dynamics: compensatory and additive effects. In: Herbivores between plants and predators/ed. by H. Olff, VK Brown & RH Drent.-Oxford: Blackwell Science, 1999 (1999).
Blumenthal, D. M. & Hufbauer, R. A. Increased plant size in exotic populations: a common-garden test with 14 invasive species. Ecology 88, 2758–2765 (2007).
Stein, C., Auge, H., Fischer, M., Weisser, W. W. & Prati, D. Dispersal and seed limitation affect diversity and productivity of montane grasslands. Oikos 117, 1469–1478 (2008).
Breitschwerdt, E., Jandt, U. & Bruelheide, H. Do newcomers stick to the rules of the residents? Designing trait‐based community assembly tests. J. Veg. Sci. 26, 219–232 (2015).
Bennett, J. A. et al. Species pools, community completeness and invasion: disentangling diversity effects on the establishment of native and alien species. Ecol. Lett. 19, 1496–1505 (2016).
Thorpe, A. S., Aschehoug, E. T., Atwater, D. Z. & Callaway, R. M. Interactions among plants and evolution. J. Ecol. 99, 729–740 (2011).
Pfenninger, M., Weigand, A., Bálint, M. & Klussmann-Kolb, A. Misperceived invasion: the Lusitanian slug (Arion lusitanicus auct. non-Mabille or Arion vulgaris Moquin-Tandon 1855) is native to Central Europe. Evol. Appl. 7, 702–713 (2014).
Warton, D. I. & Hui, F. K. The arcsine is asinine: the analysis of proportions in ecology. Ecology 92, 3–10 (2011).
Woodward, J. A. & Bonett, D. G. Simple main effects in factorial designs. J. Appl. Stat., 255–264 (1991).
Schabenberger, O., Gregoire, T. G. & Kong, F. Collections of simple effects and their relationship to main effects and interactions in factorials. Am. Stat. 54, 210–214 (2000).
Oksanen, J. Multivariate analysis of ecological communities in R: vegan tutorial. R package version 1 (2011).
De Cáceres, M., Legendre, P. & Moretti, M. Improving indicator species analysis by combining groups of sites. Oikos 119, 1674–1684 (2010).
Acknowledgements
We thank the many persons who helped with the collection of field data. Particularly, we thank the staff of the field experimental station of the Helmholtz Centre for Environmental Research - UFZ as well as C. Rosche, F. Walter and R. Schmidt. We acknowledge L. Petter for assistance with slug identification in Germany. We are grateful for valuable comments on the manuscript by C. Roscher and M. Schädler. This research was supported by the graduate school HIGRADE (32-G02110-532) and institutional funds of the Institute of Biology / Geobotany and Botanical Garden at Martin Luther University Halle-Wittenberg to LK.
Author information
Authors and Affiliations
Contributions
L.K., I.H., H.A. and H.B. designed the experiment. L.K. and B.L. performed the experiment and analyzed the data. L.K. wrote the manuscript and B.L., I.H., H.A., and H.B. contributed to the revision of the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
A correction to this article is available online at https://doi.org/10.1038/s41598-018-22518-z.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Korell, L., Lang, B.R., Hensen, I. et al. Interactions count: plant origin, herbivory and disturbance jointly explain seedling recruitment and community structure. Sci Rep 7, 8288 (2017). https://doi.org/10.1038/s41598-017-08401-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-08401-3
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.