Abstract
Endocarpon species are key components of biological soil crusts. Phenotypic and systematic molecular analyses were carried out to identify samples of Endocarpon collected from the southeast edge of the Tengger Desert in China. These morphological and molecular analyses revealed two previously undescribed species that form highly supported independent monophyletic clades within Endocarpon. The new taxa were named Endocarpon deserticola sp. nov. and E. unifoliatum sp. nov. Furthermore, our results indicated that the newly developed protein coding markers adenylate kinase (ADK) and ubiquitin-conjugating enzyme h (UCEH) are useful for assessing species boundaries in phylogenic analyses.
Similar content being viewed by others
Introduction
Biological soil crusts (BSCs) are intimate association between soil particles and biological communities composed of mosses, lichens, cyanobacteria and heterotrophs living at the soil surface1, 2. Soil particles are aggregated through the presence and activity of the biota mentioned above, and the resultant living crusts cover more than 40% of the Earth’s terrestrial surface as a coherent layer1, 2. BSCs play an important role in carbon and nitrogen fixation and soil stabilization of desert ecosystems2,3,4. According to the existence of different dominant species during the development of BSCs, it could be mainly divided into algae crust, lichen crust and moss crust5, among which the lichen crust is more compact and has stronger ability in carbon and nitrogen fixation6.
The lichen-forming fungal genus Endocarpon Hedw. is a key component of BSCs in the arid and semiarid regions, which can aggregate soil particles using squamulose thallus or rhizines at their lower surface1. Endocarpon crusts play important ecological roles in desert ecosystems, such as stabilising sand dunes7, preventing soil wind erosion8, and promoting soil fertility4. In China, this genus has been reported as dominant group growing in Shapotou Region9, where is the southeast edge of the Tengger Desert (Fig. 1). As dominant species, Endocarpon pusillum Hedw. has been intensively studied from many aspects, such as physiology under desiccation and starvation stress10, photosynthetic rate11, genome12, transcriptome13 and stress resistance functional protein14. And all the above results supported that Endocarpon pusillum has good drought resistant ability, which could be one potential species used in the prospective ‘desert biocarpet engieering’ (DBCE)15, which has been proposed by Wei16, referring to developing during a short period into a dominant protection system instead of the naked desert with the help of the artificial inoculation of the microorganisms.
The location of sampling site and field overview. (A) The location of the sampling site in China, highlighted with orange color (created using R3.4.0); (B) Partial magnification of the detailed sampling site marked by a solid red triangle, situated in Ningxia Hui Autonomous Region and the southeast edge of the Tengger Desert (created using the drawing tool software Microsoft Paint (Windows 8.0); (C) Field overview of the sampling site; (D) Detailed view of Endocarpon spp. in the BSC.
The genus Endocarpon belongs to the Verrucariaceae, Verrucariales, Chaetothyriomycetidae, Eurotiomycetes, and Ascomycota17 and is characterised by a squamulose thallus, immersed perithecioid ascomata with hymenial algae, and asci with (1-)2(-8) muriform ascospores. The key characteristics delimiting Endocarpon species generally include squamule morphology and colour, presence or absence and colour of rhizines, amounts of perithecia and ascospores, and ascospore size. Although molecular data have greatly strengthened our understanding of the evolution and phylogenetic relationships of lichen-forming fungi, resulting in dramatic changes in their classification over the past few decades18,19,20,21,22,23,24, the phylogenetic relationships in Endocarpon are still not well understood. Until now, only a few studies have provided insight into the molecular phylogeny of a limited number of Endocarpon species, including E. pusillum, E. adscendens (Anzi) Müll. Arg., E. crystallinu J.C. Wei & Jun Yang, E. tenuissimum (Degel.) Lendemer & E.A. Tripp, E. psorodeum (Nyl.) Th. Fr., E. petrolepideum (Nyl.) Nyl. and E. pallidulum Ach9, 25,26,27,28,29,30. Globally, the genus comprises approximately 50 species31 based on studies conducted in China15, 16, 32,33,34,35,36 and other regions of the world37,38,39,40,41,42,43,44,45.
In order to seek for new species resource with sand-stabilisation potential besides Endocarpon pusillum, we carried out taxonomic study on Chinese Endocarpon and two specific species drew our attention because they were not assignable to any known species based on phenotypic characteristics. In view of insufficiency of phylogenetic data in the genus Endocarpon, we aim to study the two species based on both phenotypic traits and phylogeny and accumulate more DNA data for the further study.
Nowadays, besides nrDNA ITS region, which is often used in fungal species delimitation46, some protein-coding genes (e.g., RPB1, MCM7) have also been successfully used47. In this study, two protein-coding genes (ADK and UCEH) were developed and used for the first time for phylogenetic analyses. ADK is a phosphotransferase enzyme that catalyses the interconversion of adenine nucleotides, thus playing an important role in cellular energy homeostasis48. UCEH is a subunit of ubiquitin-conjugating enzymes and performs the second step of the ubiquitination reaction that targets a protein for degradation by the proteasome49. We newly designed the primers for ADK and UCEH based on the cDNA library of E. pusillum 50 in this study. Our major goals are (1) to describe the phenotypic characteristics of these two species, (2) understand the phylogenetic relationships in the genus Endocarpon and (3) as well as develop two new protein coding markers (ADK, UCEH) to strengthen the phylogenetic information.
Results
Molecular phylogeny
The aligned matrix contained 476 unambiguous nucleotide position characters for the internal transcribed spacer (ITS), 679 for the adenylate kinase (ADK), and 279 for the ubiquitin-conjugating enzyme h (UCEH). The final alignment of the concatenated data set was 1434 positions in length. Seventy-six sequences were newly generated for this study (Table 1).
The single-locus gene trees for the three markers are illustrated in Figures S1–S3. The topologies of the single-locus phylogenies did not exhibit obviously supported conflicts (i.e. bootstrap values ≥75%), and thus they were analysed in a concatenated data matrix. The best-fitting models corresponding to the three single-locus markers are listed in Table 2.
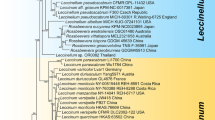
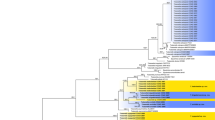
The concatenated three-locus data sets contained 40 sequences (1434 nucleotides sites), comprising 11 Endocarpon species. The maximum likelihood (ML) tree for the concatenated data sets constructed using partitioned models are presented in Fig. 2. The maximum likelihood analyses (ML, RAxML) shows the same highly supported clades as the Bayesian analyses. Both analyses were merged in one phylogenic tree, and the respective values (bootstrap values ≥75, posterior probability values ≥95) were plotted directly on the branches (Fig. 2). Based on the phylogenetic results the genus Endocarpon forms a strongly supported monophyletic clade and is obviously separated from the other genera within Verrucaricaceae, i.e., Dermatocarpon spp., Staurothele spp., Verrucaria spp. and Willeya spp. Within the Endocarpon clade, all 11 studied species formed highly supported lineages.
The maximum likelihood tree of Endocarpon species based on the concatenated ITS, ADK and UCEH sequences using the partition model. The numbers in each node represent bootstrap support (BS) and posterior probability (PP) values based on Bayesian analysis; numbers lower than 70 (BS) and 0.95 (PP) are not shown. Bootstrap values ≥75 and posterior probability values ≥95 were plotted on the branches of the RAxML tree. Newly generated sequences are marked with the symbol. combined with closely related sequences downloaded from GenBank. Scale = 0.05 substitution per site.
Scanning electron microscope (SEM) images of rhizines in the two new species
Both of the new Endocarpon species may fulfil potentially important roles by stabilising soils via sand particles consolidation with their rhizines, as inferred from SEM observations (Fig. 3). The sand particle surface is covered by the squamose thalli of the Endocarpon species (Fig. 1C), and sand crystals are wrapped in their branched rhizines (Fig. 3).
Discussion
Phenotype
According to our morphological assessment, some samples collected from the Tengger Desert in China were not able to be categorised as any previously described Endocarpon species9, 15, 31,32,33,34,35,36,37,38,39,40,41,42,43,44,45. Samples corresponding to the newly described Endocarpon deserticola are characterised by abundant perithecia dispersed throughout nearly all squamules, and the perithecia reach 15–60 (up to 100) in number (Fig. 4A). This species is most similar to E. helmsianum Müll. Arg. of Australia, which is also characterised by abundant perithecia41; however, E. helmsianum exhibits wider squamules (5–25 mm), a more contiguous to overlapping thallus, and much larger ascospores41.
Endocarpon deserticola: (A) Upper surface of squamae with abundant perithecia (holotype, Z07090), scale bar = 1 mm; (B) an ascus containing two ascospores (paratype, SPT3–10), scale bar = 10 µm. Endocarpon unifoliatum: (C) upper surface of unifoliate squama with slightly upturned margins (holotype, Z10020), the arrow pointing to white portion of thallus, scale bar = 0.5 mm; (D) muriform ascospores (paratype, SPT10063), scale bar = 10 µm. (E) Anatomic structure of perithecia of Endocarpon deserticola (holotype, Z07090), the arrow pointing to ascospores, scale bar = 50 µm. (F) Anatomic structure of perithecia of Endocarpon unifoliatum (holotype, Z10020), scale bar = 100 µm; (G) Anatomic structure of thallus (holotype, Z10020), the arrow pointing to white part of upper cortex, scale bar = 50 µm. (H) Partial magnification of anatomic structure of thallus (holotype, Z10020), the arrow pointing to white portion of the upper cortex and indicating less to absence of algal cells in this part, scale bar = 20 µm.
Samples corresponding to Endocarpon unifoliatum are characterised by a unifoliate, concave, and lobated thallus with slightly upturned margins (Fig. 4C). This species is similar to E. pusillum Hedw., but distinguished by plane, fully adnate thallus and tightly aggregated, nearly inseparable squamules in E. pusillum 42.
Molecular phylogeny
The genus Dermatocarpon with foliose umbilicate thallus was strongly supported (BS = 100%, PP = 1) separating from other genera with crustose or squamulose thallus (Endocarpon, Staurothele and Verrucaria). Another genus with hymenial algae besides Endocarpon, Staurothele, clustered closer to Verrucaria (BS = 77%, PP = 0.99) than to Endocarpon.
Within the Endocarpon clade was no explicit interspecific relationship reflected in the phylogenetic tree (Fig. 2), which may be the results of the limited number of species and gene loci included in the analyses. Nevertheless, each species was highly supported and obviously separated from others, e.g. the two new putative species Endocarpon deserticola and Endocarpon unifoliatum, formed two strongly supported clades separated from other species. The two newly developed protein coding markers (ADK and UCEH) show a quite high phylogenetic informative signal on species level and can be useful for future phylogenetic analyses, not only for Endocarpon, but also for other fungal genera (Figures S1–S3). This finding confirms the reliability of gene trees for phylogenetic analyses based on concatenated data sets.
The species Endocarpon tenuissimum is nested within the monophyletic Willeya diffractella (Nyl.) Müll. Arg. and has been placed in synonymy30, which is also supported by the ML tree based on ITS sequences in the present study (Figure S1). However, within the monophyletic Willeya diffractella clade, neither morphology nor geography was found to be corresponding to the main infraspecific groupings except the nature of the substrate (calcareous vs non-calcareous). As known Endocarpon tenuissimum shared the same character, i.e. non-calcareous substrate, with one group of Willeya diffractella, but there are some subtle differences in thallus color, ecology and ascospore size between Endocarpon tenuissimum and Willeya diffractella 30. More samples and genes are required to further explore the consistency between phenotype and phylogeny within Willeya diffractella.
Role of rhizines in soil stabilisation
Both new species of Endocarpon may fulfil important roles by stabilising soils by sand particle consolidation with their rhizines, which were inferred from SEM observations (Fig. 3). This finding is consistent with previous studies investigating lichenised BSC fungal communities in desert ecosystems15, 51. Lichens are more effective than cyanobacteria at reducing soil erosion because the fungal hyphae of the lichen thallus penetrate more deeply and the tissue extends above the soil surface52.
In summary, based on morphological and molecular phylogenetic data, two new putative species, Endocarpon sp. 1 and Endocarpon sp. 2, have been described under the names Endocarpon deserticola and Endocarpon unifoliatum, respectively. In previous studies, the lichen species Endocarpon pusillum exhibited drought resistance15 and sand and carbon fixation9, 11, and thus served as an important species for desert bio‐carpet engineering and the study of stress tolerance mechanisms in lichens in China12,13,14. The two new species, E. deserticola and E. unifoliatum, exhibit different morphological and phylogenetic characteristics from those of E. pusillum and may also play important roles in desert sand stabilisation. Further characterisation of features such as stress tolerance and carbon fixation should be carried out in the near future. Additionally, genomic studies should be performed to learn more about the mechanisms of stress tolerance in lichens.
Taxonomy
Endocarpon deserticola T. Zhang, X. L. Wei & J. C. Wei, sp. nov., Fig. 4A,B
Fungal Name: FN570287
TYPUS: China, Ningxia Hui Autonomous Region, Zhongwei city, Shapotou north experimental zone, on soil crust, January 2007, Zhangtao, Z07090 (holotypus-HMAS-L-135062).
Etymology: The epithet of the new species ‘deserticola’ is a Latin compound consisting of the Latin noun ‘desertum: desert’ and the Latin adjective suffix ‘-colus: inhabiting’, meaning that the new species grows in the desert.
Diagnosis: This species is characterised by its abundant perithecia, which is dispersed throughout almost all squamules, up to 100 or more in number.
Morphology: Thallus terricolous, squamulose; squamules solitary or contiguous, with slightly upturned margins, rounded, elongate or irregular, 1–3 (−4) mm in width; upper surface pale brownish to brownish; lower cortex well-developed, dark brown to black, with brown to black rhizines, 4–6 mm long, irregularly branching in the terminal region.
Upper cortex 19.5–27 (−37.5) µm thick, consisting of three layers: amorphous layer 15–22.5 (−30) µm thick; middle layer pale brown, 7.5–15 (−18) µm thick, paraplectenchymatous; lower layer hyaline, 22.5–37.5 µm thick; algal layer 45–60 (−67.5) µm thick, consisting of coccoid green microalgae, algal cells globose (diameter 3–6 µm) or subglobose (4.5–7.5 × 3–4.5 (−6) µm) and bright green; medulla pale whitish, 15–45 (−60) µm thick, without sharp demarcation between it and algal layer; lower cortex (from 22.5 to) 30–45 µm thick, dark brown to black.
Ascomata perithecioid, subglobose, immersed in thallus, 15–60 (−100) per squamule, brown to dark brown. Perithecia obpyriform, 200–275 (−325) × 200–250 µm; excipulum dark brown, 30–45 µm thick at the base and sides, pale brown at the apex near the ostiole; periphyses mostly simple, 22.5–37.5 (−45) µm long; hymenial algal cells globose (diameter 3–4.5 µm) to subglobose (3–4.5 × 1.5–3 µm), green; asci bisporous, clavate, 67.5–75 × 15–24 µm; ascospores muriform, two per ascus, hyaline to slightly brownish, matured spores brown, elongate-ellipsoid to subcylindrical, with 2–5 (−6) transverse divisions and 6–12 (−15) longitudinal divisions; upper spore generally broader and shorter than lower spore; upper spore: 28.5–39 × 18–22.5 µm; lower spore (from 31.5 to) 37.5–45 × 13.5–18 µm.
Pycnidia not seen.
Chemistry: K-, C-, KC-, P-; no lichen substances detected using thin-layer chromatography (TLC).
Substrate: Calcareous sands.
Additional specimens examined: China, Ningxia Autonomous region: Zhongwei city: Shapotou north experimental zone: soil crust, April 8, 2010, T. Zhang, Z10010 (HMAS-L-134712); soil crust, August 31, 2010, T. Zhang, SPT3–10 (HMAS-L-134716); soil crust, April 10, 2010, T. Zhang, SPT10078 (HMAS-L-121580); soil crust, April 18, 2007, J. Yang & T. Zhang, SPT295 (HMAS-L-134714); soil crust, August 6, 2003, J. C. Wei, SPT005 (HMAS-L-134713).
Comments: This species is most similar to E. helmsianum found in Australia, which is also characterised by abundant perithecia. However, E. helmsianum has wider squamules (5–25 mm), a more contiguous-to-overlapping thallus, and much larger ascospores.
Endocarpon unifoliatum T. Zhang, X. L. Wei & J. C. Wei, sp. nov., Fig. 4C–F
Fungal Name: FN570274
TYPUS: China, Ningxia Hui Autonomous Region, Zhongwei city, Shapotou north experimental zone, on soil crust, April 8, 2010, T. Zhang, Z10020 (holotypus-HMAS-L-134711).
Etymology: The epithet of the new species ‘unifoliatum’ is the nominative singular neuter of the Latin adjective ‘unifoliatus: with one thallus’.
Diagnosis: This species is characterised by its unifoliate, concave, lobate thallus with slightly upturned margins.
Morphology: Thallus terricolous, squamulose, concave, and lobate, sometimes greyish-white to white at the central part of thallus, brown at the thallus edges; squamules mostly solitary, not contiguous, with slightly upturned margins, rounded, elongate or irregular, 1–2 (−4) mm wide; upper surface pale to yellowish brown; lower cortex well developed, dark brown to black, with black rhizines 2–3 mm long, irregularly branching in the terminal region.
Upper cortex 19.5–27 (−37.5) µm thick, consisting of three layers: amorphous layer 1.5–3 µm or absent; middle layer dark brown, 12–15 µm thick, paraplectenchymatous; the lower layer hyaline, 7.5–12 (−22.5) µm thick. Algal layer 30–45 (−52.5) µm thick, consisting of coccoid green microalgae, algal cells globose (diameter 6–7.5 µm) or subglobose (6–7.5 × 3–4.5 (−6) µm) and bright green. Medulla pale whitish, 15–37.5 µm thick, merging indistinctly to the algal layer. Lower cortex 7.5–15 (−18) µm thick, dark brown to black.
Ascomata perithecioid, subglobose, immersed in thallus, 0–10 (−20) per squamule, brown to dark brown. Perithecia obpyriform, 225–275 (−300) × 175–250 µm; excipulum dark brown, 20–37.5 µm thick at the base and sides, pale brown at the apex near the ostiole; periphyses mostly simple, 22.5–34.5 µm long; hymenial algal cells globose (diameter 2–3 µm) to subglobose (3 × 1.5 µm), green; asci bisporous, clavate, 51–67.5 (−75) × 18–19.5 µm; ascospores muriform, two per ascus, hyaline to slightly brownish, matured spores brown, elongate-ellipsoid to subcylindrical, with 2–4 (−5) transverse divisions and 6–9 longitudinal divisions, upper spore generally broader and shorter than lower spore; upper spore 22.5–30 (−37.5) × 13.5–18 µm; lower spore 27–34.5 (−37.5) × 12–15 µm.
Pycnidia not seen.
Chemistry: K-, C-, KC-, P-; no lichen substances detected by TLC.
Substrate: Calcareous sands.
Additional specimens examined: China, Ningxia Autonomous region: Zhongwei city: Shapotou north experimental zone: soil crust, April 9, 2010, T. Zhang, SPT10062 (HMAS-L-134709); soil crust, April 9, 2010, T. Zhang, SPT10063 (HMAS-L-121315); soil crust, April 9, 2010, T. Zhang, SPT10047 (HMAS-L-134668); soil crust, April 19, 2007, J. Yang & T. Zhang, SPT187 (HMAS-L-134708).
Comments: This species is similar to E. pusillum, but E. pusillum is delimited by its plane, fully adnate thallus, and tightly aggregated, nearly inseparable squamules.
Materials and Methods
Lichen collection and ethics statement
Lichen specimens were collected from the Shapotou region (37°32′N, 105°02′E) on the southeast fringe of the Tengger Desert (Fig. 1). The investigation areas are located at an elevation of 1339 m in the steppified desert zone, which is also a transitional zone between desert and oasis53. The area has a mean annual precipitation of 180.2 mm, a mean annual evaporation of 3000 mm, a mean annual air temperature of 10.0 °C (minimum −25.1 °C, maximum 38.1 °C), an annual sunshine duration of 3264 h, a mean annual wind velocity of 2.9 ms−1, and 59 annual dust-storm days54. Ethical approval for lichen collection was obtained from the Shapotou Desert Research and Experimental Station. All species were deposited in the Herbarium Mycologicum Academiae Sinicae - Lichenes (HMAS-L).
Morphological and anatomical analyses
A dissecting microscope (ZEISS Stemi SV11) and compound microscope (ZEISS Axioskop 2 plus) were used to study the phenotypic traits of the specimen. Colour test reagents (10% aqueous KOH, saturated aqueous Ca(OCl)2, and concentrated alcoholic p-phenylenediamine) and TLC (solvent system C) were used to detect lichen substances55, 56.
DNA extraction, PCR amplification, and sequencing
Thirty-six specimens, including seven Endocarpon species, were chosen for DNA extraction, as shown in Table 1. The extraction procedure followed the modified CTAB method57. Three gene loci were used for PCR amplification: the nrDNA ITS region and two protein-coding genes, ADK and UCEH. The primer pairs ITS4 and ITS558 were used to amplify the nrITS regions, and the primers for ADK and UCEH were newly designed in this study (Table 3) based on the cDNA library of E. pusillum 50. The PCR reaction was carried out as follows: pre-denaturation at 95 °C for 8 min, followed by 35 cycles of amplification [95 °C for 50 s, 53 °C (50 °C for UCEH) for 50 s, 72 °C for 1 min], and finally followed by extension for 8 min at 72 °C. The PCR products were purified and sequenced by Genewiz Inc., Beijing, China.
Phylogenetic analyses
The sequences generated for this study were complemented with sequences from GenBank representing additional specimens or species, as listed in Table 1. The gene sequences of three loci, specifically nrDNA ITS, ADK and UCEH, were used for phylogenetic analyses. Sequences were aligned using ClustalW Multiple Alignment59 in BioEdit 7.2.560 and introns were manual excluded. The alignment files were transformed into both phylip and nexus formats using SeaView version 461, 62. The best model for the three single genes used in the phylogenetic analysis was identified in advance with jModelTest-2.1.963, 64.
Congruence among loci
To test the level of congruence among loci, highly supported clades (equal to or more than 75% bootstrap) from single-gene trees were compared and assessed65, 66. Each locus was subjected to a randomised accelerated maximum likelihood (RAxML) analysis involving 1000 pseudoreplicates with RAxML-HPC BlackBox 8.2.6 (Stamatakis 2014) on the Cipres Science Gateway (http://www.phylo.org)67. The results were visualised with FigTree 1.4.2. When there was no conflict using a 75% bootstrap value threshold, in situations where a monophyletic group was supported with bootstrap values ≥75% at one locus and the same group of taxa was supported (≤75%) as non-monophyletic with another locus, the group was assumed to be congruent and the data set was concatenated66.
Phylogeny of the genus Endocarpon
Phylogenetic analyses of Endocarpon were performed using the concatenated data set, which was analysed using RAxML-HPC BlackBox 8.2.668 and MrBayes 3.2.669, 70 on the Cipres Science Gateway (http://www.phylo.org)67. For the ML analysis, the GTR+G+I model was used as the substitution model with 1000 pseudoreplicates. The data were partitioned according to the different genes. The best model for the three single genes used in the Bayesian analysis was obtained in advance with jModelTest-2.1.9. Data sets for the two protein-coding genes (ADK and UCEH) were also partitioned by codon position. Two parallel Markov chain Monte Carlo runs were performed, each using 8000000 generations and sampling every 1000 steps. A 50% majority rule consensus tree was generated from the combined sampled trees of both runs after discarding the first 25% as burn-in.
Scanning electron microscopy
Rhizines of the samples were observed by performing SEM. Samples were sputter-coated with gold particles using a Bio-Rad SEM coating system (Sputter Coater BALTEC SDC 005, Leica Microsystems, Liechtenstein), and SEM images were recorded using a scanning electron microscope (SEM Quanta-200, FEI, Czech Republic) with a secondary electron detector operated at 10.0 kV.
Nomenclature
The electronic version of this article in Portable Document Format (PDF) in a work with an ISSN or ISBN will represent a published work according to the International Code of Nomenclature for algae, fungi, and plants. In addition, new names contained in this study have been submitted to Fungal Names (FN) from where they will be made available to the Global Names Index. The unique FN number can be resolved and the associated information viewed through any standard web browser by appending the FN number contained in this publication to the prefix http://www.mycobank.org/MB/.
References
Belnap, J., Büdel, B., Lange, O. L. Biological soil crusts: characteristics and distribution. p. 3–30 in: Belnap & Lange, O. L., (eds), Biological Soil Crusts: Structure, Function, and Management. (Springer, Berlin Heidelberg, 2001).
Ferrenberg, S., Reed, S. C. & Belnap, J. Climate change and physical disturbance cause similar community shifts in biological soil crusts. Proceedings of the National Academy of Sciences of the United States of America 112, 12116–12121 (2015).
Evans, R. D., Lange, O. L. Biological soil crusts and ecosystem nitrogen and carbon dynamics. p. 263–279 in: Belnap, J. & Lange, O. L. (eds), Biological Soil Crusts: Structure, Function, and Management. (Springer, Berlin Heidelberg, 2003).
Housman, D. C., Powers, H. H., Collins, A. D. & Belnap, J. Carbon and nitrogen fixation differ between successional stages of biological soil crusts in the Colorado Plateau and Chihuahuan Desert. J. Arid Environ. 66, 620–634 (2006).
Redfield, E., Barns, S. M., Belnap, J., Daane, L. L. & Kuske, C. R. Comparative diversity and composition of cyanobacteria in three predominant soil crusts of the Colorado Plateau. FEMS. Microb Ecol 40, 55–63 (2002).
Wu, L., Lan, S. B., Zhang, D. L. & Hu, C. X. Small-scale vertical distribution of algae and structure of lichen soil crusts. Microb Ecol 62, 715–724 (2011).
Veste, M., Littmann, T., Breckle, S. W. & Yair, A. The role of biological soil crusts on desert sand dunes in the northwestern Negev, Israel. In Sustainable Land Use in Deserts (eds Breckle, S. W. et al.) 357–367 (Springer, Berlin, 2001).
Zhang, Y. M., Wang, H. L., Wang, X. Q., Yang, W. K. & Zhang, D. Y. The microstructure of microbiotic crust and its influence on wind erosion for a sandy soil surface in the Gurbantunggut Desert of Northwestern China. Geoderma 132, 441–449 (2006).
Yang, J. & Wei, J. C. Endocarpon crystallinum, the new lichen species from semiarid deserts in China. Mycotaxon 106, 445–448 (2008).
Zhang, T. & Wei, J. C. Survival analyses of symbionts isolated from Endocarpon pusillum Hedwig to desiccation and starvation stress. Sci. China Life Sci. 54, 480–489 (2011).
Ding, L. P., Zhou, Q. M. & Wei, J. C. Estimation of Endocarpon pusillum Hedwig carbon budget in the Tengger Desert based on its photosynthetic rate. Sci. China Life Sci. 56, 848–855 (2013).
Wang, Y. Y. et al. Genome characteristics reveal the impact of lichenization on lichen-forming fungus Endocarpon pusillum Hedwig (Verrucariales, Ascomycota). BMC Genomics 15, 34–52 (2014).
Wang, Y. Y., Zhang, X. Y., Zhou, Q. M. & Wei, J. C. Comparative transcriptome analysis of the lichen-forming fungus Endocarpon pusillum elucidates its drought adaptation mechanisms. Sci. China Life Sci. 58, 89–100 (2015).
Li, H. & Wei, J. C. Functional analysis of thioredoxin from the desert lichen-forming fungus, Endocarpon pusillum Hedwig, reveals its role in stress tolerance. Sci Rep. 6, 27184 (2016).
Yang, J. & Wei, J. C. Desert lichens in Shapotou region of Tengger Desert and bio-carpet engineering. Mycosystema 33, 1025–1035 (2014).
Wei, J. C. The Desert Biocarpet Engineering - A new way to control the ariddesert. Arid Zone Research 2, 287–288 (2005).
Lumbsch, H. T. & Huhndorf, S. M. Outline of Ascomycota - 2007. Myconet 13, 1–58 (2007).
DePriest, P. T. Early molecular investigations of lichen-forming symbionts: 1986–2001. Ann.Rev.of Microbiol. 58, 273–301 (2004).
Lumbsch, H. T. Recent trends in phylogeny and classification of lichen-forming ascomycetes. p. 153–168 in: Ganguli, B. N. & Deshmukh, S. K., (eds), Fungi. Multifaceted Microbes. (Anamaya Publishers, New Delhi, 2006).
Miadlikowska, J. et al. New insights into classification and evolution of the Lecanoromycetes (Pezizomycotina, Ascomycota) from phylogenetic analyses of three ribosomal RNA- and two protein-coding genes. Mycologia 98, 1088–1103 (2006).
Printzen, C. Lichen Systematics: The Role of Morphological and Molecular Data to Reconstruct Phylogenetic Relationships. Pp. 233–275 in: Lüttge, U., Beyschlag, W., Büdel, B. & Francis, D., (eds), Progress in Botany (Springer, Berlin Heidelberg, 2010).
Miadlikowska, J. et al. Multigene phylogenetic synthesis for 1307 fungi representing 1139 infrageneric taxa, 312 genera and 66 families of the class Lecanoromycetes (Ascomycota). Mol. Phylogenet. Evol. 79, 132–168 (2014).
Jaklitsch, W. M., Baral, H. O., Lücking, R. & Lumbsch, H. T. Ascomycota. Pp. 1–150 in: Frey, W., (ed), Syllabus of Plant Families - Adolf Engler’s Syllabus der Pflanzenfamilien, 13 (Gebr. Borntraeger Verlagsbuchhandlung, Stuttgart, 2016).
Lücking, R., Hodkinson, B. P. & Leavitt, S. D. The 2016 classification of lichenized fungi in the Ascomycota and Basidiomycota – Approaching one thousand genera. Bryologist 119, 361–416 (2016).
James, T. Y. et al. Reconstructing the early evolution of Fungi using a six-gene phylogeny. Nature 443, 818–822 (2006).
Gueidan, C., Roux, C. & Lutzoni, F. Using a multigene phylogenetic analysis to assess generic delineation and character evolution in Verrucariaceae (Verrucariales, Ascomycota). Mycol. Res. 111, 1145–1168 (2007).
Weerakoon, G. et al. New molecular data on Pyrenulaceae from Sri Lanka reveal two well-supported groups within this family. Lichenologist 44, 639–647 (2012).
Prieto, M., Martínez, I., Aragón, G., Gueidan, C. & Lutzoni, F. Molecular phylogeny of Heteroplacidium, Placidium, and related catapyrenioid genera (Verrucariaceae, lichen-forming Ascomycota). Am. J. Bot. 99, 23–35 (2012).
Gueidan, C., Truong, V. D. & Lu, N. T. Phylogeny and taxonomy of Staurothele (Verrucariaceae, lichenized ascomycetes) from the karst of northern Vietnam. Lichenologist 46, 515–533 (2014).
Gueidan, C. & Lendemer, J. C. Molecular Data Confirm Morphological and Ecological Plasticity within the North-American Endemic Lichen Willeya diffractella (Verrucariaceae). Sys. Bot. 40, 369–375 (2015).
Kirk, P. M., Cannon, P. F., David, J. C. & Stalpers, J. A. Ainsworth & Bisby’s Dictionary of the Fungi (10th Edition) (CABI Publishing, Egham, 2008).
Magnusson, A. H. Lichens from central Asia Part I. In Reports Scientific Exped. Nort-west. provinces of China (the Sino-Swedish expedition) 13, XI. Botany, 1. (ed. Hedin, S.). (Aktiebolaget Thule, Stockholm, 1940).
Magnusson, A. H. Lichens from central Asia. Part II. In Reports Scientific Exped. Northwest. provinces of China (the Sino-Swedish Expedition) 22, XI, Botany, 2. (ed. Hedin, S.) (Aktiebolaget Thule, Stockholm, 1944).
Wei, J. C. An enumeration of lichens in China (International Academic Publishers, Beijing, 1991).
Abbas, A. & Wu, J. N. Lichens of Xinjiang. (Sci-Tec & Hygiene Publishing House of Xinjiang, Urumqi, 1998).
Harada, H. & Wang, L. S. Two new freshwater species of Verrucariaceae from Yunnan, China. Lichenologist 28, 297–305 (1996).
Fink, B. The Lichen Flora of the United States. (University of Michigan Press, 1935).
Lamb, I. M. N. rare or interesting lichens from the Southern Hemisphere. Lilloa 14, 203–251 (1948).
Kopachevskaya, E. G., Makarevich, M. F. & Ocksner, A. N. Opredeliditel’ Lishainikov SSSR. Vol. 4. Verrucariaceae-Pilocarpaceae (Nauka, Leningrad, 1977).
Singh, A. & Upreti, D. K. The lichen genus Endocarpon from India. Candollea 39, 539–548 (1984).
McCarthy, P. M. The lichen genus Endocarpon Hedwig in Australia. Lichenologist 23, 27–52 (1991).
Harada, H. A taxonomic study of the lichen genus Endocarpon (Verrucariaceae) in Japan. Nova Hedwigia 45, 335–353 (1993).
Harada, H. Contributions to the lichen flora of the Mariana Islands, Micronesia (1). Genus Endocarpon Hedw. (Verrucariaceae). J. Nat. Hist. Mus. Inst. Chiba 4, 97–99 (1997).
Breuss, O. & McCune, B. Additions to the pyrenolichen flora of North America. Bryologist 97, 365–370 (1994).
Breuss, O. Endocarpon. In Lichen flora of the greater Sonoran desert region. Vol. 1. (eds Nash, T. H. et al.) 181–187 (Arizona State University, Tempe, 2002).
Nilsson, R. H., Kristiansson, E., Ryberg, M., Hallenberg, N. & Larsson, K. H. Intraspecific ITS variability in the kingdom Fungi as expressed in the international sequence databases and its implications for molecular species identification. Evol. Bioinform. 4, 193–201 (2008).
Truong, C., Divakar, P. K., Yahr, R., Crespo, A. & Clerc, P. Testing the use of ITS rDNA and protein-coding genes in the generic and species delimitation of the lichen genus Usnea (Parmeliaceae, Ascomycota). Mol. Phylogenet. Evol. 68, 357–372 (2013).
Whitford, P. C., Miyashita, O., Levy, Y. & Onuchic, J. N. Conformational transitions of adenylate kinase: switching by cracking. J. Mol. Biol. 366, 1661–1671 (2007).
Nandi, D., Tahiliani, P., Kumar, A. & Chandu, D. The ubiquitin-proteasome system. J. Bioscience. 31, 137–155 (2006).
Wang, Y. Y., Zhang, T., Zhou, Q. M. & Wei, J. C. Construction and characterization of a full-length cDNA library from mycobiont of Endocarpon pusillum (lichen-forming Ascomycota). World J. Microb. Biot. 27, 2879–2884 (2011).
Abed, R. M., Al-Sadi, A. M., Al-Shehi, M., Al-Hinai, S. & Robinson, M. D. Diversity of free-living and lichenized fungal communities in biological soil crusts of the Sultanate of Oman and their role in improving soil properties. Soil Biol. Biochem. 57, 695–705 (2013).
Barger, N. N., Herrick, J. E., Van Zee, J. & Belnap, J. Impacts of biological soil crust disturbance and composition on C and N loss from water erosion. Biogeochemistry 77, 247–263 (2006).
Li, X. R., Zhang, J. G., Wang, X. P., Liu, L. C. & Xiao, H. L. Study on soil microbiotic crust and its influences on sand-fixing vegetation in arid desert region. Acta Bot. Sin. 42, 965–970 (2000).
Li, X. R. Influence of variation of soil spatial heterogeneity on vegetation restoration. Sci. China Ser. D. 48, 2020–2031 (2005).
Culberson, C. F. & Kristensson, H. A standardized method for the identification of lichen products. J. Chromatogr. 46, 85–93 (1970).
Culberson, C. F. Improved conditions and new data for the identification of lichen products by a standardized thin-layer chromatographic method. J. Chromatogr. 72, 113–125 (1972).
Cubero, O. F., Crespo, A., Fathi, J. & Bridge, P. D. DNA extraction and PCR amplification method suitable for fresh, herbarium-stored, lichenized and other fungi. Plant Sys. Evol. 216, 243–249 (1999).
White, T. J., Bruns, T. D., Lee, S. & Taylor, J. Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In PCR protocols: a guide to methods and applications (eds Innis, M. A. et al.) 315–322 (Academic Press, London, 1990).
Thompson, J. D., Higgins, D. G. & Gibson, T. J. CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position specific gap penalties and weight matrix choice. Nucleic Acids Res. 22, 4673–4680 (1994).
Hall, T. A. BioEdit: a user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucl. Acids. Symp. 41, 95–98 (1999).
Galtier, N., Gouy, M. & Gautier, C. SEAVIEW and PHYLO_WIN: two graphic tools for sequence alignment and molecular phylogeny. Comput. Appl. Biosci. 12, 543–548 (1996).
Gouy, M., Guindon, S. & Gascuel, O. SeaView version 4: a multiplatform graphical user interface for sequence alignment and phylogenetic tree building. Mol. Biol. Evol. 27, 221–224 (2010).
Guindon, S. & Gascuel, O. A. simple, fast and accurate method to estimate large phylogenies by maximum-likelihood. Systematic Biol. 52, 696–704 (2003).
Darriba, D., Taboada, G. L., Doallo, R. & Posada, D. jModelTest 2: more models, new heuristics and parallel computing. Nat. Methods 9, 772 (2012).
Mason-Gamer, R. J. & Kellogg, E. A. Testing for phylogenetic conflict among molecular data sets in the tribe Triticeae (Graminaeae). Systematic Biol. 45, 524–545 (1996).
Prieto, M., Martínez, I., Aragón, G., Gueidan, C. & Lutzoni, F. Molecular phylogeny of Heteroplacidium, Placidium, and related catapyrenioid genera (Verrucariaceae, lichen-forming Ascomycota). Am. J. Bot. 99, 23–35 (2012).
Miller, M. A., Pfeiffer, W. & Schwartz, T. Creating the CIPRES Science Gateway for inference of large phylogenetic trees. In Proceedings of the Gateway Computing Environments Workshop (GCE) 1–8 (New Orleans, LA, 2010).
Stamatakis, A. RAxML Version 8: A tool for phylogenetic analysis and post-analysis of large phylogenies. Bioinformatics 30, 1312–1313 (2014).
Huelsenbeck, J. P. & Ronquist, F. MRBAYES: Bayesian inference of phylogenetic trees. Bioinformatics 17, 754–755 (2001).
Ronquist, F. & Huelsenbeck, J. P. MrBayes 3: Bayesian phylogenetic inference under mixed models. Bioinformatics 19, 1572–1574 (2003).
Acknowledgements
This project was supported by the Knowledge Innovation Program of the Chinese Academy of Sciences (KSCX2-EW-Z-9), the Youth Innovation Promotion Association of the Chinese Academy of Sciences and the National Natural Science Foundation of China (31470149). The authors sincerely thank Prof. Pradeep K. Divakar for helping with the phylogenetic analysis and providing constructive suggestions and comments on the draft. Ms. H. Deng kindly provided assistance during the HMAS-L studies. The authors also thank Dr. R. L. Jia of the Shapotou Desert Research and Experimental Station and the staff of the National Herbarium of Victoria (MEL), Royal Botanic Gardens Melbourne, for their assistance.
Author information
Authors and Affiliations
Contributions
T.Z. and M.L. conducted the main lab work; Y.Y.W. and Z.J.W. conducted portions of the lab work; X.L.W. and T.Z. wrote the manuscript; X.L.W. and J.C.W. designed the study and revised the manuscript. All authors reviewed the manuscript.
Corresponding authors
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zhang, T., Liu, M., Wang, YY. et al. Two new species of Endocarpon (Verrucariaceae, Ascomycota) from China. Sci Rep 7, 7193 (2017). https://doi.org/10.1038/s41598-017-07778-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-07778-5
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.