Abstract
The myostatin (MSTN) gene is important because of its role in regulation of skeletal muscle growth in all vertebrates. In this study, CRISPR/Cas9 was utilized to successfully target the channel catfish, Ictalurus punctatus, muscle suppressor gene MSTN. CRISPR/Cas9 induced high rates (88–100%) of mutagenesis in the target protein-encoding sites of MSTN. MSTN-edited fry had more muscle cells (p < 0.001) than controls, and the mean body weight of gene-edited fry increased by 29.7%. The nucleic acid alignment of the mutated sequences against the wild-type sequence revealed multiple insertions and deletions. These results demonstrate that CRISPR/Cas9 is a highly efficient tool for editing the channel catfish genome, and opens ways for facilitating channel catfish genetic enhancement and functional genomics. This approach may produce growth-enhanced channel catfish and increase productivity.
Similar content being viewed by others
Introduction
Although fish comprise the most diverse group among the vertebrates, a much smaller number of fish species is used for research and commercial purposes. The major species cultured in the world include salmonids, tilapias, carps and catfishes. There are about 40 extant families, and more than 3,700 extant species of catfishes that have been identified and described, living in marine and fresh waters1. This makes the Siluriformes the second most diverse vertebrate order; in fact, one of every twenty vertebrate species is a catfish. Members of the family Ictaluridae, especially the genus Ictalurus and their hybrids, are the primary fish species used in aquaculture and sport fishing in the United States. Recently, the production and the water surface area devoted to catfish culture in the United States declined2 due to several factors, including increasing fuel and feed cost and competition from foreign imports. Genetic improvement of catfish offers a promising approach for this industry to maintain profitability and sustainability via development of high performance fish3.
Myostatin (MSTN), or growth and differentiation factor 8, is one of the well-known examples of economically important genes. Mutations linked with the double-muscled phenotype in Belgian Blue and Piedmontese cattle were found in the MSTN gene, resulting in a significant increase in muscling compared to conventional cattle4,5,6,7. MSTN was identified as a member of the transforming growth factor β (TGF-β) superfamily that initiate their roles in regulation of skeletal muscle mass through the cell surface receptor, activin receptors type II (ACVR2)8,9,10. Myostatin is a key regulator of skeletal muscle growth in all vertebrates and regulates myoblast differentiation in vitro 11. Altering myostatin, through gene knockout12 or overexpression of inhibitors9, prominently increases muscle mass. Mutations of myostatin were also found in other cattle breeds13, 14 and mammalian species15,16,17,18. Whereas the mammalian myostatin is exclusively expressed from one gene copy and limited to skeletal muscle6, 12, 19, two or four copies have been found in fishes20,21,22,23,24,25,26 and differentially expressed in many tissues27,28,29,30,31,32,33,34,35,36 such as muscle, eye, stomach, skin, brain, gonads, kidney, intestine, liver, spleen, gill and heart. In channel catfish (Ictalurus punctatus), the MSTN protein is actively expressed only from one copy on chromosome 6 in different tissues30, 37. The MSTN protein is conserved in teleosts, but its functions in fish remain to be revealed because of the lack of spontaneous MSTN null mutation. The knockout of MSTN would further the study on the roles of this gene in fish species.
Different molecular editing tools have been used to target MSTN in teleosts, such as zinc-finger nucleases (ZFNs)38 and transcription activator-like effector nucleases (TALENs)39, 40. Although the use of such engineered nucleases made the production of gene-edited animals possible, they are difficult to engineer and showed low specificity to target DNA sequences41. Recently, a new gene editing system has been applied with higher targeting efficiency and lower cell toxicity, known as clustered regularly interspaced short palindromic repeats (CRISPR)/CRISPR-associated protein 9 (Cas9). Based on the type II prokaryotic CRISPR from Streptococcus pyogenes 42, 43, the co-delivery of endonuclease Cas9 combined with a synthetic small guide RNA (sgRNA) targeting certain gene(s) into eukaryotic cells can edit the genome by stimulating a double-strand break (DSB) at a desired site(s). Then the non-homologous end-joining (NHEJ) process re-ligates the DSBs, generating insertion/deletion (indel) mutations and enabling existing genes to be edited, deleted or new ones inserted. Unlike ZFNs and TALENs, sgRNA is the only component that needs designation for each genomic target, thereby significantly simplifying the design and lowering the cost of gene editing compared to the protein-based target recognition platforms. To date, CRISPR/Cas9 has been applied extensively in many species and promises to transform the fields of genome editing in plants and animals. MSTN knockout strains utilizing zygote microinjection of CRISPR/Cas9 have been produced recently for laboratory animals44,45,46 and livestock47,48,49,50. Multiplex genome editing using Cas9 endonuclease also can be applied easily by co-delivering a combination of sgRNAs to target multiple genomic loci simultaneously51,52,53,54. Recently, CRISPR mutagenesis was performed to edit various genes (including MSTN) in some aquaculture species55,56,57,58,59.
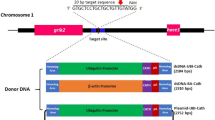
The channel catfish MSTN gene has been cloned and characterized30, 37, and has only one orthologous MSTN gene in their genome. Like other MSTN genes in mammals and teleost, the I. punctatus MSTN gene consists of three exons and two introns, and encodes a protein consisting of 389 amino acids that span three conserved domains with two proteolytic sites in between dibasic (RX) and tetrabasic (RXXR) at amino acids (55–56) and (295–298), respectively (Fig. 1A). The roles of myostatin in channel catfish, however, still need further investigation. In this study, we used CRISPR zygote microinjection to knockout MSTN gene in channel catfish, and determined the effects of knockout on growth. Due to the long generation time of channel catfish, we limited our study to early growth. We aim to produce growth-enhanced lines of channel catfish with CRISPR technology. These gene-edited lines would then be fully characterized for growth rate, feed efficiency and disease resistance and compared to genotypes currently used in aquaculture.
Design of the CRISPR/Cas9 system. (A) Schematic diagram of channel catfish MSTN; a signal sequence (N-terminal secretory signal), a propeptide domain (propeptide) and a bioactive domain (C-terminal domain). RX (55–56) is a proteolytic site to remove the signal sequence and RXXR (295–298) is a proteolytic processing site (RSSR) to produce C- terminal bioactive form of MSTN. These 3 domains (389 amino acids) are encoded from (B) the three exons of the channel catfish MSTN gene; Exons I, II and III consist of 418, 371 and 381 base-pairs encoding 139, 124 and 129 amino acids residues, respectively. CRISPR/Cas9 target sites in Exon I; The Exon and Introns are indicated by upper and lower case, the underlined Bold uppercase are the start and stop codons, The primers used in PCR are indicated in red, The guide RNA target sites are indicated in green followed by PAM (Protospacer adjacent motif, NGG) in Blue.
Results
Design and Delivery of CRISPR/Cas9 System
CRISPRscan online tool was used to design oligos to generate MSTN edited channel catfish. Three guide RNAs were designed targeting three different MSTN sites that were followed by the PAM (Protospacer Adjacent Motif, NGG) sequence along exon I to alter the expression of the channel catfish MSTN gene (Fig. 1B) (Table 1). The designed sgRNA(s) were co-injected with Cas9 nuclease individually and also mixed with the other sgRNAs, resulting in four groups, MSTN-1, MSTN-2, MSTN-3 and MSTN-Mix (a combination of -1, -2 and -3). Additionally, two control groups were included; one a buffer-injected control without any sgRNA (iCTRL), and the other a normal non-injected control (nCTRL). For each treatment group, embryo mortality, hatchability, fry survival and mutation rate were recorded and compared statistically (Table 2).
Embryo Mortality, Hatchability, Fry Survival Rates
Embryo mortality
The mortality began at the first day post-fertilization (dpf) and continued until eight dpf. Embryo mortality was lowest in nCTRL embryos, followed by iCTRL embryos (Fig. 2A). Mean survival time ranged from 4.6 dpf in MSTN-2 group to 7.6 dpf in nCTRL group. Overall comparisons revealed significant differences in the mean survival time among at least two groups (p < 0.0001). With pairwise comparisons, the mean survival time for the nCTRL group was significantly longer than for all other groups (p < 0.0001). Mean survival time was different for the following groups (p < 0.005): MSTN-1 and MSTN-2, MSTN-1 and MSTN-Mix, MSTN-2 and MSTN-3, MSTN-2 and iCTRL, MSTN-Mix and iCTRL, and iCTRL and nCTRL. All other pairwise comparisons of mean survival time were not significantly different (p > 0.05) (Table 2).
Plots of (A) embryo survival, (B) mean time to hatch, and (C) fry survival; embryos microinjected at the one-cell stage with sgRNAs/Cas9 protein targeting the channel catfish MSTN gene. Three sgRNAs were microinjected individually (MSTN-1, MSTN-2 and MSTN-3) and multiplexed (MSTN-Mix). Two controls were used; Injected control embryos (iCTRL) were full-siblings to the treatment groups and were injected with the same solution and volume but without sgRNA or Cas9 protein. The second control was not injected (nCTRL). Hatch rate was calculated as the number of embryos that hatched at a given time-point (day post-fertilization, dpf) compared to the total number of embryos that hatched.
Embryo hatch
Embryos began to hatch at six dpf, and hatch was completed at eight dpf. Embryo mean time to hatch ranged from 6.9 dpf in MSTN-3 group to 7.4 dpf in nCTRL group (Fig. 2B, Table 2). Overall comparison of embryo mean time to hatch revealed significant differences among at least two groups (p < 0.0001). The nCTRL group had the longest hatch time when compared to all other groups (p < 0.05). MSTN-3 group hatched earlier than the MSTN-1 (p = 0.001), MSTN-Mix (p = 0.0003) and iCTRL (p = 0.001) groups. All other pairwise comparisons of mean time to hatch were not different (p > 0.05). Fry mean survival time ranged from 18.3 to 20.0 dpf (Fig. 2C). No significant differences in fry survival time were detected among all groups (p = 0.078) (Table 2). No deformed or abnormal fry were seen in any treatments both pre- and post-hatch.
Analysis of Mutagenesis Efficiencies of CRISPR/ Cas9
The surveyor mutation detection assay was performed to detect the mutated individuals among both dead embryos and hatched fry. Digested PCR products revealed that non-edited fish showed one distinct band (482 bp), while the individuals carrying mutated genes showed two or more bands (<482 bp). Gel electrophoresis revealed various banding patterns depending on the type of mutations in the four treatment groups (Fig. 3Aa). Fin clips are the most desirable tissue to assay, as they cause minimal damage to the fish and they regenerate. If fin clips mirror the mutation type found in all tissues, the fish could be sorted based on mutation rates scored in fin clips only without sacrificing the fish. The mutations in every tissue tested, such as barbel, muscle, intestine, and eye, were found to have identical banding patterns with those found in the fin clips (Fig. 3Ab).
Analysis of mutagenesis efficiencies of CRISPR/ Cas9. (A) Identification of edited myostatin (MSTN) gene sequences in channel catfish using the surveyor mutation detection assay. Minus sign indicates the negative control without template [first lane] and the control with wild-type DNA as template [2nd and 3rd lanes] (482 bp). L indicates 1 kb plus DNA ladder (Invitrogen, Carlsbad, CA). (a) surveyor mutation detection of MSTN gene in fin-clip samples from 10 individuals from different treatments. Two or more bands indicate that mutations occurred ( < 482 bp). (b) surveyor mutation detection of MSTN gene in different tissues from two mutated channel catfish: B Barbel; E Eye; G Gills; I Intestine; M Muscle. Electrophoretic results were cropped from the original images shown in Supplementary Fig. S1. (B) Insertion (INS) and deletion (DEL) mutation frequencies in the treatment groups with in-frame (IF) and frame-shift (FS) mutation percentages. (C–F) CRISPR-Cas9-induced mutagenesis efficiencies for the MSTN gene in different treatment groups; (C) MSTN-1 (D) MSTN-2 (E) MSTN-3 (F) MSTN-Mix. (a) Sequences of channel catfish MSTN gene with co-delivered sgRNA(s) and Cas9 protein induced mutations. The wild-type channel catfish MSTN gene sequences are shown on the top. Sequences in green are the target sites of guide RNA followed by PAM sequence (Blue); Red arrows indicate the expected sites of cleavage by Cas9. Red dashes and letters indicate the deletion/insertion of nucleotides. Numbers in brackets shows the number of nucleotides deleted (−) or inserted (+) in the edited MSTN gene. (b) Schematic diagrams show predicted truncated proteins that would be produced from the mutated catfish (see C–Fa). The numbers show the positions of amino acid residues. Single blue lines in the domains show loss of amino acids. Amino acid sequences (black bold upper-case letters) show newly inserted amino acid fragments. Amino acid sequences following incomplete domain were due to frame-shift reading, resulting in a premature stop (red color) codon.
All dead embryos from all treatments were mutated, while mutagenesis frequencies in fry among treatment groups MSTN-1, MSTN-2, MSTN-3 and MSTN-Mix were: 88.6%, 100%, 88% and 96.3%, respectively. The PCR amplicons from mutated individuals were cloned and sequenced to confirm and characterize the mutation. The alignment of mutated DNA sequences against the wild type sequence revealed multiple forms of indels caused by the CRISPR/Cas9 system at target sites (Fig. 3C–Fa). Mutated individuals from the MSTN-1 group exhibited two types of deletions, 15 and 36 bases, and a 6-base insertion (Fig. 3Ca). However, Cas9 nuclease cut at the target site, but there was no frame shift because the base deletions and insertion were triplets, resulting in missing and/or additional amino acid (Fig. 3Cb). In the MSTN-2 treatment, only deletions were found (Fig. 3Da); 4, 13, 19, 22, 25, 26, 130 and 207 nucleotides that all should lead to frame shifts (Fig. 3Db). The MSTN-3 group had four types of deletions (2, 8, 9, and 30 bp) and three types of insertions (5, 6 and10 bp) leading to frame shift mutations in two deletion and insertion types and in-frame mutations in the rest (Fig. 3E). The most variable indel mutations (including large deletions) occurred in the MSTN-Mix Group. Exon I of MSTN was almost deleted. While exon I involves 418 bp (Fig. 1B), five long deletion types; 153,165, 238, 240 and 316 bases were deleted between the target sites of the sgRNA(s) (Fig. 3F). Mutation frequencies as well as predicted gene expression from mutated sequences revealed that most forms of insertions and deletions led to frame shift, resulting in a premature stop codon in the transcribed mRNA and a truncated nonfunctional protein product (Fig. 3B–F).
Evaluation of growth in MSTN mutant fry
The average body weight of mutated fry at 40 days post-microinjection was 29.7% larger than wild-types (234.3 ± 6.9 mg vs 180.6 ± 2.77 mg, n = 330, Fig. 4A). The mutants also showed 6.6% longer average body length than wild-types (22.7 ± 0.24 mm vs 21.3 ± 0.1 mm, n = 330, Fig. 4B). The Shapiro–Wilk normality test revealed that the body weight and body length data were not normally distributed (W = 0.95, p < 0.0001). Therefore, Mann–Whitney U-tests were used to compare differences in body weight and body length between groups. This test revealed that the mutants were significantly heavier (U = 5399, p < 0.001) than wild-types. The length of mutants was significantly greater than wild-types (U = 7633.5, p < 0.001).
Evaluation of growth in myostatin (MSTN)-mutated one-month-old channel catfish fry. Body weight (A) and body length (B) of mutant (blue) and wild type (red) (n = 330). (C,D) Representative images of the ventral cross-sectional area of the epaxial muscle of wild-type (WT) (C) and mutant (individuals with frame-shift mutation from MSTN-Mix group) (D), shown by Hematoxylin and Eosin (H&E) staining. Scale bar in (C,D): 25 µm. Numbers of muscle fibers (E), the average area of the muscle fibers (F) and the total area of fibers (G) of mutant (blue) and wild-type (red), quantified H&E staining images (see C,D) with ImageJ. Approximately 32 stained images for each treatment were quantified. Distribution percentage of the fibers (H) was calculated as the total area of fibers per cross-sectional area multiplied by 100. Statistical significance was set at p < 0.05, and all data were presented as the mean ± standard error (SEM).
Histological Analysis
The dorsal muscle of mutated channel catfish in Fig. 4D exhibited 33.7% more muscle fibers (809.5 ± 7.9) than wild-types (605.2 ± 13.7), t-test: t (49.3) = 12.90, p < 0.001. The average size of muscle fibers was larger in mutated channel catfish compared to wild-types (30.4 ± 0.2 µm2 vs 29.8 ± 0.2 µm2), t-test: t (62) = −2.22, p = 0.015 (Fig. 4C–F). Additionally, the average total area of muscle fibers was increased in the mutant (24668.6 ± 394.8 µm2) as compared to the wild-type channel catfish (18109.7 ± 467.4 µm2), t-test: t (62) = −10.72, p < 0.001. The percentage of muscle fiber area was also higher in the mutants (32 ± 0.3%) than in wild types (26.6 ± 0.3%), t-test: t (62) = −11.91, p < 0.001 (Fig. 4G,H). Altogether, these findings indicate that mutated channel catfish showed hyperplasia as well as hypertrophy.
Discussion
The sgRNA(s) and Cas9 protein were directly co-injected into one-cell embryos, and gene editing of the channel catfish MSTN gene was successfully accomplished. We utilized CRISPRscan to design the sgRNAs and selected the sgRNAs with the highest scores to target the I. punctatus MSTN gene. As CRISPRscan was originally designed based upon the zebrafish (Danio rerio) genome60, we assume that the phylogenetically close relationship between the genomes of channel catfish and zebrafish61 had an important impact on the successful targeting of these designed sgRNAs in channel catfish. All designed sgRNAs worked effectively at their target sites on the MSTN gene with high mutagenesis efficiencies. The mutation rate in teleosts using CRISPR/Cas9 system varies, fluctuating from 2%55 to 99%52. Although the CRISPR/Cas9 system (Cas9 plasmid) was previously microinjected in channel catfish in our laboratory with a 100% mutation rate58, embryo hatch and early fry survival rates were only 10% and 45%, respectively. In the present study, these rates were raised prominently to about 42% for hatching and 90% for early fry survival with high mutation rates (88–100%) (Table 2). Also, the overall mutation rates were higher than those obtained in Atlantic salmon (Salmo salar)56, 62, Common carp (Cyprinus carpio)59 and Nile tilapia (Oreochromis niloticus)57.
To assess the effects of injection and CRISPR/Cas9 system on embryo survival, hatch rate and early fry survival, we included an injected control group as well as a normal control group using injection solution devoid of CRISPR/Cas9 system components. We did not observe any abnormalities pre- or post-hatching. The higher embryo mortality in injected groups compared to the nCTRL group may be due to the microinjection procedures since all embryos were full-siblings, exposed to the same handling stress, and reared using the same environmental conditions.
Oligonucleotides are most active when injected directly into the embryo cell rather than the yolk63. However, this injection procedure needs careful orientation of the embryos, and is more time consuming. We co-microinjected Cas9 protein and sgRNA(s) directly into the yolk at the one-cell stage so that it would be transferred to the cytoplasm through cytoplasmic streaming64, 65. This approach proved adequate to edit the genome, requiring less time and effort during microinjection with less disruption of embryos, leading to high mutation and survival rates. This approach could explain why the embryo hatch and early fry survival rates were high in this study compared to that of Qin et al.58 using microinjection of the blastodisc or electroporation. Also, using Cas9 protein instead of its plasmid eliminated the time required for expression of Cas9 plasmid, making genome targeting during the one-cell stage (about 90 minutes in channel catfish) of embryonic development more likely and with high mutation rate.
CRISPR/Cas9 was highly effective as a large percentage of the embryos were mutated within the target sites along the open reading frame, and no mutations were detected nearby and outside the target site. The genome was not examined for off-target mutations. Most mutations should lead to a frame-shift that results in a premature stop codon, early termination in translation, and disrupt the molecular functions of the protein. Fortunately, about two-thirds of the deletions and insertions were frame-shift mutations which most likely caused gene truncations (Fig. 3B–F). Compared to wild-type, the patterns and lengths of amplicon bands revealed with gel electrophoresis differed, depending on the treatment group of the mutated fry. Unsurprisingly, the mutated samples of MSTN-Mix group showed the most variable banding patterns as revealed by gel electrophoresis. In the same manner, the alignment of MSTN-Mix group sequences revealed the most variable types of mutation.
Another important finding was the large deletion in exon I of the MSTN gene resulting from the injection of the three sgRNAs in combination (the MSTN-Mix group). CRISPR/Cas9 system could be used to achieve large genomic deletions, not only in one gene, but also in different chromosomal loci by delivering two pairs or more of sgRNAs together, with Cas9 nuclease targeting different genomic sites simultaneously51,52,53,54.
Since applied genetics research focuses upon economically important traits, hyper-muscularity after targeting the MSTN gene has been studied in many mammals44,45,46,47,48. The average body weights of MSTN-mutant mammals were 15–30% higher than wild-types46,47,48. Regarding MSTN-mutated teleosts, the growth rate enhancement of MSTN mutants was 10–15%39 to 39–45%66, 67. These results were with the small model species, zebrafish, which does not always translate into the same results in larger fishes. In the present study, we evaluated the phenotypic effects on muscle mass growth in the earliest weeks post-hatching. The mean body weight of mutated fry was increased by 29.7%. The mutant genotypes exhibited hyperplasia (33.7% increase in fiber number) and hypertrophy (2% increase in fiber diameter) of muscle fibers as compared to wild-type, and was the likely explanation of the growth differences between the mutants and wild-types. The number and size of muscle fibers are highly important factors in determining body size in teleosts68,69,70.
This is the first time that Cas9 protein has been directly delivered to edit genes in this biologically and commercially interesting teleost species, channel catfish. We anticipate that this technology will be the principal tool for further molecular and functional studies, especially for channel catfish since its genome is now known61. Our study goes beyond vertebrate models (zebrafish and medaka) and addresses the utility of the CRISPR/Cas9 as a tool for generating gene-edited channel catfish, and potentially other aquaculture species, with high efficiency and accompanied with significant phenotypic change. This technology is also especially relevant for teleosts with extended generation times, 2-4 years, like channel catfish, as the extensive mutation in virtually all individuals and especially in all tissues allows solid phenotypic evaluation in the founding generation of channel catfish without having to wait to generate subsequent generations. Further study is still needed to evaluate the carcass composition and meat-quality traits of mutated individuals when they reach commercial food size (400–700 g). Other physiological parameters and immune status should also be considered39, 71, 72 and linked to the productivity of these gene-modified lines.
Materials and Methods
Design and preparation of sgRNA and CRISPR/Cas9 System
Using the CRISPRscan online tool60, three small guide RNAs were customized targeting the I. punctatus MSTN gene (GenBank Accession No. AF396747.1). The cloning-free (PCR-based) method to generate sgRNA templates was used; The universal primer containing the sgRNA scaffold as well as ssDNA templates for sgRNAs containing the T7 promoter and the 20-nt gene-specific target sequence without the PAM were manufactured by Invitrogen (Carlsbad, CA) (Table 1). The sgRNAs were generated by T7 run –off as described previously73, 74 with some modifications; the universal primer and ssDNA templates were annealed and filled by Platinum™ Taq DNA Polymerase (Invitrogen). In RNAse-free environment, the resulting double-stranded DNA served as the template for in vitro transcription to generate sgRNA using the Maxiscript T7 Kit (Thermo Fisher Scientific). The obtained sgRNAs were purified using Zymo RNA Clean and Concentrator Kit (Zymo Research, Irvine, CA). The Cas9 protein was from PNA BIO Inc. (Newbury Park, CA) and reconstituted following the manufacturer’s guidelines. Four sets of injection solutions were prepared; three by mixing each individual sgRNA with Cas9 protein separately, and the fourth by combining the three sgRNAs together with Cas9 protein. Phenol red was added to color the sgRNA/Cas9 solutions by mixing sgRNA, Cas9 protein and phenol red in a 1:1:1 ratio. The final concentrations of sgRNA and Cas9 protein were 150–200 ng/µl and 300–350 ng/µl, respectively. The mixtures were then incubated for 10 minutes on ice before loading into the microinjection needle.
Ethical statement
Channel catfish were obtained from the Fish Genetic Research Unit, School of Fisheries, Aquaculture and Aquatic Sciences at Auburn University, Alabama 36849, USA. The research protocol followed all Standard Operating Procedures (SOP) approved by the Institutional Animal Care and Use Committee (IACUC) of Auburn University.
Egg collection and sperm preparation
Sexually mature Kansas Random channel catfish females were artificially spawned using luteinizing hormone releasing hormone analog (LHRHa) Reproboost® Implant (Center of Marine Biotechnology, Baltimore, MD) at 85 μg/kg body weight to facilitate ovulation, and the eggs were hand-stripped into greased spawning pan. Sexually mature Kansas Random channel catfish males were euthanized and the testes were crushed and macerated into saline (0.9% NaCl) to release sperm and prepare a sperm solution.
Fertilization, microinjection and embryo incubation
Approximately, 200–300 eggs were transferred to a greased spawning pan. Then, 1–2 mL of the normal sperm solution were added to the eggs and mixed gently. Fresh water was added to the eggs and gently swirled for 30 seconds to activate the sperm and eggs. More fresh water was added and the eggs were allowed to harden for 10–15 min before microinjection. The fertilized eggs were injected according to the procedures for zygote injection developed and modified recently in our laboratory75. Briefly, a 1.0 mm OD borosilicate glass capillary was pulled into two needles with a vertical needle puller. A very thin layer of vegetable shortening was applied to a 150 mm clean petri dish. Fifty to one-hundred eggs were transferred from the fertilization pan to the petri dish in a single layer and covered with Holtfreter’s solution (59 mmol NaCl, 0.67 mmol KCl, 2.4 mmol NaHCO3, 0.76 mmol CaCl2, 1.67 mmol MgSO4)76, 77. Using a microinjection system from Applied Scientific Instrumentation (Eugene, OR), 50 nanoliters of the mixture were directly injected into the yolk of each fertilized egg. One-cell embryos were injected through 15–90 minutes post-fertilization and just before the beginning of the first cell division78, 79. The injected control embryos were injected with the solution devoid of sgRNA/Cas9 mixture. The injected and control embryos were reared in 10-L tubs filled with Holtfreter’s solution containing 10 ppm doxycycline and incubated with continuous aeration at 27 °C for 6–8 days until hatching. Dead embryos were removed and the solution was changed daily. Channel catfish fry then were kept in 5-L containers for one month.
Mutation Analysis
Genomic DNA Extraction
Fin-clip samples were collected from one-month-old fry on ice. Five fry from each treatment were euthanized and samples from barbel, gills, muscle, intestine and eye were collected to study possible mosaicism of the mutations among different tissues. The DNA was extracted using the regular protocol80, proteinase K digestion followed by protein precipitation and iso-propanol precipitation of DNA. Quality and quantity of DNA were checked with the Nanodrop 2000 spectrophotometer (Thermo Fisher Scientific).
Polymerase Chain Reaction (PCR) and Surveyor Analysis
The primer sets for PCR were designed to cover all possible mutation sites (Fig. 1B). PCR was performed using the Expand High FidelityPLUS PCR System (Roche). The PCR amplification procedure was as follows: initial denaturation for 3 min at 94 °C; followed by 34 cycles of 94 °C for 30 s, 60 °C for 45 s, and 72 °C for 45 s; and a final elongation at 72 °C for 10 min. The resulting PCR product length was verified in a 1.5% agarose gel. The Surveyor® mutation detection kit for standard gel electrophoresis (Integrated DNA Technologies, Coralville, IA) was used to detect mutations81, 82; PCR products were denatured and re-annealed as follows: 95 °C for 10 min; 95 to 85 °C at −2 °C/s; 85 to 35 °C at −0.3 °C/s; cooling to 4 °C; and then Nuclease S was mixed with Enhancer S and MgCl2 and added to the PCR products above and incubated at 42 °C for 1 hour. The digested products were separated in a 2% agarose gel and compared to those from a non-edited channel catfish (Fig. 3A).
Cloning and Sequencing
To confirm and identify the mutations in each treatment, genomic DNA was obtained from five positive mutated individuals for each treatment and amplified with PCR, and the resulting amplicons were cloned into the TOPO® TA Cloning® Kit for Sequencing (Invitrogen, Carlsbad, CA) and then sent for sequencing to Eurofins Genomics (Louisville, KY). Alignments of nucleotides and amino acid sequences were created and interpreted using T-Coffee tool83.
Histology
To clarify the effect of MSTN knockout on skeletal muscle, we performed histological analysis and statistical quantitative analyses of muscle fibers in skeletal muscle of mutant and wild-type channel catfish. Four each of one-month-old fry from mutant (individuals with frame-shift mutation) and control groups were euthanized with tricaine methane sulfonate (MS-222) (Western Chemical Inc., Ferndale, WA). Subsequently, catfish muscles were dissected, cross sectioned and fixed in 4% paraformaldehyde at room temperature for at least 24 hours, and then dehydrated and paraffin embedded using Tissue Tek II® (Sakura Finetek USA, INC, CA). Serial sections were made at 7 μm thickness using a rotary microtome (American Optical Corporation, Southbridge, MA). The sections were mounted on glass slides and stained with the regressive staining method using Harris hematoxylin (VWR International, PA) and eosin with phloxine (Sigma-Aldrich, Inc., MO). Muscle fibers were counted. Cell numbers were calculated as the number of fibers per cross-sectional muscle area using the “Cell Counter” features of ImageJ program84 and used for evaluating fiber size.
Statistical Analysis
Statistical analysis of microinjected embryo survival, hatching and early fry survival were performed with SPSS 23.0 software (IBM Corporation, Armonk, NY). Dead embryos were collected, recorded and assigned a value representing the time of death (days post-fertilization, dpf). Mortality % was calculated as the number of dead embryos in each treatment divided by the total number of embryos in the same treatment and multiplied by 100. Survival curves for embryos and fry and the time to hatch for all treatment and control groups were compared using Kaplan-Meier test. Pairwise comparisons of mean survival and hatch time were performed using Log Rank (Mantel-Cox) test.
Independent samples t-tests were used to compare body weight and body length (growth parameters) between mutant (all positive fish from treatment groups) and non-mutated (all negative fish and controls) depending on the surveyor analysis. Independent samples t-tests also were performed to compare between mutants and wild types in terms of muscle fiber density (fibers number and size). The protocol for calculating the t-value took into account whether variances were homogeneous85. The Shapiro–Wilk test was utilized for analysis of normality of the data. The data that were not normally distributed were analyzed with the Mann–Whitney U-test (two-tailed). Analyses were performed with SAS® version 9.4 (SAS Institute, Cary, NC). Statistical significance was set at p < 0.05, and all data were presented as the mean ± standard error (SEM).
Data availability statement
All data generated or analysed during this study are included in this published article (and its Supplementary Information file).
References
Nelson, J. S., Grande, T. C. & Wilson, M. V. Fishes of the World. (John Wiley & Sons, 2016).
Hanson, T. & Sites, M. D. 2011 US Catfish Database. USDA National Agricultural Statistics Service (NASS), Mississippi Agricultural Statistics Service (MASS), (2012).
Abdelrahman, H. et al. Aquaculture genomics, genetics and breeding in the United States: current status, challenges, and priorities for future research. BMC Genomics 18, 191 (2017).
Grobet, L. et al. A deletion in the bovine myostatin gene causes the double-muscled phenotype in cattle. Nature Genetics 17, 71–74 (1997).
Grobet, L. et al. Molecular definition of an allelic series of mutations disrupting the myostatin function and causing double-muscling in cattle. Mammalian Genome 9, 210–213 (1998).
Kambadur, R., Sharma, M., Smith, T. P. & Bass, J. J. Mutations in myostatin (GDF8) in double-muscled Belgian Blue and Piedmontese cattle. Genome Research 7, 910–915 (1997).
McPherron, A. C. & Lee, S.-J. Double muscling in cattle due to mutations in the myostatin gene. Proceedings of the National Academy of Sciences of the United States of America 94, 12457–12461 (1997).
De Caestecker, M. The transforming growth factor-β superfamily of receptors. Cytokine & Growth Factor Reviews 15, 1–11 (2004).
Lee, S.-J. & McPherron, A. C. Regulation of myostatin activity and muscle growth. Proceedings of the National Academy of Sciences of the United States of America 98, 9306–9311 (2001).
Rebbapragada, A., Benchabane, H., Wrana, J., Celeste, A. & Attisano, L. Myostatin signals through a transforming growth factor β-like signaling pathway to block adipogenesis. Molecular and Cellular Biology 23, 7230–7242 (2003).
Souza, T. A. et al. Proteomic identification and functional validation of activins and bone morphogenetic protein 11 as candidate novel muscle mass regulators. Molecular Endocrinology 22, 2689–2702 (2008).
McPherron, A. C., Lawler, A. M. & Lee, S.-J. Regulation of skeletal muscle mass in mice by a new TGF-beta superfamily member. Nature 387, 83 (1997).
Dunner, S. et al. Towards interbreed IBD fine mapping of the mh locus: double-muscling in the Asturiana de los Valles breed involves the same locus as in the Belgian Blue cattle breed. Mammalian Genome 8, 430–435 (1997).
Smith, J., Lewis, A., Wiener, P. & Williams, J. Genetic variation in the bovine myostatin gene in UK beef cattle: allele frequencies and haplotype analysis in the South Devon. Animal Genetics 31, 306–309 (2000).
Clop, A. et al. A mutation creating a potential illegitimate microRNA target site in the myostatin gene affects muscularity in sheep. Nature Genetics 38, 813–818 (2006).
Mosher, D. S. et al. A mutation in the myostatin gene increases muscle mass and enhances racing performance in heterozygote dogs. PLoS Genet 3, e79 (2007).
Xianghai, Y. et al. Associations of myostatin gene polymorphisms with performance and mortality traits in broiler chickens. Genet. Sel. Evol 39, 73–89 (2007).
Zhang, C. et al. Polymorphisms of myostatin gene (MSTN) in four goat breeds and their effects on Boer goat growth performance. Molecular Biology Reports 39, 3081–3087 (2012).
Carlson, C. J., Booth, F. W. & Gordon, S. E. Skeletal muscle myostatin mRNA expression is fiber-type specific and increases during hindlimb unloading. American Journal of Physiology-Regulatory, Integrative and Comparative Physiology 277, R601–R606 (1999).
Garikipati, D. K., Gahr, S. A., Roalson, E. H. & Rodgers, B. D. Characterization of rainbow trout myostatin-2 genes (rtMSTN-2a and-2b): genomic organization, differential expression, and pseudogenization. Endocrinology 148, 2106–2115 (2007).
Kerr, T., Roalson, E. H. & Rodgers, B. D. Phylogenetic analysis of the myostatin gene sub‐family and the differential expression of a novel member in zebrafish. Evolution & Development 7, 390–400 (2005).
Maccatrozzo, L., Bargelloni, L., Cardazzo, B., Rizzo, G. & Patarnello, T. A novel second myostatin gene is present in teleost fish. FEBS Letters 509, 36–40 (2001).
Østbye, T.-K. K. et al. Myostatin (MSTN) gene duplications in Atlantic salmon (Salmo salar): evidence for different selective pressure on teleost MSTN-1 and-2. Gene 403, 159–169 (2007).
Rescan, P.-Y., Jutel, I. & Rallière, C. Two myostatin genes are differentially expressed in myotomal muscles of the trout (Oncorhynchus mykiss). Journal of Experimental Biology 204, 3523–3529 (2001).
Rodgers, B. D., Roalson, E. H., Weber, G. M., Roberts, S. B. & Goetz, F. W. A proposed nomenclature consensus for the myostatin gene family. American Journal of Physiology, Endocrinology and Metabolism 292, E371–E372 (2007).
Zheng, G.-D. et al. Two myostatin genes exhibit divergent and conserved functions in grass carp (Ctenopharyngodon idellus). General and Comparative Endocrinology 214, 68–76 (2015).
De Santis, C., Evans, B. S., Smith-Keune, C. & Jerry, D. R. Molecular characterization, tissue expression and sequence variability of the barramundi (Lates calcarifer) myostatin gene. BMC Genomics 9, 82 (2008).
Kanjanaworakul, P., Sawatdichaikul, O. & Poompuang, S. cDNA sequence and protein bioinformatics analyses of MSTN in African catfish (Clarias gariepinus). Molecular Biology Reports 43, 283–293 (2016).
Kanjanaworakul, P., Srisapoome, P., Sawatdichaikul, O. & Poompuang, S. cDNA structure and the effect of fasting on myostatin expression in walking catfish (Clarias macrocephalus, Günther 1864). Fish Physiology and Biochemistry 41, 177–191 (2015).
Kocabas, A. M., Kucuktas, H., Dunham, R. A. & Liu, Z. Molecular characterization and differential expression of the myostatin gene in channel catfish (Ictalurus punctatus). Biochimica et Biophysica Acta (BBA)-Gene Structure and Expression 1575, 99–107 (2002).
Liu, L., Yu, X. & Tong, J. Molecular characterization of myostatin (MSTN) gene and association analysis with growth traits in the bighead carp (Aristichthys nobilis). Molecular Biology Reports 39, 9211–9221 (2012).
Maccatrozzo, L., Bargelloni, L., Radaelli, G., Mascarello, F. & Patarnello, T. Characterization of the myostatin gene in the gilthead seabream (Sparus aurata): sequence, genomic structure, and expression pattern. Marine Biotechnology 3, 224–230 (2001).
Pan, J. et al. Molecular cloning and expression pattern of myostatin gene in yellow catfish (Pelteobagrus fulvidraco). DNA Sequence 18, 279–287 (2007).
Radaelli, G., Rowlerson, A., Mascarello, F., Patruno, M. & Funkenstein, B. Myostatin precursor is present in several tissues in teleost fish: a comparative immunolocalization study. Cell and Tissue Research 311, 239–250 (2003).
Rodgers, B. D., Weber, G. M., Sullivan, C. V. & Levine, M. A. Isolation and characterization of myostatin complementary deoxyribonucleic acid clones from two commercially important fish: Oreochromis mossambicus and Morone chrysops. Endocrinology 142, 1412–1418 (2001).
Ye, H.-Q., Chen, S.-L., Sha, Z.-X. & Liu, Y. Molecular cloning and expression analysis of the myostatin gene in sea perch (Lateolabrax japonicus). Marine Biotechnology 9, 262–272 (2007).
Gregory, D., Waldbieser, G. & Bosworth, B. Cloning and characterization of myogenic regulatory genes in three Ictalurid species. Animal Genetics 35, 425–430 (2004).
Dong, Z. et al. Heritable targeted inactivation of myostatin gene in yellow catfish (Pelteobagrus fulvidraco) using engineered zinc finger nucleases. PloS One 6, e28897 (2011).
Chiang, Y.-A. et al. TALENs-mediated gene disruption of myostatin produces a larger phenotype of medaka with an apparently compromised immune system. Fish & Shellfish Immunology 48, 212–220 (2016).
Dong, Z. et al. Generation of myostatin B knockout yellow catfish (Tachysurus fulvidraco) using transcription activator-like effector nucleases. Zebrafish 11, 265–274 (2014).
Maggio, I. & Goncalves, M. A. Genome editing at the crossroads of delivery, specificity, and fidelity. Trends in Biotechnology 33, 280–291 (2015).
Horvath, P. & Barrangou, R. CRISPR/Cas, the immune system of bacteria and archaea. Science 327, 167–170 (2010).
Ran, F. A. et al. Genome engineering using the CRISPR-Cas9 system. Nature Protocols 8, 2281–2308 (2013).
Fujii, W., Onuma, A., Sugiura, K. & Naito, K. One-step generation of phenotype-expressing triple-knockout mice with heritable mutated alleles by the CRISPR/Cas9 system. Journal of Reproduction and Development 60, 324–327 (2014).
Lv, Q. et al. Efficient generation of myostatin gene mutated rabbit by CRISPR/Cas9. Scientific Reports 6 (2016).
Wang, K. et al. Efficient generation of myostatin mutations in pigs using the CRISPR/Cas9 system. Scientific Reports 5, 16623 (2015).
Crispo, M. et al. Efficient generation of myostatin knock-out sheep using CRISPR/Cas9 technology and microinjection into zygotes. Plos One 10, e0136690 (2015).
Guo, R. et al. Generation and evaluation of myostatin knock-out rabbits and goats using CRISPR/Cas9 system. Scientific Reports 6 (2016).
Hongbing, H. et al. One-step generation of myostatin gene knockout sheep via the CRISPR/Cas9 system. Frontiers of Agricultural Science and Engineering 1, 2–5 (2014).
Wang, X. et al. Generation of gene-modified goats targeting MSTN and FGF5 via zygote injection of CRISPR/Cas9 system. Scientific Reports 5 (2015).
Cong, L. et al. Multiplex genome engineering using CRISPR/Cas systems. Science 339, 819–823 (2013).
Jao, L.-E., Wente, S. R. & Chen, W. Efficient multiplex biallelic zebrafish genome editing using a CRISPR nuclease system. Proceedings of the National Academy of Sciences of the United States of America 110, 13904–13909 (2013).
Wang, H. et al. One-step generation of mice carrying mutations in multiple genes by CRISPR/Cas-mediated genome engineering. Cell 153, 910–918 (2013).
Xiao, A. et al. Chromosomal deletions and inversions mediated by TALENs and CRISPR/Cas in zebrafish. Nucleic Acids Research 41, e141 (2013).
Chakrapani, V. et al. Establishing targeted carp TLR22 gene disruption via homologous recombination using CRISPR/Cas9. Developmental & Comparative Immunology 61, 242–247 (2016).
Edvardsen, R. B., Leininger, S., Kleppe, L., Skaftnesmo, K. O. & Wargelius, A. Targeted mutagenesis in Atlantic salmon (Salmo salar L.) using the CRISPR/Cas9 system induces complete knockout individuals in the F0 generation. PloS One 9, e108622 (2014).
Li, M. et al. Efficient and heritable gene targeting in tilapia by CRISPR/Cas9. Genetics 197, 591–599 (2014).
Qin, Z. Gene editing of luteinizing hormone, follicle-stimulating hormone and gonadotropin-releasing hormone genes to serilize Channel Catfish, Ictalurus punctatus, using zinc finger nuclease, transcription activator-like effector nuclease and clustered regularly interspaced short palindromic repeats/Cas9 technologies. PhD Dissertation. Auburn University, AL, USA (2015).
Zhong, Z. et al. Targeted disruption of sp7 and myostatin with CRISPR-Cas9 results in severe bone defects and more muscular cells in common carp. Scientific Reports 6 (2016).
Moreno-Mateos, M. A. et al. CRISPRscan: designing highly efficient sgRNAs for CRISPR-Cas9 targeting in vivo. Nature Methods 12, 982–988 (2015).
Liu, Z. et al. The channel catfish genome sequence provides insights into the evolution of scale formation in teleosts. Nature Communications 7 (2016).
Wargelius, A. et al. Dnd knockout ablates germ cells and demonstrates germ cell independent sex differentiation in Atlantic salmon. Scientific Reports 6 (2016).
Westerfield, M. & Zon, L. I. Methods in Cell Biology: Genetics, Genomics, and Informatics. The Zebrafish. Elsevier Academic Press (2004).
Abraham, V. C., Gupta, S. & Fluck, R. A. Ooplasmic segregation in the medaka (Oryzias latipes) egg. The Biological Bulletin 184, 115–124 (1993).
Leung, C. F., Webb, S. E. & Miller, A. L. On the mechanism of ooplasmic segregation in single‐cell zebrafish embryos. Development, Growth & Differentiation 42, 29–40 (2000).
Acosta, J., Carpio, Y., Borroto, I., González, O. & Estrada, M. P. Myostatin gene silenced by RNAi show a zebrafish giant phenotype. Journal of Biotechnology 119, 324–331 (2005).
Lee, C.-Y. et al. Suppression of myostatin with vector-based RNA interference causes a double-muscle effect in transgenic zebrafish. Biochemical and Biophysical Research Communications 387, 766–771 (2009).
Biga, P. R. & Goetz, F. W. Zebrafish and giant danio as models for muscle growth: determinate vs. indeterminate growth as determined by morphometric analysis. American Journal of Physiology-Regulatory, Integrative and Comparative Physiology 291, R1327–R1337 (2006).
Silva, P. et al. Hyperplastic and hypertrophic growth of lateral muscle in blackspot seabream Pagellus bogaraveo from hatching to juvenile. Journal of Fish Biology 74, 37–53 (2009).
Veggetti, A., Mascarello, F., Scapolo, P. & Rowlerson, A. Hyperplastic and hypertrophic growth of lateral muscle in Dicentrarchus labrax (L.). Anatomy and Embryology 182, 1–10 (1990).
Lyons, J.-A., Haring, J. S. & Biga, P. R. Myostatin expression, lymphocyte population, and potential cytokine production correlate with predisposition to high-fat diet induced obesity in mice. PloS One 5, e12928 (2010).
Singh, N. K., Singh, S., Jain, S. K. & Sarkhel, B. C. Evaluation of interferon response induced by anti-myostatin shRNA constructs in goat (Capra hircus) fetal fibroblasts by quantitative real time-polymerase chain reaction. Animal Biotechnology 23, 174–183 (2012).
Gagnon, J. A. et al. Efficient mutagenesis by Cas9 protein-mediated oligonucleotide insertion and large-scale assessment of single-guide RNAs. PloS One 9, e98186 (2014).
Varshney, G. K. et al. High-throughput gene targeting and phenotyping in zebrafish using CRISPR/Cas9. Genome Research 25, 1030–1042 (2015).
Elaswad, A. Genetic technologies for disease resistance research and enhancement in catfish. PhD Dissertation. Auburn University, AL, USA (2016).
Armstrong, J., Duhon, S. & Malacinski, G. Raising the axolotl in captivity in Developmental Biology of the Axolotl, Oxford University Press, New York, 220–227 (1989).
Bart, A. & Dunham, R. Effects of sperm concentration and egg number on fertilization efficiency with channel catfish (Ictalurus punctatus) eggs and blue catfish (I. furcatus) spermatozoa. Theriogenology 45, 673–682 (1996).
Saksena, V. P., Riggs, C. D. & Yamamoto, K. Early development of the channel catfish. The Progressive Fish-Culturist 23, 156–161 (1961).
Tucker, C. C. & Robinson, E. H. Channel Catfish Farming Handbook. Springer Science & Business Media, (1990).
Kurita, K., Burgess, S. M. & Sakai, N. TransgeniTc zebrafish produced by retroviral infection of in vitro-cultured sperm. Proceedings of the National Academy of Sciences of the United States of America 101, 1263–1267 (2004).
Gerard, G., Shandilya, H., Qiu, P., Shi, Y. & Lo, J. Genetic variance detection using Surveyor Nuclease. Genetic Variance Detection-Technologies for Pharmacogenomics (ed. Hecker, KH) DNA Press, Eagleville, PA, 95–129 (2006).
Qiu, P. et al. Mutation detection using Surveyor™ nuclease. Biotechniques 36, 702–707 (2004).
Notredame, C., Higgins, D. G. & Heringa, J. T-Coffee: A novel method for fast and accurate multiple sequence alignment. Journal of Molecular Biology 302, 205–217 (2000).
Schneider, C. A., Rasband, W. S. & Eliceiri, K. W. NIH Image to ImageJ: 25 years of image analysis. Nature Methods 9, 671 (2012).
Steel, R. G. & Torrie, J. H. Principles and Procedures of Statistics: A Biometrical Approach. Vol. 633 McGraw-Hill, New York, (1980).
Acknowledgements
This project was supported via PhD scholarship to Karim Khalil from Culture Affairs and Mission Sector, Ministry of Higher Education, Government of Egypt.
Author information
Authors and Affiliations
Contributions
K.K., M.E., E.K., S.D., A.E. and R.D. conceived and designed the experiment; K.K., A.E., Z.Y., R.O., D.D., K.V., K.G., W.B. and D.R. conducted the experiments; M.M. provided materials; A.E. and H.A. performed the statistical analyses; K.K. and R.D. analyzed the results and wrote the manuscript. All authors critically revised and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing Interests
We declare that we have no significant competing financial, professional or personal interests that might have influenced the performance or presentation of the work described in this manuscript.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Khalil, K., Elayat, M., Khalifa, E. et al. Generation of Myostatin Gene-Edited Channel Catfish (Ictalurus punctatus) via Zygote Injection of CRISPR/Cas9 System. Sci Rep 7, 7301 (2017). https://doi.org/10.1038/s41598-017-07223-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-07223-7
This article is cited by
-
Activin Signaling Pathway Specialization During Embryonic and Skeletal Muscle Development in Rainbow Trout (Oncorhynchus mykiss)
Marine Biotechnology (2024)
-
The key role of myostatin b in somatic growth in fishes derived from distant hybridization
Science China Life Sciences (2024)
-
Genetic improvement in edible fish: status, constraints, and prospects on CRISPR-based genome engineering
3 Biotech (2024)
-
Pangenomics of the cichlid species (Oreochromis niloticus) reveals genetic admixture ancestry with potential for aquaculture improvement in Kenya
The Journal of Basic and Applied Zoology (2023)
-
Development of a chub mackerel with less-aggressive fry stage by genome editing of arginine vasotocin receptor V1a2
Scientific Reports (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.