Abstract
Remora fishes adhere to, and maintain long-term, reversible attachment with, surfaces of varying roughness and compliance under wetted high-shear conditions using an adhesive disc that evolved from the dorsal fin spines typical of other fishes. Evolution of this complex hierarchical structure required extensive reorganization of the skull and fin spines, but the functional role of the soft tissues of the disc are poorly understood. Here I show that remora cranial veins are highly-modified in comparison to those of other vertebrates; they are transposed anteriorly and enlarged, and lie directly ventral to the disc on the dorsum of the cranium. Ancestrally, these veins lie inside the neurocranium, in the dura ventral to the brain, and return blood from the eyes, nares, and brain to the heart. Repositioning of these vessels to lie in contact with the ventral surface of the disc lamellae implies functional importance associated with the adhesive mechanism. The position of the anterior cardinal sinus suggests that it may aid in pressurization equilibrium during attachment by acting as a hydraulic differential.
Similar content being viewed by others
Introduction
Remoras (Echeneoidea) comprise a clade of fishes that have evolved an adhesive disc on the dorsal aspect of the head which permits them long-term reversible attachment to a variety of hosts of different roughness and body compliance (including sharks, rays, whales, dolphins, marlin, turtles, other remoras, divers, and boats)1,2,3,4,5,6.
Whereas previous research has investigated the development of the remora disc7, adhesive force measurements8,9, and the skeletal components contributing to the hierarchical mechanics of adhesion8,10, little attention (with the exception of the anatomical description of the epithelium, muscles, and nerves of the remora disc by Houy11) has been paid to the soft tissues that contribute to remora adhesion mechanics12.
Here I describe a previously unidentified circulatory structure in the remora adhesive mechanism that may contribute to the versatile attachment abilities of remoras.
Results and Discussion
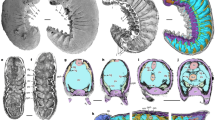
Dissection of a euthanized, fresh remora revealed large diameter blood vessels lying dorsal to the skull and in contact with the ventral side of the lamellae and associated musculature of the adhesive disc. This vasculature was better visualized through reconstruction of computed microtomography (µCT) scans of remora specimens perfused with iodixanol, which revealed that these exceptionally large cranial veins (approximately 2.8–3.3 mm in diameter, or 11.2–13.2% of head width) are located within a space situated between the pectinated lamellae of the disc and the dorsal aspect of the cranium (Fig. 1), indicating that when the lamellae are erected, the cranial veins and their overlying epithelium constitute the roof of each lamellar compartment when attached to a host organism. The large blood vessels are flanked laterally and medially by the erector and depressor muscles that control the rotation of the lamellae.
Cranial veins of the remora, Echeneis naucrates, post-perfusion with iodixanol (blue). (a) Lateral view. (b) Dorsolateral view, disc digitally removed. ACS, anterior cardinal sinus; DA, dorsal aorta; DC, ductus Cuvieri; DCV, dorsal collector vein; OS, orbital sinus; SV, sinus venosus. (c) Diagram of transverse view or remora cranium at mid-orbit, left lamellae is depressed, right lamellae is erected. CE central erector muscle; D, depressor muscle; LE, lateral erector muscle; ME, medial erector muscle; SP, spinules. In this orientation, spinules are not visible on the depressed lamellae. An interactive model of the micro CT scan is available both as a 3D PDF in Supplementary Materials and online here: https://skfb.ly/6oLuR.
In the remora (Fig. 1), blood leaves the neurocranium anteriorly through the dorsal collecting vein (DCV) and from the eyes via the orbital sinus (OS), and passes through the anterior cardinal sinus (ACS), through the ductus Cuvieri (DC) to return to the sinus venosus (SV) of the heart. Blood is then pumped through the gills to be oxygenated and returns to the body through the dorsal aorta (DA).
Notably, in other vertebrates13,14,15, the ACS is located primarily inside the neurocranium ventral to the brain in the dura. In humans it is known as the cavernous sinus; the primary function of cavernous sinus is to return blood from the brain, eyes, and nares through the superior and inferior petrosal sinus to the jugular vein (homologous to the ductus Cuvieri16), back to the heart. In cobia (Rachycentron canadum), the closest living relative of remoras, the ACS is in the dura, ventral to the brain, and measures 0.84 mm in diameter (3.4% of head width) at its largest point as it exits the neurocranium through the jugular foramen.
The dorsally and anteriorly transposed position of the cranial vessels of remoras shows considerable morphological deviation from the normal, protected position of the thin-walled ACS within the neurocranium as compared to all other vertebrates, indicating that this vessel has an important functional role driving its anomalous location. Divergence from the general vertebrate cranial vasculature condition is not entirely unique to remoras; highly-modified cranial vessels form the brain-warming rete in mobulids17 and eye-warming rete in lamnid sharks18. However, remoras adhere to both exothermic and endothermic hosts, thereby making function as a heat-sink unlikely as a driving factor in this evolutionary novelty. Instead, it is quite likely that the highly-modified remora ACS has a functional role in the cranial adhesive mechanism.
Remora adhesion is primarily achieved by friction and suction. Frictional force is generated by the small spinules of the pectinated lamellae (SP, Fig. 2) that interact with host surface to overcome, by an order of magnitude8, the drag experienced by remoras. Therefore, the most probable cause of failure over long periods of hold time is not slippage but loss of suction as a result of seep12. Suction under the disc is achieved by rotation of the pectinated lamellae when the disc is in contact with the host – this creates a relatively negative, sub-ambient pressure space under the disc8,9. The edge of the disc is sealed by a fleshy lip of thick epithelium, as are the edges of the lamellae and the median septum, thereby creating a sealed compartment between two neighboring lamellae (Fig. 2a). Suction failure results when fluid seeping under the fleshy edge of the disc causes equalization between under-disc and ambient pressures19. Host organisms to which remoras adhere have curved bodies with varying ranges of surface roughness, resulting in uneven attachment surfaces. Lamellar rotation is individually controlled to permit the disc to shape its contact surface to the host, which results in unequal volume compartmentalization between lamellae such that the initial pressure within a lamellar compartment would not be equal to that of its neighbors (Fig. 3a). Unequal pressurization in neighboring lamellar compartments would promote seep from outside the disc leading to equilibrium12; thereby causing loss of suction adhesion.
Disc of a remora attached to glass; tissue appears lighter in color when it is sealed to the glass. The fleshy epithelial lip (FL) surrounds the edge of disc and creates the suction seal. The pectinated lamellae (PL) rotate to contact the host, generating friction when spinules interact with local surface asperities. A lamellar compartment (LC) is created between two neighboring rotated lamellae and is sealed on four sides by the thick epithelium on the distal edge of the lamellae (a), fleshy lip, and surface of the medial septum (MS). Note that in the more posterior region of the disc (white dashed circle), the lamellae do not create a full seal with the host surface (b).
The remora anterior cardinal sinus as a hydrostatic differential. (a) Remora attached to a host. (b) Remora approaching a host, lamellae depressed before contact is made. (c) Host organisms all have some degree of surface roughness or curvature, therefore, in attachment to a host, not all lamellae rotate to the same degree, thus not all lamellar compartments are equal in volume or pressure. The thin-walled anterior cardinal sinus (ACS) is deformed by lamellar rotation and thereby could equalize pressure differences among lamellar compartments within the disc. Numerical values illustrate hypothesized variation in pressure differentials within the disc. (d) Bulging of the ACS into the lamellar compartment (AT) occurs passively during lamellar rotation when compartment height (h) increases, causing compartmental volume to increase (V), whereas the host surface area of any given lamellar compartment (b) remains constant.
I hypothesize that the antero-dorsally transposed position of the ACS allows it to function as a hydraulic differential20, promoting equalization of pressure among lamellar compartments at the time of attachment, thereby eliminating localized relative pressure gradients that would increase fluid seep and cause suction failure. Blood vessels have non-linearly elastic properties and exhibit strain-dependent increases in elasticity and stiffness21,22,23. The ACS is a thin-walled vein unlikely to resist deformation as a result of pressurization. Functioning as a hydraulic differential, the dorsal wall of the ACS would bulge into the lamellar compartment in proportion to the sub-ambient pressure within any given compartment (Fig. 3), thereby rendering the average pressure throughout the space between the disc and the host relatively uniform. Eliminating the pressure differential among lamellar compartments resists seepage by removing focal weak points along the disc edge. This mechanism may be especially important when the host surface is irregular or curved, as it is on host organisms to which remoras adhere, in which case, variation in volume among lamellar compartments would be greater. Incidentally, the hierarchical adhesive mechanism of some geckos also relies upon a fluid pressure differential; the reticular network of enlarged blood vessels in the lamellar plates allows deformation of scansors along multiple axes at a single time, thereby promoting adhesive contact on irregular surfaces20,24,25.
It is worth noting, however, that the ACS only lies deep to the anterior 70% of the disc; the more caudal 30% of the disc lies dorsal to the body. If this posterior 30% of the disc was also functionally divided into separate lamellar compartments, it is reasonable to expect that this portion of the disc would be more susceptible to seep because it lacks the hydraulic differential system for equilibration. However, during adhesion, the posterior region of disc is not divided into discrete lamellar compartments, because those lamellae do not create a seal with the host surface (Fig. 2,b). By injecting food dye under the disc of a live, attached remora, I found that fluid is contained within any of the individual lamellar compartments in the anterior 70% of the disc, but dye injected between lamellae in the posterior 30% of the disc circulates within this posterior region. This suggests functional regionalization within the disc; the posterior-most lamellae are not as tightly associated with the host and the posterior region acts as one continuous suction component. The large, posterior suction region of the remora disc is advantageous in two ways: the fleshy lip is wider and it has a larger cavity volume. The fleshy lip of the disc is wider in the posterior region than the anterior region (Fig. 2) and, following Darcy’s law26, this requires a larger pressure gradient to induce seep across a longer seal distance. And, because the larger posterior cavity takes longer to fill with a given seep rate, it extends the time to attachment failure.
The remora adhesive disc is a functional novelty that is the product of extensive modulation of pre-existing morphology in its evolution. In cobia, the closest living relative of remoras27, the dorsal fin spines are located approximately 40–45% of total length, measured from the rostrum; the remora adhesive disc evolved from fin spines like those in cobia. The most basal remora (†Opisthomyzon) possessed a disc-like structure with fewer lamellae (which lacked spinules) and was located farther caudally than modern remoras27,28,29. This suggests that basal remoras may have had adhesive abilities, albeit potentially limited, before the disc was transposed anteriorly and superficial to the cranium. Therefore, reorganization of the cranial vasculature was independent of the exaptation of the dorsal fin elements in forming the adhesive disc. Development of the remora adhesive disc does not appear to modify the development of the neurocranium7, suggesting that modulation of the cranial veins was independently derived. The evolution of this unique morphology, that would otherwise seem constrained given the nature of its physiology, and which places a large, thin-walled vein in a very superficial position anatomically, underlies the functional importance of the ACS in the remora adhesive mechanism.
Experimental Procedures
Remoras (Echeneis naucrates) were kept in 1135 L aquaculture tanks with 12:12 hour light:dark cycle in the aquatic animal facility in the Department of Biological Sciences at the New Jersey Institute of Technology, and in the Lauder Laboratory at Harvard University during pilot studies. They were fed a custom fish, shrimp meal, and vegetable mixture daily. All animals were handled ethically according to institutional animal care and use protocols approved by the New Jersey Institute of Technology (14–036) and Harvard University (20–03).
One remora was euthanized with an overdose of tricaine methanesulfonate (Tricaine-S, Western Chemical, Inc., Ferndale, WA) and dissected in preparation for an electromyography study, which led to my initial discovery of the novel vein structure described here.
Two remora (24 and 33 cm TL) were used for imaging cranial vasculature. Transcardiac perfusion was performed after fish had been euthanized by an overdose of Tricaine-S. An incision was made between the dentaries and pectoral girdle and the skin reflected to reveal the heart. A 26 g needle attached to polyethylene outlet tubing of a small peristaltic pump (P720, Instech Laboratories, Plymouth Meeting, PA) was inserted into the conus arteriosus and 30–50 ml of heparinized saline (1 mg/ml)30 perfused, depending upon the size of the fish. A small cut was made in the sinus venosus to drain blood and fluids so as not to overinflate vessels and risk bursting. For contrast, 50–70 ml of iodixanol (270 mg/ml, Visipaque 270, GE Healthcare Inc., USA) was then perfused and either µCT scanned immediately or placed in formalin and µCT scanned within 18 hours (dependent upon scanner availability) in a Skyscan 1173 (125 kV, 48 µA, 35.17 µm pixel size, MicroPhotonics, Inc., USA) or Skyscan 1275 (80 kV, 125 µA, 31.00 µm pixel size, MicroPhotonics, Inc., USA). Reconstruction and segmentation of µCT scans was performed in Mimics 18 (Materialise USA).
One remora (37 cm TL) was lightly anaesthetized using Tricaine-S to permit handling without inducing stress, and placed onto a piece of acrylic into which a 1.5 mm hole had been drilled and polyethylene tubing had been glued using cyanoacrylate. The remora was positioned so that a single lamellar chamber was lying over the hole and blue food dye was injected into the lamellar compartment. The dye was observed for 1 minute and then the remora was moved off the plate to clear the dye. The procedure was replicated for each lamellar chamber in the disc. The remora was under light sedation for 28 minutes, was observed in a revival tank for one hour, and returned to its tank without incident.
Two cobia (Rachycentron canadum), one preserved (30 cm TL) and one received fresh frozen (28 cm TL; Rosenstiel School of Marine and Atmospheric Science, University of Miami), were dissected to measure the ACS for comparison to the remora. In both cases, a midsagittal incision was made in the cranium using a Dremel rotary tool and the ACS located at the jugular foramen in the posterior neurocranium.
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
Change history
12 February 2018
A correction to this article has been published and is linked from the HTML version of this paper. The error has been fixed in the paper.
References
Williams, E. H. Jr et al. Echeneid–sirenian associations, with information on sharksucker diet. J. Fish Biol. 63, 1176–1183 (2003).
Sazima, I. & Grossman, A. Turtle riders: remoras on marine turtles in Southwest Atlantic. Neotrop. Ichthyol. 4, 123–126 (2006).
Weihs, D., Fish, F. E. & Nicastro, A. J. Mechanics of remora removal by dolphin spinning. Mar. Mammal Sci. 23, 707–714 (2007).
Silva, J. M. Jr & Sazima, I. Whalesuckers and a spinner dolphin bonded for weeks: Does host fidelity pay off? Biota Neotrop. 2003, 2 (2003).
Cressey, R. F. & Lachner, E. A. The parasitic copepod diet and life history of diskfishes (Echeneidae). Copeia 1970, 310–318 (1970).
Fertl, D. & Landry, A. M. Sharksucker (Echeneis naucrates) on a bottlenose dolphin (Tursiops truncatus) and a review of other cetacean-remora associations. Mar. Mammal Sci. 15, 859–863 (1999).
Britz, R. & Johnson, G. D. Ontogeny and homology of the skeletal elements that form the sucking disc of remoras (Teleostei, Echeneoidei, Echeneidae). J. Morphol. 273, 1353–1366 (2012).
Beckert, M., Flammang, B. E. & Nadler, J. H. Remora fish suction pad attachment is enhanced by spinule friction. J. Exp. Biol. 218, 3551–3558 (2015).
Fulcher, B. A. & Motta, P. J. Suction disk performance of echeneid fishes. Can. J. Zool. 84, 42–50 (2006).
Beckert, M., Flammang, B. E., Anderson, E. J. & Nadler, J. H. Theoretical and computational fluid dynamics of an attached remora (Echeneis naucrates). Zoology (2016).
Huoy, R. B. zur Kenntnis der Haftscheibe von Echeneis. Zool Jb Anat Ontog Tiere 29, 101–138 (1909).
Beckert, M., Flammang, B. E. & Nadler, J. H. A model of interfacial permeability for viscoelastic seals in marine-organism, suction-based adhesion. MRS Advances 1, 2531–2543 (2016).
Gilbert, S. G. Pictorial anatomy of the dogfish. (University of Washington Press, 1973).
Satchell, G. H. in Fish Physiology Vol. XII, Part A (ed William Stewart Hoar) 141–184 (Academic Press, Inc., 1992).
Gray, H. Gray’s anatomy. (Running Press, 1901).
Liem, K. F., Bemis, W. E., Walker, W. F. & Grande, L. Functional anatomy of the vertebrates: an evolutionary perspective. 3rd edn, (Thomson Learning, 2001).
Alexander, R. Evidence of brain‐warming in the mobulid rays, Mobula tarapacana and Manta birostris (Chondrichthyes: Elasmobranchii: Batoidea: Myliobatiformes). Zool. J. Linnean Soc. 118, 151–164 (1996).
Block, B. A. & Carey, F. G. Warm brain and eye temperatures in sharks. J. Comp. Physiol. B 156, 229–236 (1985).
Beckert, M., Flammang, B. E. & Nadler, J. H. A model of interfacial permeability for soft seals in marine-organism, suction-based adhesion. MRS Advances (2016).
Russell, A. P. Descriptive and functional anatomy of the digital vascular system of the tokay, Gekko gecko. J. Morphol. 169, 293–323 (1981).
Shadwick, R. E. Mechanical design in arteries. J. Exp. Biol. 202, 3305–3313 (1999).
Storm, C., Pastore, J. J., MacKintosh, F. C., Lubensky, T. C. & Janmey, P. A. Nonlinear elasticity in biological gels. Nature 435, 191–194 (2005).
Wesly, R., Vaishnav, R. N., Fuchs, J., Patel, D. J. & Greenfield, J. C. Static linear and nonlinear elastic properties of normal and arterialized venous tissue in dog and man. Circ res 37, 509–520 (1975).
Hiller, U. Untersuchungen zum feinbau und zur funktion der haftborsten von reptilien. Z. Morph. Tiere 62, 307–362 (1968).
Russell, A. P. A contribution to the functional analysis of the foot of the Tokay, Gekko gecko (Reptilia: Gekkonidae). J. Zool. 176, 437–476 (1975).
Dullen, F. A. L. Porous Media Fluid Transport and Pore Strcture. 2nd edition edn, (Academic Press, Inc., 1992).
Friedman, M., Johanson, Z., Harrington, R. C., Near, T. J. & Graham, M. R. in Proc. R. Soc. B. 20131200 (The Royal Society).
Storms, R. X. The adhesive disk of Echeneis. Journal of Natural History 2, 67–76 (1888).
Friedman, M. & Johanson, Z. † Opisthomyzon glaronensis (Wettstein, 1886)(Acanthomorpha,† Opisthomyzonidae), a junior synonym of† Uropteryx elongatus Agassiz, 1844. Journal of Vertebrate Paleontology 32, 1202–1206 (2012).
Hattingh, J. Heparin and ethylenediamine tetra-acetate as anticoagulants for fish blood. Pflügers Archiv 355, 347–352 (1975).
Acknowledgements
B.E.F. wishes to thank E.J. Anderson, J.H. Nadler, M. Beckert, A.P. Summers, and G.V. Lauder for their encouraging discussions. A.P. Russell offered tremendous insight that greatly improved the quality of this manuscript. K. Hartel and A. Williston (Museum of Comparative Zoology, Harvard University) generously loaned specimens for comparative observation. S. Crofts, C. Crawford, K. Gamel, L. Winn, and A. Tam assisted in the dye injection experiments. K. Suri and K. Stanchak assisted in several perfusion experiments and CT scans. D. Benetti and J. Fiorentino (RSMAS, University of Miami) provided the cobia. B. Ache and B. Walters (MicroPhotonics, Inc.) assisted in μCT scanning.
Author information
Authors and Affiliations
Contributions
B.E.F. discovered the anatomy, conceived and conducted the experiments, analysed the data, developed the functional theory, prepared the figures, and wrote the manuscript. C.P.K. provided the contrast agent and operated the CT scanner for the experiment that resulted in Figure 1.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Change History: A correction to this article has been published and is linked from the HTML version of this paper. The error has been fixed in the paper.
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
A correction to this article is available online at https://doi.org/10.1038/s41598-018-20761-y.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Flammang, B.E., Kenaley, C.P. Remora cranial vein morphology and its functional implications for attachment. Sci Rep 7, 5914 (2017). https://doi.org/10.1038/s41598-017-06429-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-06429-z
This article is cited by
-
The design and working effect evaluation of the conical surface-mounted end-effector
Journal of the Brazilian Society of Mechanical Sciences and Engineering (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.